Abstract
In exercise, little is known about local cerebral glucose utilization (LCGU), which is an index of functional neurogenic activity. We measured LCGU in resting and running (≈85% of maximum O2 uptake) rats (n = 7 in both groups) previously equipped with a tail artery catheter. LCGU was measured quantitatively from 2-deoxy-D-[1-14C]glucose autoradiographs. During exercise, total cerebral glucose utilization (TCGU) increased by 38% (p < 0.005). LCGU increased (p < 0.05) in areas involved in motor function (motor cortex 39%, cerebellum ≈110%, basal ganglia ≈30%, substantia nigra ≈37%, and in the following nuclei: subthalamic 47%, posterior hypothalamic 74%, red 61%, ambiguus 43%, pontine 61%), areas involved in sensory function (somatosensory 27%, auditory 32%, and visual cortex 42%, thalamus ≈75%, and in the following nuclei: Darkschewitsch 22%, cochlear 51%, vestibular 30%, superior olive 23%, cuneate 115%), areas involved in autonomic function (dorsal raphe nucleus 30%, and areas in the hypothalamus ≈35%, amygdala ≈35%, and hippocampus 29%), and in white matter of the corpus callosum (36%) and cerebellum (52%). LCGU did not change with exercise in prefrontal and frontal cortex, cingulum, inferior olive, nucleus of solitary tract and median raphe, lateral septal and interpenduncular nuclei, or in areas of the hippocampus, amygdala, and hypothalamus. Glucose utilization did not decrease during exercise in any of the studied cerebral regions. In summary, heavy dynamic exercise increases TCGU and evokes marked differential changes in LCGU. The findings provide clues to the cerebral areas that participate in the large motor, sensory, and autonomic adaptation occurring in exercise.
Pronounced motor, sensory, and autonomic adaptation is known to occur in response to both static and dynamic exercise. Such changes must be thought to evoke marked regional changes in cerebral functional activity, but little is known about the effect of exercise on regional cerebral blood flow and in particular on regional cerebral metabolism, which are both indexes of the functional activity of regional neurons (Sokoloff, 1991, 1992). Results from investigations on cerebral blood flow during dynamic exercise are conflicting. Thus, in response to dynamic exercise, cerebral blood flow measurements using the 133Xe washout technique, which primarily measures cortical blood flow, have shown increased flow (Herholz et al., 1987; Thomas et al., 1989) or no change in flow (Globus et al., 1983). The radioactive microsphere method shows no change in total cerebral blood flow during dynamic exercise (Gross et al., 1980; Musch et al., 1987), whereas regional redistribution of cerebral flow has been demonstrated (Gross et al., 1980).
The quantitative 2-deoxy-D-[1-14C]glucose (2DG) method to measure local cerebral glucose utilization (LCGU) was introduced by Sokoloff et al. in 1977. With this technique 2DG is trapped intracellularly according to the metabolic activity of the cells, and the method is therefore a powerful tool for mapping regional differences in tissue glucose uptake with a high resolution. The method has been applied to study LCGU during a variety of physiological and pathophysiological conditions (Sokoloff, 1992) and has been modified to study local muscular glucose utilization during dynamic exercise (Vissing et al., 1988a). The effect of dynamic exercise on LCGU, however, has not been examined. Evaluation of LCGU during exercise may give important insight into which regions of the brain are involved in evoking the pronounced muscular, sensory, and autonomic adaptation occurring during exercise. In the present study, regional and total glucose metabolism in the brain was investigated during dynamic exercise using the quantitative 2DG method. Experiments were performed in rats at rest and during running on a treadmill at ≈85% of maximum oxygen consumption.
METHODS
The experiments were performed in 14 male Wistar rats weighing between 353 and 408 g. The rats were kept in a room with a constant temperature of 23°C, with lights on from 6 a.m. to 6 p.m., and with free access to water and ordinary rat chow. The rats were accustomed to running unforced on a rodent treadmill at a speed of 28 m/min. The rats ran for 35 min daily in the 7 days preceding the experiment. Rats were excluded if they were unwilling to run unforced or lost weight during the training period.
Surgery
Approximately 20 h before the experiment, the rat was anesthetized with Immobilon Vet (0.01 ml/100 g body wt s.c., a neuroleptic anesthetic containing 0.125 mg etorphine and 0.4 mg acepromazine/ml; Pharmacia, Denmark), and the caudal artery at the base of the ventral part of the tail was cannulated. Ampicillin (5 mg/100 g body wt) was administered to prevent infection and the cannula was flushed with heparin (0.3 ml, 500 IE/ml). The cannula was a modified version of a Venflon (0.8 mm O.D., length 25 mm; Viggo AB, Helsingborg, Sweden). A thin metal net was wrapped around the tail to protect the catheter. An Immobilon antidote, Revivon Vet (0.01 ml/100 g body wt s.c., containing 6 mg diprenorphine/ml; Pharmacia) was administered postoperatively. This limited the duration of the anesthesia to <15 min. In contrast to carotid artery cannulation in the rat (Vissing et al., 1988b), the rats cannulated in the tail artery in the present study maintained body weight postoperatively. Twenty hours after the tail artery cannulation, the rats had gained 6 ± 4 g (mean ± SD) in weight.
Experimental protocol
All experiments were started at 9–10 a.m. The rats were randomized to a resting or a running group. When performing resting experiments, a transparent plexiglass platform was positioned a few millimeters above the moving track of the treadmill, so that the rats could be studied inside the treadmill while the track was running. In this way, differences in LCGU between resting and running rats due to audiovisual and mental influence from the moving treadmill were minimized. The arterial catheter was extended so that injections and blood sampling could be performed without disturbing the rat. A bolus injection of 35 μCi 2DG (Amersham) was given intraarterially and the cannula was subsequently flushed twice with saline. In the running group, the rats ran for 5 min prior to the injection to avoid measurement of tracer uptake into tissues in the transitory period from rest to exercise where cardiovascular and metabolic variables are far from being in steady state. Following the 2DG injection, the rats were either resting or running for 30 min, and arterial blood was sampled for analysis of plasma 2DG and glucose at the times depicted in Fig. 2. The running speed was 28 m/min, which corresponds to ≈85% of maximum oxygen consumption in the Wistar rat (Sonne, 1989). After the 30-min blood sample, the rats were anesthetized with pentobarbital sodium (5 mg/100 g body wt intraarterially) and decapitated. The brains were removed within 3 min and frozen in isopentane, cooled to −60°C by dry ice in acetone.
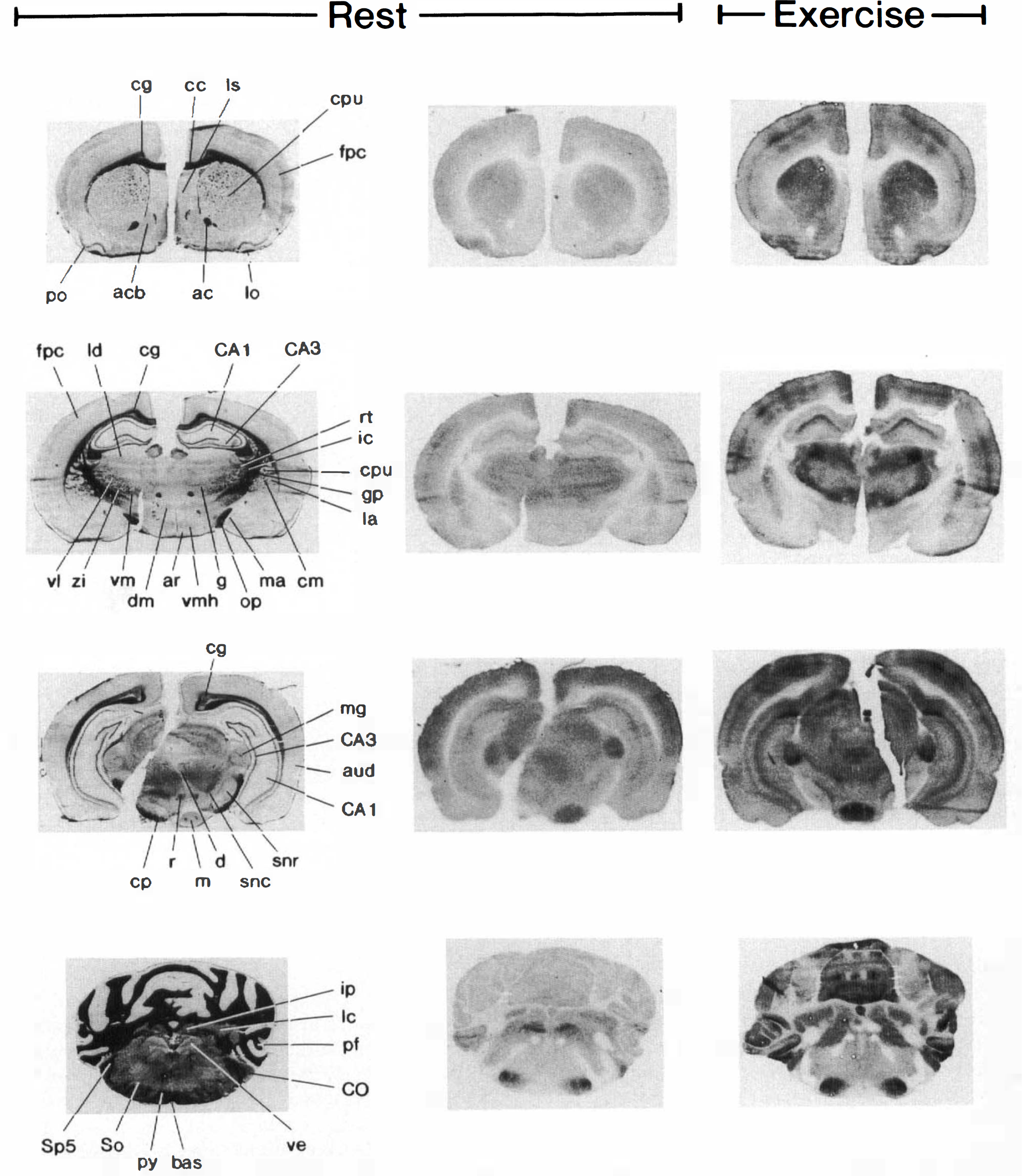
Klüver–Barrera-stained histological sections
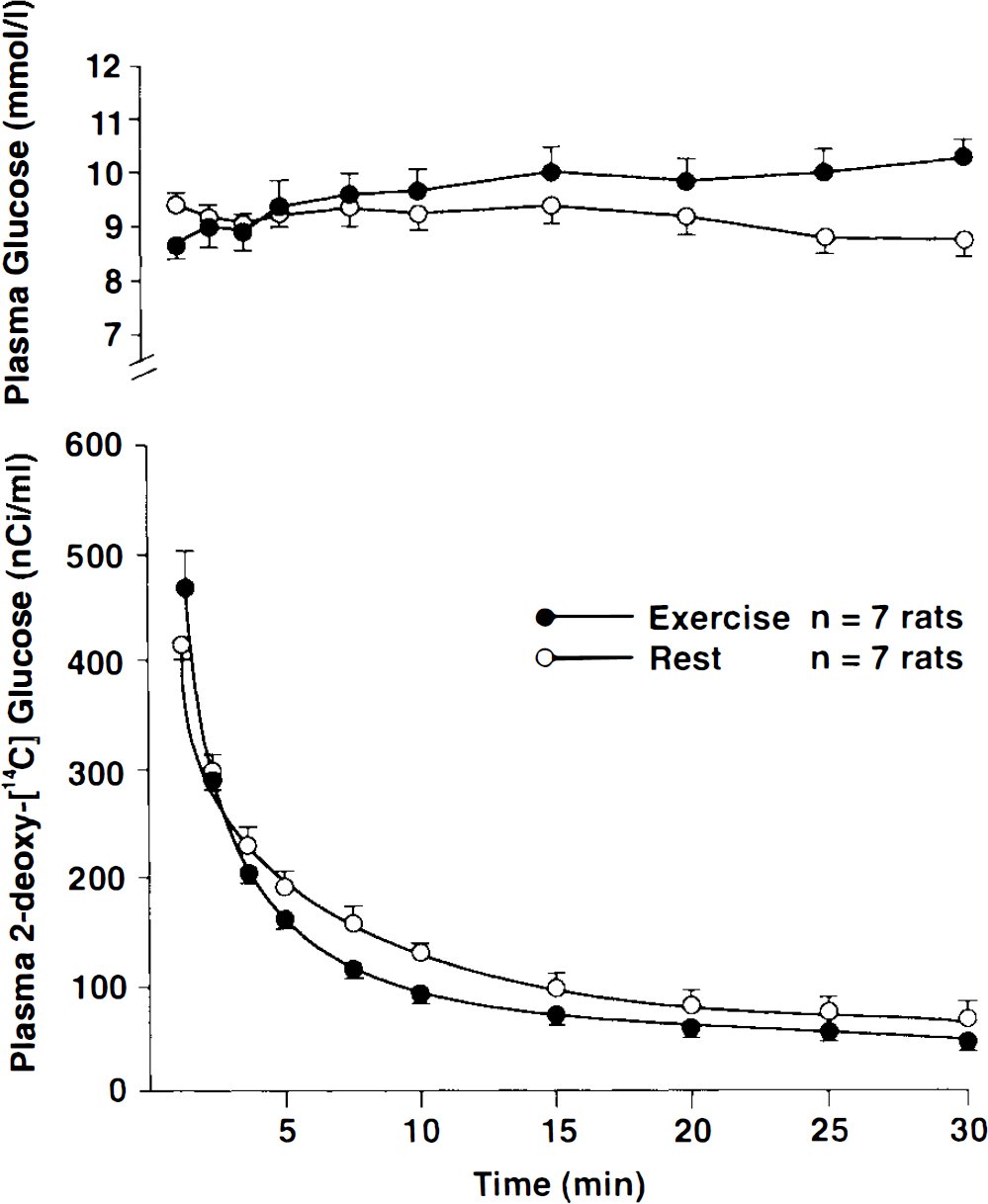
Plasma glucose concentration and 2-deoxyglucose activity in rats at rest and during running at 28 m/min. Values are mean ± SD.
Blood sampling and analytical procedures
The blood samples were immediately transferred to precooled microtubes and centrifuged in a Beckman microfuge. Plasma glucose was determined by the glucose oxidase method (glucose analyzer model 23A; Yellow Springs Instruments). Plasma activity of 2DG was determined by counting plasma samples in a Mark III liquid scintillation system (model 6880; Searle Analytic, Skokie, IL, U.S.A.). Correction for counting efficiency was carried out by comparing known amounts of tracer in plasma with plasma samples from the experiment.
Autoradiography
Serial 20-μm-thick coronal sections of the brain were cut at −22°C in a cryostat. Every tenth section was dried on a glass coverslip placed on a hot plate at 60°C. The dried brain slices were placed on x-ray films along with a set of 14C-calibrated methylmethacrylate standards for 3 weeks. Adjacent brain sections were stained a.m. Klüver—Barrera for positive identification of the corresponding anatomical structures seen in the autoradiographs. The atlas of Paxinos and Watson (1982) was used as reference. The density of the autoradiographs was measured with a Leitz TAS-plus computerized image analyzer and was converted to radioactivity by a third degree polynomial standard curve established for each film (Gjedde and Diemer, 1985). The density was determined in 73 regions and in the whole brain. Each region was measured in both hemispheres. In addition, the density of large regions was measured in all consecutive autoradiographs in which they were present. No regions showed significant differences in LCGU between the two hemispheres. Accordingly, reported values of LCGU are presented as the average values for both hemispheres, and for larger regions present on several autoradiographs, values are averages for all measured sections. When a region was present in more than one section, the area of the region in each section was weighted before an average LCGU was calculated. Total cerebral glucose utilization (TCGU) was measured separately and was calculated from the measured density of the whole-brain autoradiograph. As for LCGU, the contribution of each brain section to TCGU was weighted according to the area of the brain section. Cerebral glucose utilization was calculated as described by Sokoloff et al. (1977), using the same lumped constant and gray and white matter rate constants as published then.
Statistical evaluation
Values are presented as means ± SD. Differences between resting and exercising rats were evaluated by the Mann–Whitney rank sum test for unpaired data; p < 0.05 was considered significant (two-tailed testing).
RESULTS
Cerebral glucose utilization (Table 1; Fig. 1)
Local cerebral glucose utilization (μmol 100 g−1 min−1) in resting and running (28 m/min) rats
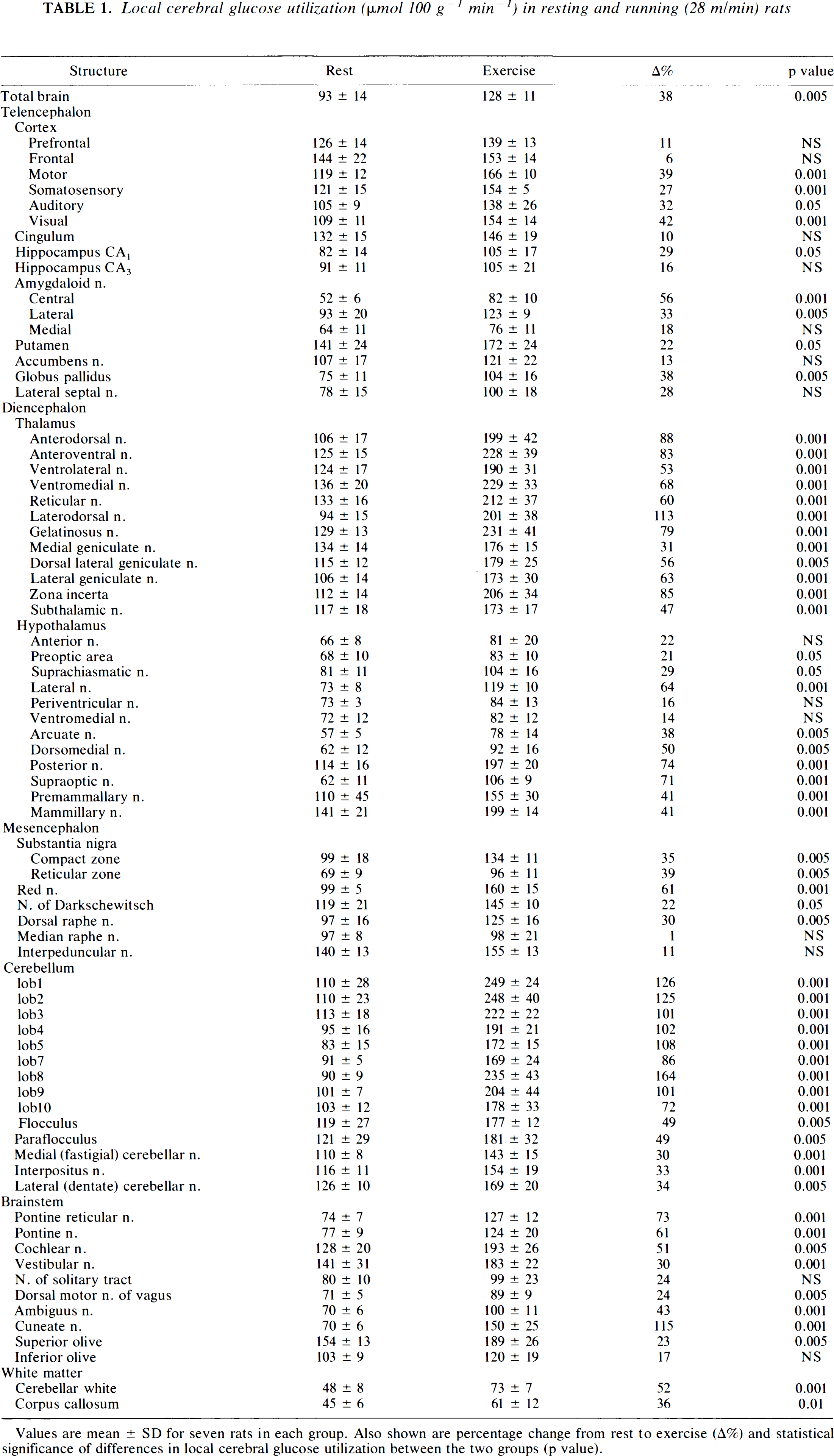
Values are mean ± SD for seven rats in each group. Also shown are percentage change from rest to exercise (A%) and statistical significance of differences in local cerebral glucose utilization between the two groups (p value).
TCGU increased by 38% during running at ≈85% of maximum O2 consumption. Glucose utilization did not decrease during exercise in any of the brain regions studied. Glucose utilization increased in both gray and white matter structures during exercise. However, regional differences in LCGU responses to exercise were pronounced. LCGU increased during exercise in cerebral areas involved in eliciting and coordinating motor function (cerebellum, basal ganglia, substantia nigra, motor cortex, and the following nuclei: subthalamic, posterior hypothalamic, red, ambiguus, and pontine), in areas involved in sensory function (somatosensory, auditory, and visual cortex, the thalamus, and the following nuclei: Darkschewitsch, cochlear, vestibular, superior olive, and cuneate), in white matter of the cerebellum and corpus callosum, and in areas involved in the regulation of autonomic function (dorsal raphe n. and areas in the hypothalamus, amygdala, and hippocampus). The largest increases in LCGU were seen in areas involved in motor control and processing of afferent information (cerebellum, thalamus, cuneate nucleus). Not all areas that can be expected to be functionally related to exercise showed increases in LCGU during exercise (i.e., inferior olive, prefrontal and frontal cortex, ventromedial hypothalamus, median raphe n.).
Plasma glucose concentration and plasma disappearance of 2DG (Fig. 2)
In resting rats, the plasma glucose concentration was stable during the experimental period. In running rats, the plasma glucose concentration also remained stable in the first 10 min of exercise, but increased (p < 0.05) from t = 15 min to the end of exercise compared with levels at rest. Except for t = 30 min, where plasma glucose was higher in running than resting rats (p < 0.05), plasma glucose did not differ between the two groups.
The rate of disappearance of 2DG tended to be higher (p < 0.10) in the exercising group than in the resting group, probably as a result of a higher 2DG uptake in muscles during exercise.
DISCUSSION
The principal new finding in the present study is the increase in TCGU and the marked redistribution of LCGU occurring during running. Marked exercise-induced increases in LCGU were found in cerebral gray matter structures involved in motor, sensory, and autonomic function as well as in white matter structures in the cerebellum and corpus callosum. The findings provide clues to the functionally active areas in the brain that participate in the pronounced motor, sensory, and autonomic adaptation occurring during exercise.
The effect of dynamic exercise on LCGU has not been studied extensively before. LCGU during static and low-intensity dynamic arm exercise has been studied in monkeys, and for both types of exercise, increases in LCGU were observed in sensorimotor cortex in the hemisphere contralateral to the moving forelimb (Schwartzman et al., 1981; Savaki et al., 1993). LCGU has also been studied in mice during two stages of the light-dark cycle in which the mice are “drowsy” and “active,” respectively (Jay et al., 1985). In parallel with findings in the present study, LCGU increased in active versus drowsy mice in the thalamus, amygdaloid n., and sensorimotor and auditory cortex. The differences in LCGU between active and drowsy mice, however, were smaller than those observed between resting and running rats in the present study. Furthermore, LCGU of the cerebellum, visual cortex, and lateral hypothalamic, red, subthalamic, and pontine reticular nuclei, which was significantly elevated by exercise in the present study, did not differ significantly in active versus drowsy mice, although qualitatively the changes in LCGU were the same as those in the present study. Some of these differences in LCGU between the two studies undoubtedly reflect variations in work intensity and the lack of quantitative assessment of work intensity in active and drowsy mice.
Cerebral 2DG uptake has also been studied during high levels of mental stress in swimming rats (Sharp, 1976). Neither exercise intensity nor quantitative LCGU was assessed. However, in corroboration with the present findings, the highest density of 2DG was found in the cerebellum, and the density of 2DG never decreased in any region of the brain during swimming, indicating an increase in TCGU during exercise.
The measured resting values of LCGU in the present study are comparable with those found by others in resting rats (Sokoloff et al., 1977). The small increase in plasma glucose concentration occurring late in the exercise period in running rats has no significant influence on the calculations of LCGU (Schuier et al., 1990).
Apart from the moderate decrease in TCGU that has been demonstrated during deep sleep (Kennedy et al., 1982), impaired consciousness (Sokoloff, 1989), and anesthesia (Sokoloff et al., 1977) and the increased levels of TCGU during epileptic seizures (Duncan, 1992), TCGU is thought to change very little with physiological alterations in cerebral functional activity (Sokoloff, 1991). It is therefore interesting that TCGU increases during dynamic exercise of high intensity, as demonstrated by the present findings. TCGU was calculated with gray matter rate constants. The presented values of TCGU are therefore probably underestimated by a few percent.
In contrast to the increase in TCGU during exercise in the present study, total cerebral blood flow during light, moderate, and heavy dynamic exercise does not increase significantly in dogs when measured by the radioactive microsphere method (Gross et al., 1980; Musch et al., 1987). However, significant local increases in flow were seen in the sensorimotor cortex and cerebellum during moderate exercise but not during light exercise (Gross et al., 1980). In accordance with this, cortical flow, as measured by the 133Xe washout technique, increases during moderate and heavy exercise (Herholz et al., 1987; Thomas et al., 1989), but not during light dynamic exercise (Globus et al., 1983) in humans. The apparent discrepancy between the highly significant increase in TCGU in the present study and the lack of significant increases in total cerebral blood flow during exercise in dogs (Gross et al., 1980; Musch et al., 1987) does not support the theory of a close coupling between cerebral blood flow and metabolism. However, a coupling may have been masked by methodological differences in the measurements of flow and metabolism, species differences, and different duration and intensity of exercise in the studies.
At rest, white matter structures generally had a two to three times lower glucose utilization than gray matter structures. During exercise, increases in gray matter LCGU were accompanied by increases in white matter LCGU. Parallel changes in LCGU of white and gray matter have previously been demonstrated with increases in hypoxia (Torbati et al., 1986) and decreases in starvation (Hasselbalch et al., 1994), anesthesia (Sokoloff et al., 1977), and hypothermia (Palmer et al., 1989).
Glucose utilization in some of the cerebral regions in the present study did not increase during exercise, although it would be expected to do so based on their known afferent and efferent connections. These regions are the inferior olive that projects sensory information to the cerebellum, the prefrontal and frontal cortex involved in modulating motor output from the brain, and the ventromedial hypothalamus and median raphe nucleus involved in autonomic adjustments to exercise. A possible explanation for this could be that large concomitant increases and decreases in neuronal activity may occur within small volumes of the brain without being detected by the 2DG method. In concordance with this proposal, we have found evidence for an increased sympathetic and decreased parasympathetic outflow from the ventromedial hypothalamus during exercise (Vissing et al., 1989), which could explain the lack of overall change in LCGU in this brain region in the present study.
In contrast, LCGU increased during exercise in the visual and the auditory cortex, which are not thought to be functionally related to running. All experimental rats in the present study were very familiar with the treadmill. As a result of this, all resting rats were very calm and fell into a slumber during the experiment. They did not fall asleep, as evidenced by the opening and closing of the eyes throughout the experiment. The noise from the moving treadmill in both rest and exercise experiments significantly exceeded any sound the rats could have produced while running. The higher LCGU in auditory and visual cortex during exercise is therefore presumably not related directly to the exercise per se, but to a higher mental alertness in exercising versus resting rats. It is conceivable that other observed differences in LCGU between resting and exercising rats in the present study may not be attributed exclusively to exercise. Studies in which LCGU during exercise is compared with LCGU in physiological conditions other than rest can help to clarify the specificity of LCGU responses to exercise.
In summary, the present findings demonstrate an increase in TCGU during dynamic exercise of high intensity. The increase in TCGU is associated with large differences in glucose utilization among various brain regions. The findings provide clues to the functionally active areas in the brain participating in the pronounced motor, sensory, and autonomic adaptation occurring in exercise.
Footnotes
Acknowledgment:
The study was financially supported by the Danish Medical Association and the Danish Health Science Research Council. Excellent technical assistance was performed by Marianne Nielsen.
