Abstract
AMPA receptor potentiators enhance AMPA receptor-mediated glutamatergic neurotransmission and may have therapeutic potential as cognitive enhancers or antidepressants. The anatomical basis for the action of AMPA receptor potentiators is unknown. The aim of this study was to determine the effects of the biarylpropylsulfonamide AMPA receptor potentiator, LY404187 (0.05 to 5 mg/kg subcutaneously), upon cerebral glucose utilization and c-fos expression using 14C-2-deoxglucose autoradiography and c-fos immunocytochemistry. LY404187 (0.5 mg/kg) produced significant elevations in glucose utilization in 28 of the 52 anatomical regions analyzed, which included rostral neocortical areas and the hippocampus, as well the dorsal raphe nucleus, lateral habenula, and locus coeruleus. No significant decreases in glucose utilization were observed in any region after LY404187 administration. The increases in glucose utilization with LY404187 (0.5 mg/kg) were blocked by pretreatment with the AMPA receptor antagonist LY293558 (25 mg/kg), indicating that LY404187 acts through AMPA receptor-mediated mechanisms. LY404187 (0.5 mg/kg) also produced increases in c-fos immunoreactivity in the cortex, locus coeruleus, and the dorsal raphe nucleus. These studies demonstrate neuronal activation in key brain areas that are associated with memory processes and thus provide an anatomical basis for the cognitive enhancing effects of AMPA receptor potentiators.
AMPA (α-amino-3-hydroxy-5-methyl-4-isoxazolepropanoic acid) receptors, a subtype of ionotropic glutamate receptor, mediate fast excitatory neurotransmission and are ubiquitously distributed throughout the central nervous system. Activation of AMPA receptors contributes to the induction of long-term potentiation (LTP), which is suggested to be a major neural substrate for the formation and development of learning and memory (Bliss and Collingridge, 1993; Lynch and Baudry, 1984). Deficits in glutamatergic neurotransmission and AMPA receptor antagonism often result in cognitive deficits (Filliat et al., 1998; Segovia et al., 2001; Tamminga, 1998). In contrast, enhancing glutamatergic neurotransmission or AMPA receptor-mediated transmission appears to enhance cognition and may therefore be therapeutically beneficial where cognitive function is impaired (Lynch et al., 1997; Staubli et al., 1994a, b ). Indeed, many early nootropic drugs (piracetam, aniracetam) were reported to positively modulate AMPA receptors (Copani et al., 1992).
AMPA receptor potentiators are positive allosteric modulators that increase current flux through the ion channel in the presence of an agonist, by reducing desensitisation and/or deactivation (Partin et al., 1994; Yamada and Tang, 1993). A number of different classes of AMPA receptor potentiator exist: pyrrolidinones, such as piracetam and aniracetam (Gouliaev and Senning, 1994; Ito et al., 1990); structurally related benzamide compounds, such as CX-516 and 1-BCP (Granger et al., 1993; Staubli et al., 1994a, b ); and thiazide derivatives, such as cyclothiazide, diazoxide and IDRA-21 (Yamada and Rothman, 1992; Yamada and Tang, 1993; Zivkovic et al., 1995). As predicted from their ability to enhance LTP in vivo (Staubli et al., 1994b), many of these compounds have demonstrated cognitive enhancing effects in rodent models of cognition (Granger et al., 1993; Staubli et al., 1994a, b ; Zivkovic et al., 1995). In addition, these compounds are suggested to be efficacious in animal models of depression (Knapp et al, 2002; Li et al, 2001).
A novel series of biarylpropylsulfonamide AMPA receptor potentiators has recently been developed (Ornstein et al., 2000), which are highly potent, selective, and centrally active (Linden et al., 2001; Vandergriff et al., 2001). LY404187 is a representative compound of this series, which has efficacy in rodent models of depression and cognition (O'Neill et al., 2004a; Quirk and Nisenbaum, 2002) and Parkinson's disease (O'Neill et al., 2004b). These molecules also appear to increase brain-derived neurotrophic factor (BDNF) expression in the hippocampus (Mackowiak et al., 2002), which may contribute to the activity observed in the in vivo models, but the overall anatomical basis of the action of AMPA receptor potentiators is not known. Therefore, the aim of the current study was to determine the effects of LY404187 upon cerebral glucose utilization using 14C-2-deoxyglucose autoradiography and c-fos expression to gain insight into the specific anatomical sites of action of AMPA receptor potentiators.
MATERIALS AND METHODS
Preparation of animals for 2-deoxyglucose autoradiography
Thirty-eight adult male Sprague Dawley rats (Harlan Olac, UK) (345–405 g) were anesthetized initially in a perspex box filled with 5% halothane in a 30% oxygen/70% nitrous oxide mixture. When deeply anesthetized, rats were transferred to a face mask where anesthesia was maintained with 2 to 2.5% halothane. Polythene catheters (external diameter 0.96 mm, internal diameter 0.58 mm diameter; SIMS Portex Ltd) containing heparinized saline were inserted into the femoral vein and artery to allow for the injection of 14C-2-deoxyglucose and the sampling of blood, respectively. Local anesthetic gel (xylocaine) was applied to the incision site, which was sutured. A bandage and loose fitting plaster cast was carefully wrapped around the body and hind limbs, which were taped to a lead brick to restrain the animal. A rectal probe was inserted to allow temperature to be monitored. At this stage, the anesthetic was discontinued, and the animals were left for 2 hours before the start of the 2-deoxyglucose procedure. Animals were maintained at 37°C with the aid of heating lamps. Arterial blood pressure was monitored throughout the subsequent procedure.
Measurement of local cerebral glucose utilization
Local cerebral glucose utilization was measured in conscious rats using an experimental design described by Sokoloff et al. (1977). At time zero, 50 μCi [14C]-2-DG dissolved in 0.5 mL of saline (specific activity 51 mCi/mol, Sigma, UK) was administered into the venous catheter at a constant rate for 30 seconds. Fourteen arterial blood samples (approximately 100 μL) were taken over the 45-minute experimental period, beginning at the onset of the administration of 14C-2-DG. Arterial samples were immediately centrifuged. Plasma samples were then assayed for glucose levels, using a semiautomated glucose oxidase enzyme assay (Glucose Analyser 2, Beckman, UK) and for 14C-2-DG concentration by means of liquid scintillation counting. Forty-five minutes subsequent to the isotope administration, the animals were decapitated, and each brain was dissected and frozen in isopentane maintained at −42°C for 10 minutes. Coronal sections (20 μm) were cut in a cryostat at −15°C. Three sections were retained from every 13 sections cut throughout the brain and cerebellum; however, extra sections were retained from regions of interest. Triplicate sections were retained on coverslips and dried quickly on a hotplate at 60°C. Autoradiograms were generated by exposing the brain sections to x-ray film (Kodak BioMax MR-1 film, Sigma, UK), with a set of 14 predetermined 14C standards, for 3 days. Local tissue concentrations of 14C, and local rates of cerebral glucose utilization, were calculated in 52 brain areas using quantitative densitometric analysis with an MCID image analyser (MCID-M4, Imaging Research, Canada), based upon the operational equation derived by Sokoloff et al. (1977).
Drug administration for 2-deoxyglucose studies
Previous studies have indicated that peak plasma levels of the AMPA receptor potentiator LY404187 are observed for 1 to 2 hours after subcutaneous administration. The AMPA receptor potentiator LY404187 (0.05, 0.5, 5 mg/kg) or vehicle (30% ethanol, 35% hydroxy-propyl-β-cyclodextrin) were therefore administered subcutaneously 15 minutes before administration of the 14C-2-deoxyglucose (n = 28). Ten animals received intravenous administration of the AMPA receptor antagonist, LY293558 (25 mg/kg), dissolved in NaOH and 5.5% glucose solution (final pH 8.5, isotonic solution), 5 minutes before subcutaneous administration of the AMPA receptor potentiator, LY404187 (0.5 mg/kg), or vehicle. The dose of AMPA antagonist was selected on the basis of effects of LY293558 upon glucose utilization demonstrated in a previous study (Browne and McCulloch, 1994). Previous studies suggest that the AMPA receptor antagonist LY293558 has maximal effects 60 minutes after administration.
Drug administration for c-fos studies
The effects of AMPA receptor administration upon c-fos immunocytochemistry were determined in a separate group of animals, to confirm if metabolic activation occurred in animals that had not been anesthetized, cannulated, and restrained. C-fos is an immediate early gene transiently expressed by physiologically active cells (Sagar et al., 1988). C-fos immunocytochemistry is commonly used to determine cellular activation after pharmacologic drug administration (reviewed in Sharp et al., 1993). Adult male Sprague-Dawley rats (Harlan Olac, UK) (n = 32) received a single subcutaneous injection of the AMPA receptor potentiator LY404187 (0.05, 0.5, 5 mg/kg), or vehicle (30% ethanol, 35% hydroxy-propyl-β-cyclodextrin).
C-fos immunocytochemistry
Six hours after injection, the rats were anesthetized with isoflurane and transcardially perfused with 0.9% saline followed by 10% phosphate-buffered formalin (pH 7.4). The brains were postfixed in formalin for 1 to 2 days, then 5-mm coronal blocks were processed and embedded in paraffin wax. Serial 8-μm sections were cut on a microtome from each block and mounted onto Snowcoat X-tra slides (Surgipath, UK). Sections from the level of the frontal cortex, parietal cortex (bregma 1.2 mm), hippocampus, medial habenula (bregma −3.30 mm), substantia nigra (bregma −4.3 mm), dorsal raphe (bregma −7.3 mm), and locus coeruleus (bregma −9.8 mm) were selected for c-fos immunostaining (as described previously by Dawe et al., 2001). Briefly, sections were placed in xylene to remove the wax, then rehydrated and incubated in 0.3% H2O2 for 30 minutes to quench endogenous peroxidase. After three washes in 0.01 M phosphate buffered saline (PBS), the sections were incubated with 0.4% pepsin (Sigma, UK) in 0.01 M HCl, washed in PBS a further two times, then incubated with anti–c-fos (goat polyclonal c-fos, sc-52, Santa Cruz Biotechnology, UK 1:400) for 24 hours at 4°C. The next day, the sections were washed in PBS and then incubated in biotinylated goat anti-rabbit (1:200, Vector Labs, UK) for 20 minutes. After a further two washes in PBS, the sections were incubated with avidin-biotin complex (Vector Labs, UK), then visualized using 0.02% diaminobenzide with 0.02% hydrogen peroxide. The sections were then dehydrated and placed in xylene, and coverslips were mounted.
Quantification of c-fos immunoreactivity
Images of c-fos immunopositive cells from selected areas of interest were captured digitally at a magnification of x250. The images were opened using the image analysis software (ImagePro Plus, UK), and the number of c-fos immunopositive cells was counted in the anatomical regions of interest. The software allowed the user to click on each cell counted, and this was then automatically tagged. Counts were made in triplicate on both left and right hemisphere and then averaged for each brain area.
Statistical analysis
All 2-deoxyglucose and c-fos data for each anatomic region were analyzed for statistical significance by analysis of variance, then post hoc Student's t-test. For the 2-deoxyglucose studies, a Bonferroni correction factor of 6 was applied to the probability values, to take into consideration the multiple comparisons between drug treated and control groups.
RESULTS
Effect of AMPA receptor potentiator LY404187 upon physiologic parameters
Before drug administration, rats were generally inquisitive, with periods of movement and grooming. LY404187 produced no overt changes in this behavioral pattern. In contrast, administration of the AMPA receptor antagonist LY293558 produced sedative effects, reduced motor activity, and a loss of responsiveness to auditory stimuli. This sedative effect continued after the administration of the AMPA receptor potentiator, LY404187, 5 minutes after administration of LY293558. LY404187 produced no significant changes in rectal temperature, blood glucose levels, or arterial blood pressure (Table 1).
Physiologic parameters in 2-deoxyglucose experiments
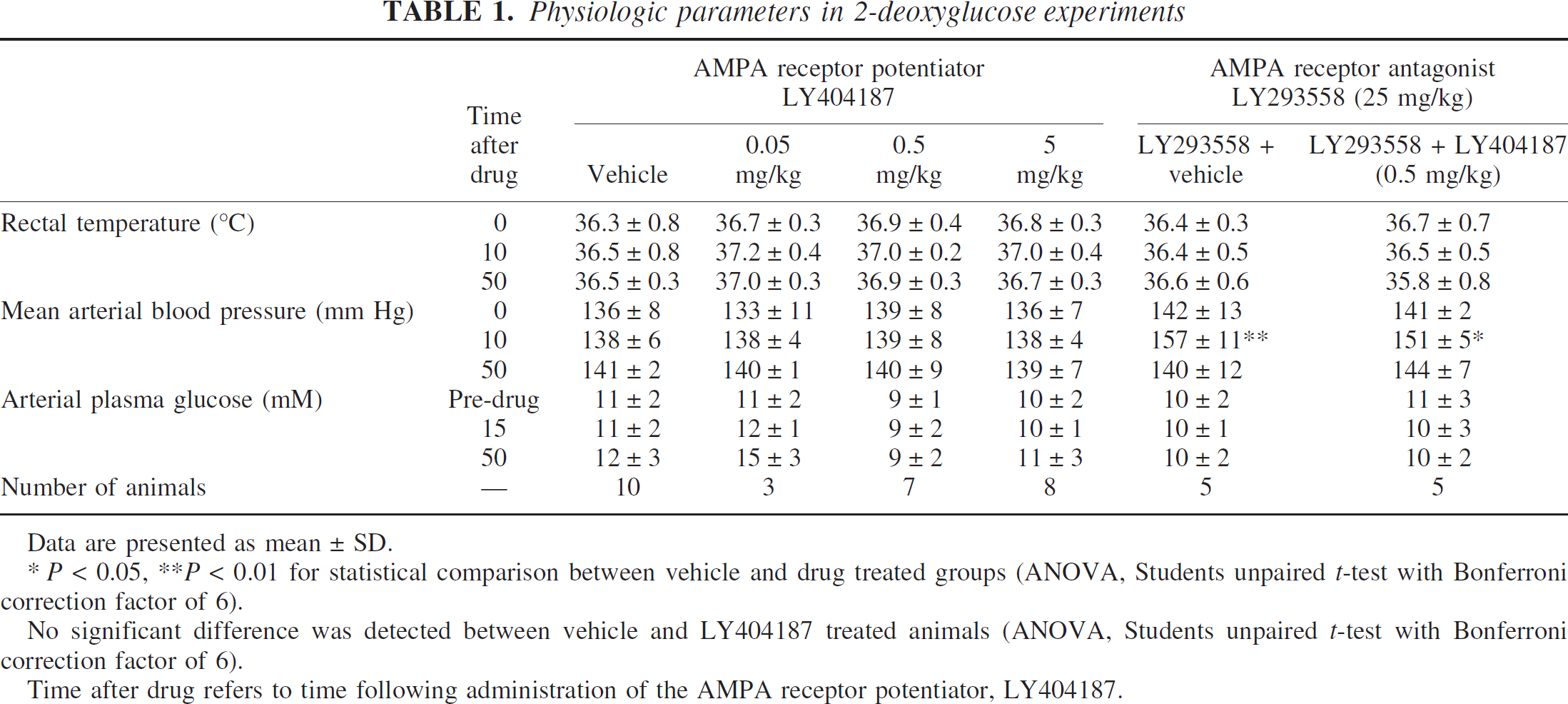
Data are presented as mean ± SD.
* P < 0.05, **P < 0.01 for statistical comparison between vehicle and drug treated groups (ANOVA, Students unpaired t-test with Bonferroni correction factor of 6).
No significant difference was detected between vehicle and LY404187 treated animals (ANOVA, Students unpaired t-test with Bonferroni correction factor of 6).
Time after drug refers to time following administration of the AMPA receptor potentiator, LY404187.
LY404187 and cerebral glucose utilization
The effects of the AMPA receptor potentiator LY404187 (0.05, 0.5, 5 mg/kg) compared with administration of the vehicle are presented in Tables 2–6. LY404187 (0.05, 0.5, 5 mg/kg) produced dose-dependent alterations in glucose utilization. Significant alterations in glucose utilization were observed only at the middle dose examined (0.5 mg/kg), which produced increases in 28 of the 52 anatomical areas examined. In contrast, the other doses of LY404187 (0.05 or 5 mg/kg) did not produce any significant increases in glucose utilization, with the exception of the cerebellar cortex, where glucose utilization was significantly increased after administration of the highest dose of LY404187 (5 mg/kg). Administration of LY404187 (5.05, 0.5 or 5 mg/kg) did not produce any significant decreases in glucose utilization in any of the anatomic areas studied. The areas in which LY404187 (0.5 mg/kg) caused elevations in glucose utilization included the hippocampus, several of the cortical areas examined (Figs. 1 and 2), the locus coeruleus, dorsal raphe nucleus, and the lateral habenula (Figs. 2 and 3). Other brain areas such as the hypothalamus (Fig. 1D) were relatively unaffected by LY404187 (0.5 mg/kg).
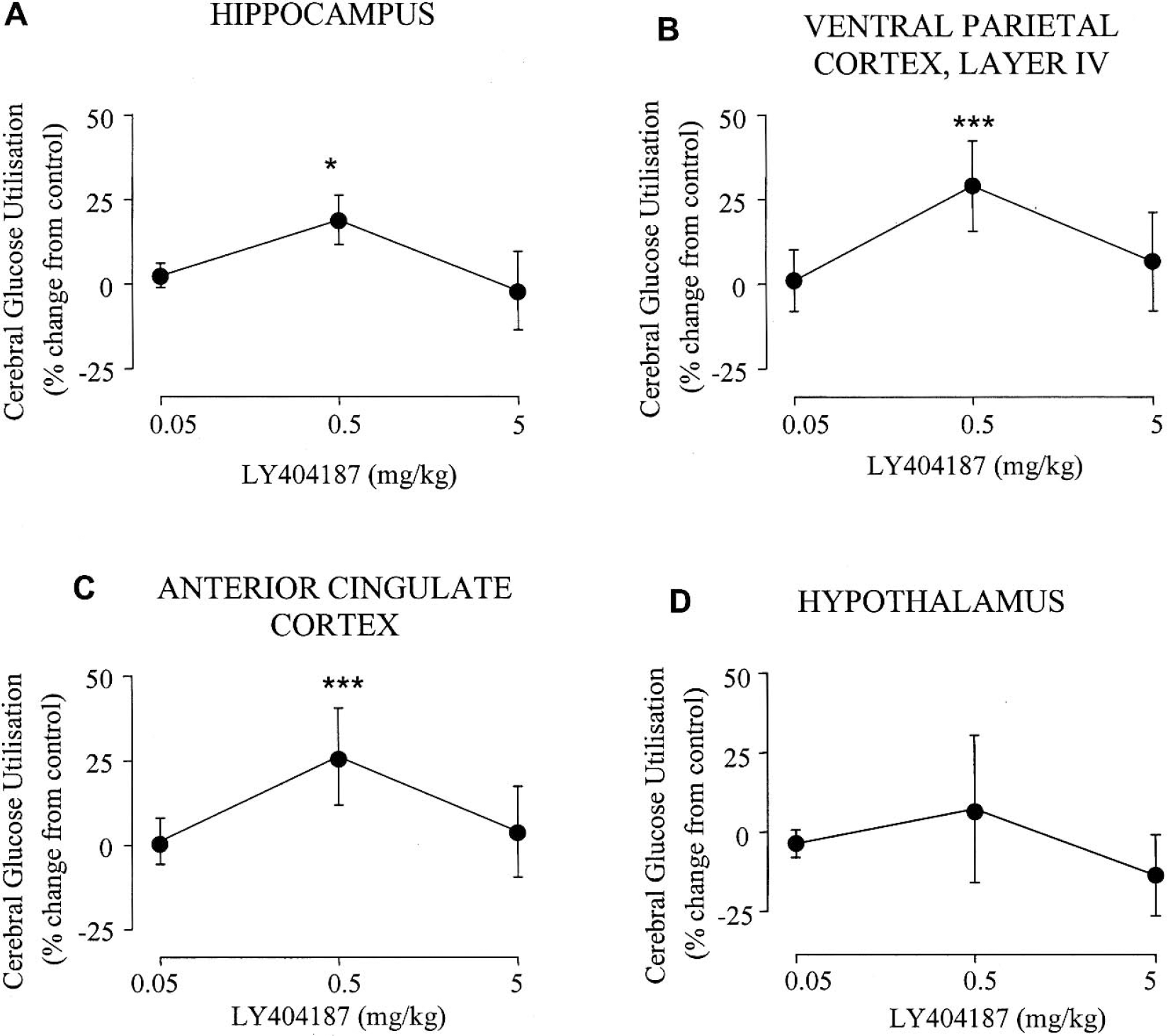
Dose-dependent effects of the AMPA receptor potentiator in four anatomic brain regions. The data are presented as percentage change in glucose utilization relative to the mean glucose utilization in vehicle-treated control animals (mean ± SD). The AMPA receptor potentiator LY404187 (0.5 mg/kg) caused increases in glucose utilization in the hippocampus (A), layer four of the parietal cortex (B), and the anterior cingulate cortex (C). In contrast, LY404187 (0.5 mg/kg) did not cause significant increases in glucose utilization in other brain areas such as the hypothalamus (D). *P < 0.05, ***P < 0.001 for comparison between vehicle and drug treated groups.
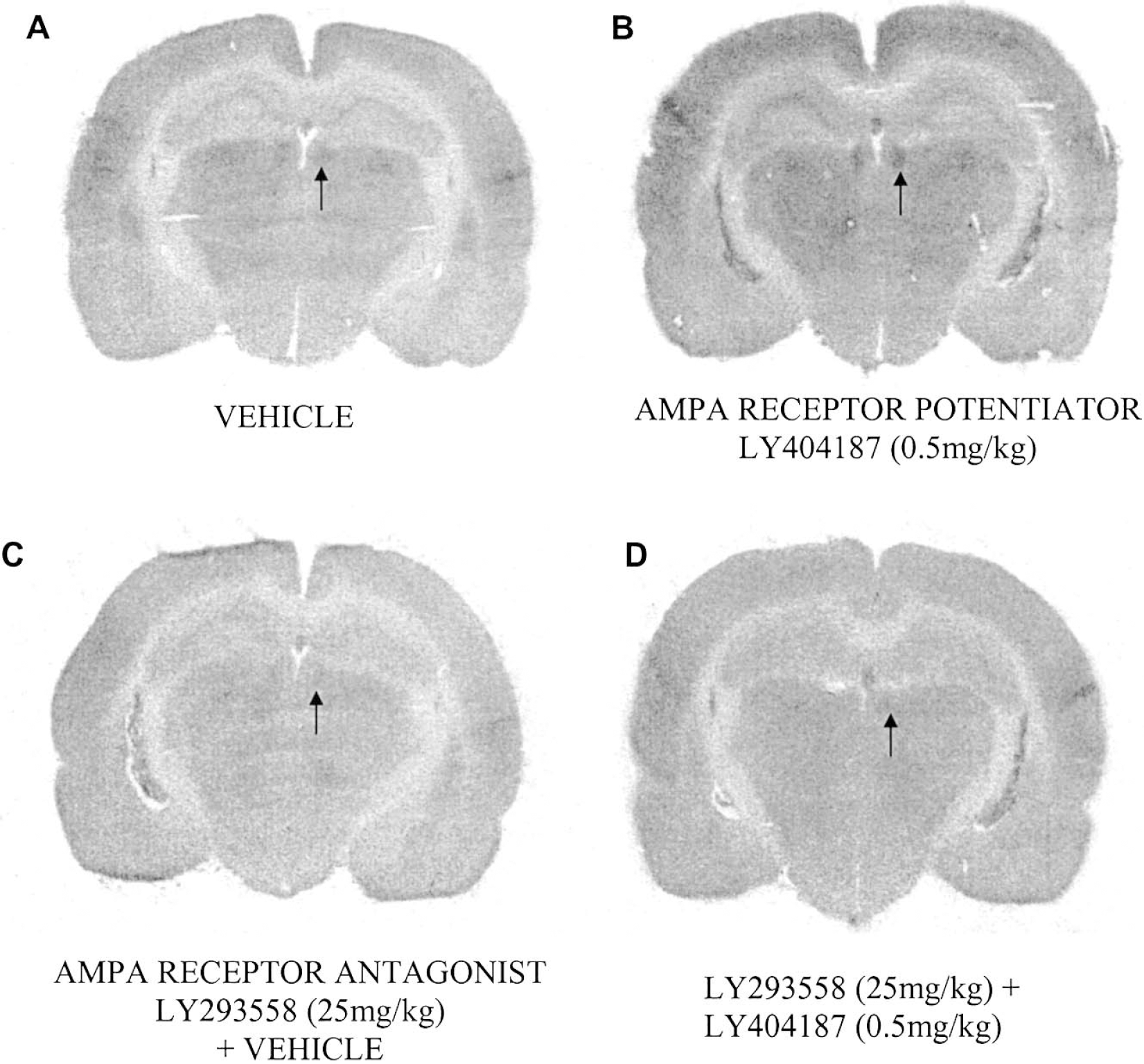
Representative autoradiograms illustrating changes in local cerebral glucose utilization in the hippocampus, parietal cortex and lateral habenula. Increases in glucose utilization were evident in the parietal cortex, hippocampus, and lateral habenula after administration of the AMPA receptor potentiator, LY404187 (0.5 mg/kg) (B) compared with administration of the vehicle (A). In contrast, administration of the AMPA receptor antagonist, LY293558, caused decreases in glucose utilization in these brain areas (C). Administration of the AMPA receptor antagonist LY293558 (25 mg/kg) before administration of the AMPA receptor potentiator LY404187 (0.5 mg/kg) attenuated the effects of this drug upon glucose utilization (D)
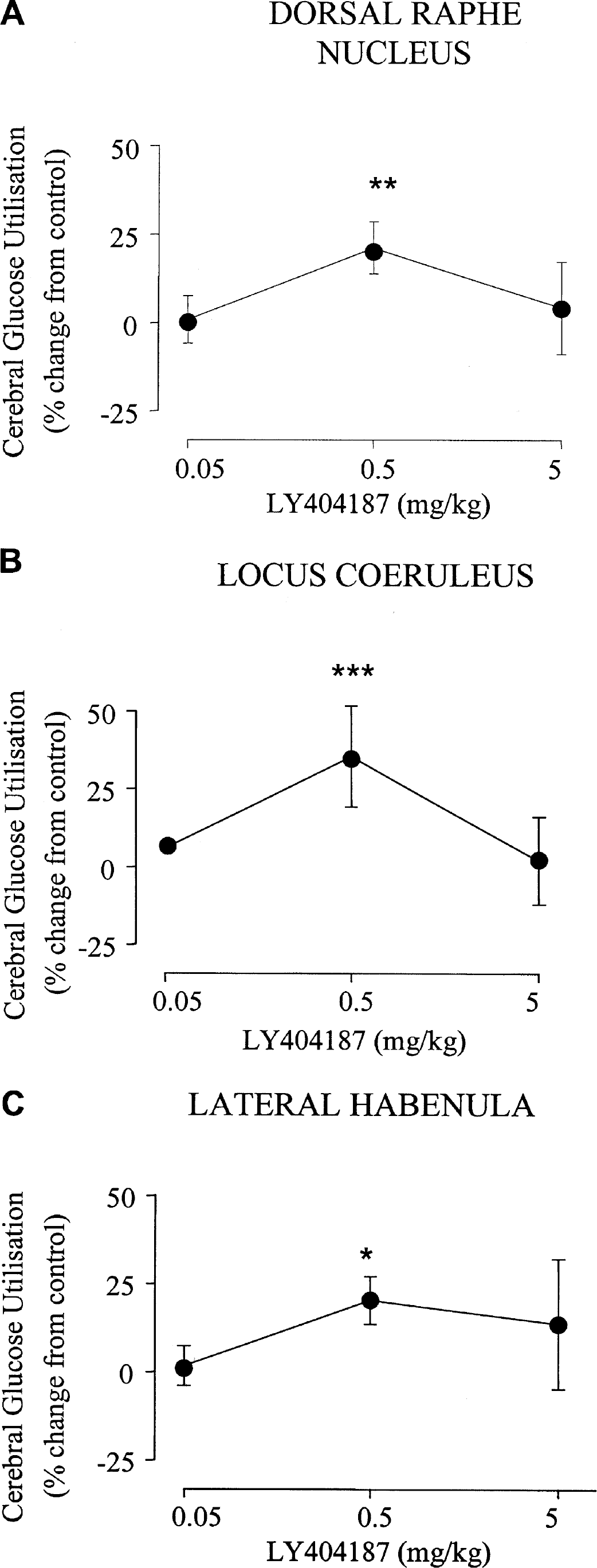
Dose-dependent effects of the AMPA receptor potentiator in the locus coeruleus, raphe nucleus, and lateral habenula. The data are presented as percentage change in glucose utilization relative to the mean glucose utilization in vehicle-treated control animals (mean ± SD). The AMPA receptor potentiator LY404187 (0.5 mg/kg) caused increases in glucose utilization in the dorsal raphe (A), the locus coeruleus (B), and the lateral habenula (C). *P < 0.05, **P < 0.01, ***P < 0.001 for comparison between vehicle and drug treated groups.
Effects of the AMPA receptor potentiator LY404187 upon cerebral glucose use (μmol/100 g/min) in anatomic areas of the auditory and visual system
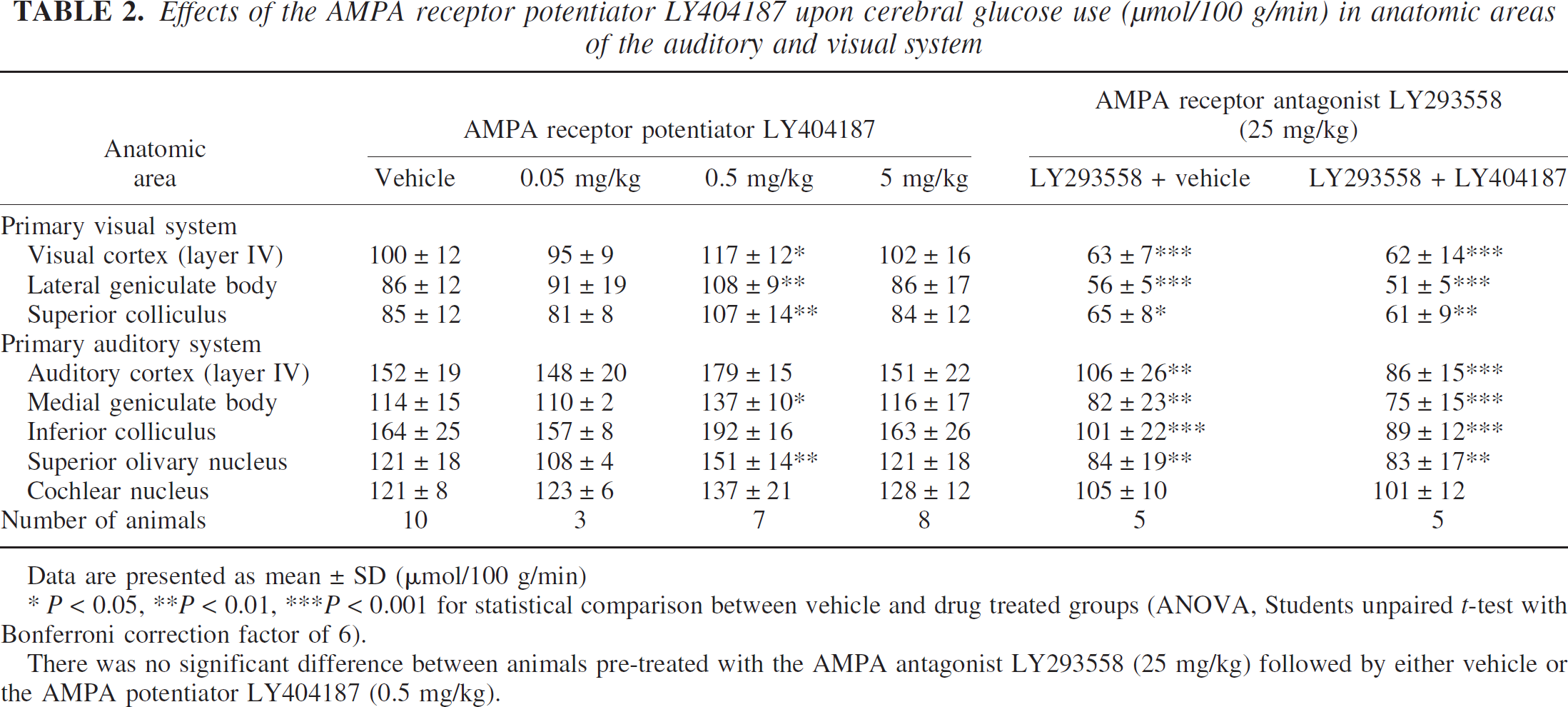
Data are presented as mean ± SD (μmol/100 g/min)
* P < 0.05, **P < 0.01, ***P < 0.001 for statistical comparison between vehicle and drug treated groups (ANOVA, Students unpaired t-test with Bonferroni correction factor of 6).
There was no significant difference between animals pre-treated with the AMPA antagonist LY293558 (25 mg/kg) followed by either vehicle or the AMPA potentiator LY404187 (0.5 mg/kg).
Effects of the AMPA receptor potentiator LY404187 upon cerebral glucose use (μmol/100 g/min) in cerebral cortical areas
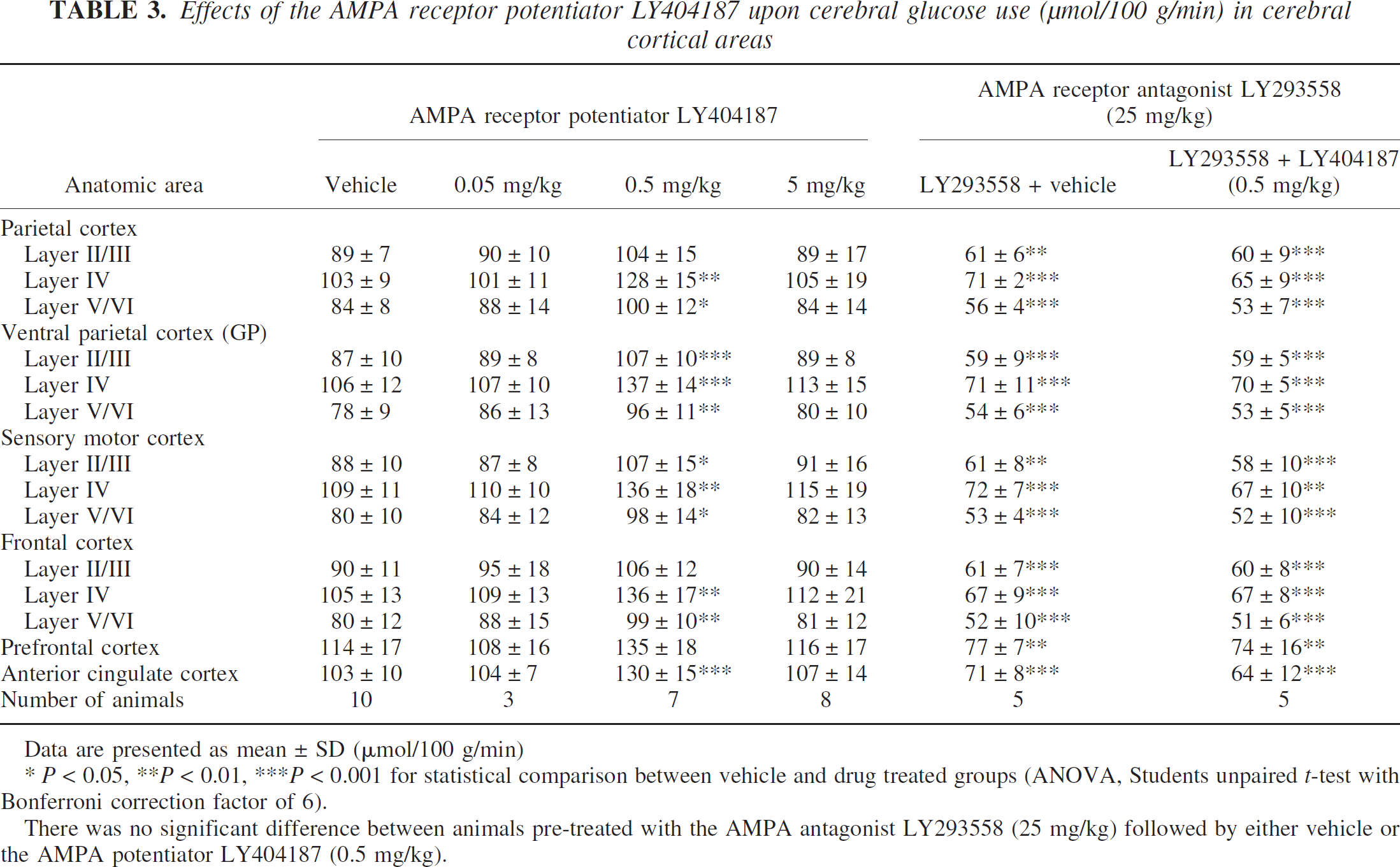
Data are presented as mean ± SD (μmol/100 g/min)
* P < 0.05, **P < 0.01, ***P < 0.001 for statistical comparison between vehicle and drug treated groups (ANOVA, Students unpaired t-test with Bonferroni correction factor of 6).
There was no significant difference between animals pre-treated with the AMPA antagonist LY293558 (25 mg/kg) followed by either vehicle or the AMPA potentiator LY404187 (0.5 mg/kg).
Effects of the AMPA receptor potentiator LY404187 upon cerebral glucose use (μmol/100 g/min) in extrapyramidal and sensory-motor areas
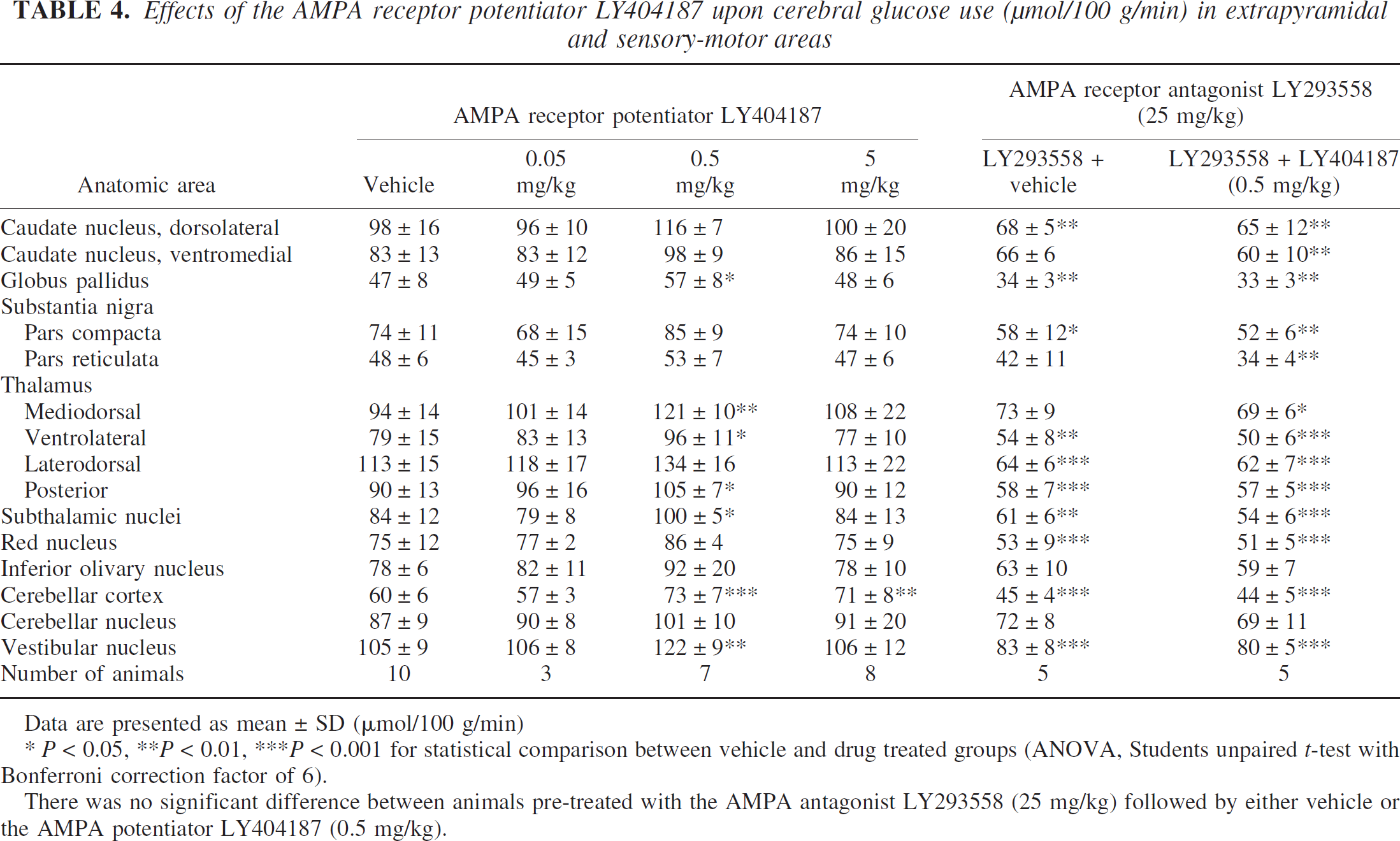
Data are presented as mean ± SD (μmol/100 g/min)
* P < 0.05, **P < 0.01, ***P < 0.001 for statistical comparison between vehicle and drug treated groups (ANOVA, Students unpaired t-test with Bonferroni correction factor of 6).
There was no significant difference between animals pre-treated with the AMPA antagonist LY293558 (25 mg/kg) followed by either vehicle or the AMPA potentiator LY404187 (0.5 mg/kg).
Effects of the AMPA receptor potentiator LY404187 upon cerebral glucose use in anatomic areas of limbic and functionally non-specific areas
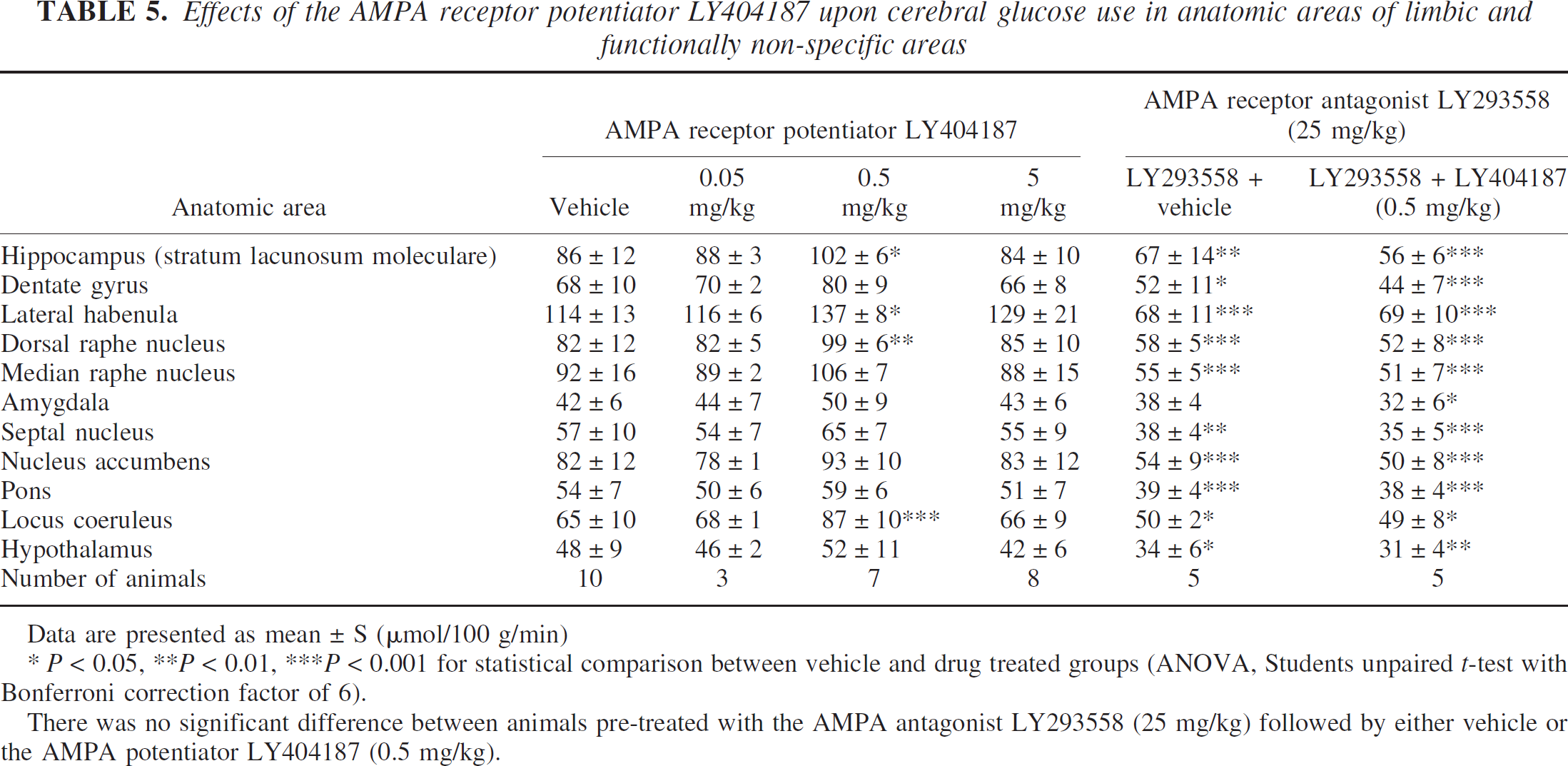
Data are presented as mean ± S (μmol/100 g/min)
* P < 0.05, **P < 0.01, ***P < 0.001 for statistical comparison between vehicle and drug treated groups (ANOVA, Students unpaired t-test with Bonferroni correction factor of 6).
There was no significant difference between animals pre-treated with the AMPA antagonist LY293558 (25 mg/kg) followed by either vehicle or the AMPA potentiator LY404187 (0.5 mg/kg).
Effects of the AMPA receptor potentiator LY404187 upon cerebral glucose use in white matter tracts

Data are presented as mean ± SD (μmol/100 g/min)
* P < 0.05, **P < 0.01, ***P < 0.001 for statistical comparison between vehicle and drug treated groups (ANOVA, Students unpaired t-test with Bonferroni correction factor of 6).
There was no significant difference between animals pre-treated with the AMPA antagonist LY293558 (25 mg/kg) followed by either vehicle or the AMPA potentiator LY404187 (0.5 mg/kg).
Administration of the AMPA receptor antagonist LY293558 before treatment with the AMPA receptor potentiator, LY404187
Pretreatment with the AMPA receptor antagonist LY293558 (25 mg/kg intravenously) significantly reduced glucose utilization in 43 of the 52 anatomical areas examined (Tables 2–6). There were no significant differences in glucose utilization in any of the anatomical areas in the animals treated with LY293558 (25 mg/kg) and vehicle compared with administration of LY293558 (25 mg/kg) and the AMPA receptor potentiator LY404187 (0.5 mg/kg).
Effects of LY404187 upon c-fos immunoreactivity
The effects of LY404187 upon c-fos immunoreactivity in the parietal cortex, dorsal raphe, locus coeruleus, and hypothalamus are presented in Fig. 4. The data indicated that LY404187 increased the number of c-fos positive cells in the parietal cortex (Figs. 4 and 5), dorsal raphe, and locus coeruleus (Fig. 4). The effect was most pronounced with LY404187 (0.5 mg/kg), and although the magnitude of the effect was small, it was significant in both the parietal cortex and dorsal raphe. The highest dose of LY404187 (5 mg/kg) also appeared to produce an increase in c-fos immunoreactivity, but this failed to reach statistical significance. In contrast to other brain regions, there was no change in the number of c-fos positive cells in the hypothalamus after any dose of LY404187 (Fig. 4D).
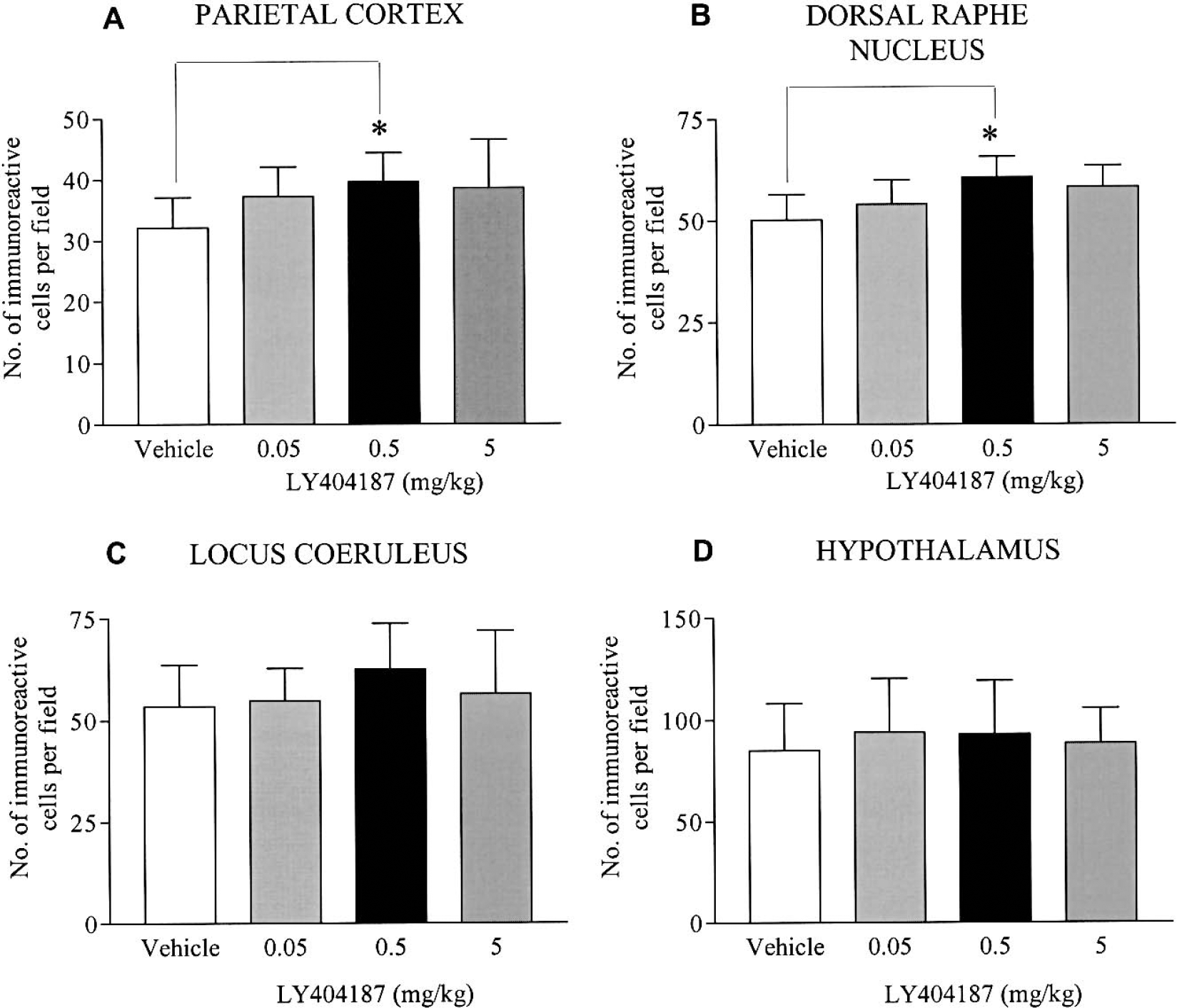
Quantification of the number of c-fos immunopositive cells in four anatomic areas after administration of the AMPA receptor potentiator, LY404187 or vehicle. Comparison of the effects of LY404187 (0.05, 0.5, and 5.0 mg/kg subcutaneously) upon the number of c-fos poitive cells in the parietal cortex (A), dorsal raphe (B), locus coeruleus (C), and hypothalamus (D). There were significant increases (P < 0.05) in c-fos immunoreactivity in the parietal cortex and dorsal raphe with 0.5 mg/kg dose of LY404187. Data are presented as mean ± SD (n = 8 per group). *P < 0.05 versus vehicle control.
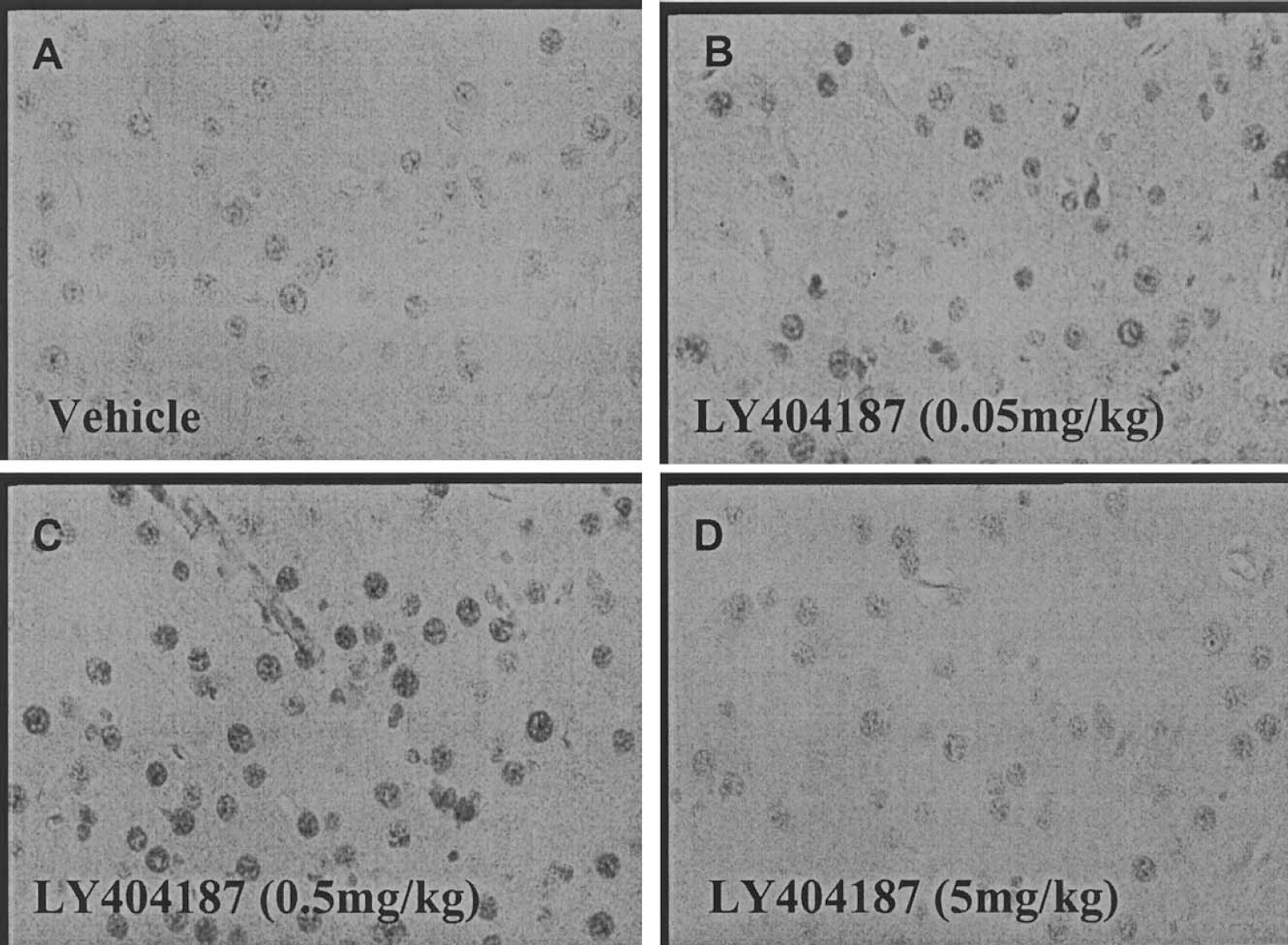
Images taken from parietal cortex in c-fos immunostained sections, after administration of vehicle, or the AMPA receptor potentiator, LY404187. Parietal cortex images from vehicle-treated (A) and LY404817-treated rats (B–D). There was a clear increase in c-fos immunoreactive cells with LY404187 (0.5 mg/kg).
DISCUSSION
Our results demonstrate that the AMPA receptor potentiator, LY404187 (0.5 mg/kg), significantly increased glucose utilization in 28 anatomically discrete regions, including the hippocampus, layer IV of the cerebral cortex, the raphe nucleus, and the locus coeruleus. This correlated with an increase in c-fos protein expression in these areas 6 hours after administration of LY404187 (0.5 mg/kg). These data provide an anatomical basis for the cognitive enhancing effects of LY404187 observed in other studies.
The cellular site of the local increases in glucose utilization caused by LY404187 is currently unclear. LY404187 has previously been shown to enhance ion influx in recombinant GLUA1–4 and to selectively enhance AMPA-mediated responses in native hippocampal neurons in vitro and in vivo (Gates et al., 2001; Vandergriff et al., 2001). We have also shown that the AMPA receptor antagonist LY293558 (25 mg/kg) attenuated the effects of LY404187 (0.5 mg/kg). The AMPA receptor potentiating effects of LY404187 at both recombinant and several types of native receptors can be completely blocked by AMPA receptor antagonists, such as NBQX and LY300168 (Baumbarger et al., 2001; Gates et al., 2001; Miu et al., 2001). Furthermore, LY404187 does not directly enhance currents at other ionotropic glutamate receptors, including GluR5 recombinant, or native kainate and NMDA receptors (Baumbarger et al., 2001; Gates et al., 2001). Collectively, these studies suggest that the effects of LY404187 upon glucose utilization are mediated via AMPA receptors. In the present studies LY404187 appeared to produce a bell-shaped response for both c-fos expression and glucose utilization. Even at the highest concentrations of LY404187, there was no evidence of the intense elevations in glucose utilization observed with AMPA or kainate receptor agonists (Browne et al., 1998). This is in agreement with pharmacologic studies that have reported activity in a range of rodent models with 0.5 mg/kg of LY404187 or at similar doses with the related compounds, LY392098 and LY451646 (Li et al., 2001; Mackowiak et al., 2002; O'Neill et al., 2004a, b ). The anatomical distribution of altered glucose utilization with the AMPA receptor potentiator LY404187 is also distinct from that observed previously with agents acting on other aspects of glutamatergic transmission, such as metabotropic glutamate agonists or NMDA receptor antagonists (Kurumaji et al., 1989; Lam et al., 1999).
AMPA receptors are widely distributed throughout the brain; however, in contrast to AMPA receptor antagonists, which produce widespread depression in glucose utilization (Browne and McCulloch, 1994; Suzdak and Sheardown, 1993), the AMPA receptor potentiator LY404187 produced increases in glucose utilization in only 28 out of the 52 brain areas analyzed. Anatomic areas such as the superficial layers of the cortex and stratum lacunosum-moleculare of the hippocampus, which contain the highest levels of AMPA receptors (Monaghan et al., 1984; Rainbow et al., 1984), did have significant elevations in glucose utilization after LY404187 administration. In contrast, LY404187 had no significant influence upon glucose utilization in other areas (caudate nucleus, nucleus accumbens, and the amygdala) with high levels of AMPA receptors. In addition, LY404187 was found to activate areas such as the lateral habenula and the lateral geniculate nucleus, which were reported to contain lower levels of [3H]AMPA binding sites (Monaghan et al., 1984). The anatomical distribution of altered glucose utilization after drug administration does not reflect the distribution of receptors but rather the polysynaptic circuits that can be modulated by the drug. The heterogeneity of AMPA receptor subunit composition in the brain may also contribute to the observed anatomical pattern of altered glucose utilization. LY404187 and related biarylpropylsulfonamides have different profiles (potency and efficacy) at potentiating recombinant GLUA1–4 (Miu et al., 2001) and on rat native neurones from various brain regions (E. S. Nisenbaum, personal communication, January, 2004). The distribution of the individual AMPA receptor subunits, GLUA1–4 in the rat brain, also varies from the pattern of [3H]AMPA binding (Petralia and Wenthold, 1992). For example, high levels of the glutamate receptor subunits GluR2/3 have been reported in the lateral habenula and lateral geniculate nucleus (Petralia and Wenthold, 1992). LY404187 is most potent at recombinant receptors composed of the GLUA2 subunit (GLUA2 > GLUA4 > GLUA3 > GLUA1) (Miu et al., 2001).
Administration of LY404187 produced activation of the locus coeruleus, the dorsal raphe nucleus, and the lateral habenula, a key relay nucleus from the forebrain to the midbrain raphe. The locus coeruleus and raphe nucleus are the origin of noradrenergic and serotonergic projections, which may have a modulatory influence over both memory processes (Buhot et al., 2000; Kobayashi and Yasoshima, 2001; Meneses, 1999) and may also contribute to the antidepressant effects of the drug. In recent years several new drug targets and animal models for development of antidepressants are emerging (Nestler et al., 2002). Of direct relevance to our current studies, LY404187 has been reported to increase levels of brain-derived neurotrophic factor BDNF in the hippocampus (Legutko et al., 2001, Mackowiak et al., 2002), at regions in which glucose utilization was also increased. BDNF is upregulated by many conventional antidepressants and may exert a range of trophic and protective effects upon monoaminergic neurons (Altar, 1999; Skolnick et al., 2001). It has also been demonstrated that direct infusion of BDNF into the hippocampus produces effects in animal models of depression (Shirayama et al., 2002). LY404187 and related AMPA potentiators are efficacious in the same rodent models (forced swim and tail suspension tests) of depression (Li et al., 2001; Quirk and Nisenbaum, 2002). It is also of interest to note that low doses of monoamine-based antidepressants can increase the efficacy of AMPA potentiators in the forced swim test (O'Neill et al., 2004a). Our present findings suggest that in addition to modulating BDNF, LY404187 also has actions on the biogenic amine neurotransmitter systems, and this may help provide a neurochemical basis for the antidepressant-like action of LY404187.
Multiple brain areas may contribute to the anatomical basis of memory; however there is a general consensus that the cortex and hippocampus play a pivotal role in the formation, storage, or retrieval of memories (Buckner et al., 1999; Fletcher et al., 1997; Milner et al., 1998). 14C-2-deoxglucose autoradiography studies have revealed activation of the hippocampus and cerebral cortex after a variety of cognitive tasks, aimed to test different memory entities in the nonhuman primate (Bontempi et al., 1999; Davachi and Goldman-Ravic, 2001; Sybirska et al., 2000). Intrahippocampal injections of AMPA antagonists markedly and selectively reduce glucose utilization in this region and disrupt encoding, retrieval, and storage of spatial memory (Riedel et al., 1999). The present study shows that the AMPA receptor potentiator LY404187 causes activation of the hippocampus and the frontal, anterior cingulate, and sensory motor cortex in the rodent, suggesting that this compound activates brain areas that are associated with memory formation.
The cognitive-enhancing effect of AMPA receptor potentiators has been demonstrated in a number of animal models of cognition. The cognitive enhancing effects of aniracetam and piracetam in delayed-nonmatching-to-sample and maze exploration tasks (Pontecorvo and Evans, 1985; Verloes, 1988) were demonstrated before the AMPA receptor mediated action of theses pyrrolidinones were discovered. Benzoylpiperidine compounds have since demonstrated positive effects in rodents in the radial arm maze, delayed-nonmatching-to-sample tests and the Morris water maze (Hampson et al., 1998; Staubli et al., 1994a, b ). IDRA-21 also improves rodent performance in the water maze (Zivkovic et al., 1995) and prevents pharmacologically induced cognitive impairments in nonhuman primates (Thompson et al., 1995). The active isomer of LY404187, LY451646, enhances rodent performance in two versions of the radial arm maze (Fowler et al, 2003; O'Neill et al., 2004a). The active dose range (0.1–0.3 mg/kg subcutaneously) equates to 0.2 to 0.6 mg/kg LY404187, and these doses produce activation of the hippocampus and cortex in the 2-deoxyglucose studies. Previous studies have indicated that LY404187 and related biarylpropylsulfonamides also improve performance in the water maze (Quirk and Nisenbaum, 2002) and passive avoidance tasks. Our results demonstrate that activation of the hippocampus and cerebral cortex may contribute to the anatomical circuitry involved in memory processes engaged during these tasks. As predicted from the effects of LY404187 upon glucose utilization and c-fos expression, the cognitive enhancing and antidepressant actions of structurally related AMPA receptor potentiators, LY392098 and LY451646, are absent when using higher doses of the drug (Li et al, 2001; D. L. McKinzie, personal communication, January, 2004). This bell-shaped response phenomenon also extends to other effects of AMPA receptor potentiators, such as their ability to enhance BDNF expression and neurogenesis in the rat brain (Bai et al., 2003; Mackowiak et al., 2002).
Administration of AMPA receptor potentiators to healthy humans is well tolerated, and positive cognitive effects in healthy young (Ingvar et al., 1997) and elderly (Lynch et al., 1997) have been reported. Deficits in glutamatergic transmission may contribute to cognitive decline in the elderly (Segovia et al., 2001). Disruption in glutamatergic transmission may also be a factor in the cognitive deficits observed in Alzheimer's disease and schizophrenia (Carlsson et al., 1999; Tamminga, 1998) and preliminary data suggest AMPA receptor potentiators may be beneficial in these diseases (Goff et al., 2001; Lee and Benfield, 1994). It has recently been reported that a related biarylpropylsulfonamide, LY451395, is tolerated in both healthy volunteers and elderly patients with probable Alzheimer's disease (Jhee et al., 2002). In the current studies we provide for the first time to our knowledge an anatomical basis for the preclinical effects of LY404187 in models of depression and cognition. These studies provide further evidence that LY404187, and related molecules may have therapeutic use in the treatment of cognitive deficits in certain neurologic conditions where glutamatergic hypofunction occurs.
Footnotes
Acknowledgment:
The authors thank Mrs. Joan Stewart at the Wellcome Surgical Institute.
