Abstract
The authors studied the differences between heat-shock/stress protein 70 (hsp70) gene expression and protein synthesis in the unilateral middle cerebral artery (MCA) microsurgical direct occlusion (Tamura's) model and the unilateral intraluminal occlusion model. In Tamura's model, expression of hsp70 mRNA and HSP70 protein and decreased protein synthesis were detected in the ischemic areas, including the ipsilateral cortex and caudate. These phenomena, however, were not observed in the areas outside the MCA territory, including the ipsilateral thalamus, hippocampus, and substantia nigra. These results were consistent among the experimental rats. In the intraluminal occlusion model, however, induction of both hsp70 mRNA and HSP70 protein and impairment of protein synthesis were noted in the areas outside the MCA territory, including the ipsilateral thalamus, hypothalamus, hippocampus, and substantia nigra, as well as in the MCA territory, including the ipsilateral cortex and caudate. These results were not consistent among the experimental rats. These different results might be due to widespread damage resulting from internal carotid artery (ICA) occlusion in the intraluminal occlusion model. Accordingly, the authors suggest that this model be called an ICA occlusion model, rather than a pure MCA occlusion model.
The most commonly encountered type of stroke in humans is cerebral ischemia. Occlusion of a single arterial trunk, particularly the middle cerebral artery (MCA), is the most frequent of the many causes of cerebral ischemia. Therefore, many cerebral ischemic models in nonhuman primates (Harvey and Rasmussen, 1951; Hudgins and Garcia, 1970; Sundt and Waltz, 1966), cats (O'Brien and Waltz, 1973) and rats (Albanese et al., 1980; Hirakawa et al., 1994; Koizumi et al. 1986; Longa et al. 1989; Robinson, 1981; Shigeno et al., 1985; Tamura et al., 1981) have been developed in order to investigate the pathophysiology of cerebral ischemic injury and the efficacy of various treatment modalities. Two procedures, microsurgical direct occlusion of the MCA by electrocoagulation (Tamura's model) and intraluminal thread occlusion of the MCA (intraluminal occlusion model), are now widely used methods of MCA occlusion in rats. There is no published report, however, concerning the comparison of these two models.
Heat-shock/stress proteins (HSPs) are various proteins encoded by specific groups of genes that are not constitutively expressed (Lindquist, 1986). After transient ischemia in the gerbil, HSPs are synthesized and cells survive, even though these cells stop synthesizing other proteins (Nowak, 1990, 1993). However, the interpretation of HSP70 protein expression in the rat is subtly different from that in the gerbil. In the rat, Gonzalez et al. (1989) observed that HSP72 immunostaining was induced in and around areas of infarction after focal cerebral ischemia and suggested that HSP72 immunohistochemistry may be used as a marker of cellular injury. Li et al. (1992) also suggested that HSP70 protein expression might be a sensitive and early marker of ischemic cell damage.
It is known that induction of protein synthesis in neurons exposed to the direct effect of ischemia is strongly suppressed after cerebral ischemia (Kirino and Sano, 1984; Kleihues and Hossmann, 1971; Thilmann et al., 1986). Hata et al. (2000
In this article we describe the differences between expression of hsp70 mRNA and HSP70 protein, and protein synthesis in Tamura's model and the intraluminal occlusion model. The aim of this study was to clarify the differences in the extent of primary ischemic damage in these two models, particularly from the standpoint of the cerebrovascular anatomy.
MATERIALS AND METHODS
The Animal Research Committee of Teikyo University School of Medicine approved all the experimental protocols. Male Wistar rats weighing 250 to 350 g were assigned to the following experimental groups: normal (untreated), sham operation (controls), Tamura's model, and the intraluminal occlusion model.
Tamura's model
The MCA coagulation was carried out using the method of Tamura et al. (1981), which was modified and involved a transretroorbital approach (Tamura et al., 1986a; Yamamoto et al., 1988). Anesthesia was induced with 4% halothane and maintained during the operation with 2% halothane in 30% oxygen and 70% nitrogen delivered via a facemask. Each animal was placed in a lateral position and a curved, vertical, 2-cm skin incision was made at the midpoint between the left orbit and the external auditory canal. Under an operating microscope, the incised skin and temporal muscle were simply retracted pos-teroinferiorly, a small craniectomy was made, and the stem of the MCA was electrocauterized and cut to ensure vascular occlusion was complete. The soft tissues were allowed to fall back into place and the skin was sutured. Each operation lasted approximately 10 minutes. During the operation, the body temperature was regulated as close to 37.5°C as possible by means of a servo-controlled heat pad and a heat lamp.
Intraluminal occlusion model
We used the 2-hour occlusion model because mortality in the permanent intraluminal occlusion model was very high in a preliminary experiment. The MCA occlusion was performed using a slightly modified method of Koizumi et al. (1986). In brief, anesthesia was induced and maintained as described previously. The right common carotid artery was exposed through a midline incision, the external carotid artery (ECA) was dissected further distally, the internal carotid artery (ICA) was isolated and carefully separated from the adjacent vagus nerve, and the pterygopalatine artery was ligated. A 5-cm length of nylon suture (number 0.8, 0.148 mm in diameter), one tip of which was covered with silicone filler to form an embolus 0.2 or 0.3 mm in diameter and 5-mm long and the other of which was rounded using a soldering iron to form a stopper, was then gently advanced from the ECA to the ICA lumen. After occlusion for 2 hours, MCA blood flow was restored by removing the nylon suture. Each operation lasted approximately 15 minutes, and body temperature was regulated during the operation as described previously.
In situ hybridization
Anesthesia was induced with pentobarbital (50 mg/kg body weight) 4 hours and 1 and 4 days after ischemia. The animals (Tamura's model, n = 4 per group; intraluminal occlusion model, n = 3 per group) were fixed by transcardiac perfusion with 0.9% (w/v) saline followed by 4% paraformaldehyde in 0.1-mol/L phosphate buffer (pH 7.4). Brains were postfixed with the same fixative for 1 hour at 4°C, removed, transferred to 0.1-mol/L phosphate-buffered saline (PBS, pH 7.4) containing 20% sucrose overnight at 4°C, and stored at −80°C until analyzed.
Brain sections (15-μm thick) containing the striatum, thalamus, or substantia nigra were cut on a cryostat at −20°C, collected on gelatin-coated slides, and processed for
Immunohistochemistry
Anesthesia was induced with pentobarbital (50 mg/kg body weight) 4 hours and 1 and 4 days after ischemia. The animals (Tamura's model, n = 4 per group; intraluminal occlusion model, n = 3 per group) were fixed and their brains were postfixed and removed, as described previously, and transferred to 0.1-mol/L PBS (pH 7.4) and stored at 4°C until required for use. Brain sections (50-μm thick) containing the striatum, thalamus, or substantia nigra were cut using a vibratome and immunostained using the procedure described by Vass et al. (1988). We used a mouse monoclonal antibody raised against human HSP70-related protein (RPN 1197), biotinylated antimouse IgG (RPN 1001), and streptoavidin-biotinylated peroxidase complex (RPN 1051; Amersham Bio-sciences Corp., Piscataway, NJ, U.S.A.) and diaminobenzidine (Sigma Chemical, St. Louis, MO, U.S.A.). We then confirmed the specific and nonspecific expression in the cells using light microscopy (Table 1).
Expression of HSP70 protein on the ipsilateral and contralateral sides
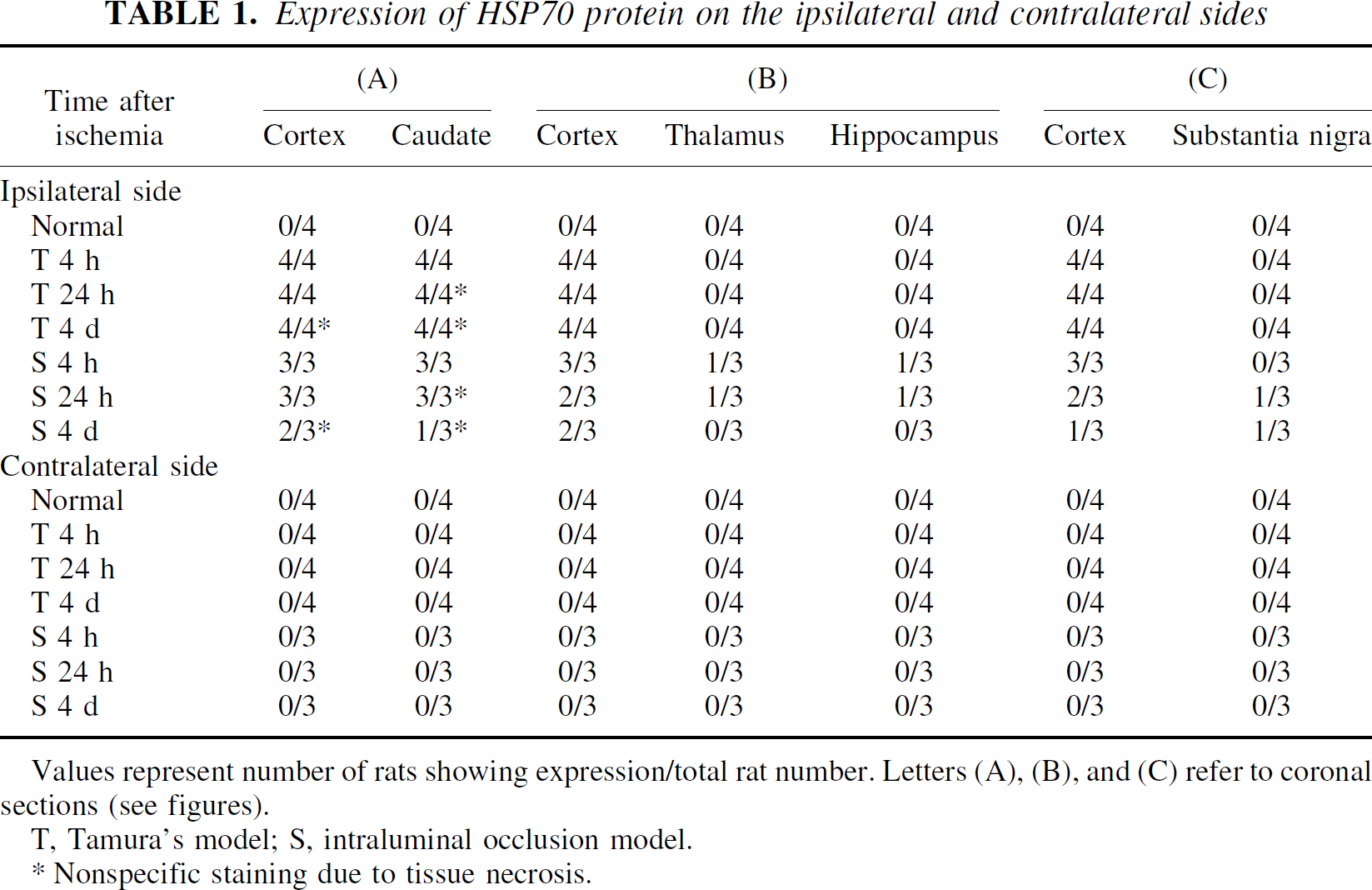
Values represent number of rats showing expression/total rat number. Letters (A), (B), and (C) refer to coronal sections (see figures).
T, Tamura's model; S, intraluminal occlusion model.
Nonspecific staining due to tissue necrosis.
Protein synthesis
Autoradiography to quantify protein synthesis was performed according to the method of Thilmann et al. (1986). Solvent from L-[2,3−3H]valine (PerkinElmer Life Science Products, Boston, MA, U.S.A.) was evaporated and dissolved in 0.9% saline to produce a concentration of 5 mCi/mL. Three hours before isotope administration, each rat was anesthetized with pentobarbital (50 mg/kg) and a polyethylene catheter was inserted into a femoral vein, as described by Nakagomi et al. (1993). Six hours after MCA occlusion, 13 animals (Tamura's model, n = 8; intraluminal occlusion model, n = 5) each received a single intravenous injection of a tracer dose of L-[2,3- H]valine (6.7 mCi/kg). The rats were anesthetized with pentobarbital and perfused with ice-cold isotonic saline to remove the blood from the cerebral vasculature. The brains were fixed with 4% (w/v) formaldehyde solution containing 0.5% (w/v) trichloroacetic acid and 0.1% (w/v) unlabeled L-valine at 4°C and postfixed in the same fixative at 4°C overnight. Brains were removed and placed in fresh fixative solution, and the acid-soluble precursors were removed by repeatedly changing the fixative. Finally, the brains were dehydrated and paraffin embedded using standard techniques, and 5-μm-thick coronal sections containing the striatum, thalamus, or substantia nigra were autoradiographed using Amersham Hyper- H film, which was exposed for 2 months. Untreated normal rats (n = 2) and rats subjected to sham operation (n = 4) served as controls.
RESULTS
In situ hybridization
No hsp70 mRNA was detected in any brain areas containing the cortex, caudate putamen, thalamus, hypothalamus, hippocampus, or substantia nigra of the four control rats (Table 2, Fig. 1).
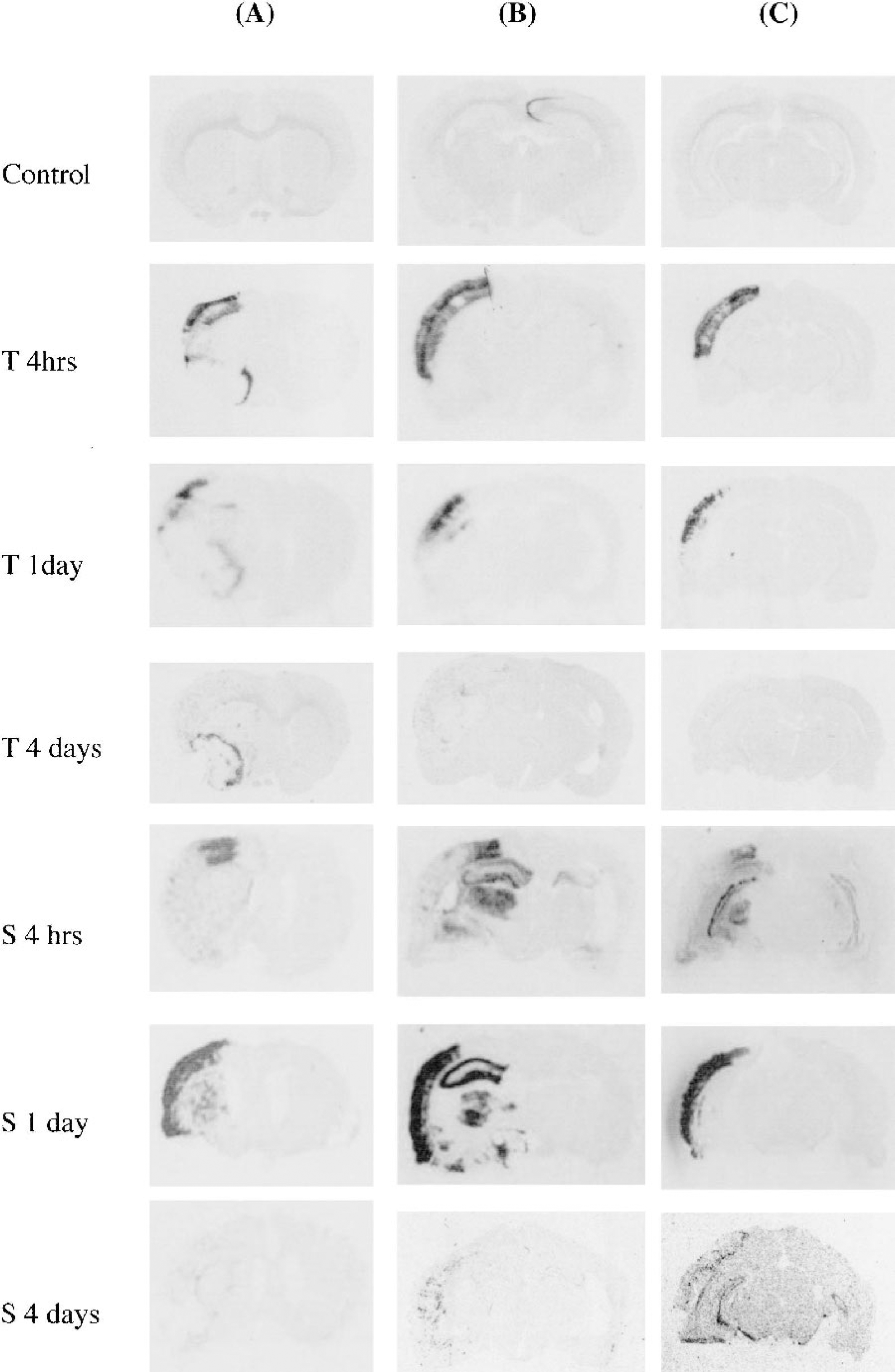
Induction of hsp70 mRNA in coronal sections containing
Expression of heat-shock protein 70 mRNA on the ipsilateral and contralateral sides
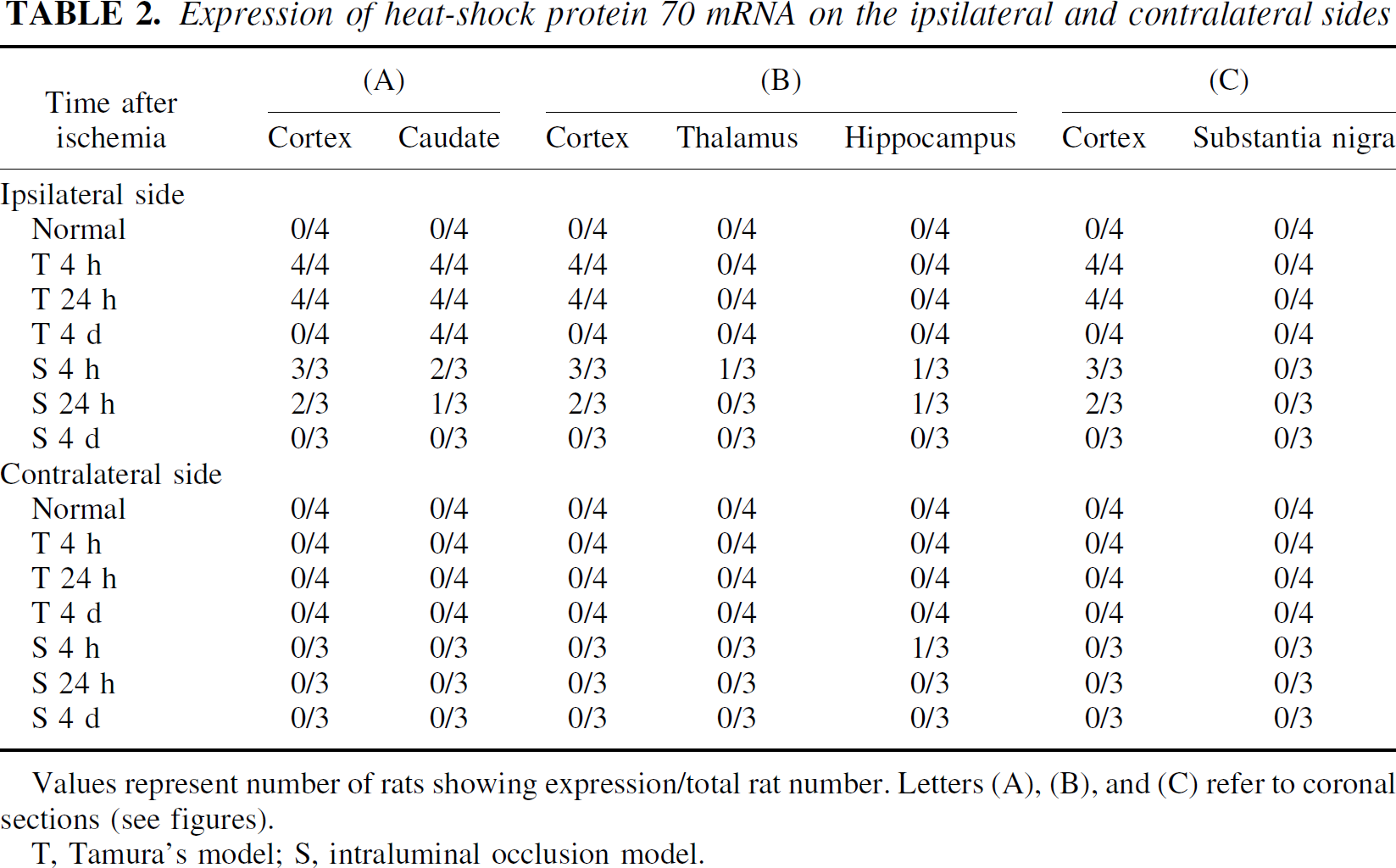
Values represent number of rats showing expression/total rat number. Letters (A), (B), and (C) refer to coronal sections (see figures).
T, Tamura's model; S, intraluminal occlusion model.
Tamura's model
Induction of hsp70 mRNA was consistent among the experimental rats (Table 2). Four hours (n = 4) and 1 day (n = 4) after MCA occlusion, hsp70 mRNA induction was detected in the ipsilateral cortex and caudate putamen of the MCA territory (Table 2, Fig. 1). Four days (n = 4) after occlusion, hsp70 mRNA induction was demonstrated only in the ipsilateral caudate and had disappeared from the cortex (Table 2, Fig. 1). No hsp70 mRNA induction was observed in the thalamus, hippocampus, or substantia nigra (Table 2, Fig. 1).
Intraluminal occlusion model
The pattern of hsp70 mRNA expression was not consistent among the experimental rats (Table 2). Four hours after occlusion, hsp70 mRNA induction was seen in the ipsilateral cortex of all three rats (Table 2). However, hsp70 mRNA was induced in the ipsilateral caudate putamen, but not in the thalamus, hippocampus, or substantia nigra, of two of these three rats (Table 2), whereas hsp70 mRNA was induced in the ipsilateral thalamus and the ipsilateral and contralateral hippocampi, but not in the substantia nigra of the other rat (Fig. 1). One day after occlusion, hsp70 mRNA induction was seen in the ipsilateral cortex, caudate putamen, thalamus, and hippocampus, but not in the substantia nigra, of one of the three rats (Fig. 1); one rat showed hsp70 mRNA induction only in the ipsilateral cortex and not in the caudate putamen, thalamus, hippocampus, or substantia nigra, and another showed no hsp70 mRNA induction in any brain area (Table 2, Fig. 1). Four days after ischemia, no hsp70 mRNA induction was detected in any brain area of the three rats (Table 2, Fig. 1).
Immunohistochemistry
In control rats (n = 4), no HSP70 protein induction was detected in any areas containing the cortex, caudate putamen, thalamus, hippocampus, or substantia nigra (Table 1, Fig. 2).
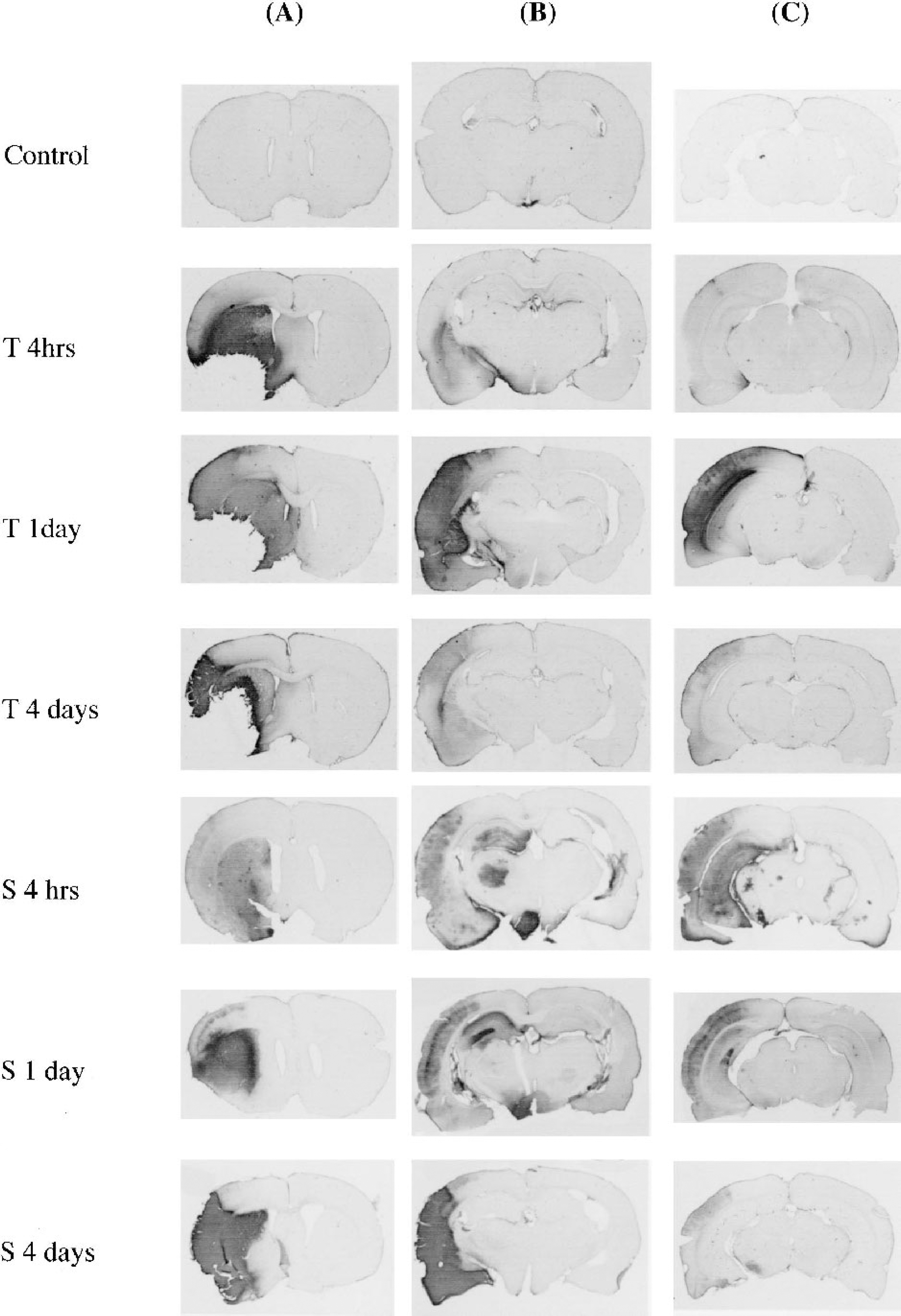
Immunostaining of HSP70 protein in coronal sections containing
Tamura's model
The pattern of HSP70 protein induction was consistent among the rats (Table 1). Four hours and 1 and 4 days after MCA occlusion, specific (neuronal and/or endothelial) and nonspecific HSP70 protein immunostaining was seen in the ipsilateral cortex and caudate putamen of the MCA territory (Table 1, Fig. 2), but not in the thalamus, hippocampus, or substantia nigra (Table 1, Fig. 2). Specific staining was induced in the caudate putamen 4 hours after ischemia and in the cortex 4 hour and 1 day after ischemia. Nonspecific staining due to tissue necrosis was observed in the caudate putamen 1 and 4 days after ischemia and in the cortex 4 days after ischemia.
Intraluminal occlusion model
The pattern of HSP70 protein induction in the cortex and caudate putamen was almost identical to that found in Tamura's model, but 4 days after occlusion no HSP protein induction was detected in any brain areas of one of the three rats. However, the pattern of HSP70 protein induction in the thalamus, hypothalamus, hippocampus, and substantia nigra was not consistent among the rats (Table 1).
Four hours after occlusion, HSP70 protein was induced in the ipsilateral hippocampus (CA1-CA4 and dentate gyrus), thalamus, and hypothalamus, but not in the substantia nigra of one rat (Fig. 2), and no HSP70 protein induction was detected in the thalamus, hippocampus, or substantia nigra of the other two rats. One day after occlusion, HSP70 protein induction was detected in the thalamus, hypothalamus, hippocampus (CA1-CA4 and dentate gyrus), and substantia nigra of one of the three rats (Table 1, Fig. 2). No HSP70 protein induction was detected in the thalamus, hippocampus, or substantia nigra of the other two rats. Four days after occlusion, HSP70 protein immunostaining was present in the substantia nigra, but not in the thalamus or hippocampus, of one of the three rats (Table 1, Fig. 2), whereas no HSP70 protein induction was detected in the thalamus, hippocampus, or substantia nigra of the other two rats.
Protein synthesis
Tamura's model
In the ischemic core, which included the lateral striatum and anterior cortex, incorporation of [3H]-valine into the brain was strongly suppressed (Fig. 3). No decrease in protein synthesis was observed in the brain areas outside the MCA territory, which included the thalamus, hippocampus, and substantia nigra (Fig. 3).
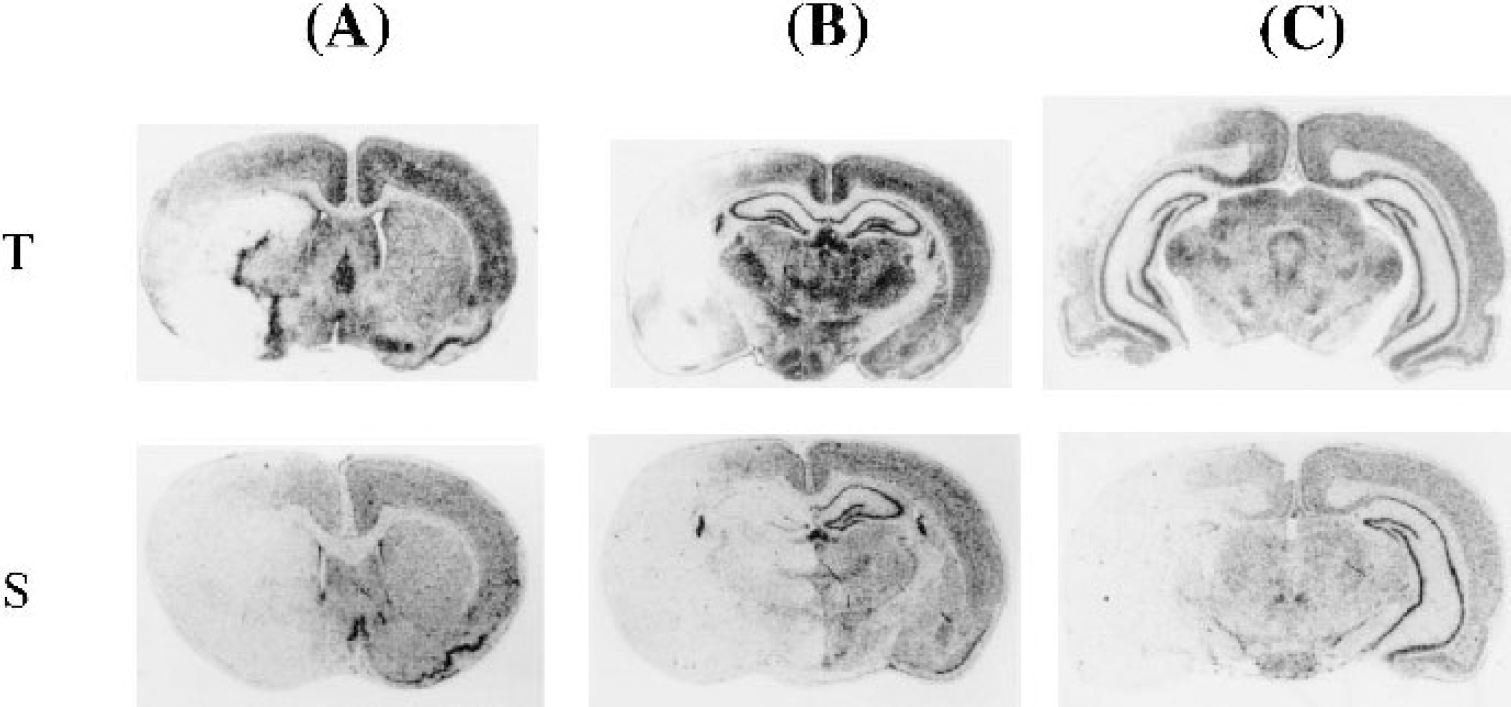
Incorporation of [3H]-valine in coronal sections containing
Intraluminal occlusion model
In all five animals, incorporation of [3H]-valine into the brain was strongly suppressed in the ischemic core, which included the lateral striatum and anterior cortex (Fig. 3). In four of the five animals, protein synthesis was suppressed in the brain areas outside the MCA territory, including the ipsilateral thalamus, hypothalamus, hippocampus, and substantia nigra, which are territories of branches of the ICA (Fig. 3).
DISCUSSION
We showed the differences between hsp70 mRNA and HSP70 protein expression, and protein synthesis in Tamura's and the intraluminal occlusion models. In Tamura's model, hsp70 mRNA and HSP70 protein expression was induced only in the MCA territory, never in brain regions outside it (Figs. 1 and 2). In the intraluminal occlusion model, hsp70 mRNA and HSP70 protein expression was simultaneously induced not only in the MCA territory, but also in brain regions beyond it, including the thalamus, hypothalamus, hippocampus, and substantia nigra. To date, there are many reports that show similar findings in the intraluminal occlusion model (Kinouchi et al., 1994; Li et al., 1992, 1993; States et al., 1996; Welsh et al., 1992). Proposed explanations for this phenomenon were that hsp70 mRNA and/or HSP70 protein expression in brain regions outside the MCA territory might be caused by secondary responses to ischemia, such as spreading depression (Nedergaard and Astrup, 1981), diaschisis (Feeney and Barron, 1986; Ginsberg et al., l977), deafferentation (Yamada et al., 1994), or transsynaptic responses (Kinouchi et al., 1994; States et al., 1996).
Recently, Sharp et al. (2000) reviewed mRNA and/or protein expression in areas outside the MCA territory, including the thalamus, hippocampus, and substantia nigra, after MCA occlusion in the intraluminal occlusion model. They pointed out that activation of entorhinal cortical inputs to the hippocampus could account for the induction of many genes in the hippocampus after MCA occlusion in the intraluminal occlusion model, and that gene induction in the other remote regions may be caused in part by acute ischemia-induced depolarization during the period immediately after a focal stroke. It is difficult to credit these explanations because they do not consider occlusion of the ICA in the intraluminal occlusion model.
The human posterior communicating artery (PcomA) is a slender blood vessel, not infrequently absent on one side, that joins the ICA and the terminal branch of the basilar artery. In rats, however, this vessel is of a comparable size to the middle and anterior cerebral arteries and anastomoses with the posterior cerebral artery (PCA) (Scremin, 1995). The PCA in rats, which arises from the terminal division of the basilar artery or from a trunk shared in common with the superior cerebellar artery and the proximal part of the PCA (which lies between the site of origin of the PCA and the site of its junction with the PcomA), is more slender than the PcomA. According to Brown (1966), functionally, the PcomA appeared to continue beyond its junction with the PCA and the PcomA appeared to produce an extension, providing a direct pathway for blood from the ICA to the caudal part of the cerebral hemisphere. Distal to joining the PcomA, the PCA ramifies into three trunks: the longitudinal hippocampal artery, the continuation of the PCA, and the transverse collicular artery (Coyle, 1975). The longitudinal hippocampal artery supplies branches that nourish the fascia dentate and hippocampus, as well as subicular and entorhinal structures. Terminal branches of the PCA supply some of the thalamic arteries, and the transverse collicular artery gives off perforating branches that supply the substantia nigra (Coyle, 1975). The hypothalamus is supplied by dorsomedially directed perforating vessels that originate from the PCA, ICA, and anterior cerebral arteries (Scremin, 1995). Therefore, the thalamus, hypothalamus, hippocampus, and substantia nigra are territories of the branches of the ICA, although the size of these branches varies from rat to rat. If occlusion of the ICA occurred in the intraluminal occlusion model, these areas could be involved in ischemia, according to the size of the vessels.
The present study clearly demonstrates that protein synthesis in brain areas outside the MCA territory in Tamura's and the intraluminal occlusion models differs. Decreased protein synthesis in the intraluminal occlusion model occurs not only in the MCA territory, but also in the brain region beyond it. Six hours after MCA occlusion, protein synthesis in the brain areas outside the MCA territory was suppressed in four of five animals subjected to the intraluminal occlusion procedure, and there was no decrease in protein synthesis in the former. Hata et al. (2000
Judging from these results, it would appear that hsp70 mRNA and HSP70 protein expression in the thalamus, hypothalamus, hippocampus, and substantia nigra during the acute ischemic phase in the intraluminal model is due to a direct ischemic insult and is not a secondary effect.
In previous studies we demonstrated atrophy of the ipsilateral thalamus and substantia nigra during the chronic phase more than 2 weeks after diathermic MCA occlusion (Fujie et al., 1990; Tamura et al., 1990). The most important difference between atrophy in remote areas and ischemic neuronal damage in the MCA territory is that tissue damage is never seen in the thalamus and substantia nigra during the acute phase of ischemia. Therefore, we concluded that these phenomena are caused by secondary ischemic neuronal damage, which takes a long time to develop. Thalamic atrophy after permanent MCA occlusion may be the result of retrograde degeneration (Fujie et al., 1990; Tamura et al., 1991), because loss of cerebral cortex and atrophy of the substantia nigra may be attributable to disinhibition hyperexcitation due to striatal damage (Saji and Reis, 1987; Tamura et al., 1990). There was a lag time between the phenomena caused by direct ischemic damage and secondary neuronal damage (Fujie et al., 1990; Saji and Reis, 1987; Tamura et al., 1990, 1991; Yamada et al., 1994). The results of this and other studies show that hsp70 mRNA and/or HSP70 protein expression in the thalamus, hippocampus, and substantia nigra occurs simultaneously with that in the cortex and caudate in the intraluminal occlusion model, indicating that hsp70 mRNA and/or HSP70 protein expression in these areas during the acute ischemic phase in the intraluminal model is due to a direct ischemic insult, not a secondary effect.
The infarcted area in Tamura's model is also different from that in the intraluminal thread model. In Tamura's model, the infarcted area was in the MCA territory only (Tamura et al., 1981,1986b), whereas in the intraluminal thread model the infarcted area was in the MCA territory, thalamus, hippocampus, and substantia nigra, which lie outside it (Koizumi, 1986; Memezawa et al., 1992a,b). As a thread is inserted into the MCA through the ICA in the intraluminal occlusion model, it is inevitable that the thread will influence the blood supply from the ICA to the PCA. Accordingly, taking into consideration that the size of infarction was not consistent in the intraluminal thread model, the influence depends on the shape and placement of the thread and the size of the communicating artery between the ICA and the PCA (McAuley, 1995).
The intraluminal occlusion model was recently developed and used in the mouse, and the same findings have been reported in mouse models (Barone et al., 1993; Hata et al., 2000a, b ; Kitagawa et al., 1998). There is a significant difference among mouse strains in terms of mortality and sensitivity to cerebral ischemia (Barone et al., 1993; Kitagawa et al., 1998). Intraluminal occlusion in C57/BL/6 mice resulted in a severe reduction of local cerebral blood flow, not only in the caudate putamen and the cerebral cortex, but also in the thalamus, hypothalamus, and hippocampus. Those mice subjected to intraluminal occlusion for 30 minutes showed neuronal death in the hippocampus, caudate putamen, and cerebral cortex after reperfusion (Kitagawa et al., 1998). Therefore, the vascular anatomy at the circle of Willis is highly variable in mice, and the patency of the PcomA is a crucial determinant of the ischemic effects of intraluminal occlusion (Barone et al., 1993; Kitagawa et al., 1998).
In summary, impairment of protein synthesis, induction of hsp70 mRNA, and induction of HSP70 protein expression were noted in brain areas outside the MCA territory, including the ipsilateral thalamus, hypothalamus, hippocampus, and substantia nigra, as well as in the MCA territory in some animals of the intraluminal occlusion model. In Tamura's model, impairment of protein synthesis and hsp70 gene expression were never observed in these areas outside the MCA territory during an acute phase of ischemia. Therefore, we consider that the thalamus, hypothalamus, hippocampus, and substantia nigra are included in the ischemic areas, not in remote or nonischemic areas, in the intraluminal occlusion model. In view of these results, we strongly believe that the intraluminal occlusion model should be referred to as an ICA occlusion model, not an MCA occlusion model.
Footnotes
Acknowledgements
The authors thank Dr. K. Takagi for scientific help and Ms. N. Kishino, Ms. K. Tatebe, and Ms. T. Iwasawa for their excellent assistance.
