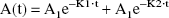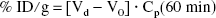Abstract
Aβ1–40 is a potential peptide radiopharmaceutical that could be used to image the brain Aβ amyloid of Alzheimer disease in vivo, should this peptide be made transportable through the blood–brain barrier in vivo. The blood–brain barrier transport of [125I]-Aβ1–40 in a transgenic mouse model was enabled by conjugation to the rat 8D3 monoclonal antibody to the mouse transferrin receptor. The Aβ1–40–8D3 conjugate is a bifunctional molecule that binds the blood–brain barrier TfR and undergoes transport into brain and binds the Aβ amyloid plaques of Alzheimer disease. App SW /Psen1 double-transgenic and littermate control mice were administered either unconjugated Aβ1–40 or the Aβ1–40–8D3 conjugate intravenously, and brain scans were obtained 6 hours later. Immunocytochemical analysis showed abundant Aβ immunoreactive plaques in the brains of the App SW /Psen1 transgenic mice and there was a selective retention of radioactivity in the brains of these mice at 6 hours after intravenous administration of the conjugate. In contrast, there was no selective sequestration either of the conjugate in control littermate mouse brain or of unconjugated Aβ1–40 in transgenic mouse brain. In conclusion, the results show that it is possible to image the Aβ amyloid burden in the brain in vivo with an amyloid imaging agent, provided the molecule is conjugated to a blood–brain barrier drug-targeting system.
The Aβ amyloid burden in Alzheimer disease (AD) correlates with dementia and memory deficit in humans (Cummings and Cotman, 1995) and AD transgenic mice models (Hsiao et al., 1996). The accumulation of Aβ amyloid plaque in the brain is a specific neuropathologic hallmark of AD (Glenner et al., 1981; Selkoe, 1986). There is no premortem diagnostic test that is specific for AD at present, nor is there a brain scan that will allow for quantitation of the Aβ amyloid burden in AD. The latter would be useful not only in the diagnosis of AD, but also in evaluating the therapy of AD with drugs intended to reduce the Aβ-amyloid burden in brain.
The Aβ amyloid in the brain could be imaged either with single-photon emission computed tomography and a radiopharmaceutic imaging agent, or with magnetic resonance imaging and a magnetopharmaceutic imaging agent. Three types of amyloid imaging agents are presently being developed: (1) small molecules that are derivatives of Congo Red and bind to the β-pleated amyloid structure (Dezutter et al., 1999; Skovronsky et al., 2000), (2) anti-Aβ monoclonal antibodies that bind to specific amino acid sequences within the Aβ1–42/43 peptide comprising the amyloid plaques (Majocha et al., 1992; Walker et al., 1994), and (3) Aβ1–40 peptide radiopharmaceuticals that bind to preexisting Aβ amyloid by a nucleation-polymerization mechanism (Maggio et al., 1992; Jarrett and Lansbury, 1993). However, none of these three classes of central nervous system amyloid imaging agents crosses the brain capillary endothelial wall, which forms the blood–brain barrier (BBB) in vivo. Because the BBB is intact in AD (Schlageter et al., 1987; Munoz et al., 1997; Vorbrodt et al., 1997), and because the amyloid imaging agents do not cross the BBB, no amyloid brain scan has been developed to date. Conversely, if the amyloid imaging agent was conjugated to a BBB drug-targeting system, (Saito et al., 1995; Wu et al., 1997), and the amyloid imaging agent was able to enter brain from blood, then it might be possible to develop an AD brain scan for the semiquantitation of the Aβ amyloid burden in the brain of patients with AD.
The present investigation uses a BBB drug-targeting technology to deliver a [125I] Aβ1–40 peptide radiopharmaceutical across the BBB in double-transgenic mice that overexpress the amyloid peptide precursor (APP) with the Swiss mutation (App SW ) and presenilin-1 (Psen1) (Takeuchi et al., 2000). Chimeric peptides are formed when a nontransportable peptide (e.g. the amyloid imaging agent) is conjugated to a BBB transport vector (Pardridge, 2001). The latter is an endogenous peptide, modified protein, or peptidomimetic monoclonal antibody (MAb) that undergoes receptor-mediated transcytosis across the BBB in vivo. Previous work in rats (Saito et al., 1995) or Old World primates (Wu et al., 1997) has shown that Aβ1–40 peptide radiopharmaceuticals can be made transportable across the BBB by conjugation of the peptide to a peptidomimetic MAb that crosses the BBB. The MAb–Aβ1–40 conjugate binds to the amyloid plaques in postmortem tissue sections of the AD brain (Saito et al., 1995; Wu et al., 1997), in a manner similar to that of unconjugated Aβ1–40 (Maggio et al., 1992). Previous studies in rats used the murine OX26 MAb to the rat transferrin receptor (TfR) as a BBB transport vector for the Aβ1–40 (Saito et al., 1995). However, there are no transgenic-brain Aβ rat models available, and the OX26 MAb is not active in mice (Lee et al., 2000). Either the RI7–217 or the 8D3 rat MAb to the mouse TfR are active as brain drug-delivery vectors in mice (Lee et al., 2000), and could be used to facilitate Aβ1–40 transport into the mouse brain.
Nontransportable drugs, such as Aβ peptide radiopharmaceuticals, are conjugated to the targeting MAb with the use of avidin-biotin technology (Pardridge, 2001). In this approach, the Aβ peptide radiopharmaceutical is monobiotinylated on the amino terminus in parallel with production of a conjugate of streptavidin and the targeting MAb. The streptavidin–MAb conjugate is designated MAb–streptavidin or streptavidin–MAb, and the delivery to mouse brain of Aβ1–40 with a conjugate of streptavidin and the RI7–217 MAb has been investigated (Lee and Pardridge, 2000). However, the complex of [125I]-Aβ1–40-biotin/streptavidin-RI7–217 was rapidly degraded in vivo in mice because of biotinidase activity (Lee and Pardridge, 2000). In the present work, a conjugate of streptavidin and the 8D3 MAb was prepared and the [125I]-Aβ1–40-biotin peptide radiopharmaceutical was captured by the 8D3–streptavidin conjugate (Fig. 1A). This central nervous system amyloid imaging agent is a bifunctional molecule that binds both the BBB TfR, to mediate uptake into the brain, and the Aβ amyloid plaques in the brain of the transgenic mice. The initial studies on the pharmacokinetics and in vivo metabolic stability of the Aβ1–40–8D3 conjugate indicated that there was minimal degradation of this conjugate by biotinidase in mice in vivo, and enabled the subsequent development of an Aβ-amyloid brain scan in the double-transgenic mouse model.
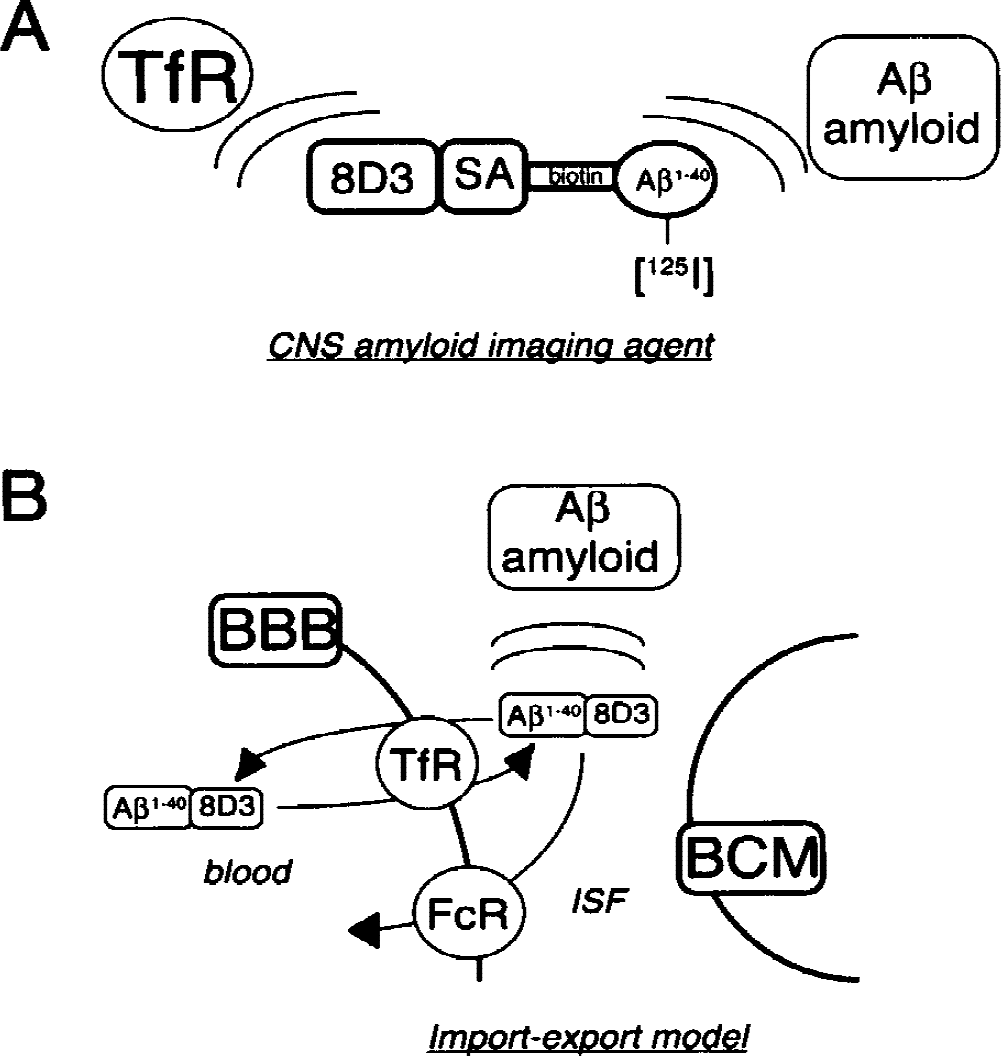
(A) The central nervous system amyloid imaging agent is a bifunctional molecule that binds to both the blood–brain barrier (BBB) transferrin receptor (TfR) and to Aβ-amyloid plaque in the brain. The Aβ1–40 is radiolabeled with 125-iodine on the tyrosine residue and is conjugated at the amino terminus with biotin to allow for high-affinity capture by a conjugate of the rat 8D3 monoclonal antibody (MAb) to the mouse TfR and streptavidin.
A further goal of the present study was to determine the efflux kinetics of the amyloid imaging agent from the brain to the blood that occurs after the initial transport of the agent from the blood to the brain. The imaging of the Aβ amyloid in the brain is based on the import-export model shown in Fig. 1B. Initially, there is import of the amyloid imaging agent into all parts of brain from blood because of receptor-mediated transport across the BBB. Once in brain interstitial fluid, the Aβ-amyloid imaging agent may then deposit at preexisting Aβ-amyloid plaques, which causes selective sequestration of radioactivity in the region of the brain amyloid. In parallel, there must be export of the amyloid imaging agent back to blood from the nonamyloid-bearing brain, so that the selective sequestration of the amyloid imaging agent in the brain may be observed. This export from the brain to the blood occurs via receptor-mediated exocytosis of the conjugate that is mediated either by the BBB TfR or by the BBB Fc receptor. The BBB TfR mediates the bidirectional movement of either transferrin or anti-TfR MAbs between the blood and brain compartments (Zhang and Pardridge, 2001a). In addition, the immunoglobulin G molecules are also ligands for the BBB Fc receptor, which selectively mediates the transport of immunoglobulin G in the brain-to-blood direction only (Zhang and Pardridge, 2001b). The imaging of the amyloid in brain is performed at delayed imaging times because of the need for the export of the amyloid imaging agent from the normal brain. Therefore, the present study describes the kinetics of the import from the blood to the brain and the export from the brain to the blood of the amyloid imaging agent.
MATERIALS AND METHODS
Materials
[125I]-Sodium iodine, autoradiographic [125I] microscales (20-μm thick), and Superose 12 High Resolution 10/30 fast protein liquid chromatography columns were obtained from Amersham-Pharmacia Biotech (Piscataway, NJ, U.S.A.). Amino-terminal biotinyl-Aβ1–40 (Aβ1–40-bio) was purchased from Quality Controlled Biochemicals (Hopkinton, MA, U.S.A.). MBS (m-maleimidobenzoyl-N-hydroxysuccinimide ester) was supplied by Pierce Chemical (Rockford, IL, U.S.A.). Recombinant streptavidin, chloramine T, and all other reagents were purchased from Sigma-Aldrich (St. Louis, MO, U.S.A.). Male BALB/c mice (weight, 20–30 g) were supplied by Harlan Sprague-Dawley (San Diego, CA, U.S.A.). Female nude mice (age, 10 weeks) were purchased from Charles River Laboratories (Wilmington, MA, U.S.A.). App SW /Psen1 double-transgenic and littermate control mice 7 to 11 months in age have been described previously (Takeuchi et al., 2000), and were provided under the Material Transfer Agreement by the Nathan Kline Institute and New York University.
Synthesis of the 8D3–streptavidin conjugate
The rat hybridoma line secreting the 8D3 MAb was provided by Dr. Britta Engelhardt, as described previously (Kissel et al., 1998). 8D3 MAb was purified by protein G affinity chromatography from ascites generated in nude mice after growth of the hybridoma line on a feeder layer of newborn mouse thymocytes and peritoneal cells in Dulbecco modified Eagle medium with 10% fetal bovine serum (Kissel et al., 1998). A 1:1 conjugate of the 8D3 MAb and streptavidin was prepared by stable thiolether linkage using 8D3 thiolated with Traut reagent and streptavidin activated with MBS, as described previously (Saito et al., 1995). A mixture of 2.2 mL 8D3 MAb (10 mg) in 0.05 mol/L sodium borate, 1 mmol/L edetic acid (pH 8.0), and 76 μL 2-iminothiolane (4 mg/mL) in 0.05 mol/L sodium borate and 0.1 mmol/L edetic acid (pH 8.0) was added, and the mixture was rocked for 60 minutes at room temperature. The reaction was quenched by adding 200 μL glycine (7.5 mg/mL) and rocked for 30 minutes at room temperature. The solution was then dialyzed for 2 hours at room temperature with stirring against 2 L 0.01 mol/L PBS and 0.1 mmol/L edetic acid (pH 7.4) using a 12-KDa molecular weight cutoff dialysis tubing. Streptavidin (6 mg) was dissolved in 2 mL 0.05 mol/L sodium borate (pH 8.5) and 32 μL MBS (24 mg/mL) in dimethylformamide was added. The solution was mixed by stirring and was incubated for 60 minutes at room temperature. The reaction was stopped by the addition of 100 μL glycine (7.5 mg/mL) and was rocked for 30 minutes at room temperature. The reaction solution was dialyzed for 2 hours at room temperature with stirring against 2 L 0.01 mol/L PBS, 0.1 mmol/L edetic acid (pH 7.0), and 12-KDa molecular weight cutoff dialysis tubing. The MBS-activated streptavidin and the thiolated 8D3 MAb were then mixed immediately and incubated for 3 hours at room temperature. [3H]-Biotin (0.093 MBq) was added, along with Tween-20 for a final concentration of 0.05%. The conjugate was purified with a 2.6 × 93 cm Sephacryl S300 column, followed by elution in 0.01 mol/L PBS and 0.05% Tween-20 (pH 7.4) at a flow rate of 30 mL/h, and 3 mL fractions were collected. The absorbance at 280 nm and [3H]-radioactivity of each fraction were measured, and the 8D3 MAb–streptavidin conjugate was separated from unconjugated 8D3 MAb and streptavidin. The fractions 70 to 86 were pooled, concentrated, and protein was measured with the bicinchoninic acid protein assay. The 8D3–streptavidin conjugate was stored at −20°C until further use.
Iodination of N-biotinyl-Aβ1–40
N-biotinyl-Aβ1–40 (10 μg, 2.1 nmol) was iodinated with [125I]-sodium (74 MBq, 1.1 nmol) and chloramine T (44 nmol) at room temperature. The reaction was quenched by the addition of sodium metabisulfite (125 nmol), and 50 μL 0.01 mol/L PBS 7.4 containing 0.05% Tween-20 (PBST) was added to the reaction solution. [125I]-N-biotinyl-Aβ1–40 was purified by Sephadex G25 size-exclusion chromatography in the presence of PBST. The trichloroacetic acid (TCA) precipitability of the [125I]-N-biotinyl-Aβ1–40 was more than 96%, and the specific activity was 5.5 MBq/μg.
Pharmacokinetics and organ uptake in BALB/c mice
Adult male BALB/c mice were anesthetized with 100 mg/kg ketamine and 2 mg/kg xylazine intraperitoneally, and divided into five groups of three mice each; 0.30 MBq unconjugated [125I]bio-Aβ1–40 or the [125I]bio-Aβ1–40/streptavidin–8D3 conjugate in PBS was injected via the jugular vein of each mouse in a total volume of 0.05 mL. A total of 30 mice were used in these studies. Groups of three mice each were killed at 0.25, 2, 5, 15, and 60 minutes after the isotope administration, and arterial blood was sampled from the descending aorta. The blood samples were centrifuged and the collected serum was analyzed for either total [125I]-radioactivity using a 7-counter (Beckman Instruments, Inc., Fullerton, CA, U.S.A.) or precipitation by 10% cold TCA. Six organs (brain, liver, kidney, heart, lung, and spleen) were also removed from the 60-minute group. Total [125I]-radioactivity of each organ was measured and normalized by organ weight. The measurement of serum TCA precipitable radioactivity provides an index of the uptake and degradation of the [125I]bio-Aβ1–40 by peripheral tissues. This degradation results in the release to blood of TCA-soluble metabolites, such as [125I]-tyrosine.
Pharmacokinetic parameters were estimated by fitting the TCA-precipitable serum radioactivity data to the following biexponential equation with a weighting factor of 1/A(t)2 using a derivative-free nonlinear regression analysis (PARBMDP, Biomedical Computer P-Series, developed at UCLA Health Science Computing Facilities):
where A(t) = percent injected dose per milliliter serum. The area under the serum concentration curve, the area under the moment curve, the serum clearance, the steady-state volume of distribution (Vss), and the mean residence time were calculated from the injected dose, the slopes (K1 and K2), and intercepts (A1 and A2) of the biexponential equation (Gibaldi and Perrier, 1982). The organ uptake was expressed as percentage of injected dose per gram organ, and was calculated as follows:
where Cp(60 minutes) is the terminal serum concentration, Vd is the organ volume of distribution, and V0 is the plasma volume of each organ for adult mice (Lee and Pardridge, 2000). The Vd is the ratio of counts/min per gram organ at 60 minutes divided by the counts/min per microliter serum 60 minutes after the injection.
Gel-filtration fast protein liquid chromatography
The serum stability of [125I]bio-Aβ1–40 with or without conjugation to 8D3–streptavidin was investigated by gel-filtration fast protein liquid chromatography using two Superose 12 High Resolution 10/30 fast protein liquid chromatography columns in series, as described previously (Kurihara et al., 1999). A 25-μL aliquot of 60-minute terminal serum from each of three mice was pooled and injected on to the columns with elution in PBST at a flow rate of 0.5 mL/min for 80 minutes. [125I]-Radioactivity was counted from each fraction (0.5 mL) using a γ-counter.
Immunocytochemistry of transgenic mouse brain
Avidin-biotin immunocytochemistry was performed with the ABC Elite kit from Vector Labs (Burlingame, CA). Frozen sections (10 μm) transgenic mouse brain were fixed with 10% formalin for 10 minutes at 4°C. After washing in PBS, sections were treated with 50% formic acid for 15 minutes at room temperature, and endogenous peroxidase activity was blocked with 0.3% hydrogen peroxide for 30 minutes. Nonspecific binding was blocked with 10% goat serum for 30 minutes. The sections were incubated for 60 minutes at room temperature with the primary antibody, a 1:1000 dilution of the rabbit anti-Aβ1–28 antiserum, as described previously (Vinters et al., 1990), after absorption of the antiserum with bovine thyroglobulin (Vinters et al., 1990). After washing in PBS, sections were incubated with biotinylated goat antirabbit immunoglobulin G 1:200 in 1% goat serum. Control sections were treated with preimmune serum.
Quantitative autoradiography in transgenic mice and littermate controls
App SW /Psen1 double-transgenic mice and littermate controls were injected either with the unconjugated [125I] bio-Aβ1–40 or with the [125I] bio-Aβ1–40/streptavidin–8D3 conjugate via the jugular vein with anesthesia using a 30-g needle. A total of five transgenic mice and five littermate controls were examined. The injected dose of the unconjugated [125I] bio-Aβ1–40 was 0.34 micrograms per mouse. For the mice receiving the conjugate, the injected dose of streptavidin–8D3 was 15 micrograms per mouse (0.08 nmol/mouse), and this was conjugated to [125I] bio-Aβ1–40 (0.34 micrograms per mouse). These dosages represented a 1:1 molar ratio of the [125I] bio-Aβ1–40 and the streptavidin–8D3. At 2, 4, and 6 hours after the intravenous injection, the brain of each mouse was removed, rapidly frozen in powdered dry ice, and dipped in Tissue-Tek Optimal Cutting Temperature medium (Sakura, Torrance, CA, U.S.A.). Then, the frozen brain specimens were sectioned at 20-μm thickness using a Mikron HM 505E cryostat (Micron Instruments, San Diego, CA), placed on glass slides, and dried at room temperature. For film autoradiography, the slides were placed in apposition to X-Omat Blue XB-1 film and exposed for 7 days at −70°C in parallel with 20-μm 125I microscale standard strips. The films were scanned with a 1200 dpi PowerLookIII scanner with transparency adapter (UMAX Technologies, Fremont, CA, U.S.A.), and cropped in Photoshop 5.5 (Adobe Systems, San Jose, CA, U.S.A.) on a G4 Power Macintosh computer, as described previously (Kurihara and Pardridge, 1999). Quantitation of the integrated density over the whole brain section or the 125I-microscale standard was performed with NIH Image 1.62 software (National Institutes of Health, Bethesda, MD, U.S.A.), as described previously (Kurihara and Pardridge, 1999), and normalized by pixel area of the scanned region. A standard curve was constructed from the 125I-standards (0.7, 1.5, 2.5, 5.0, and 10 μCi/g), and this curve was used to convert the normalized integrated density to tissue radioactivity (μCi/g).
The use of quantitative autoradiography in these studies is considered an in vivo imaging modality because the peptide radiopharmaceutical is administered in vivo 2 to 6 hours before animals are killed and frozen sectioning of the brain is performed. In contrast, with in vitro autoradiography, the radiolabeled peptide is applied to tissue sections postmortem.
RESULTS
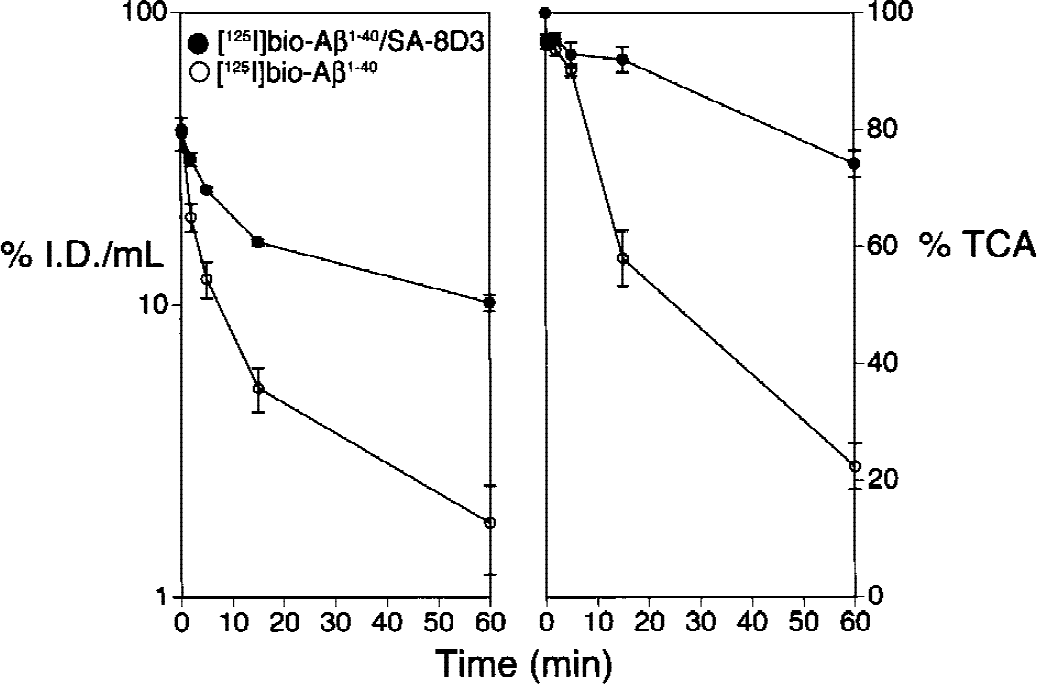
The rate of removal of [125I]-bio-Aβ1–40 from blood in mice in vivo was rapid (Fig. 2, left panel) with a high clearance rate of 9.5 ± 0.8 mL·min−1·kg−1 (Table 1). This high clearance was associated with the rapid formation of TCA-soluble metabolites in serum, as shown in Fig. 2, right panel. The conjugation of [125I]-bio-Aβ1–40 to 8D3–streptavidin resulted in a fivefold reduction in the plasma clearance to a value of 1.9 ± 0.9 mL·min−1·kg−1 (Fig. 2, Table 1). This reduced plasma clearance was also associated with increased metabolic stability as shown by the TCA precipitability of serum radioactivity (Fig. 2). The metabolic stability of the [125I]bio-Aβ1–40/streptavidin–8D3 conjugate was also shown with gel-filtration fast protein liquid chromatography. As shown in Fig. 3, the principle peak of radioactivity in serum comigrated with the intact [125I]-bio-Aβ1–40/streptavidin–8D3 conjugate (peak 2). Peak 1 radioactivity represents binding of the conjugate to high molecular weight proteins, such as lipoproteins or α2-macroglobulin (Du et al., 1997). Peak 3 radioactivity represents binding of [125I]bio-Aβ1–40 to serum albumin (Biere et al., 1996). Peak 4 corresponds to unconjugated [125I]-Aβ1–40, and peak 5 corresponds to [125I]-tyrosine or [125I]-iodide (Lee and Pardridge, 2000). Peaks 1 to 4 are precipitated by TCA, whereas peak 5 is TCA soluble.
Pharmacokinetic parameters in BALB/c mice
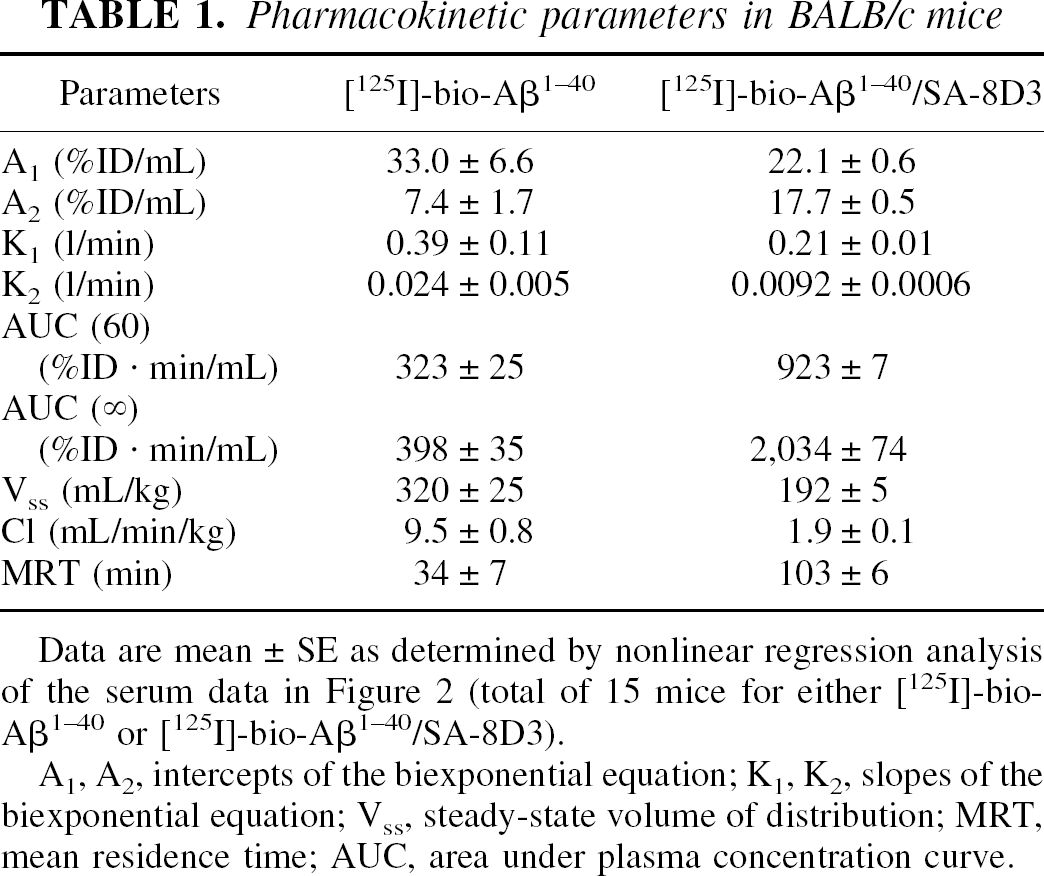
Data are mean ± SE as determined by nonlinear regression analysis of the serum data in Figure 2 (total of 15 mice for either [125I]-bio-Aβ1–40 or [125I]-bio-Aβ1–40/SA-8D3).
A1, A2, intercepts of the biexponential equation; K1, K2, slopes of the biexponential equation; Vss, steady-state volume of distribution; MRT, mean residence time; AUC, area under plasma concentration curve.
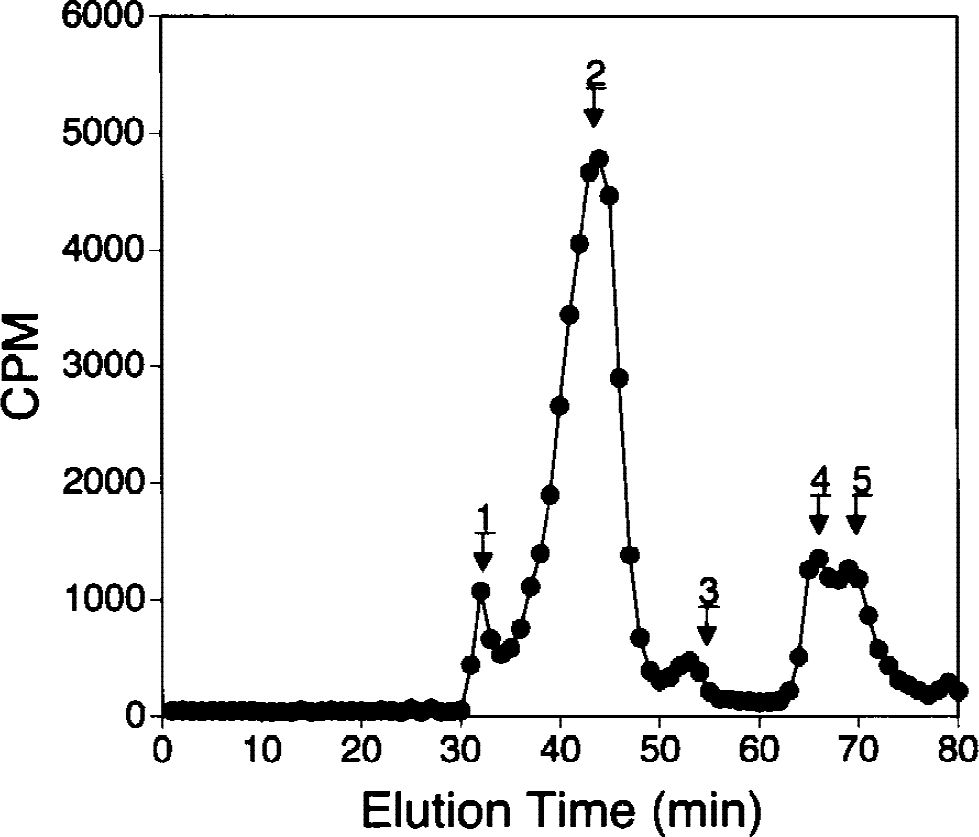
The fast protein liquid chromatography gel-filtration elution profile of serum taken from mice 60 minutes after intravenous injection of [125I]-bio-Aβ1–40/streptavidin–8D3 conjugate is shown. Radioactivity is associated with high molecular weight complexes such as lipoprotein or α2-macroglobulin (peak 1), intact conjugate (peak 2), albumin (peak 3), unconjugated [125I]-bio-Aβ1–40 separated from the streptavidin–8D3 conjugate (peak 4), or unconjugated [125I]-tyrosine or [125I]-iodide (peak 5).
Conjugation of the [125I]-bio-Aβ1–40 to the 8D3 MAb resulted in a redistribution in organ uptake of the peptide radiopharmaceutical. The unconjugated peptide was rapidly cleared by kidney. In contrast, conjugation of the Aβ peptide to the 8D3 MAb resulted in an increase in uptake by TfR-rich organs such as the spleen, liver, and brain (Fig. 4). There was a decrease in renal clearance caused by conjugation of the peptide to the MAb, and there was minimal change in peptide clearance by organs such as the lung or heart (Fig. 4). The enhanced import of the peptide radiopharmaceutical into the brain caused by conjugation to the 8D3 MAb is also evident in the auto radiography study shown in Fig. 5. Although there is minimal uptake of radioactivity in brain after the intravenous injection of unconjugated [125I]-bio-Aβ1–40, there is a high level of brain radioactivity 2 hours after the intravenous injection of the [125I]-bio-Aβ1–40 conjugated to the 8D3 MAb (Fig. 5). The differences in uptake between the conjugate and the unconjugated peptide are also visible 4 hours after a single intravenous injection, but after 6 hours there is nearly complete export of [125I]-bio-Aβ1–40/streptavidin-8D3 conjugate from the control brain bearing no Aβ amyloid (Fig. 5). The autoradiography studies performed in the transgenic littermate controls (Fig. 5) were corroborated by other studies measuring the efflux of radioactivity from brains of adult BALB/c mice injected intravenously with the [125I]-bio-Aβ1–40/streptavidin–8D3 conjugate. The percent of injected dose per gram brain 1, 2, 5, and 8 hours after injection was 0.90 ± 0.06, 0.67 ± 0.10, 0.31 ± 0.03, and 0.17 ± 0.03, respectively (mean ± standard error, n = 3 mice per time point). These data were fit to a monoex-ponential decay function using nonlinear regression analysis to yield the maximum percent brain uptake of 1.15 ± 0.04% injected dose per gram and an efflux rate constant of 0.26 ± 0.02 hours with an efflux t1/2 of 2.7 ± 0.2 hours. On the basis of these combined studies in both control BALB/c mice and the littermate controls (Fig. 5), 6 hours was chosen as the optimal time for brain scanning of transgenic mice because this period represents more than two halftimes of conjugate efflux from the brain of nonamyloid-bearing animals.
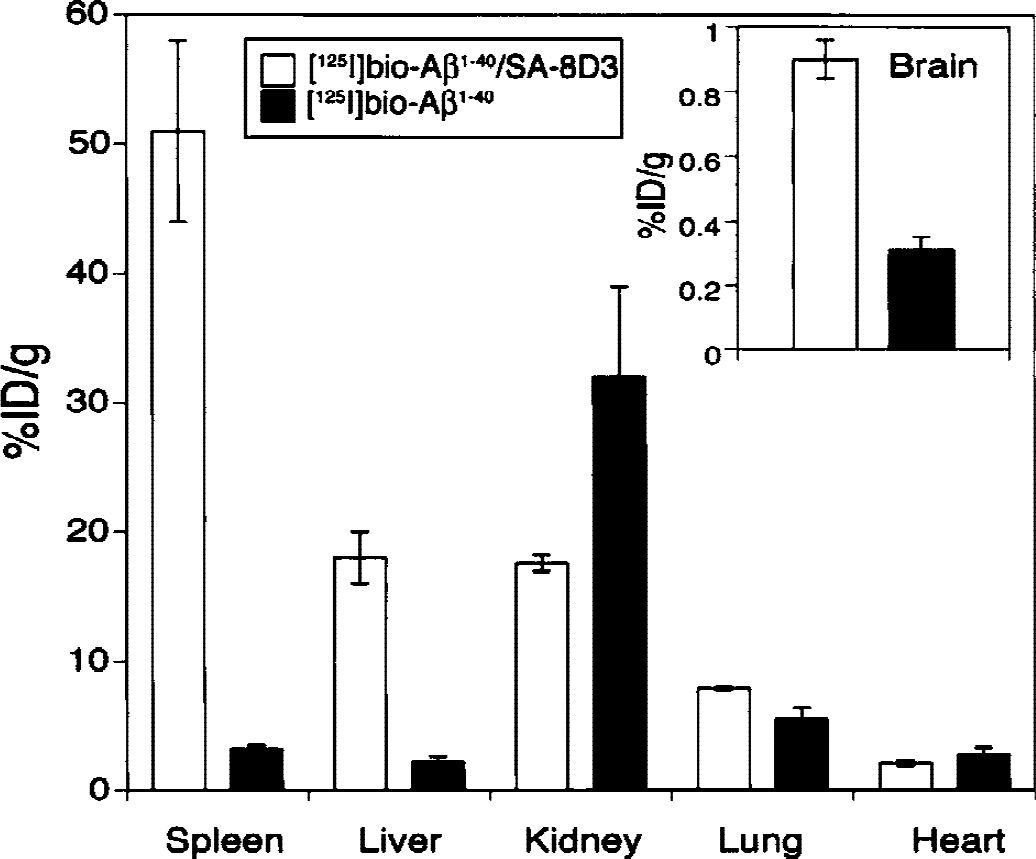
The organ uptake, expressed as a percent of injected dose per gram organ for the spleen, liver, kidney, lung, heart, or brain is shown 60 minutes after intravenous injection of [125I]-bio-Aβ1–40/streptavidin–8D3 (open bars) or [125I]-bio-Aβ1–40 (closed bars) in control mice. Data are mean ± standard error (n = 3 mice per time point).
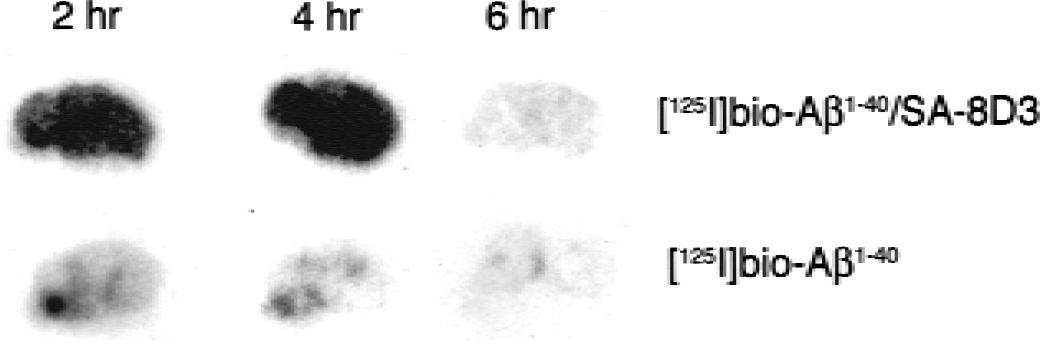
Film autoradiography of 20-μm frozen sections of brain removed 2, 4, or 6 hours after intravenous injection of [125I]-bio-Aβ1–40/streptavidin–8D3 conjugate or the unconjugated [125I]-bio-Aβ1–40 in littermate controls. Films were exposed for 7 days.
Immunocytochemistry with an anti-Aβ1–28 antiserum showed diffuse APP immunoreactivity throughout the cerebrum and cerebellum of the transgenic mice, with a higher immunoreactivity in gray matter relative to white matter (Figs. 6A and 6C). Multiple amyloid plaques were also visible in the cortical and subcortical gray matter of the cerebrum of the transgenic mice (Figs. 6A and 6C) and high-magnification microscopy showed the presence of Aβ-immunoreactive amyloid plaques in the transgenic mouse brain (Figs. 6E and 6F). The 6-hour brain scans of double-transgenic mice and littermate controls injected either with the [125I]-bio-Aβ1–40/streptavidin–8D3 conjugate or with the unconjugated [125I]-bio-Aβ1–40 are shown in Fig. 7. There is minimal brain radioactivity in double-transgenic mice (Fig. 7B) or littermate controls (Fig. 7D) injected with unconjugated [125I]-Aβ1–40, a finding that is consistent with the minimal BBB transport of this peptide. There is also minimal radioactivity in the littermate controls injected with the [125I]-bio-Aβ1–40/SA-8D3 conjugate, which is consistent with the export of radioactivity from nonamyloid-bearing brain 6 hours after intravenous injection (Fig. 7C). However, in the transgenic mouse injected with the [125I]-bio-Aβ1–40/streptavidin–8D3 conjugate, there is a selective sequestration of radioactivity throughout the cerebrum and cerebellum (Fig. 7A), which parallels the increased APP/Aβ immunoreactivity in the brain (Figs. 6A and 6C). This 6-hour brain scan was quantified by comparison of the brain radioactivity with the 20-μm [125I] microscale standard shown in Fig. 7E, and the standard curve allowed computation of the radioactivity in the brains of the four groups at 6 hours (Fig. 7F).
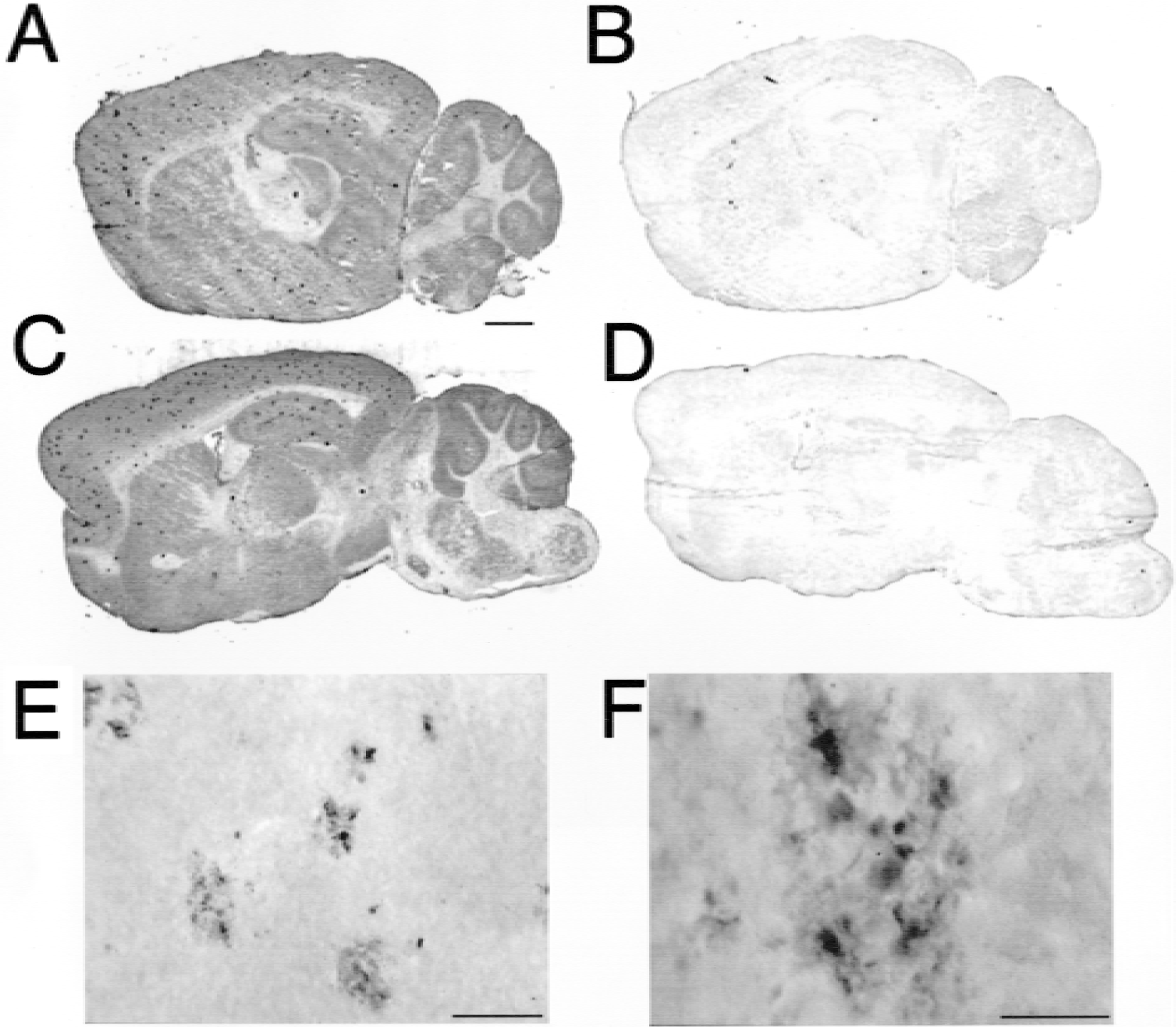
Immunocytochemical analysis of the double-transgenic mouse brain using (
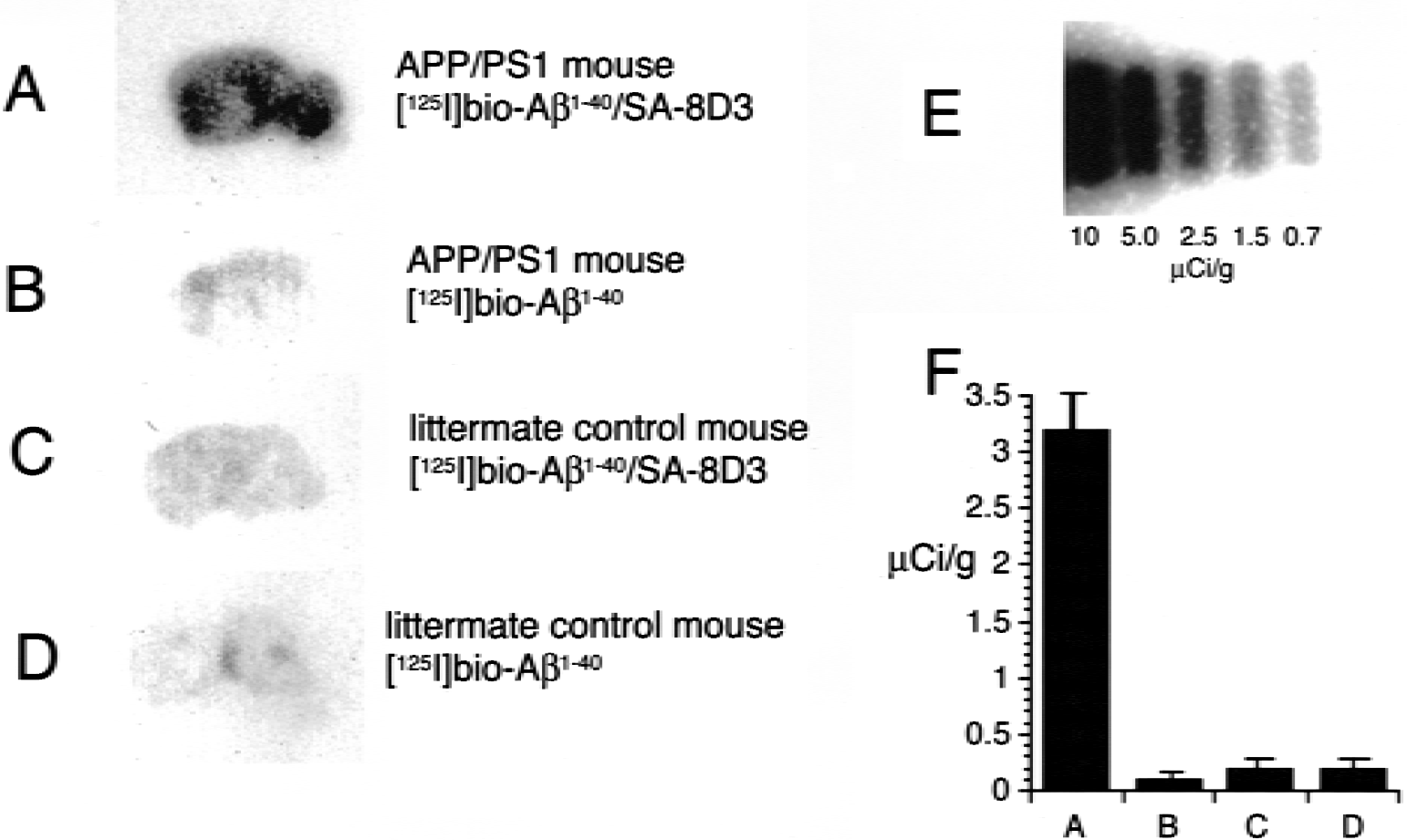
The 6-hour brain quantitative autoradiogram is shown for App/Psen1 double-transgenic mice injected with
DISCUSSION
The results of these studies are consistent with the following conclusions. First, conjugation of the Aβ1–40 peptide radiopharmaceutical to the 8D3 targeting MAb confers enhanced metabolic stability, prolonged plasma residence time, and biotinidase resistance of the biotinylated peptide in mice (Figs. 2 and 3). Second, conjugation of the [125I]-Aβ1–40 to the 8D3 MAb results in a redirection of organ clearance of the peptide radiopharmaceutical from the kidney to organs rich in TfR, such as liver, spleen, or the BBB (Fig. 4). The brain uptake of the [125I]-bio-Aβ1–40/streptavidin–8D3 conjugate 60 minutes after intravenous injection is approximately 1% injected dose per gram brain, and this approximates the brain uptake of the unconjugated 8D3 MAb in mice (Lee et al., 2000). Third, following the initial import of the radioactive conjugate into brain, there is subsequent export of radioactivity from the brain back to the blood that is nearly complete 6 hours after intravenous injection in the littermate controls (Fig. 5). This import and export of radioactivity provides the basis for scanning the brains of transgenic mice 6 hours after intravenous administration. Fourth, the double-transgenic App SW /Psen1 mouse (Takeuchi et al., 2000) expresses significant Aβ-amyloid plaque in the cortical and subcortical structures of the cerebrum at 9 months of age based on immunocytochemistry (Fig. 6). This global increase in brain APP/Aβ immunoreactivity correlates with the selective sequestration of radioactivity in the transgenic mouse brain administered [125I]-Aβ1–40, providing this peptide radiopharmaceutical is conjugated to a BBB drug-targeting system (Figs. 7A and 7F).
Aβ1–40 peptide radiopharmaceuticals deposit onto preexisting Aβ-amyloid plaques in autopsy tissue sections of AD brain (Maggio et al., 1992). However, Aβ1–40 peptide radiopharmaceuticals have not been successfully used in the in vivo imaging of brain amyloid because of the lack of transport of this neuropeptide across the BBB in vivo (Saito et al., 1995; Wu et al., 1997). In addition, Aβ1–40 peptide radiopharmaceuticals are rapidly degraded in vivo with a concomitant increase in plasma TCA-soluble radioactivity (Fig. 2). The TCA-soluble radioactive metabolites include [125I]-tyrosine, which enters the brain from the blood on the BBB-neutral amino acid transport system (Pardridge, 2001). The brain uptake of radioactivity after intravenous injection of Aβ peptide radiopharmaceuticals is inversely related to the TCA precipitability of the serum radioactivity (Poduslo et al., 1999). Therefore, the radioactivity in the brain after intravenous injection of unconjugated [125I]-Aβ1–40 largely reflects the brain uptake of metabolites, and not the brain uptake of [125I]-Aβ1–40 per se. The intravenous injection of unconjugated Aβ peptide radiopharmaceuticals does not lead to the labeling of amyloid plaques in the brain of double-transgenic mice because of the lack of transport of this peptide across the BBB in vivo (Wengenack et al., 2000). Similarly, Congo Red derivatives, such as chrysamine G (Dezutter et al., 1999) or (trans, trans)-1-bromo-2–5-bis (3-hydroxy carbonyl-4-hydroxy) styryl benzene (Skovronsky et al., 2000) cross the BBB poorly (Zhuang et al., 2001) because of the multiple highly charged anionic groups on these compounds. Thioflavin T derivatives are more lipid soluble and are potential small-molecule amyloid imaging agents (Zhuang et al., 2001; Klunk et al., 2001), though these molecules have not been shown to image experimental brain amyloid in vivo. Similar to Aβ peptide radiopharmaceuticals and Congo Red derivatives, anti-Aβ monoclonal antibodies do not cross the BBB, and these antibodies must be administered by invasive procedures such as cranial window (Bacskai et al., 2001) or intracerebral ventricular administration (Walker et al., 1994).
Aβ peptide radiopharmaceuticals, Congo Red derivatives, or anti-Aβ monoclonal antibodies could be used to image brain amyloid in AD if these molecules are made transportable through the BBB with brain drug-targeting systems (Pardridge, 2001). The conjugation of Aβ1–40 peptide radiopharmaceuticals to BBB transport vectors results in a level of brain uptake of the peptide that is in excess of the brain uptake of a neuroactive small molecule, morphine (Pardridge, 2001). Conjugation of the Aβ peptide radiopharmaceutical to the BBB targeting MAb does not inhibit binding of the Aβ to amyloid plaques in autopsy tissue sections of AD brain (Saito et al., 1995; Wu et al., 1997). Aβ peptide radiopharmaceuticals bind to the AD amyloid plaque at tracer concentrations as low as less than 0.1 nmol/L (Ghilardi et al., 1996). Therefore, tracer concentrations of the Aβ1–40 peptide radiopharmaceutical delivered to brain in vivo will bind the amyloid. The placement of an amino terminal biotin residue on the Aβ1–40 does not impair binding of the peptide to amyloid plaques (Prior et al., 1996). The increased brain uptake of [125I]-bio-Aβ1–40 in the mouse is enabled with conjugation of the biotinylated peptide to the 8D3-streptavidin BBB transport vector (Figs. 4 to 6). The conjugation of the peptide to the MAb transport vector is performed with a biotin-streptavidin linker (Fig. 1A). The biotin bond was degraded by biotinidase activity in mice in vivo when the RI7–217 MAb is used as the mouse brain transport vector (Lee and Pardridge, 2000). However, the fast protein liquid chromatography study shows that the conjugate and the biotin linker is intact in mice with the 8D3 MAb (Fig. 3). Because of the stability of the linker attached to the 8D3 MAb, the brain uptake of the [125I]-bio-Aβ1–40/streptavidin–8D3 conjugate (Fig. 4) is 100% higher than the brain uptake of the [125I]-bio-Aβ1–40/streptavidin-RI7–217 conjugate in mice (Lee and Pardridge, 2000).
Subsequent to the import of Aβ peptide radiopharmaceuticals from the blood to the brain by receptor-mediated transcytosis across the BBB, there must also be an export of radioactivity from the brain to the blood in the regions of brain lacking Aβ amyloid (Fig. 1B). If there is no export of radioactivity in the nonamyloid-bearing regions of brain, then there would be no selective sequestration of radioactivity in brain expressing Aβ amyloid. Macromolecular efflux from the brain to the blood across the BBB does occur and is receptor mediated. The BBB TfR mediates the bidirectional transcytosis of either transferrin (TfR) or the anti-TfR MAbs in the blood-to-brain or brain-to-blood directions (Zhang and Pardridge, 2001a). Anti-TfR MAbs are also immunoglobulin G molecules and are substrates for the unidirectional export from the brain to the blood via the BBB Fc receptor (Zhang and Pardridge, 2001b), as depicted in Fig. 1B. The kinetics of efflux of the conjugate from mouse brain (t1/2 = 2.7 hours, Results) are similar to the kinetics of efflux from rat brain of holotransferrin via the BBB TfR (Zhang and Pardridge, 2001a). Based on the kinetics of efflux of the radioactive conjugate from control mouse brain (t1/2 = 2.7 hours, Results), a time of 6 hours was chosen for brain scanning of transgenic mice.
The App SW /Psen1 double-transgenic mouse has both discrete Aβ plaques (Figs. 6E and 6F) and diffuse APP/Aβ immunoreactivity in the brain (Figs. 6A and 6C), whereas there is no immunoreactivity detected in transgenic brain with the preimmune antiserum (Figs. 6B and 6D). The selective sequestration of the [125I]-bio-Aβ1–40/streptavidin–8D3 in the double-transgenic mouse brain (Figs. 7A and 7F) correlates with the overall increase in APP/Aβ immunoreactivity in the transgenic mouse brain (Fig. 6). The selective sequestration of the radioactivity in the transgenic mouse brain is enabled by the transport of the amyloid imaging agent across the BBB using brain drug-targeting technology.
In summary, the present studies show that A β peptide radiopharmaceuticals may be used for the imaging of brain amyloid in vivo, provided the amyloid imaging agent is conjugated to a BBB drug-targeting system. The amyloid plaque burden in the AD brain increases with age and may occupy up to 15% of the total brain volume of patients with AD (Kuo et al., 1996). It may be possible to quantitate the Aβ amyloid burden in patients with AD using an amyloid imaging agent conjugated to a BBB drug-delivery system. Studies in humans would necessitate the use of genetically engineered antibodies that are not immunogenic in humans. A genetically engineered human insulin receptor MAb retains 100% of the receptor-binding activity compared with the original murine MAb. The genetically engineered human insulin receptor MAb is rapidly transported across the BBB in the living rhesus monkey in vivo, and 2% of the injected dose is delivered to the primate brain after an intravenous injection (Coloma et al., 2000). Because of the need to allow sufficient time for export of the radioactive conjugate from nonamyloid-bearing regions of the brain, it is necessary to image brain at delayed times after intravenous administration of the radiopharmaceutical. Aβ1–40-antibody conjugates efflux from control primate brain in vivo with a t1/2 of 16 hours (Wu et al., 1997). The efflux of Aβ1–40-antibody conjugates from the brain is slower in primates, and presumably humans, than in mice (efflux t1/2, 2.7 hours; Results). Therefore, optimal brain images in humans may necessitate brain scanning 24 or 48 hours after intravenous administration of the radiopharmaceutical. Because of this requirement for delayed imaging times, it may be necessary to use a radionuclide with a long t1/2 of radioactive decay, such as 111-indium for single-photon emission computed tomography imaging or 124-iodine for positron emission tomography imaging. The 111In radionuclide is preferred over an iodine radionuclide because there is no export back to the blood of 111In-labeled metabolites formed by the peripheral degradation of peptide radiopharmaceuticals (Kurihara et al., 1999). Much higher signal/noise ratios are obtained with 111In, relative to 125I (Kurihara et al., 1999), and the use of Aβ1–40 peptide radiopharmaceuticals labeled with 111In would be expected to give brain scans with a resolution higher than those obtained in Fig. 7 with the 125I labeled peptide radiopharmaceutical. Aβ1–40 can be double conjugated with a biotin group at the amino terminus, and a diethylenetriaminepentacetic acid moiety at the ɛ-amino group of an internal lysine residue. This double-conjugated Aβ1–40 still binds to Aβ-amyloid plaques in autopsy sections of the AD brain (Kurihara and Pardridge, 2000). The double conjugation of Aβ1–400 with biotin and diethylenetriaminepentacetic acid enables high-affinity capture of the biotinylated peptide by a MAb/avidin fusion protein for delivery through the BBB in humans in vivo, and high-affinity capture of 111In for production of a radiopharmaceutical. Alternatively, the diethylenetriaminepentacetic acid moiety could chelate gadolinium for production of a peptide magneto-pharmaceutical for brain scanning with magnetic resonance imaging.
Footnotes
Acknowledgments:
The authors thank Daniel Jeong for his skillful preparation of the manuscript.