Abstract
The expression of the large amino acid transporter, LAT1, was investigated in brain of adult Long-Evans rats. The LAT1 transcript was readily detected in brain microvessels and choroid plexus by reverse transcription polymerase chain reaction analysis using three different gene specific primer pairs. A polyclonal affinity purified antibody against the N-terminus of LAT1 was generated in chickens and used in immunoblot and immunocytochemical analyses of brain tissue sections of adult rats. On immunoblots, the antibody detected a peptide-inhibitable 45 kDa band in a rat brain microvessel membrane preparation. It also identified the same protein band in membrane preparations of different brain structures, as well as in heart and testis, whereas the protein was absent or only faintly detectable in muscle, kidney, and liver. In brain sections, the antibody intensely labeled the luminal and abluminal membranes of brain microvessel endothelial cells in all brain areas examined including cerebral cortex, cerebellum, hippocampus, and in gray and white matter regions. These results suggest that LAT1 is involved in transcellular transport and may play an important role in large, neutral amino acid transfer across the blood–brain barrier.
The brain depends upon a supply of various nutrients for maintenance of physiologic function and metabolic homeostasis. Although not a major source of metabolic energy, amino acids are critically important for essential metabolic pathways and for performing specialized functions. Amino acids serve as integral units in the synthesis of all central nervous system (CNS) proteins during brain maturation and steady-state turnover of CNS proteins. Some amino acids (glutamate, aspartate, glycine, and others) serve as chemical messengers for cell–cell communication, and others (tyrosine, glutamine, tryptophan, histidine, and arginine) are precursors of major CNS neurotransmitters (dopamine, norepinephrine, GABA, serotonin, histamine, nitric oxide) (Cooper et al., 1996). At least two amino acids (glutamate, glutamine) are known to play a significant role in eliminating excess ammonium ion from brain and in maintaining osmotic equilibrium between neuronal and glial cells and their environment (Lee et al., 1998). Although neural cells are capable of amino acid biosynthesis, many amino acids, including the essential ones, are delivered to the brain through the circulating plasma.
A hallmark characteristic of the brain vasculature is an endothelium with tight occluding junctions at positions of cell–cell contact resulting in the so-called blood–brain barrier (Brightman and Reese, 1969). For transcellular transport of amino acids across the brain vasculature to occur, transport systems must be located in the luminal and abluminal plasma membranes of the vascular endothelial cells (Pardridge, 1983). In vivo and in vitro approaches have been used to determine the functional characteristics of blood–brain amino acid transport, and several sodium-dependent and sodium-independent systems have been detected. A system with a relatively high transport capacity was identified and exhibited the characteristics of System L, the sodium independent transporter for large, neutral amino acids (Oldendorf, 1971). Recent advances by application of molecular techniques have led to the isolation, cloning, and identification of two closely related System L amino acid transporter mRNA, LAT1 and LAT2 (Kanai et al., 1998; Pineda et al., 1999; Segawa et al., 1999). Although evidence has been presented that the mRNA transcript for LAT1 is present in brain (Kanai et al., 1998), and more precisely at the blood-brain barrier (Boado et al., 1999), the exact cellular location of this transport protein has not yet been reported. In the current study, an affinity-purified antibody, raised in chicken against rat LAT1, was used to localize this transporter in the brain by the use of immunoblotting and immunocytochemistry techniques. The authors report that LAT1 protein is abundantly expressed by rat brain capillary endothelial cells and is located at the luminal and abluminal membranes, thus suggesting that LAT1 is the primary carrier of large, neutral amino acids across the blood–brain barrier. Thus, transcellular transport of amino acids through the brain vascular endothelium by LAT1 is an important process in overall brain metabolism and integral to normal brain function. Demonstration of LAT1 transcript and protein in rat brain endothelium and other brain structures now enables experimental approaches for investigating alterations of this transporter under pathologic conditions.
MATERIALS AND METHODS
All procedures using animals followed a protocol approved by the University of Minnesota Animal Care Committee.
cDNA, reverse transcription, and polymerase chain reaction procedures
Polymerase chain reaction analysis was used to detect the presence of LAT1 transcripts in whole rat brain, choroid plexus, and cerebral microvessels. For whole brain, total RNA was extracted from fresh tissue (Qiagen, Valencia, CA, U.S.A.) and used to prepare cDNA according to the manufacturer's instructions (cDNA Cycle Kit; Invitrogen, San Diego, CA, U.S.A.). Rat brain microvessel and choroid plexus cDNA libraries were obtained by isolating brain microvessels (Gerhart et al., 1988) and choroid plexuses and subsequently extracting poly(A)+ RNA (Invitrogen). The respective libraries were constructed commercially (Invitrogen) according to the modified procedure of Gubler and Hoffman (1983) using the plasmid vector pYES II.
Five nanograms purified cDNA was diluted into a PCR reaction mix consisting of 20 mmol/L Tris-HCl, 50 mmol/L KCl, 2 mmol/L MgCl2, deoxynucleotide triphosphates each at 0.4 mmol/L, 20 pmol of each primer, and 1.5 U Taq polymerase in a total volume of 50 μL. Based on the sequence of rat LAT1 (GenBank accession number AB015432), three pairs of primers were designed to amplify LAT1 cDNA and produce amplicons from near the 5′ end of the coding region, from near the 3′ end of the coding region, and from nearly the full length of the coding region. The LAT1 primer pairs were as follows: forward (5′-ATC GGT TCG GGC ATC TTC GT-3′), reverse (5′-CAG CGA GCC GTA GAC CTC TAG CAT-3′); forward (5′-ATC GGT TCG GGC ATC TTC GT-3′), reverse (5′-AAC TTG ACC CAA ATG AAC GCT ACA-3′); and forward (5′-CCG TGA AAG AAA CCT GGT ACG A-3′), reverse (5′-AAC TTG ACC CAA ATG AAC GCT ACA-3′). The latter primer pair is specific for LAT1 and a truncated form of LAT1 known as TA1 (Sang et al., 1995). Amplification conditions were 30 seconds at 95°C and 60 seconds at 68°C for 35 cycles. Polymerase chain reaction products were subjected to electrophoresis in 2% agarose gel and visualized under ultraviolet light after staining with ethidium bromide.
Antibodies
Peptides consisting of the 15 N-terminal amino acids (MAVAGAKRRAVAAPAC) and the 11 C-terminal amino acids (CQKLMQVVPQET) of rat LAT1 were synthesized with a carboxyl-terminal and an amino-terminal cysteine residue, respectively. Each peptide was conjugated to keyhole limpet hemocyanin and injected (0.5 mg) with Freund's complete adjuvant into the breast muscle of two chickens. Booster injections followed at weekly intervals. One week after the fourth injection, immunoglobulin (Ig) Y was extracted from egg yolks (EGGstract IgY Purification System, Promega, Madison, WI, U.S.A.) and affinity-purified using a 2-mL support column with the LAT1 peptide covalently attached (AminoLink Plus Immobilization Kit; Pierce, Rockfort, IL, U.S.A.).
Immunoblots
After 3% halothane anesthesia and decapitation, tissues from adult Long-Evans rats were quickly removed and homogenized by mechanical disruption (Ultra-Turrax T25; Ikaworks, Cincinnati, OH, U.S.A.) in hypotonic buffer (20 mmol/L Tris-HCl, 1 mmol/L dithiothreitol, pH 7.4) containing a protease inhibitor cocktail (Complete Protease Inhibitor; Roche Diagnostics, Indianapolis, IN, U.S.A.). Homogenates were centrifuged at 200×g for 5 minutes at 4°C to remove debris. The supernatants were centrifuged at 200,000×g for 30 minutes at 4°C. The membrane pellets were resuspended in 62.5 mmol/L Tris HCl, pH 6.8, and protein concentrations were determined (BCA Protein Assay Kit; Pierce). Finally, samples were adjusted to 4 μg/μL and diluted 1:1 with sample buffer (62.5 mmol/L Tris-HCl, pH 6.8, 2% sodium dodecyl sulfate, 25% glycerol, 0.01% bromophenol blue, 5% β-mercaptoethanol).
Solubilized proteins were subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis in 4% to 15% gradient gels (Bio-Rad) and transferred to nitrocellulose membranes (Hybond; Amersham, Arlington Heights, IL, U.S.A.). Membranes were blocked at room temperature for 1 hour in Sea Block (Pierce) on an orbital shaker, and subsequently incubated for 18 hours with N-terminal LAT1 antibody that was diluted 1:15,000 in TBS-T (50 mmol/L Tris, pH 8.0, 0.9% NaCl, 0.1% Tween20). After five washes in TBS-T, the membranes were incubated for 1 hour with rabbit anti-chicken IgY conjugated to horseradish peroxidase (Pierce) diluted 1:10,000 in TBS-T. Detection was accomplished with chemiluminescence (Super Signal, Pierce and Hyperfilm ECL; Amersham). Negative control blots consisted of membranes probed with antibody that had been preabsorbed with 0.1 μg/mL, 1 μg/mL, or 10 μg/mL of LAT1 peptide.
Immunocytochemistry
Four Long-Evans rats were anesthetized with 5% halothane and perfused transcardially with formal-acetic fixative (4% formaldehyde, 2% acetic acid) for 12 minutes. Brains were removed and postfixed overnight in fixative before embedding in paraffin. Coronal paraffin sections (7 μm) were cut at the level of the anterior hippocampus and at approximately 11 mm from bregma through the cerebellum and the medulla. Slices were mounted on Biobond-treated microscopic slides (Goldmark Biologicals, Philipsburg, NJ, U.S.A.). After deparaffinizing in xylene and alcohols, tissue sections were blocked with PBS containing 0.1% bovine serum albumin (BSA) and 1.5% normal goat serum. Both primary antibodies (N-terminal and C-terminal) were diluted 1:2,000 in PBS containing 0.1% BSA and applied to the sections for 1 hour at room temperature. For negative controls, the appropriate LAT1 peptide (20 μg/mL) was added to the antibody solution before application. Sections then were incubated 30 minutes with biotinylated goat anti-chicken IgG (5 μg/mL in blocking solution) and 30 minutes with avidin-biotin-peroxidase complex (ABC) reagent (both reagents from Vector Laboratories, Burlingame, CA, U.S.A.). Color development was from 1 to 6 minutes in 0.6 mg/mL 3,3′-diaminobenzidine (Dako, Carpinteria, CA, U.S.A.).
RESULTS
Transcript analysis by polymerase chain reaction
Transcript for the amino acid transporter LAT1 was readily detected in cDNA libraries constructed from rat brain microvessels and choroid plexus and in whole rat brain RNA extracts (Fig. 1A). A long (1691 bp) and two short (182 bp, 192 bp) amplicons were produced by PCR using LAT1 specific primers. The sizes of the products corresponded to the predicted sizes based on the primer pairs. The identity of each was verified by isolation of the products from the gels and sequencing. Because the primers for the 182 bp product are also capable of hybridizing to cDNA from TA1, a truncated form of LAT1 that was previously reported (Sang et al., 1995), this product may have been amplified from either LAT1 or TA1.
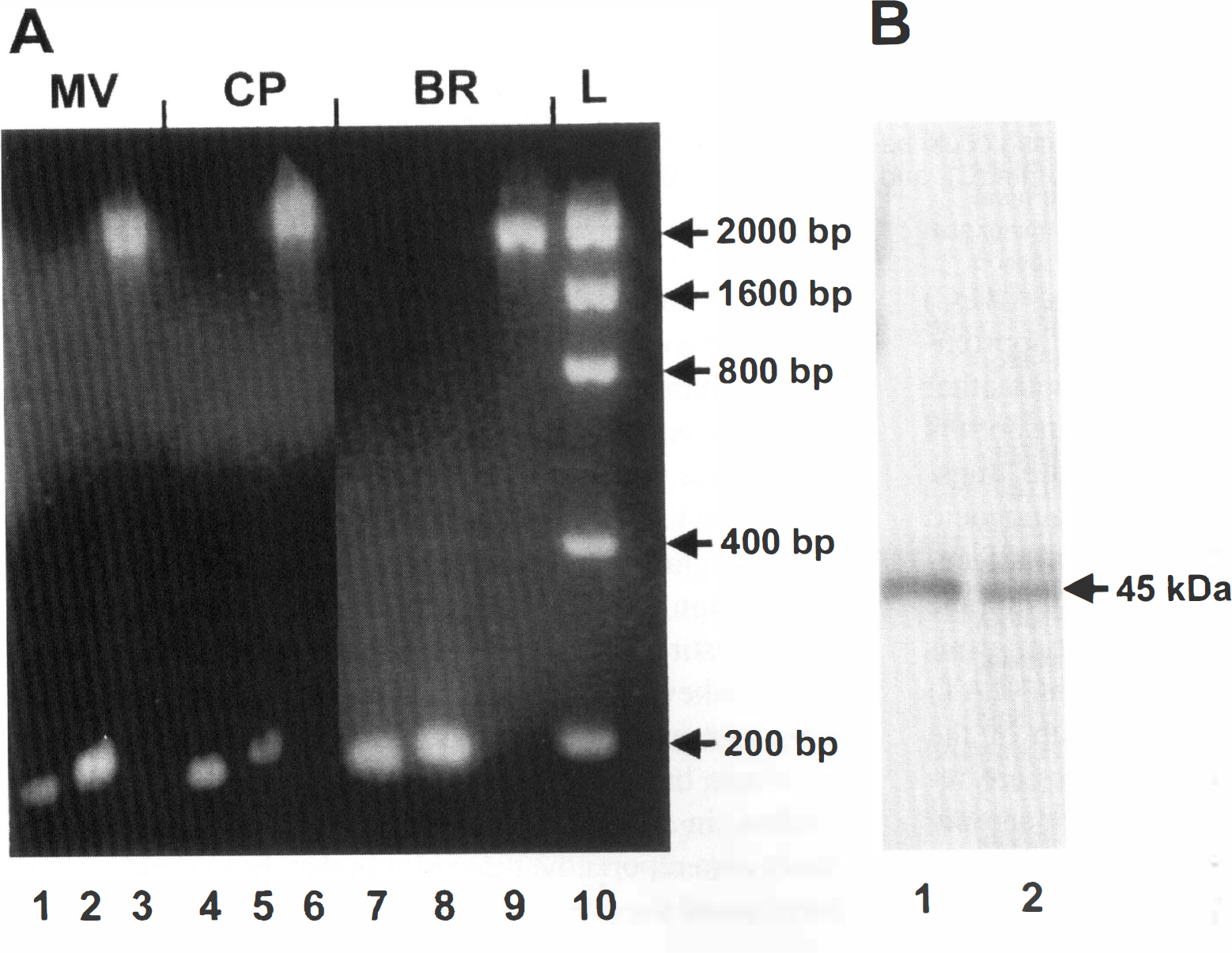
Polymerase chain reaction and immunoblot analysis for LAT1. (A) cDNA from whole rat brain (BR) and from rat brain microvessel (MV) and choroid plexus (CP) cDNA libraries were used as templates. LAT1 specific products were predicted and detected at 182 bp (lanes 1, 4, and 7), 192 bp (lanes 2, 5, and 8), and 1691 bp (lanes 3, 6, and 9). The standard molecular weight DNA ladder is also shown (lane 10). Note that the truncated version of LAT1, called TA1, also encodes the 182 bp template and may produce this product. (B) Immunoblot of a rat brain cortex (lane 1) and cerebellum (lane 2) membrane preparation detecting the N-terminal end of LAT1 at the expected size of approximately 45 kDa.
Immunoblot analysis of LAT1
The N-terminal LAT1 antibody recognized a protein migrating with an apparent molecular mass of ∼45 kDa on immunoblots of brain membranes from cerebral cortex and cerebellum of adult rats (Fig. 1B). This protein was not observed when the antibody was preabsorbed with any of the tested concentrations of LAT1 peptide. A similar protein was observed in immunoblots of membrane preparations from all CNS regions investigated—that is, the frontoparietal cortex, hippocampus, hypothalamus, cerebellum, brainstem, and spinal cord. LAT1 was also detected in heart and testis, but was absent or only faintly detected in kidney, liver, and muscle (data not shown).
The antibody raised against the C-terminal end of LAT1 was expected to detect the C-terminal of LAT1 and the C-terminal of TA1, the approximately 28 kDa truncated form of LAT1 (Sang et al., 1995). However, the C-terminal peptide was an unsatisfactory antigen, and this antibody was not further evaluated.
Immunocytochemistry
Using the N-terminal LAT1 antibody, the most intense LAT1 staining in sections of cerebral cortex was found in brain capillary endothelium where it appeared to stain all microvessels (Fig. 2). The staining was relatively uniformly distributed within the capillaries and examination under high magnification suggested that LAT1 was located in the luminal and abluminal membranes of capillary endothelium (Figs. 2B and 2C). Capillaries were similarly stained in sections of rat cerebellum, including those in the molecular layer, granular layer, and in the cerebellar white matter (Fig. 2D). Microvessels in hippocampus and other brain regions were also stained. LAT1 was also detected in the microvascular endothelium of rat pup brain (data not shown).
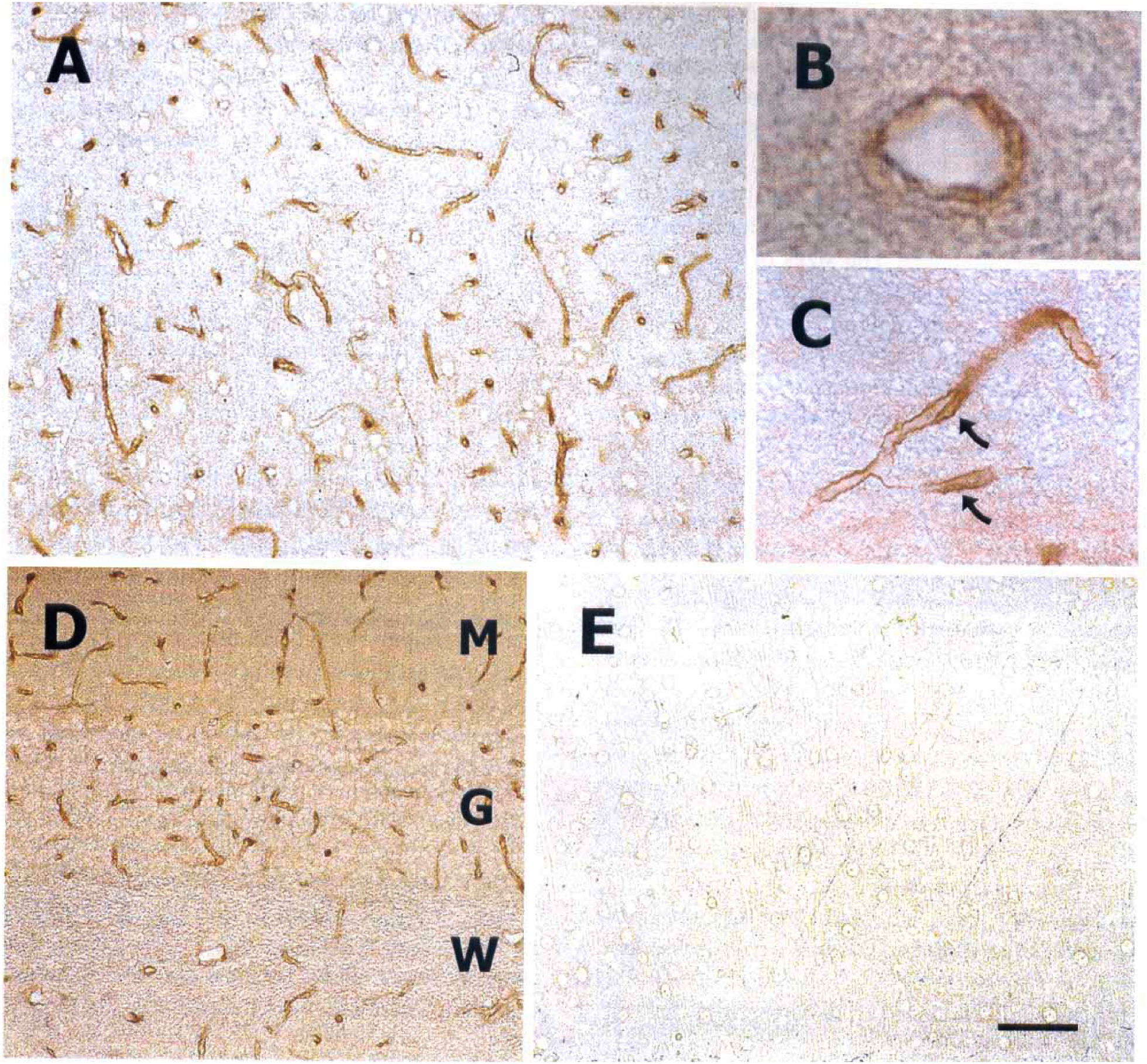
LAT1 immunocytochemistry.
In contrast to the vascular endothelium, immunostaining of LAT1 in the neuropil was weak (Fig. 2A). However, the staining in the neuropil disappeared when the antibody was preabsorbed with peptide.
DISCUSSION
To transport amino acids across their plasma membranes, cells are equipped with integral membrane proteins that function to recognize, bind, and translocate specific amino acids from one side of the membrane to the other. These amino acid transporters belong to a large superfamily of amino acid/polyamine/choline (APC) transporters (Verrey et al., 1999), of which approximately 300 members have been sequenced. As with other transport proteins (for example, the glucose transporters (Mueckler et al., 1985) or the monocarboxylate transporters (Halestrap and Price, 1999)), the amino acid transporters that have been characterized at the molecular level exhibit a topology consisting of 12 membrane spanning segments with both ends of the protein terminating in the cytoplasm. The large amino acid (LAT) transporter family was identified as a subgroup of the APC transporter superfamily. These transporters are responsible for the exchange of neutral, cationic, or anionic amino acids. One member of the System L family, LAT1, was the subject of the present investigation.
These studies demonstrate the development and application of tools for detection of the LAT1 transcript and protein in the rat. Thus, future studies using in situ, in vitro, and in vivo assays to investigate alterations in LAT1 expression in this experimental animal during development and under pathologic conditions will be possible.
LAT1 mRNA was found in brain microvessels, a preparation enriched in cells of the vascular endothelium and constituting the blood–brain barrier. Boado et al. (1999) likewise found an approximately 100-fold greater transcript expression in bovine brain microvessels than in whole brain extracts. In addition to detecting LAT1 mRNA in microvessels and whole brain, the current study also reports the transcript in the choroid plexus, the location of the blood–cerebral spinal fluid barrier.
The current study reports that not only is the mRNA for the amino acid transporter LAT1 present in brain, but also that the LAT1 protein is abundantly expressed in the luminal and abluminal membranes of capillary endothelial cells of all brain regions examined. Transport of blood-borne amino acids into brain is important for maintaining metabolic homeostasis and normal functioning of the brain. Blood–brain transport of amino acids was first characterized by Oldendorf using the brain uptake index method (Oldendorf, 1971). Subsequent studies reported that this transport was saturable, stereospecific, sodium-independent and that it exhibited specificity for large, neutral amino acids such as phenylalanine, tyrosine, tryptophan, leucine, isoleucine, and valine (Oldendorf and Szabo, 1976; Smith, 1991; Smith et al., 1987). Although these in vivo studies of rat brain concluded that System L transporters were located on the luminal side of the endothelial cell, in vitro studies using freshly isolated microvessels also demonstrated System L on the abluminal side (Betz and Goldstein, 1978; Audus and Borchardt, 1986; Choi and Pardridge, 1986; del Pino et al., 1995). These properties and observations are consistent with the characteristics of LAT1 and the immunocytochemical localization reported here.
The pattern of membrane localization for LAT1 in luminal and abluminal endothelial membranes is similar to that for glucose and monocarboxylic acid transport systems that function in transcellular transport of their respective brain nutrients (Gerhart et al., 1989; Pardridge et al., 1990; Lee et al., 1997; Gerhart et al., 1998). Transport studies in other species, including humans and dogs (Betz et al., 1975; Oldendorf, 1977), suggest that the presence of System L transporters on luminal and abluminal membranes of the brain microvasculature is likely to be broadly conserved among species. More recent and detailed studies of brain endothelium (Smith and Stoll, 1998) have demonstrated the presence of at least seven additional amino acid transport systems, each with distinct specificities, including some that require sodium for cotransport and some with asymmetric distribution in the endothelial cell membranes.
The functional properties of the LAT1 protein have been described by Kanai et al. (1998). The transport of amino acids by LAT1 is sodium-independent, and the preferential substrates are neutral amino acids with branched or aromatic side chains. To be functional, LAT1 requires association with the membrane glycoprotein known as 4F2hc (Broer et al., 1997; Kanai et al., 1998). 4F2hc forms a heterodimer complex with LAT1 and is involved in the control of intracellular trafficking of the complex, although the nature of the interaction remains unclear. Other homologues of LAT1 may also form heterodimers with the membrane gloycoprotein 4F2hc. Mastroberardino et al. (1998) hypothesized that the heavy chain of 4F2hc is linked by an extracellular disulfide bond to LAT1 and that its single transmembrane segment is located near the second and third transmembrane domains of LAT. Previous reports indicate that LAT1 activity is regulated by hormones or bioactive molecules (Smith, 1991) such as arginine-vasopressin and β-adrenergic agents (Brust, 1986; Eriksson and Carlsson, 1988). Whether associated proteins such as 4F2hc are involved in this modulation of transport activity remains to be determined.
Little is currently known about the regulation of LAT1 expression. Based on the current results and other studies, it seems likely that brain endothelial cells express high levels of LAT1 for transcellular transport of amino acids rather than for supplying amino acids for their own consumption. Therefore, the high constitutive expression of LAT1 by endothelial cells may be influenced by factors in the environment of these cells. For example, induction of LAT1 expression may result from the influence of the surrounding astrocytes much like has been suggested for other proteins characteristic of the brain endothelial cell. In addition, pathologic conditions such as hepatic encephalopathy or the amino acidurias may involve altered expression of blood–brain amino acid transporters such as LAT1 (Pardridge, 1998). With the newly available probes for LAT1 mRNA and protein, molecular analysis of these mechanisms will be possible in future studies.
