Abstract
The selective cyclooxygenase-2 (COX-2) inhibitor has been reported to have antiinflammatory, neuroprotective, and antioxidant effects in ischemia models. In this study, the authors examined whether a selective COX-2 inhibitor (celecoxib) reduces cerebral inflammation and edema after intracerebral hemorrhage (ICH), and whether functional recovery is sustained with longer treatment. ICH was induced using collagenase in adult rats. Celecoxib (10 or 20 mg/kg) was administered intraperitoneally 20 minutes, 6 hours, and 24 hours after ICH and then daily thereafter. Seventy-two hours after ICH induction, the rats were killed for histologic assessment and measurement of brain edema and prostaglandin E2. Behavioral tests were performed before and 1, 7, 14, 21, and 28 days after ICH. The brain water content of celecoxib-treated rats decreased both in lesioned and nonlesioned hemispheres in a dose-dependent manner. Compared with the ICH-only group, the number of TUNEL-positive, myeloperoxidase-positive, or OX42-positive cells was decreased in the periphery of hematoma and brain prostaglandin E2 level was reduced in the celecoxib-treated group. Celecoxib-treated rats recovered better by the behavioral tests at 7 days after ICH throughout the 28-day period, and the earlier the drug was administered, the better the functional recovery. Evidence of similar effects in an autologous blood–injected model showed that direct collagenase toxicity was not the major cause of inflammation or cell death. These data suggest that celecoxib treatment after ICH reduces prostaglandin E2 production, brain edema, inflammation, and perihematomal cell death in the perihematomal zone and induces better functional recovery.
Intracerebral hemorrhage (ICH) represents at least 15% of all strokes in Western populations and a considerably higher proportion (20–30%) in Asian and black populations (Qureshi et al., 2001). The prognosis of patients after ICH is poor, often much worse than that of patients with ischemic strokes of similar size (Jorgensen et al., 1995). Some patients with ischemic stroke (up to 30%) undergo hemorrhagic transformation (Lyden and Zivin, 1993; Sacco and Mayer, 1994) and a considerable number of ischemic infarct patients develop hemorrhagic conversion or symptomatic hematoma after thrombolysis (The NINDS t-PA Stroke Study Group, 1997). Currently, there is no available medical therapy for patients with ICH, and treatment is limited to supportive care or invasive neurosurgical evacuation of hematoma in selective patients (Qureshi et al., 2001).
Inflammation is observed in ICH and contributes to brain injury in several ways (Gong et al., 2000; Xi et al., 2001). Treatment of inflammation with immunosuppressive agent (FK-506) reduces edema formation and tissue injury and improves outcome in an ICH model (Peeling et al., 2001), and an antiinflammatory strategy using adenosine A2A receptor activation decreases cell death after ICH (Mayne et al., 2001).
The cyclooxygenases (COXs) are enzymes that metabolize arachidonic acid to prostaglandins and thromboxanes (FitzGerald and Patrono, 2001; Graham and Hickey, 2003). Three isoforms of COX have been identified: COX-1, COX-2, and the COX-1 splice variant, COX-3 (Vane et al., 1998). COX-1 is constitutively expressed in many organs and is thought to synthesize prostanoids involved in normal cellular activities, such as gastric secretion, endothelial function, and platelet aggregation. COX-2, although constitutively active in some organs such as brain and kidney, is highly inducible in response to a wide variety of stimuli, including inflammatory mediators and growth factors (Brian et al., 1998; Drachman et al., 2002; FitzGerald and Patrono, 2001; Graham and Hickey, 2003; Gong et al., 2000; Iadecola, 2003). COX-2 is present in the central nervous system in neurons and astrocytes as well as macrophages and microglia (Graham and Hickey, 2003). COX-2 is the predominant isoform within the neuron itself, has a special role in regulating synaptic activity, and contributes to neuronal death in a variety of disease states (Graham and Hickey, 2003; Iadecola, 2003). Administration of NS-398 or SC58236, relatively selective COX-2 inhibitors, or genetic deletion of COX-2 reduces the ischemic cell death in models of focal or global cerebral ischemia (Araki et al., 2001; Iadecola et al., 2001; Nagayama et al., 1999; Nogawa et al., 1997a,b). Chronic overexpression of COX-2 increases ischemic brain damage (Dore et al., 2003). In ICH models, COX-2 increases maximally after 1 or 3 days and mediates inflammatory changes; however, previous experiments using NS-398 in the autologous blood–injected ICH model could not reduce brain edema formation after 24 hours (Gong et al., 2001).
In the present study, we examined the hypothesis that COX-2 inhibition by celecoxib treatment may reduce brain edema and the associated inflammation and perihematomal cell death after ICH while improving functional outcome.
MATERIALS AND METHODS
Induction of intracerebral hemorrhage with collagenase infusion
All procedures were performed following an institutionally approved protocol in accordance with NIH Guide for the Care and Use of Laboratory Animals. Male Sprague-Dawley rats (Genomics, Seoul, R.O.K.) weighing 200 to 220 g were used in these experiments. Experimental groups consisted of rats subjected to ICH (n = 150) and normal control rats (n = 30). Experimental ICH was induced by the stereotaxic intrastriatal administration of bacterial collagenase type IV (Sigma, St. Louis, MO, U.S.A.), as described elsewhere (Jeong et al., 2003; Rosenberg et al., 1990; Song et al., 2003). In brief, after intraperitoneal injections of 1% ketamine (30 mg/kg; Sigma) and xylene hydrochloride (4 mg/kg; Sigma), rats were placed in a stereotaxic frame (David Kopf instruments, Tujunga, CA, U.S.A.). A burr hole was made and a 30-gauge Hamilton syringe needle was inserted through the burr hole into the striatum (location: 3.0 mm left lateral to the midline, 0.2 mm posterior to bregma, 6 mm below the skull) (Paxinos and Watson, 1982). ICH was induced by the administration of 1 μL containing 0.23 collagen digestion units of collagenase type IV (Sigma) over a 5-minute period. After collagenase infusion, craniotomies were sealed with bone wax, wounds were sutured, and rats were allowed to recover. Physiologic parameters, including mean arterial blood pressure, blood gases, and glucose concentration, were measured during the experiment. During the recovery period, rats were assessed for forelimb flexion and contralateral circling to confirm the effectiveness of the procedures. No cases of seizure were observed during the experiments or at any time after the procedure. Rectal temperature was maintained at 37°C ± 0.5°C using a thermistor-controlled heating blanket. Free access to food and water was allowed after recovery from anesthesia. Rats were kept in air-ventilated cages at 24°C ± 0.5°C for the duration of the experiment.
Experimental group and celecoxib treatment
Rats were grouped as follows: collagenase-induced ICH only (n = 51), celecoxib-treated ICH (n = 89), normal control (n = 18), and celecoxib-treated normal (n = 12). Celecoxib (Pfizer Korea, Seoul, R.O.K.) was administered intraperitoneally, dissolved in 10% dimethyl sulfoxide (DMSO) and saline, at a dosage of 10 mg/kg (n = 24) or 20 mg/kg (n = 65) daily for 3 days or 14 days (to the rats assigned for the behavioral testing; n = 9 respectively). Initial celecoxib treatment was started 20 minutes after ICH induction for the histologic and biochemical studies, and at 20 minutes, 6 hours, and 24 hours for the time window study. ICH rats injected with DMSO alone comprised the ICH-only group (n = 51), and normal rats with DMSO alone (n = 18) or 20 mg/kg celecoxib (n = 12) comprised the normal control or celecoxib-treated normal groups, respectively.
Morphometric measurement of hemorrhage volume
Rats were assigned to one of two experimental groups: ICH only (n = 12) and celecoxib-treated (20 mg/kg) ICH group (n = 12). 72 hours after the operation, rats were anesthetized using the previously described methods and killed by decapitation. The brain was removed immediately and fixed with 4% phosphate-buffered paraformaldehyde. Fixed brains were cut coronally through the needle entry site (identifiable on the brain surface), and then serial slices (1-mm thick) both anterior and posterior to the needle entry site were obtained. Digital photographs of the serial slices were taken and the hemorrhage size was measured using an image analyzer program (Image ProPlus, Silver Spring, MD, U.S.A.). Total hematoma volume was calculated by summing the clot area in each section and multiplying by the distance between sections (Song et al., 2003).
Measuring brain water contents
Rats were assigned to one of four experimental groups: ICH only (n = 12), celecoxib-treated (10 mg/kg) ICH (n = 12), celecoxib-treated (20 mg/kg) ICH (n = 12), normal control (n = 12), and celecoxib-treated (20 mg/kg) normal (n = 12). Three days after operation, rats were anesthetized and killed by decapitation. The brains were immediately removed, divided into two hemispheres along the midline, and the cerebellum was removed. The brain samples were immediately weighed on an electronic analytical balance to obtain the wet weight. The samples were then dried in a gravity oven at 100°C for 24 hours to obtain the dry weight. Water content was expressed as a percentage of wet weight as follows: (wet weight − dry weight) / (wet weight) × 100 (%) (Song et al., 2003).
Histologic analysis
Rats (ICH-only group, n = 12; celecoxib-treated (20 mg/kg) ICH group, n = 12; celecoxib-treated (10 mg/kg) ICH group, n = 6) were perfused by cardiac puncture with 100 mL cold saline and 100 mL of 4% paraformaldehyde in 0.1-mol/L phosphate-buffered saline. Brains were removed and cryopreserved for cryostat sectioning at 30-μm thicknesses. Immunostains were processed as described previously (Jeong et al., 2003; Song et al., 2003). We performed immunostaining with antibodies to myeloperoxidase (MPO; dilution, 1:200, Dako, Carpenteria, CA, U.S.A.), and OX42 (dilution, 1:500, Chemicon, Temecula, CA, U.S.A.) followed by fluorescent microscopy (Nikon, MD, U.S.A.) for evaluation of neutrophils and microglias, respectively. The axial sections through the center (three sections per animal; 1 mm width) of the hemorrhagic lesion were analyzed. Only morphologically intact myeloperoxidase-positive or OX42-positive cells were included in these counts (Zhang et al., 2001). We counted marker-specific cells in the whole section (three sections per each antibody staining). The quantitative analysis was performed by two independent investigators who were blinded to the rat treatment status (Gong et al., 2000). Total counts in these sections were converted into cell densities for comparison between the groups.
TUNEL assay
Rats used for terminal deoxynucleotidyl transferase-mediated 2′-deoxyuridine 5′-triphosphate-biotin neck end labeling (TUNEL) staining were killed with an overdose of sodium pentobarbital at 72 hours after collagenase injection. The TUNEL procedure for in situ detection of DNA fragmentation was performed as described elsewhere (Gavrieli et al., 1992; Matsushita et al., 2000; Song et al., 2003). Cell density counts were performed on sections counterstained with toluidine blue. Three axial sections (1 mm width) through the center of the hemorrhagic lesion were analyzed by two independent investigators who were masked to the rat treatment status. Total counts in the measured sections were converted into cell densities for quantitation and comparison between the groups.
Prostaglandin E2 levels
To evaluate the effectiveness of COX-2 inhibition, we measured prostaglandin E2 (PGE2) levels in the brains of rats treated for 3 days. Groups included six ICH-only rats, six celecoxib-treated (20 mg/kg) ICH rats, six celecoxib-tread (10 mg/kg) ICH rats, and six normal control rats. We rapidly removed and froze their brains and used them for determination of PGE2. We measured the content of PGE2 spectrophotometrically in duplicate samples with an immunoassay kit (Cayman Chemical, Ann Arbor, MI, U.S.A.), according to the manufacturer's instructions, as described elsewhere (Araki et al., 2001; Drachman et al., 2002; Salvemini et al., 1995).
Behavioral testing
Behavioral testing (n = 9, respectively) was performed weekly using the rotarod and modified limb placing tests (MLPT), which were monitored by two individuals masked to rat treatment status. For the time window study, the celecoxib-treated group was divided to three subgroups according to drug commencing time (20 minutes, 6 hours, and 24 hours after ICH induction; n = 9, respectively). In the rotarod test (Jeong et al., 2003), the rats were placed on the accelerating rotarod cylinder, and the time the animals remained on the rotarod was measured. The animals were trained for 3 days before the stereotaxic operation. The maximum duration (in seconds) on the device was recorded with three rotarod measurements 1 day before ICH induction. Motor test data are presented as percentages of the maximal duration compared with the baseline control (before ICH). The MLPT is the a modified version of a test previously described in the literature (Jeong et al., 2003; Puurunen et al., 2001; Song et al., 2003). The test consists of two limb-placing tasks that assess the sensorimotor integration of the forelimb and the hindlimb by checking responses to tactile and proprioceptive stimulation. First, the rat is suspended 10 cm over a table and the stretch of the forelimbs towards the table is observed and evaluated: normal stretch, 0 points; abnormal flexion, 1 point. Next, the rat is positioned along the edge of the table, with its forelimbs suspended over the edge and allowed to move freely. Each forelimb (forelimb-second task, hindlimb-third task) is gently pulled down and retrieval and placement is checked. Finally, the rat is placed towards the table edge to check for lateral placement of the forelimb. These latter three tasks are scored in the following manner: normal performance, 0 points; performance with a delay (2 seconds) and/or incomplete, 1 point; and no performance, 2 points. A score of 7 points indicates maximal neurologic deficit and 0 points indicates a normal performance. The body weight of all animals was checked weekly for 4 weeks.
Autologous blood–injected ICH model
Additional experiments were also performed using direct infusion of autologous blood into the rat striatum as an alternative model (Matsushita et al., 2000; Wagner et al., 1996). A small craniotomy was performed using the same stereotactic coordinates, and 300 μL of autologous unheparinized blood (from the femoral artery) was infused into the left striatum. We analyzed the brain water content (n = 6 respectively) and histologic change (n = 6 respectively) in the ICH-only and celecoxib-treated (20 mg/kg) ICH groups that were initially treated 20 minutes after ICH induction, and verified whether the effects of celecoxib would be comparable with those in the collagenase-induced ICH models.
In vitro assay of collagenase-induced PGE2 production
To further clarify that the inflammatory responses obtained from the collagenase-induced ICH models were not due to the collagenase itself, we performed studies to examine the PGE2 production by collagenase using in vitro culture models, such as A1G11 human cortical neuron (Nagai et al., 2002), U393 human astrocyte (Hong et al., 2003), and PC12 rat pheochromocytoma (Kim et al., 2004) cell lines. The cells were suspended into Dulbecco's modified Eagle's medium supplemented with 10% fetal bovine serum and plated on 12-well plates coated with poly-L-lysine at a density of 2 × 106 cells per plate. Growth medium was changed every 2 or 3 days. Experiments were performed at 7 days in culture. As previously described (Matsushita et al., 2000), we investigated a range of collagenase concentrations in vitro from 5 to 50 U/mL. All cultures were exposed for 24 hours. PGE2, concentration was measured triplicate and compared with control cultures not exposed to collagenase.
Statistical analysis
All data in this study are presented as mean ± SD. Data were analyzed by repeated measures analysis of variance (ANOVA), and unpaired Student's t-test if they were normally distributed (Kolmogorov-Smirov test, P > 0.05). Otherwise, we used the Mann-Whitney test and specified the test used. Two-tailed probability value of <0.05 was considered significant.
RESULTS
Physiologic parameters
All animals survived the experiments. The physiologic parameters, including MABP, blood gases, serum glucose, and body temperatures, were not significantly different in any experimental group before, during, or 30 minutes after ICH (Table 1, ANOVA).
Physiologic parameters before and 30 minutes after ICH induction
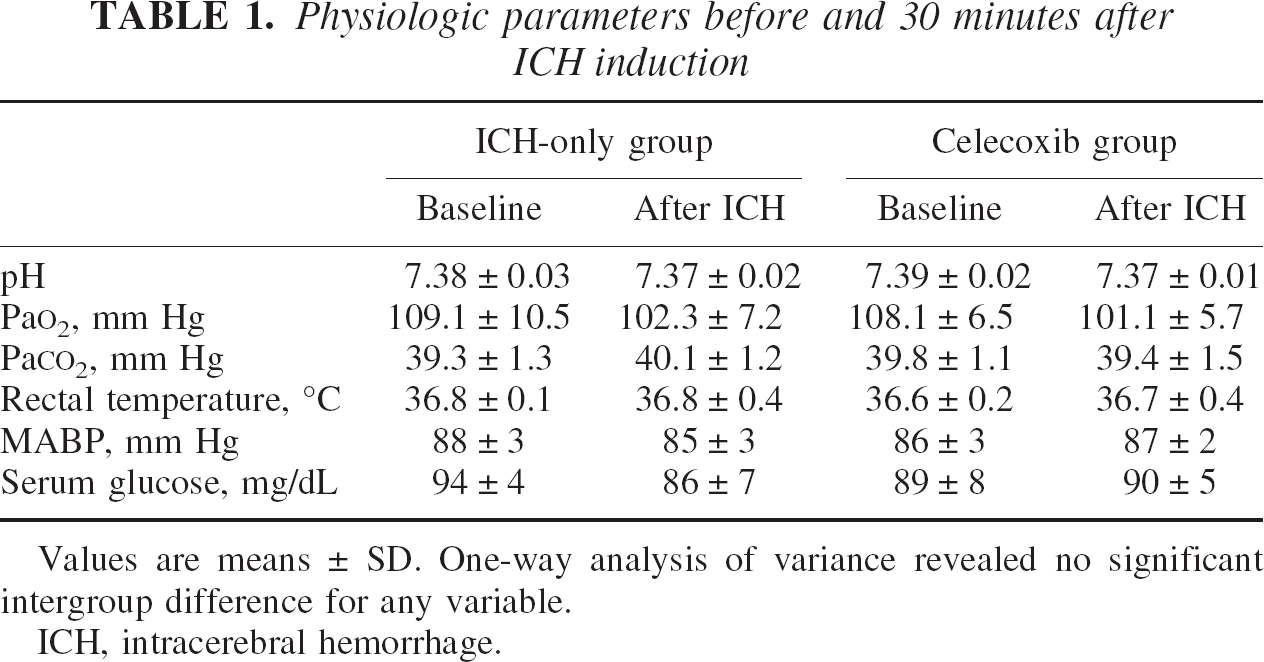
Values are means ± SD. One-way analysis of variance revealed no significant intergroup difference for any variable.
ICH, intracerebral hemorrhage.
Hemorrhage size was not significantly different between groups
Rats infused with 0.23 CDU of collagenase developed hemorrhages in the striatum that were evident by 24 hours (Fig. 1A). The hemorrhages were 19.09 ± 4.37 mm3 (n = 12; ICH-only group) and 18.97 ± 5.67 mm3 (n = 12; celecoxib-treated [20 mg/kg] group at 20 minutes after ICH), respectively, by 72 hours. There was no significant difference between groups (P = 0.95, Student's t-test). Minor intraventricular hemorrhage (IVH) occurred in a few rats in each group (n = 5/12, ICH-only group; 4/12, celecoxib-treated ICH group), but did not significantly affect the behavioral test results measured at 3 days after ICH induction (MLPT score: 6.7 ± 0.3 in the ICH plus IVH group, n = 9; 6.5 ± 0.4 in the ICH-only group, n = 9; P = 0.97, Mann-Whitney test).
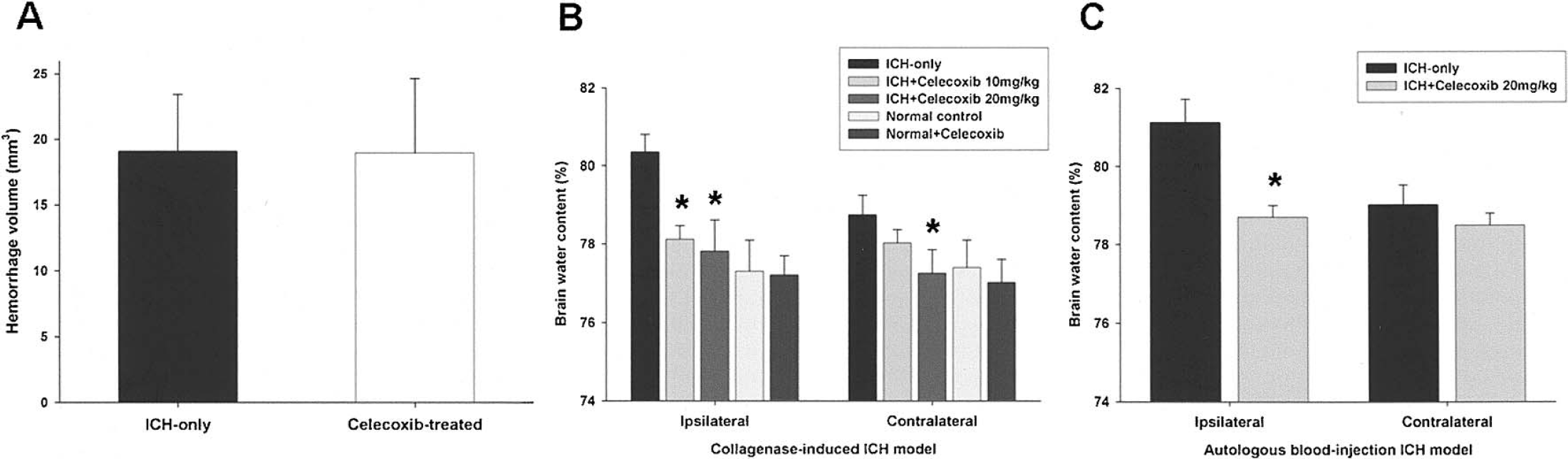
Hematoma volume and brain water content 72 hours after intracerebral infusion of collagenase. (
Brain water content is decreased in the bilateral hemispheres by celecoxib treatment
Brain water content of the lesioned (left) hemisphere was 80.35% ± 0.45% in the ICH-only group (n = 12) and 77.81% ± 0.82% in the celecoxib-treated (20 mg/kg) ICH group, and corresponding values in the nonlesioned (right) hemisphere were 78.74% ± 0.51% and 77.25% ± 0.59%, respectively. Brain water content of the lesioned and nonlesioned hemispheres was dose-dependently reduced in the celecoxib-treated ICH group (n = 12; P < 0.05, unpaired t-test; Fig. 1B). Brain water content of both hemispheres in the normal control (n = 12) and celecoxib-treated normal (n = 12) groups was not significantly different from that of the celecoxib-treated ICH group (Fig. 1B). Similar patterns of brain water content were observed in an alternative model of ICH in which 300 μL of autologous blood was infused into the rat striatum (n = 6; Fig. 1C).
Inflammatory cell density and TUNEL-positive cells after ICH are decreased by celecoxib treatment
Three days after ICH induction, the myeloperoxidase-positive or OX42-positive cells were clustered but generally in a ringlike distribution in the striatal tissue around the hematoma (Figs. 2A–2D). Quantitative analysis showed significant, dose-dependent reduction of myeloperoxidase-positive or OX42-positive cell infiltration in celecoxib-treated ICH groups (Fig. 2G; n = 12; P < 0.01, unpaired t-test).
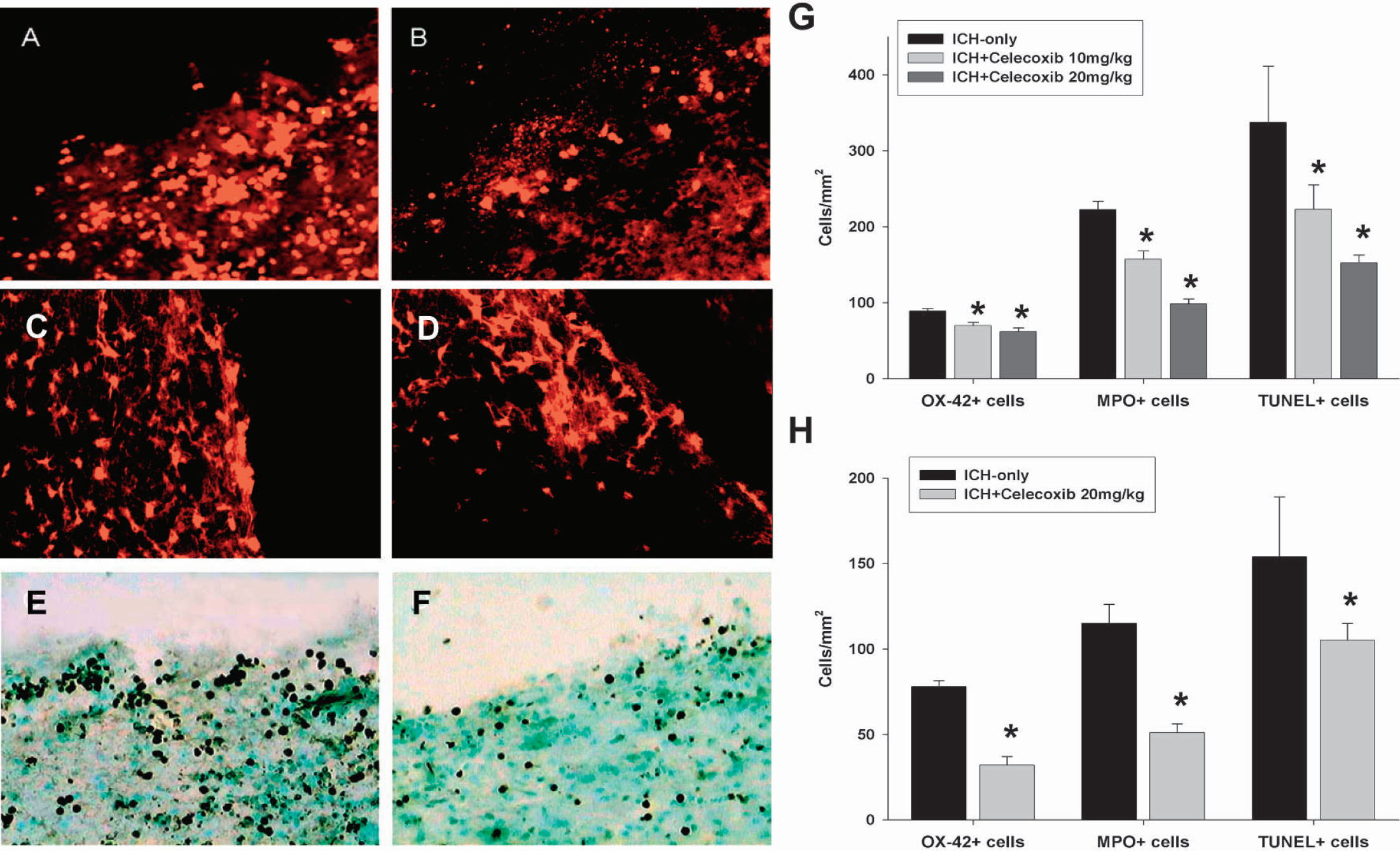
Evaluation of inflammatory and perihematomal cell death 3 days after intracerebral infusion of collagenase in the ICH-only group (
The TUNEL staining revealed a high density of positively stained cells within the hemorrhage lesion itself as well as in the surrounding periphery (Figs. 2E and 2F). Brain sections that were counterstained with toluidine blue showed that the TUNEL-positive cells were not likely to be infiltrating leukocytes. Quantitative analysis showed regional TUNEL-positive cell differences between the groups (Fig. 2G). The ICH-only group (337 ± 74 cells/mm2) showed significantly more TUNEL-positive cells than the celecoxib-treated ICH group (152 ± 10 cells/mm2; P < 0.01, unpaired t-test).
Similar effects of celecoxib on inflammation or cell death after ICH were observed in an alternative model of ICH in which 300 μL of autologous blood was infused into the rat striatum (n = 6; Fig 2H).
Celecoxib reduces PGE2 production in the brains of ICH and collagenase itself does not affect the PGE2 content
PGE2 formation due to COX-2 activation was markedly increased in ICH-only rats. Intraperitoneal administration of celecoxib effectively inhibited brain PGE2 levels in the lesioned side to the level of normal brain within 3 days on treatment (Fig. 3A). Brains from untreated ICH rats had more than three times the normal concentration of PGE2 (P < 0.001, Mann-Whitney test); those in the celecoxib-treated ICH group had significantly lower levels of PGE2 with dose dependency (P < 0.01).
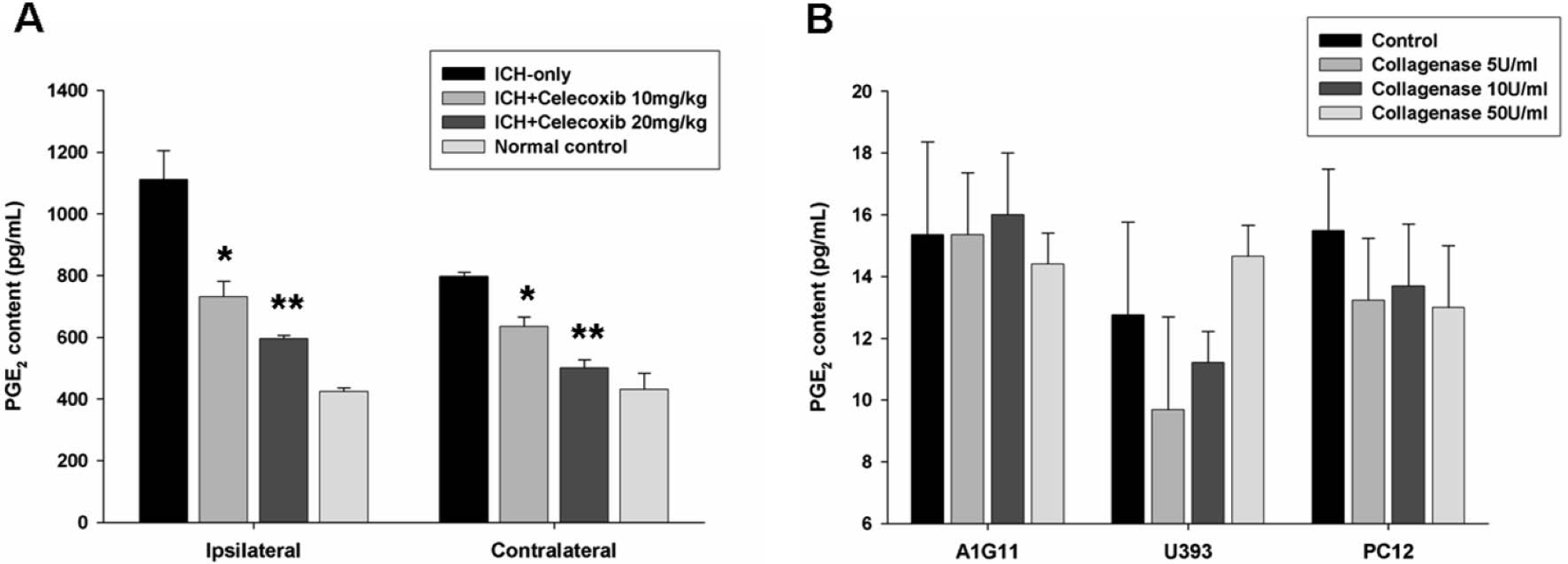
Celecoxib decreases brain PGE2. (
We also performed in vitro assays using three different cell lines. These experiments showed that collagenase concentrations (5–50 U/mL) within the estimated range of those used in our in vivo studies did not affect the PGE2 level in vitro (Fig. 3B).
COX-2 inhibition improves sensorimotor outcome in ICH rats in a time-dependent manner
The celecoxib-treated ICH group (20-minute group) showed better performance on the rotarod and MLPT after 1 week compared with the ICH-only group (Fig. 4; P < 0.01, ANOVA), and this effect persisted for up to 4 weeks. The earlier celecoxib treatment was started, the better the functional outcome (P < 0.01, ANOVA). The 6-hour-treatment group showed better outcome compared with the 24-hour-treatment group at 4 weeks (Fig. 4, P < 0.05), and the 24-hour-treatment group showed better behavioral test results at 28 days after ICH (P < 0.05) compared with the ICH-only group. Initial body weights were similar (P = 0.89, Student's t-test), as were those measured after 4 weeks (P = 0.91). The body weights among all four groups were not significantly different during the entire 4-week period (data not shown).
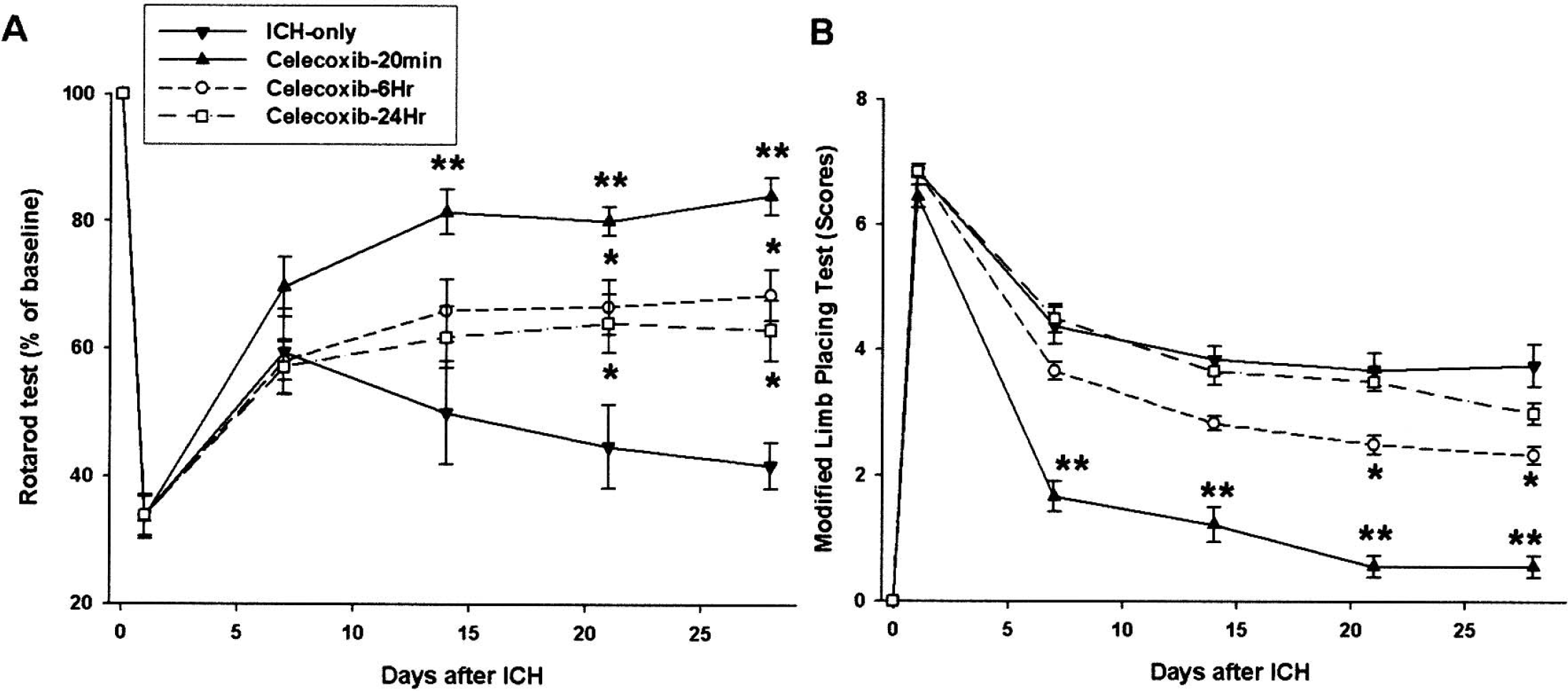
Results of rotarod and modified limb-placement tests. In the rotarod (
DISCUSSION
These experiments were undertaken to test our hypothesis that inhibition of COX-2 activity may have a therapeutic effect in ICH. In this study, we have shown initial evidence suggesting that a selective COX-2 inhibitor, celecoxib, reduces brain edema in the hemispheres bilaterally. The number of TUNEL-positive cells and inflammatory cells in celecoxib-treated rats was significantly decreased within and around the hemorrhagic lesion 3 days after ICH. COX-2 inhibition reduced PGE2 synthesis in the lesioned hemisphere in a dose-dependent manner and improved behavioral outcome. The earlier celecoxib was started, the more robust the sensorimotor recovery. Evidence of similar effects on inflammation and brain edema were obtained from an autologous blood–injected model, suggesting that direct collagenase toxicity was not the major cause of inflammation or cell death. Furthermore, equivalent concentrations of collagenase did not affect the PGE2 production in cultured neurons or astrocytes.
In our study, we observed that the PGE2 level after ICH with celecoxib treatment was decreased in the lesioned hemisphere, and this decrease may be attributable to COX-2 inhibition. The COXs catalyze the rate-limiting steps in the synthesis of prostaglandins (Kelley et al., 1999). The first-order product of both COXs is prostaglandin H2, which is subsequently metabolized into PGD2, PGE2 (the most potent prostanoid for astroglial glutamate release), PGF2α, prostacyclin, or thromboxane A2, depending on which specific prostaglandin synthases are expressed in that cell (Kelley et al., 1999; Smith et al., 1991). Our results support the concept that the beneficial effect of celecoxib is caused by its inhibition of COX-2 in the CNS: PGE2, the major product of COX-2 enzymatic activity, was markedly reduced in the brains of celecoxib-treated ICH rats, as compared with those of untreated ICH-only rats.
It is assumed that activated macrophages and other inflammatory cells could mediate the pathologic effects of COX activity (Chen et al., 1995). COX-2 is rapidly induced in inflamed tissues, and its reactive products are responsible for many cytotoxic effects of inflammation. Proinflammatory cytokines, including interleukin 1β, upregulate the expression of COX-2 in astrocytes as well as in other cells (Bezzi et al., 1998). Our study shows that COX-2 inhibition by celecoxib treatment can reduce the infiltration of inflammatory cells and brain edema, resulting in the reduction of TUNEL-positive perihematomal cell death.
COX-2 messenger RNA and protein expression are upregulated 12 to 24 hours after cerebral ischemia in rodents (Miettinen et al., 1997; Nogawa et al., 1997a, b; Sugimoto et al., 1998). COX-2 expression in rodents is observed in neurons at the periphery of the infarct, in vascular cells, and possibly in microglia (Miettinen et al., 1997; Nogawa et al., 1997). COX-2 is also expressed in the human brain after ischemic stroke (Iadecola et al., 1999; Sairanen et al., 1998). COX-2 immunoreactivity is observed in ischemic neurons at the border of ischemic territory, and in neutrophils and vascular cells. The upregulation in COX-2 immunoreactivity is confined to the area of damage (Iadecola et al., 1999). Administration of NS-398, a relatively selective COX-2 inhibitor, 6 hours after ischemia reduced infarct volume by 20% in 30% in a model of focal ischemia in rats (Nagayama et al., 1999; Nogawa et al., 1997). Recently, the beneficial effect of NS-398 on reduction of infarct volume and improvement of neurologic deficits was noted more than 24 hours after the injection, regardless of the administration timing (Sugimoto and Iadecola, 2003). The observation that delayed administration of NS-398 reduces infarct volume supports the hypothesis that COX-2 is involved in the late stages of ischemic injury.
COX-2 is maximally increased 1 or 3 days after ICH and it mediates inflammatory changes (Gong et al., 2001); however, administration of NS-398 in ICH showed no reduction of brain edema after 24 hours (Gong et al., 2001). The peak time for brain edema in the autologous blood–injection model and collagenase-injection model is the third day after ICH induction (Song et al., 2003; Xi et al., 2001). In addition, NS-398 and celecoxib are 168 times and 375 times selective, respectively, for COX-2 versus COX-1 (IC50 for COX-1 and COX-2: 16.8 and 0.1 μmol/L, respectively, for NS-398, and 15.0 and 0.04 μmol/L for celecoxib) (Vane et al., 1998). In the present study, we used four total doses of a different COX-2 inhibitor (celecoxib) with collagenase- and autologous blood–induced ICH models, and measured the water content at 72 hours after ICH, which might explain the underlying differences of the results between these two studies. Collagenase itself, which was used in this study for the induction of ICH, did not cause cell death (apoptosis) in vitro (Matsushita et al., 2000). However, the triggering events that are responsible for initiating the apoptotic cascade after ICH remain to be defined (Kitaoka et al., 2002).
The data presented here provide initial evidence that celecoxib may reduce inflammation and brain edema with perihematomal cell death. These results suggest that inhibition of COX-2 could have therapeutic effects by altering the cascade of pathogenic processes in the experimental ICH rat model.
Footnotes
Acknowledgment:
The authors thank Yon-Jae C. Chu for preparing the figures and providing editorial support.
