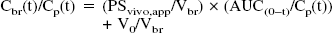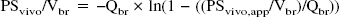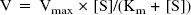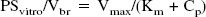Abstract
Although creatine plays a pivotal role in the storage of phosphate-bound energy in the brain, the source of cerebral creatine is still unclear. The authors examined the contribution made by the creatine transporter (CRT) at the blood–brain barrier in supplying creatine to the brain from blood. An in vivo intravenous administration study suggested that creatine is continuously transported from the blood to the brain against the creatine concentration gradient that exists between brain and blood. Conditionally immortalized mouse brain capillary endothelial cells (TM-BBB) exhibited creatine uptake, which is Na+ and Cl− dependent and inhibited by CRT inhibitors, such as β-guanidinopropionate and guanidinoacetate. Northern blot and immunoblot analyses demonstrated that CRT is expressed in TM-BBB cells and isolated mouse brain microvessels. Moreover, high expression of CRT was observed in the mouse brain capillaries by confocal immunofluorescent microscopy. These results suggest that CRT plays an important role in supplying creatine to the brain via the blood–brain barrier.
Keywords
Creatine is an essential molecule in cellular ATP homeostasis, particularly in tissues subject to high metabolic demands, such as brain and muscle. Oral administration of creatine protects the neurons in animal models of amyotrophic lateral sclerosis (ALS) and Huntington and Parkinson disease (Klivenyi et al., 1999; Matthews et al., 1998, 1999), and it is important to elucidate the mechanisms governing the neuroprotective effects of creatine as a high-energy phosphate buffering system after oral administration.
Although creatine is synthesized in the liver and pancreas from L-arginine and glycine (Walker, 1979), the creatine concentration in the mammalian brain is 4- to 20-fold greater than that in the liver (Marescau et al., 1992). Nevertheless, oral administration of 20 g creatine per day for 4 weeks produces only about a 9% increase in total creatine in human brain (Dechent et al., 1999). Using in situ hybridization, Braissant et al. (2001) suggested creatine synthesis in the brain because of mRNA expression of the enzymes involved in creatine synthesis. In contrast, creatine is transported to muscle cells from the circulating blood via a creatine transporter (CRT) (Murphy et al., 2001).
The blood–brain barrier (BBB) separates the circulating blood from the brain interstitial fluid and regulates the supply of nutrients to the brain from the circulating blood (Cornford, 1985). It is possible that CRT is expressed at the BBB and mediates the passage of creatine from the blood to the brain. Oral creatine supplementation is an effective treatment for creatine-deficient patients (Stöckler et al., 1994, 1996), and the creatine concentration in the brain appears to be dependent on the BBB function and the blood concentration of creatine. It is therefore important to identify the origin of the creatine in the brain for the treatment of such neurodegenerative diseases, since creatine supplementation appears to have a neuroprotective effect in animal models of ALS and Huntington and Parkinson disease (Klivenyi et al., 1999; Matthews et al., 1998, 1999).
We report evidence supporting the hypothesis that creatine in the brain is transported from the blood across the BBB. The characteristics and functions of creatine transport at the BBB were examined in vivo and in an in vitro BBB model. The localization of CRT at the BBB was analyzed by Northern blot and immunohistochemical studies. The identification and characteristics of the creatine transporter at the BBB will increase our understanding of how creatine is supplied to the brain.
MATERIALS AND METHODS
Animals
Adult male ddY mice (25 to 30 g) and female New Zealand White rabbits were purchased from SLC Ltd. (Shizuoka, Japan). The Animal Care Committee of Graduate School of Pharmaceutical Sciences, Tokyo University, approved all experiments.
Determination of [14C]creatine uptake in vivo
After mice were anesthetized by intramuscular injection of ketamine-xylazine, 3 μCi [14C]creatine (55 mCi/mmol; American Radiolabeled Chemicals, St. Louis, MO, U.S.A) dissolved in 50 μL saline solution was administrated via the tail vein, and blood samples were collected at predetermined time points. Mice were killed and tissues were excised immediately. All samples were weighed and dissolved in Soluene-350 (Packard, Meriden, CT, U.S.A.) at 55°C for 5 hours. The radioactivity in each sample was measured by liquid scintillation counting. As an index of the tissue distribution characteristics of creatine, the apparent tissue-to-plasma concentration ratio (Kp,app) was used. This ratio was defined as the amount of [14C]creatine per gram tissue divided by that per milliliter plasma, calculated over the time period of the experiment (Yang et al., 1997). The apparent influx permeability clearance of [14C]creatine was determined by integration plot analysis (Yang et al., 1997). In brief, the brain uptake rate of [14C]creatine can be described by the following equation:
The parenchymal and capillary fractions of mouse brain were separated by a modification of the procedure reported by Triguero et al. (1990). Briefly, the cerebrum was excised 1 hour after [14C]creatine injection and homogenized using extra cellular fluid (ECF) buffer (122 mmol/L NaCl, 3 mmol/L KCl, 0.4 mmol/L K2HPO4, 25 mmol/L NaHCO3, 1.4 mmol/L CaCl2, 1.4 mmol/L MgSO4, 10 mmol/L D-glucose, 10 mmol/L HEPES [pH 7.4], and 290 mOsm/kg) in a Potter-Elvehjem homogenizer. The homogenate was added to the same volume of 32% dextran and centrifuged at 4,500 g at 4°C for 15 minutes. The pellet containing capillaries and the supernatant, which represents the brain parenchyma, were carefully separated and the radioactivity of each was measured.
High-performance liquid chromatography analysis
The metabolism of [14C]creatine in the brain after intravenous injection was determined by high-performance liquid chromatography (HPLC). One hour after intravenous injection (10 μCi/50 μL saline), the cerebrum was excised and then dipped in liquid nitrogen. The frozen sample was homogenized in 0.3 mol/L HClO4 and centrifuged at 12,550 g at 4°C for 5 minutes. The supernatant was neutralized with KHCO3 and centrifuged at 12,550 g at 4°C for 5 minutes. The supernatant (200 μL) was subjected to HPLC using a system equipped with a Capcell Pak C18 UG120 (Shiseido, Tokyo, Japan) column. The mobile phase consisted of 14.7 mmol/L KH2PO4 and 1.15 mmol/L tetrabutylammonium phosphate (pH 5.0) at a flow rate of 1.0 mL/min. The eluent was collected in vials and the radioactivity in each fraction was determined by liquid scintillation counting.
[14C]Creatine uptake by TM-BBB4 cells
The conditionally immortalized mouse brain capillary endothelial cell line (TM-BBB4), which was established from a transgenic mouse harboring temperature-sensitive simian virus 40 large T-antigen gene, was used as an in vitro BBB model (Hosoya et al., 2000; Terasaki and Hosoya, 2001). TM-BBB4 cells (passage number 18–23) were cultured at 33°C in Dulbecco modified Eagle medium (Nissui Pharmaceutical Co., Tokyo, Japan) under 5% CO2/air as described previously (Hosoya et al., 2000). For the uptake study, TM-BBB4 cells were seeded on a 24-well plate (Iwaki, Tokyo, Japan) at a density of 3 × 105 cells/cm2 and cultured for 24 hours at 33°C. The [14C]creatine uptake by TM-BBB4 cells was measured as described previously (Hosoya et al., 2000). In brief, after discarding the culture medium, TM-BBB4 cells were washed with ECF buffer at 37°C. Uptake was initiated by applying 200 μL ECF buffer containing 0.1 μCi [14C]creatine (9.1 μmol/L) in the presence or absence of inhibitors at 37°C. Na+-free ECF buffer was prepared by equimolar replacement of NaCl and NaHCO3 with N-methylglucamine chloride and KHCO3, respectively. Cl−-free ECF buffer was prepared by equimolar replacement of NaCl, KCl, and CaCl2 with sodium gluconate, potassium gluconate, and calcium gluconate, respectively. After a predetermined time period, uptake was terminated by removing the solution and cells were immersed in ice-cold ECF buffer. The cells were then solubilized in 200 μL 5 mol/L NaOH, and the radioactivity was measured in a liquid scintillation counter. The protein content of cells was determined using a DC protein assay kit (Bio-Rad, Hercules, CA, U.S.A.) with bovine serum albumin as a standard. The degree of uptake by the cells was estimated from the radioactivity in the sample and expressed as either the cell-to-medium ratio per milligram cell protein (μL/mg protein) or as a percentage of the control. The [14C]inulin (PerkinElmer Life Sciences, Boston, MA, U.S.A.) uptake study was performed to estimate the volume of adhering water. The resulting cell-to-medium ratio was 0.1 to 0.2 μL/mg protein and 100-fold less than that of [14C]creatine. Therefore, adhering water was ignored when calculating the cell-to-medium ratio of [14C]creatine.
Data analysis
For kinetic studies, the Michaelis-Menten constant (Km) and the maximal rate (Vmax) of creatine uptake were calculated from the following equation using the nonlinear least-square regression analysis program, MULTI (Yamaoka et al., 1981):
To estimate the BBB uptake clearance (PSvitro) from the in vitro uptake study (Fig. 2C), the amount of protein in TM-BBB4 cells is 17.6 cm2/mg, and the surface area of the cerebrum capillary is 100 cm2/g brain (Pardridge et al., 1990b). The BBB uptake clearance per gram brain (PSvitro/Vbr) of creatine was estimated according to the following equation:
All data represent mean ± SD values. An unpaired, two-tailed Student's t-test was used to determine the significance of differences between two group means. Statistical significance among means of more than two groups was determined by a one-way analysis of variance followed by the modified Fisher least-square difference method.
cDNA cloning of mouse creatine transporter cDNA
The mouse CRT cDNA fragment was isolated by reverse transcription–polymerase chain reaction (RT–PCR) from TM-BBB4 cell total RNA. Total RNA was prepared using Trizol reagent according to the manufacturer's instructions (Invitrogen, Carlsbad, CA, U.S.A.). Primers were designed from rat CRT/CHOT1 (GenBank Accession Number X66494) as the following sequences: sense, 5′ -GAAATGGTGCTGGTCCTTCTTCAC-3′ (2193–2216); antisense, 5′-GTCACATGACACTCTCCACCACGA-3′ (2247–2545). The amplified product was sequenced and its sequence has over 90% homology with that of rat, rabbit and human CRT. This mouse cDNA fragment was used as a probe, and the TM-BBB4 cDNA library was screened by plaque hybridization. The TM-BBB4 cDNA library was constructed using a λ-Zap cDNA library constructing kit (Stratagene, La Jolla, CA, U.S.A.). The isolated mouse CRT cDNA was subcloned into pBluescript SKII+ (Stratagene) by in vivo excision, and sequenced from both directions using a DNA sequencer (model 4200; Li-COR, Lincoln, NE, U.S.A.).
Northern blot analysis
Total RNA was prepared from adult mouse tissues, isolated brain microvessels, and TM-BBB4 cells using Trizol reagent. Fifteen micrograms total RNA was electrophoresed on a formaldehyde (18%) and agarose (1%) gel and electrotransferred to a nylon membrane (GeneScreen Plus; PerkinElmer Life Sciences). Blots were prehybridized (5x SSPE, 50% formamide, 5x denhalt solution, 0.5% SDS, and 20 μg/mL salmon sperm DNA) for 2 hours at 42°C. A mouse CRT cDNA fragment (corresponding to nucleotide residues 133–2401 based on the nucleotide sequence of mouse CRT cDNA isolated from TM-BBB4 cells) was labeled with [32P]γ-dCTP using a random primer labeling kit (TaKaRa, Shiga, Japan). The labeled probe was then incubated with blots in prehybridization buffer overnight at 42°C, followed by washes in 2x SSPE, 0.1% SDS (2 × 10 minutes, room temperature), 1x SSPE, 0.1% SDS (2 × 20 minutes, 65°C) and 0.1x SSPE, 0.1% SDS (1 × 20 minutes, 65°C), and exposed to films (X-OMAT; Kodak, Rochester, NY, U.S.A.). The blots were also probed with [32P]β-actin cDNA as an internal control.
Antibody preparation
Polyclonal antibody was raised against the mouse CRT C-terminal region (CRT-c; corresponding to nucleotide residues 1736–1909 based on the nucleotide sequence of mouse CRT cDNA isolated from TM-BBB4 cells). CRT-c was amplified from the cloned mouse CRT cDNA by RT–PCR, and inserted into pGEX4T-2 expression vector (Amersham Biosciences, Piscataway, NJ, U.S.A.). CRT-c protein conjugated with glutathione S-transferase (CRT-c/GST) was expressed in the BL21 pLys strain of Escherichia coli (Stratagene) with induction by isopropyl-1-thio-β-D-galactoside. Because bacterially expressed CRT-c/GST is insoluble, a soluble fraction was obtained by solubilization in 50 mmol/L Tris-HCl (pH 8.0), 8 mol/L urea, 1 mmol/L DTT, 1 mmol/L EDTA, and stepwise dialysis against 10 mmol/L sodium phosphate buffer (pH 7.5), 10 mmol/L EDTA, 10 mmol/L EGTA, and 1% Triton X-100, and used for both immunization and affinity gel preparation. CRT-c fusion protein was emulsified with Freund's complete adjuvant (Difco, Detroit, MI, U.S.A.) and injected subcutaneously into a female New Zealand White rabbit (70 μg CRT-c per injection) at intervals of 2 to 4 weeks. Two weeks after the sixth injection, the immunoglobulin fraction was purified from antisera using protein-G Sepharose (Amersham Biosciences). Immunoglobulins specific to CRT-c were affinity purified using CNBr-activated Sepharose 4B (Amersham Biosciences) carrying CRT-c free of GST.
Immunoblot analysis
Tissue and cell lysates of adult mouse brain, isolated brain microvessels, and TM-BBB4 cells were prepared in buffer containing 10 mmol/L HEPES-NaOH (pH 7.4), 250 mmol/L sucrose, 1 mmol/L EDTA, 1 mmol/L phenylmethylsulfonyl fluoride, and a protease-inhibitor cocktail (Sigma Chemical Co., St. Louis, MO, U.S.A.) by nitrogen cavitation (800 psi at 4°C for 30 minutes). To prepare subcellular protein fractions, the lysates were centrifuged at 1,000 g for 10 minutes to remove nuclei. Then, the enucleated supernatants were centrifuged at 10,000 g for 10 minutes and the supernatants were collected. These supernatants were successively centrifuged at 100,000 g for 1 hour, and a crude membrane fraction was obtained from the pellets. To prepare a microsomal fraction, the lysates were enucleated by centrifugation at 1,000 g for 10 minutes. Then, the enucleated supernatants were centrifuged at 100,000 g for 1 hour, and a microsomal fraction was obtained from the pellets. Protein concentrations were determined by a DC protein assay kit (Bio-Rad). Protein samples (20 μg) were fractionated on 7.5% SDS-polyacrylamide gel and electrotransferred to a nitrocellulose membrane. Blotted membranes were incubated with affinity-purified anti-CRT antibody at 1 μg/mL in blocking agent solution (Block Ace; Dainippon Pharmaceutical, Osaka, Japan), and visualized with an enhanced chemiluminescence kit (Supersignal west pico chemiluminescent substrate; Pierce, Rockford, IL, U.S.A.).
Immunohistochemical analysis
Under deep pentobarbital anesthesia (100 mg/kg body weight, intraperitoneally), the brains of adult mice were perfused transcardially with 4% paraformaldehyde in 0.1 mol/L sodium phosphate buffer (pH 7.2). Cryostat sections (20 μm in thickness; CM1900; Leica, Heidelberg, Germany) and microslicer sections (50 μm; VT1000S; Leica) were prepared for immunofluorescence. Sections were incubated with rabbit anti-CRT antibody (2 μg/mL) overnight, and then incubated with FITC-conjugated secondary antibody (Jackson ImmunoResearch, West Grove, PA, U.S.A.) for 2 hours at room temperature. The immunofluorescent-stained sections were viewed under a confocal laser scanning microscope (Fluoview; Olympus, Tokyo, Japan).
RESULTS
Blood-to-brain transport of creatine across the blood–brain barrier
In order to determine whether creatine is transported into the brain, the in vivo tissue accumulation of creatine was examined for 24 hours after intravenous injection of [14C]creatine in mice. The apparent tissue-to-plasma concentration (Kp,app) of the cerebrum and cerebellum was 30.8 ± 4.3 and 35.1 ± 8.3 mL/g tissue, respectively (Fig. 1A). The Kp,app value of the cerebrum was 1.7-and 7.7-fold higher than that of the kidney and liver, respectively, whereas it was 2.7-fold lower than that of skeletal muscle. These results demonstrate that creatine is transported to and accumulated in the brain as well as skeletal muscle.
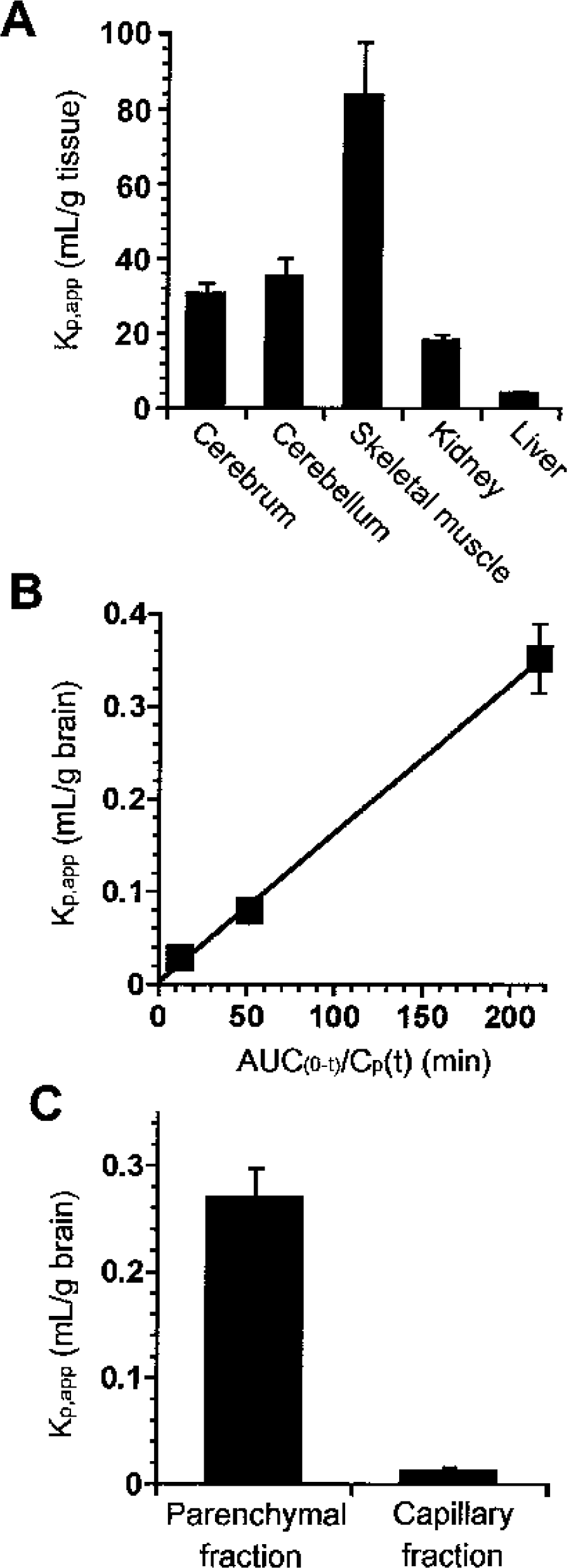
Blood-to-brain transport of creatine.
The apparent influx permeability clearance of [14C]creatine from the circulating blood to the brain through the BBB was determined by measuring the amount of [14C] activity in the brain by integration plot analysis (Fig. 1B). The influx permeability clearance per gram brain (PSvivo/Vbr) of [14C]creatine was determined to be 1.61 ± 0.15 μL/(min · g brain) from the slope representing the apparent influx permeability clearance using Eq. 1 and a correction for the cerebral plasma flow rate using Eq. 2. Moreover, HPLC analysis revealed that over 90% of the recovered [14C] activity was detected at the same retention time as creatine and no metabolites were detected 1 hour after intravenous injection (data not shown). These results indicate that [14C]creatine is transported in intact form from the blood to the brain across the BBB, but does not accumulate in the brain capillary endothelial cells, since the [14C] activity in the capillary fraction was only 5% that in the brain parenchymal fraction (Fig. 1C).
Na+- and Cl−-dependent carrier-mediated transport of creatine
To characterize creatine transport at the BBB, TM-BBB4 cells were used as an in vitro mouse BBB model (Hosoya et al., 2000; Terasaki and Hosoya, 2001). [14C]Creatine uptake by TM-BBB4 cells exhibited a time-dependent increase and was linear for at least 10 minutes, with a cell-to-medium ratio of 32.9 ± 8.6 μL/mg protein (Fig. 2A). In contrast, the absence of either Na+ or Cl− reduced the [14C]creatine uptake by more than 90% (Fig. 2B). [14C]Creatine uptake by TM-BBB4 cells took place in a concentration-dependent manner with a Km of 16.2 ± 2.8 μmol/L and a Vmax of 1.05 ± 0.05 nmol/(mg protein · 10 min) (Fig. 2C), indicating that creatine is transported by an Na+- and Cl−dependent carrier-mediated transport system in TM-BBB4 cells. To characterize [14C]creatine uptake by TM-BBB4 cells, we examined the effects of several inhibitors of CRT and other compounds on [14C]creatine uptake by TM-BBB4 cells (Table 1). β-Guanidinopropionate and guanidinoacetate, potent inhibitors of CRT, markedly inhibited [14C]creatine uptake by 95.5% and 69.8%, respectively, whereas phosphocreatine, creatinine, L-arginine, glycine, γ-aminobutyric acid (GABA), and choline did not have any significant effect. This type of inhibition of [14C]creatine uptake suggests that CRT is involved in the uptake process by TM-BBB4 cells.
Effect of several compounds on [14C]creatine uptake by TM-BBB4 cells

[14C]Creatine uptake (9.1 μmol/L) by TM-BBB4 cells was performed in the absence (control) or presence of 1-mmol/L inhibitors at 10 minutes and 37°C. Each value represents the mean ± SD values (n = 3 to 4).
P < 0.01, significantly different from control.
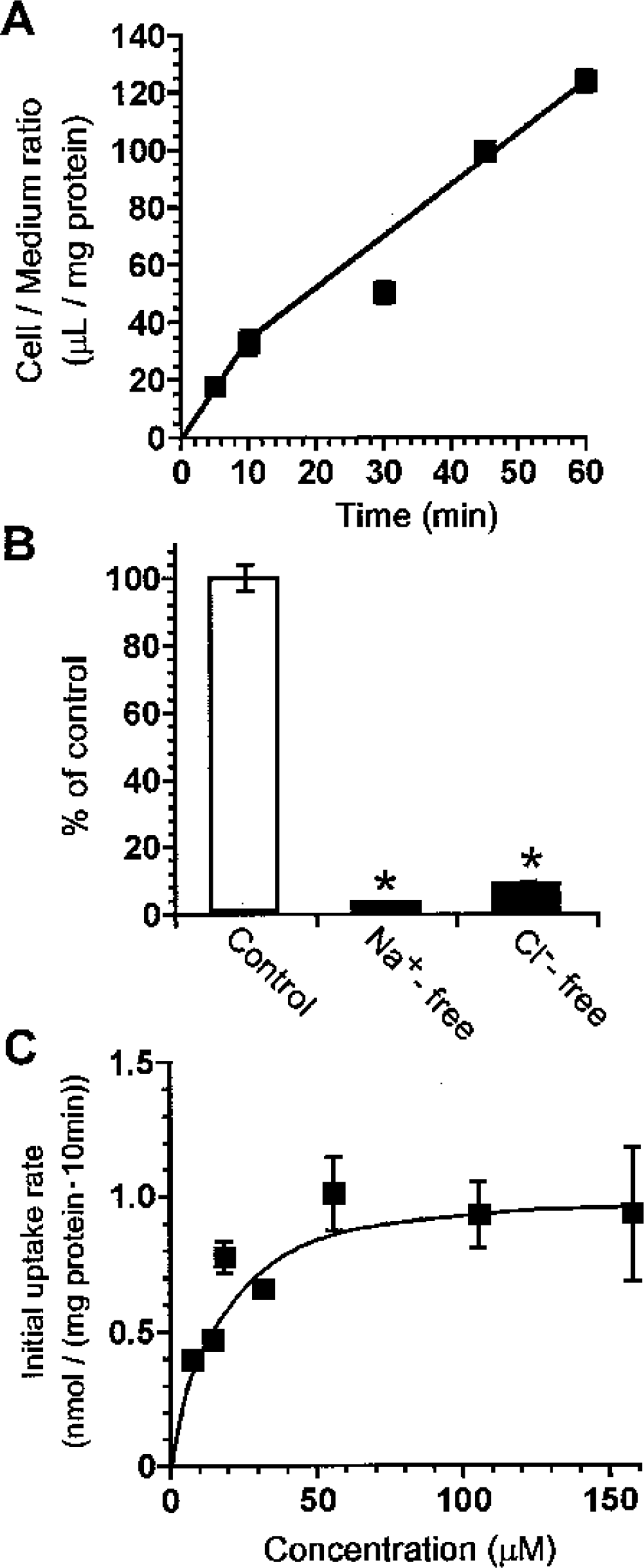
Characteristics of creatine transport at the blood–brain barrier.
Expression of creatine transporter
Because mouse CRT has not been cloned yet, the mouse CRT cDNA was isolated from the TM-BBB4 cell cDNA library. The mouse CRT cDNA includes an open reading frame of 1,905 nucleotides, which correspond to 635 amino acid residues (Fig. 3). Its deduced amino acid sequence is 99.2%, 98.7%, and 97.6% identical with rat, human, and rabbit CRT, respectively (Guimbal et al., 1993; Saltarelli et al., 1996; Sora et al., 1994). Northern blot analysis was performed to examine the expression of CRT mRNA in brain microvessels and TM-BBB4 cells (Fig. 4). Bands at 3.7 kb, corresponding to mouse CRT cDNA, were detected in TM-BBB4 cells, isolated mouse brain microvessels as used in vivo BBB, whole mouse brain, and mouse skeletal muscle as a positive control, whereas no similar band was detected in mouse liver.

Nucleotide and deduced amino acid sequences of mouse creatine transporter (CRT). The deduced amino acid sequence is shown below the nucleotide sequence. The C-terminal region (CRT-c) used for preparing antimouse CRT antibody is underlined, and the asterisk shows the termination codon. A poly(A) addition signal is boxed. GenBank Accession Number: AB077327.
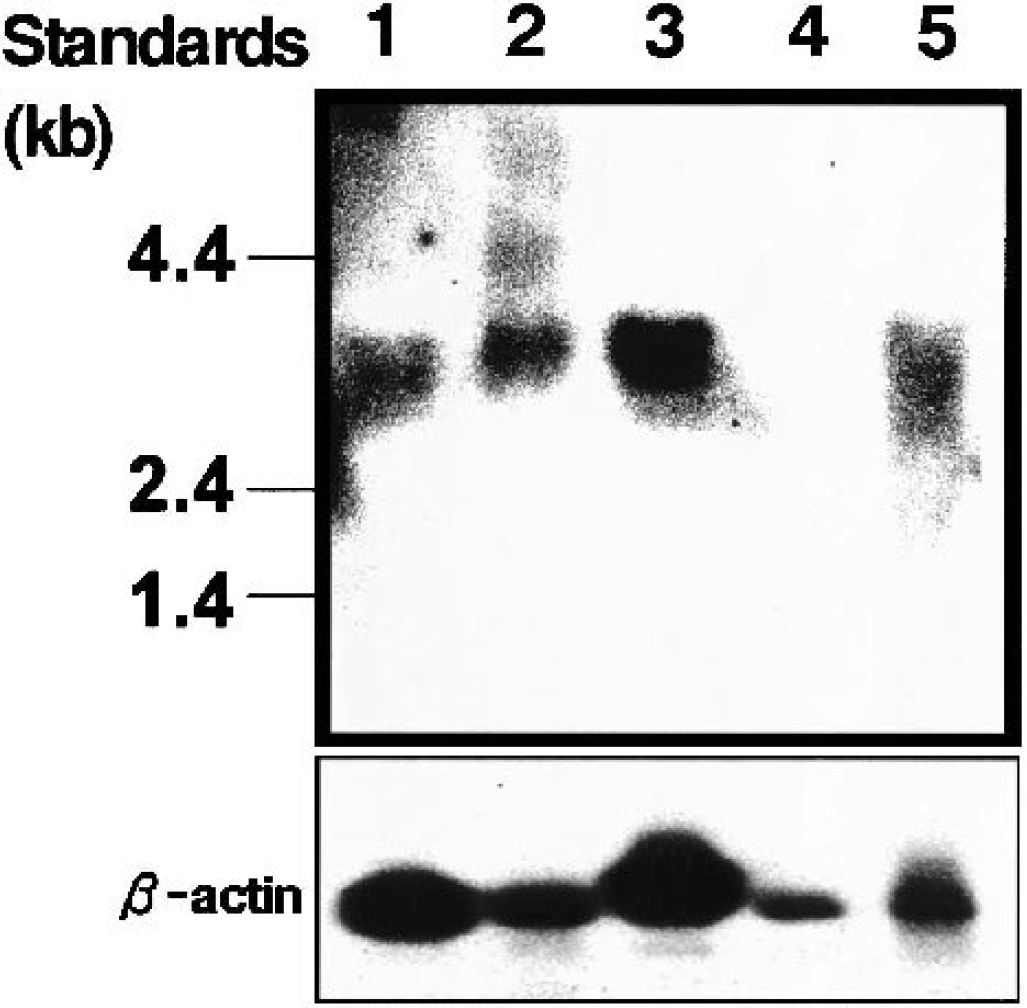
Northern blot analysis of creatine transporter mRNAs in TM-BBB4 cells, isolated mouse brain microvessels, and mouse peripheral tissues. Lane 1, TM-BBB4 cells; lane 2, total brain; lane 3, skeletal muscle; lane 4, liver; lane 5, isolated brain microvessels. The size of markers is indicated on the left-hand side.
Immunoblot analysis with an affinity-purified polyclonal antibody against mouse CRT was conducted using a crude membrane fraction of adult mouse brain, isolated mouse brain microvessels, and TM-BBB4 cells (Fig. 5). Bands at 84 kd, which were detected in all samples, represent the glycosylated form of CRT reported elsewhere (Dodd and Christie, 2001). On the other hand, an additional band at 74 kd was detected in the microsomal fraction of TM-BBB4 cells (lane 3), suggesting a nonglycosylated form of CRT. These bands were abolished when primary antibody was preabsorbed with CRT peptide (100 μg/mL; data not shown).
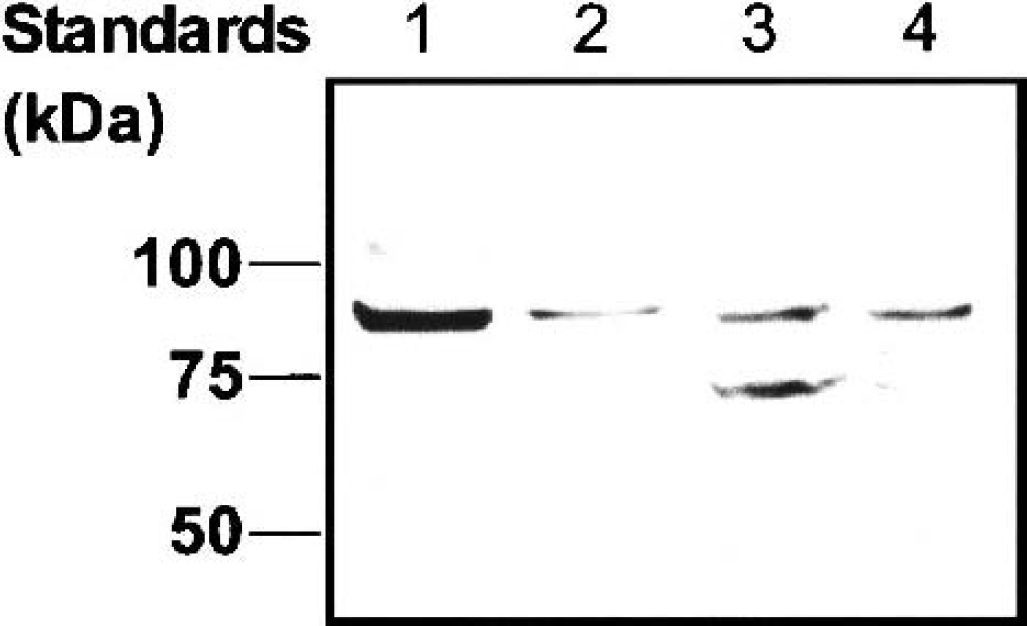
Expression of creatine transporter protein in subcellular fractions of TM-BBB4 cells and isolated mouse brain microvessels. Lane 1, crude membrane fraction of adult mouse brain; lane 2, crude membrane fraction of isolated brain microvessels; lane 3, microsomal fraction of TM-BBB4 cells; lane 4, crude membrane fraction of TM-BBB4 cells. The size of marker proteins is indicated on the left-hand side.
Localization of creatine transporter in mouse brain
The localization of CRT was determined in the cerebral cortex by immunohistochemical analysis (Fig. 6). The CRT immunoreactivity was strongly detected in brain capillaries, which are branched in all cortical layers (Fig. 6A). Immunostaining was observed along the surface of capillaries (arrows in Fig. 6B). In addition, neuronal perikarya in the cortical laminae II-VI were also stained at low to moderate levels (arrowheads in Fig. 6B). Such characteristic immunostaining was not seen after the use of preimmune rabbit immunoglobulin or primary antibody preabsorbed with CRT peptide (100 μg/mL; data not shown). These features indicate that, in the adult mouse brain, CRT is highly expressed in capillaries with additional expression in neurons.
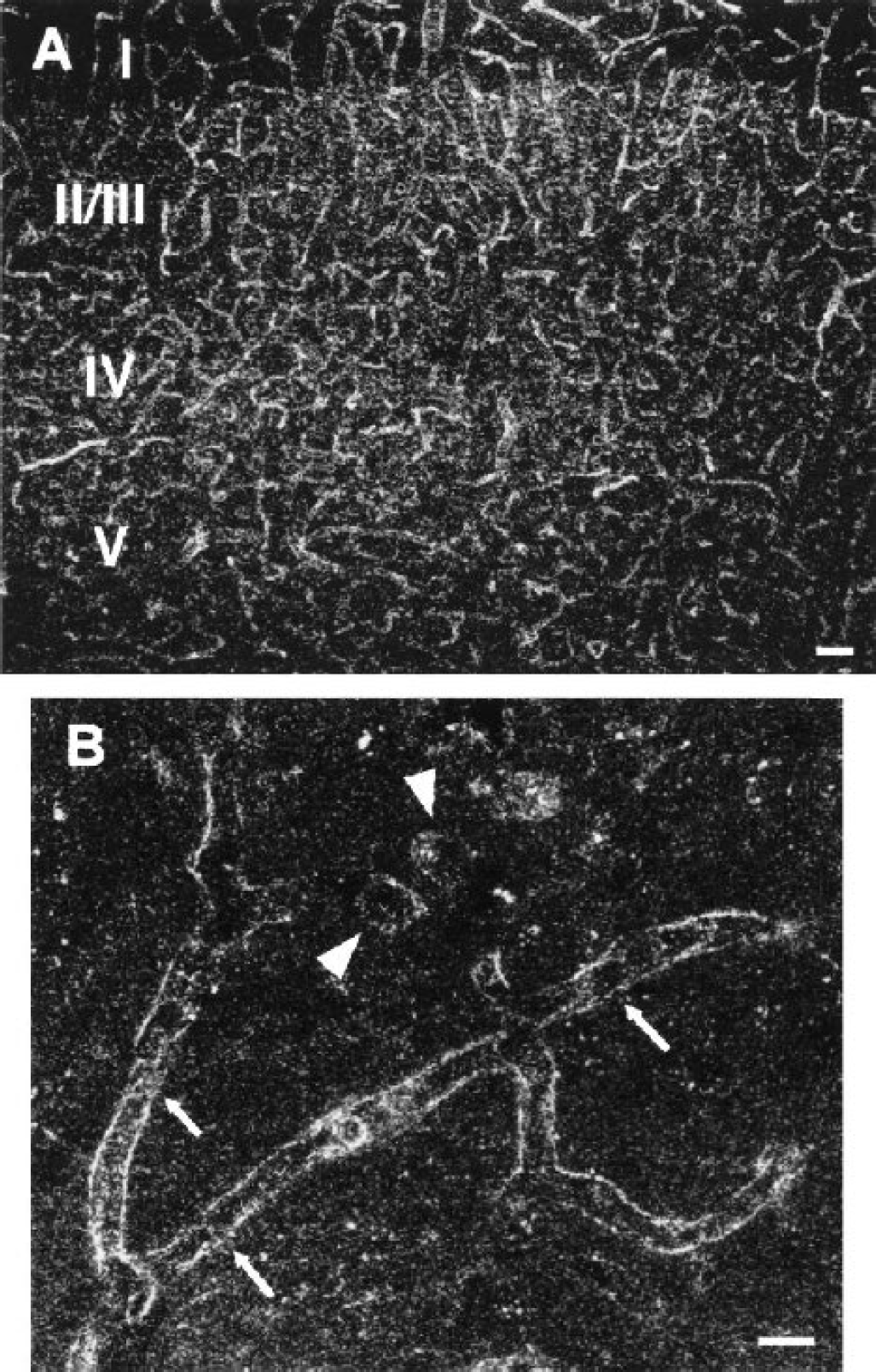
Localization of creatine transporter in the cerebral cortex of adult mouse brain (parasagittal sections). Low (
DISCUSSION
In this study, we have demonstrated creatine transport from the blood to the brain across the BBB and expression and localization of CRT in mouse capillaries. We have also characterized the key features of creatine transport in TM-BBB4 cells used as an in vitro BBB model. Creatine was transported and accumulated in the brain with a cerebrum Kp,app of 30.8 mL/g tissue (Fig. 1A), suggesting that creatine is supplied from the circulating blood to the brain across the BBB. This evidence is consistent with the 30-fold higher creatine concentration in mouse brain compared with that in serum (Marescau et al., 1992), supporting the hypothesis that the BBB is a major pathway supplying creatine to the brain. In other words, creatine is transported from the circulating blood to the brain against the concentration gradient between brain and blood. The influx permeability clearance per gram brain (PSvivo/Vbr) of [14C]creatine through the BBB was 1.61 μL/(min · g brain) (Fig. 1B). This number is about 1,600-fold greater than that of albumin, which is a marker for fluid-phase endocytosis at the BBB (Yang et al., 1997).
The characteristics of [14C]creatine uptake by TM-BBB4 cells show that it is an Na+- and Cl−-dependent carrier-mediated transport process (Fig. 2). Although [14C]creatine may be metabolized in the cells, the initial uptake rate reflects intact [14C]creatine transport from the medium into the cells. The Km and Vmax values for [14C]creatine uptake by TM-BBB4 cells are 16 μmol/L and 1.05 nmol/(mg protein · 10 minutes), respectively (Fig. 2C). This Km value is in good agreement with an apparent Km of 15 and 30 μmol/L in human and rat CRT, respectively (Saltarelli et al., 1996; Sora et al., 1994), and is 17-fold lower than the plasma creatine concentration (272 μmol/L) in the mouse (Marescau et al., 1992). This suggests that creatine transport is saturated by endogenous plasma creatine. Therefore, oral administration of a relatively large amount of creatine for 4 weeks produces only about a 9% increase in total creatine in human brain (Dechent et al., 1999). The BBB uptake clearance per gram brain from the in vitro uptake study (PSvitro/Vbr) can be estimated to be 2.1 μL/(min · g brain) according to Eq. 4. This is in good agreement with the PSvivo/Vbr value (1.6 μL/[min · g brain]) (Fig. 1B), suggesting that the in vitro uptake study using TM-BBB4 cells reflects the in vivo situation. These results suggest that the creatine transport process plays a role in continuously supplying creatine to the brain.
The characteristics of [14C]creatine uptake by TM-BBB4 cells suggest that creatine is transported via the CRT, since β-guanidinopropionate and guanidinoacetate produce marked inhibition of [14C]creatine uptake as reported elsewhere (Guimbal and Kilimann, 1993), but not of creatine metabolites (e.g., phosphocreatine and creatinine) and precursors (e.g., glycine and L-arginine) (Table 1). Mouse CRT cDNA was isolated from TM-BBB4 cells (Fig. 3) and Northern blot and immunoblot analysis showed that CRT is expressed in TM-BBB4 cells as well as isolated mouse brain microvessels (Figs. 4 and 5). Moreover, immunohistochemical studies revealed that CRT is intensely localized on capillaries (Fig. 6). CRT is also expressed in neurons (Fig. 6) and a previous study has reported that CRT mRNA can be detected in neurons by in situ hybridization (Braissant et al., 2001), suggesting that creatine is transported into neuronal cells by CRT following BBB transport. This is the first evidence that CRT is located in the brain capillary and plays a role in transporting creatine from the circulating blood to the brain across the BBB.
The physiologic role of CRT expressed at the BBB involves supplying creatine to the brain, as well as to skeletal muscle, since CRT expressed in skeletal muscle (Fig. 4) takes up creatine from the circulating blood (Fig. 1A). CRT at the BBB plays a key role in maintaining a constant supply of creatine to the brain and contributing to ATP homeostasis in neurons required for high metabolic demands. It remains to be seen whether creatine is synthesized from L-arginine and glycine in the brain. Braissant et al. (2001) used in situ hybridization and suggested that the mRNAs for enzymes involved in creatine synthesis, such as L-arginine–glycine amidinotransferase and S-adenosyl-L-methionine–N-guanidinoacetate methyltransferase, are expressed in the rat brain. The signal for CRT mRNA was absent in astrocytes, particularly those contacting capillary endothelial cells. Thus, Braissant et al. (2001) suggested that the brain is dependent on its own creatine synthesis. In N-guanidinoacetate methyltransferase–deficient patients, oral administration of creatine increased the creatine level in the brain, and this increase was accompanied by an improvement in the patient's neurologic symptoms (Stöckler et al., 1994). In contrast, patients with a nonsense mutation of creatine transporter 1 (SLC6A8, CT1) did not display any creatine signal in the brain according to proton magnetic-resonance spectroscopy (Cecil et al., 2001; Salomons et al., 2001), strongly suggesting a contribution from creatine transport at the BBB. Moreover, oral administration of creatine is a potential treatment for neurodegenerative diseases owing to the neuroprotective effects found in animal models of ALS and Huntington and Parkinson diseases (Klivenyi et al., 1999; Matthews et al., 1998, 1999). Our results indicate that CRT is involved in the carrier-mediated blood-to-brain transport of creatine across the BBB.
The BBB has been investigated to examine transport functions as a dynamic regulatory interface (Cornford, 1985; Terasaki and Hosoya, 1999). Although there has been an investigation of the mechanism of transport at the BBB and identification of the responsible molecules, including GLUT1, MCT1, and LAT1 (Boado et al., 1999; Gerhart et al., 1997; Pardridge et al., 1990a; Takanaga et al., 2001; Tsuji et al., 1992), the transport system of creatine at the BBB has not been elucidated. It is important to clarify the origin of creatine, which plays an important role in the high-energy buffering system in the brain consuming large amounts of energy. Our findings regarding CRT expression and creatine transport at the BBB provide new evidence to increase our understanding of the physiologic role of the BBB and the regulation of creatine concentration in the brain.
Oral supplementation of creatine is crucial in creatine-deficient patients (Stöckler et al., 1994, 1996), and the creatine treatment of neurodegenerative diseases may depend on CRT expression at the BBB. The present study shows that CRT at the BBB plays a pivotal role in regulating the creatine concentration as a major pathway in the brain. These findings regarding the creatine transport and CRT expression at the BBB are important in increasing our understanding of the mechanism governing the supply of creatine to the brain and the neuroprotective effect produced by oral supplementation of creatine.
Footnotes
Acknowledgments:
The authors thank Ms. N. Funayama for secretarial assistance.