Abstract
Although macrophages represent the major inflammatory cells in cerebral ischemia, the kinetics of macrophage infiltration are largely unknown. To address this issue, we injected superparamagnetic iron oxide (SPIO) particles into the circulation of rats at different time points after focal photothrombotic cerebral infarction and performed magnetic resonance imaging (MRI) 24 hours later. Infarcts appeared as hyperintense lesions on T2-w and CISS MR images during all stages. At days 5.5 and 6, an additional rim of signal loss indicative of local accumulation of SPIO particles appeared at the outer margin of the hyperintense ischemic lesions, which was not present at days 1 to 5. Areas of signal loss corresponded to local accumulation of iron-loaded macrophages in histologic sections. At day 8, signal loss became restricted to the inner core of the lesions and ceased thereafter. Macrophages, however, were still present in late ischemic brain lesions, but they were iron-negative. Thus SPIO-induced signal loss indicates active macrophage transmigration into ischemic infarcts but not their mere presence. SPIO-induced signal loss was independent from the disturbance of the blood-brain barrier. In conclusion, we have shown by in vivo monitoring that macrophages enter photothrombotic infarcts at late stages of infarct development, suggesting a role in tissue remodeling rather than neuronal injury.
Focal cerebral ischemia evokes a profound inflammatory response involving granulocytes, T-cells, and hematogenous macrophages (Kochanek and Hallenbeck, 1992; Stoll et al., 1998). Immune mediators produced by these inflammatory cells have been inflicted in secondary infarct expansion (Allen and Rothwell, 2001; Del Zoppo et al., 2000; Hallenbeck, 2002). In contrast with granulocytes and T-cells that can be unequivocally identified by morphologic criteria and cell-type specific immunologic markers (Jander et al., 1995; Kochanek and Hallenbeck, 1992; Schroeter et al., 1994), both the cellular origin of phagocytic cells and their timing of infiltration into ischemic brain lesions are a matter of debate (Lehrmann et al., 1997; Schroeter et al., 1997; Stoll and Jander, 1999). Resting microglia in the normal central nervous system (CNS) parenchyma are ramified and easily discernible from macrophages. Upon activation, however, microglia transform into phagocytes and become indistinguishable from hematogenous macrophages on morphologic grounds. In addition, macrophages share most phenotypic markers with phagocytic microglia, including the complement-3 receptor and the lysosomal antigen ED-1 (Flaris et al., 1993). Because of these difficulties, the exact timing of macrophage entry and the relative contribution of microglia to the pool of phagocytic cells in ischemic brain lesions is largely unknown.
To address this issue, we injected superparamagnetic iron oxide (SPIO) particles systemically into the blood circulation of rats at different time points after focal cerebral ischemia and performed magnetic resonance imaging (MRI) 24 hours later. Tissue accumulation of SPIO particles appears on T2-weighed (T2-w) MR images as focal signal loss because of their paramagnetic effect (Chen et al., 1999). As a model of focal cerebral ischemia, rat photothrombosis as originally described by Watson and colleagues (1985) was chosen because the location and size of the lesions are highly reproducible, and cellular responses develop in a predictable sequence (Jander et al., 1995; Schroeter et al., 1997, 1999). In this model, a photosensitive dye, rose bengal, is injected intravenously and subsequentially activated locally by a light beam through the intact skull, which leads to free-radical formation and consecutive thrombosis of small vessels of the illuminated cerebral cortex (Dietrich et al., 1987). In contrast with middle cerebral artery occlusion (MCAO) models, photothrombosis produces a circumscribed infarction and breakdown of the blood-brain barrier without a penumbra, which may interfere with clear delineation of the infarct boundaries on MRI (Dietrich et al., 1987). Patterns of postischemic inflammation and cytokine induction in photothrombotic cerebral infarcts are similar to focal ischemia induced by permanent MCAO, as shown in numerous studies (Jander et al., 1995, 2000; Schroeter et al., 1994, 1997, 1999). We demonstrate in vivo that hematogenous macrophages are the predominant inflammatory cells infiltrating ischemic lesions within a tight time frame and at late stages of infarct development.
MATERIALS AND METHODS
Animal experiments
Focal cortical infarctions were induced in 58 adult male Wistar rats (220 to 250 g) by photothrombosis (PT) of cortical microvessels under inhalation anesthesia with enflurane in a 2:1 nitrogen/oxygen atmosphere, as described in detail elsewhere (Jander et al., 1995; Watson et al., 1985). During anesthesia, rectal temperature was kept at 36.5°C to 37.5°C by a servo-controlled heating blanket. Briefly, a fiber optic bundle of a cold light source was centered stereotactically 4 mm posterior and 4 mm lateral from Bregma on the skull exposed via a dorsal midline incision of the skin. Via a femoral vein catheter, 0.4 mL of a sterile-filtered rose bengal solution (10 g/L; Sigma, Deisenhofen, Germany) was given, and the brain was illuminated for 20 minutes. Afterwards, the skin was sutured, and the rats were allowed to recover. This procedure resulted in cone-shaped cortical infarctions without clinically visible symptoms. All animal experiments were performed in accordance with institutional guidelines and were approved by Bavarian state authorities.
Magnetic resonance imaging
In a first set of experiments, 0.2 mmol Fe/kg body weight superparamagnetic iron oxide (SPIO) particles (Resovist, Schering, Berlin, Germany) were applied systemically into the circulation 24 hours before every MR examination. All measurements were performed on a clinical 1.5 Tesla MRT unit (Magnetom Siemens Vision, Erlangen, Germany). Rats were anesthetized with intraperitoneal (IP) injections of 100 mg/kg body weight ketamin (Ketanest) and 10 mg/kg body weight xylazin (Rompun) and MRI performed at days 1 (n = 3), 2 (n = 3), 3 (n = 3), 4 (n = 3), 5 (n = 2), 5.5 (n = 2), 6 (n = 6), 7 (n = 6), 8 (n = 5), 9 (n = 4), 11 (n = 3), 12 (n = 3), and 14 (n = 3) after photothrombosis. A total of six control animals that had received bengal rose but were not illuminated (three) or were illuminated after injection of saline instead of the dye (three) received SPIO particles at day 5 and were scanned by MRI on day 6. In addition, three animals underwent MRI without SPIO application at day 6 after PT to obtain unenhanced CISS images and subsequently received systemic Gd-DTPA.
To specifically assess the relation between SPIO-induced signal loss and the disruption of the blood-brain barrier (BBB), groups of rats received SPIO particles on days 5 (n = 3) or 8 (n = 3). They were scanned by MRI on days 6 or 9, respectively, for iron deposition on CISS images and immediately thereafter received additional systemic Gadolinium-DTPA injections (Gd-DTPA, Magnevist, Schering, 0.2 mmol/kg body weight) as a marker for BBB integrity. Animals were then scanned a second time to assess Gd-DTPA-enhancement on T1-w images. Thereafter, CISS and T1-w images were compared in individual animals. In a third set of experiments, three animals received SPIO particles once at day 5 after PT and were scanned subsequentially twice at days 6 and 10 by MRI to study the dynamics of signal loss after a single injection.
For all MR measurements, animals were lying in supine position with their heads fixed in a round surface coil (40 mm diameter). The MR protocol included T1-w (TR 460 milliseconds, TE 14 ms) and T2-w (TR 2,500 milliseconds, TE 80 ms) sequences in the coronal plane with a slice thickness of 3 mm. Moreover, a 3D CISS (constructed interference in steady state, TR 16.4 milliseconds, TE 8.2 ms) sequence was applied in the coronal plane with a slice thickness of 1 mm (Casselmann et al., 1993). For postprocessing MR data were transferred to an external workstation (Leonardo, Siemens, Erlangen, Germany). We then determined the overall lesion volume (mm3) in each animal by manually delineating the lesion borders in subsequent slices. Moreover, in animals of days 5 to 8 after PT, the extent of the hypointense area caused by iron-accumulation was calculated and related to the overall lesion volume. In addition, the mean signal intensity of the hypointense and hyperintense areas on CISS images were determined and normalized to cerebrospinal fluid signals to allow for interindividual comparison. Mean values were statistically compared using one-way ANOVA.
Histology
After MRI rats were killed by CO2 narcosis, the brains were rapidly removed and immersion-fixed overnight in 4% paraformaldehyde, washed in phosphate-buffered saline (PBS), and embedded in paraffin. Subsequently, 5-μm thick paraffin sections were cut at multiple levels through the infarcts, deparaffinized with xylene, rehydrated, and washed in water and PBS. For iron detection, tissue sections were rinsed in deionized water and immersed in Perl's solution containing 2% potassium ferrocyanide and 2% HCl at a 1:1 concentration for 30 minutes. Sections were then rinsed in deionized water and either dehydrated and coverslipped or further processed for ED-1 immunocytochemistry. Additional sections were stained by immunocytochemistry using the antibody ED-1 at a dilution of 1:1000, as a marker for monocytes/macrophages and phagocytic microglia (Serotec, Oxford, UK). Binding of ED-1 antibodies to cells was visualized by the avidin-biotin-peroxidase method with diaminobenzidine as chromogen according to routine procedures (Stoll et al., 1989). For unequivocal identification of iron-labeled cells, we performed Perl's stain and ED-1 immunocytochemistry upon the same sections. Sections were examined with a Zeiss-Axiophot microscope.
RESULTS
In a first set of experiments, all animals received systemic SPIO particles injections 24 hours before MRI. No changes on T1-w, T2-w, or CISS images were seen in control animals after SPIO application that had been sham-illuminated after IV injection of saline nor in rats that had received rose bengal without brain illumination. In the experimental group, photothrombotic infarcts appeared as hyperintense lesions on T2-w and CISS MR images during all stages of infarct development (Fig. 1A). Whereas no areas of signal loss were present in ischemic lesions at early stages 1 to 5 days after photothrombosis and SPIO injection the day before (Fig. 1A), a small rim of signal loss regularly appeared at the outer margin of the hyperintense ischemic lesions on T2-w and CISS images at days 5.5 and 6 (Fig. 1B), which is indicative of local accumulation of SPIO particles. The extent of the signal loss could best be demonstrated on high resolution CISS images. At days 7 and 8, the outer rim became hyperintense again, and more central areas now revealed signal loss (Fig. 1C). At later stages of infarct development, such as days 9 to 14, the lesions were shrunken and appeared entirely hyperintense on T2-w images. No more focal areas of signal loss were seen. These findings indicate that SPIO particle-induced signal loss in ischemic brain lesions is a transient event.
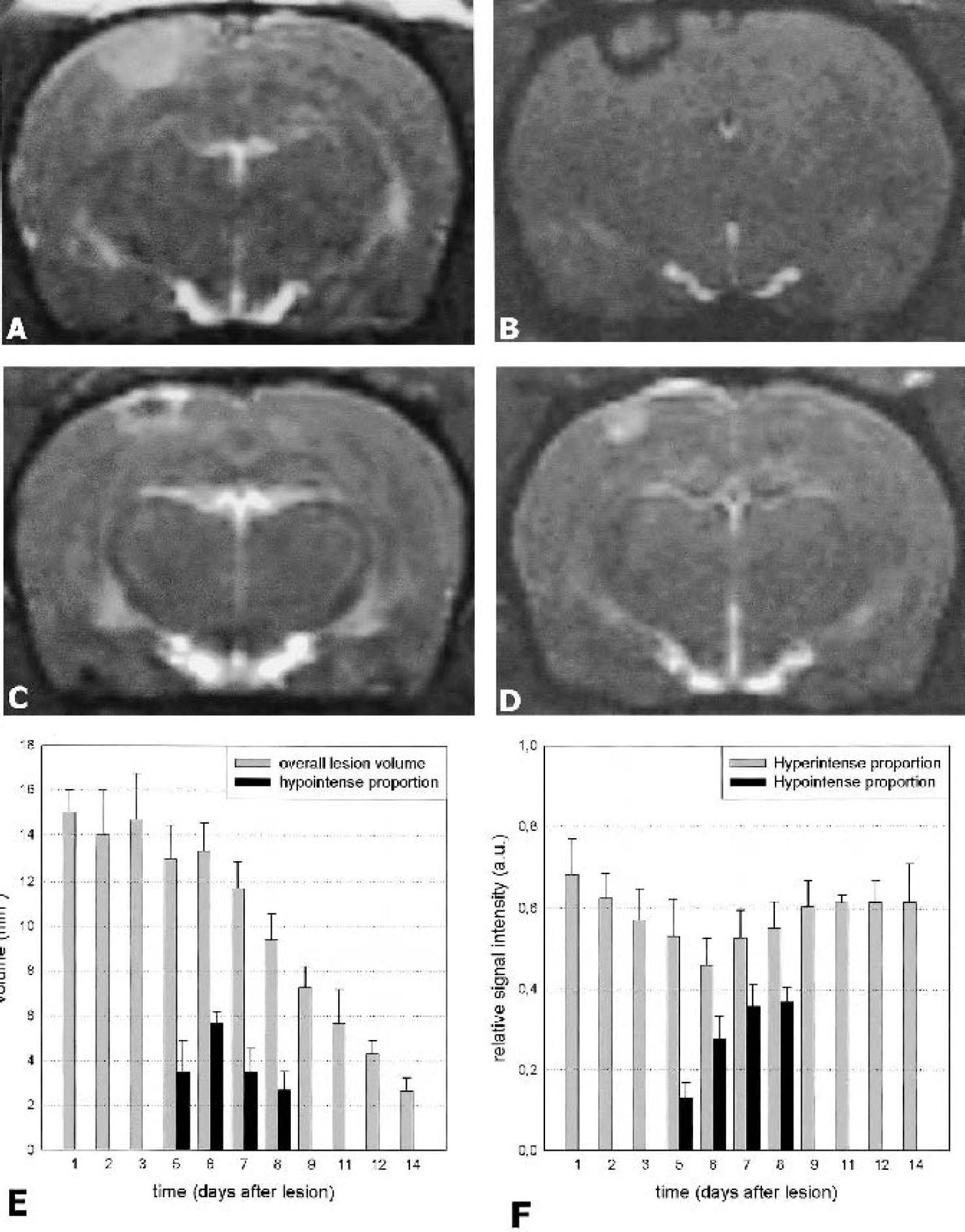
In vivo assessment of macrophage infiltration by magnetic resonance imaging as indicated by focal signal loss: coronal CISS-weighed images (slice thickness 1 mm) of photothrombotic infarcts at day 3
Lesion volumes did not significantly differ between days 1 to 6 after PT (P > 0.05, one-way ANOVA) (Fig. 1E) but were significantly smaller from days 7 onwards compared with day 1, indicating shrinkage of the lesions caused by tissue remodeling (P < 0.05) (Fig. 1E). Signal intensity measurements of the hyperintense lesion proportions exhibited a lower mean signal at day 6 after PT compared with days 1 (P = 0.002) and 3 (P = 0.047) (Fig. 1F). The proportion of the hypointense area in relation to total infarct volume was greatest at day 6 after PT and already decreased at days 7 (P = 0.004) and 8 (P = 0.002) (Fig. 1F). Moreover, quantitatively, the hypointense signal reached lowest values indicating peak iron deposition at day 5.5, which significantly differed from the values at days 6 (P = 0.012), 7 (P < 0.001), and 8 (P < 0.001). In control animals with infarcts but no SPIO-injections, ischemic lesions did not exhibit signal loss on CISS or T2-w images as examined on day 6 and reported before (Schroeter et al., 2001) but showed Gd-DTPA enhancement (Fig. 2A and Fig. 2B). To further assess the relation between SPIO-induced signal loss and disruption of the BBB, we investigated the temporal course of Gd-DTPA enhancement on T1-w images in individual animals. Whereas Gd-DTPA enhancement was present at all time points examined, such as days 6 and 9 after PT (Fig. 2D and Fig. 2F), SPIO-induced signal loss was restricted to day 6 (Fig. 2C) but was absent on day 9 (Fig. 2E). This indicates that breakdown of the BBB and macrophage infiltration are unrelated events.
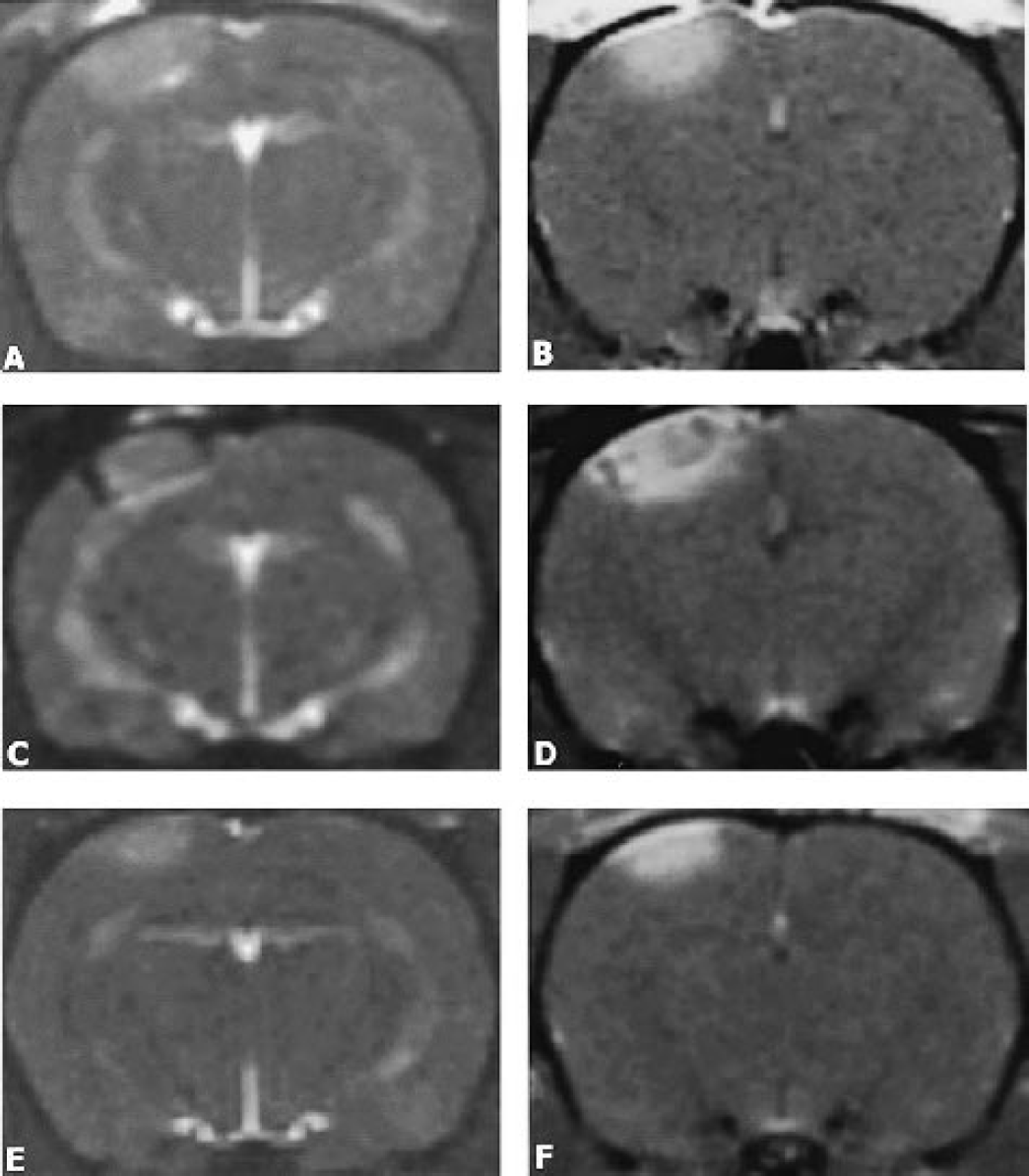
Relationship between SPIO-induced signal loss and breakdown of the BBB as indicated by gadolinium enhancement in focal ischemic lesions: CISS image of an infarct at day 6 without previous SPIO particle application shows no hypointense areas
In confirmation of our MRI findings, we could demonstrate focal iron deposition in histologic sections stained with Perl's solution. In the ischemic lesions up to day 5, only occasional cells on the pial surface were iron-positive. At days 5.5 and 6 after photothrombosis, a large number of iron-laden cells were located at the outer margin of ischemic brain lesions (Fig. 3A and Fig. 3B), whereas at days 7 and 8, more central parts of the ischemic brain lesions contained iron-laden cells. At later stages of infarct development, that is, beyond day 9, iron-positive cells were virtually absent in ischemic lesions. Thus, our histologic findings corresponded to the MRI results and confirmed the transient nature of the iron-induced signal loss. The nature of iron-laden cells in ischemic brain lesions could be identified by immunocytochemistry. Identical tissue sections were immersed in Perl's stain for iron detection and subsequently processed for ED-1 immunostaining, a marker for phagocytic monocytes/macrophages revealing extensive colocalisation at days 5.5 to 8 after photothrombosis (Fig. 3C). Of importance is the fact that numerous ED-1+ macrophages were still present in the ischemic lesions at days 9 to 14 after photothrombosis, but macrophages no longer exhibited intracellular iron deposition at this time and in this location (Fig. 3D). Lack of iron-labeling of phagocytes at later stages of infarct development when the blood-brain-barrier (BBB) is still disturbed (see above) clearly indicates that SPIO particles did not passively diffuse through the BBB. Rather, SPIO particles must have been phagocytosed by monocytes in the circulation and transported cell-bound into the brain during phases of active infiltration. This could be proved by histologic examination, showing that iron was always cell-associated and not found in the extracellular space. On the meningeal surface of ischemic brain lesions, a small number of iron-laden macrophages were present through all stages of infarct development, indicating a continuous turnover of meningeal macrophages.
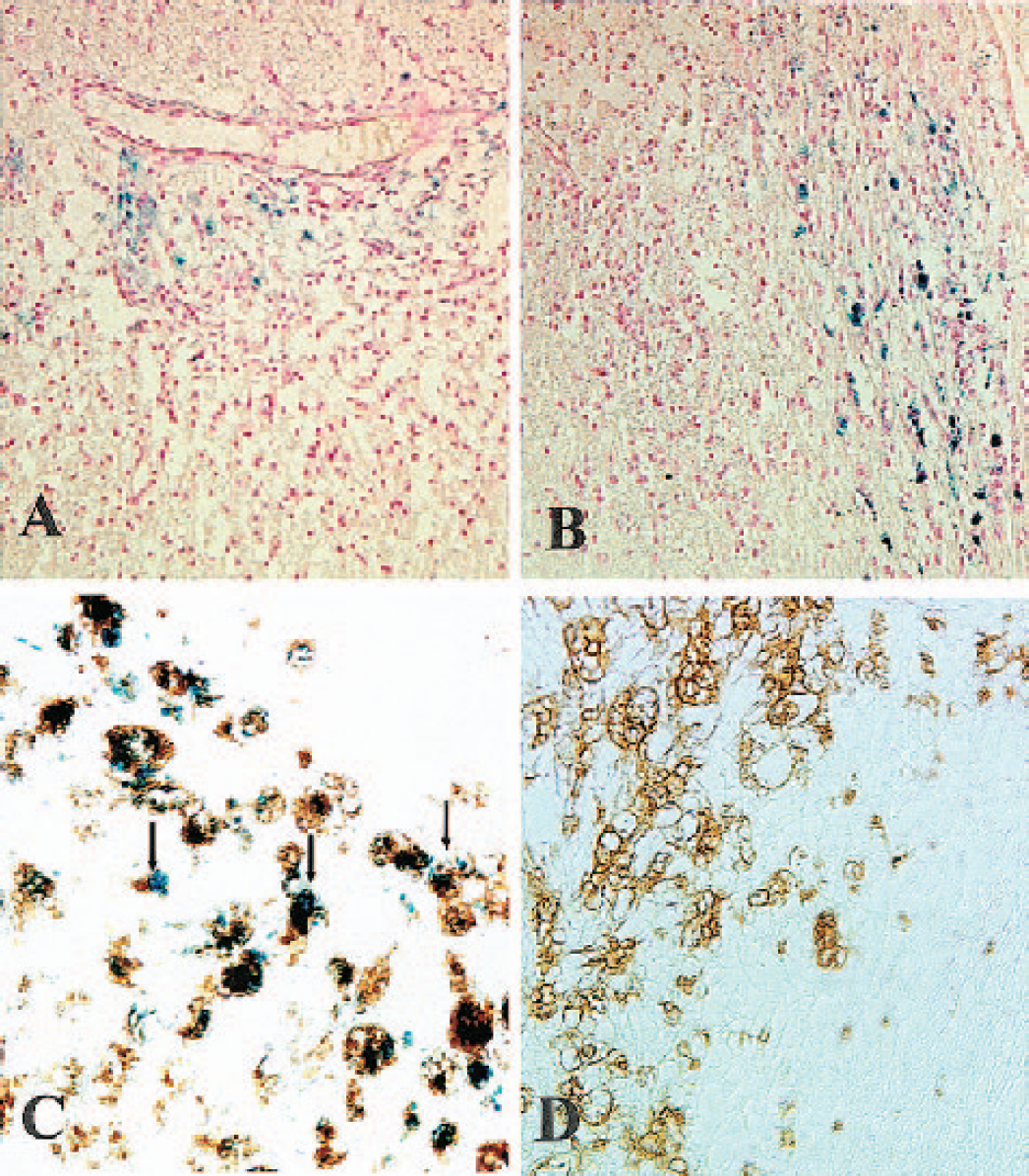
Localization and identification of iron-laden cells in ischemic brain lesions: paraffin sections were stained for iron (blue in
In a third set of experiments, we assessed if iron-labeled macrophages further migrate towards the core once they have infiltrated the rim of the lesion. To demonstrate this, photothrombotic infarcts were induced, SPIO were applied into the circulation at a fixed time point at day 5, and consecutive MRI scans were performed in these animals at days 6 and 10. In accordance with our first experiments, animals showed signal loss at the outer margin of their ischemic lesions at day 6 (Fig. 4A). Almost the same distribution of signal loss was seen on the second MRI scans at day 10, indicating that macrophages remained sessile at the site of their initial infiltration (Fig. 4B).
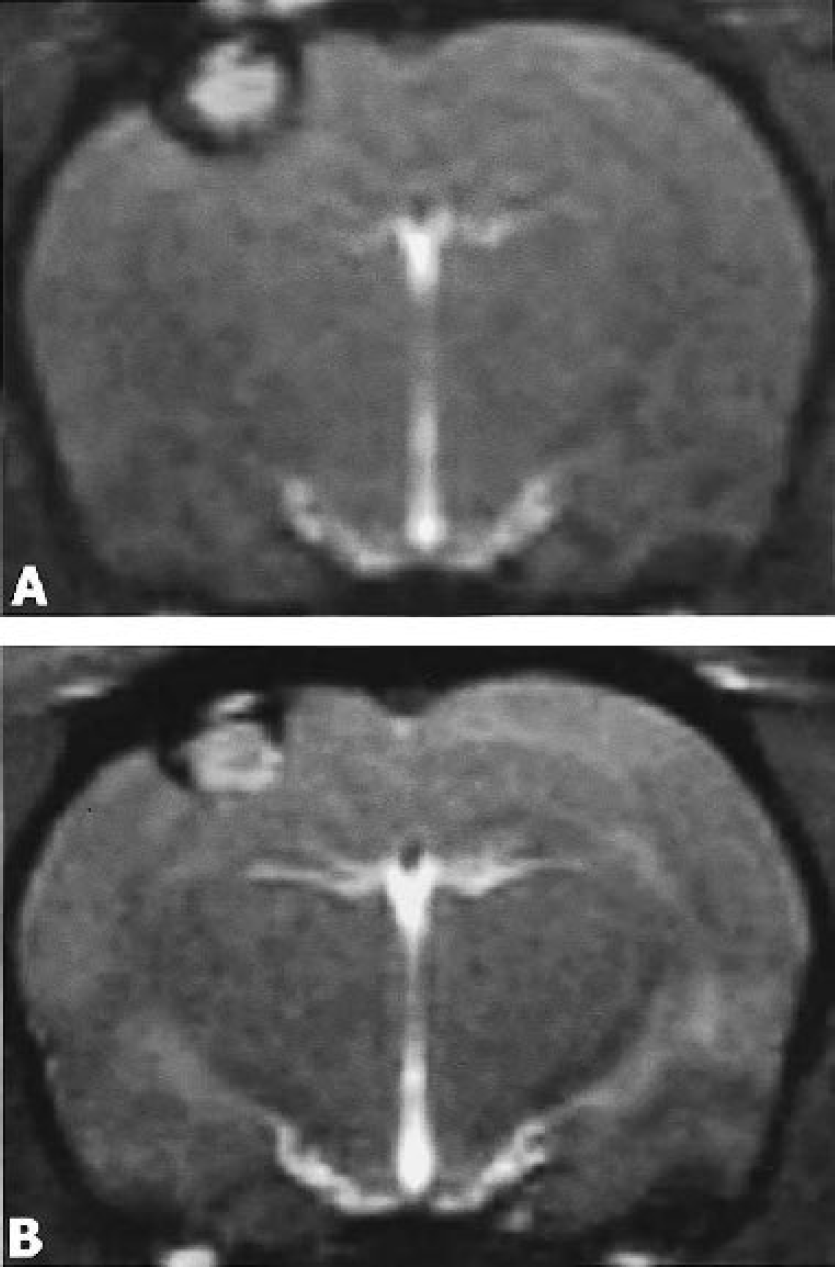
Two subsequent MR scans (coronal view, CISS sequence) in an individual rat at day 6
DISCUSSION
As principal findings, we show that macrophages infiltrate focal ischemic brain lesions at late stages of infarct development. Macrophage recruitment occurred within a very tight time frame and in a spatial order starting at the outer rim of infarcts and further proceeding into the core. To visualize macrophage infiltration in vivo, we systemically injected SPIO particles 24 hours before MRI. Signal loss on T2-w and CISS images in ischemic brain lesions was caused by the presence of numerous iron-laden cells in corresponding locations as revealed by Perl's staining of tissue sections. Iron-positive cells could unequivocally be identified as ED-1 positive macrophages by combining Perl's stain and immunocytochemistry. At later stages of infarct development, that is, beyond day 9 after photothrombosis, macrophages were still abundant in the lesions but were iron-negative. These findings indicate that SPIO-enhanced MRI depicts the actual migration of iron-laden monocytes from the circulation into the brain and strongly argue against passive diffusion of SPIO particles through a defective BBB and consecutive uptake by local phagocytes. Accordingly, SPIO-induced signal loss was unrelated to breakdown of the BBB that occurs immediately in this model (Dietrich et al., 1987; Van Bruggen et al., 1992) and lasts for at least 2 weeks, as evidenced by persistent gadolinium enhancement of the lesions (Schroeter et al., 2001; present study). By follow-up MRI scans in individual animals, we could further show that once macrophages have infiltrated a particular infarct zone, they did not further migrate.
The delayed macrophage invasion of photochemically induced cortical infarcts, as shown in the present study, is in full accordance with our previous histologic investigation (Schroeter et al., 1997), which used a macrophage-depletion paradigm and ED-1 immunoreactivity as a measure for the phagocytic activity of microglia and macrophages (Flaris et al., 1993). We had compared the number of ED1+ phagocytes in the border zone of infarcts between rats in which hematogenous macrophages had been depleted before by toxic liposomes and sham-treated rats with normal peripheral macrophage counts, and we found a major difference at day 6 but not at day 3. Taken together, both studies suggest that the major wave of macrophage infiltration is delayed by 5 to 6 days and occurs at a stage in which neuronal cell death is mostly completed (Braun et al., 1996; Isenmann et al., 1998). Thus it appears that infiltrating macrophages play a role in tissue remodeling rather than in neuronal injury (Jander et al., 2002).
With modern imaging techniques, the development of cerebral infarcts can be monitored (Neumann-Haefelin et al., 2000). However, various MR parameters, such as the apparent diffusion coefficient (ADC) and the T2 relaxation time, only reflect consecutive changes in the amount and mobility of protons but fail to delineate areas of specific cellular responses, such as astrogliosis, microglial activation, and macrophage recruitment (Schroeter et al., 2001). There have been previous attempts to specifically visualize macrophage responses in models of cerebral ischemia by use of ultrasmall superparamagnetic iron oxide (USPIO) particles, but the data have been inconclusive so far. Rausch and colleagues (2001) injected USPIO into five rats 5.5 hours after permanent MCAO and scanned these animals by MRI on days 1, 2, 4, and 7. A patchy signal loss was observed on T2-w images that declined from day 2 onward. No systematic histologic analysis of iron deposition during infarct development has been performed in parallel to MRI, and only some iron positive monocytes could be localized in the boundary zone on day 7, the end-point of their study. In a subsequent study using the transient MCAO model, the same authors could not detect signal loss on T2-weighted images in evolving ischemic lesions, and, again, iron staining on tissue sections revealed inconsistent results (Rausch et al., 2002). The lack of iron accumulation in transient MCAO, however, could be because of the fact that in this model microglial activation prevails over macrophage infiltration which is sparse (Lehrmann et al., 1997). In contrast with these previous studies, we always injected SPIO particles 24 hours before MRI scans were performed at various stages of infarct development, rather than applying iron particles at a fixed time point 5 hours after infarct induction. In conclusion, SPIO-enhanced MRI provides a reliable in vivo tool to follow macrophage invasion into focal ischemic infarcts and may help to further elucidate the role of these cells in stroke development, tissue remodeling, and repair.
Footnotes
Acknowledgment
C.K. and M.B. equally contributed to the study, which was supported by funds of the state of Bavaria. We thank Tanja Horn and Katharina Treutlein for expert technical assistance. M.B. is a stiftungsprofessor supported by a grant from Schering Deutschland GmbH, Berlin.
