Abstract
Although the interleukin (IL)-1 receptor is densely distributed in the leptomeninges constituting the blood/cerebrospinal fluid barrier, its physiologic significance has remained unclear. In the present study, we show that in cultured leptomeningeal cells, IL-1β, tumor necrosis factors, or lipopolysaccharide causes a prominent increase in the synthesis and release of prostaglandin (PG) D synthase, which catalyzes the final step in the biosynthesis of PGD2. Although significant increases in the amount of PGD synthase were also observed with cells exposed to somatostatin, thrombin, or ciliary neurotrophic factor, these were much smaller than were those induced by the proinflammatory cytokines. Other agents tested including IGF-I had no effect upon the enzyme levels in the culture media. Furthermore, we found that the increased secretion of PGD synthase by IL-1β was completely inhibited by 10−7 M PGE2. The same dose of PGD2 or 15-deoxy-Δ 12–14 PGJ2 had no effect upon the IL-1β action. In addition, PGE2 increased the level of fibronectin and eliminated the expression of zonula occludentes-1, a tight junction-associated protein from cultured cells, effects likely reflecting a loss of barrier integrity. These results demonstrate the importance of inflammatory stimuli as a physiologic regulator of the leptomeningeal cell function.
The choroid plexus and leptomeninges constituting the blood/cerebrospinal fluid barrier (BCSFB) have been shown to express, synthesize, and secrete biologically important proteins (Ohe et al., 1996), which are beneficial for brain parenchymal cells. They include insulin-like growth factor-binding protein (IGFBP)-2 and transferrin, among others (Berger and Hediger, 2000;Carder et al., 1990;Durkin et al., 1995;Evans et al., 1996;Ohe et al., 1996;Senjo et al., 1986;Stenvers et al., 1994;Sullivan and Feldman 1994;Yamaguchi et al., 1994;Zetterstrom et al., 1994). Based upon the wide spectrum of their protein products, the choroid plexus/leptomeninges has been thought to be involved in the maintenance of brain parenchymal cells or contribute to the scavenging of wasted materials and the detoxifying of a wide range of toxic substances. In addition to such roles, the presence of rich networks of dendritic cells and a dense population of resident tissue macrophages in the choroid plexus and leptomeninges (McMenamin, 1999) suggest their importance in the brain response to inflammation. This hypothesis is further supported by the finding that tumor necrosis factor (TNF)-α injected into the brain causes a prominent inflammatory reaction in the leptomeninges (Glabinski et al., 1998). The brain structure expresses high levels of mRNA for kininogens, which may be a precursor for kinins (Takano et al., 1999). Furthermore, the receptor for interleukin (IL)-1, a proinflammatory cytokine, is highly concentrated in the leptomeninges/choroid plexus, and its binding activity decreases after peripheral injection of bacterial lipopolysaccharide (LPS) (Ban, 1994;Marquette et al., 1995). IL-1β infusion into the brain induces a rapid recruitment of granulocytes into cerebral spinal fluid (CSF), which precedes the invasion into the brain parenchyma (Schoening et al., 2002). Based on the finding that the BCSFB is more fragile than is the blood/brain barrier (BBB) when exposed to IL-1β (Anthony et al., 1997;Schnell et al., 1999), it has been hypothesized that granulocytes infiltrate via the BCSFB (the choroid plexus), CSF, the Virchow-Robin spaces, and the brain parenchyma. The IL-1β -mediated breakdown of the barrier is well known to underlie, at least in part, the pathogenesis of many neurodegenerative diseased or injury, including ischemia (Kuroiwa et al., 1988). Intracerebroventricular injection of IL-1β has been shown to dramatically exacerbate ischemic brain damage, whereas IL-1 receptor antagonist injection significantly reduces ischemic injury (Loddick and Rothwell, 1996). Deletion of IL-1 in mice reduces ischemic brain damage by approximately 80% (Boutin et al., 2001). Therefore, the choroids plexus/leptomeninges may be one of the chief targets of the pro-inflammatory cytokines released under inflammation, and clarification of the physiologic significance of such receptors on the BCSFB may be important biomedically.
Inflammatory stimuli such as peripheral LPS injection rapidly induce cyclooxygenase (COX) type 2 (Cao et al., 1997;Yamagata et al., 2001) and prostaglandin (PG) E synthase expression (Mancini et al., 2001) in endothelial cells that line the blood/brain barrier (BBB) or macrophages in the BCSFB. The PGE2 thereby produced diffuses into the brain parenchyma, where it elicits systemic responses such as fever (Saper, 1998). Because the number of COX-2 expressing-cells and fever follow a similar time course, the BBB is thought to be the main site at which the activation of arachidonic acid cascade producing PGE2 takes place. On the other hand, there are few data available for understanding the physiologic role of the BCSFB during inflammation. Leptomeningeal cells constituting the BCSFB produce and secrete a large amount of PGD synthase (Ohe et al., 1996), which catalyzes the final step in the biosynthesis of PGD2. The PG and its nonenzymatic metabolites, notably 15-deoxy-Δ PGJ212–(15-dPGJ2), have been proposed to participate in antiinflammatory processes such as inhibition of the production of proinflammatory cytokines and reduction of inducible nitric oxide synthase (iNOS) (Bernardo et al., 2000;Colville-Nash et al., 1998), which catalyzes the formation of NO. These findings raise the possibility that the leptomeninges serve as a modulator of PG metabolism during inflammation at the border between the brain and the peripheral circulation. To clarify this further, we have examined the effects of growth factors and proinflammatory cytokines including IL-1β on the synthesis and secretion of PGD synthase in cultivated leptomeningeal cells. In the present study, we show that IL-1β remarkably increases the synthesis and release of PGD synthase. The stimulation of PGD synthase production and secretion by IL-1β was completely inhibited by PGE2. Administration of PGE2 also eliminated the expression of ZO (zonula occludentes)-1, a tight-junction associated protein, and stimulated the secretion of fibronectin from cultured leptomeningeal cells.
METHODS
Leptomeningeal cell culture
Leptomeningeal cell cultures were prepared as described previously (Ohe et al., 1996). In brief, 1-day-old Wistar rats were killed by decapitation. The brains were removed from the skull and immersed in ice-cold Dulbecco's modified Eagle's medium (DMEM) containing 15 mM HEPES, 24 mg/L penicillin G, 50 mg/L streptomycin, 50 mg/L gentamicin, and 10% fetal bovine serum (FBS). The cortical leptomeninges were taken out with a pair of fine forceps under a microscope and cut into small pieces after being washed with the fresh medium. The tissue fragments were suspended in HEPES-free DMEM containing 10% FBS and antibiotics. The suspended fragments were plated on 75-cm2 culture dishes coated with poly-D-lysine (MW 261,000; Sigma Chemical, St. Louis, MO, U.S.A.) and incubated in a humid atmosphere of 5% CO2 at 37°C. After incubation for approximately 1 week, the outgrowing cells were treated with 0.125% trypsin (Sigma, Type XIII)/0.06% collagenase (Wako, Osaka, Japan) solved in Ca2+/Mg2+-free sterile isotonic buffer (SIB) consisting of 15 mM HEPES, 122mM NaCl, 5mM KCl, 0.7 mM KH2PO4, 25 mM glucose, 0.3% BSA, and 59 mM sucrose. The treated cells were triturated with a disposable pipette, filtered through a 75-μm mesh, collected by centrifugation (500 g for 5 minutes), and suspended in fresh DMEM containing 10% FBS and antibiotics. The dissociated cells were grown in 10% FBS-containing DMEM for 1 additional week and harvested with trypsin/collagenase solution, following vigorous shaking to remove contaminating microglial cells. The purity of the cells was assessed by immunocytochemistry with antibodies against rat PGD synthase (Ohe et al., 1996) and GFAP (Ishimaru et al., 1996). More than 90% of cells obtained were PGD synthase-positive and showed large, flat, polygonal cells in cultures. Contamination with GFAP-positive cells and cells with neuronal cell morphology were negligible. Finally, the cells were suspended in a preservative (Cellbanker II, NZH Biochemicals, Japan) and frozen in liquid nitrogen until used for experiments.
Measurement of prostaglandin D synthase
The levels of PGD synthase in media were estimated by a specific and highly sensitive fluoroimmunoassay with 96-well polystyrene microtiter strips, as previously described (Ishizaka et al., 2001). In brief, 50 μL of each medium was appropriately diluted in the assay buffer (Delfia Perkin Elmer, Wellesley, MA, U.S.A.) and incubated in wells coated with rabbit anti-rat PGD synthase serum. After washing with a plate washer (Delfia), the wells were further incubated with Eulabeled anti-PGD synthase. Each well was then washed, and the fluorescence was measured in a time-resolved mode with a platefluorometer (Delfia) after adding the enhancement solution (Delfia). The assay of all calibrators was performed in duplicate. Between-group comparisons for all data were made with Student's t- test or Chochran-Cox test after one-way analysis of variance. P < 0.05 was considered significant.
Determination of mRNA levels
Reverse transcription (RT)-polymerase chain reaction (PCR) analysis was performed as described elsewhere (Fujimori et al., 2003). Briefly, the first-strand cDNAs were synthesized from 1 μg of total RNA with Oligo(dT) Adapter Primer (Takara Shuzo, Kyoto Japan) and RNase H- SuperScript II reverse transcriptase (Invitrogen, California, U.S.A.) at 42°C for 60 minutes after denaturation of the RNA at 72°C for 3 minutes. PCR amplification was performed using gene-specific primer sets of L-PGDS-T (5′-GACAAGTTCCTGGGGCGCTGGT-3′) and L-PGDS-C (5′-GCTGTAGAGGGTGGCCATGCGG-3′) for PGD synthase (Urade et al., 1989) or G3PDH-5′ (5′-ACCACAGTCCATGCCATCAC-3′) and G3PDH-3′ (5′-TCCACCACCCTGTTGCTGTA-3′) for glyceraldehyde-3-phosphate dehydrogenase (Tso et al., 1985). The amplified products were separated by electrophoresis on 2% agarose gel and stained with ethidium bromide. The stained products were scanned and analyzed by ChemiImager system (Alpha Innotech, San Leandro, CA, U.S.A.).
Incubation with cytokines or growth factors
To examine the effects of cytokines and growth factors on the PGD synthase production of leptomeningeal cells, the preserved leptomeningeal cells were used. The cells were suspended in 10% FBS-containing DMEM (HEPES-free) and plated on 6-well culture dishes at a density of 2.5 × 105 cells/well. After 5 days, when leptomeningeal cell cultures reached confluence, the cultures were washed thoroughly and further incubated in FBS-free DMEM with or without of the test sample. The PGD synthase levels in the FBS-free media were monitored. The final concentration of each test sample used was calculated from the dose at which the sample was reported to produce its maximal biologic effect. The test samples included LPS (20 μg/mL, Escherichia coli 055:B5, List Biological Laboratories, Campbell, CA, U.S.A.), recombinant rat TNF-α and -β (100 ng/mL, Pepro Tech EC, London, England), recombinant IGF-I (50 ng/mL, Bachem, U.S.A.), somatostatin (1 μM, Peptide Institute, Japan), thrombin (40 ng/mL), recombinant murine leptin (200 nM, Pepro Tech EE, England), recombinant murine IL-6 (10 ng/mL, Pepro Tech EE, England), recombinant murine IL-1β (10 ng/mL, Pepro Tech EC, England), nerve growth factor (NGF) (100 ng/mL), recombinant ciliary neurotrophic factor (CNTF) (100 ng/mL, Genzyme, Cambridge, MA, U.S.A.), acidic and basic fibroblast growth factors (FGF) (100 ng/mL, R & D, Minneapolis, MN, U.S.A.), epidermal growth factor (EGF) (100 ng/mL, Biomedical Technologies, Stoughton, MA, U.S.A.), human recombinant transforming growth factor (TGF) -β1 and -α (10 ng/mL, Boehringer Mannheim Biochemica, Germany), and recombinant human platelet derived growth factor (PDGF)-AA (100 ng/mL, Pepro Tech EE, England). To examine the effects of PGD2 (Wako, Japan), PGE2 (Wako, Japan), PGI2 (Sigma, U.S.A.), and 15-dPGJ2 (Cayman, Ann Arbor, MI, U.S.A.) on the IL-1β action, each PG was solved in ethanol or water and added to FBS-free medium at the onset of incubation.
Western blotting
To quantify the major protein constituents in the same preparation as used for the measurement of PGD synthase levels, each culture medium was concentrated and desalted with a centrifugal filter device (Microcon YM-10 or −3, Millipore, Bedford, MA, U.S.A.) and then dried and dissolved in sodium dodecyl sulfate sample buffer. Transfer onto nitrocellulose membrane (Advantec, Tokyo, Japan) after electrophoresis was performed as described elsewhere (Ohe et al., 1996). To block nonspecific binding, the membranes were treated with 5% non-fat dry milk in Tris-buffered saline containing 0.1% Tween-20 (TBS-T) for 1 hour at room temperature and then incubated with each 1st antibody (diluted 1:1,000–2,000) overnight at room temperature. The antibodies used included rabbit anti-rat fibronectin serum (Biogenesis, U.S.A.), guinea pig anti-rat follistatin-related protein (FRP) serum (made at our laboratory), guinea pig anti-rat β2-microglobulin serum (Shinoda et al., 1996), guinea pig anti-rat secreted protein acidic and rich in cystein (SPARC) serum (made at our laboratory), rabbit anti-rat transferrin serum (Ishimaru et al., 1996b), guinea pig anti-rat cystatin C serum (Ishimaru et al., 1996a), guinea pig anti-rat IG-FBP-2 serum (Ishikawa et al., 1995), and guinea pig anti-rat apolipoprotein E serum (Ishimaru et al., 1996a). After the incubation with biotinilated 2nd antibodies, the complexes were visualized with 3,3′-diaminobenzidine (DAB), following the manufacturer's instruction (ABC kit, VectaStain, Burlingame, CA, U.S.A.).
Immunocytochemistry
For immunocytochemical staining for NF-κB, leptomeningeal cells were fixed with 4% paraformaldehyde in phosphate buffer, pH 7.5. The fixed cells were treated with acetone/methanol (1:1) for 2 minutes and then incubated in phosphate-buffered saline containing 2% normal horse serum and 0.3% tritonX-100 (PBS-T) for 1 to 2 hours. After rinsing with PBS-T, the cells were incubated with rabbit anti-NF-κB (P-65) serum (IBL, Gunma, Japan) overnight at room temperature. The reaction products were visualized with avidin/biotin peroxidase staining by the ABC technique (Vectastain/Elite ABC kit, Vector Laboratories, Burlingame, CA, U.S.A.). The immunoperoxidase reaction was developed with 0.02% H2O2 using 0.05% DAB as a chromogen. For the staining for ZO-1, cultured cells were fixed with 3% acetic acid in methanol for 10 minutes at −20°C. After washing with PBS-T, the cells were incubated with rabbit anti-ZO-1 serum (Zymed Lab., CA, U.S.A.) overnight. The reaction products were visualized with avidin/biotin staining by the ABC technique, as described above.
RESULTS
Stimulation of PGD synthase secretion by proinflammatory cytokines
The data on the effects of cytokines and growth factors on PGD synthase secretion from cultivated leptomeningeal cells are summarized in Fig. 1. The PGD synthase levels in the media were noticeably increased by proinflammatory cytokines (TNF-α, TNF-β, and IL-1β) and LPS. In the control, the level at day 5 was 1.18 ± 0.70 ng/mL (n = 3). The enzyme levels in the presence of TNF-α, TNF-β, and IL-1β were 73.5 ± 30 ng/mL, 98.0 ± 36 ng/mL, and 16.8 ± 7.0 ng/mL, respectively. The addition of LPS to the culture medium caused a similar increase in the enzyme levels. In addition to such cytokines, SRIF, thrombin, and CNTF caused a slight increase in the enzyme levels. Other agents tested, including IGF-1, leptin, IL-6, NGF, FGFs, EGF, TGFs and PDGF-AA, had no effects on the enzyme levels in the culture media. The PGD synthase levels in the media containing PGD2 were significantly lower than those in the controls.
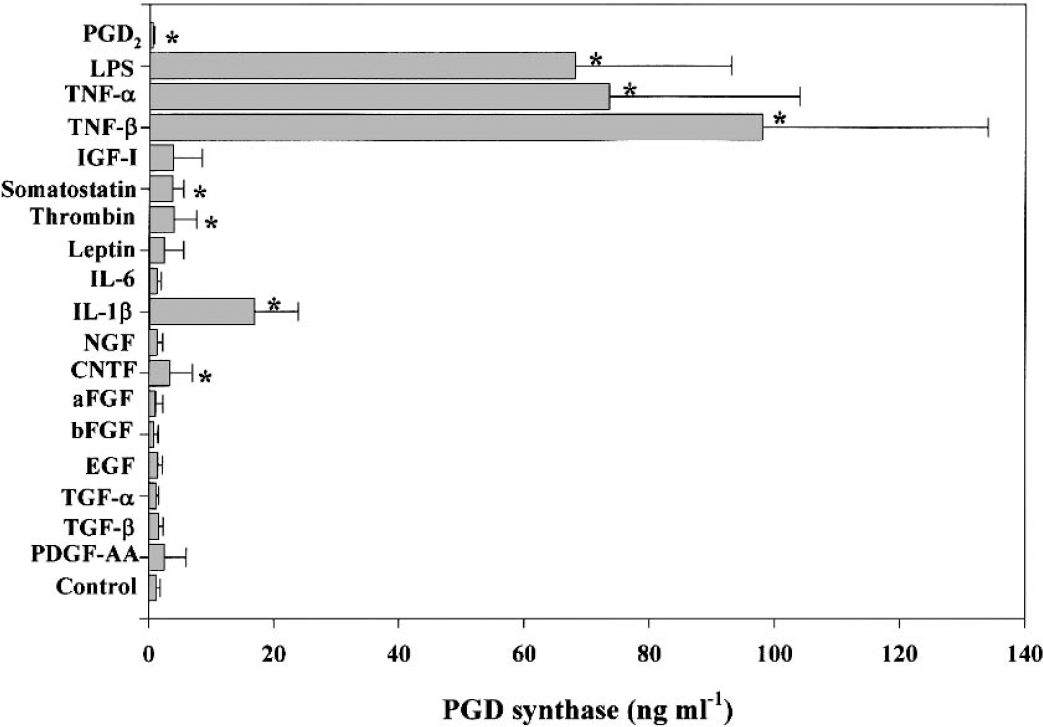
Stimulation of PGD synthase secretion from cultured leptomeningeal cells by proinflammatory stimuli (IL-1β, TNFs, and LPS). Cells were incubated for 5 days in the absence (control) or presence of the indicated factors (see Methods for concentrations), after which the concentration of PGD synthase in the culture medium was determined. Data are means ± SD of values from three independent experiments. *P < 0.05 versus control value (Student's unpaired t-test). PGD, prostaglandin D; IL, interleukin; TNF, tumor necrosis factor; LPS, lipopolysaccharide.
Time and dose dependency
In the next experiment, we examined the time and dose dependency of the cytokine action. Administration of LPS resulted in an increase in the level in a time and dose dependent manner (Fig. 2a). Within 1 day of incubation, there was no significant difference in the levels between the LPS group and the control. At 3 days, 10 to 80 μg/mL of LPS increased the levels significantly (P < 0.05 versus control; Student's t-test). The highest level was observed at 7 days in the presence of 40 μg/mL of LPS. TNF-α and IL-1β caused a similar increase in the enzyme level. The increase in the level induced by TNF-α was dose-dependent, and the highest level was observed at a dose of 500 ng/mL. The action of IL-1β on the level was also time- and dose-dependent: the enzyme level increased steadily during incubation. The maximum level (58.0 ± 9.8 ng/mL at day 7) was observed at a dose of 4 ng/mL, and the levels at 20 to 500 ng/mL of IL-1β were much lower than those at 4 ng/mL.
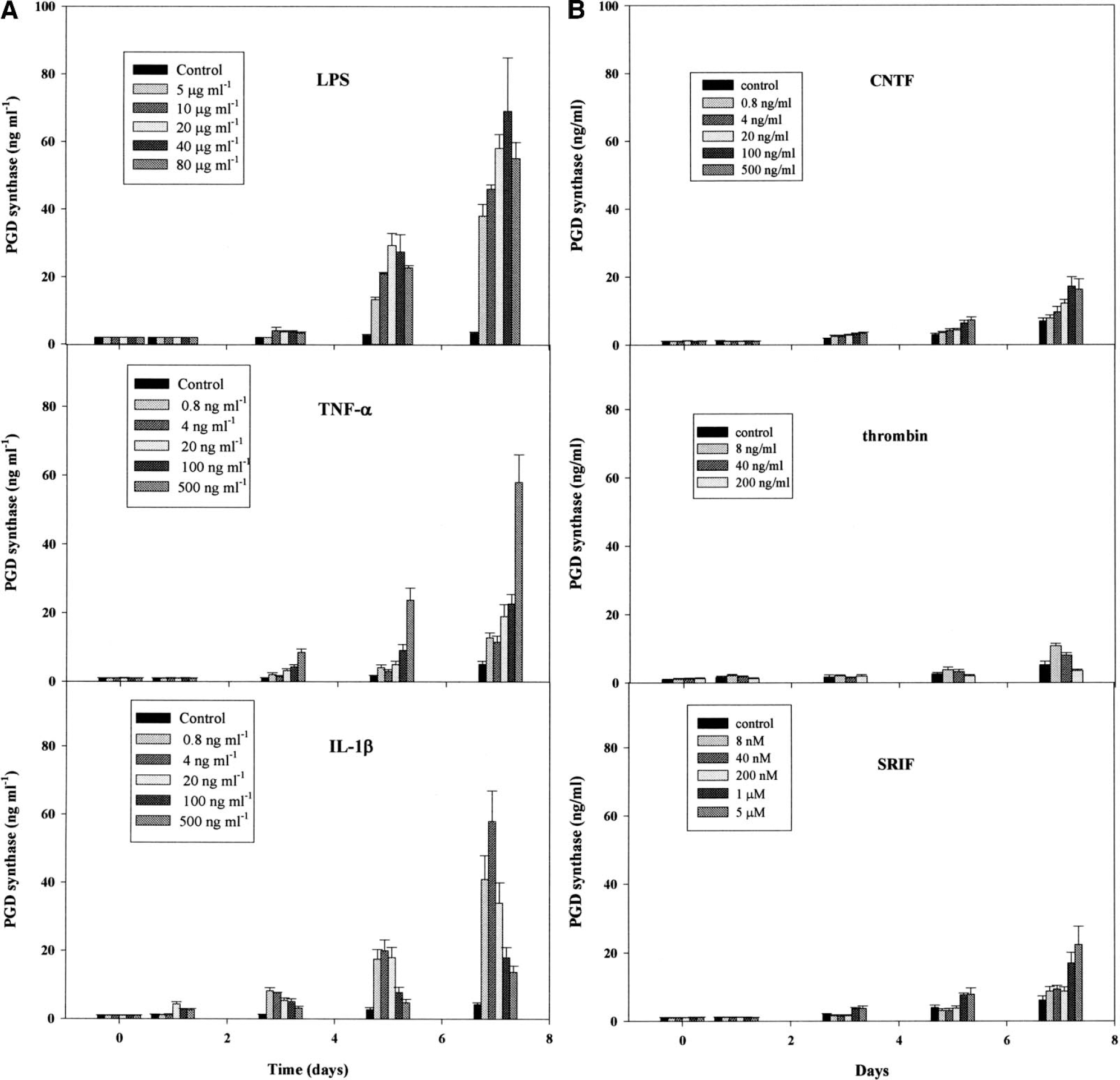
Thrombin and CNTF caused a small (but significant) increase in the enzyme level. Eight ng/mL of thrombin resulted in an increase in the level (10.8 ± 1.0 ng/mL at day 7), but little difference was observed at a larger dose of thrombin (Fig. 2b). CNTF action on the enzyme level was time- and dose-dependent, and the maximum levels (18.5 ± 2.0 ng/mL at day 7) were observed in the presence of 100 to 500 ng/mL of CNTF. The modification caused by SRIF was almost the same as that by CNTF, and the highest level (25.0 ± 5.0 ng/mL at day 7) of the enzyme was observed in the presence of 5 μM of SRIF. These increases induced by SRIF, CNTF, or thrombin were much smaller than were those by the proinflammatory cytokines at all doses examined.
MRNA levels
PGD synthase mRNA levels in the cultivated leptomeningeal cells were monitored and found to be increased by IL-1β. In the control, mRNA levels decreased slightly during incubation (Fig. 3). At 48 to 72 hours, the levels were significantly lower than those at the beginning of the incubation. On the other hand, 10 ng/mL IL-1β caused dramatic increases in the mRNA levels. During incubation, the levels increased and reached a peak at 24 hours and gradually decreased thereafter, but the level at 72 hours was still higher than that of the control. G3PDH gene expression was not altered by the IL-1β treatment (data not shown).
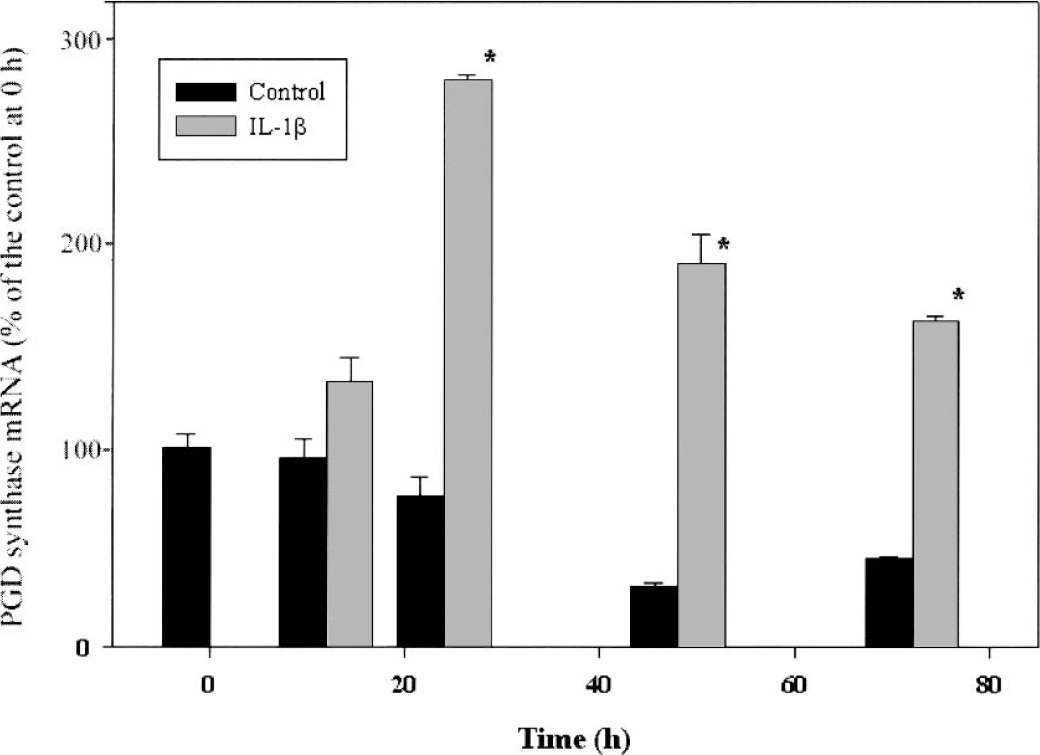
Time course of the effect of IL-1β on the abundance of PGD synthase mRNA in cultured leptomeningeal cells. Cells were incubated for the indicated times in the absence (control) or presence of IL-1β (10 ng mL−1), after which the amount of PGD synthase mRNA in cells was determined by RT-PCR. Data are expressed as 107 molecules of PGD synthase mRNA per microgram of total RNA and are means ± SD of values from three independent experiments. *P < 0.05 versus corresponding control value (Student's unpaired t-test). PGD, prostaglandin D; IL, interleukin; RT-PCR, reverse transcription PCR.
Inhibitory action of PGs
Exposure of leptomeningeal cells to 10 mM PGD2 was found to reduce the secretion of PGD synthase into the culture medium (Fig. 1). To characterize this further, we compared the effects of PGE2, PGD2, 15dPGJ2, and PGI2 upon the IL-1β -induced secretion of PGD synthase. Although PGI2 had no effect on the enzyme levels (data not shown), three PGs (PGD2, PGE2, and 15dPGJ2) inhibited the stimulation of PGD synthase release by IL-1β. PGE2 was the most potent inhibitor; the median inhibitory concentration of PGE2 was <10 nM, compared with values of 5 μM for PGD2 and 15dPGJ2 (Fig. 4).
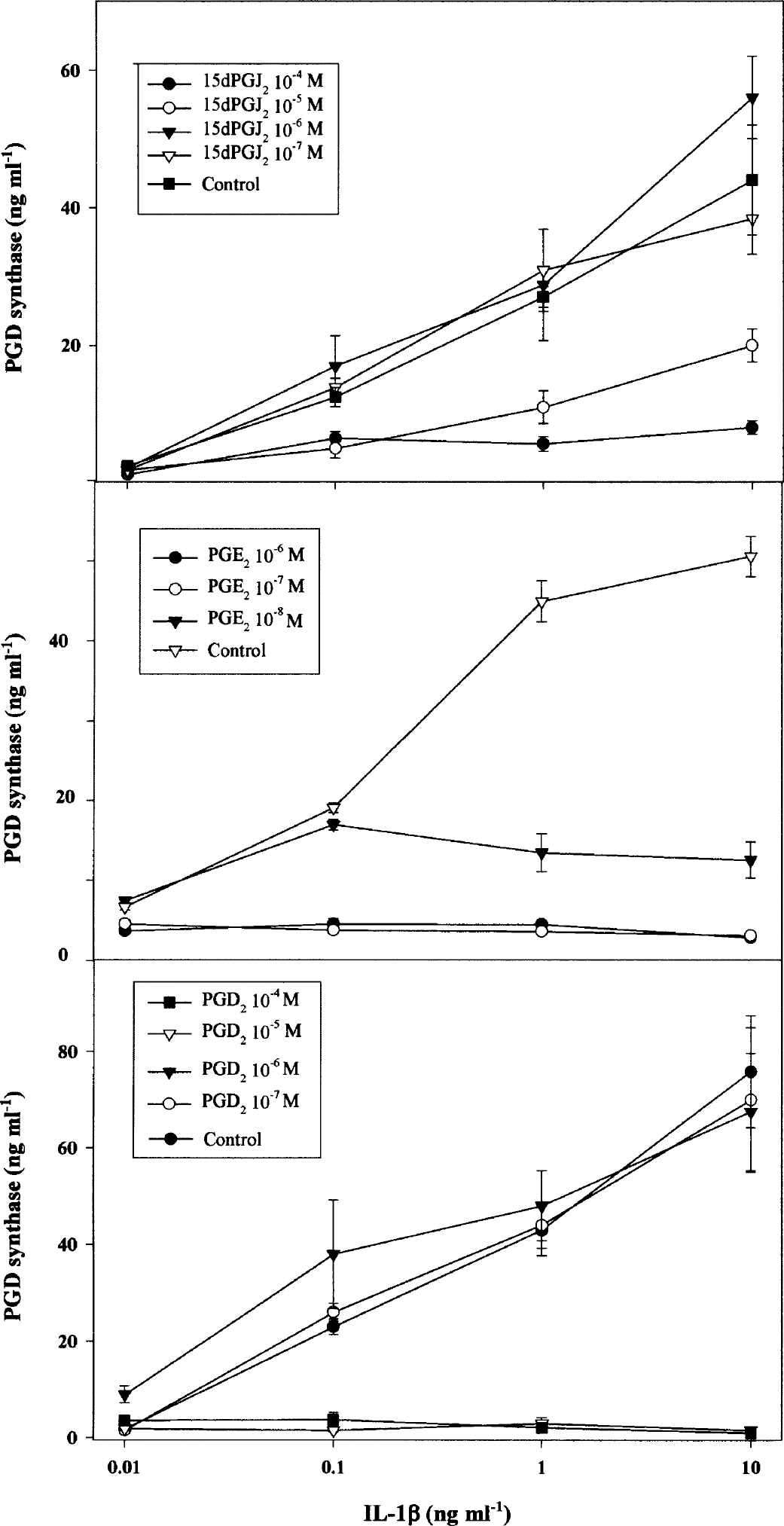
Inhibition of IL-1β –induced PGD synthase secretion by 15dPGJ2, PGE2, and PGD2. Leptomeningeal cells were cultured for 5 days with IL-1β (0.01–10 ng/mL−1) in the absence (control) or presence of either 15dPGJ2, PGE2, or PGD2 at the indicated concentrations, after which the concentration of PGD synthase in the culture medium was determined. Data are means ± SD of values from three independent experiments. PG, prostaglandin; IL, interleukin.
NF-kB translocation
In control cultures, NF-κB immunoreactivity was diffusely distributed throughout the cytoplasm of leptomeningeal cells (Fig. 5). After incubation of the cells with IL-1β (10 ng/mL) for 3 hours, intense NF-κB immunostaining was apparent within the cell nuclei, indicative of the nuclear translocation of this transcription factor. Exposure of cells to 0.1 mM PGE2 had no effect upon the pattern of NF-κB immunostaining in the absence or presence of IL-1β, indicating that the inhibitory effect of -induced increase in PGD synthase is not PGE2 on IL-1β mediated at the level of NF-κB translocation.
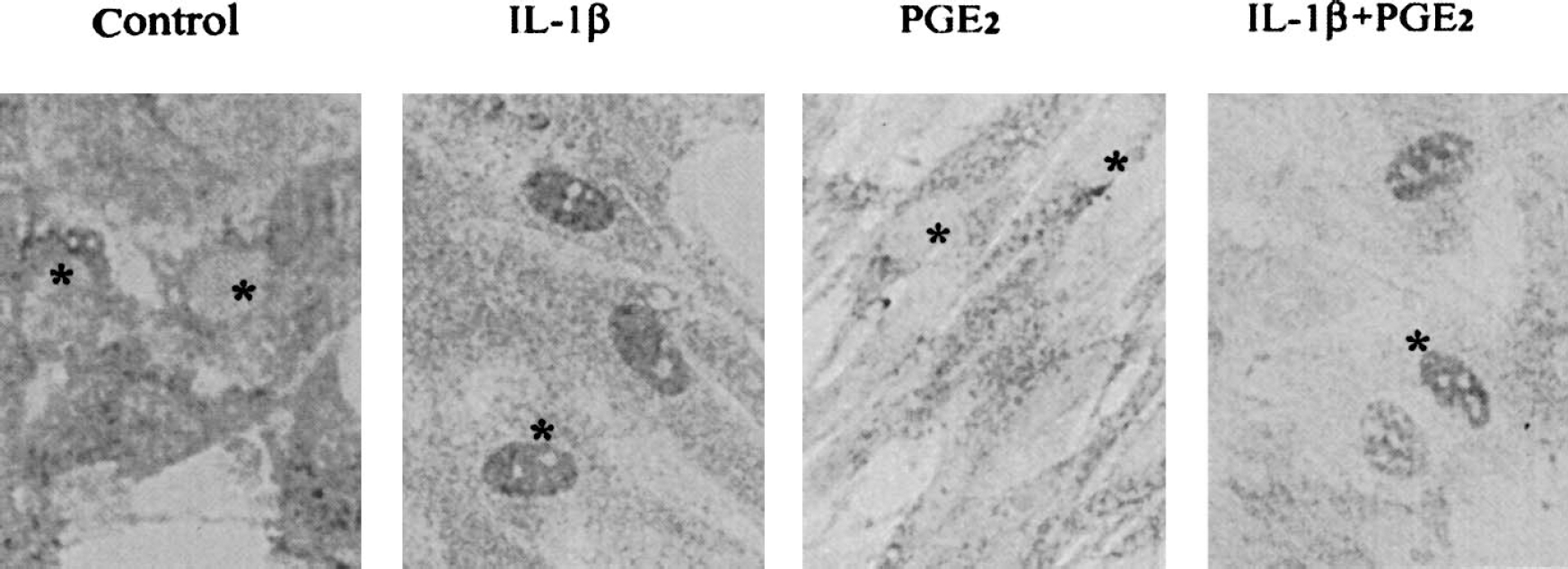
Effects of IL-1β and PGE2 upon the subcellular distribution of NF-κB in cultured leptomeningeal cells. Cells were incubated for 3 hours in FBS-containing medium in the absence (control) or presence of IL-1β (10 ng ml−1), 0.1 μM PGE2, or IL-1β plus PGE2. They were then fixed and subjected to immunocytochemical staining for NF-κB. *Nuclei. IL, interleukin; PG, prostaglandin; FBS, fetal bovine serum.
Western blot analysis
To investigate whether PGE2 or IL-1β also modifies the secretion of other bioactive proteins from leptomeningeal cells, the major protein components such as β2-microglobulin, IGFBP-2, and fibronectin in the media were analyzed by means of western blotting. IL-1β did not induce substantial changes in the secretion of the major protein constituents except PGD synthase (data not shown). On the other hand, incubation of cells for 5 days with 0.1 mM PGE2 resulted in a marked increase in the intensity of fibronectin bands (>150 kDa) (Fig. 6). The identity of these positive bands was confirmed by their individual excision from the membrane and determination of the partial amino acid sequence of each protein (data not shown) according to the method described previously (Ohe et al., 1996). PGE2 induced relatively small increases in the release of FRP and β2− microglobulin and had essentially no effect on that of SPARC, transferrin, cystatin C, IGFBP-2, or apolipoprotein E.
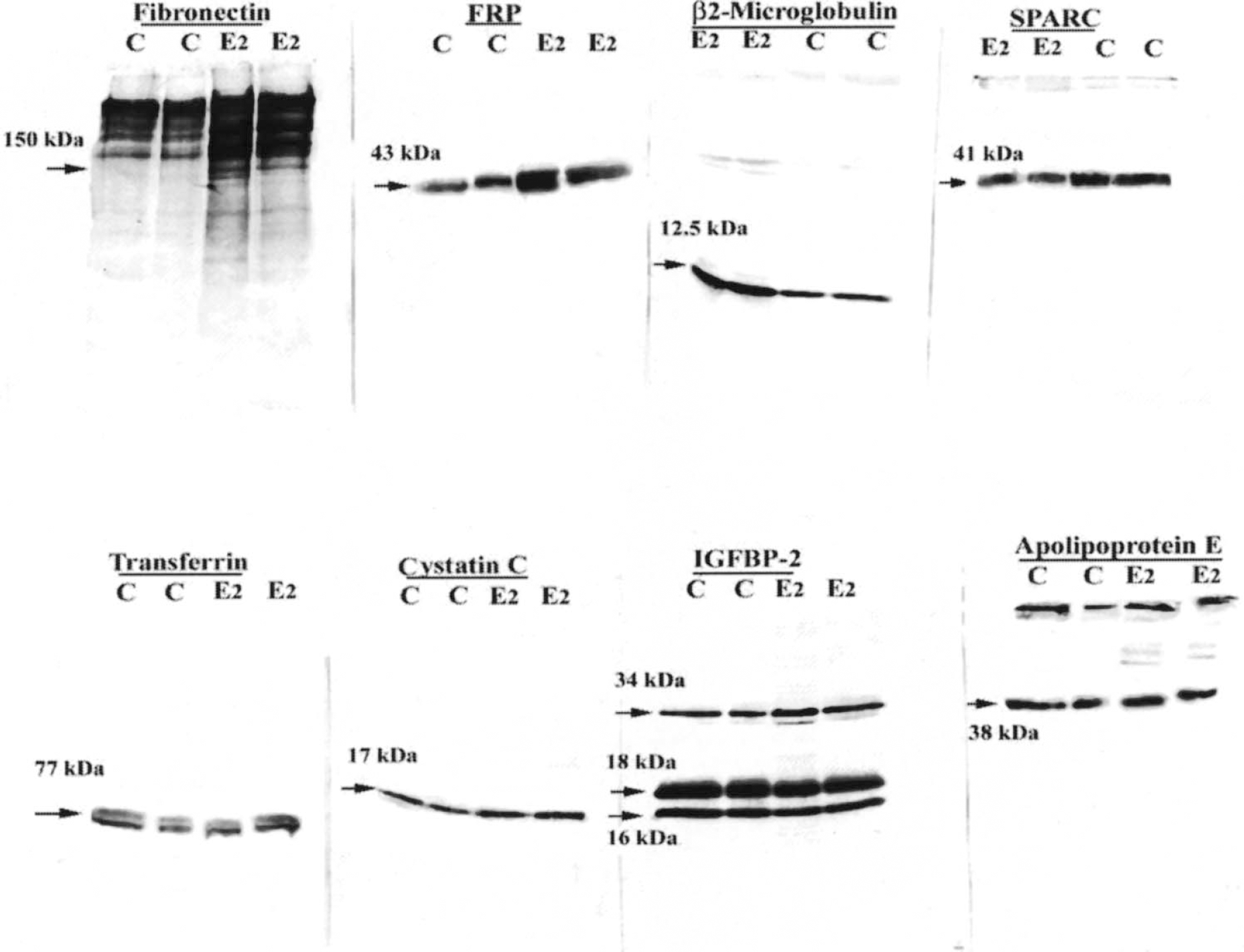
PGE2-induced increase in fibronectin secretion from leptomeningeal cells. Cells were incubated for 5 days in the absence (C) or presence (E2) of 0.1 μM PGE2, after which the culture medium was subjected to immunoblot analysis with antibodies to the indicated proteins. Arrows, positions and sizes of the immunoreactive proteins. PG, prostaglandin.
ZO-1 immunoreactivity
To examine the possibility that PGE2 affects the formation of BCSFB, we subjected cultured leptomeningeal cells to immunocytochemical staining with an antibody to ZO-1, which is localized to the networks of tight junctions between cells in the brain vasculature and the leptomeninges (Smith and Shine, 1992). Loss of ZO-1 from vascular endothelium is associated with breakdown of the BBB (Dallasta et al., 1999). Control cultures of leptomeningeal cells exhibited an epithelial cell-like morphology, with immunostaining for ZO-1 restricted to the cell periphery (Fig. 7). Exposure to 0.1 mM PGE2 for 3 days induced the cells to adopt a more elongated shape and resulted in the complete loss of ZO-1 staining.
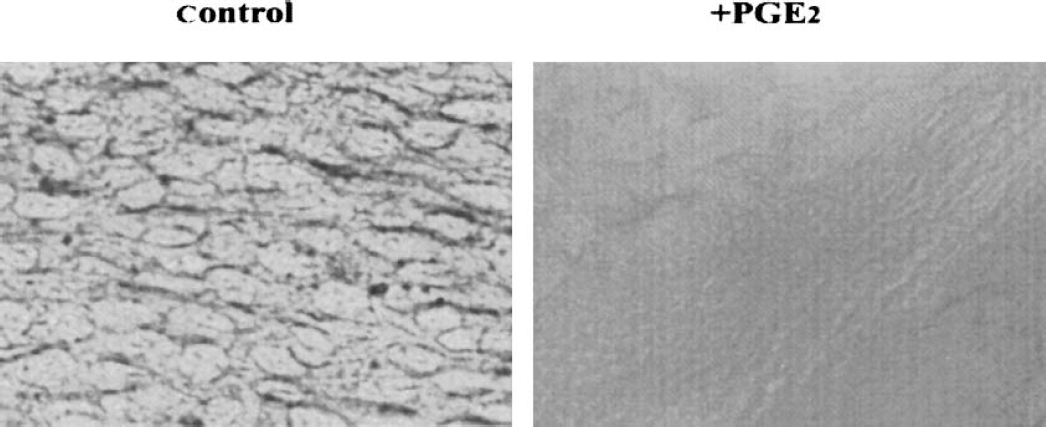
Loss of ZO-1 immunoreactivity from leptomeningeal cells exposed to PGE2. Cells were incubated for 3 days in FBS-containing medium in the absence (control) or presence of 0.1 μM PGE2. They were then fixed and subjected to immunocytochemical staining for ZO-1. ZO, zonula occludentes; PG, prostaglandin; FBS, fetal bovine serum.
DISCUSSION
In the present study, we examine the effects of a wide range of compounds, including growth factors, cytokines, peptides, and hormones, on PGD synthase secretion. Among them, only proinflammatory cytokines (IL-1β and TNFs) and LPS caused a prominent increase in the release of PGD synthase from cultivated leptomeningeal cells. Characterization of the dose-response relation for the effect of IL-1β on PGD synthase release revealed a bell-shaped curve, with the maximal response apparent at a concentration of 4 ng/mL (Fig. 2). On the other hand, a TNF-α concentration of 500 ng/mL was required to elicit an effect of similar magnitude. Because the physiologic concentration of proinflammatory cytokines in CSF is in the pg/mL to ng/mL range (Schoening et al., 2002), these data appear consistent with the presence of a high concentration of IL-1 receptor in the leptomeninges (Ban, 1994;Marquette et al., 1995). Similar results have been observed in human fetal astrocytes: the proinflammatory cytokines and LPS increase colony stimulating factor mRNA levels in the order of IL-1 > TNF-α > LPS (Lee et al. 1993). It has been also shown that IL-1β is more effective than TNF-α in inducing meningitis or BBB injury (Anthony et al., 1997;Quagliarello et al., 1991;Schoening et al., 2002). Immunoblot analysis of the culture medium revealed that IL-1β did not induce substantial changes in the abundance of any other major proteins released by leptomeningeal cells (data not shown), suggesting that its effect on the secretion of PGD synthase is specific.
These results were unexpected, given that IL-1β is well known to induce an increase in PGE synthase and COX-2 expression in vascular endothelial cells that line the BBB, resulting in release of the rather inflammatory PGE2 (Mancini et al., 2001;Yamagata et al., 2001). RT-PCR analysis confirmed that IL-1β increases the amount of PGD synthase mRNA in cultured leptomeningeal cells (Fujimori et al., 2003). This effect was apparent after a lag period of approximately 24 hours and persisted for at least 3 days (Fig. 3). This time course was consistent with that for the release of PGD synthase into the culture medium (Fig. 2). The amount of β-trace protein (PGD synthase) in rat CSF was previously shown to peak at 2 days and to have returned to basal levels by 5 days after an intraperitoneal injection of LPS (Ishizaka et al., 2001). In contrast, the events that lead to the production of PGE2 by brain vascular endothelial cells are apparent as soon as 1 hour and have almost returned to basal levels within 1 day of exposure of animals to LPS. For example, PGE synthase mRNA is first detected in the blood vessels of rat brain 1 hour after intraperitoneal injection of LPS, reaching a maximum at 2.4 to 4 hours and returning to normal levels within 5 hours (Yamagata et al., 2001). Taken together with these previous observations, our data suggest that, although IL-1β initiates events leading to the expression of PGE synthase in endothelial cells and to that of PGD synthase in leptomeningeal cells at about the same time, the actual production of PGD synthase in the BCSFB occurs much later than that of PGE synthase in the BBB. This is in agreement with the finding (Gilroy et al., 1999) that COX-2 has antiinflammatory properties; the synthesis of PGD2 is increased in late phases of inflammation and is associated with resolution, and PGE2 is produced during the early phases of inflammation.
In the first experiment, we found that exposure of leptomeningeal cells to 10 μM PGD2 significantly reduced the secretion of PGD synthase into the culture medium (Fig. 1). The induction of COX-2 and subsequent synthesis of PGE2 in endothelial cells by IL-1β is known to be dependent upon the translocation of nuclear factor-κB (NF-κB) to the nucleus (Newton et al., 1997). The antiinflammatory activity of the PGD2 metabolite 15dPGJ2 is mediated through the modification of the IκB kinase linked to NF-κB activation (Rossi et al., 2000) or through the activation of peroxisome proliferation-activated receptor-γ, a nuclear receptor that interferes with the translocation of NF-κB (Jiang et al., 1998). In addition, a subtype of PGD2 receptors is localized exclusively in the leptomeninges (Oida et al., 1997). From these observations, we first inferred that PGD2 or the metabolite 15dPGJ2 might inhibit the increase in PGD synthase by IL-1β through an effect upon NF-κB translocation. Unexpectedly, however, not only PGD2 but also PGE2 inhibited the stimulation of PGD synthase release by IL-1β. PGE2 was the most potent; the median inhibitory concentration of PGE2 was <10 nM, compared with values of 5 mM for PGD2 and 15dPGJ2 (Fig. 4). This is in good agreement with the presence of a subtype of PGE2 receptors on the leptomeninges (Zhang and Rivest, 1999). Given that the physiologic concentration of PGs in body fluid is in the picomolar to nanomolar range (Ram et al., 1997;Serhan et al., 1996), the concentration of PGD2 required for inhibition of PGD synthase secretion is most likely not achieved in the brain, even during inflammation. These results also suggest that inhibition of the IL-1β –induced increase in PGD synthase by PGE2 is not mediated through the modification of the translocation of NF-κB induced by the cytokine. Indeed, we found that exposure of cells to 0.1 mM PGE2 had no effect on the distribution pattern of NF-κB immunostaining in the absence or presence of IL-1β. To clarify the signaling pathway leading to the modification of PGD synthase production by PGE2, further analysis is in progress at our laboratory.
The present finding that PGE2 inhibited IL-1β-induced PGD synthase production is in agreement with those in carrageenin-induced pleurisy in rats. In this model, levels of PGD2 and 15d-PGJ2 are high at 2 hours, decrease as inflammation increases, but increase again at 48 hours (Gilroy et al., 1999). COX-2 expression peaks initially at early phases, associated with maximal PGE2 synthesis. A second increase in COX-2 expression at 48 hours is associated with minimal PGE2 synthesis and coincides with resolution. Because PGD2 and 15d-PGJ2 participate in antiinflammatory processes (Bernardo et al., 2000; Colviller-Nash et al., 1998), these prostaglandins have been thought to aid the resolution at the later phase. Thus our findings suggest that the inhibition of PGD synthesis by PGE2 takes part in the inflammatory action of PGE2.
PGE2 modified not only PGD synthase secretion but also the release of fibronectin from leptomeningeal cells, although the change in the amount of the other major proteins examined was rather small. Fibronectin is known to contribute to regulation of the migration, proliferation, and phenotype expression of many cell types, including brain endothelial cells (Isogai et al., 2001;Tilling et al., 1998). Taken together with the finding that the treatment with PGE2 caused the complete loss of ZO-1 staining (Fig. 7), this suggests that the PG is able to induce a loss of barrier function of the BCSFB. ZO-1 is localized to the networks of tight junctions between cells in the brain leptomeninges and the vasculature (Smith and Shine, 1992). Loss of ZO-1 from vascular endothelium is known to be associated with breakdown of the BBB (Dallasta et al., 1999). Overall, our data are consistent with the previous observations that IL-1β induces disruption of the barrier between the brain and the peripheral circulation through COX activation (Laflamme et al., 1999) and that PGE2 is the principal PG formed by brain cells including endothelial cells in response to IL-1β (DeVries et al., 1995). Furthermore, the time course of changes in the concentration of PGE2 in CSF is very similar to that of the infiltration of sucrose into the brain parenchyma of rats treated with LPS (Jaworowicz et al., 1998). The inflammatory mediator PGE2 thus seems to contribute, at least in part, to the IL-1β -mediated impairment of BCSFB, which is a dynamic rather than static brain structure and readily adapts to a variety of developmental, physiologic, and pathologic circumstances. It has been shown that administration of PGs such as PGE2 to the ischemic brain increases neural damage (Thornhill and Asselin, 1999). Further experiments are required to clarify this hypothesis.
Infection or inflammation causes the activation of macrophages, which are densely distributed in the BCSFB (McMenamin, 1999;Polfliet et al., 2001), and microglia within the brain and also results in the release of proinflammatory cytokines including IL-1β. The PGD synthase secreted into the CSF from the BCSFB in response to IL-1β during the later phases of inflammation may diffuse along preferential pathways such as Virchow-Robin spaces and act on distant cells, such as reactive astrocytes and activated microglia within the brain, as well as macrophages. The direction of the arachidonic acid cascade to the production of PGD2 by PGD synthase might represent part of a feedback mechanism that results in cessation of inflammatory brain responses. This feedback mechanism is probably not operative in the presence of PGE2, which is produced during the early phases of inflammation. In addition to its inhibitory action upon the secretion of PGD synthase, PGE2 most likely is involved in the breakdown of the BCSFB. The increase in the synthesis of PGE2 may be prolonged during severe infection or inflammation and contribute, at least in part, to cerebral edema, leukocyte recruitment, and extravasation of blood, which are often associated with illness. Our present results should provide a basis for the development of improved diagnostic techniques and treatments for infectious and inflammatory diseases.
