Abstract
In acute ischemic stroke, the diffusion-weighted imaging (DWI) lesion is widely held to represent the core of irreversible damage and is therefore crucial in selecting patients for thrombolysis. However, recent research suggests it may also represent penumbra. An illustrative patient was imaged 7 hours after stroke onset with back-to-back 3T diffusion tensor imaging and quantitative positron emission tomography, which showed a DWI lesion and misery perfusion, respectively. Using previously validated voxel-based probabilistic CBF, CMRO2, and Oxygen Extraction Fraction (OEF) thresholds, the authors show that the DWI lesion contained not only core but also substantial proportions of penumbra. Also, severe apparent diffusion coefficient reductions were present within the potentially salvageable penumbra as well as in the core. These findings have potential implications regarding treatment decisions.
In acute ischemic stroke, the hyperintense diffusion-weighted imaging (DWI) lesion has been widely held to represent irreversibly damaged tissue (i.e., “core”), and is therefore critical in selecting patients for thrombolysis (Fisher, 1997;Hasegawa et al., 1994). However, recent research suggests it may also represent reversible ischemia (“penumbra”), and an absolute apparent diffusion coefficient (ADC) threshold for infarction may not exist in the early hours (Fiehler et al., 2002;Kidwell et al., 2000).
Positron emission tomography (PET) studies in acute ischemic stroke have led to the determination of validated probabilistic voxel-based thresholds defining core and penumbra (Furlan et al., 1996;Marchal et al., 1999). Probabilistic thresholds have the unique property of allowing objective classification of voxels in the individual patient (Heiss et al., 2001), acknowledging the fact that by definition, not all voxels can be classified using this approach. This offers a unique opportunity to study how DWI and ADC relate to these components.
By combining PET and magnetic resonance imaging (MRI) in acute stroke we asked two questions. First, does the acute DWI lesion contain penumbra in addition to core (and in what proportions)? And second, can penumbra have severely reduced ADC equivalent to that found in core?
We present data from a particularly illustrative patient who had an extensive DWI lesion as well as substantial penumbra and core defined by quantitative PET.
MATERIALS AND METHODS
A 53-year-old, right-handed, healthy man presented 5 hours after onset of acute mixed aphasia, right-sided hemiparesis, and right homonymous hemianopia. He was a nonsmoker with no other known vascular risk factors. After obtaining fat-suppressed T1 axial neck views of his carotids, left carotid dissection was diagnosed. Initial computed tomography showed only left-sided early ischemic signs. Magnetic resonance imaging was commenced at 7 hours and followed immediately by PET at 9 hours. Transcranial-Doppler findings were consistent with distal middle cerebral artery branch occlusion both before and between the two imaging modalities. The local ethics committee approved this imaging protocol. Informed consent was obtained.
Diffusion tensor imaging was performed on a 3T whole-body magnet (Med-spec s300; Bruker, Ettlingen, Germany). The MRI technique, parameters, and calculation of the mean diffusivity of the diffusion tensor have been published previously (Green et al., 2002). Briefly, diffusion tensor data sets were calculated from the ADC measured in 12 non—collinear-gradient directions from 5 b values distributed equidistantly in the interval 300 to 1,570 s/mm2, which effectively characterizes the fast diffusing water population (Mulkern et al., 2000), avoids contamination of ADC values by a perfusion component, and reduces CSF partial-volume effects (Le Bihan et al., 1991). Axial field-of-view was limited to 4 cm. A structural T2 was also acquired at the same time point.
The PET study was performed according to a procedure detailed elsewhere (Coles et al., 2004;Steiner et al., 2003) on a GE Advance scanner (General Electric, Milwaukee, WI, U.S.A.), with a 15.3-cm axial field-of-view. This involved a continuous infusion of H215O and inhalation of 15O2 (both to steady state) and brief inhalation of C15O. Maps of CBF, cerebral blood volume, Oxygen Extraction Fraction (OEF), and CMRO2 were generated using arterial data and standard modeling incorporated into a custom-designed software package (PETAN) (Smielewski, 2003). A final MR outcome study (high-resolution three-dimensional volume acquisition spoiled gradient echo sequence) was carried out on day 30.
Image analysis
The PET parametric maps were coregistered to the T2 scan using a routine from SPM99 (www.fil.ion.bpmf.ac.uk/spm) incorporated into PETAN. The average image from the b0 DWI preparation scans was coregistered to the T2 using the normalized mutual information algorithm in vtk-CISG software package (www.image-registration.com). The matrix transformation file so created was applied to the b945 DWI images and ADC map, thus coregistering DWI, ADC, CBF, OEF, CMRO2, and the structural T2. Because of the limited axial extent of DWI and differences in head orientation between the two imaging modalities in this acutely ill patient, the coregistration process resulted in the DWI and ADC images appearing “cut” (see figures). Using ANALYZE 5.0 (AnalyzeDirect Inc., Lenexa, Kansas, U.S.A.), all image modalities were set to PET voxel size (2.34 × 2.34 × 4.25mm) for further analysis.
To address our first question (i.e., Does the acute DWI lesion contain penumbra?), an experienced neuroradiologist (J.H.G.) defined the DWI lesion region-of-interest (ROI) on the b945 images by using a semiautomated routine that applies the same threshold voxel value throughout the image volume. Two further ROIs were created from the PET data, based on the probabilistic 95% thresholds for core (Marchal et al., 1999) and penumbra (Furlan et al., 1996) defined previously for patients studied within the same time frame as this patient, as follows: (1) “core”: voxels in the middle cerebral artery territories with CBF values below 8.4 mL·100 mL−1·min−1or CMRO2 values below 0.87 mL·100 mL−1·min−1 (using both thresholds together slightly but significantly increases the sensitivity; see discussion in (Marchal et al., 1999); and (2) “penumbra”: voxels with CBF between 8.4 and 20 mL·100 mL−1·min−1and OEF above 70%. The DWI ROI was superimposed onto the core and penumbra regions.
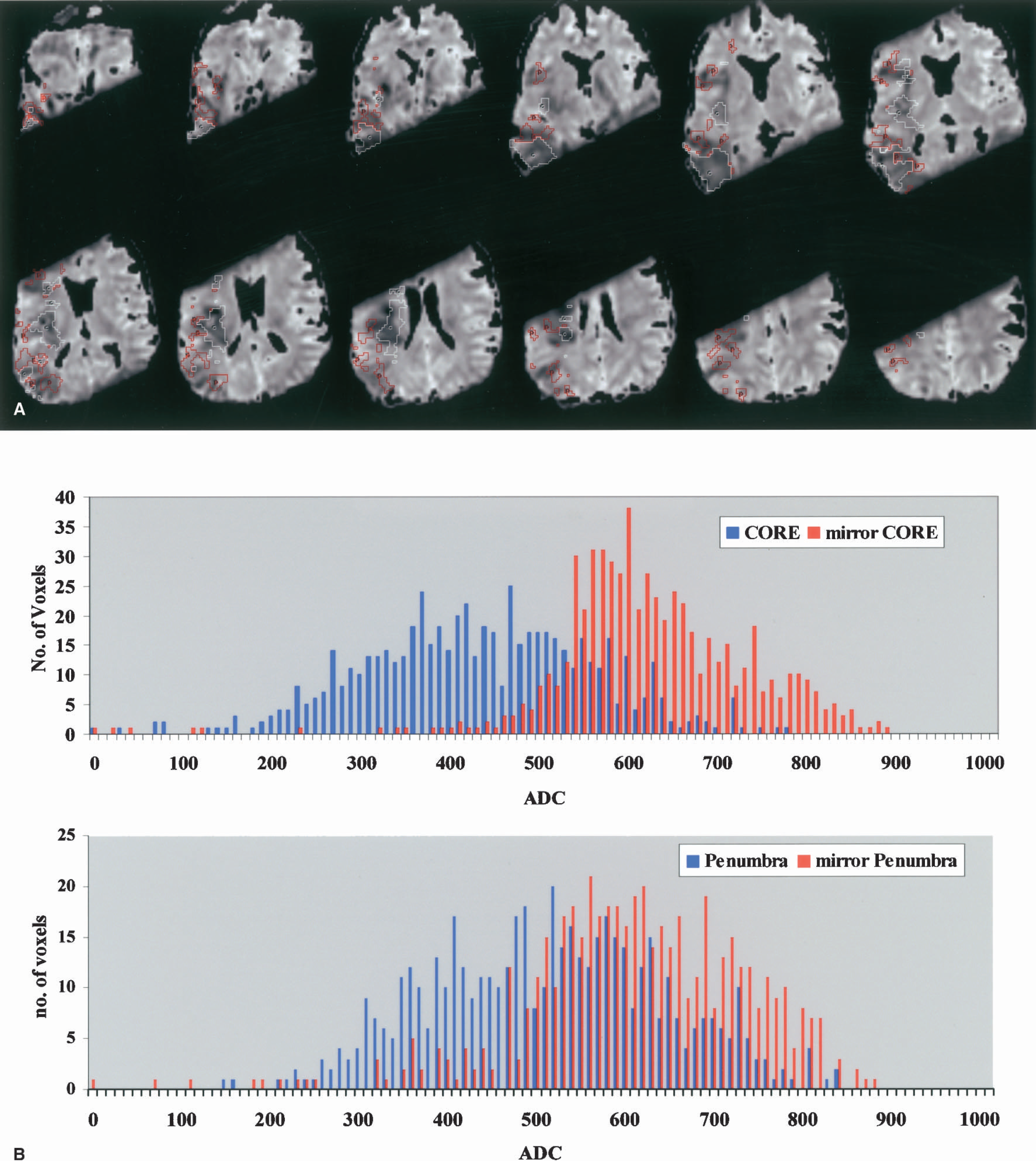
To address the second question (i.e., Can penumbra have severely reduced ADC equivalent to that of core?), the ADC map was thresholded at 1.00 × 10−3 mm2·s−1 to eliminate CSF voxels (Fiehler et al., 2002;Latour and Warach, 2002). The core and penumbra ROIs were transferred to the coregistered ADC map and histograms produced of the ADC values within each ROI. The ROIs were “flipped” to form mirror ROIs on the contralateral hemisphere. Because the images appeared “cut,” some clusters from core and penumbra did not have a corresponding contralateral cluster for comparison; these clusters were not used in the analysis.
Simple descriptive statistics were used throughout (parametric tests only, except with nonnormal distributions as assessed with Kolmogorov-Smirnov test).
RESULTS
On admission, the patient had normal chest x-ray and ECG findings. Blood glucose and cholesterol levels were within normal limits. His score on the National Institute of Health Stroke Scale (NIHSS) was 16 when undergoing acute MRI/PET. Some good clinical recovery was made by day 30 (outcome scan), when his NIHSS score was 9. He was now able to walk with assistance but had moderate expressive dysphasia and some mild receptive component.
Regarding question 1, both penumbra and core were found within the DWI lesion (N = 281 and 651 voxels representing 14.6% and 34% of the whole DWI lesion, respectively; remaining voxels were not classifiable with our probabilistic thresholds). The corresponding values for the mirror ROIs were 1.8% and 3.3% (N = 34 and N = 64), respectively. These differences in geographic distribution of both core and penumbra voxels between hemispheres were highly significant by chi-square test (both P < 0.005). Fig. 1 illustrates the findings and shows that most voxels classified as core fell within the DWI lesion, whereas penumbra voxels fell both within and outside it.
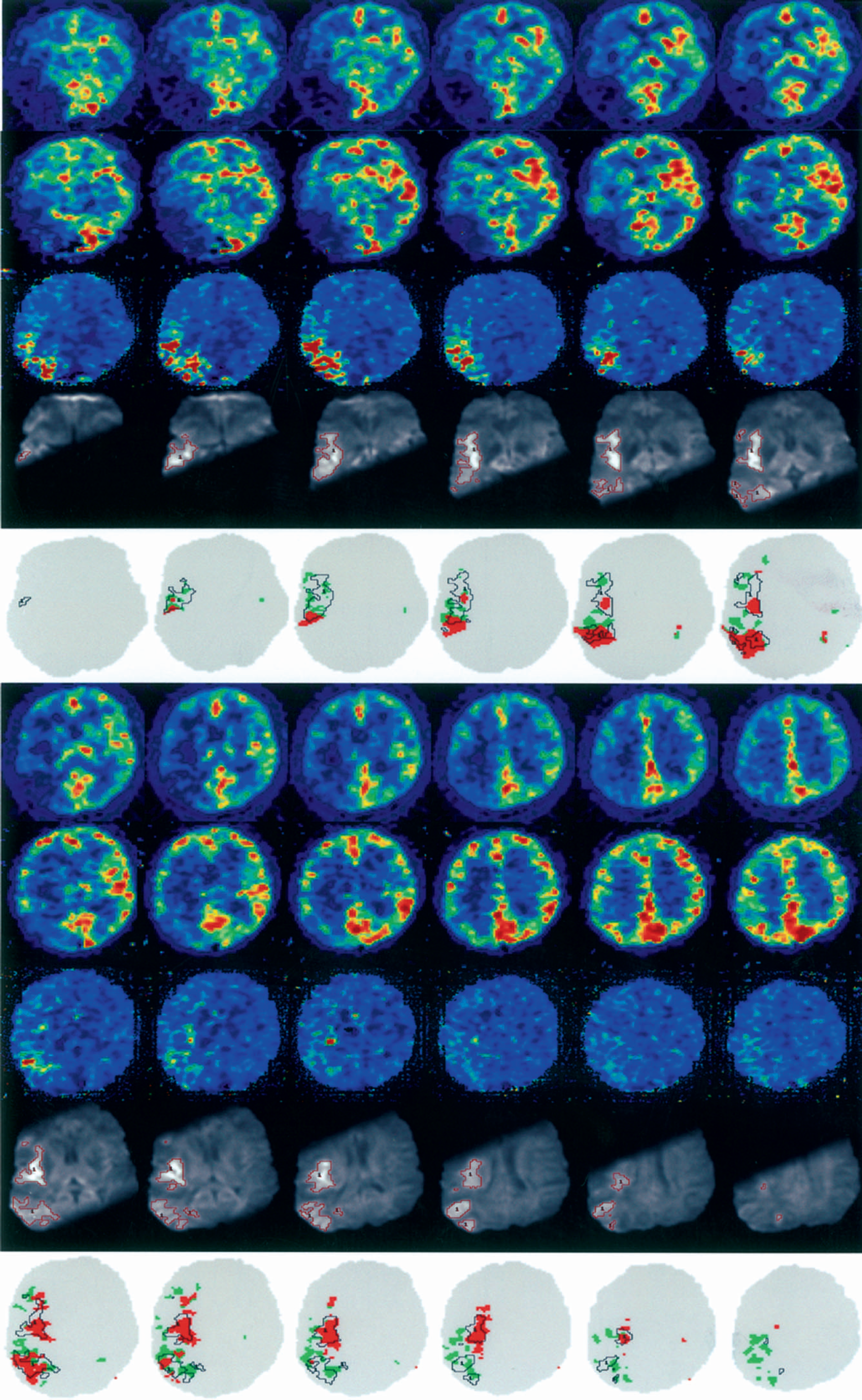
Combined information from coregistered PET and DWI. Rows 1 and 6 show the PET CBF parametric maps, rows 2 and 7 show CMRO2 maps, and rows 3 and 8 show OEF maps, rows 4 and 9 show the 12 contiguous cuts encompassing the DWI lesion (with the DWI lesion ROI shown in red contours), and rows 5 and 10 illustrate the core and penumbra voxels as defined by PET (red and green contours, respectively) with the DWI ROI superimposed (black contours). (See Materials and Methods for details.) This figure illustrates the main finding that the DWI lesion contains both core and penumbral tissue. (See Discussion regarding the apparently variable DWI lesion intensity.) As expected, the relative proportion of penumbra was highest at the dorsal- and ventral-most regions of the middle cerebral artery territory, and lowest in its center (Markus et al., 2003). In these images, the right hemisphere is on the right.
Regarding question 2, the ADC for PET-classified core and penumbra voxels was 428 × 10−6 (±127) and 510 × 10−6 (±131) mm2·s−1 respectively, a nonsignificant difference (P = 0.2; t-test). However, when compared with their mirror ROIs, both were significantly reduced (P < 10−3; Mann-Whitney test). These findings are illustrated in Fig. 2. Accordingly, substantial amounts of severely reduced ADC voxels (defined as <60% of mean contralateral hemisphere (Fiehler et al., 2002)) were found in both core and penumbra voxel populations (33.4% and 15.1%, respectively).
(
Fig. 3 illustrates the final infarct as defined on the day 30 high-resolution spoiled gradient echo sequence.
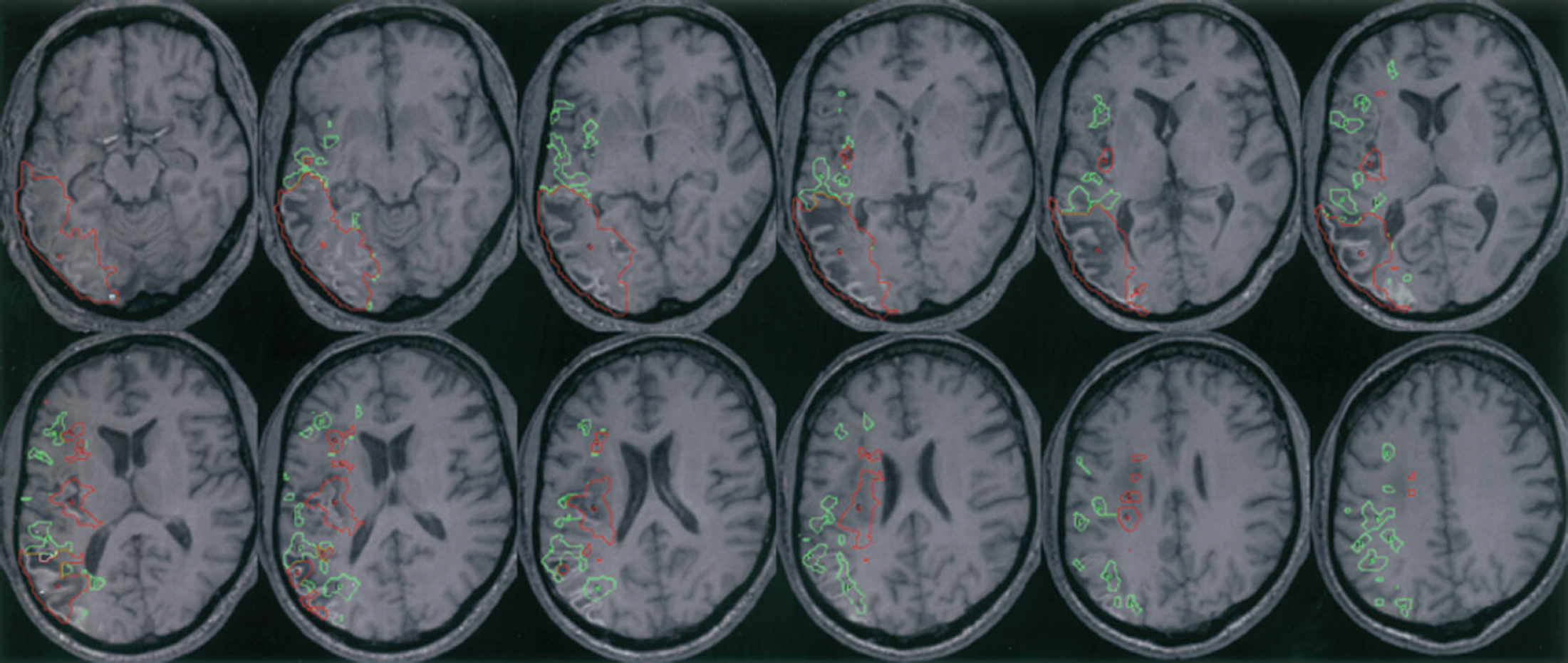
Coregistered day 30 high-resolution spoiled gradient echo sequence scan showing final infarct (equivalent slices to Fig. 1). Superimposed are the entire PET defined core (red) and Penumbra (green). ROIs (i.e., not truncated as in Fig. 1, which was required because of the limited number of DWI slices). The PET-derived ROIs were interpolated to the higher-resolution magnetic resonance image. Visual assessment confirms that, bearing in mind a 95% probabilistic threshold (see Discussion), the core ROI translates into infarcted tissue, whereas the penumbra ROIs (as expected) have a mixed outcome, which would fit with the documented associated clinical improvement from an NIHSS of 16 to 9. Regarding the outcome of core voxels in white matter (see Discussion), inspection confirms that the outcome is also infarction. Note the striatum is unaffected. Comparing the DWI lesion ROI from Fig. 1, it can be seen that most of it progresses to infarction, but note the variable DWI lesion intensity (see Discussion).
DISCUSSION
This is the first report of combined PET/MRI analysis in acute ischemic stroke using state-of-the-art diffusion tensor imaging and fully quantitative PET, as applied to a patient particularly appropriate in relation to our questions. We found that in addition to the expected core, the acute DWI lesion also contained a substantial proportion of penumbral tissue. Furthermore, severe ADC reductions were present within the PET-defined penumbra, consistent with the recent notion that an ADC threshold for infarction does not exist at early time points (Fiehler et al., 2002;Kidwell et al., 2000).
Consistent with the use of 95% thresholds, a few voxels within the unaffected hemisphere fell below threshold but within the expected 5% proportion. That a subset of voxels within the DWI lesion was not classified as either core or penumbra is expected when using probabilistic thresholds (see Introduction), and reflects the conservative nature of the PET thresholds used here (Furlan et al., 1996;Marchal et al., 1999). Therefore, with these validated thresholds there is a 95% chance that voxels meeting these criteria will belong in that defined tissue compartment. If tissue does not meet these stringent criteria, it cannot be defined with this certainty (i.e., 95%). For instance, some tissue may be in the process of changing from penumbra to core, and in doing so we cannot predict with this level of certainty to which tissue compartment it belongs. This approach therefore provides a validated operational procedure to address the question asked in this study. Using a more liberal definition of the penumbra (e.g., based on higher CBF or on OEF alone) would, if anything, have resulted in a greater proportion of the DWI lesion being categorized as penumbral. Since our questions relate to the meaning of DWI and ADC in acute stroke, we retained the voxel size of the original PET as common denominator. However, our findings were replicated if the voxel size used in earlier work (i.e., 8 × 8 × 9 mm) was implemented here (data not shown).
Because of the unavoidable time delay (approximately 1.5 hours) from end of MRI to beginning of PET, changes in the DWI lesion could have occurred in that interval. However, the MRI preceded PET, and penumbra was still present at the time of PET. Furthermore, both the transcranial-Doppler findings and the very low CBF at the time of PET imaging indicate that recanalization had not occurred between scans, so the DWI lesion should have, if anything, enlarged (Barber et al., 1998).
There is no widely accepted method for defining the “DWI lesion.” We used a semiautomated technique where the same threshold was applied throughout the image volume. Because our DWI lesion was defined conservatively, more penumbra would have contributed to the DWI lesion had a more liberal threshold been used (see Fig. 1). Applying a number of different thresholds did not alter the main findings (data not shown).
Regarding question 1, three additional salient findings emerge from scrutiny of Fig. 1. Firstly, individual DWI lesion clusters have varying proportions of core and penumbra, indicating highly heterogeneous local hemodynamics and bioenergetics. Secondly, that penumbral voxels lay both within and outside the DWI lesion suggests that some of the penumbral voxels may have a CBF below an electrical failure threshold but not below a putative threshold for significant restriction of water diffusion. Thirdly, the intensity of the DWI lesion has no relationship to core; even within particular slices, bright DWI lesions could correspond to core or penumbra, and less intense DWI lesions to core. These observations suggest caution in the interpretation of DWI maps for treatment decisions (Coutts et al., 2003). Further work will address these three issues.
Regarding question 2, the wide range of ADC values within core and penumbra, and the finding that severe ADC reductions were present in the latter, further support the idea that ADC alone does not predict irreversibly infarcted tissue within the first hours. Accordingly, even very low ADC may be reversed with early reperfusion and therefore can also represent penumbra (Hasegawa et al., 1994;Hossmann et al., 1994;Kidwell et al., 2003;Li et al., 2000).
There is potentially a difference in infarction and penumbral thresholds between white and gray matter, and possibly between striatum and cortex. Because of the well-known partial volume effect of PET, one has to bring the resolution of MRI to PET for a valid analysis, and therefore it becomes invalid to want to segment out gray from white matter. This issue was discussed by Marchal et al. (1999), who justified why thresholds were calculated for mixed gray and white matter. In addition, the data shown for ADC of PET-defined core and penumbra are compared with mirror ADC ROIs (Figs. 2A and 2B); therefore, the same amount of white and gray matter can be compared. Finally, the striatum was unaffected in this particular patient (see Fig. 3), so we cannot comment on this area.
Applied to a particularly illustrative patient, the approach taken in this work has given further insight into the newer functional MRI findings of acute stroke, with potential clinical implications regarding treatment decisions.
Footnotes
Acknowledgments
The authors thank Diana Day, Peter S. Jones, Dr. Hadrian Green, the radiographers at the WBIC, Drs. R. Salvador and John C. Clark, and Professors D. K. Menon and J. D. Pickard for their help in this project.
