Abstract
The objectives of the present study were to describe the clinical characteristics of patients with severe familial non-hemiplegic migraine with aura (NHMA) and to compare these data to those from cases in previous population-based Danish studies using the same methodology. NHMA families were recruited from the Danish patient registry and from Danish neurology practices. A total of 362 NHMA patients were diagnosed according to the 1988 International Headache Society criteria using a validated semistructured physician-conducted interview. Visual aura occurred in almost every NHMA attack. In aura without headache visual aura occurred primarily in isolation. Aura without headache was most common in older, male patients. Several clinical characteristics of familial NHMA differed from migraine with aura in the general population: firstly, the age at onset was lower, secondly, the age at cessation was higher, thirdly, aura symptoms were more severe and finally, the co-occurrence of migraine without aura was higher in familial NHMA. There seems to be a correlation between more severe symptoms and familial aggregation. These results have both clinical and scientific implications.
Introduction
Migraine with aura (MA) is characterized by recurrent attacks of visual, sensory, motor, aphasic or basilar symptoms. The presence of a headache related to the aura is a variable feature. A detailed knowledge of the aura symptoms is crucial when diagnosing MA because it is a clinical entity with no diagnostic biological markers. Most descriptions of MA are from studies of modest size or questionnaire studies (1–4). However, only studies using a validated, structured interview done by physicians with experience in headache diagnoses are likely to increase the understanding of the symptomathology of MA (5, 6). Previously the characteristics of MA patients from two Danish population-based studies have been presented (1, 7, 8).
The aim of the present study was to identify families with familial occurrence of MA in order to describe the clinical characteristics of familial MA and subsequently use the material for molecular genetic studies. In this report we present the methodology and the clinical data. Furthermore, we compare the present data representing severe, clinic-type MA, to the clinical characteristics in MA from previous Danish population-based studies that have used the same methodology (1, 7, 8). The present study describes exclusively MA patients without motor weakness, that is non-hemiplegic migraine with aura (NHMA), since NHMA and familial-(FHM) and sporadic (SHM) hemiplegic migraine are separate entities in the International Classification of Headache Disorders 2nd Edition (9).
Materials and methods
The two sources of ascertainment
Our search strategy included patients from the entire Danish population of 5.2 million inhabitants. The strategy aimed at identifying probands with nonhemiplegic migraine with aura (NHMA) for the present study and probands with hemiplegic migraine (HM) for other studies (10–12). Two sources of ascertainment were used: From the National Patient Register of all hospitalized patients, we extracted records of all patients admitted to hospital or seen as outpatients at a department of neurology or paediatrics in a Danish hospital in the years 1994–97 and discharged with a diagnosis of MA or migraine with complication (ICD 10 diagnosis DG431 or DG433) (10). Additionally, more than 27 000 case records were screened for patients with NHMA and a history of siblings with NHMA and for patients with HM or NHMA with more widespread sensory symptoms (10). About 6000 of these case records were from two headache clinics and a SPECT-scanning clinic, whereas 21 000 case records were from practising neurologists (10). The recruited patients received a posted letter stating that the objectives of the survey were to study the inheritance of migraine. The patients were asked to tick on a return slip whether they agreed to participate in a telephone interview and return the slip within 5 days in a prepaid envelope. Patients recruited from the practicing neurologists had to reply confirmatively on the return slip before we were allowed to contact them by telephone (10). The other patients were contacted, unless they returned the slip stating that they did not want to participate. Those not responding had a reminding letter.
Ascertainment of probands with NHMA
A total of 1831 patients were recruited: 1101 from the National Patient Register and 730 from the case records. Of 1831 patients, 85 were non-contactable and 381 did not participate (10). A total of 1365 patients participated in the study. These patients were labelled probands. The probands with a history of HM or more widespread sensory symptoms recruited from the screening of case records were contacted for an extensive semistructured telephone interview by a physician (10) whereas the remainder probands took part in an initial screening telephone interview by three medical students trained in headache diagnoses. The validated extensive semi-structured telephone interview was based on the diagnostic criteria of the International Headache Society (IHS) (6, 13, 14) and the patients were diagnosed according to the IHS 1988 criteria (15). If a proband turned out to have HM according to the screening intreviw this proband was also contacted for an extensive semistructured telephone interview by a physician (10). A total of 128 probands had HM (33 FHM, 64 SHM, 31 unclassifiable HM patients) (10), 980 had NHMA and 257 did not have any kind of MA. The probands for the present study were recruited among the 980 NHMA probands. The ascertainment of NHMA probands and their response pattern are shown in Fig. 1.

Ascertainment of probands with nonhemiplegic migraine with aura (NHMA). In total 105 families with NHMA sib pairs were identified. The 105 probands were diagnosed with NHMA in an extensive interview. M: F, male to female ratio.
Ascertainment of relatives with NHMA
During the screening telephone interview the NHMA probands were asked about affected relatives. Out of 980 NHMA probands, 189 probands had a family history of at least one sib pair with both siblings affected with NHMA (NHMA sib pair) in the family. These families were further extended. Selected relatives of the 189 probands were contacted: relatives suspected of being part of a NHMA sib pair, siblings or parents of a NHMA sib pair, or close relatives to NHMA patients in families with many affected members. The relatives were contacted using the same procedure as used when contacting the probands except that all relatives took part in an extensive telephone interview performed by a physician with experience in headache diagnoses. The ascertainment of relatives and their response pattern are shown in Fig. 2. Contact was desirable in 736 living relatives. However, 25 relatives were noncontactable due to emigration or lost family contact (n = 19) or because the proband did not agree to contact relatives (n = 6), and 68 did not participate because contact was impossible to obtain (n = 13) or the relative declined (n = 55). In total 643 relatives were interviewed of whom 272 had NHMA. Out of 272 relatives with NHMA 257 were parts of a family with a NHMA sib pair: 208 first-degree relatives, 26 second-degree relatives, and 23 third-degree or more distant relatives.

Ascertainment of relatives with nonhemiplegic migraine with aura (NHMA). In total 643 relatives participated in an extensive interview of whom 272 had NHMA. Out of 272 relatives with NHMA, 257 were part of a family with a NHMA sib pair. M: F, male to female ratio.
The probands of the 189 families were also contacted for an extensive telephone interview by a physician as appropriate if affected siblings or relatives were identified. Contact was desirable in 135 probands. However, in 3 probands contact was impossible to obtain, one proband was deceased and 4 declined. In total 127 probands participated in an extensive telephone interview: An IHS diagnosis of NHMA (IHS 1.2) was confirmed in 120 probands, 3 probands had HM and 4 probands did not fulfil the IHS criteria of NHMA. We succeeded in finding 105 families with at least one NHMA sib pair (Fig. 1). The reasons for not identifying a NHMA sib pair in the remainder 76 participating families were that the proband had HM (n = 3), the proband did not fulfil the IHS criteria of NHMA (n = 4), the sibling had no NHMA (n = 40), the sibling had HM (n = 4), the proband did not agree to contact sibling (n = 11), the sibling declined (n = 5), the siblinCivil Registration Systemg was deceased (n = 1), the sibling had emigrated (n = 4), the sibling was noncontactable (n = 3), or the siblings were monozygotic twins (n = 1), respectively.
Participation pattern
We recruited 362 NHMA patients from 105 families. The number of NHMA patients in each family was 2 (40 families), 3 (32 families), 4 (18 families), 5 (1 family), 6 (8 families), 7 (1 family), 8 (1 family), 9 (1 family), 12 (2 families), or 13 (1 family) patients, respectively – with a mean of 3.4. The overall participation rate among probands recruited from the two recruitment sources was 78% (1357/1746): 87% among probands recruited from the National Patient Register and 65% among probands from the case records. The overall participation rate among relatives was 90% (643/711): 93% among first-degree relatives, 94% among second-degree relatives and 80% among third-degree or more distant relatives. The interviews took place from February 1999 to March 2002. The project was approved by the Danish Ethical Committees.
The previous Danish population-based studies
The present data were compared to the data from two previous Danish population-based studies (1, 7, 8). In both population-based studies the patients were interviewed by a physician with experience in headache diagnosis using a similar semistructured interview as used in the present study. In one population-based study (1, 7) the patients were recruited among 3000 males and 1000 females drawn from the Danish National Civil Registration System. This study was used for comparison whenever possible. A total of 163 MA patients (male:female (M:F) ratio 1.4 : 1) aged 40 years were recruited. In the other cross-sectional population-based study (8) the patients were recruited among 1000 persons drawn from the Danish National Civil Registration System. A total of 38 MA patients (M:F ratio 1 : 2) aged 25–64 years were recruited.
Data processing and statistical analysis
The χ2 test, t-test or Mann–Whitney U-test were used as appropriate. P < 0.05 was considered significant. Statistical analysis was performed using SPSS Base System 11.5 for Windows XP Professional and SAS 8.02 TS Level 02M0.
Results
Sex distribution and diagnosis
The 362 NHMA patients comprised 99 males and 263 females. The M:F ratio was 1 : 2.7 (vs 1 : 2 in general population (8)) and the mean age ± SD was 46 ± 16 years (range 12–90) with no difference between genders. A total of 64% (234/362) of patients had migraine aura with headache in every attack, 30% (107/362) had attacks of both migraine aura with and without headache, and 6% (21/362) had exclusively migraine aura without headache. Males had exclusively aura without headache more often than females (11%; 11/99 vs 4%; 10/263, P = 0.008, χ2 test).
Age at onset and age at cessation of attacks
The age at onset of aura with headache was 21 ± 12 years (mean ± SD) (range 5–77) and approximately followed a unimodal normal distribution in both males and females. The previous Danish population-based study revealed a nonsignificant bimodal distribution of the age at onset composed of two normal distributions and different in males and females (1). The age at onset was analysed separately in each family: Exclusively the three families each comprising ≥12 NHMA patients had a lower mean age at onset than the remainder families. The age at onset was 15 ± 6 years (mean ± SD) in patients from families with ≥12 NHMA patients compared to 21 ± 12 years in patients from families with <12 HNMA patients (P = 0.001, Mann–Whitney U-test). The age at onset of aura without headache in patients exclusively with aura without headache was 36 ± 15 years (mean ± SD) (range 13–73) in both males and females and was higher than the age at onset of aura with headache (P < 0.0001, Mann–Whitney U-test). The age at onset of aura without headache was 40 ± 15 years (mean ± SD) in patients who previously had attacks of aura with headache.
The age at cessation of attacks of aura with headache (defined as no attacks for at least two years) was nonsignificantly higher in females than males (40 ± 15 vs 34 ± 16 years (mean ± SD), n = 92) and was not influenced by the age at onset. In females from families with ≥12 NHMA patients the age at cessation was nonsignificantly higher than in families with <12 NHMA patients (54 ± 9 vs 39 ± 15 years (mean ± SD), n = 59); and correspondingly in males (40 ± 20 vs 33 ± 14 years (mean ± SD), n = 33). The age at cessation of aura without headache was nonsignificantly higher in males than females (49 ± 20 vs 32 ± 11 years (mean ± SD), n = 13). Among patients with aura with and without headache 29% (31/107) had cessation of attacks of aura with headache but 77% (24/31) continued to have attacks of aura without headache.
Lifetime number of attacks and attack frequency
The lifetime number of NHMA attacks is shown in Table 1. The lifetime number of attacks in patients exclusively with aura without headache (n = 21) was 2–4 attacks in 5 patients, 5–9 attacks in one patient, 10–49 attacks in 7 patients, 50–100 attacks in 6 patients, and >100 attacks in two patients. The attack frequency of aura with headache (n = 337) within the last year was no attacks in 31%, vs 34% in population-based study (8); 1–6 attacks in 48%, vs 53% (8); 7–12 attacks in 8%, vs 8% (8); 13–24 attacks in 7%, vs 5% (8); 25–36 attacks in 2%, vs 0% (8); >36 attacks in 4%, vs 0% (8) of patients.
Number of attacks of nonhemiplegic migraine with aura (NHMA), pain characteristics and accompanying symptoms

Subjects exclusively with migraine aura without headache not included.
Subjects with valid values for statistical analyses.
The data were recorded in the semistructured interview as categorical data.
Significantly different values between males and females (P < 0.05).
Co-occurrence of migraine with aura and other headaches
The co-occurrence of migraine without aura (MO) was 20% (20/99; 95% CI 12–28%) in males and 35% (92/263; 95% CI 32–41%) in females and higher than in the population-based study where the co-occurrence was 11% (95% CI 4–17%) in males (P = 0.058, χ2 test) and 16% (95% CI 7–25%) in females (P = 0.0028, χ2 test) (1) with NHMA. A total of 3% (10/362) of the NHMA patients had migrainous disorder without aura (IHS 1.7), 43% (155/362) had episodic tension-type headache (vs 66% of all persons in the general population) (14), 1.4% (5/362) had chronic tension-type headache (vs 3%) (14), and 0.6% (2/362) had episodic cluster headache (vs 0.1%) (14).
Headache and accompanying symptoms related to aura
The headache characteristics and accompanying symptoms are shown in Table 1. Males had more attacks of 30 min−4 h duration (P = 0.021, χ2 test) and fewer attacks of 24–72 h duration (P = 0.016, χ2 test) than females. More females than males had unilateral (P = 0.0002, χ2 test) or pulsating headache (P = 0.0095, χ2 test) or phonophobia (P = 0.0005, χ2 test). Generally the headache related to aura fulfilled the IHS criteria of MO regarding number of attacks in 94% (317/338; vs 89% in population-based study) (1), duration in 69% (234/339; vs 72%) (1), pain characteristics in 90% (293/327; vs 92%) (1), and accompanying symptoms in 84% (285/340; vs 86%) (1) of patients. Overall the headache and accompanying symptoms fulfilled the IHS criteria of MO in 57% (189/329) of patients (vs 95% in population-based study (8), P < 0.0001, χ2 test) – nonsignificantly more often in females than males.
The headache began after the onset of the aura in 82% (278/341) of patients vs 93% in population-based study (7), during the aura or ≤ 30 min after the cessation of the aura in 96% (266/278), 30–60 min after in 4% (11/278) and 60–120 min after in <1% (9/278) of patients. The headache began simultaneously with the aura in 11% (37/341) of patients vs 5% (7). The headache began before the onset of the aura in 8% (26/341) of patients vs 3% (7); ≤ 30 min before in 85% (22/26), 30–60 min before in 12% (3/26) and 60–120 min before in 4% (1/26) of patients. The sequence of headache and aura was different from the results of the population-based study (7) (P = 0.0036, χ2 test on three groups).
Distribution of aura symptoms
The distribution of the various aura symptoms are shown in Fig. 3. Visual aura occurred at least in some attacks in 99% (358/362; 95% CI 98–100%) of patients vs 99% (95% CI 97–100%) in the population-based study (7), sensory aura in 54% (196/362; 95% CI 49–59%) vs 31% (95% CI 24–38%, P < 0.0001, χ2 test) (7) and aphasic aura in 32% (116/362; 95% CI 27–37%) vs 18%, 95% CI 12–24%, P = 0.0007, χ2 test) (7). Nine patients from the population-based study had motor aura (7) and were consequently excluded from the above calculations and Fig. 3. When more than one aura symptom was observed, they occurred in succession in 96% (149/155) and simultaneously in 4% (6/155) of patients (65 missing values or patients uncertain). The distribution of the various aura symptoms was the same in patients from the large and small families. Table 2 shows the percentage of attacks with occurrence of specific aura symptoms. Visual aura occurred in almost every attack whereas sensory and aphasic aura occurred in only a fraction of an individual's total number of attacks. The results were not significantly different from the results of the population-based study (7). The aura symptoms of males and females are not presented separately since no differences by gender were found in the population-based study (7). Aura without headache occurred in 35% (128/362) of patients (vs 42% in population-based study) (7) and in 16% (21/128) of these patients vs 10% (7) aura occurred without headache in every attack.

Venn diagram illustrating the distribution of the various aura symptoms in 362 patients with nonhemiplegic migraine with aura (NHMA) from the present study (bold) and 154 patients from a Danish population-based study (1) (not bold).
The percentage of attacks with occurrence of specific aura symptoms

Sensory aura occurred in 196 patients but values on the percentage of attacks were missing in 24 subjects.
Aphasic aura occurred in 116 patients but values on the percentage of attacks were missing in 29 subjects.
Visual aura
The characteristics of the visual aura are shown in Table 3. The visual aura was predominantly unilateral/homonymous with flickering light, scotoma or zig-zag lines. The visual aura more often started peripherally than centrally in the visual field (P < 0.0001, χ2 test, only valid values included) and comprised a scotoma (P < 0.0001, χ2 test) but less commonly had preserved central vision (P < 0.0001, χ2 test) and zig-zag lines (P < 0.0001, χ2 test) than in the population-based study (7). The visual aura developed gradually in 81% (280/345) of patients vs 97% in population-based study (7), (P < 0.0001, χ2 test) with a mean ± SD duration of the gradual development of 18 ± 13 min (range 3–60 min). The duration of the gradual development was more often ≤30 min than in the population-based study (7) (P = 0.032, χ2 test). Among the 65 patients classified as acute onset visual aura two patients reported a gradual development of 3 min duration. Among the remainder 63 patients 30 patients had aphasic or sensory aura developing gradually or occurring in succession with the visual aura. The median duration of the visual aura was 30 min (interquartile range (IQR) 15–45 min, range 2–360 min) and was not influenced by the presence of other aura symptoms.
Visual aura symptoms

Subjects with valid values for statistical analyses.
The data were recorded as numerical data.
Significantly different values (P < 0.05).
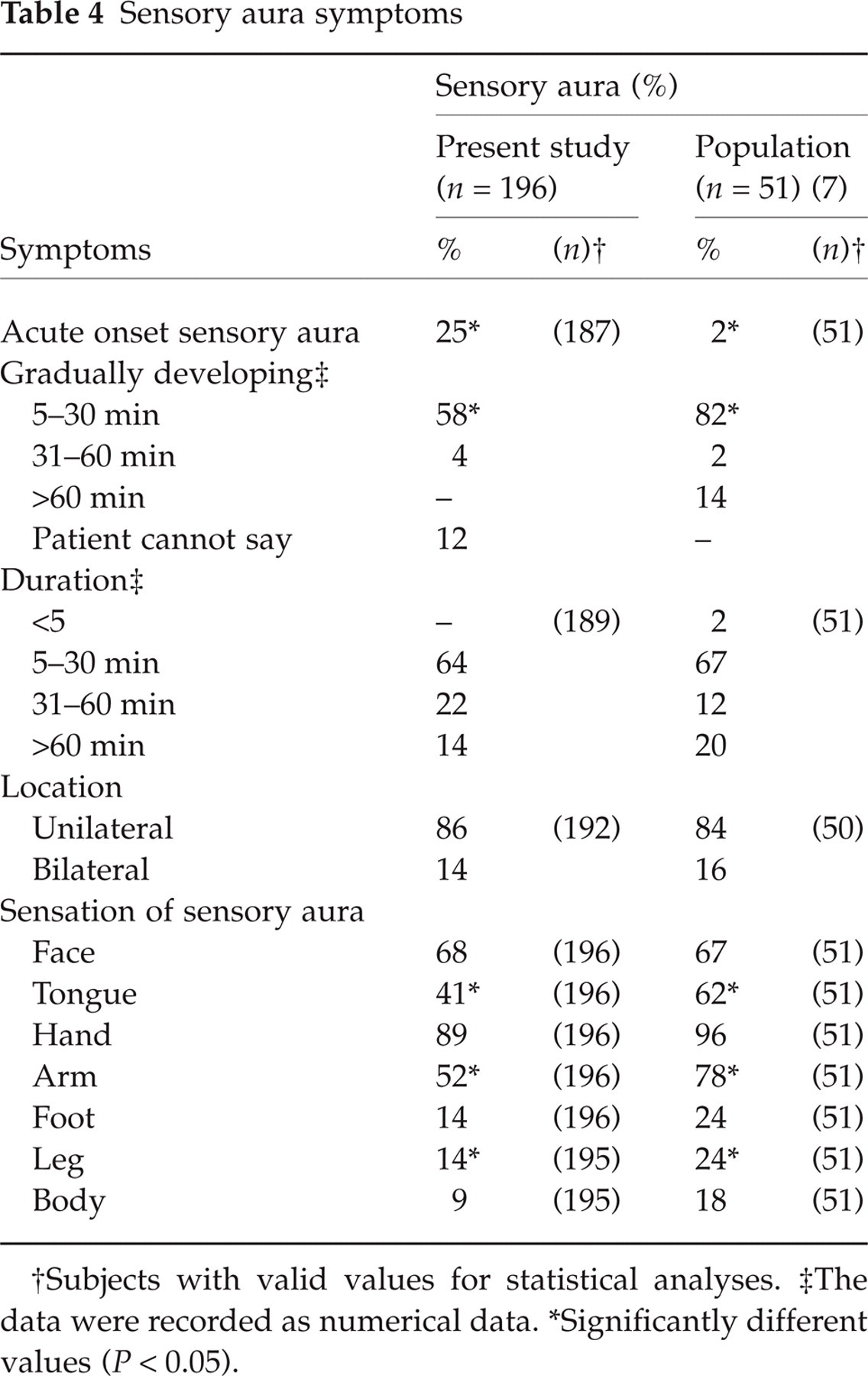
Sensory aura
The characteristics of the sensory aura are shown in Table 4. The sensory aura was predominantly unilateral and affected the face and hand. The sensory aura less commonly affected the tongue (P < 0.012, χ2 test), arm (P < 0.0007, χ2 test) and leg (P < 0.011, χ2 test) than in the population-based study (7). The sensory aura developed gradually in 75% (140/187) of patients vs 98% in the general population (7) (P < 0.0003, χ2 test) with a mean ± SD duration of the gradual development of 19 ± 15 min (range 5–60 min). The duration of the gradual development was more often ≤30 min than in the population-based study (7) (P = 0.065, χ2 test). Among the 47 patients classified as acute onset sensory aura one patient reported a gradual development of 2 min duration. Among the remainder 46 patients 43 patients had a visual aura developing gradually or occurring in succession with the sensory aura. The median duration of the sensory aura was 30 min (IQR 9–51 min, range 5–999 min).
Sensory aura symptoms
Subjects with valid values for statistical analyses.
The data were recorded as numerical data.
Significantly different values (P < 0.05).
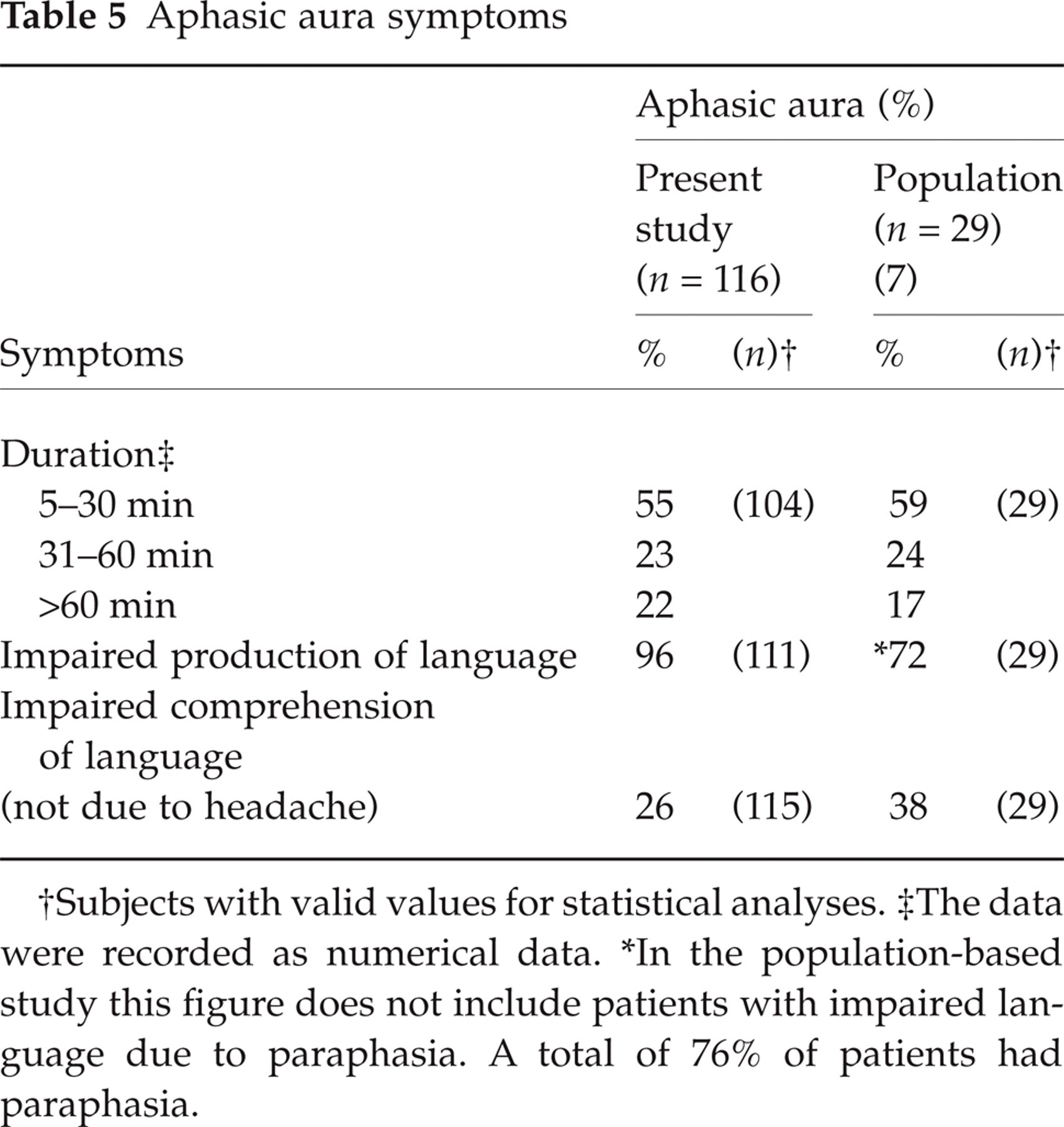
Aphasic aura
The characteristics of the aphasic aura are shown in Table 5. Aphasic aura was defined as impaired production of language (not due to dysarthria) or impaired comprehension of language. Most patients with impaired production of language had paraphasia. Dysarthria was experienced by 46% (53/116) of patients with aphasic aura (52% in population-based study) (7) and by 15% (16/104) of patients with sensory aura but without aphasic aura. The median duration of the aphasic aura was 30 min (IQR 8–53 min, range 5–1440 min) vs a mean ± SD of 43 ± 43 min in population-based study (7).
Aphasic aura symptoms
Subjects with valid values for statistical analyses.
The data were recorded as numerical data.
In the population-based study this figure does not include patients with impaired language due to paraphasia. A total of 76% of patients had paraphasia.
Basilar-type aura
In total, 10% (35/362) of patients had at least two of the following basilar-type aura symptoms: bilateral visual symptoms, dysarthria, vertigo, tinnitus, decreased hearing, double vision, bilateral parestesias or decreased level of consciousness. The telephone interview was not considered valid for diagnosing ataxia. Patients with basilar-type symptoms were equally distributed among the families and among the large and small families.
Migraine aura without headache
Among the patients exclusively with aura without headache 76% (16/21) had exclusively visual aura, 10% (2/21) had visual and sensory aura, and 14% (3/21) of patients had visual, sensory and aphasic aura. Among the patients with attacks of both aura with and without headache, in attacks of aura without headache 83% (98/107) had exclusively visual aura vs 84% in population-based study (7), 9% (10/107) had visual and sensory aura vs 11% (7), 3% (3/107) had visual sensory and aphasic aura vs <1% (7), <1% (1/107) visual and aphasic aura vs <1% (7), and 4% (4/107) of patients had exclusively sensory aura vs <1% (7). Among the patients with attacks of both aura with and without headache 97% (101/104) of patients who had visual aura related to headache had visual aura without headache in attacks of aura without headache, 28% (17/61) of patients who had sensory aura related to headache had sensory aura without headache in attacks of aura without headache, and 3% (2/74) of patients who had aphasic aura related to headache had aphasic aura in attacks of aura without headache. The median duration of the visual aura in patients exclusively with aura without headache was 18 min (IQR 13–22 min, range 5–120 min) vs a mean ± SD of 25 ± 13 min in population-based study (7) and was shorter than the median duration of 30 min in patients with attacks of aura with headache (P = 0.003, Mann–Whitney U-test). The aura characteristics did not differ in other aspects.
Discussion
The present study is the so far largest study using a validated, physician conducted semistructured interview presenting the clinical characteristics of a clinic based sample of patients with nonhemiplegic migraine with aura (NHMA). Our findings are in agreement with the main clinical characteristics of NHMA in previous studies and additionally demonstrate the distinctive characteristics of familial NHMA. The participants did not have a physical or neurological examination since previous studies have shown that this rarely alters the diagnosis of migraine in epidemiological studies (14). The fact that the present study and the previous Danish population-based studies used for comparison (1, 7, 8) were done in the same headache centre using similar diagnostic tools ensures a high degree of validity when comparing the clinical data from these studies. Studies using lay interviews or self-administered questionnaires can be valuable for a number of purposes but are not valid for this type of analysis. The previous Danish population-based studies (1, 7, 8) included nine and six patients with hemiplegic migraine (HM), respectively. Since it has subsequently been shown that HM patients may have symptoms different from NHMA patients (11, 12, 16) this may cause a small bias. However, this would be in the direction of increasing the severity in the previous materials and would thus diminish the observed difference between the previous and the present studies. Moreover, the recording of symptoms in the studies was retrospective and therefore implies a risk of recall bias.
Clinical implications of our findings
The characteristics of typical NHMA attacks and atypical variations of NHMA attacks are highlighted in Tables 6, 7. In clinical practice it is important to know the clinical features that prompt further investigation. We found that visual aura occurred in almost every attack of NHMA and often in isolation whereas sensory and aphasic aura mostly occurred in combination with visual aura. Therefore, other diagnoses should be considered in patients with parestesias and/or aphasia without visual symptoms. In aura without headache, visual aura occurred primarily in isolation. Thus, multiple aura symptoms without headache may require diagnostic tests. The findings confirm the results of a population-based study (7) and other studies (2, 3, 17). The age at onset of aura without headache was higher than in aura with headache. Furthermore, many patients with aura with headache experienced a cessation of their headache but continued to have attacks of aura without headache. That is, aura without headache is often seen in the older segment of NHMA patients and more often in males. The differential diagnostic possibilities should always be considered in patients without headache.
Characteristics of typical nonhemiplegic migraine with aura

Characteristics of atypical but likely variations of nonhemiplegic migraine with aura and symptoms requiring further investigations

The aura characteristics determine the diagnosis of NHMA according to the operational diagnostic criteria of the International Headache Society (IHS) (15). The NHMA criteria are based on the description of the aura (gradual development ≥5 min and duration 5–60 min) and on the free interval to the following headache (≤60 min). Therefore, the analyses of these aura characteristics are of special interest. Our patients reported acute onset of an aura symptom more often than reported in the population-based study (7) and other studies (18, 19). However, if the patients reporting acute onset of an aura symptom had additional aura symptoms they mostly had a gradual development or occurred in succession. Prospective recordings have previously reduced but not eliminated acute onset aura (18, 19). The free interval to the headache was less than one hour in all but one patient with aura with headache.
The distribution of the sensory aura was less widespread in patients from the present study than in patients from the population-based study (7) but the finding is probably biased by the fact that the population-based study included patients with HM (12). The headache related to aura reportedly begins after the aura (7, 18, 19) but in the present study the headache began before the aura in a few patients. In these cases the headache was however, often mild before the onset of aura and worsened after the aura. It is therefore possible that these patients had a tension-type headache before the aura but a mild migraine cannot be excluded.
Scientific implications of our findings
The present study has implications regarding future molecular genetic studies. NHMA is a multifactorial, genetically heterogeneous disorder with a complex mode of inheritance (20, 21) but the genes involved have not yet been identified (22). Examining the age at onset of NHMA may give clues to underlying differences in genetic aetiology. The mean age at onset of NHMA in our patients was influenced by the probable genetic load in the family, i.e. patients had an earlier age at onset in families with many affected members than in families with few affected members. The large families were expanded to the younger generations more often than the small families, but repeating the test for statistical significance of our result excluding patients below age 38 (arbitrary value of the mean age minus half the value of the SD) does not change the significance. Our results show the existence of NHMA families with an early age at onset. The overall mean age at onset of familial NHMA was similar to the results from other clinic materials (3, 23, 24) but our findings did not correspond to the bimodal distribution of age at onset reported in one of the Danish population-based studies (1). Stratifying NHMA patients by age at onset may be considered as an approach in future genetic studies. By narrowing the definition of the trait one will be including only individuals likely to have a similar genetic aetiology leading to the disease phenotype (25). The age at cessation of NHMA attacks was higher in patients from large families than in patients from small families. The result may be biased by the fact that the mean age of the patients was nonsignificantly higher in the large families than in the small families but variation in age at cessation of attacks more likely reflects underlying differences in genetic aetiology.
Regarding phenotype delineation for genetic studies it is important to consider the severity of a disease. Patients with familial NHMA had more sever attacks, i.e. they experienced a combination of aura symptoms more often than patients from the population-based study (1) and from other selected patient materials where sensory aura has been reported in only 26–40% of NHMA patients (vs 54% in present study) and aphasia in 9–20% (vs 32% in present study) (2, 3, 17). The lifetime number of NHMA attacks of patients from the present study was not directly comparable to the figures from the population-based studies but our results suggest that patients with familial NHMA did not have more attacks than patients with migraine with aura (MA) from the general population (7, 8). Our finding, that patients with familial NHMA had more severe, though not more frequent, NHMA attacks may reflect that individuals with familial NHMA represent extreme phenotypes more likely to be the result of genetic influence (25). In future genetic studies of NHMA stratifying patients by the severity of aura may be considered (extreme genetics). This perspective is supported by the fact, that NHMA patients with sensory or aphasic aura besides their visual aura have a more unfavourable prognosis than patients exclusively with visual aura (26–28). Subgroups of NHMA patients with an unfavourable prognosis may be highly influenced by inherent factors and lesser influenced by environmental factors.
Different NHMA characteristics in males and females may give clues to underlying differences in aetiology as well. The prognosis of NHMA is less favourable in females than males (26, 28) and the prevalence of NHMA is higher among females than males at all ages (7, 8, 29, 30). We found, that the age at cessation of NHMA was higher in females than males (not biased by age differences). Furthermore, the headache related to aura was more severe in females than in males who also more often had exclusively aura without headache. The genetic constitution necessary to express the NHMA phenotype may thus be different in males and females suggesting to analyse them separately for example in genetic association studies.
The co-occurrence of migraine without aura (MO) and NHMA was higher in patients with familial NHMA than in MA patients from the general population. Our results agree with a previous clinic material (3) and are probably caused by a higher load of genes involved in both NHMA and MO in these more severely affected patients.
In conclusion, the clinical characteristics of familial NHMA differ in several aspects from MA in the general population. Generally there seems to be a correlation between more severe symptoms and familial aggregation and our results may be helpful in clinical diagnosis as well as in future genetic studies.
