Abstract
Quantification of damage in both grey and white matter is required for comprehensive assessment of neuroprotective drug efficacy. Although methods for quantification of neuronal perikaryal damage after ischemia are well established, assessment of axonal damage has been limited. This article describes a new method for quantitation of axonal injury after middle cerebral artery (MCA) occlusion in rats and its application to the study of the antioxidant ebselen. The methodology is based on immunohistochemical detection of amyloid precursor protein (APP) accumulation in deformed, swollen axons in zones of ischemia. Sixty-five axon-rich sites throughout the MCA territory are assessed for the presence (scored 1) or absence (scored 0) of accumulated APP in axonal swellings. Scores for individual sites are summated in predefined neuroanatomic regions (e.g. corpus callosum), stereotaxic levels, or for a total hemisphere APP score. Both intra-rater and inter-rater reproducibility were high (r = 0.87 and 0.80, respectively). Ebselen (1 mg kg−1 hr−1, intravenously) significantly reduced the volume of neuronal perikaryal damage (24%, P < 0.01) and axonal damage (total APP score reduced from 27 [23.9 to 35.1, 95% CI] to 21.5 [18.2 to 23.3], P = 0.002 with ebselen treatment). In conclusion, a robust and generally applicable method is described for assessing pathologic features in myelinated fiber tracts that is sensitive for detection of drug effects on axonal damage.
The importance of both white matter and grey matter damage in experimental brain injury is being increasingly recognized (Stys, 1998; Dewar et al., 1999; Petty and Wettstein, 1999). Historically, grey matter damage induced by cerebral ischemia has attracted much more attention than white matter damage. One reason for this imbalance is the lack of a quantitative methodology for the assessment of white matter damage in vivo. Quantitative histopathology, examining neuronal perikarya in multiple hematoxylin- and eosin-stained sections, was established for the volumetric assessment of ischemic damage in grey matter after middle cerebral artery (MCA) occlusion (Osborne et al., 1987) and rapidly became the predominant method for assessing drug efficacy. This method, however, is insensitive in detecting axonal damage. Consequently, attention has focused almost exclusively on the damage to neuronal perikarya in rodent models of brain injury while the assessment of damage to axons, nerve terminals, and glial cells (oligodendrocytes and astrocytes) has been neglected. In the development of neuroprotective drugs for clinical use in stroke, the efficacy of a drug, particularly with regard to functional recovery, is ultimately dependent on its ability to protect against both grey and white matter damage.
Injury to white matter, which results in a disruption of axoplasmic flow, can be detected immunohistochemically within a few hours by the accumulation of transported proteins in axonal swellings or bulbs. Amyloid precursor protein (APP), one such protein, is carried along the nerve by fast axoplasmic flow and has been extensively validated as a sensitive marker for the detection of axonal injury (Kawarabayashi et al., 1991; Kalaria et al., 1993; Blumbergs et al., 1994; McKenzie et al., 1996). Comparative studies of transported proteins as potential markers for axonal damage (e.g., APP, ubiquitin, SNAP-25, neurofilament proteins) have also found APP to produce the most sensitive and reliable staining for axonal damage (Sherriff et al., 1994; Gentleman et al., 1995). The accumulation of APP at sites of injury, accompanied by morphologic evidence of axonal damage in the form of axonal swellings or bulbs, is regarded as evidence for irreversible axonal injury (Gentleman et al., 1995). Accumulation of APP (in axonal tracts exhibiting morphologic abnormalities) has consequently been used to characterize axonal injury after head injury (Pierce et al., 1996; Bramlett et al., 1997; Gentleman et al., 1999; Graham et al., 2000) and ischemic injury (Yam et al., 1997; Dietrich et al., 1998) in man and animals.
Previous studies have quantified the extent of axonal damage in terms of a single, global APP score (rat focal ischemia; Yam et al., 1998; Valeriani et al., 2000), APP load (human head injury; Gentleman et al., 1999), or sector score (human head injury; Blumbergs et al., 1995) in each brain. For interventional studies in experimental stroke (e.g., drug treatment) where an increase or reduction in axonal damage is to be investigated, detailed information on regional heterogeneity in the severity of axonal damage and in any change induced by the intervention is also required. We have, therefore, identified the individual strengths of the existing methods and combined them to provide a straightforward and reproducible method specifically for assessing drug action in rodent models of focal cerebral ischemia. The method, which provides information on both the neuroanatomic location and extent of axonal injury, has been validated for both permanent and transient MCA occlusion models and has been applied to the study of a new intravenous formulation of the antioxidant ebselen to examine the sensitivity of the method in detecting drug-induced changes in axonal damage in permanent focal cerebral ischemia.
MATERIALS AND METHODS
General surgical preparation
Adult male Sprague-Dawley rats (300 to 370 g) had free access to laboratory pellet diet and water until the day of operation. All procedures were carried out under license from the Home Office and the approval of the University Ethical Review Panel and were subject to the Animals (Scientific Procedures) Act 1986. Anesthesia was induced with 5% halothane and subsequently maintained with 1.0% to 1.9% halothane in a nitrous oxide-oxygen (2:1) mixture. The rats were intubated transorally and artificially ventilated via a small animal respirator pump. The right femoral artery and vein were cannulated for continuous physiologic monitoring and, in the drug study, for administration of ebselen or vehicle. Arterial pressure was monitored throughout the experiment and arterial blood samples were taken at regular intervals for assessment of respiratory status using a direct-reading electrode system (Bayer, Newbury, Berkshire, U.K.). Rats were maintained normotensive (MABP > 80 mm Hg), normocapnic (36 < Pa
Production of focal cerebral ischemia
The left MCA was exposed using a modification of the method described by Tamura et al. (1981). Briefly, through a 2-cm skin incision, the temporalis muscle was incised and stripped subperiosteally from the lateral and ventral aspects of the temporal bone to enter the infratemporal fossa from the foramen opticum rostrally to the foramen ovale caudally. A small subtemporal craniectomy was made, centered 3-mm rostral to the foramen ovale, and the dura opened by a linear incision using a 25-gauge needle. Cerebral ischemia was then induced by electrocoagulation of the MCA from a point proximal to the origin of the lenticulostriate artery to a distal point where it crosses the inferior cerebral vein. The MCA was then transected at the olfactory tract to ensure completeness of the occlusion. The time of transection was taken as the exact time of MCA occlusion. The craniectomy wound was then sutured and the animal was allowed to recover from the anesthesia. A subcutaneous injection of 2 mL of saline was given to prevent postanesthetic dehydration. During the development phase of the method, histologic material from the intraluminal thread model was also used to assess the method (Imai et al., 2001).
Ebselen study
Rats were divided into two groups to receive either ebselen or the vehicle (control group). Intravenous ebselen (1 mg kg−1 hr−1, n = 8) or vehicle infusion (n = 7) was started 45 minutes before permanent MCA occlusion and maintained until the end of the experiment, 4 hours after MCA occlusion. During this period, physiologic variables were continuously monitored and the animal's behavior observed.
Tissue processing and histologic quantification of ischemic damage
The rats were perfusion-fixed for neuropathologic analysis with PAM (4% paraformaldehyde in phophate-buffered saline [PBS]). Briefly, the rats were deeply anesthetized with 5% halothane and placed in a supine position so that the thorax could be opened through a bilateral incision. A catheter was inserted into the left ventricle, the right atrium was incised, and heparinized saline was infused at a pressure equal to the MABP (90 to 110 mm Hg) of the animal until the perfusate from the right atrium was bloodless. The saline was followed by approximately 300 mL of PAM. The rat was decapitated immediately after perfusion fixation, and the head stored in the fixative for at least 24 hours. The brain was then removed. After detaching the hindbrain, the forebrain was processed, embedded in paraffin wax, and sectioned at multiple levels. Sections, stained with hematoxylin and eosin, were examined by light microscopy by one of us (D. I. G.) without knowledge of the experimental protocol. Areas of ischemic damage to neuronal perikarya (Fig. 1A) were delineated at eight preselected levels from anterior 10.50 mm to anterior 1.02 mm (Brierley and Graham, 1987; Osborne et al. 1987), drawn on scale diagrams (x3.36 actual size; Fig. 2A) of forebrain based on the atlas of Konig and Klippel (1963), and measured on an image analyser (MCID, M4; Imaging Research, St Catherine's, Ontario, Canada). Areas were then integrated, with the known distance between each coronal level, to determine the total volume of neuronal perikaryal damage in each specimen.
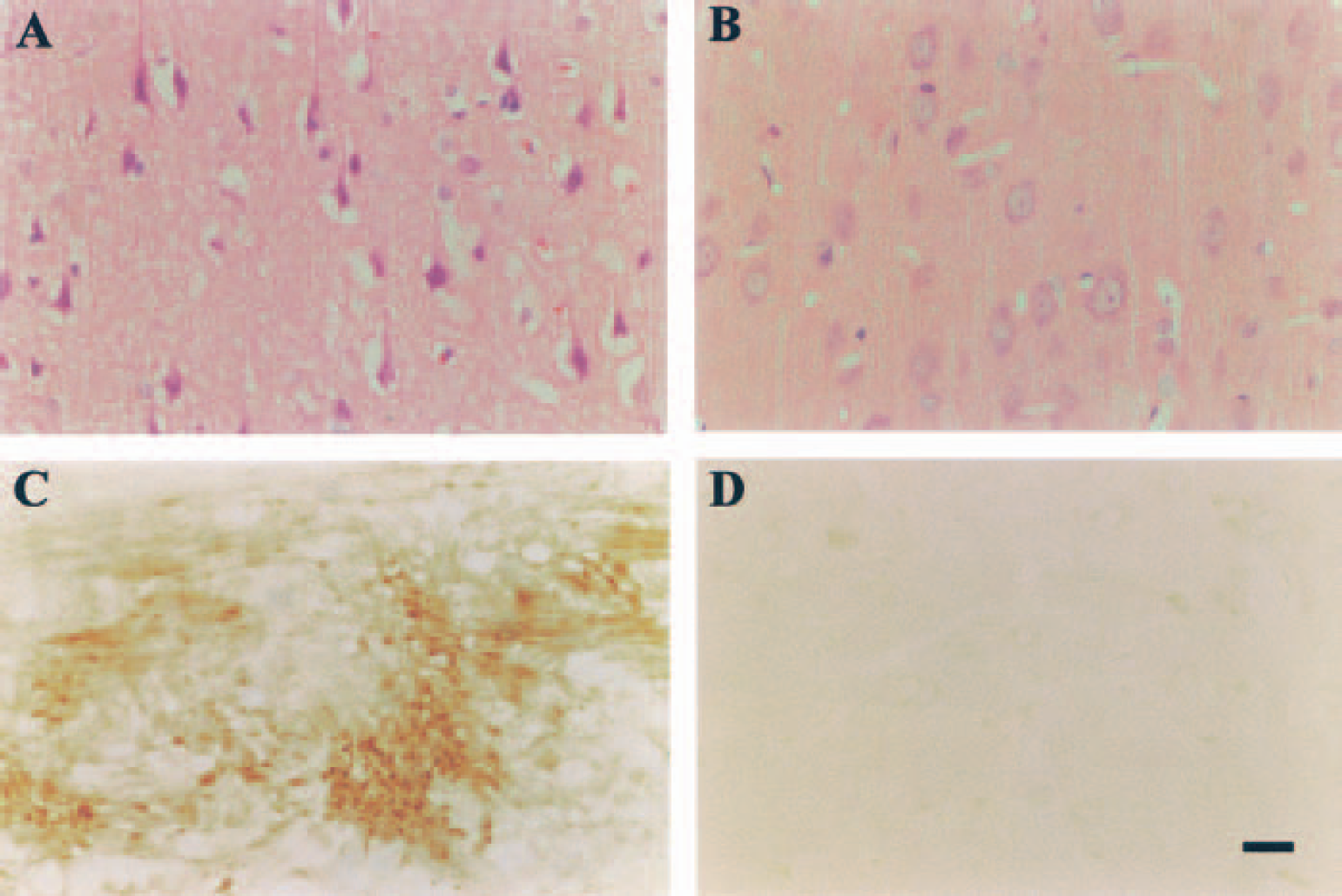
Histologic features.
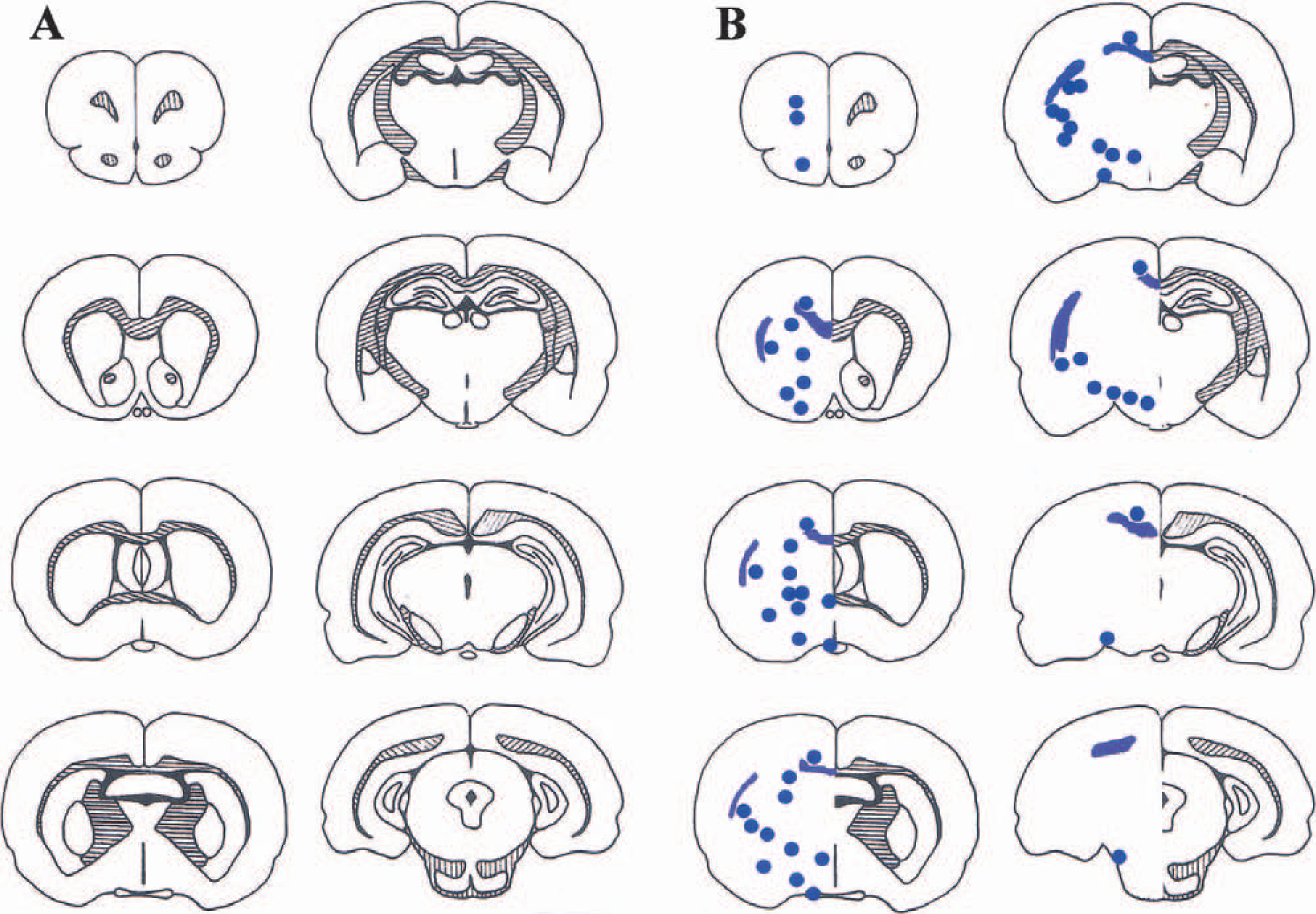
Neuronal perikaryal and axon damage assessment.
Immunohistochemistry
Adjacent sections at the eight preselected coronal levels (Osborne et al., 1987) were processed for immunohistochemistry as follows. Sections were mounted on Poly-L-Lysine (Sigma-Aldrich Company Ltd., Poole, Dorset, U.K.)-coated slides and dried at 37°C overnight. To remove the wax, the sections were placed in Histoclear (National Diagnostics, Hull, U.K.) for 20 minutes, then dehydrated by placing them in absolute alcohol for 20 minutes. Sections were microwaved for 10 minutes in 10 mmol/L citric acid (pH 6.0), allowed to cool at room temperature for 60 minutes, incubated in 3% H2O2 in methanol for 30 minutes, followed by 50 mmol/L PBS (pH 7.2) containing 0.5% bovine serum albumin and 10% normal horse serum for one hour. Monoclonal antibody to APP (Clone 22C 11; Roche Molecular Biochemicals, Mannheim, Germany) was diluted 1:300, applied to sections, and incubated overnight at 4°C. The sections were washed in PBS (2 × 20 minutes), the secondary antibody (biotinylated horse antimouse, 1:100; Vector Laboratories, Burlingame, CA, U.S.A.) was applied for 1 hour, and the sections were washed again (2 × 20 minutes). The avidin/biotinylated horseradish peroxidase complex (ABC kit; Vector Laboratories) was then applied for 1 hour. The washing procedure with PBS was repeated and the sections were allowed to develop in 3,3‘-diaminobenzidine solution (Vector Laboratories) for 3 minutes. Finally, the sections were dehydrated, cleared, and mounted for light microscopical analysis. A negative control, in which the primary antibody was omitted from the procedure, was included in the protocol, and minimal staining was detected. All further analysis was performed by an investigator unaware of the treatment in each animal.
Production of the data collection sheet and quantification of axonal damage
Predefined regions, rich in axons, were selected, that is, corpus callosum (CC), external capsule (EC), internal capsule (IC), anterior commissure (AC), median forebrain bundle (Mfb), optic tract (Opt), fornix (Fnx), caudate putamen (CP), and globus pallidus (GP). Large regions were subdivided according to the classification in the stereotaxic atlas of Paxinos and Watson (1986) or divided into dorsal, ventral, medial, or lateral quadrants. For example, CC was subdivided into 5 regions (forceps minor, genus corpus callosum, cingulum, splenium corpus callosum, and forceps major) and CP was subdivided into 4 quadrants (dorsal, ventral, medial, and lateral) per coronal section. Each region was further assessed within each of the 8 stereotaxic coronal levels (Osborne et al., 1987) where that region was present. Overall, 65 individual sites contributed to the assessment of axonal injury in each brain (Fig. 2B). A score of 0 (no APP accumulation) or 1 (APP accumulation in axonal swellings, e.g. Fig. 1C) is assigned to each site and marked onto a data collection sheet for each animal (Fig. 3). Amyloid precursor protein scores were calculated for the entire hemisphere (total APP score), individual regions (adding scores along horizontal rows), or a particular coronal level (adding scores in the appropriate vertical column). Each data collection sheet represents the axonal damage for a single animal. Animals from the vehicle group in the ebselen study were used to generate the data to characterize axonal damage after proximal MCA occlusion.
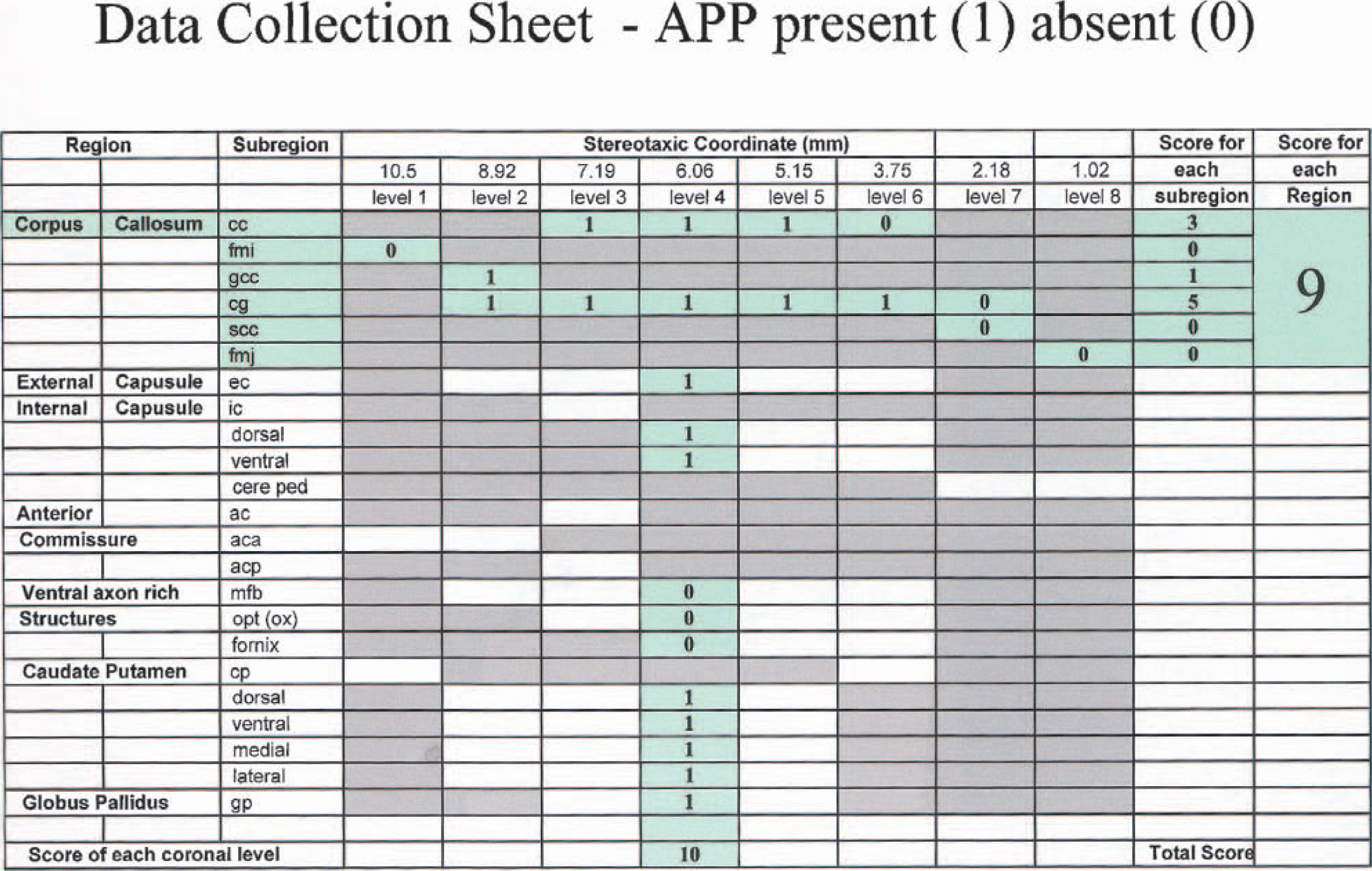
Data collection sheet for collection of information on axonal damage. The example demonstrates how scores for a particular neuroanatomic region (corpus callosum) or a particular coronal level (6.06 mm from the interaural line) are calculated. Summation of scores at all 65 sites provides the total amyloid precursor protein score for the hemisphere.
Reproducibility
The reproducibility of the method was first tested by an intra-rater study in 9 animals exposed to focal cerebral ischemia. Independent scores were made by the same investigator on two occasions, and the first rating was compared with the second rating by analysis of two randomly selected regions—the CC and IC. Stratified statistical analysis was then carried out to study the variability in scoring between the first and second occasion for the APP score for the 2 regions (n = 18), the score for each of the component subregions (n = 90), and the score for each individual site (n = 207).
An inter-rater study followed, where the IC was chosen and ranked by a second rater. Reproducibility was assessed from the correlation of the severity of the axonal pathology within the IC for rater 1 compared with rater 2. All reproducibility analyses (intra- and inter-rater) were performed in a “blinded” manner.
Statistical analysis
The hypothesis that was examined was whether ebselen altered axonal damage; total APP score was the primary end point. Axonal damage is presented as median (95% confidence intervals) and analyzed by the Mann-Whitney test. Intra-rater and inter-rater comparisons of axonal damage were assessed by Spearman rank correlation. Neuronal perikaryal damage is presented as mean (± SD) and analyzed by Student's unpaired two-tailed t-test. A P value of 0.05 was considered to be significant.
RESULTS
Neuronal perikaryal damage
All brains were well perfusion-fixed, as evidenced by good neuronal morphology in the contralateral hemisphere (Fig. 1B), the absence of intravascular blood, and the lack of cytological artefacts such as “dark cells.”
Ischemic damage to neuronal perikarya was observed only within the territory of the occluded MCA (i.e., the ventrolateral cerebral cortex and caudate nucleus). Neuronal perikarya within these regions showed the morphologic characteristics of ischemic damage, that is, micro-vacuolation, shrinkage, and triangulation of nucleus and cytoplasm, and increased eosinophilia of cytoplasm (Fig. 1A). A sharp boundary could be readily distinguished between areas where neuronal perikaryal damage was present and areas where it was absent, and this boundary was delineated onto eight preselected scale drawings (Fig. 2A). The volume of neuronal perikaryal damage induced by proximal occlusion of the MCA was 130 ± 9.6 mm3 in the cerebral hemisphere, of which damage of 94 ± 10.3 mm3 was in the cerebral cortex and 28 ± 2.4 mm3 in the caudate nucleus.
Axonal damage
The hemisphere contralateral to the occluded MCA displayed diffuse APP immunoreactivity with no evidence of APP accumulation or axonal pathology (Fig. 1D). In the hemisphere ipsilateral to the occluded MCA, intense APP immunoreactivity was present in myelinated fiber tracts permeating the caudate nucleus, in the EC, and dorsal IC inter alia (Fig. 1C). The intense APP immunoreactivity was associated with gross segmental axonal swellings or bulb formation.
The presence or absence of intense APP immunoreactivity in deformed axons was assessed at 65 predefined sites (Fig. 2B) and data collected on a scoring sheet (Fig. 3). In animals exposed to MCA occlusion the total APP score for the ipsilateral hemisphere was 27 (23.9 to 35.1; median, 95% CI), indicating that intense APP staining was present in just under half of the 65 sites examined. When the eight coronal planes were examined, the highest median APP score of 7 (5.52 to 7.33, 95% CI) was observed at the coronal level 6.06 mm anterior to the interaural line (Fig. 2B, 4th level on LHS), indicating that intense APP immunoreactivity was present at 7 of the 13 sites examined at this coronal level. When the individual regions were examined throughout the MCA territory, the highest median APP score of 13 (11.7 to 15.2, 95% CI) was observed in the caudate nucleus (Fig. 2B), indicating that intense APP staining in axons was present at 13 of the 17 caudate nucleus sites examined. In the CC, the median APP score was 5 (3.7 to 5.7, 95% CI), indicating that intense APP immunoreactivity was present in 5 of the 14 sites examined within the CC.
Excellent intra-rater reproducibility was achieved for the generation of APP scores. Using CC and IC data, APP scores for region and subregion were r = 0.87 (P < 0.0001) and r = 0.89 (P < 0.0001), respectively, with a slightly lower correlation coefficient for replication at a single site (r = 0.82, P < 0.0001; Fig. 4). Of the 207 sites that were examined, the same result (presence or absence of APP immunoreactivity in deformed axons) was obtained in 189 (91%) of the replications performed.
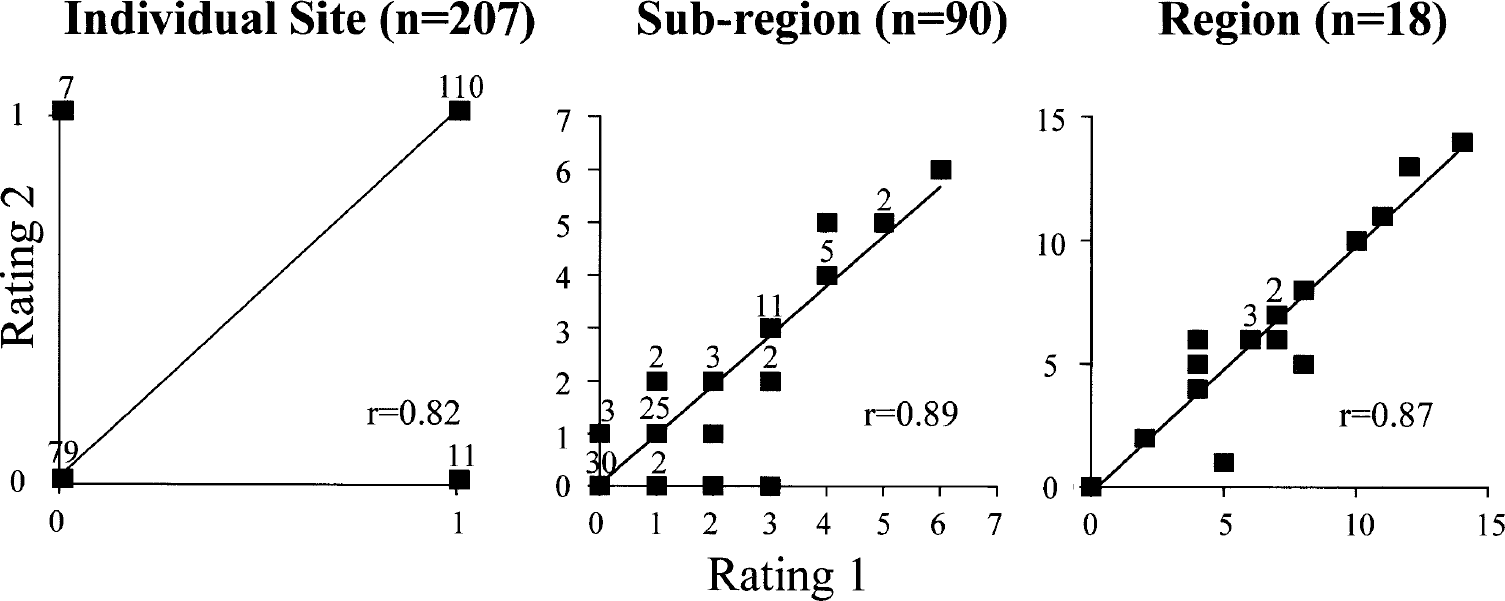
Intra-rater reproducibility. Two independent scores were made by the same investigator on two occasions; the first rating was compared with the second rating by linear regression analysis. Data are presented for two randomly selected regions, corpus callosum and internal capsule (n = 9 per region), their component subregions, and individual sites (as detailed in Fig. 3). Numbers above each symbol (■) indicate the number of data points at that position.
Inter-rater reproducibility between 2 investigators also demonstrated good agreement in scoring (r = 0.80, P < 0.0001). Reproducibility was slightly poorer than for the equivalent intra-rater value (r = 0.87), most probably reflecting differences in defining the threshold for APP presence between investigators (i.e., the level of APP staining and axon pathology set by the investigator to merit a score of 1).
Ebselen and ischemic pathology in axons and perikarya
Physiologic variables
Physiologic variables, monitored for arterial blood gases, plasma glucose, and body temperature, were within the normal range and were comparable between ebselen- and vehicle-treated animals. Blood pressure during anesthesia and in the conscious animal was not affected by ebselen. Overt behavioral effects of ebselen were not present in conscious animals, and deterioration in general condition was not observed in animals of either group during the experiment.
Neuronal perikaryal damage
The intravenous administration of ebselen (1 mg kg−1 hr−1) significantly reduced the volume of neuronal perikaryal pathology after MCA occlusion by 24% (P < 0.01). The reduction in damage in the ipsilateral hemisphere was due to a reduction in cerebral cortex damage (Fig. 5A). Neuronal perikaryal damage in the caudate nucleus after MCA occlusion was minimally altered by ebselen.
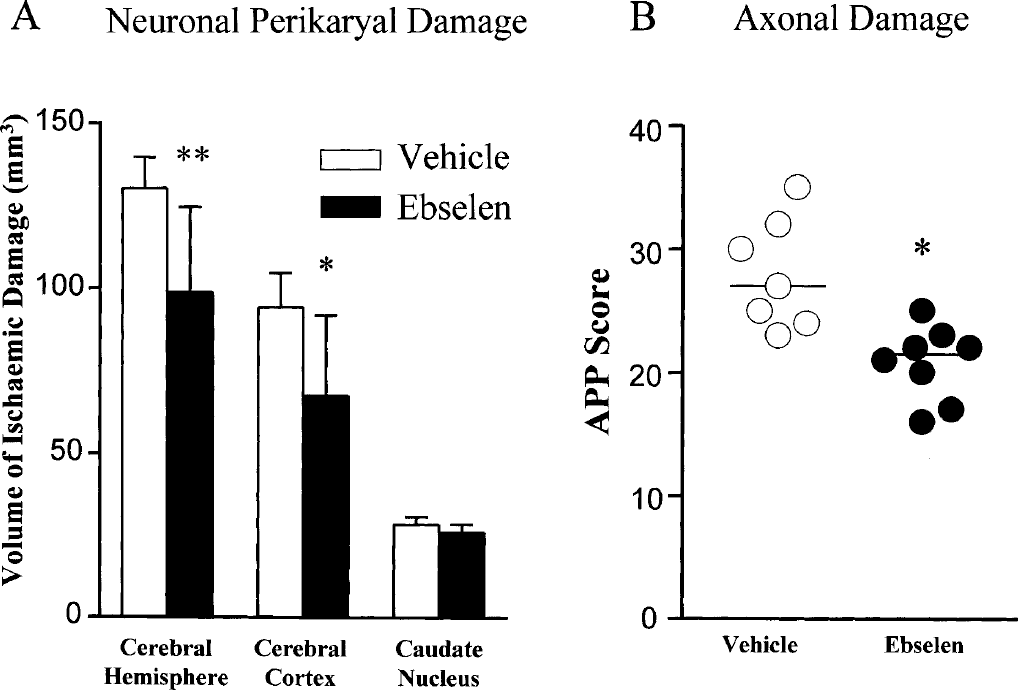
(A) Ebselen significantly reduced the volume of neuronal perikaryal damage in the cerebral hemisphere and cortex compared with the vehicle control group (mean ± SD, *P < 0.02; **P < 0.01, two-tailed unpaired Student's t-test).
Axonal damage
The intravenous administration of ebselen significantly reduced the total hemisphere APP score from 27 (23.9 to 35.1, 95% CI) to 21.5 (18.23 to 23.27, 95% CI) (P = 0.002; Fig. 5B). This attenuation in axon pathology in the hemisphere ipsilateral to the occluded MCA was mainly due to a reduction in APP scores in the CC, the dorsal IC, and the GP (Fig. 6B). Axonal damage (and APP scores) in the caudate nucleus and EC was minimally altered by ebselen. Regional assessment of axonal damage (Fig. 6B) provided greater insight into the location of ebselen-induced protection of white matter than did analysis of the rostrocaudal distribution of axonal pathology (Fig. 6A) in this study.
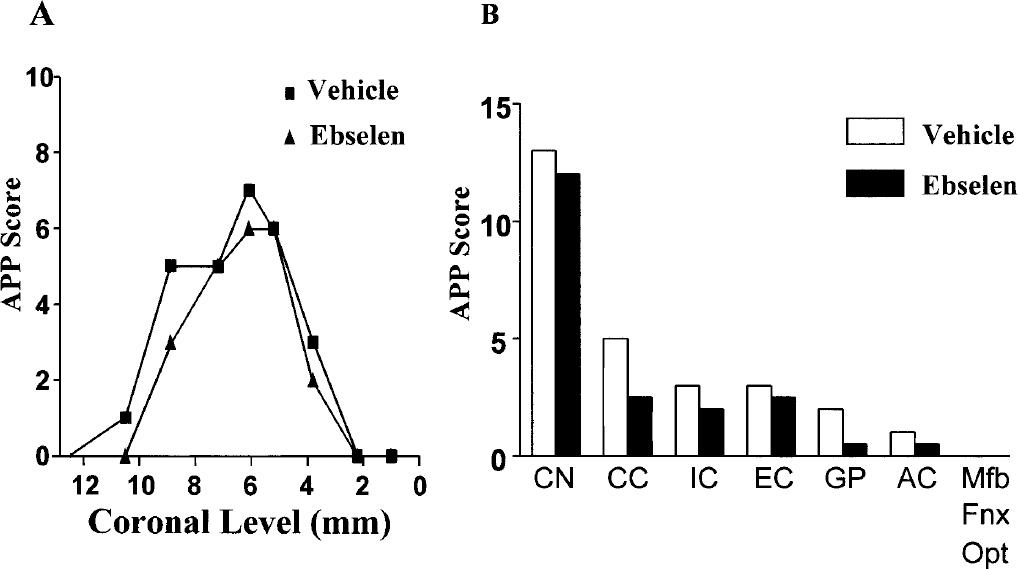
Axonal damage in ebselen- and vehicle-treated rats: extent of axonal damage
DISCUSSION
The failure of many classes of neuroprotective drugs in clinical stroke trials has led to reappraisal of the pertinence of animal models for the preclinical testing of drugs (De Keyser et al., 1999; STAIR, 1999). One valid criticism of the assessment of ischemic damage in animal models is the omission of white matter damage. The lack of a generally applicable, objective, and reproducible methodology for the assessment of ischemic damage to white matter has focused research efforts on grey matter damage and drugs specifically designed to protect neuronal perikarya (e.g., glutamate antagonists and uptake enhancers). Many of these drugs (e.g., NMDA antagonists), however, may be ineffective in attenuating ischemic damage to axons and nerve terminals (Yam et al., 2000).
In white matter, oligodendrocytes and axons comprise the functional unit of the myelinated fiber. Although a variety of approaches for assessing axonal damage have been described, to date none have been entirely satisfactory and no single method has become widely used in experimental stroke research.
Two general approaches have been followed. The first approach identifies white matter pathology by selective staining of myelin (e.g., Luxol fast blue-periodic acid Schiff), axons, (Bielschowsky's silver stain) (Wakita et al., 1995; Schäbitz et al., 2000), or oligodendrocytes (proteolipid protein mRNA or tau-1 immunohistochemistry for injured oligodendrocytes) (Dewar and Dawson, 1995; Mandai et al., 1997). Assessment of the severity of white matter changes is made by optical density measurements, graded scoring systems of axon pathology, or the extent of oligodendrocyte pathology. The second approach identifies accumulation of proteins (commonly APP) normally transported along the axon. Amyloid precursor protein, transported by fast axoplasmic flow, accumulates at the point of axonal damage and microtubule disruption in swollen axon profiles and can be detected within 4 hours of permanent MCA occlusion (Fig. 1C). In terms of detecting the damaged axon, accumulation of APP in axon bulbs is presently regarded as the most sensitive approach for assessing ischemic and traumatic axonal injury (Graham et al., 2000) and has been used previously for detection of axonal damage in the rat and cat after focal ischemia (Yam et al., 1997, 2000; Valeriani et al., 2000). The quantification of axonal damage associated with stroke and head injury has previously been assessed by either manual or automated methods in terms of a total “APP load” (Gentleman et al., 1999) or total APP score (Yam et al., 1997; Valeriani et al., 2000). These approaches, however, do not provide detailed information on the particular white matter tracts that have been affected by injury. The advantage of the present method is that, in addition to quantifying axonal damage in terms of a total APP score, it can be mapped out topographically and APP scores generated for specific white matter regions or subregions and at different rostrocaudal levels. These features are extremely useful because—as is clear from studying CBF levels in white matter after MCA occlusion (Komjati et al., 2001)—the severity of ischemia and extent of axonal pathology varies considerably in different white matter regions. The topographical profile of ischemia is also different in the various models of focal ischemia (e.g., electrocoagulation of the MCA versus the intraluminal filament approach). The intraluminal filament model may also induce ischemia and axonal pathology in the territory of additional arteries that are more proximal branches of the internal carotid artery (e.g., the anterior choroidal artery, which arises just proximal to the MCA bifurcation) (He et al., 1999). When the APP method was used to study the pattern of axonal damage induced by transient intraluminal filament-induced ischemia (Imai et al., 2001), axonal pathology was identified in additional white matter tracts (e.g., ventral internal capsule, median forebrain bundle, fornix, and optic tract), which were not affected after diathermy occlusion of the MCA as used in the present study.
To study the consequences of an intervention on axonal damage (e.g., drug treatment), therefore, a method is required that allows assessment of the overall severity and the neuroanatomic location of axonal damage. Using APP immunohistochemistry, Blumbergs et al. (1995) devised a sector scoring method for the assessment of axonal injury in human head injury, dividing the whole brain into 116 sectors. We have applied a similar approach in the rat forebrain, carefully identifying 65 individual sites to give an optimal assessment of white matter damage in both permanent and transient MCA occlusion models involving direct occlusion (e.g., electrocoagulation) or blockade of the origin (intraluminal filament) of the MCA.
The strengths of this new method include: (1) Neuroanatomic added value. In addition to quantitation of total white matter damage, it (the method) provides information on the pathology in discrete white matter tracts, which is advantageous for the study of neuroprotective drugs, behavioral outcomes, and functional recovery (Fig. 7). (2) Simplicity of the scoring system. For axonal damage at each site examined, the decision of whether APP accumulation is present (1) or not (0) is straightforward. (3) Reproducibility. Reproducibility is very high both for an individual rater examining the same material on a number of occasions or for different raters examining the same material. Reproducibility also improves as values for the 65 individual sites are added together to provide data on the 23 subregions and the 9 regions (Fig. 4). (4) Sensitivity. It can detect, for example, a reduction in axonal damage in discrete axonal tracts with drug treatment (Fig. 6). (5) Flexibility. Topographical maps of neuronal perikaryal and axonal damage from an individual animal can be viewed simultaneously and the maps used to assess the influence of a particular intervention or drug (Fig. 7). Adding up the APP score horizontally or vertically on the APP data collection sheet provides data on individual neuroanatomic regions or data for each coronal level, respectively (Fig. 3). The latter facility, used routinely to display grey matter damage, can therefore be used to compare the effects of a drug on the amount and rostrocaudal extent of both grey and white matter damage. The total APP score, generated by adding the 65 individual scores together, provides a global value for white matter damage within the hemisphere, which can complement data on the total volume of neuronal perikaryal damage. (6) Applicability. The individual sites have been selected specifically to allow sensitive assessment of white matter damage in a range of MCA occlusion models and can be used to highlight differences in axonal pathology between different models (e.g., diathermy occlusion of the MCA and intraluminal thread model).
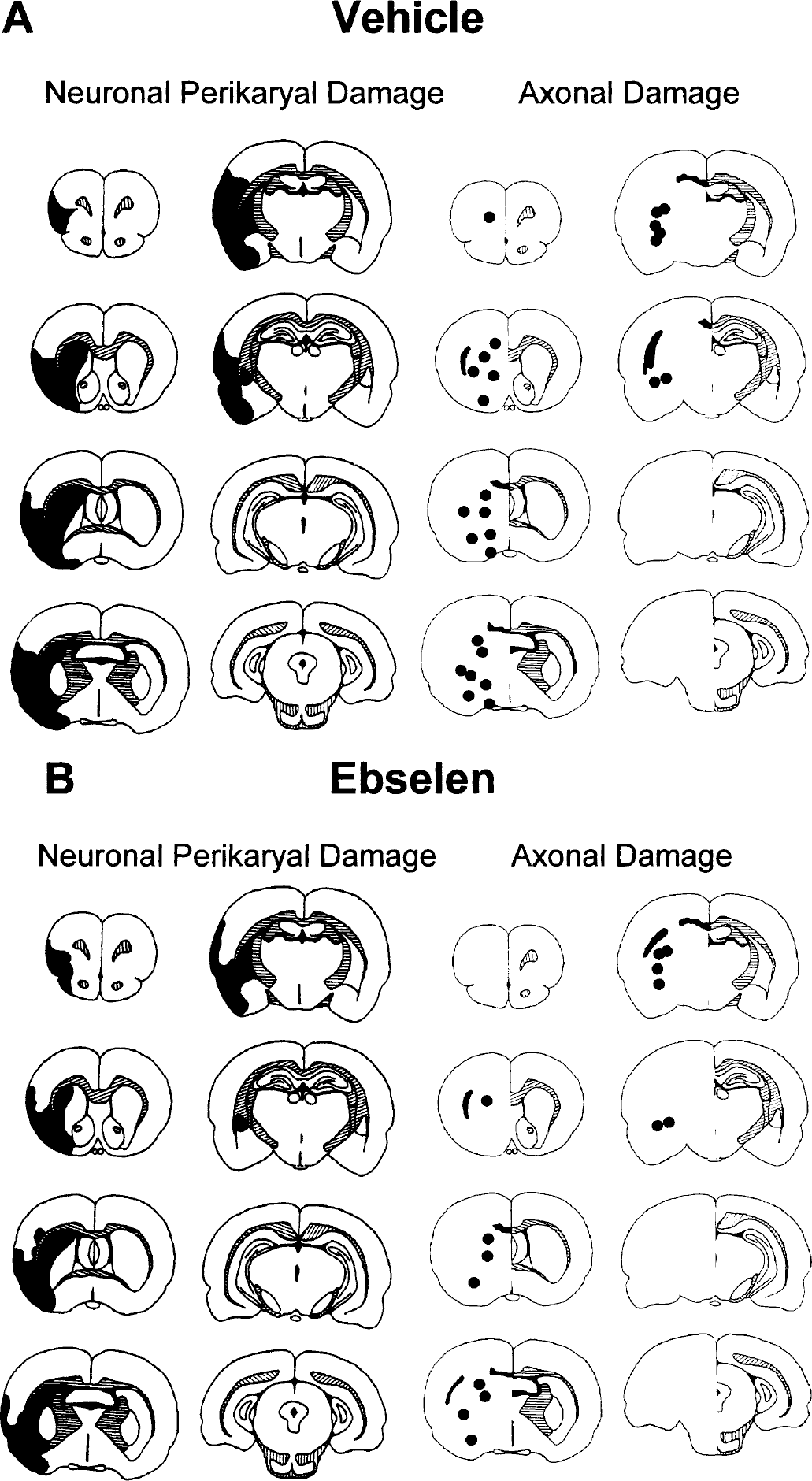
Topography of neuronal perikaryal damage and axonal damage in a representative animal from the vehicle group (
The method has been applied to the study of a new, intravenous formulation of the antioxidant ebselen in a standard model of permanent MCA occlusion. Free radicals are generated in both grey and white matter in the ischemic core, and in periischemic, penumbral regions, which may receive supplementary flow from collateral blood vessels. In addition, the high lipid content of myelinated axon tracts increases their vulnerability to lipid peroxidation produced by free radical attack. Cytotoxic by-products of free radical damage, such as 4-hydroxynonenal, are known to be toxic to both axons and oligodendrocytes (McCracken et al., 2000). Antioxidants, therefore, have the capacity to protect both grey and white matter from oxidative stress. Assessment of APP accumulation in the present study showed that ebselen was able to protect axons from an ischemic insult. The most marked reductions in white matter damage were found in periinfarct regions such as CC, IC, and GP, whereas damage in white matter regions experiencing the most severe ischemia (Komjati et al., 2001), such as in the fiber tracts of the CP, the EC, and AC, were influenced less by the drug (Fig. 6B). Ebselen also produced a significant attenuation of neuronal perikaryal damage (24% salvage of hemisphere), which was principally due to a reduction in cortical damage (28%; Fig. 5A). A failure to protect tissue in the severely ischemic dorsolateral caudate nucleus is a common finding with this model and is consistent with a previous study using the oral formulation of ebselen in the same experimental model (Takasago et al., 1997).
For axonal damage, therefore, zones that can be salvaged by pharmacologic intervention and zones that cannot be salvaged appear to be present in white matter. Whether the presence of salvageable and nonsalvageable areas within oligemic white matter reflects differences in the severity of CBF reduction (as is the case in grey matter) (Heiss, 2000) remains to be established.
The main neuroprotective mechanism of ebselen has been linked to its interaction with the thioredoxin system (Zhao et al., 2002) and its ability to mimic glutathione peroxidase and phospholipid hydroperoxide glutathione peroxidase (Sies, 1993; Sies and Parnham, 2000), reducing hydrogen peroxide (H2O2) to H2O and O2. Moreover, in vitro studies have revealed ebselen-induced inhibition of a number of free radical-producing enzymes including inducible nitric oxide synthase (Hattori et al., 1994), 5- and 15-lipoxygenases (Schewe et al., 1994), and NADPH oxidase (Cotgreave et al., 1989). Mitochondria, as well as representing a source of free radicals in ischemia, are also at risk of injury from reactive oxygen species with peroxidation of membrane lipids, a possible key component in mitochondrial dysfunction (Bindoli, 1988). Ebselen has been shown to protect mitochondria from lipid peroxidation through its direct antioxidant properties (Boireau et al., 1999). Ebselen attenuates the toxic effects of 4-HNE generated by lipid peroxidation (Malecki et al., 2000) and reduces glutamate excitotoxicity by acting as a modulator of the NMDA receptor redox site in neurones in culture (Herin et al., 2001). In a recent in vivo focal ischemia study, further evidence of the antioxidant potential of ebselen was revealed by the significant reduction in the volume of tissue immunopositive for the DNA-damage marker 8-hydroxy-2‘-deoxyguanosine and the lipid peroxidation marker 4-hydroxynonenal in ebselen-treated animals (Imai et al., 2001). Ebselen therefore displays a range of actions that attenuate the production of, and the damage induced by, all the major free radicals and oxidizing agents generated in ischemic tissues.
In conclusion, this article describes a reproducible, flexible, and sensitive method for the detection of axonal damage in focal cerebral ischemia. It has been applied to the study of the antioxidant ebselen and demonstrates that pretreatment with this antioxidant is capable of significantly reducing (by 26%) white matter damage, principally in the CC, IC, and GP. This, combined with the significant reduction in grey matter damage (24%), demonstrates ebselen-induced antioxidant effects consistent with significant protection of both cell bodies and axons, and without evidence of significant adverse effects, in a recognized model of MCA occlusion.
Footnotes
Acknowledgements
The authors are grateful to Mrs. L. Gallagher and other staff from the Wellcome Surgical Institute and Department of Neuropathology for technical support.
