Abstract
Activation flow coupling, increases in neuronal activity leading to changes in cerebral blood flow (CBF), is the basis of many neuroimaging methods. An early rise in deoxygenation, the “initial dip,” occurs before changes in CBF and cerebral blood volume (CBV) and may provide a better spatial localizer of early neuronal activity compared with subsequent increases in CBF. Imaging modality, anesthetic, degree of oxygenation, and species can influence the magnitude of this initial dip. The observed initial dip may reflect a depletion of mitochondrial oxygen (O2) buffers caused by increased neuronal activity. Changes in CBF mediated by nitric oxide (NO) or other metabolites and not caused by a lack of O2 or energy depletion most likely lead to an increased delivery of capillary O2 in an attempt to maintain intracellular O2 buffers.
WHAT IS THE “INITIAL DIP”?
A coupling exists between changes in neuronal activity and changes in cerebral blood flow (CBF) caused by functional stimulation (Villringer and Dirnagl, 1995). This coupling, also known as activation flow coupling (AFC), is the basis of many functional neuroimaging methods including positron emission tomography (PET) and functional magnetic resonance imaging (fMRI). These neuroimaging techniques use large increases in signal intensity of the hemodynamic response as a marker of neural activity. However, debate exists regarding the use of the spatial and temporal characteristics of the hemodynamic response as a surrogate of neural activity (Logothetis et al., 2001).
Over the past few years, a blossoming has occurred in our understanding of the multiple components involved in changes in the hemodynamic response caused by functional stimulation. Malonek and Grinvald (1996), using optical imaging in cats, measured the spatial and temporal dynamics of the changes in blood deoxyhemoglobin concentration (HbR), oxyhemoglobin (Hb-O2), cerebral blood volume (CBV), and CBF in response to a checkerboard visual stimulus. A bi-phasic Hb-O2 response was present with an initial decrease, the “initial dip,” followed by a subsequent larger increase. This initial dip in Hb-O2 occurred before any observed changes in CBV and CBF. These authors postulated that an initial increase in neuronal activity could lead to focal upregulation of oxidative metabolism, which is subsequently overwhelmed by slower, more coarse increases in CBF and CBV that are not as tightly regulated. The result is a “watering of the garden for the sake of one thirsty flower” (Malonek and Grinvald, 1996). Most functional magnetic resonance imaging (fMRI) studies that employ the blood oxygen level dependent (BOLD) method use the large increase in CBF for localization of particular areas involved within a task (Buxton, 2001). However, studies using the initial dip (Duong et al., 2000; Kim and Duong, 2002; Kim et al., 2000) strongly suggest that it may provide a better spatial localizer of early neuronal activity because the draining vein effect seen with the subsequent increases in CBF makes localization more problematic (Buxton, 2001). A greater understanding of this initial dip is relevant for both experimental and clinical functional neuroimaging techniques as well as for the basic mechanisms underlying neurometabolic and neurovascular coupling.
IS THERE A DIP? EVIDENCE FOR AND AGAINST THE DIP USING DIFFERENT MODALITIES
Based upon the initial optical imaging studies described above, an initial dip in the magnetic resonance signal was hypothesized to occur and was observed in a functional magnetic resonance spectroscopy (MRS) study in humans (Ernst and Hennig, 1994). Subsequent functional magnetic resonance imaging (fMRI) at high field strengths (>2 Tesla) have noted an initial dip (Duong et al., 2000; Hu et al., 1997; Janz et al., 1997, 2000; Kohl et al., 2000; Logothetis et al., 1999; Menon et al., 1995; Shtoyerman et al., 2000; Yacoub and Hu, 1999, 2001; Yacoub et al., 1999). However, not all fMRI studies have demonstrated the initial dip. A number of high field strength fMRI studies performed on anesthetized animals were unable to detect an initial dip (Jezzard et al., 1997; Mandeville et al., 1999; Marota et al., 1999; Silva et al., 2000). The presence of the initial dip using fMRI has been shown during pathophysiologic conditions presence (Rother et al., 2002; Roc et al., 2004).
Controversy also exists concerning the presence of the initial dip within optical imaging. Whereas initial optical studies demonstrated the presence of the initial dip (Malonek and Grinvald, 1996; Nemoto et al., 1999), subsequent critiques have suggested a dependence upon the type of analysis performed (Lindauer et al., 2001; Mayhew et al., 2000). Malonek and Grinvald (Malonek and Grinvald 1996) used a simple linear decomposition method to estimate the stimulus-evoked changes in oxyhemoglobin and deoxyhemoglobin. However, the pathlength of the light penetrating the cortex may be wavelength dependent (Jones et al., 2001; Mayhew et al., 2000, 2001). Optical imaging studies using a pathlengthdependent analysis have produced conflicting results; one group was unable to detect an initial dip (Lindauer et al., 2001), whereas another has observed it (Jones et al., 2001; Mayhew et al., 2001). Criticism of the optical imaging technique has also involved the interrelationship between changes in oxygen metabolism, and CBF as a concomitant decrease in oxygenated hemoglobin (Hb-O2) was not observed with initial increases in HbR (Buxton, 2001).
Because of some of the above criticisms, other techniques have been used in an attempt to confirm the presence of the initial dip. Vanzetta and Grinvald (1999) used oxygen phosphorescence quenching to measure changes in oxygen concentration within the microvasculature caused by functional activation. The initial dip was observed with increases in HbR occurring before both CBV and CBF changes. However, Lindauer and colleagues (2001) (Lindauer et al., 2001), using this same technique in rats, were unable to replicate these results. Recently direct measurements of changes in partial pressure of tissue oxygen (pO2), using an O2 microelectrode, during functional stimulation have been performed by several laboratories (Ances et al., 2001; Thompson et al., 2003). In rats anesthetized with α-chloralose during a forepaw stimulation paradigm, a transient decrease in partial pressure of tissue pO2, an initial dip was repeatedly observed before CBF changes, as measured by laser Doppler (Ances et al., 2001). The magnitude of the initial dip as measured by tissue pO2 electrodes can also be influenced by variations in forepaw stimulation parameters (Kanno, 2003). The initial dip has been observed in the cat visual cortex using tissue pO2 microelectrode. Simultaneous measurements of changes in tissue pO2 and neuronal activity demonstrated that early increases in neuronal spike activity were accompanied by an immediate decrease in tissue oxygenation (Thompson et al., 2003).
WHY DO SOME OBSERVE THE INITIAL DIP WHEREAS OTHERS DO NOT?
A number of factors including differences in methodology, anesthetic, degree of oxygenation, species, or system studied may account for some the differences observed among studies investigating the initial dip. Methodologic differences may exist not only depending upon the modality (fMRI, spectroscopy optical imaging, oxygen phosphorescence, O2 microelectrode) but also in technical differences such as field strength, optical wavelength range, or mode of analysis. Each of the imaging studies that have been used is more sensitive to particular vascular compartments. BOLD-fMRI primarily reflects oxygen changes within the venous system (Frahm et al., 1994; Hall et al., 2002; Turner, 2001), whereas optical imaging measures changes within capillaries (Obrig and Villringer, 2003; Pouratian et al., 2003). Oxygen phosphorescence imaging can obtain measurements of oxygen changes within capillaries and precapillaries, whereas O2 microelectrode measurements primarily reflect tissue oxygen changes (which lie somewhere between mitochondrial and capillary O2) (Villringer and Dirnagl, 1995; Wilson, 1992). Within each of these compartments, temporal as well as spatial differences in oxygen metabolism may exist, leading to the presence or absence of an initial dip. As highlighted above, even within a particular technique, such as optical imaging, the mode of analysis can lead to conflicting results (Jones et al., 2001; Lindauer et al., 2001; Mayhew et al., 2001). Differences observed within this technique may be caused by optical wavelength or field strength used. However, as seen in Table 1, these differences alone cannot account for the large number of studies that have observed the initial dip at varying field strengths as well as across a range of wavelengths (Shtoyerman et al., 2000).
Studies showing the presence or absence of the “initial dip”
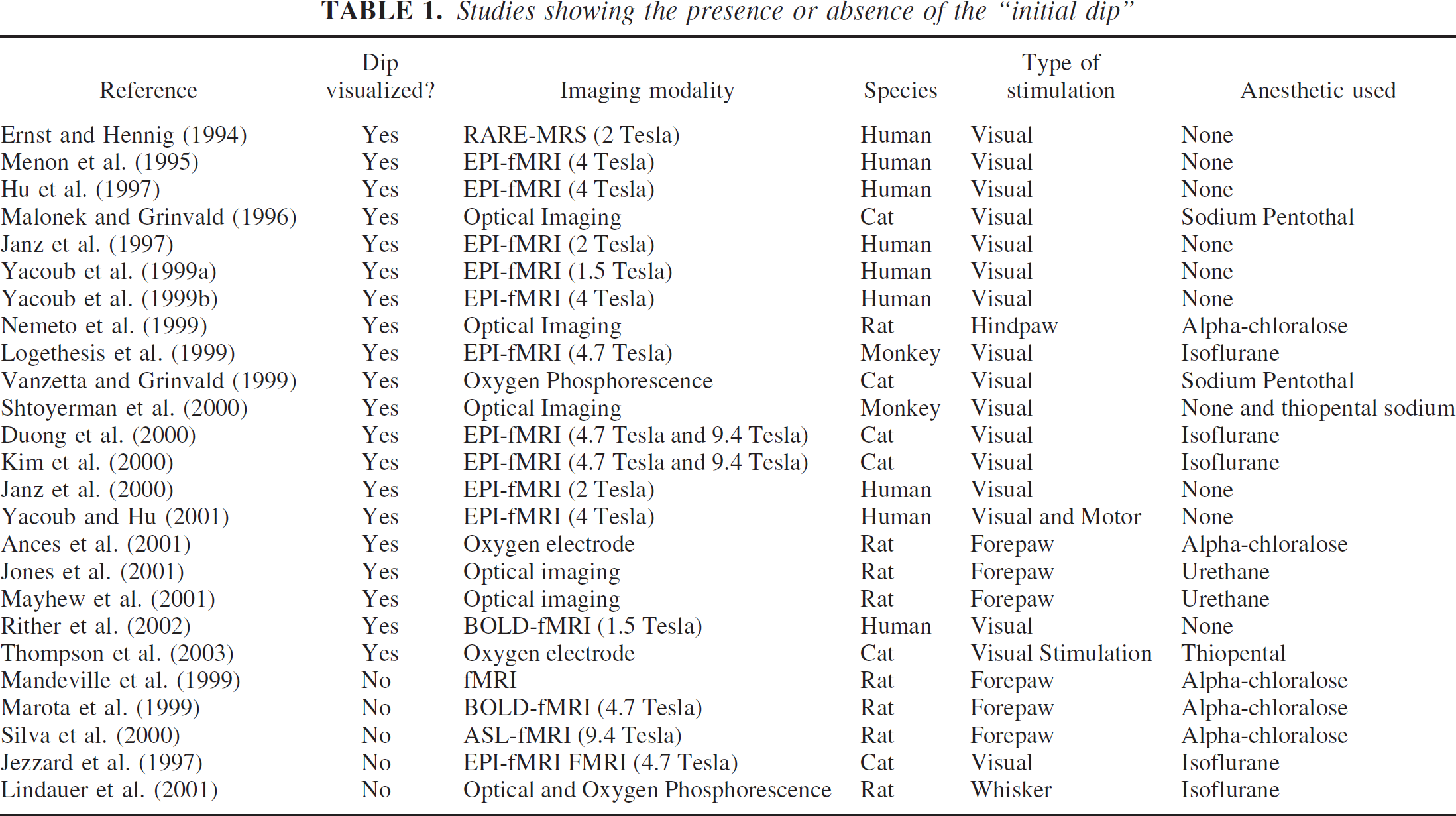
The degree of anesthesia could affect the magnitude of the initial dip; early fMRI studies in awake humans did not observe the initial dip, whereas subsequent studies in anesthetized animals have observed the initial dip. More recently a number of optical imaging studies have attempted to determine the effect of anesthesia on the initial dip. Both in monkeys (Shtoyerman et al., 2000) and rats (Jones et al., 2001) the overall magnitude of the initial dip was significantly greater in awake compared with anesthetized animals. Anesthesia did not influence the temporal dynamics of the initial dip. These results suggest that the degree of anesthesia may affect the coupling between neuronal activity and blood circulation. The exact mechanism remains unknown, but a number of studies have shown that anesthetics can influence the production of possible mediators, such nitric oxide (NO), involved in AFC (Baumane et al., 2002; Galley et al., 2001; Nakao et al., 2001).
Not only anesthesia but also oxygen blood saturation may influence the amplitude of the initial dip. Preliminary experiments by Mayhew et al. (2001) have demonstrated that increasing oxygen concentrations of inspired air reduced the amplitude of the initial dip. This may account for some of the differences observed between recent conflicting optical imaging studies; one group was unable to observe the initial dip but used a higher fraction of O2 in the inspired air (Lindauer et al., 2001). Changes in pCO2 may also influence the magnitude of the initial dip. At pCO2 levels of greater than 40 mm Hg, the initial dip was observed in the primary visual cortex in cats because of visual stimulation (Kemna and Posse, 2001). However at lower pCO2 values, the initial dip could not be detected (Cohen et al., 2002; Harel et al., 2002). These results suggest that hypercapnic conditions could lead to an increase in oxygen consumption (Hyder et al., 1998, 2000) and may augment the initial dip. CO2-induced vasodilation may partially compensate by allowing for the diversion of CBF from areas of less activity to areas of increased demand (Woolsey et al., 1996).
Differences in species have also been proposed for the presence or absence of the initial dip (Silva et al., 2000). The initial dip has been observed in cats (Malonek and Grinvald, 1996; Malonek et al., 1997; Thompson et al., 2003) with subsequent confirmatory studies in both humans (Yacoub and Hu, 1999) and monkeys (Logothetis et al., 2001). However, an initial dip was not seen in rats using fMRI (Silva et al., 2000). More recent studies in this species using tissue pO2 microelectrodes (Ances et al., 2001) as well as optical imaging (Jones et al., 2001) have demonstrated the initial dip. The relatively smaller amplitude of the initial dip compared with subsequent larger hyperoxygenation increases, varies across species. Smaller species have a smaller ratio for the magnitude of initial deoxygentation changes, initial dip, compared with the subsequent hyperoxygenation changes mediated by changes in CBF. These hyperoxygenation changes may mask the initial dip for certain imaging modalities, especially in smaller species. Variations observed across species may also reflect differences in the cerebral transit time required for the passage of blood from the larger arteries to draining veins. Smaller species such as rats have higher blood flow and subsequently lower cerebral transit times that may not be temporally resolved with some imaging modalities. Shorter cerebral transit times could lead to reduction in the lag period between the onset of increases mitochondrial oxygen demand and CBF changes and therefore diminish the magnitude of the initial dip (Lindauer et al., 2001).
Differences in the systems studied have also been postulated to affect the presence or absence of the initial dip (Silva et al., 2000). As seen in Table 1, a number of anatomic systems, including the visual, motor, and sensory systems, have been studied. The initial dip has been observed in each of these model systems, but its amplitude has varied. The observed differences may also reflect the content of cytochrome oxidase metabolism within each of these systems (Fujita et al., 1999; Vafaee et al., 1999).
A POSSIBLE UNIFYING THEORY BEHIND THE INTIAL DIP
Stimulus-induced neural activity is associated with an increase in sodium (Na+) flow into the cell. Increases in intracellular Na+ activate a sodium-potassium pumps (Na+/K+) requiring consumption of adenosine triphosphate (ATP), the energy building block of the cell (Clarke and Sokoloff, 1994; Gjedde, 1997). ATP energy required for cellular function can be generated via three different mechanisms with different time constraints and regulation mechanisms: (1) a buffering effect of phosphocreatine that reacts with adenosine diphosphate (ADP) to produce ATP and creatine, (2) glycolysis that converts intracellular glucose into intracellular pyruvate and lactate and leads to the production of two ATPs, and (3) mitochondrial respiration that consumes intracellular oxygen (O2) and pyruvate via the tricarboxyclic acid cycle to produce 15 ATPs. Mitochondrial respiration depends upon the ATP/ADP ratio and the intracellular concentrations of pyruvate and oxygen present (Aubert and Costalat, 2002).
Increases in neuronal activity lead to regional CBF changes within capillaries. A “steal phenomenon” may occur during AFC with CBF changes directed towards metabolically activate neurons with certain feeding arterioles dilated while others are constricted (Harel et al., 2002; Woolsey et al., 1996). The result will be CBF increases to particular metabolically active areas to provide necessary nutrients, including glucose and O2, required for ATP production.
A number of mediators or modulators has been postulated to be involved in AFC. One mediator that has recently received a great deal of attention is NO because it is a rapidly diffusable vasoactive substance with a relatively short half-life. Studies have shown that inhibition of NO using certain NO synthase antagonists can lead to an attenuation but not elimination of the CBF response because of functional stimulation in both animals (Gotoh et al., 2001; Lindauer et al., 1999) and humans (White et al., 1999). Simultaneous measurements of CBF, using laser Doppler, and tissue NO, using tissue microelectrodes, have been obtained in rats anesthetized with α-chloralose during repeated forepaw stimulation (Buerk et al., 2003). Superposition of these results upon previously obtained simultaneous measurements of CBF and microelectrode tissue O2 (Ances et al., 2001) are shown in Fig. 1. After the onset of stimulation, tissue NO immediately rose, while an initial dip in tissue O2 was present. Tissue changes in NO and O2 occurred before measured CBF or CBV changes and are similar to those seen by other laboratories using different stimulation paradigms and modalities (Grinvald et al., 2000; Thompson et al., 2003; Vanzetta and Grinvald, 1999). These results strongly suggest that the initial dip is primarily caused by changes in deoxygenation and not CBF or CBV changes. Subsequent rises in CBF lead to a reduction in tissue NO because of a possible wash-out effect (Buerk et al., 2003), whereas a delayed peak in tissue O2 occurs as an increase in nutrients are provided to the activated neurons (Ances et al., 2001).
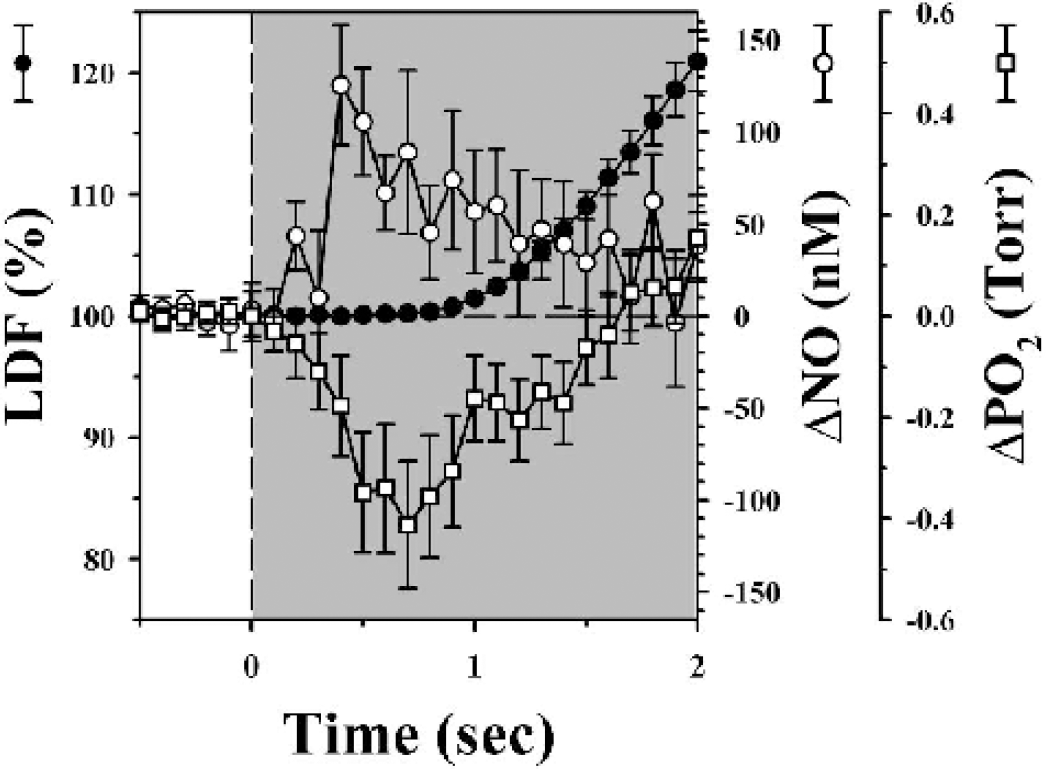
Studies showing presence or absence of the initial dip. Superimposed signal averaged changes in tissue NO (Beurk et al., 2003) and pO2 (Ances et al., 2001) and cerebral blood flow (CBF) caused by 4 seconds of forepaw stimulation with a 20-second interstimulus interval in rats anesthetized with α-chloralose. Change in tissue NO (open circles ± SE, scale at right) and tissue pO2 (open squares ± SE, scale at far right), as measured by tissue microelectrodes, preceded changes in CBF (solid circles ± SE, scale at left), as measured by laser Doppler, during forepaw stimulation (shaded regions). An initial dip in tissue pO2 was observed while tissue NO quickly rose prior to changes in CBF.
At the cellular level, an increase in neuronal activity leads to a rise in energy requirements to maintain Na+/K+pumps, reduce free radical production, and allow for the recycling of neurotransmitters (Atwell and Iadecola, 2002; Ido et al., 1997). Initial increases in ATP required for homeostasis are derived primarily from presynaptic mitochondrial respiration and phosphocreatine reserves (Aubert and Costalat, 2002). The observed initial dip in tissue pO2 may reflect a utilization of mitochondrial O2 stores in an attempt to replenish diminishing ATP reserves that occur with increase in neuronal activity (Aubert and Costalat, 2002). Most of the energy is expended on reversing ion fluxes underlying excitatory postsynaptic currents and action potentials (Atwell and Laughlin, 2001; Atwell and Iadecola, 2002), with only a small percentage of this O2 metabolism, approximately 10%, based upon stoichiometric reactions, used for the production and release of NO (Thomas et al., 2001) or other mediators involved in coupling. Thus, changes in CBF are most likely mediated by NO or other metabolites and not the lack of O2 (Mintun et al., 2001) or a decrease in energy (Powers et al., 1996). These changes in CBF most likely will lead to an increase in delivery of capillary O2 in an attempt to maintain the intracellular O2 levels. A gradient for O2 will exist with O2 easily diffusing from the blood into the presynaptic neuron, in particular the mitochondria. As the O2 gradient is reduced and other sources of ATP are used, primarily phosphocreatine and glycolysis, the O2 extraction fraction will decrease (Hyder et al., 1996). Larger CBF changes may therefore be required to provide increases in glucose for glycolysis as well as maintain a gradient for O2 for continued diffusion into the mitochondria (Buxton, 2001).
CONCLUSION
This review has attempted to summarize existing studies that have investigated the initial dip using different imaging modalities and suggests that the initial dip is present. A unifying physiologic hypothesis has also been proposed concerning why the initial dip occurs as well as why certain studies have been unable to detect the initial dip. Further studies investigating the mediators involved as well as the initial dip under pathophysiologic states are required.
Footnotes
Acknowledgment
The author thanks Drs. Buxton, Bandettini, Detre, Hyder, Kim, and Mayhew, as well as the reviewers, for their thoughtful comments and discussions.
