Abstract
Alzheimer disease (AD) is an illness that can only be diagnosed with certainty with postmortem examination of brain tissue. Tissue samples from afflicted patients show neuronal loss, neurofibrillary tangles (NFTs), and amyloid-β plaques. An imaging technique that permitted in vivo detection of NFTs or amyloid-β plaques would be extremely valuable. For example, chronic imaging of senile plaques would provide a readout of the efficacy of experimental therapeutics aimed at removing these neuropathologic lesions. This review discusses the available techniques for imaging amyloid-β deposits in the intact brain, including magnetic resonance imaging, positron emission tomography, single photon emission computed tomography, and multiphoton microscopy. A variety of agents that target amyloid-β deposits specifically have been developed using one or several of these imaging modalities. The difficulty in developing these tools lies in the need for the agents to cross the blood-brain barrier while recognizing amyloid-β with high sensitivity and specificity. This review describes the progress in developing reagents suitable for in vivo imaging of senile plaques.
The diagnosis of Alzheimer disease (AD) currently depends on the patient having impairments in memory function and at least one other cognitive domain, to the extent that it impairs daily function. This behavioral definition subsumes essentially all causes of dementia, and clinical evaluation is necessary to differentiate AD from the numerous other conditions that may impair cognitive function. Generally this evaluation is targeted at “ruling out” other, potentially treatable causes of dementia. Laboratory studies are used to evaluate for metabolic disorders, and frequently a neuroimaging study is used to evaluate the possibility of, for example, stroke, brain tumor, and normal pressure hydrocephalus. Atrophy in a neuroimaging study is viewed as consistent with AD, but is generally not diagnostic. Functional studies of cerebral blood flow and metabolism including single photon emission computed tomography (SPECT) and positron emission tomography (PET) are not routinely used at present for clinical evaluation. Currently, no single test can be considered alone as definitive evidence of the presence of AD.
Why is diagnosing AD so difficult? The major issue is that the neuropathologic changes of AD are microscopic lesions, too small for standard neuroimaging approaches to detect. Structural magnetic resonance imaging (MRI) and blood flow studies depend on gross changes in brain structure and function that suggest major atrophy or a functional alteration that has already occurred; these markers are secondary phenomena and may not be ideal for following patients over time or for diagnosing very early, subtle changes. For example, while whole brain MRI can detect widespread atrophy or even provide insight into enlargement of the lateral ventricle at the expense of the hippocampus and entorhinal cortex (Fox et al., 1996; Killiany et al., 2000), clinical-pathologic studies show that substantial neuronal loss has already occurred by the time gross atrophy is detectable by MRI (Gomez-Isla et al., 1996). PET measures of cerebral metabolism show promise as a biomarker. PET [18F]fluorodeoxyglucose studies have been reported to add to diagnostic and prognostic accuracy in the clinical evaluation of AD (Silverman et al., 2001), but still detect an indirect measure of disease presence and progression.
The ideal imaging agent for AD would directly reflect the disease process, have high signal-to-noise ratio so that even small amounts of neuropathologic abnormality can be detected, and be useful in tracking patients over time (Klunk et al., 1998). Indeed, an imaging agent that reflected the amount of either amyloid-β or neurofibrillary tangle (NFT) burden, or both, would be advantageous, especially because such a test could be used to evaluate individuals with the earliest stage of the neuropathologic alterations of AD (Arriagada et al., 1992) rather than waiting until behavioral manifestations can be detected. Moreover, a biomarker that closely tracked with the neuropathologic alterations of the disease would be useful for evaluating the efficacy of treatments aimed at reversal or stabilization of lesions directly (van Dyck, 2001). Since current therapeutic trials depend on waiting for several years to determine whether treatment strategies alter the rate of progression of dementia, such an imaging strategy would have the effect of jump-starting the drug discovery process for AD. Hence, a good deal of effort has gone into developing strategies for Alzheimer diagnostics, and for amyloid-β imaging in particular.
Hydrophobic molecular imaging probes have been studied in order to develop such a diagnostic test (Friedland et al., 2000; Kurihara and Pardridge, 2000; Skovronsky et al., 2000; Agdeppa et al., 2001; Bacskai et al., 2001b; Klunk et al., 2001; Schmidt et al., 2001; Zhuang et al., 2001a; Lee et al., 2002; Marshall et al., 2002; Mathis et al., 2002). While many probes are able to bind to the relatively hydrophobic amyloid-β senile plaques in slides of tissue from patients with AD, the challenge of developing probes that both specifically bind amyloid-β and are able to cross the blood-brain barrier (BBB) has proven formidable. In this review, we outline our approaches to this problem, bringing to bear the recently developed tool of multiphoton microscopy to analyze staining of plaques in vivo in transgenic models of cerebral amyloid-β deposition.
TECHNIQUES FOR IMAGING AMYLOID-β IN VIVO
Imaging techniques suitable for detection of senile plaques are listed in Table 1, and will be described briefly. First, positron emission tomography (PET) is a very sensitive in vivo imaging technique capable of detecting nanomolar quantities of receptors throughout the body (Jacobs and Cherry, 2001; Silverman and Phelps, 2001). If the radioligand-amyloid–β interaction is analogous to that of classic radioligand-receptor interactions, then PET imaging techniques should be easily capable of detecting amyloid-β at the micromolar amounts found in human AD brains. The requirements for successful amyloid detection and quantification include the availability of a suitable amyloid-β-specific radioligand that incorporates a positron-emitting radionuclide within its structure (e.g., C-11 or F-18). The production of most positron-emitting radionuclides requires an on-site cyclotron, although longer-lived (110-minute half-life) F-18-labeled radiopharmaceuticals can be (and presently are) shipped from regional distribution sites to PET imaging centers. The spatial resolution of PET is relatively low, on the order of 6 mm, and approaches 2 mm with micro-PET animal scanners. The average diameter of a senile plaque in AD tissue is 0.06 mm, and a single plaque contains many millions of potential binding sites for amyloid-imaging tracers. Therefore, much like PET receptor imaging is unable to detect individual receptors, PET amyloid imaging will not be able to resolve individual plaques. Instead, PET imaging should provide a measure of the average amyloid density in different brain regions and demonstrate differences between regions containing high and low densities of amyloid deposits. An advantage of PET is that imaging results in quantitative measurements suitable for detecting subtle changes in disease states.
Comparison of techniques for in vivo imaging of amyloid-β
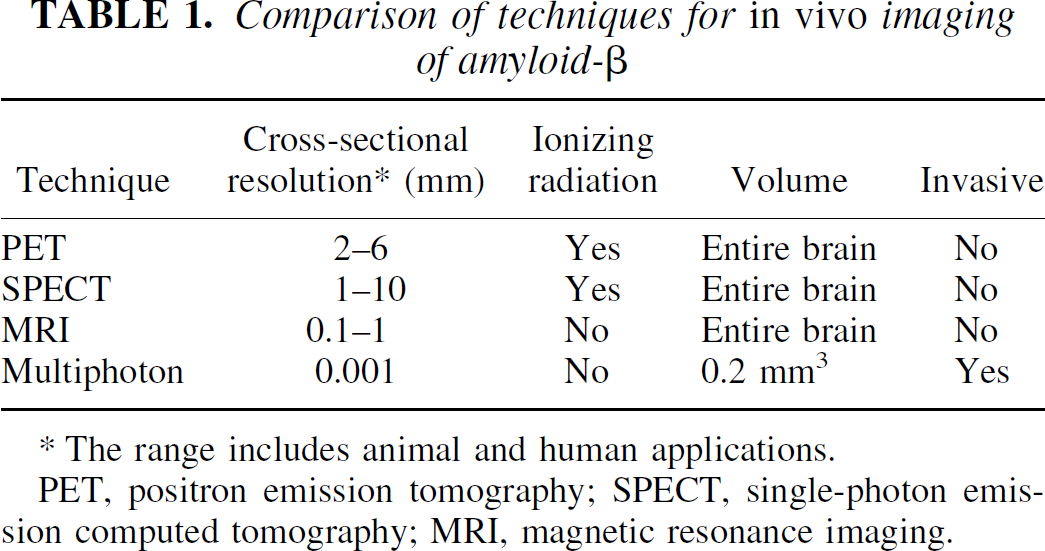
The range includes animal and human applications.
PET, positron emission tomography; SPECT, single-photon emission computed tomography; MRI, magnetic resonance imaging.
A more widely available technique, SPECT, relies on radionuclides with longer half-lives (e.g., Tc-99m and I-123) (Camargo, 2001; Wirrwar et al., 2001). The resolution of SPECT cameras has approached that of PET, and SPECT pinhole imaging techniques have achieved resolutions less than 1 mm (Weber et al., 1994; Ishizu et al., 1995). The sensitivity of these techniques is lower than that of PET, and SPECT data are not as readily quantifiable as those for PET.
MRI can lead to noninvasive, high-resolution structural imaging without the need for administration of exogenous material, or may require the injection of contrast enhancement media (Cavagna et al., 1997). The spatial resolution of MRI is much higher than PET or SPECT, being about 1 mm for human applications and under 0.1 mm for high-field animal scanners. Sophisticated manipulation of pulse sequences has allowed imaging cerebral vasculature, diffusion processes, and indirect measures of cerebral blood flow. The major disadvantage of MRI is low sensitivity, usually requiring the imaged molecule to be present in the high micromolar to low millimolar range. One way to partially circumvent this is to detect regional differences in the relaxation time (T2*) of water molecules locally perturbed by exogenous paramagnetic contrast agents such as gadolinium (Gd) and monocrystalline iron oxide nanoparticles (MION). In addition, endogenous brain pathology can cause local T2* changes that may be detectable. Several large technological hurdles need to be crossed before MR technology can be applied to in vivo amyloid imaging in animals or humans.
Multiphoton microscopy allows very high resolution (<0.001 mm) imaging of microscopic structures with high sensitivity (Gratton et al., 2001; Williams et al., 2001). This light microscope-based technique depends on detecting fluorescence (either autofluorescence or from a contrast reagent), so no radioactive tracers are necessary. The technique uses a pulsed near-infrared laser to excite fluorescence for deeper penetration into scattering tissue than visible light microscopy, and optical tomography with a restricted excitation volume. The technique depends on an invasive technique to allow access to the brain, and imaging depths of about 500 μm are achievable. This leads to a very small imaging volume within the cortex compared with the size of the brain. Fluorescent contrast reagents with high specificity result in very high contrast images of immunohistochemically defined microscopic structures. No clinical applications of multiphoton microscopy have been developed, but recent progress in imaging of animal models may provide a springboard to clinical use.
AGENTS FOR IN VIVO IMAGING OF AMYLOID-β
Imaging of senile plaques may be possible without contrast reagents, that is, by taking advantage of the intrinsic properties of the amyloid-β deposit, or the local microenvironment of the plaque (i.e., T2* changes of local water molecules). Benveniste et al. (1999) reported detection with MRI of amyloid-β deposits in postmortem AD brain by a special T2* pulse sequence not requiring an exogenous contrast agent. This MRI study was done with postmortem tissue, but suggests that in vivo imaging may be possible with significant future technical advances.
Agents that recognize amyloid-β deposits should improve imaging contrast and detection sensitivity. Suitable agents for in vivo detection of amyloid-β deposits must meet several important criteria. First, the compound must bind to amyloid deposits with high specificity. Second, the reagent must get into the brain in sufficient amounts to allow detection. This generally requires that the compound be relatively small and lipophilic. The reagent should bind with high enough affinity to allow retention of the compound specifically bound to amyloid deposits while the free and nonspecifically bound compound clears from brain. A list of contrast reagents successfully used for detecting amyloid-β in vivo is presented in Table 2.
Agents for in vivo imaging of amyloid-β
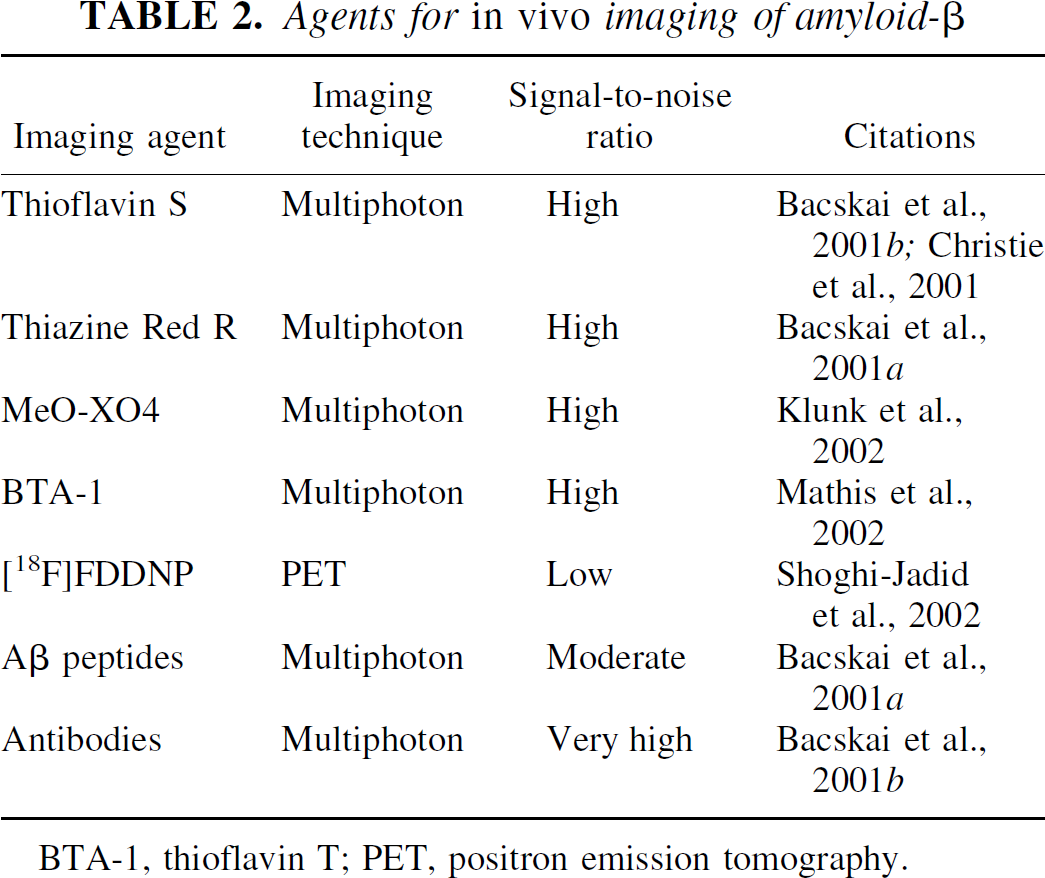
BTA-1, thioflavin T; PET, positron emission tomography.
The development of specific contrast reagents generally starts with existing compounds that target amyloid-β. These compounds are then modified to be suitable for in vivo imaging. Senile plaques are composed primarily of amyloid-β, a 40- to 42-aa self-aggregating peptide derived from the amyloid precursor protein (APP). The amyloid-β peptide itself, therefore, is a plaque-binding compound. Modified amyloid-β, or peptide fragments of amyloid-β that bind to plaques in vitro, have been evaluated as contrast reagents for in vivo imaging (Maggio et al., 1992; Ghilardi et al., 1996; Lee et al., 2002). Unfortunately, peptides are rapidly degraded in the blood, and entry across the BBB is poor. Putrescine modifications of these peptides, and the use of all D-enantiomers of the amino acids, improves brain access and decreases degradation, respectively, but not to an extent to allow sensitive detection of plaques in vivo (Poduslo et al., 1999; Wengenack et al., 2000; Wisniewski et al., 2001). Wisniewski et al. (2001) have labeled Aβ with MION, for detection with MRI. While no in vivo studies have been reported, this group has presented preliminary data from transgenic APP mice that have had their BBB disrupted to allow entry of the large MION-Aβ complex. After killing the animals and removing the brain, they were able to detect brain areas that partially corresponded to amyloid-β deposits in immunostained brain sections. This study required both transient disruption of the BBB for brain entry and detection using postmortem tissue.
Brain entry of I-125-labeled amyloid-β was also demonstrated in a transgenic mouse, but detection required killing the animal, sectioning the brain, and exposing the tissue sections to film for long times (Wengenack et al., 2000). Using multiphoton microscopy, senile plaques were targeted and detected in vivo after direct application of a fluorescein-labeled amyloid-β (1–40) to the cortex of a live transgenic mouse (Bacskai et al., 2001a). Although this approach required preparation of a craniotomy, the results indicate that exogenous amyloid-β peptide can target existing plaques in vivo if brain delivery is sufficient. Together, these studies indicate that advances in brain entry, detection sensitivity, or both are needed for practical in vivo imaging with amyloid-β peptides.
Attempts have been made to image amyloid using a small molecule labeled with 18F based on a 6-dialkylamino-2-naphthylethylidene derivative. This compound, [18F]FDDNP, labeled senile plaques and NFTs in tissue sections from AD brain (Agdeppa et al., 2001); however, specificity for these neuropathologic lesions in vivo is not clear. When administered to humans, PET scanning revealed good brain entry, and reduced exit of the probe in AD patients as compared to controls (Shoghi-Jadid et al., 2002). Unfortunately, nonspecific accumulation of the very lipophilic [18F]FDDNP is high in white matter areas. [18F]FDDNP initially accumulates most in pons (an area without significant numbers of plaques or tangles) and least in hippocampus (an area of severe pathology). Five to 10 minutes after injection, the radioactivity in the pons exceeded that in the hippocampus by 1.6-fold. Differential clearance then resulted in a reversal to a 1.4-fold increase in residual radioactivity in hippocampus over the pons. This ratio is very low for a PET imaging probe, especially in a patient in an advanced stage of the disease, leaving little room for detection of early stages of amyloid-β deposition. While this is the only reported PET imaging study of amyloid-β in humans to date, the very low specific to nonspecific binding ratio of this tracer will likely limit its practical application.
In vivo imaging in experimental animal models has been accomplished with fluorescent derivatives of thioflavin T and Congo red. Both of these compounds have been used as histochemical stains for fibrillar amyloid-β for decades (Kelenyi and Thioflavin, 1967). Derivatives of Congo red [methoxy-XO4 (Klunk et al., 2002)] and thioflavin T [BTA-1 (Mathis et al., 2002)] were engineered to cross the BBB while retaining their specificity for amyloid-β deposits. Both derivatives are small, lipophilic, and enter the brain rapidly. Multiphoton microscopy was used to image the labeled plaques in transgenic mouse brains 30 minutes after intravenous injection, or 24 hours after intraperitoneal injection. The fluorescent reagents specifically labeled senile plaques and cerebrovascular amyloid (no NFTs are present in this mouse model). Figure 1 is a collapsed projection of a three-dimensional volume of the brain of a living transgenic mouse 24 hours after intraperitoneal injection of methoxy-XO4, imaged with multiphoton microscopy, using 750-nm light to excite the probe. This image reveals numerous senile plaques and cerebrovascular amyloid angiopathy. Figure 2 shows a side-on view of a three-dimensional reconstruction of a volume of the brain in a 20-month-old transgenic mouse 24 hours after an intraperitoneal injection of methoxy-XO4. The fluorescent probe is detectable for up to 3 days after a single administration, and can be reapplied for serial imaging studies. These reagents have near ideal properties for in vivo imaging of amyloid-β deposits in the brain, and multiphoton microscopy demonstrates directly that the compounds label immunohistochemically defined amyloid-β deposits in transgenic mice overexpressing mutant human APP. Radiolabeled versions of methoxy-XO4 and BTA-1 may be excellent candidates for PET imaging in AD patients.
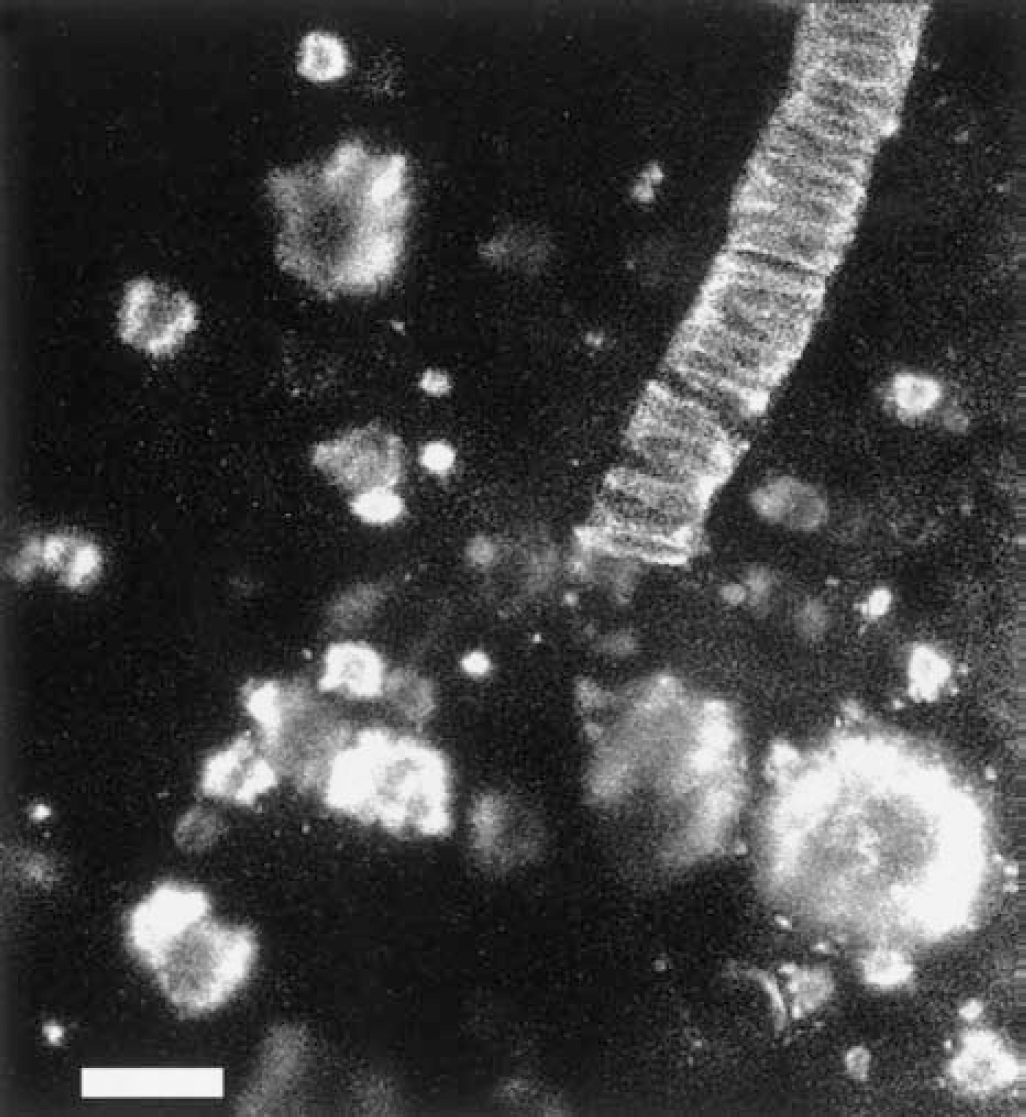
Multiphoton microscopy was used to detect amyloid-β deposits in living transgenic mice after systemic administration of a Congo red derivative, methoxy-XO4 (Klunk et al., 2002). Twenty-four hours after intraperitoneal injection (10 mg/kg) the mouse was anesthetized and prepared for imaging, by creating a “thin-skull” bone window, as previously described (Christie et al., 2001). Using 750-nm light from a femtosecond-pulsed Ti:sapphire laser, images of methoxy-XO4 fluorescence were acquired up to 250 μm deep from the surface of the brain. This image is a collapsed projection of 60 images at progressively deeper planes of focus. The fluorescent dye was bound to numerous amyloid-β plaques as well as amyloid angiopathy in this 20-month-old PS-APP transgenic mouse (Klunk et al., 2002). (Scale bar = 35 μm.)
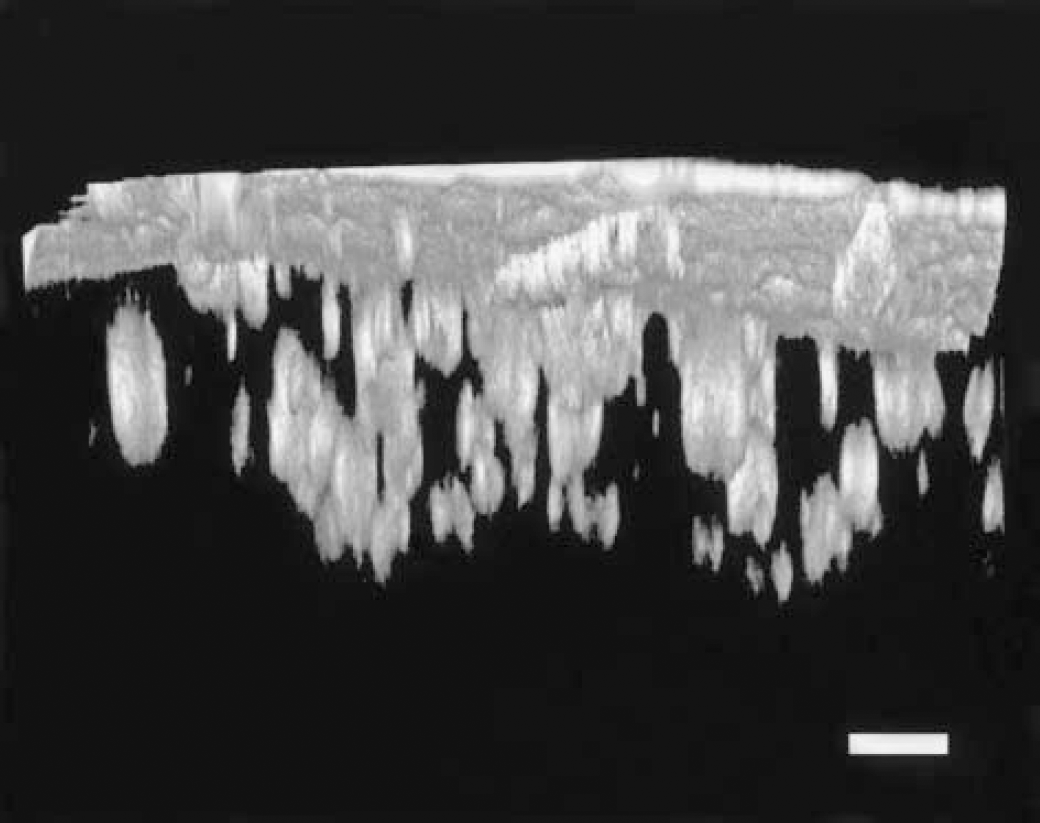
Three-dimensional imaging of a volume of the brain can be obtained with multiphoton microscopy in a living transgenic mouse. A 20-month-old PS-APP mouse was given an intraperitoneal injection of 10 mg/kg methoxy-XO4 24 hours prior to imaging. The figure is a side-on view of an imaging volume obtained with a 20x objective (NA = 0.45, Olympus) that extends to 300 μm deep from the surface of the cortex. Autofluorescence from the thinned skull is seen as a solid plane at the top of the figure. Note the resolution of multiple amyloid-β plaques at various depths. Cerebrovascular amyloid angiopathy can be detected at the surface, just underneath the thinned skull. (Scale bar = 50 μm.)
Related derivatives of Congo red, with less ideal in vivo properties, have also been described. One compound, termed BSB, shows specific staining of NFTs, senile plaques, and cerebrovascular amyloid deposits in tissue sections, and was also able to target plaques in transgenic mice when administered peripherally (Skovronsky et al., 2000). This study also relied on postmortem detection of the plaques in processed tissue, and was therefore subject to the same caveats as was discussed above for the detection of I-125-labeled amyloid-β peptides.
Very specific detection of amyloid-β deposits can be obtained with antibodies targeted against the amyloid-β peptide. Antibodies enter the CNS in very small amounts when administered peripherally and have not yet been successfully used for in vivo brain imaging studies. However, fluorescently labeled anti-amyloid-β antibodies were applied directly to the brains of transgenic mice and were detected with multiphoton microscopy. The antibodies labeled amyloid-β deposits sensitively and specifically within a few hundred micrometers from the surface of the cortex (Bacskai et al., 2001b). Surprisingly, this approach also led to rapid clearance of the labeled deposits, confirming the utility of an immunotherapeutic approach to treating AD. It is possible that manipulations that increase the peripheral access of labeled antibodies into the central nervous system would allow the use of an antibody for in vivo imaging.
DISCUSSION
Several different approaches to neuroimaging have been used to aid in the diagnosis of AD and to act as additional markers of progression of illness. To date, MRI volumetric imaging, PET [18F]fluorodeoxyglucose, and SPECT cerebral blood flow studies have had the most clinical use. Under appropriate conditions, these approaches can provide very strong diagnostic, and even predictive, information (de Leon et al., 1997; Juottonen et al., 1998; Bobinski et al., 1999; De Toledo-Morrell et al., 2000; Killiany et al., 2000; Xu et al., 2000; Dickerson et al., 2001; Silverman and Phelps, 2001). Regional cerebral blood flow, glucose utilization, and functional MRI measurements are able to distinguish Alzheimer from control patients (de Leon et al., 2001; De Santi et al., 2001; Kato et al., 2001; Obrien et al., 2001; Silverman and Phelps, 2001). These remain important technologies and, with improvements in techniques and resolution, no doubt will provide additional information regarding cerebral changes in dementias and in AD.
Application of these noninvasive imaging techniques to the evaluation of AD may be significantly enhanced by the development of probes that specifically and selectively detect amyloid-β in the brain. While amyloid-imaging agents are in various stages of development for MRI, SPECT, and PET, all of these imaging modalities have resolutions that are insufficient to distinguish individual senile plaques. Imaging studies with these modalities would therefore provide some average measure of plaque density in various brain regions, similar to current PET neuroreceptor studies.
Amyloid-β imaging probe development has been hampered by the need to have a small molecule that crosses the BBB and remains selective and specific in its binding to amyloid-β. Some compounds have been developed, based on the observation that amyloid-β has a very high affinity interaction with itself. Thus probes based on amyloid-β structure itself may well be attractive, and limited success has been described using modified amyloid-β peptides (Kurihara and Pardridge, 2000; Bacskai et al., 2001a; Lee et al., 2002). A second approach uses histologic dyes that are known to bind amyloid, such as Congo red or thioflavin T. Several such molecules, modified to enhance BBB permeability, show promise as PET or SPECT ligands (Skovronsky et al., 2000; Agdeppa et al., 2001; Klunk et al., 2001; Schmidt et al., 2001; Zhuang et al., 2001a, b ; Mathis et al., 2002).
In this review we highlight the use of modified derivatives of fluorescent histologic dyes, and adapt the new technology of multiphoton microscopy to the challenge of imaging senile plaques in vivo, in transgenic models of AD. Multiphoton microscopy has the advantage of having an inherent resolution of about 1 μm, far surpassing MRI, SPECT, or PET. The long wavelength light is not harmful to tissues, and allows deeper penetration into tissue than standard visible or UV approaches. We are able to obtain dramatic images of well-resolved, individual plaques, and use this technique in mouse models to test therapeutics (Bacskai et al., 2001b; Christie et al., 2001). However, the imaging requires either thinning or removing the portion of the skull through which the light passes to diminish light scattering. Therefore, this technique is not yet useful for clinical applications. Nonetheless, the in vivo resolution provided by multiphoton microscopy is unparalleled, and this technique will be critical in delineating the specific structures labeled in vivo by amyloid-imaging probes ultimately destined for clinical use with the less invasive techniques such as MRI, SPECT, and PET. It is clear that the era of direct imaging of neuropathologic changes, including senile plaques, has arrived in terms of transgenic mouse models, and may be approaching from a clinical perspective as well.
Footnotes
Acknowledgements
We thank Stephen Kajdasz and Drs. Jeffrey Stanley and Kanagasabai Panchalingam for helpful discussions of this article.
