Abstract
Prostaglandin E2 (PGE2) has been shown to dilate and constrict the systemic vascular beds, including cerebral vessels. The exact mechanism of PGE2-induced cerebral vasoconstriction, however, is less clarified. The authors' preliminary studies showed that PGE2 exclusively constricted the adult porcine basilar arteries. The present study, therefore, was designed to examine the receptor mechanisms involved in PGE2-induced constriction of large cerebral arteries in the adult pig. Results from an in vitro tissue-bath study indicated that PGE2 and its agonists 17-phenyl trinor PGE2 (17-PGE2), sulprostone (EP1/EP3 receptor agonists), and 11-deoxy-16,16-dimethyl PGE2 (11-PGE2, an EP2/EP3-receptor agonist) induced exclusive constriction, which was not affected by endothelium denudation or cold-storage denervation of perivascular nerves. The constriction induced by PGE2, 17-PGE2, and sulprostone, but not by potassium chloride, was blocked by SC-19220 (a selective EP1-receptor antagonist), AH-6809 (an EP1/EP2-receptor antagonist), and U-73122 and neomycin (phospholipase C inhibitors). AH-6809, however, did not affect 11-PGE2–induced contraction. These results suggest that the contraction was not mediated by the EP2-receptor, but was mediated by EP1- and EP3-receptors. Furthermore, EP1-receptor immunoreactivities were found across the entire medial smooth muscle layers, whereas EP3-receptor immunoreactivities were limited to the outer smooth muscle layer toward the adventitia. Western blotting also showed the presence of EP1- and EP3-receptor proteins in cultured primary cerebral vascular smooth muscle cells. In conclusion, PGE2 exclusively constricts the adult porcine large cerebral arteries. This constriction is mediated by phosphatidyl–inositol pathway via activation of EP1- and EP3-receptors located on the smooth muscle cells. These two receptor subtypes may play important roles in physiologic and pathophysiologic control of cerebral vascular tone.
The role of prostaglandins in the maintenance of vascular functions is well recognized (Narumiya et al., 1999; Wright et al., 2001). Prostaglandin E2, an important and ubiquitously present vasoactive eicosanoid, has been reported to be a potent vasodilator in most peripheral (Coleman et al., 1994; Lawrence and Jones, 1992; Lydford et al., 1996) and cerebral (Armstead et al., 1989; Ellis et al., 1979; Hayashi et al., 1985; Li et al., 1994) vascular beds by directly relaxing the vascular smooth muscle cells (Lawrence and Jones, 1992) in both humans and experimental animals.
Evidence, however, has shown that PGE2 causes both cerebral vasoconstriction and relaxation in several species. For example, PGE2 has been shown to induce both constriction and relaxation of middle cerebral arteries of the adult baboon (Hayashi et al., 1985), adult cat basilar and middle cerebral arteries (Whalley et al., 1989), and adult human pial (Uski et al., 1984) and basilar arteries (Parsons and Whalley, 1989). Prostoglandin E2 also has been shown to induce exclusive constriction of the cerebral intraparenchymal microvessels in the adult pig (Li et al., 1994), and cerebral arteries of the adult canine and monkey (Toda et al., 1991). The different findings in PGE2-induced vascular responses appear to be age dependent (see Discussion) due to expression of different subtypes of PGE2 receptors (EP-receptors). The exact mechanism mediating PGE2-induced vascular constriction is less explored.
The EP-receptors are heterotrimeric G protein–coupled prostanoid receptors (Coleman et al., 1994; Narumiya et al., 1999), which are grouped into three categories on the basis of their signal transduction and action. Cluster 1 consists of EP2, EP4, IP (PGI2), and DP (PGD2) receptors, which mediate smooth muscle relaxation through Gs protein. Cluster 2 consists of EP1, FP (PGF2α), and TP (thromboxane A2) receptors, which mediate smooth muscle contraction by activating Gq protein (Wright et al., 2001). Cluster 3 consists of the inhibitory EP3 receptor coupled mainly to Gi or Gq proteins (Narumiya et al., 1999; Wright et al., 2001). Our pilot studies showed that PGE2 induced exclusive constriction of isolated basilar arteries and the circle of Willis of adult pigs. The current study, therefore, was designed to determine the vasomotor action of PGE2 and its underlying receptor mechanisms in large arteries at the base of the adult porcine brain.
Prostaglandin E2 is one of the major mediators produced during certain cerebral vascular dysfunctions, such as the cerebral vasospasm after SAH (Macdonald and Weir, 1991; Toda et al., 1991, Walker et al., 1983) in animals (Chyatte, 1989) and humans (Pickard et al., 1994). Consequently, in SAH, prostaglandins such as PGE2 and HbO2 have been invoked as important causative agents (Macdonald and Weir, 1991). However, the interaction between these putative spasmogens, if any, has not been explored. Thus, using the in vitro tissue bath approach, we also further investigated the interaction between PGE2 and HbO2 in adult porcine basilar arteries.
MATERIALS AND METHODS
General procedure
Fresh heads of adult pigs (60 to 100 kg) of both sexes were collected from local packing companies (Excel Corporation, Beardstown, IL, U.S.A. and Y-T, Springfield, IL, U.S.A.). The entire brain, with the dura mater attached, was removed and placed in Krebs bicarbonate solution (in millimoles per liter: NaCl 122.0, KCl 5.16, CaCl2 1.2, MgSO4 1.22, NaHCO3 25.6, EDTA 0.03, L-ascorbic acid 0.1, and glucose 11.0, pH 7.4) equilibrated with 95% O2 and 5% CO2 at room temperature. The large cerebral arteries at the base of the brain, namely, the basilar arteries and the circle of Willis, were dissected and cleaned of surrounding tissue under a dissecting microscope.
In vitro tissue bath
Arterial ring segments (5 mm long) were cannulated with a stainless steel rod (30-gauge hemispherical section) and a short piece of platinum wire and mounted horizontally in a Plexiglas tissue bath containing 6 mL of Krebs solution. The platinum wire was bent into a U shape and anchored to the gate. The stainless steel rod was connected to a strain-gauge transducer (UC2; Gould, Cleveland, OH, U.S.A.) for isometric recording of changes in force, as described in our previous work (Zhang et al., 1998). The temperature of Krebs solution was maintained at 37°C. Tissues were equilibrated for 30 minutes and then mechanically stretched to a resting tension of 750 mg (Zhang et al., 1998).
Drugs were added directly to the tissue bath as indicated and the concentrations of drugs reported represent the final concentrations in the bath. Each ring segment served as its own control. PGE2, sulprostone, or or 17-PGE2 was added to the tissue bath in increasing concentrations (0.001 to 3.0 μmol/L). The contraction of arteries in the absence of active muscle tone obtained from this first application served as the control. These drugs were also tested in the presence of active muscle tone induced by U-46619 (a thromboxane A2 analog, 0.3 μmol/L) in separate experiments. Similarly, PGE2 in increasing concentrations (0.001–3.0 μmol/L) was also tested in endothelium-denuded arteries precontracted with endothelin (0.001 μmol/L), neuropeptide Y (0.03 μmol/L), acetylcholine (0.3 mmol/L), and bradykinin (1.0 mmol/L). All drugs were washed off with prewarmed Krebs solution (37°C) with at least four washes over 60 minutes after every set of incremental PGE2 or agonist applications. Antagonists (AH-6809, SC-19220, and SQ-29548) and inhibitors (neomycin and U-73122) were administered 20 minutes before repeating PGE2 or agonist applications at the same concentrations as before. Oxyhemoglobin (10 μmol/L) was added and the solution was incubated for 60 minutes and washed off. Prostaglandin E2 was then applied in the presence or absence of neomycin (30 mmol/L). Neomycin was administered 20 minutes before PGE2 applications. Recovery or postwash data were obtained by repeating PGE2 or agonist applications in incremental concentrations, without any antagonists or blockers, similar to control.
Because 11-PGE2 is much more potent (Karim et al., 1980) than other EP agonists, and because it was difficult to “washout” 11-PGE2-induced contraction, a different approach was used to examine its constricting effect. 11-PGE2 in increasing concentrations (0.001 nmol/L to 3.0 μmol/L) was added to the tissue bath to induce contraction of arteries in the absence of active muscle tone. After adding the final concentration of 11-PGE2, AH-6809 was added in increasing concentrations (3 to 300 μmol/L), followed by the addition of full concentrations of isoproterenol (0.001 to 3.0 μmol/L).
Potassium chloride (80 mmol/L) was administered to induce maximum contraction at the end of the experiments. The magnitude of vasoconstriction during the experiments was expressed as a percentage of the maximum response induced by KCl (80 mmol/L) for each individual segment. EC50 values (the concentration that produces 50% of the maximum contraction) were determined for each arterial ring segment. From these values, the geometric means EC50 with 95% confidence intervals were calculated (Fleming et al., 1972).
For cold-storage denervation of the arteries, tissues were stored in Krebs solution at 4°C for 7 or 8 days (Lee, 1982). The denervation of ring segments was confirmed by the absence of transmural nerve stimulation–induced dilation of the arteries in the presence of active muscle tone (Lee and Sarwinski, 1991). For transmural nerve stimulation, tissues were electrically and transmurally stimulated with a pair of electrodes through which 100 biphasic square-wave pulses of 0.6-millisecond duration and 200-mA intensity (continuously monitored on a Tektronix oscilloscope) were applied at various frequencies of 2, 4, and 8 Hz (Zhang et al., 1998).
The arterial endothelium was denuded mechanically by a standard brief gentle rubbing of the intimal surface with a serrated stainless steel rod with a diameter (25–30 gauge) equivalent to the lumen of the arteries (Lee, 1982). A complete removal of endothelial cells was verified by lack of effect of N-nitro-
Primary cultures of cerebrovascular smooth muscle cells
The basilar arteries and the circle of Willis were dissected from porcine brain, placed in ice-cold sterile PBS (pH 7.4, Gibco BRL/Life Technologies, Carlsbad, CA, U.S.A.) and denuded of the endothelium as described before (Lee, 1982; Zhang et al., 1998). The vessels were then slit open longitudinally, cut into approximately 2 × 2 mm explants, placed in Dulbecco's modified Eagle's medium (DMEM) (Fisher Scientific, Hampton, NH) and washed once by centrifugation at 1,000g for 5 minutes. The explants were plated in 35-mm, six-well cultured plates in DMEM plus 5% fetal bovine serum (Gibco BRL/Life Technologies), and placed in an incubator with an atmosphere of 5% CO2 and 95% O2 at 37°C. The cells began exiting by 10 to 12 days and the medium was changed every 4 or 5 days until they reached confluence. Cells were then passaged into polystyrene tissue culture dishes (100 × 20 mm) using 0.25% trypsin (Gibco BRL/Life Technologies) and grown in DMEM plus 5% fetal bovine serum. All solutions and media contained antibiotic-antimycotic solution containing 100 μg/ml penicillin G, 100 μg/ml streptomycin sulfate, and 0.25 μg/ml amphotericin B (Fisher Scientific). Smooth muscle cells were confirmed by positive staining with FITC-conjugated monoclonal antibody to α-smooth muscle actin (Sigma, St. Louis, MO, U.S.A.). Cells from passages 3 through 5 were used for experiments. Our primary cultures stained more than 95% positive for α-smooth muscle actin.
Immunohistochemistry
The dissected arterial segments and the cultured cerebrovascular smooth muscle cells on coverslips were fixed in 4% paraformaldehyde for 10 minutes at room temperature. After washing in ice-cold PBS, they were permeabilized and nonspecific binding sites were blocked by incubation in PBS buffer containing 0.5% Triton X-100 and 5% donkey serum for 10 minutes. Both specimens were incubated with rabbit anti–EP1- or anti–EP3-receptor polyclonal antibodies (1:50 dilution in buffer solution, Cayman Chemical, Ann Arbor, MI, U.S.A.) and mouse anti–α-smooth muscle actin (1:50 dilution in buffer solution, Sigma) or mouse anti–neurofilament-200 monoclonal antibodies (1:100 dilution in buffer solution, Sigma) at 4°C for 48 hours. After two washes with buffer solution and PBS, both specimens were incubated for one hour in FITC- or TRITC-conjugated secondary antibody (1:40 dilution in buffer solution) (Jackson Immuno Research labs, PA). After washing, the arterial segments were either cross-sectioned using Microm cryostat HM 505-E or cut open longitudinally, and mounted using Cytoseal (Richard-Allan Scientific, Kalamazoo, MI, U.S.A.). Similarly, the cultured cells on coverslips were also placed on glass slides. Both were stored in the dark before analysis using an Olympus BX50 fluorescence microscope.
Western blot
The cultured cerebrovascular smooth muscle cells (passages 3–5) were homogenized in lysis buffer containing 50 mmol/L Tris (pH 7.4), 150 mmol/L NaCl, 10 mmol/L sodium metavanadate, 5 mmol/L PMSF, 1% Triton X-100, and 0.1% 100X protease inhibitor cocktail set I (Calbiochem, La Jolla, CA, U.S.A.). The cell lysate protein content was determined by BioRad protein assay, and equal amounts of protein were denatured by boiling after adding sample buffer laemmli, followed by electrophoresis in 10% SDS-polyacrylamide gel. The protein bands were transferred electrophoretically to a nitrocellulose membrane and blocked using 3% bovine serum albumin and 0.1% Tween 20 in PBS (pH 7.4). Membrane was probed by incubation with primary polyclonal antibodies to EP1- and EP3-receptors (Cayman Chemical) at dilutions of 1:500, and followed with horseradish peroxidase-conjugated secondary antibodies (Santa Cruz Biotechnology, Santa Cruz, CA, U.S.A.) at a 1:1000 dilution. Antibody reaction was detected using the enhanced chemiluminescence detection procedures according to the manufacture's recommendation (Supersignal Pierce, Perbio, Rockford, IL, U.S.A.). Blots were then scanned into a video image capture system using Hitachi Genetic CCD camera and Genesnap/Gene tools (Syngene; Scientific Laboratory Supplies, Millville, NJ, U.S.A.).
Preparation of oxyhemoglobin
Oxyhemoglobin was prepared from purified porcine hemoglobin (Sigma) by reduction with sodium dithionite (1 mg dithionite/15 mg hemoglobin) and separated on a Sephadex G-25 column as previously described (Linnik and Lee, 1989). The concentration was then determined using a BioRad protein assay. Oxyhemoglobin was always prepared fresh on the day of experiment just before adding to tissue bath and stored on ice while handling.
Drugs and statistical analysis
Prostaglandin E2, KCl, N-nitro-
The results were computed as means ± SD. Repeated measures analyses of variance were used to evaluate concentration versus treatment interactions. Paired t-tests were used to compare control to active treatment for each experiment. Results were significant at P < 0.05.
RESULTS
Prostaglandin E2 induced exclusive constriction of porcine large cerebral arteries
Prostaglandin E2 (0.001 to 3.0 μmol/L) induced, in a concentration-dependent manner, exclusive contraction of ring segments of the basilar arteries (Figs. 1A and 1B) and the circle of Willis (n = 8, data not shown) in the absence (Figs. 1B and 2C) or presence of active muscle tone induced by U-46619 (a thromboxane A2 analogue, 0.3 μmol/L; Fig. 1A, n = 8). Similar concentration-dependent PGE2 (0.001–3.0 (mol/L) contraction was observed in endothelium-denuded basilar arteries precontracted with endothelin (0.001 μmol/L), neuropeptide Y (0.03 μmol/L), acetylcholine (0.3 mmol/L), or bradykinin (1.0 mmol/L) (n = 4 each, data not shown). Quantitative analysis of data in subsequent studies was obtained in the basilar arterial preparations in the absence of active muscle tone. The contraction induced by PGE2 was not affected by endothelium denudation (Fig. 1B, n = 8 for each group) or cold-storage denervation of perivascular nerves (n = 8, data not shown). Similarly, sulprostone and 17-PGE2 (0.001 to 3.0 μmol/L, EP1/EP3-receptor agonists) in a concentration-dependent manner contracted exclusively the endothelium-preserved (n = 6, data not shown) and endothelium-denuded porcine basilar arteries in the absence (Table 1) and presence of active muscle tone induced by U-46619 (n = 4 for each agonist, data not shown). 11-PGE2, a potent EP2/EP3-receptor agonist (Karim et al., 1980), also induced, in a concentration-dependent (0.001 nmol/L to 3.0 μmol/L) manner, contraction in both endothelium-preserved and endothelium-denuded porcine basilar arteries (Figs. 1C and 1D, n = 10 each) (geometric means EC50 values were 1.255 [0.315 to 4.988] nmol/L and 0.921 [0.181 to 4.678] nmol/L, respectively).
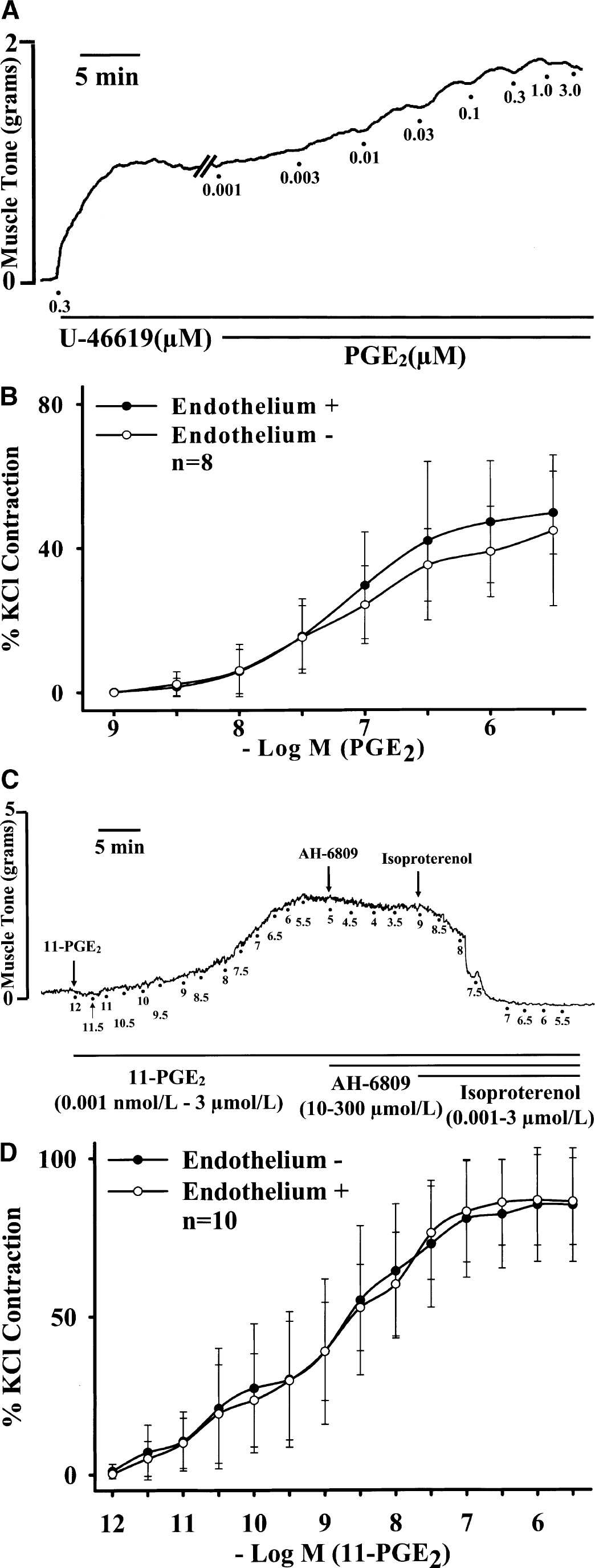
(
Prostaglandin E2 agonist–induced vasoconstriction was attenuated by EP-receptor antagonists
AH-6809 (an EP1/EP2/DP-receptor antagonist, 30 to 300 μmol/L) and SC-19220 (a selective EP1-receptor antagonist, 30 and 100 μmol/L) attenuated, in a concentration-dependent manner, the contraction induced by PGE2 (Fig. 2A, n ≥ 7 for each concentration and Fig. 2B, n = 8 for each concentration, respectively; Fig. 2C representative tracing with AH-6809, 300 μmol/L), sulprostone (Table 1, n ≥ 6 for each concentration), and 17-PGE2 (Table 1, n = 7) in endothelium-denuded basilar arteries in the absence of active muscle tone. Similar results were seen in endothelium-preserved arteries (n = 4 for each antagonist at 100 μmol/L, data not shown). None of the antagonists significantly affected the contraction induced by KCl (80 mmol/L) (100% and 97.80% ± 16.04% before and after AH-6809 treatment and 100% and 108.67% ± 9.43% before and after SC-19220 treatment, n = 4). The attenuation by antagonists was reversed after washing off the antagonists (Figs. 2A–2C).
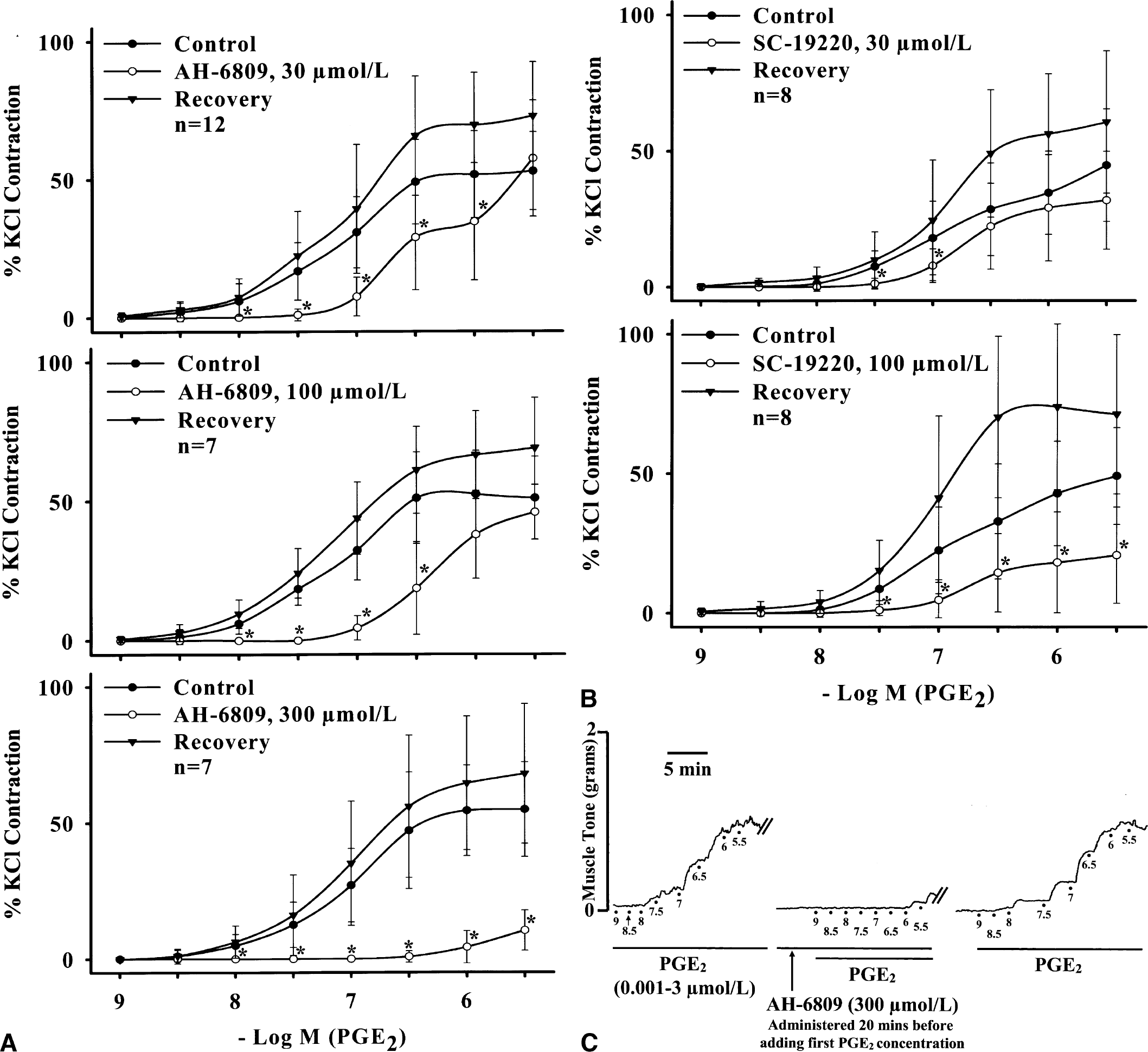
Effects of different concentrations of EP receptor antagonists, AH-6809 (an EP1/EP2/DP-receptor antagonist, panel
The 11-PGE2–induced contraction, however, was not affected by AH-6809 (3 to 300 μmol/L), but was reversed by isoproterenol (β-adrenergic agonist, 1.0 nmol/L to 3.0 μmol/L) in a concentration-dependent manner in both endothelium-denuded (Fig. 1C, representative tracing) and endothelium-preserved basilar arteries in the absence of active muscle tone (Fig. 1D, n = 10 each).
Effect of antagonists and inhibitors on PGE2-agonist–induced constriction of porcine large cerebral arteries
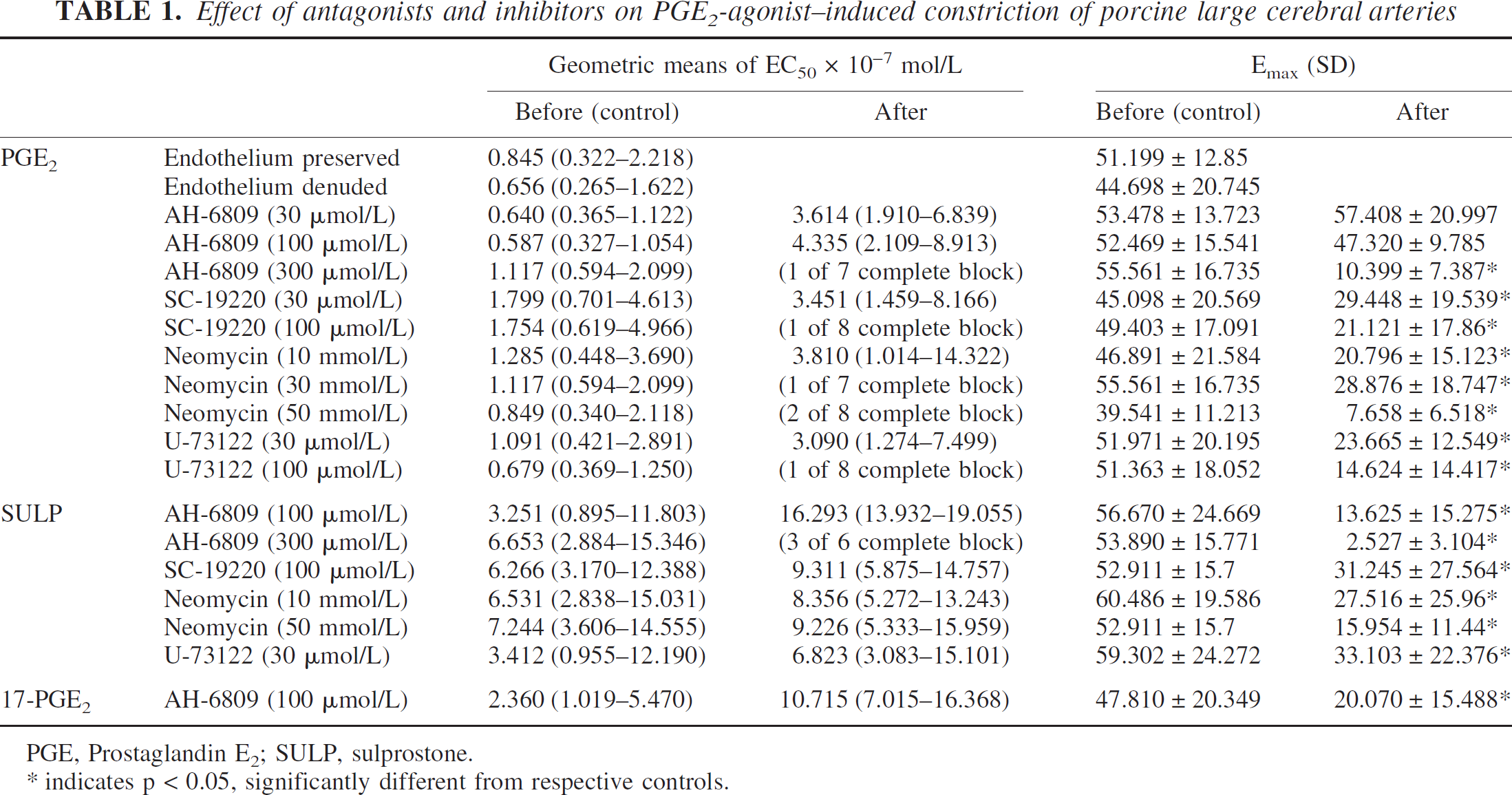
PGE, Prostaglandin E2; SULP, sulprostone.
indicates p < 0.05, significantly different from respective controls.
Previous reports (Dorn et al., 1992) have suggested the possibility of PGE2 cross-reacting at TP receptors in the rat aorta. However, SQ-29548 (30 μmol/L), a selective TP receptor antagonist (Dorn et al., 1992), did not affect PGE2-induced contraction (Emax = 74.72 ± 27.24 and 83.55 ± 26.06; geometric means EC50 values = 12.82 [6.23 to 26.41] μmol/L and 10.42 [5.81 to 18.7] μmol/L before and after SQ-29548 treatment, n = 11) in porcine basilar arteries. But, SQ-29548 abolished the contraction induced by U-46619 (a thromboxane A2 analogue, 0.001 to 3.0 μmol/L) (Emax = 94.72 ± 38.62 and 3.02 ± 3.252 before and after SQ-29548 treatment, n = 4). Emax (with SD) is indicated as percent KCl contraction.
EP1- and EP3-receptor subtypes in cerebrovascular smooth muscle cells
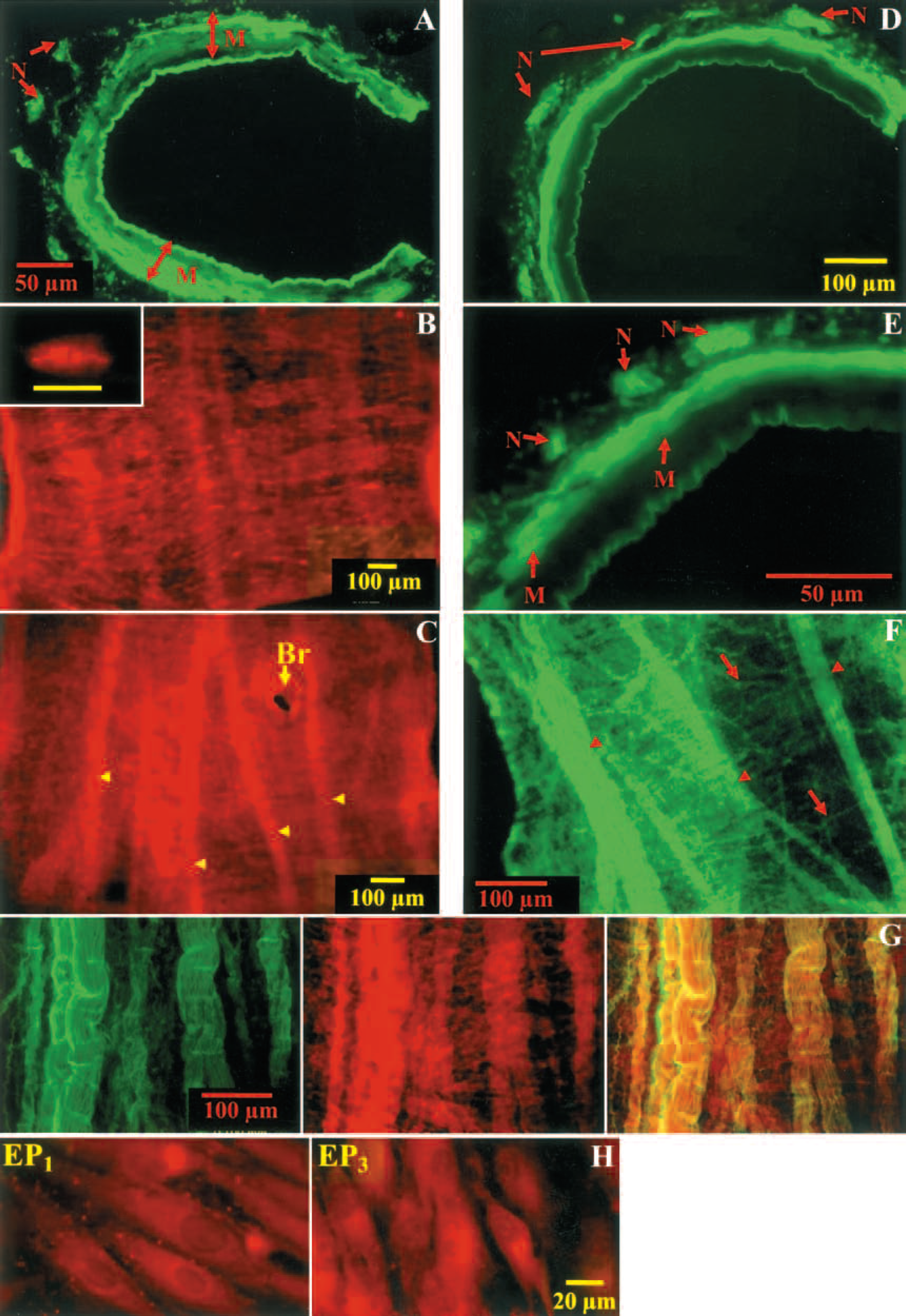
Results from immunohistochemical studies in cross section of the whole-mount porcine basilar arteries and the circle of Willis (Figs. 3A–3G, n = 8) indicated that the EP1-receptor immunoreactivities were present primarily in the smooth muscle throughout the entire medial layers (Fig. 3A). In these arteries, dense EP1-receptor–immunoreactive structures appeared elongated in shape (approximately 20 to 25 μm long) and oriented circularly around the axis of the artery in the whole-mount preparations (Fig. 3B). The EP3-receptor-immunoreactivities, however, were found on the smooth muscle of the outer layers towards the adventitia (Figs. 3D and 3E). Immunoreactivities of both EP1-receptors and EP3-receptors were found on cultured smooth muscle cells from the basilar arteries and the circle of Willis (Fig. 3H). In these cultured smooth muscle cells, EP3-receptor but not EP1-receptor immunoreactivites were observed in association with the nuclear compartment. For negative controls, no immunoreactivities of EP1- or EP3-receptor or α-smooth muscle actin were observed in whole-mount or cultured cells by omitting the respective primary antibodies (data not shown).
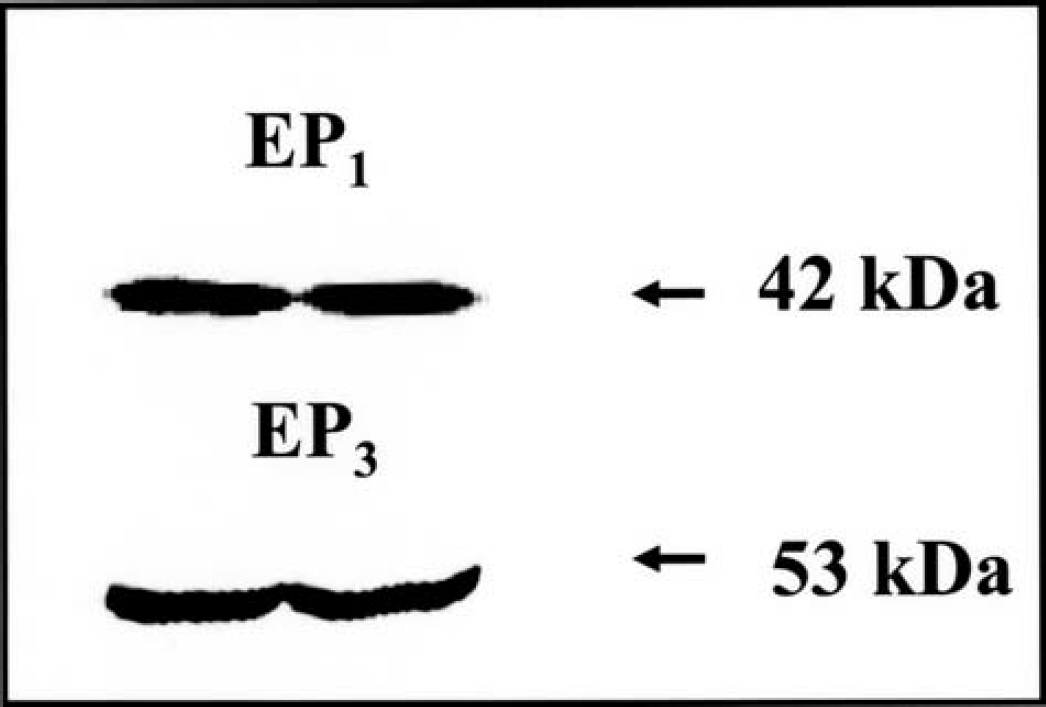
Western blots showing EP1- and EP3-receptor bands at 42 kd and 53 kd, respectively. Primary cerebrovascular smooth muscle cell cultures from passage 3 were used for this study (n = 4).
The EP3-receptor immunoreactivities were also found on the adventitial bundles and fine fibers (Figs. 3F–3G), which were coincident with neurofilament-200 immunoreactivities, a marker for perivascular neurons (Fig. 3G). The adventitial EP1-receptor–immunoreactive bundles, although relatively sparse compared with EP3-receptor–immunoreactive fibers, were also found (Fig. 3C).
EP1- and EP3-receptor proteins in cultured cerebral vascular smooth muscle cells
Results from Western blotting experiments using polyclonal antibodies against the EP1- or EP3-receptor indicated the expression of both EP1- and EP3-receptor proteins in the primary cultures of the smooth muscle cells from the large cerebral arteries (Fig. 4, n = 4). Bands were observed at 42 kd and 53 kd in EP1- and EP3-immunoblots, respectively.
Neomycin and U-73122 blocked vasoconstriction induced by PGE2 agonists
Both EP1- and EP3-receptors have been shown to signal via the phosphatidyl–inositol pathway (Narumiya et al., 1999). Neomycin (10–50 mmol/L) and U-73122 (30 and 100 μmol/L), which are phospholipase C inhibitors (Bleasdale et al., 1990; Vollrath et al., 1994), attenuated the PGE2-induced contraction in the endothelium-denuded porcine basilar arteries in a concentration-dependent manner (Figs. 5A and 5B, n ≥ 7 for each concentration). These phospholipase C inhibitors did not significantly affect KCl-induced contraction (100% and 110.35% ± 12.78% before and after U-73122 treatment, respectively, and 100% and 103.25% ± 15.01% before and after neomycin treatment, respectively, n = 4 each). Similarly, these inhibitors attenuated, in a concentration-dependent manner, the contractions induced by other PGE2 agonists, sulprostone (Table 1, n ≥ 7 for each group), 17-PGE2 (n = 3 for each inhibitor, data not shown), and 11-PGE2 (n = 3, data not shown). The attenuation by inhibitors was reversed after washing off the inhibitors (Figs. 5A and 5B).
EP1-receptor- and EP3-receptor-immunoreactivities in whole-mount large cerebral arteries and the cultured smooth muscle cells from these arteries in representative fluorescence photomicrographs with FITC (green) and TRITC (red) labeling. (
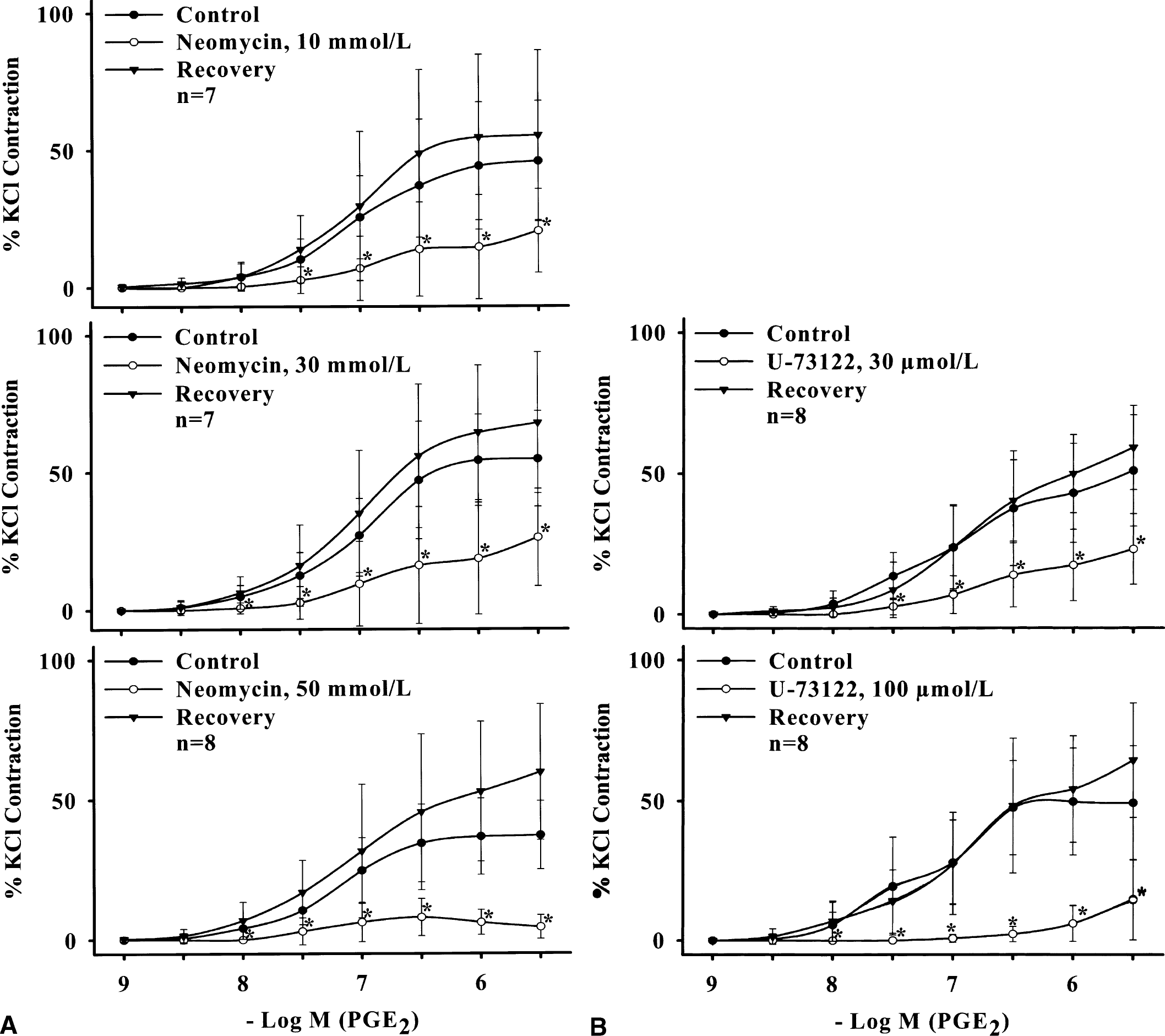
Inhibitory effects of different concentrations of neomycin (
Prostaglandin E2-induced vasoconstriction was potentiated by oxyhemoglobin
The interaction between PGE2 and HbO2, both implicated as causative agents in cerebral vasospasm after SAH (Chyatte 1989; Macdonald and Weir, 1991; Pickard et al., 1994; Walker et al., 1983), was examined by studying and comparing PGE2-induced constriction before and after incubation with HbO2. The PGE2-induced constriction in endothelium-denuded porcine basilar arteries in the absence of active muscle tone was potentiated in a concentration-dependent manner after 1-hour incubation with HbO2 (10 μmol/L) (Fig. 6A, n = 9). This potentiation was significantly blocked by neomycin (30 mmol/L) (Fig. 6B, n = 11). The selective EP1-receptor antagonist SC-19220 (30 μmol/L) also suppressed the potentiation of PGE2-induced constriction after 1 hour of HbO2 incubation (Fig. 6C, n = 7).
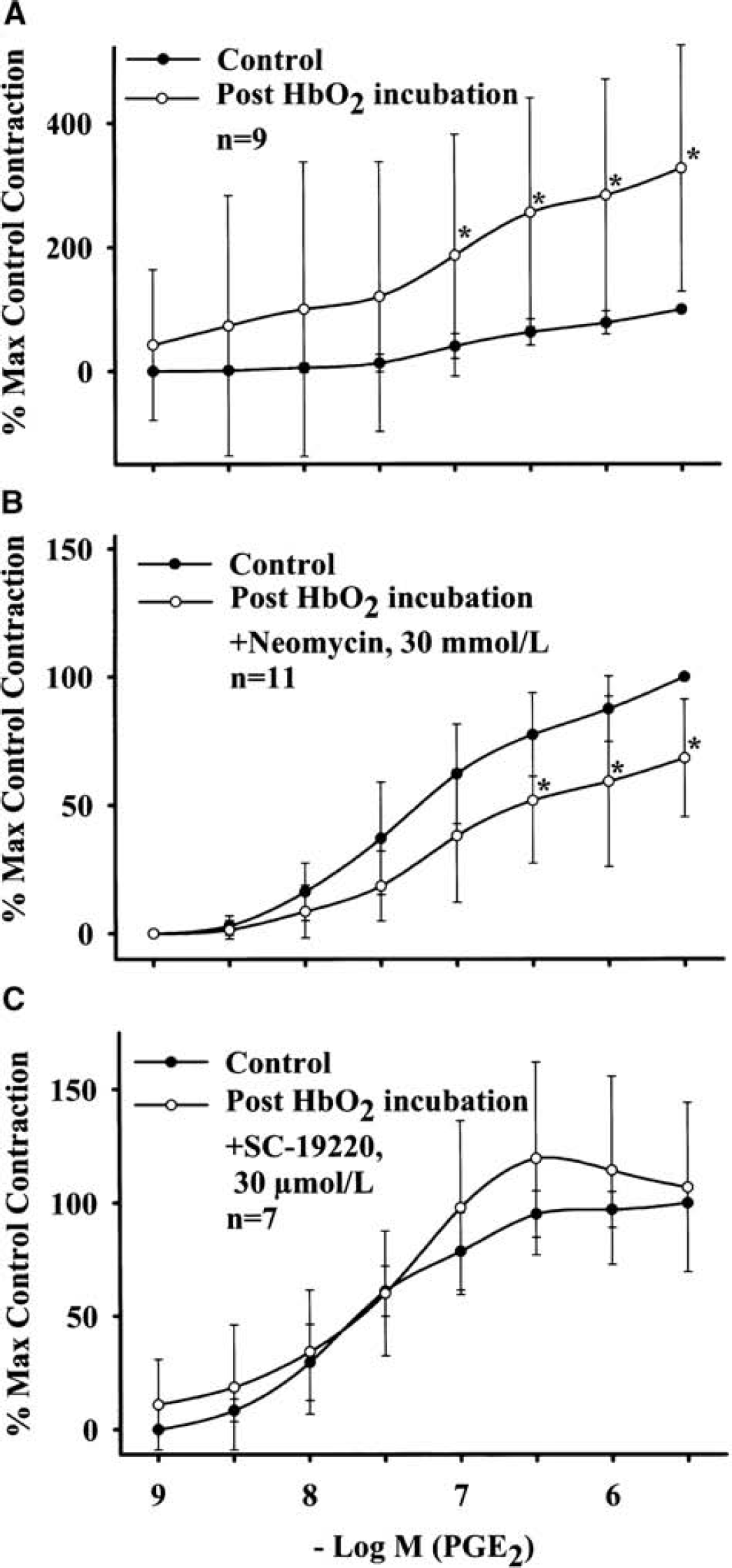
Effect of 1-hour oxyhemoglobin (10 μmol/L) incubation on PGE2-induced contraction of endothelium-denuded porcine basilar arteries. (
DISCUSSION
Prostaglandin E2, a ubiquitous vasoactive eicosanoid, has generally been considered a potent vasodilator in most organs, including the brain, in both humans and experimental animals (Coleman et al., 1994; Ellis et al., 1979; Narumiya et al., 1999). Increasing evidence, however, indicates that PGE2 induces constriction in addition to dilation in many vascular beds (Hayashi et al., 1985; Li et al., 1994; Parsons and Whalley, 1989; Toda et al., 1991; Uski et al., 1984; Whalley et al., 1989), including large cerebral arteries in the human, monkey, and dog (Parsons and Whalley, 1989; Toda et al., 1991). In the present study, PGE2 caused exclusive constriction of the basilar arteries and the circle of Willis of adult pigs. This constriction was independent of intact endothelium or perivascular nerves in that it was not affected in endothelium-denuded or cold-storage–induced perivascular nerve–denervated preparations. These results suggest that the vascular smooth muscle is the primary site of action for PGE2 to induce constriction of large arteries at the base of the porcine brain.
The physiologic significance of the dual vasoconstricting and dilating effects of PGE2 remains to be determined. Although regional and species variations may account for differences in PGE2-induced vascular response, different vascular responses to PGE2 appear to be age dependent, according to available publications. For example, pronounced vasoconstrictor effects of PGE2 on the retinal vasculature in adult versus newborn pigs are attributed to greater EP receptor subtypes mediating constriction than those for dilation (Abran et al., 1995). Age-dependent alterations in EP receptors for constriction and dilation also have been shown to be responsible for the decreased responsiveness of ductus arteriosus to the dilator effect of PGE2 in newborn pigs (Wright et al., 2001). Furthermore, PGE2 has been shown to have dual vasomotor effects on normal human corpus cavernosum (Kirkeby et al., 1993) with a preferential decrease in dilator response to PGE2 in aged patients, possibly due to altered EP receptors (Wolfson et al., 1993). Similar age-related changes in response to PGE2 in cerebral arteries have been reported. Prostaglandin E2 has been shown to relax the pial vessels (Armstead et al., 1989) and the cerebral intraparenchymal microvessels (Li et al., 1994) in newborn pigs. However, PGE2 constricts the cerebral intraparenchymal microvessels in adult pigs (Li et al., 1994). Prostaglandin E2 also causes relaxation of middle cerebral arteries in premature and newborn baboons, whereas in adult baboons it causes both constriction and relaxation of the same arteries (Hayashi et al., 1985). Similarly, PGE2 causes both constriction and relaxation of the feline basilar and middle cerebral arteries (Whalley et al., 1989) and the human pial (Uski et al., 1984) and basilar arteries (Parsons and Whalley, 1989), whereas it causes constriction of adult canine and monkey cerebral vessels (Toda et al., 1991). Furthermore, when prostaglandin concentrations are altered in newborn brain to match those of adult brain, the EP1- and EP3-receptor densities also change accordingly to match the adult levels (Wright et al., 2001). Together with results of the present findings in large cerebral arteries of adult pigs, it is very likely that PGE2-induced cerebral vascular response is age dependent, with a preferential vasodilation in the newborn and a preferential vasoconstriction in the adult. This information is obviously important in understanding the exact role of PGE2 in regulating cerebral circulation.
The PGE2-induced constriction of the porcine basilar arteries and the circle of Willis was mimicked by 17-PGE2 and sulprostone, which are PGE2 agonists for EP1/EP3-receptors. The constriction induced by these agonists was attenuated specifically by AH-6809 (an EP1/EP2/DP-receptor antagonist) and SC-19220 (a selective EP1-receptor antagonist) in a concentration-dependent manner, which, however, did not affect the KCl-induced constriction. These results suggest that EP1-receptors are mediating PGE2-induced constriction. 11-PGE2, which is a highly potent agonist for EP2/EP3-receptors (Karim et al., 1980), also caused a potent constriction of porcine basilar arteries as evidenced by its low initial concentration for constriction and EC50 values. 11-PGE2-induced constriction was not affected by AH-6809 even at high concentrations, which was shown to inhibit the PGE2-induced, EP1/EP3-receptor-mediated contraction. The 11-PGE2-induced constriction, however, was reversed in a concentration-dependent manner by β-adrenergic agonist isoproterenol, a known relaxant of porcine basilar arteries (Lee et al., 1982). These results suggest that EP2-receptors are not involved in 11-PGE2-induced constriction, which is more likely mediated by EP3-receptors. Although direct evidence is yet to be presented for the presence of a functional EP3-receptor subtype on the cerebral vascular smooth muscle due to unavailability of a selective receptor antagonist, our results suggest that EP3-receptors together with EP1-receptors are mediating PGE2-induced constriction. The possibility of PGE2 cross-reacting with thromboxane A2 (TP) receptors (Dorn et al., 1992) was ruled out because SQ-29548, a selective TP receptor antagonist (Dorn et al., 1992) that blocked constriction induced by thromboxane A2, did not affect the PGE2-induced constriction.
The presence of functional EP1- and EP3-receptors in PGE2-induced constriction is supported further by the presence of both EP1- and EP3-receptor immunoreactivities in the large porcine cerebral arteries and the cultured smooth muscle cells isolated from these arteries (Fig 3). This is consistent with the reported findings in porcine brain intraparenchymal microvessels (Li et al., 1994). Our results, however, further indicate that distribution of EP1- and EP3-receptors in the medial smooth muscle is different between these two receptor subtypes. The EP1-receptor immunoreactivities were found primarily in association with the entire layers of the medial smooth muscle cells (Fig 3A). This is consistent with the finding that the EP1-receptor–immunoreactive profile in the whole-mount porcine basilar arteries and the circle of Willis is similar in size and (elongated) shape to that of a smooth muscle cell that runs circularly akin to the smooth muscle cell arrangement in the arterial wall (Lee, 1982; Shiraishi et al., 1990) (Fig 3B). The EP3-receptor-immunoreactivities, however, were found in the smooth muscle only at the outer medial layer bordering the adventitia (Figs. 3D and 3E). These findings are also supported by the presence of EP1- and EP3-receptor proteins in cultured smooth muscle cells (Figs. 3H and 4).
The EP3-receptor immunoreactivities were associated with bundles and fine fibers in the adventitia (Figs. 3E–3G). These fibers were coincident with neurofilament-200–immunoreactive fibers, suggesting that EP3-receptors are intimately associated with the perivascular neurons. Although the EP1-receptor immunoreactivities were also associated with bundlelike structures in the adventitia, the immunoreactivities were relatively sparse and not manifested as fine fibers in the whole-mount arterial preparations. The exact role of the adventitial neuronal EP1- and EP3-receptors remains to be determined. Because perivascular nerves and endothelial cells are not obligatory for PGE2-induced constriction, both EP1- and EP3-receptors on the smooth muscle cells most likely mediate the constriction of porcine large cerebral arteries induced by PGE2 and its agonists. Our findings are consistent with the reports by others that EP1-receptors mediate constriction in human pulmonary venous smooth muscle (Walch et al., 2001) and in the nonvascular smooth muscle of the trachea, gastrointestinal tract, uterus, and bladder (Asboth et al., 1996; Coleman et al., 1994), as do the EP3-receptors in the ileum and myometrium (Asboth et al., 1996; Breyer and Breyer, 2000; Coleman et al., 1994).
The presence of EP3-receptor (but not EP1-receptor) immunoreactivites in the nuclear compartment of the cultured smooth muscle cells (Fig. 3H) is interesting and suggests a possible nucleus-associated site for EP3-receptors in cerebral vascular smooth muscle cells. The presence of perinuclear EP3-receptors has recently been shown in cerebral vascular endothelial cells where they function in regulating constitutive eNOS gene (Gobeil et al., 2002). The functional significance of this receptor subtype in the nuclear compartment of smooth muscle cells remains to be determined.
The EP1-receptor has been shown to signal via IP3 generation with an increase in cellular calcium (Båtshake et al., 1995; Tabata et al., 2002; Watabe et al., 1993). In the brain microvessels, PGE2 and 17-PGE2 have been shown to increase IP3 formation (Li et al., 1994). U-73122 and neomycin, which are phospholipase C inhibitors, have been shown to inhibit the hydrolysis of phosphoinositide to IP3 leading to a decrease in cytosolic free calcium and inhibition of the coupling of G protein-phospholipase C activation (Bleasdale et al., 1990; Vollrath et al., 1994). In the present study, U-73122 and neomycin attenuated, in a concentration-dependent manner, the constriction induced by PGE2 and its EP1/EP3-receptor agonists, but not by KCl, in the basilar arteries and the circle of Willis of pigs, suggesting that the constriction was mediated by phosphatidyl–inositol pathway, possibly via activation of Gαq protein (Wright et al., 2001). Recent oocyte studies (Tabata et al., 2002) have shown that EP1-receptors have the potential to induce Ca+2 mobilization via Gq/11. One of the EP3-receptor subtypes (EP3D) also is coupled to Gq, evoking a phosphatidyl-inositol response (Narumiya et al., 1999).
The findings of the present study may be clinically relevant, particularly because PGE2 is one of the major mediators produced during certain cerebral vascular dysfunctions such as the cerebral vasospasm after SAH (Macdonald and Weir, 1991; Toda et al., 1991). In SAH, prostaglandins and HbO2 have been invoked as causative agents (Macdonald and Weir, 1991). It has been shown that PGE2 is the eicosanoid present in the highest concentration in the CSF after SAH (Walker et al., 1983). The levels of PGE2 in the CSF have been shown to increase 90-fold in dogs (Chyatte, 1989) and up to 250-fold in humans (Pickard et al., 1994) after SAH. In addition, our present experiments showed that constriction induced by PGE2 was greatly potentiated by HbO2, supporting a potential role for PGE2 in SAH and suggesting a possible mechanism of interaction between these putative causative agents. This is consistent with the reported role of PGE2 along with hemoglobin and other eicosanoids in cerebral vasospasm (Chyatte, 1989; Macdonald and Weir, 1991; Pickard et al., 1994; Toda et al., 1991; Walker et al., 1983).
It may be interesting to note that in SAH, the outer layer smooth muscle cells bearing EP1- and EP3-receptors by the virtue of their close proximity to the adventitia would be in earlier contact with the spasmogens released from the subarachnoid blood clot than would the majority of EP1-receptors in the inner muscle layer. These latter EP1-receptors probably are more importantly involved in the physiologic control than in the pathologic control of the vascular function because they are acted upon by the PGE2 released from the endothelium. Although the exact role of these receptors remains to be elucidated, this study lays the platform to further investigate their function in the aforementioned cerebrovascular pathologies.
It is known that, albeit limitations in studying a complex phenomenon such as SAH, the cerebral vascular ring-segment–mounted tissue bath method has supported most of the isolated in vitro work in cerebrovascular spasm (Lee et al., 1984; Cook, 1995). In our study, both PGE2-induced constriction and its potentiation by HbO2 were blocked by neomycin, a phospholipase C inhibitor, and SC-19220, a selective EP1-receptor antagonist. Neomycin not only suppressed the potentiation, but also reversed significantly the PGE2-induced contraction after HbO2 incubation. This is consistent with the finding that activation of phospholipase C may be a critical step in the development of cerebral vasospasm (Laher and Zhang, 2001; Vollrath et al., 1994). On this premise, neomycin and selective EP receptor antagonists may warrant further study as likely strategies in combating conditions such as cerebral vasospasm.
To summarize, the present study shows through multifaceted approaches that PGE2 causes an exclusive constriction of adult porcine large cerebral arteries. The constriction is likely mediated by the phosphatidyl-inositol pathway via activation of EP1- and EP3-receptors on smooth muscle cells. The expression of these receptors, which is likely to be age dependent according to available reports, may play important roles in cerebral vascular tone regulation in health and disease.
Footnotes
Acknowledgements
The authors thank Steve Markwell for advice on statistical analysis and Min-Liang Si for advice on tissue bath experiments.
