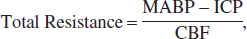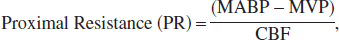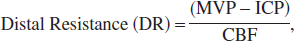Abstract
In an effort to localize the site at which increased resistance occurs after brain trauma, pial arteriole diameter and pressure were assessed after mild controlled cortical impact (CCI) injury in rats using an open cranial window technique. The authors tested the hypothesis that an increase in resistance accompanied by vasoconstriction occurs at the level of the pial arterioles within the injured cortex of the brain. At 1 hour after mild CCI injury, ipsilateral cerebral blood flow was significantly reduced by 42% compared with sham injury (n = 4; P < 0.05). Pial arteriole diameter and pressure remained unchanged. Resistance in the larger arteries (proximal resistance), however, was significantly greater after CCI injury (1.87 ± 0.26 mm Hg/[mL · 100 g · min]) compared with sham injury (0.91 ± 0.21 mm Hg/[mL · 100 g ·min]; P < 0.0001). Resistance in small vessels, arterioles, and venules (distal resistance) was also significantly greater after CCI injury (1.13 ± 0.05 mm Hg/[mL · 100 g · min]) compared with sham injury (0.74 ± 0.13 mm Hg/[mL · 100 g · min]; P = 0.0001). The authors conclude that, at 1 hour after mild CCI injury, changes in vascular resistance comprise a 53% increase in the resistance distal to the area of injury and, surprisingly, a 105% increase in resistance in the arteries proximal to the injury site.
Decreased cerebral blood flow (CBF) is a hallmark of the pathophysiology after traumatic brain injury (TBI) in clinical (Gobiet et al., 1975; Langfitt et al., 1977) and experimental settings (Yamakami and McIntosh, 1991; Yuan et al., 1988). This decrease in CBF does not itself produce permanent brain injury, but it does render the cortex more susceptible to secondary insults (Giri et al., 2000; Jenkins et al., 1989). For this reason, our laboratory has been interested in the mechanism responsible for the decrease in CBF after TBI.
We have previously shown (Golding et al., 1999) that local CBF decreases by approximately 50% at 1 hour after mild controlled cortical impact (CCI) injury. This decrease in CBF was localized to the cortical region directly beneath the impact site. According to Poiseuille's Law, which describes the flow of fluid through cylinders, a decrease in blood flow in the cortex would have to be accompanied by one or more of the following: (1) a decrease in the perfusion pressure, (2) a decrease in the radius of blood vessels, (3) an increase in the length of the cerebrovascular system, or (4) an increase in the viscosity of the blood. Changes in perfusion pressure can be ruled out because the components of perfusion pressure, namely, the mean arterial blood pressure (MABP) and intracranial pressure (ICP), are unchanged by mild CCI injury (Cherian et al., 1996). In addition, the length of the vascular system can be ruled out because it is fixed and would remain constant after injury. We therefore suspected that changes in the radius of blood vessels and viscosity were involved with the decreased CBF, with the former being most prominent because flow is directly related to the fourth power of the radius. An increase in vascular resistance may be an important contributing factor leading to the decrease in CBF that may increase the susceptibility toward secondary injury after TBI. In the present study, we tested the hypothesis that an increase in resistance, accompanied by vasoconstriction, would occur in the arterioles or venules directly within the injured cortex of the brain.
MATERIALS AND METHODS
Experiments were carried out in strict accordance with National Institutes of Health guidelines for the care and use of laboratory animals and were approved by the Animal Protocol Review Committee at Baylor College of Medicine.
Animal surgery
Adult male Long-Evans rats (300 to 380 g; n = 42) were maintained on rodent chow and water before surgery. Anesthesia was induced with a single intraperitoneal injection of sodium pentobarbital (60 mg/kg). Animals were tracheotomized (14-gauge catheter) and mechanically ventilated with room air supplemented with 100% oxygen (to maintain PO2 at approximately 100 mm Hg). The left femoral artery was cannulated with polyethylene tubing (PE-50; Intramedic, Sparks, MD, U.S.A.) to monitor systemic blood pressure and to enable sampling of blood to measure gases. The left femoral vein was cannulated with PE-50 tubing to allow sodium pentobarbital infusion (15 mg · kg−1 · h−1) during the course of the experiment. Rectal temperature was maintained at 37°C using a heating pad and a temperature controller (Digi-Sense; Cole-Parmer, Vernon Hills, IL, U.S.A.). The head of each rat was secured in a stereotaxic frame. The skull was exposed by a midline incision and the scalp (including the periosteum) and temporal muscle were reflected. Bleeding from the bone was controlled with bone wax.
Open cranial window preparation
An open cranial window was used to view the parietal cortex of rats after sham injury or mild CCI injury (Golding et al., 2000). A dam of dental acrylic was constructed surrounding the left parietal cortex with two ports (inflow and outflow). A 10-mm-diameter craniotomy was then performed in the center of the dam with a dental drill, using the bregma, lambda, and sagittal sutures as boundaries. To prevent thermal injury to the cortex, a stream of cool air was directed at the drilling site. Artificial cerebrospinal fluid (aCSF) was suffused over the surface of the brain at a rate of 2 mL/min. Under an operative microscope, the dura mater was incised with a 27-gauge needle. With the aid of fine forceps and microscissors, the dura—arachnoid complex was reflected around the pial arteriole that was to be visualized. Bleeding from the dural vessels was controlled by gel foam and occlusion of vessels with forceps.
Suffusion of artificial cerebrospinal fluid
The aCSF had the following composition (Baumbach et al., 1989): 131.9 mmol/L NaCl, 3.0 mmol/L KCl, 24.6 mmol/L NaHCO3, 6.7 mmol/L urea, 3.7 mmol/L glucose, 1.5 mmol/L CaCl2. The aCSF was continuously bubbled with 5% CO2, 10% O2, balanced in N2 and directly connected to a pump and custom-built heat exchanger. After equilibration, the CSF sampled from the dam had a pH of 7.3 ± 0.02, PCO2 of 37.9 ± 2.0 mm Hg, PO2 of 117.9 ± 3.6 mm Hg, and a temperature of 37.2 ± 0.3°C (n = 10).
Traumatic brain injury
Traumatic brain injury was applied using a CCI device (Dixon et al., 1991). This consists of a pneumatic impactor with a convex tip (diameter 9.5 mm), which is directed at 30° from the vertical. The impactor was positioned in the center of the 10-mm-diameter craniotomy (see “Open Cranial Window Preparation”). To achieve a mild level of injury, the CCI device was adjusted such that the impactor velocity was 3 m/s with a 50-millisecond duration and a 2-mm deformation. Impact velocity was monitored by a differential linear velocity transformer and recorded using physiologic acquisition software (PowerLab; AD Instruments, Castle Hill, Australia). Sham controls underwent identical surgical manipulations as the CCI-injured rats with the exception of injury.
Measurement of cerebral blood flow
Regional cerebral blood flow (rCBF) was measured using isopropyliodoamphetamine labeled with carbon 14 (14C-IPIA; ARC Inc., St. Louis, MO, U.S.A.) and quantitative autoradiography (Bryan et al., 1988; Giri et al., 2000). Male Long-Evans rats (285 to 365 g) were prepared as described previously (see “Animal Surgery”) with the following exceptions or additions. The right femoral vein and left femoral artery were catheterized (PE-50 tubing) for the infusion and sampling of blood, respectively. The tail artery was cannulated (22-G catheter) for monitoring MABP. Animals were randomly assigned into either the sham-injury (n = 3) or mild CCI-injury (n = 4) group. Ipsilateral CBF was measured 1 hour after either sham injury or mild CCI injury (as described previously).
Before measurement of CBF, 200 U of heparin was administered via the femoral vein. 14C-IPIA (0.13 μCi/g) was dissolved in 0.4 mL physiologic saline and infused as a bolus into the left femoral vein. Blood was withdrawn from the left femoral artery at a rate of 0.4 mL/min beginning at the time of 14C-IPIA infusion and continuing until the rat was decapitated 30 seconds later. The brain was rapidly removed and frozen in isopentane cooled with dry ice. Each brain was cut into 20-μm-thick cryostat sections (−18°C) that were mounted on glass slides. The glass slides containing the tissue sections along with calibrated standards were placed in contact with radiograph film. After a 15-day exposure, the film was developed to produce autoradiographic images. Concentrations of the tracer were determined by comparing the optical densities of the region of interest in the brain to that produced by the standards using an image analysis system (MCID; Imaging Research, St. Catherines, Ontario, Canada). The total radioactivity in the withdrawn blood was assessed after the sample was solubilized and counted in a liquid scintillation counter (Packard Instrument Co., Downers Grove, IL, U.S.A.). Cerebral blood flow was calculated as described elsewhere (Bryan et al., 1988; Giri et al., 2000).
Measurement of pial arteriole diameter
Ipsilateral pial arterioles with resting diameters of 28 to 59 μm were tested (n = 5). Vessel diameter was monitored using orthogonal polarization spectral imaging (Cytoscan; Cytometrics, Philadelphia, PA, U.S.A.) before and 1 hour after mild CCI injury. The technique is based on the illumination of the tissue with linearly polarized light while the reflected light is imaged through an orthogonally placed polarized analyzer (Thomale et al., 2001).
Measurement of pial artery pressure
Ipsilateral pial artery pressure was measured with a micropipette connected to a servo-null pressure-measuring device (Model 5A; Instrumentation for Physiology and Medicine, San Diego, CA, U.S.A.) (Baumbach and Heistad, 1983; Shapiro et al., 1971). Pial pressure was measured at 1 hour after either sham injury (n = 5) or mild CCI injury (n = 5) in pial arterioles with resting diameters of 28 to 59 μm. Before obtaining in vivo measurements, micropipettes were calibrated in vitro to ensure credibility and accuracy. Micropipettes connected to the servo-nulling pressure system accurately tracked changes in pressure in vitro (data not shown). Pial pressures were obtained separately in sham- and CCI-injured animals, to avoid possible compromising of vessels due to micropipette puncture.
Pipettes were made with a micropipette puller (Model P-57; Sutter Instruments, Novato, CA, U.S.A.) and sharpened to a beveled tip of 2 to 3 μm. A pipette filled with 2 mol/L NaCl was inserted into a pial arteriole using an Olympus (Tokyo, Japan) SZX zoom microscope (1.6x lens) and micromanipulator (Narishige, Tokyo, Japan). Micropipettes were calibrated with a mercury manometer before use in vivo. The servo-nulling system measures the resistance across the tip of the micropipette, where an interface exists between blood in the microvessels and NaCl in the micropipette. Because the blood and the NaCl solution have different conductances, the resistance across the tip will change as the interface moves in or out of the tip. After measuring the resistance in the tip, the servo-nulling system produces the appropriate pressure to maintain the resistance (or blood/NaCl interface) constant. The pressure required to maintain an unchanged resistance is measured and recorded. This pressure is equal to the microvascular pressure. Systemic and pial pressures were recorded using PowerLab software (AD Instruments). The diameter of the artery did not noticeably change after insertion of the micropipette into the lumen. Hemorrhage at the puncture site occurred on occasion; in such instances the experiment was terminated.
By measuring pial arteriole pressure, we were able to subdivide the vascular system into proximal and distal portions of the circulation (see “Data Analysis”). The proximal circulation comprised arteries upstream from the pial arterioles including the aorta, carotids, circle of Willis, middle cerebral artery, as well as the primary branch of the middle cerebral artery. The distal circulation comprised penetrating arterioles, capillaries, venules, and veins downstream from the pial arterioles.
Capillary perfusion
The number of perfused capillary segments was determined by infusing a fluorescent marker (Evans blue) that could be visualized in the capillaries (Gobel et al., 1990). Briefly, Evans blue dye (4% in saline at 1 mL/kg body weight) was injected into the right atrium through a catheter placed in the right jugular vein at 1 hour after sham injury (n = 5) or mild CCI injury (n = 5). Ten seconds after infusion the rat was decapitated, and the brain was rapidly removed and frozen in isopentane chilled between −40 and −50°C. Sections (3-μm thick) of the brain were obtained from the injured cortex and corresponding contralateral cortex using a cryostat (−20°C). The sections were placed on slides that were chilled to −20°C. The slides had been precoated with a mixture of egg foam and glycerol (1:1) to facilitate adherence of sections and dried between 35 to 40°C for 10 minutes before chilling. The slides were immediately immersed in acetone (−20°C) for 10 minutes. The transfer to the acetone was made in the cooling chamber of the cryostat to avoid thawing and diffusion of the Evans blue dye within the tissue sections. The slides (immersed in acetone) were removed from the cryostat and allowed to sit at room temperature for 1 to 1.5 hours.
To visualize the perfused capillaries, the slides were removed from the container and several drops of acetone were placed over the tissue section and immediately covered with a coverslip. The slide was placed on the stage of a fluorescence microscope (Nikon E600; 20x objective) and visualized using an excitation wavelength of 546 nm and fluorescent wavelength of 595 nm. Acetone was periodically added to the edge of the coverslip as the acetone under the cover slip evaporated. The number of perfused capillary segments for a given unit area was counted in 7 to 10 sections in the injured cortex and the uninjured cortex. The data were expressed as the number of capillary segments per mm2. Vessel segments larger than 15 μm in diameter were not counted.
Hematoxylin and eosin staining
In some animals, at 1 hour after sham CCI injury (n = 2) or mild CCI injury (n = 2), the aCSF was replaced with 10% formalin for 10 minutes. The animal was decapitated and the cerebral cortex within the craniotomy site was removed and placed in formalin to allow further fixation. The tissue was embedded in paraffin and 9-x m sections were cut and stained with hematoxylin and eosin. Sections were viewed on the stage of a Labophot-2 Nikon microscope (oil immersion, 100x objective).
Data analysis
Data are expressed as mean ± SD and statistical comparisons were performed using either a one-way analysis of variance (physiologic parameters, pial pressure, and resistance) or repeated measures analysis of variance (pial diameter). Post hoc comparisons were made using a Student—Newman—Keuls test. Differences were considered significant at P < 0.05.
Resistances proximal and distal to the pial arterioles, where pressure was directly measured, were calculated by the following equations (Baumbach and Heistad, 1983):
where MABP is mean arterial blood pressure, MVP the microvascular pressure measured in the pial arterioles, ICP the intracranial pressure, and CBF the cerebral blood flow. The ICP in sham and injured rats was assumed to be 9 mm Hg, as previously reported (Golding et al., 1999). Mild CCI injury as used in this study does not alter ICP (Golding et al., 1999). Equations 2 and 3 assume that the pressure in the arteriole being measured is representative of other arterioles in the vascular field for the given condition (sham injury or injury).
RESULTS
Arterial blood variables
The physiologic parameters including MABP, arterial pH, PCO2, and PO2 at 1 hour after sham injury or mild CCI injury are shown in Table 1. Similar values were observed before sham injury or mild CCI injury (data not shown).
Physiologic parameters in animals at 1 hour after sham and mild controlled cortical impact injury
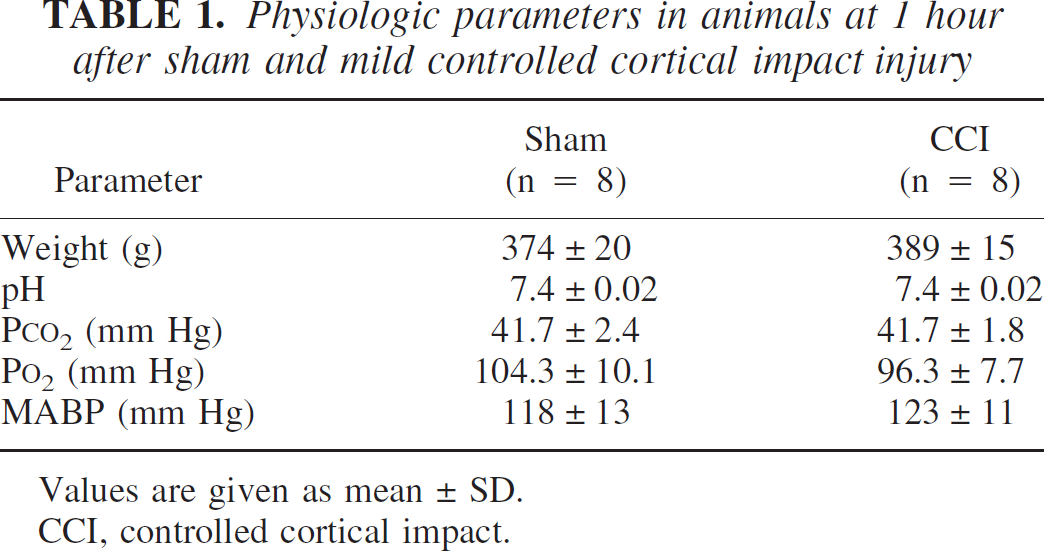
Values are given as mean ± SD.
CCI, controlled cortical impact.
Cerebral blood flow
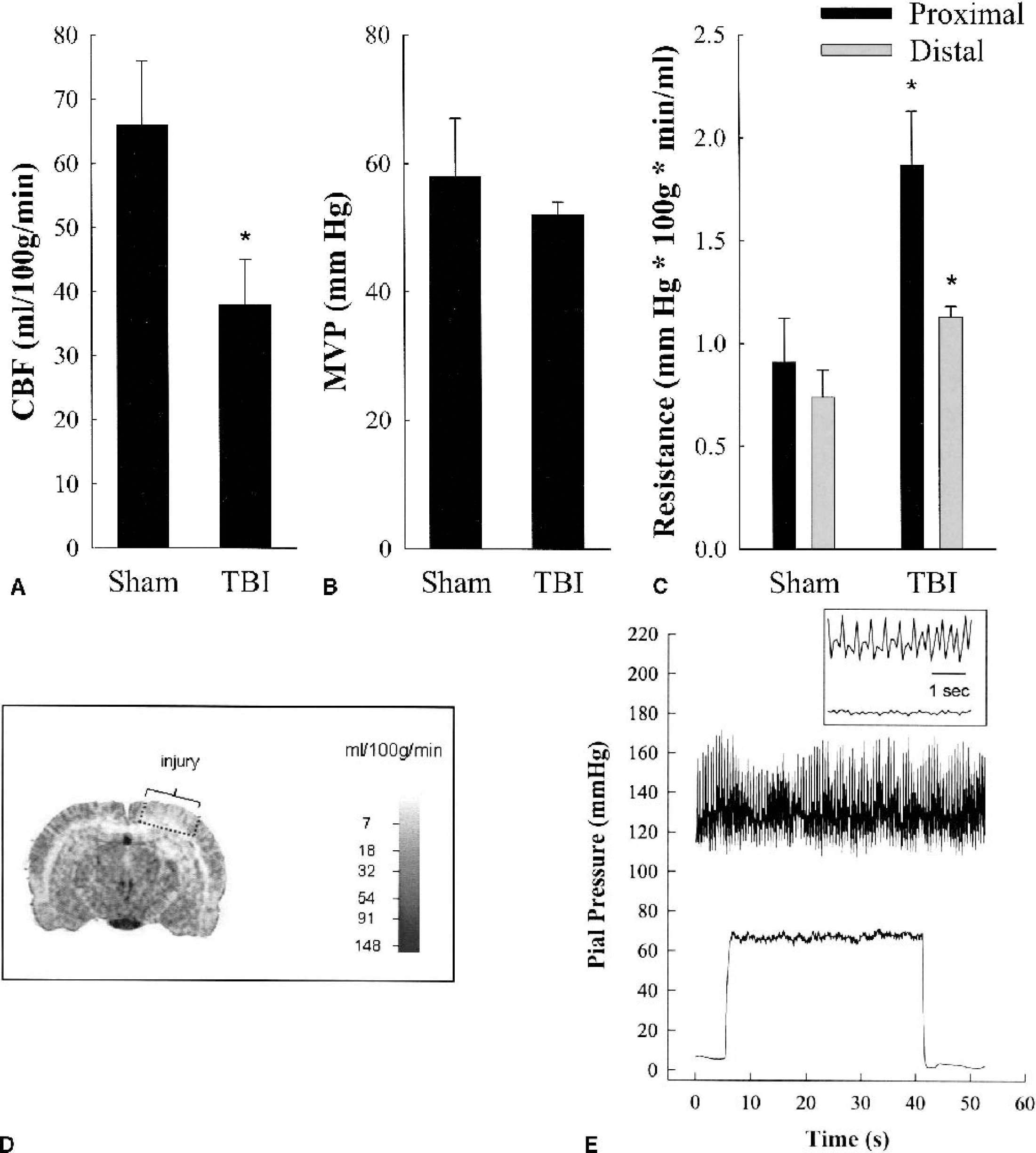
At 1 hour after sham injury, ipsilateral cortical rCBF was 66 ± 10 mL · 100 g−1 · min−1 (n = 3) as assessed by 14C-IPIA. At 1 hour after mild CCI injury, ipsilateral cortical rCBF was significantly reduced to 38 ± 7 mL · 100 g−1 · min−1 (n = 4; P <0.05 compared with sham; one-way ANOVA; see Figs. 4A and 4D). At this same time point, ipsilateral hippocampal and thalamic rCBF were 43 ± 19 mL · 100 g−1 · min−1 and 56 ± 22 · 100 g−1 · min−1, respectively, after sham injury and 39 ± 7 mL · 100 g−1 · min−1 and 61 ± 14 mL · 100 g−1 · min−1, respectively, after mild CCI injury. Therefore, the reduction in rCBF was specifically localized to the cortical region.
A cross-section of a pial arteriole after
Pial arteriole diameter
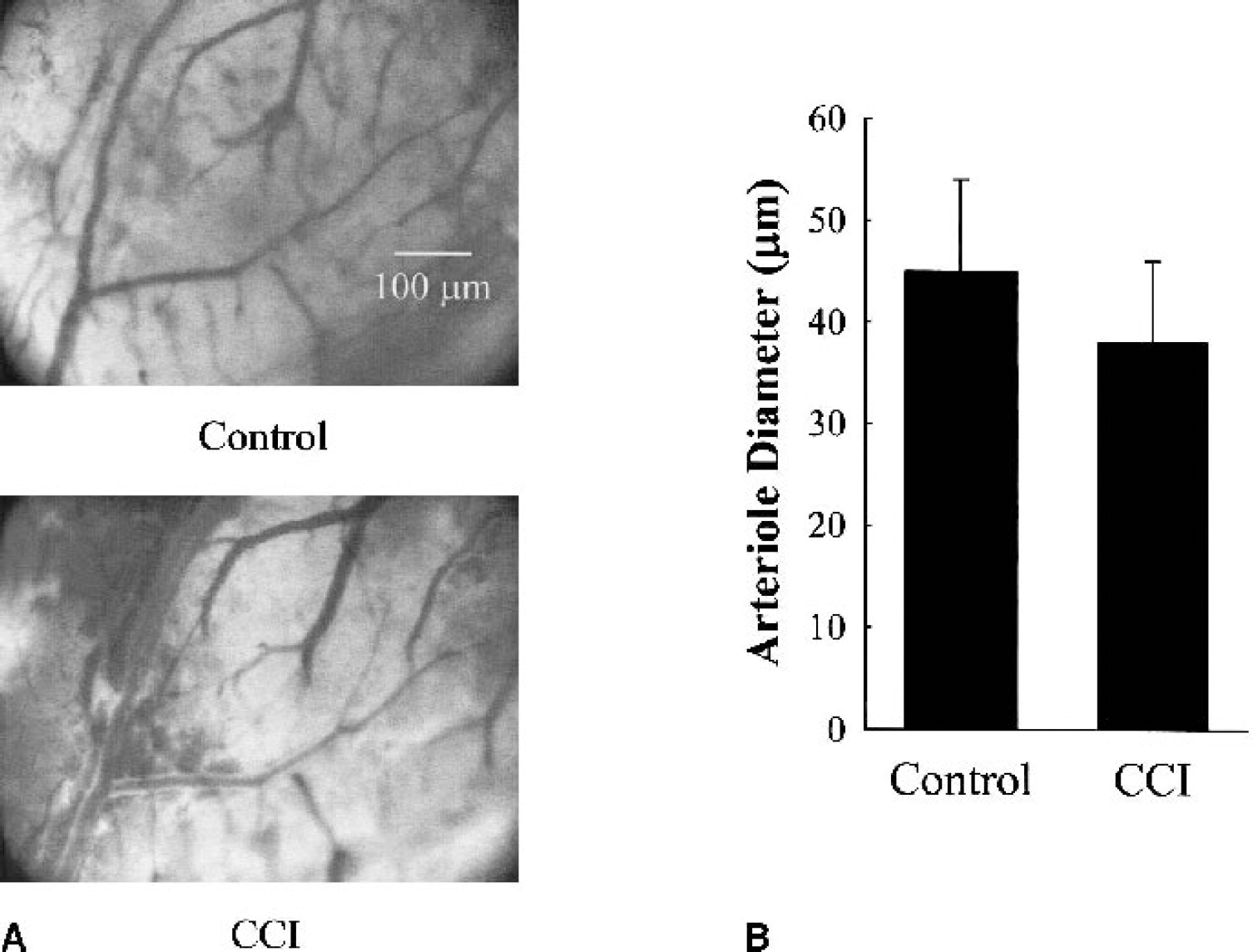
Figure 1A shows pial vessels before (control) and 1 hour after mild CCI injury. Note the appearance of extravascular blood after CCI injury. Some blood was diffuse (upper left), whereas other areas showed blood confined to borders adjacent to the arteriole. Thomale et al. (2002) have reported similar observations. We were able to visualize five arterioles both before and 1 hour after the injury. In some cases we were not able to find the same arteriole owing to excessive blood in the field of view. For those five arterioles, the diameter was 45 ± 9 μm before injury and 38 ± 8 μm 1 hour after injury (Fig. 1B; P = 0.23, paired one-way ANOVA). Even when nonpaired arterioles of the same branching order were compared before and after injury there was no difference in diameter (39 ± 14 μm before injury and 30 ± 18 μm after injury; n = 13 for each). Pial arteriole diameter change was not large enough to be statistically significant. We have previously shown (Golding et al., 2000) that, after sham injury, pial arteriole diameter is maintained.
(
Pial arteriole pressure and segmental resistances
As mentioned previously, CBF was 66 mL · 100 g−1 · min−1 after sham injury and 38 mL · 100 g−1 · min−1 after mild CCI injury. This represents an increase in total resistance to flow of 82% (Eq. 1; 1.65 mm Hg/[mL · 100 g · min] for sham and 3.0 mm Hg/[mL · 100 g · min] for CCI).
Figure 2E shows a typical pressure recording obtained from one animal. Pial arteriole pressure after sham and CCI injury was 58 ± 9 mm Hg and 52 ± 2 mm Hg, respectively (P = 0.08; one-way ANOVA; Table 2). These values are similar to those obtained by other investigators at this segment of the vasculature (Hajdu et al., 1993; Harper and Bohlen, 1984). The calculated proximal resistance (PR; Eq. 2) and distal resistance (DR; Eq. 3) are also shown in Table 2. Proximal resistance was significantly greater at 1 hour after CCI injury (1.87 ± 0.26 mm Hg/[mL · 100 g · min]) compared with sham injury (0.91 ± 0.21 mm Hg/[mL · 100 g · min]; P < 0.0001; one-way ANOVA). This represents an increase of 105% after CCI injury compared with sham injury. Studies in the rat have calculated PR in control rats to be 0.9 ± 0.3 mm Hg/(mL · 100 g · min) (Hajdu et al., 1993). Distal resistance was also significantly greater after CCI injury (1.13 ± 0.05 mm Hg/[mL · 100 g · min]) compared with sham injury (0.74 ± 0.13 mm Hg/[mL · 100 g · min]; P = 0.0001; one-way ANOVA). This is a 53% increase in resistance after injury. Although Hajdu et al. (1993) did not calculate DR, we calculate that—using their reported measurements and assuming an ICP of 9 mm Hg—DR would be 0.6, which is in line with our present findings in sham animals.
Bar graphs summarizing
Effect of sham and mild controlled cortical impact injury on segmental vascular resistance
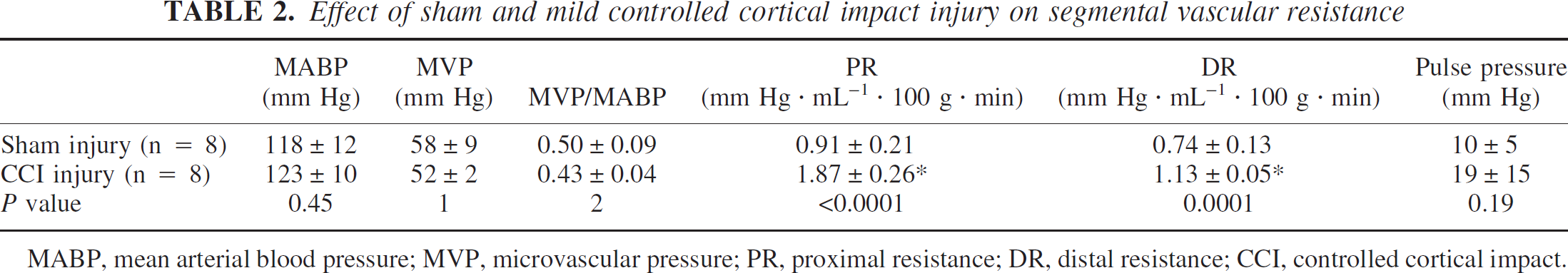
MABP, mean arterial blood pressure; MVP, microvascular pressure; PR, proximal resistance; DR, distal resistance; CCI, controlled cortical impact.
Capillary perfusion
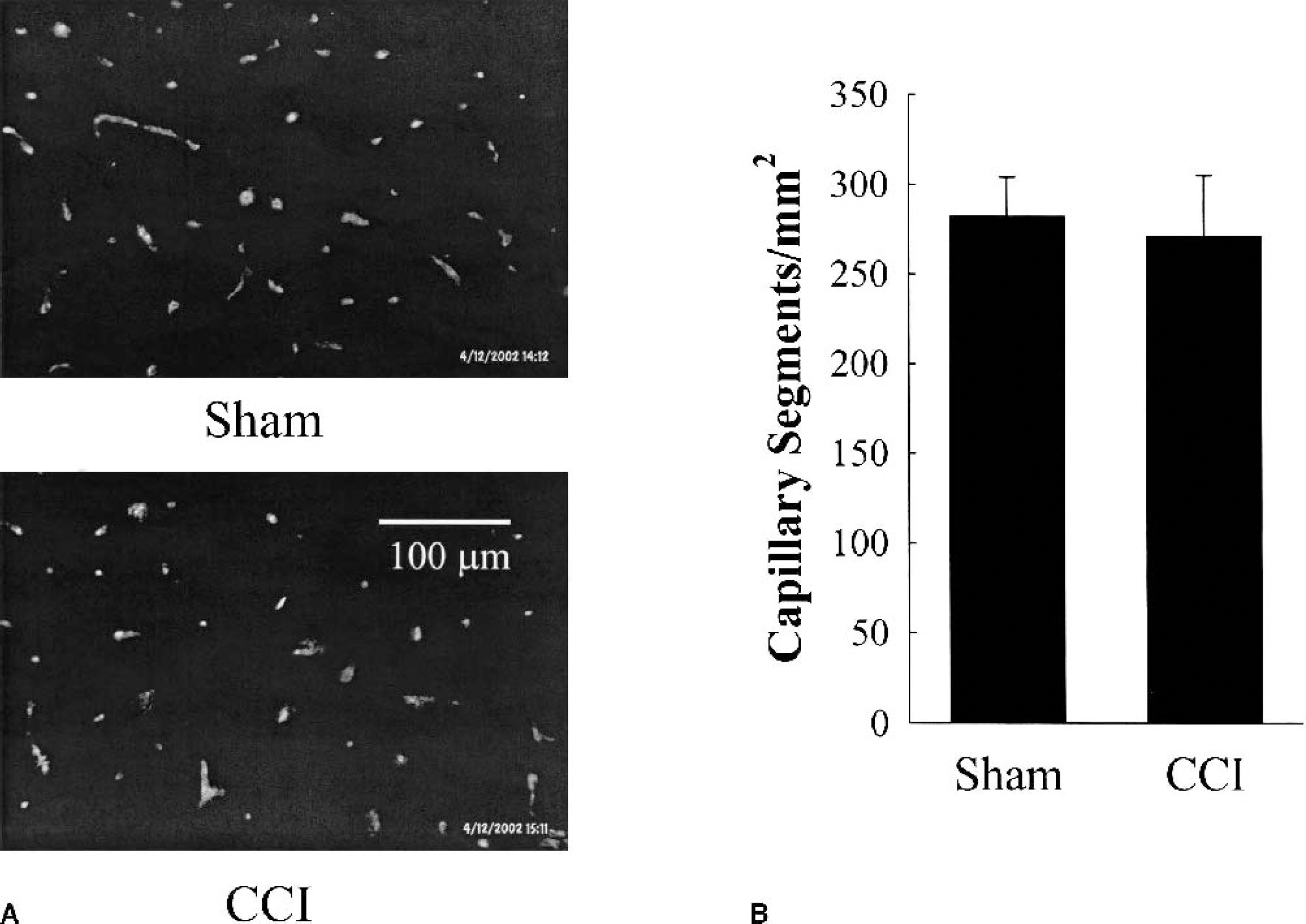
Perfused capillaries stained by Evans blue dye at 1 hour after sham and mild CCI injury are shown in Fig. 3A. Quantitative analysis (Fig. 3B) showed no differences in the number of perfused capillary segments at 1 hour after mild CCI injury (271 ± 34 capillary segments/mm2, n = 5) compared with sham injury (282 ± 22 capillary segments/mm2, n = 5).
(
Hematoxylin and eosin staining
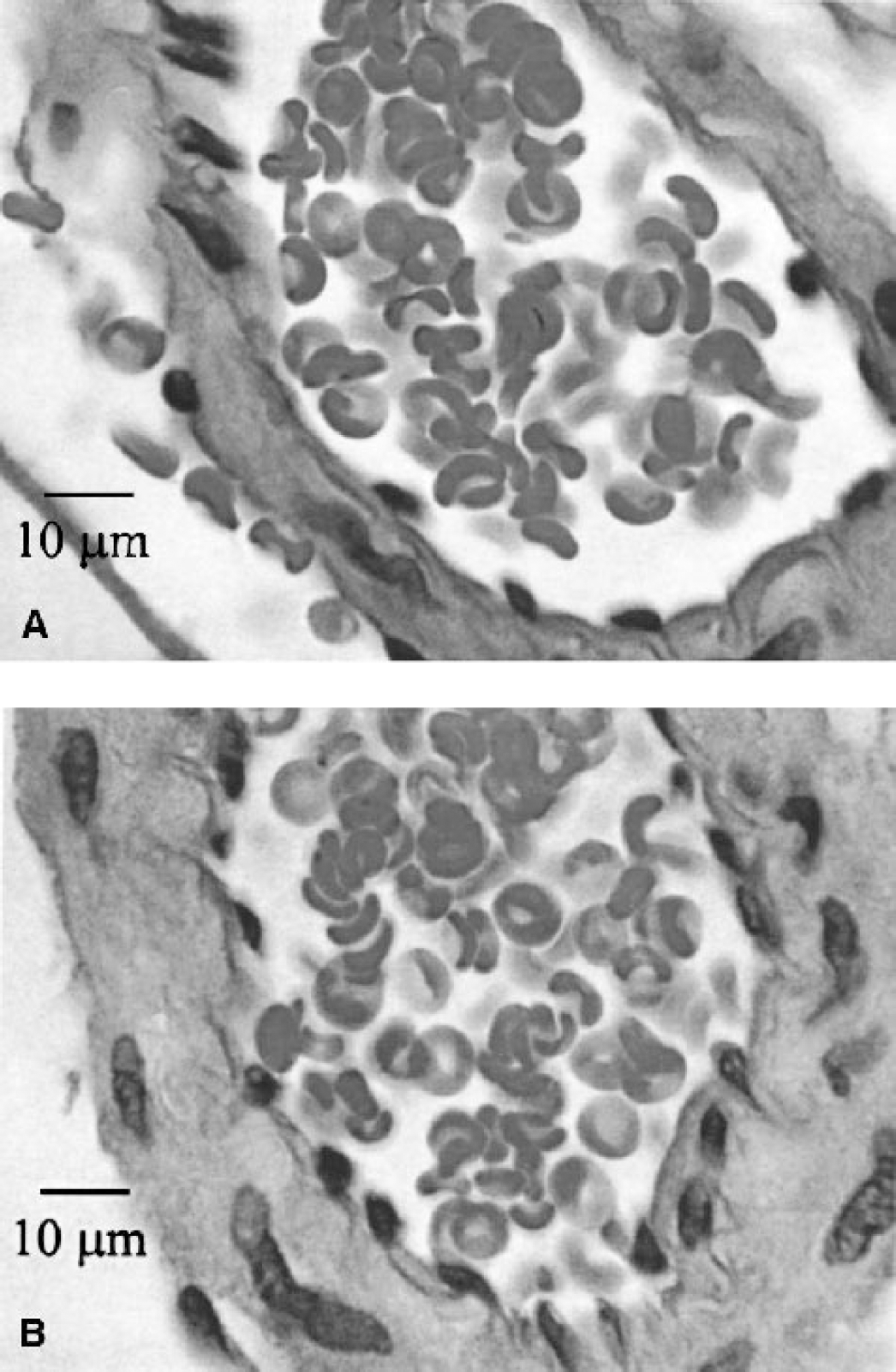
In pial arterioles directly beneath the impact site, hematoxylin and eosin staining of paraffin-embedded cross-sections showed endothelial cell integrity with no apparent swelling (Fig. 4B) when compared with arterioles in sham animals (Fig. 4A).
DISCUSSION
In the present study, we report that the decrease in CBF after mild CCI injury is a result of increased resistance of the vasculature. This is the first study to directly measure pial arteriole pressure under conditions after injury to the brain. From these measurements, we have calculated DR as having increased by 53% (from 0.74 to 1.13 mm Hg/[mL · 100 g · min]). Surprisingly, the PR increased by 105% (from 0.91 to 1.87 mm Hg/[mL · 100 g · min]). Therefore, the majority of the resistance increase occurred in vessels upstream from the injury site. Our findings correlate well with the reported increases in segmental resistance found after global ischemia in the cat (Schmidt-Kastner et al., 1987).
Pial arteriole diameter
Pial arteriole diameter was unchanged after mild CCI injury. On inspection of the literature, pial arteriole caliber has been reported as unchanged (DeWitt et al., 1986; Tompkins et al., 1999), constricted (Armstead and Kurth, 1994; Shibata et al., 1993), and dilated (Ellis et al., 1988) after TBI. These differences could be attributed to specifics regarding species, injury model, or severity of injury. Indeed, in support of the latter possibility, we did observe a reduced cell column in two animals. A similar phenomenon of streaming of red blood cells has been reported in the human microcirculation in close proximity to a brain tumor resection (Uhl et al., 2000). Endothelial swelling, or a dense layer of plasma proteins along the inner surface of the endothelium, was suggested to perhaps account for the underlying pathophysiology. Histologic analysis of these vessels using hematoxylin and eosin staining, however, did not indicate endothelial swelling (Fig. 4).
Distal resistance changes
Distal resistance increased from 0.74 to 1.13 mm Hg/(mL · 100 g · min). This increase in small-vessel resistance may be caused by the decrease in diameter of penetrating arterioles or the partial occlusion of capillaries or venules by leukocytes (Hartl et al., 1997), platelets (Dietrich et al., 1996; Hekmatpanah and Hekmatpanah, 1985), or endothelial swelling (Dietrich et al., 1984). In the present study, the complete occlusion of capillaries was not the primary mechanism contributing to the increase in distal resistance because the number of perfused capillary segments was not reduced after mild CCI injury (Fig. 3). Our data, however, do not preclude the idea that capillaries were partially occluded.
This occlusion or constriction in the distal circulation would act to reduce the effective radius of vessels and would represent the first step in a cascade leading to a further decrease in flow. When blood flow decreases, the shear rate also decreases, causing the viscosity to increase (Berne and Levy, 1988). The increase in viscosity would further decrease blood flow, according to Poiseuille's Law. The decreased shear rate could account for as much as a 74% increase in the resistance after pathologic situations (Lipowsky et al., 1980).
Proximal resistance
Proximal resistance increased from 0.91 to 1.87 mm Hg/(mL · 100 g · min). This represents a 105% increase in the large vessels supplying the injury site. This magnitude of resistance increase was surprising because it represents (for the most part) vessels that are presumably not damaged, upstream, and outside the area of injury. Furthermore, these vessels contributed more to the overall resistance increase after mild CCI than did the vessels within the injured cortex. In uninjured cortex the PR contributed to 45% of the total resistance to flow; 1 hour after mild CCI injury, however, PR accounted for approximately 61% of the total resistance to flow.
Why would large-vessel resistance increase when most of these vessels are located proximal to and outside the area of injury? We speculate that the large vessels are responding by recruiting the myogenic response as a compensatory mechanism to avoid excessive pressures in arterioles and venules. Cerebral vessels possess a characteristic whereby an increase in luminal pressure of a vessel is accompanied by a constriction (Golding et al., 1998). This phenomenon is termed the myogenic response and is considered to have a role in the autoregulatory response. The basic idea is that an increase in pressure would be accompanied by a constrictor response to increase resistance through that particular vessel segment, to maintain CBF constant over a broad range of pressure changes. The myogenic response has an added role: to protect small arterioles and capillaries from being exposed to pressures that could potentially damage the vessels. Such a protective mechanism is especially important in brain, which is encased in the cranial cavity. Unnecessary increases in capillary pressure would produce edema, brain swelling, and increased ICP. An increased ICP would further act to decrease cerebral perfusion pressure.
The clinical implications of this finding suggest that interventions that attempt to increase cerebral perfusion pressure by increasing MABP may not be beneficial as long as the myogenic response remains intact in the large vessels. We speculate that MVP after mild TBI would remain relatively constant over a wide pressure range owing to the myogenic response of uninjured upstream cerebral arteries. Raising or lowering the MABP (as long as it was above the lower autoregulatory limit (60 mm Hg) and below the upper autoregulatory limit (160 mm Hg)) would maintain MVP near constant in pial vessels. We have previously shown (Golding et al., 1999) that, after mild CCI injury, autoregulation was intact even though baseline flow was dramatically reduced. We speculate that it is the large-vessel myogenic response that is responsible for the maintenance of the autoregulatory response during mild CCI injury. After more severe head trauma, however, where autoregulation is completely abolished, the phenomenon would not hold true; rather, MVP would be directly related to MABP.
Capillary perfusion
The present study shows ipsilateral capillary patency at 1 hour after mild CCI injury, despite a significant decrease in regional CBF. The number of perfused capillary segments after sham injury is consistent with previous studies (Gobel et al., 1990). Our results after mild CCI injury are also similar to the situation after ischemia—reperfusion injury where capillary patency was maintained (Li et al., 1998; Theilen et al., 1994). Although the number of perfused capillaries was found to be unaffected after injury, we cannot discount the possibility that capillary flow is compromised. Indeed, it is possible that the number of perfused capillaries is maintained with a reduced velocity of perfusion.
In conclusion, our data suggest that the increased resistance after CCI injury does not occur at the site of the pial arterioles. Rather, the vascular changes consist of a 53% increase in the DR and an unexpected 105% increase in PR. The increase in PR has profound clinical implications and must be considered by the intensivist when caring for the head-injured patient.
Footnotes
Acknowledgments
The authors thank Tom Gerhold and Dr. Gary Baumbach (University of Iowa), for their invaluable guidance with the servo-nulling pressure system; and Drs. Herbert Lipowsky (Pennsylvania State University), Johannes Vogel and Prof. Wolfgang Kuschinsky (University of Heidelberg), for their many helpful discussions.