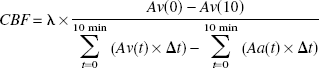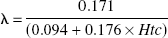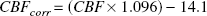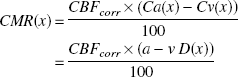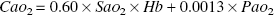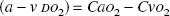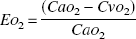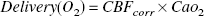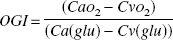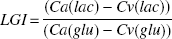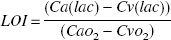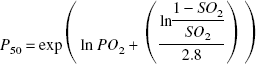Abstract
The authors investigated the effect of acclimatization to high altitude on cerebral blood flow and oxidative metabolism at rest and during exercise. Nine healthy, native sea-level residents were studied 3 weeks after arrival at Chacaltaya, Bolivia (5,260 m) and after reacclimatization to sea level. Global cerebral blood flow at rest and during exercise on a bicycle ergometer was measured by the Kety-Schmidt technique. Cerebral metabolic rates of oxygen, glucose, and lactate were calculated by the Fick principle. Cerebral function was assessed by a computer-based measurement of reaction time. At high altitude at rest, arterial carbon dioxide tension, oxygen saturation, and oxygen tension were significantly reduced, and arterial oxygen content was increased because of an increase in hemoglobin concentration. Global cerebral blood flow was similar in the four conditions. Cerebral oxygen delivery and cerebral metabolic rates of oxygen and glucose also remained unchanged, whereas cerebral metabolic rates of lactate increased slightly but nonsignificantly at high altitude during exercise compared with high altitude at rest. Reaction time was unchanged. The data indicate that cerebral blood flow and oxidative metabolism are unaltered after high-altitude acclimatization from sea level, despite marked changes in breathing and other organ functions.
Cerebral perfusion is tightly controlled to meet cerebral metabolic needs and ensure chemical homeostasis. Major determinants of cerebral blood flow (CBF) are the metabolic activity of the brain (Schmidt et al., 1945) and arterial oxygen tension (Kety and Schmidt, 1948a). On arrival at high altitude, hypobaric hypoxia changes these determinants of CBF because of a decrease in arterial oxygen tension, which is an independent mediator of cerebral arteriolar dilatation. However, acute hypoxemia also triggers acute hyperventilation and an associated decrease in arterial carbon dioxide tension, which will constrict cerebral arterioles because of an associated increase in periarteriolar pH; this effect will persist for the first few days. The vasoconstrictive effect of prolonged hypocapnia may be modified by metabolic compensation, an adaptation that occurs within hours to days. Furthermore, when hypoxemia persists for weeks, i.e., during a prolonged stay at altitude, the hematocrit increases, resulting in an increased arterial oxygen content at an unchanged oxygen tension; this change will tend to decrease CBF (Henriksen et al., 1981). Thus, cerebral hemodynamics during acclimatization to altitude are the result of these homeostatic mechanisms.
Previous studies have shown that CBF increases significantly within the first 24 hours after arrival at high altitude, followed by a gradual decrease to normal levels within days (Severinghaus et al., 1966). However, measurements of CBF have never been reported in well-acclimatized sea-level natives after several weeks at high altitude. The cerebral metabolic rate of oxygen (CMRO2) was unchanged compared with sea-level values in five subjects investigated within the first 24 hours after arrival at 3,810 m (Severinghaus et al., 1966), but knowledge regarding the cerebral oxidative metabolism after more prolonged acclimatization is unavailable. Hence, we do not know whether compensation is sufficient for maintenance of cerebral metabolism at a level similar to that at sea level. If compensation is not sufficient, this might be apparent in the resting state; alternatively, it might be unmasked by a superimposed decrease in arterial oxygenation, leading to a decrease in oxygen delivery and, hence, to a decrease in CMRO2. Exercise at altitude leads to more profound arterial hypoxemia even after acclimatization (Sutton et al., 1988); thus, unchanged cerebral function and oxidative metabolism during exercise at altitude indirectly indicate that cerebral metabolism is not decreased after acclimatization.
The Danish 1998 Chacaltaya Expedition was undertaken to study human exercise capacity at high altitude. In this context, the present study was conducted to explore cerebral function and whether it is a possible limiting factor for exercise capacity at high altitude. We measured global CBF and cerebral metabolic rates of oxygen, glucose, and lactate in native sea-level residents after several weeks of acclimatization to high altitude. In addition, cerebral function was monitored by means of an attention test. The measurements were carried out at rest and during exercise and repeated in the same subjects after reacclimatization to sea level. The null hypothesis to be tested was that CBF and cerebral metabolic rate in these well-acclimatized subjects at altitude were not significantly different from sea-level values, even during exercise.
METHODS
The study was approved by the Scientific-Ethics Committee of Copenhagen, Denmark (jr. No. 01-166/98). Nine healthy adults with a mean age of 25 ± 1.7 years (range, 22–26 years), three of whom were women, participated after giving written informed consent. All participants were native Danish sea-level residents.
Study protocol
Measurements of CBF and cerebral oxidative metabolism were carried out in Chacaltaya, Bolivia (5,260 m = 17300 ft; measured barometric pressure, 410 mm Hg) and in Copenhagen, Denmark (40 m = 130 ft; measured barometric pressure, 760 mm Hg). The altitude measurements were preceded by acclimatization during a 14-day stay in La Paz (3800 m) with intermittent ascents to increasing altitudes up to 6,088 m (Mt. Potosi), followed by a 21-day stay in Chacaltaya; thus, subjects had stayed more than 5 weeks above 3,800 m before testing was done. Sea-level measurements were performed 8 months after the return to sea level. For logistic reasons, one subject (no. 8) was studied after 15 months.
Each subject underwent two consecutive measurements of CBF: at rest in the supine position and during upright cycle exercise. After measurements at rest, moderate exercise (100 W) was performed on a standard bicycle ergometer. The interval between CBF measurements at rest and during exercise was 60 minutes. A computerized reaction-time test was used as a simple way to assess cerebral function during both rest and exercise.
Measurements
Global CBF and cerebral metabolic rates were measured by the Kety-Schmidt technique (Kety and Schmidt, 1948b) in the desaturation mode (Madsen et al., 1993). All subjects had an arterial and a jugular bulb catheter as well as a peripheral venous catheter. An intravenous infusion of133Xe dissolved in saline was used. The activity of133Xe in the infusate was adjusted to obtain similar maximum activities in blood samples in the four conditions; because133Xe is eliminated by ventilation, the dose was higher during conditions of increased ventilation (e.g., during exercise and at altitude). Thus, the following doses were administered: high altitude at rest, 800 MBq; high altitude during exercise, 3,500 MBq; sea level at rest, 450 MBq; sea level during exercise, 1,500 MBq. The effective dose of133Xe was approximately 0.2 mSv per study during all conditions. After the saturating 30-minute infusion, blood samples for measurement of133Xe activity were drawn in preweighed syringes simultaneously from the arterial and the jugular venous catheters at the time points −2, −1, 0, 0.5, 1, 2, 3, 4, 6, 8, and 10 minutes, where 0 denotes the time that the133Xe infusion was stopped. The syringes were reweighed and immediately sealed in gas-tight vials to avoid the escape of133Xe. At high altitude, counting of the paired samples in chronological order was done for 5 minutes per sample using a single shielded sodium iodide crystal (Harshaw Integral Line 2 × 2 in, type 2SF8, with a counting well [depth, 38 mm; diameter, 28.6 mm], Harshaw Chemistry, The Netherlands). Background correction was applied. At sea level, paired samples were counted in chronological order for 10 minutes per sample on a scintillation counter (Cobra II; Packard Instruments, Meriden, CT, U.S.A.) and automatically corrected for background activity and decay.
Blood samples for blood gas analysis and measurement of lactate and glucose content were obtained at -1, 4, and 8 minutes. The analyses were performed using the following equipment at high altitude (all Radiometer, Copenhagen, Denmark): ABL 5 (gas tensions), OSM 3 (oxygen saturation and hemoglobin), and EML 105 (glucose and lactate). At sea level, the analyses were performed on an ABL 610 (gas tensions, glucose and lactate content) and an OSM 3 (oxygen saturation and hemoglobin). For validation purposes, sea-level glucose and lactate concentrations were also measured by the enzymatic method, using a YSI 2700 (Yellow Spring Instruments, Yellow Springs, CO, U.S.A.). The validation showed a good agreement between absolute measurements of glucose (r2 = 0.982, P < 0.01; mean difference (YSI measurement − ABL measurement), 0.05 mmol/L (95% CI, ÷0.28 − +0.38)) and lactate (r2 = 0.926, P < 0.01; mean difference 0.06 mmol/L (÷0.05 − +0.18)) and a fair agreement between the resulting arteriovenous differences (glucose, r2 = 0.747, P < 0.01; 95% limits of agreement, ÷0.18 − +0.13 mmol/L; lactate, r2 = 0.551, P < 0.01; 95% limits of agreement, ÷0.05 − +0.08 mmol/L).
The CalCAP test (California Computerized Assessment Package, Los Angeles, CA, U.S.A.) (Waterloo et al., 1997) is a computerized digit-recognition test. While resting in the sitting position or during steady-state exercise, the subject was requested to watch a computer screen on which digits were presented in a random fashion with fixed time intervals. Subjects were told to press a button whenever the digit 7 was presented, and reaction time (in milliseconds) and number of true and false “hits” were recorded.
Calculations
Global CBF (mL · 100g−1 · minute−1) was calculated by the Kety-Schmidt equation (Kety and Schmidt, 1945) as
where Av(t) and Aa(t) denote decay-corrected and background-corrected activities of133Xe (in cpm/g blood) in the jugular venous and arterial sample, respectively, at time t. λ (i.e., the blood-to-brain partition coefficient of133Xe) was calculated from the individual hematocrit (Htc) values according to Chen (Chen et al., 1980) as
When using Equation 1, it is assumed that full saturation and desaturation is achieved (i.e., that infusion and sampling times approach infinity). With infusion and saturation times less than infinity, CBF will be overestimated. Madsen et al. (1993) have performed a computer simulation to show that the “true” CBF can be calculated from the infusion time, sampling time, and the measured CBF. Therefore, the “true” CBF in mL · 100g−1 · minute−1 (CBFcorr) was recalculated from the CBF obtained at saturation time (30 minutes) and sampling time (10 minutes) (Madsen et al., 1993)
The cerebral metabolic rates of oxygen, glucose, and lactate in μmol · g−1 · minute−1 were calculated by the Fick principle (Fick, 1870)
where Ca(x) and Cv(x) denote arterial and jugular venous concentrations, respectively, and a-v D(x) is the arterial-to-jugular concentration difference, of substance x in mmol/L. By definition, a positive value indicates consumption (or net influx), and a negative value indicates production (or net efflux) of the substance.
The arterial concentration of oxygen (CaO2) in mmol/L was calculated as
where Hb denotes the blood hemoglobin concentration in g/dl, SaO2 denotes the measured arterial saturation of oxygen (the oxyhemoglobin fraction of total hemoglobin), and PaO2 is the measured arterial partial pressure of oxygen (mm Hg) at standard temperature and pressure dry. The jugular venous concentration of oxygen (CvO2) was calculated in an identical fashion.
The arterial-to-jugular oxygen content difference v(a − v DO2) was calculated as
and the cerebral extraction fraction of oxygen (EO2) was calculated as
The cerebral oxygen delivery (Delivery [O2]; i.e., the amount of oxygen entering the cerebral circulation with the arterial blood per unit time) in μmol · g−1 · minute−1 was calculated as
The oxygen-glucose index (OGI) was calculated as
When glucose is metabolized aerobically, six molecules of oxygen are used to oxidate every molecule of glucose. If an excess of glucose is metabolized relative to oxygen, the OGI will be lower than six. Hence, an oxygen-glucose index below six indicates that some glucose is being metabolized anaerobically. In contrast, an OGI exceeding six indicates that oxygen consumption exceeds the amount necessary for metabolism of glucose (i.e., that substrates other than glucose are being metabolized aerobically).
The lactate-glucose index (LGI) was calculated as
The LGI is the fraction of glucose uptake that is metabolized and excreted from the brain as lactate. If lactate is excreted, the numerator will be negative; because the denominator is always positive, the resulting LGI will be negative. Hence, a negative index indicates that some glucose is being converted to lactate anaerobically. One molecule of glucose is metabolized to two molecules of lactate (e.g., an LGI of −0.1 indicates that 5% of the cerebral glucose uptake may be accounted for by lactate excretion). This should also lead to a OGI below six, as described previously.
The lactate-oxygen index (LOI) was calculated as
If Cv(lac) is lower than Ca(lac), then the LOI will be positive because the denominator is always positive. Thus, a positive LOI indicates that lactate is taken up by the brain, whereas a negative value indicates a net lactate release. The LOI gives a rough indication of the magnitude of cerebral anaerobic metabolism relative to oxidative metabolism. In healthy volunteers, a slight lactate efflux (i.e., a negative LOI) is usually present at rest (Hasselbalch et al., 1994). In the case of severe cerebral hypoxia, the LOI may be expected to decrease (i.e., become more negative) as the oxygen supply becomes insufficient for aerobic metabolism.
P50 (the oxygen tension at which the hemoglobin is 50% saturated) was calculated in both arterial and jugular venous blood samples from the Hill equation (Paulson et al., 1973) as
The value of P50 changes inversely with the binding affinity of oxygen to hemoglobin. Thus, a low value of P50 indicates that a large fraction of hemoglobin remains in the oxygenated form at low oxygen tensions (i.e., oxygen is relatively tightly bound to hemoglobin), whereas a high value of P50 indicates that oxygen is more readily released from the hemoglobin at low oxygen tensions. P50 was not corrected for changes in pH.
For the attention tests, the number of true-positive and false-positive responses and mean reaction time in milliseconds was calculated separately for each of the four test series, (i.e., at rest and during exercise at high altitude at rest and during exercise at sea level. Furthermore, a measure of the subject's performance (i.e., the standardized normal deviate, the z score) was calculated using the results from a large reference group of healthy young men (Miller et al., 1991).
Statistical analysis
The data are presented as means and SD;P < 0.05 was considered statistically significant.
Both unpaired and paired parametric tests were used. Thus, data sets containing data from all four conditions underwent paired testing. To this end, a repeated-measurements approach was used to analyze the effect of height (altitude or sea level), activity (rest or exercise), and the interaction of height and activity. However, because data for some measurements were missing, group means during the four conditions (altitude rest, altitude exercise, sea level rest, and sea level exercise) also underwent unpaired testing by one-way analysis of variance. If significance was found among groups, unpaired t- tests with Bonferroni correction for multiple comparisons were used to identify differences. Analysis was done using SPSS Base and Advanced Models Version 10.0 for Windows (SPSS, Chicago, IL, U.S.A.).
RESULTS
Cerebral parameters
The CalCAP test showed no significant differences in performance between conditions (Table 1). The mean z score was positive during all conditions, indicating that the average achievement was slightly better than the reference population; however, this difference was not statistically significant.
Cerebral blood flow and oxidative metabolism
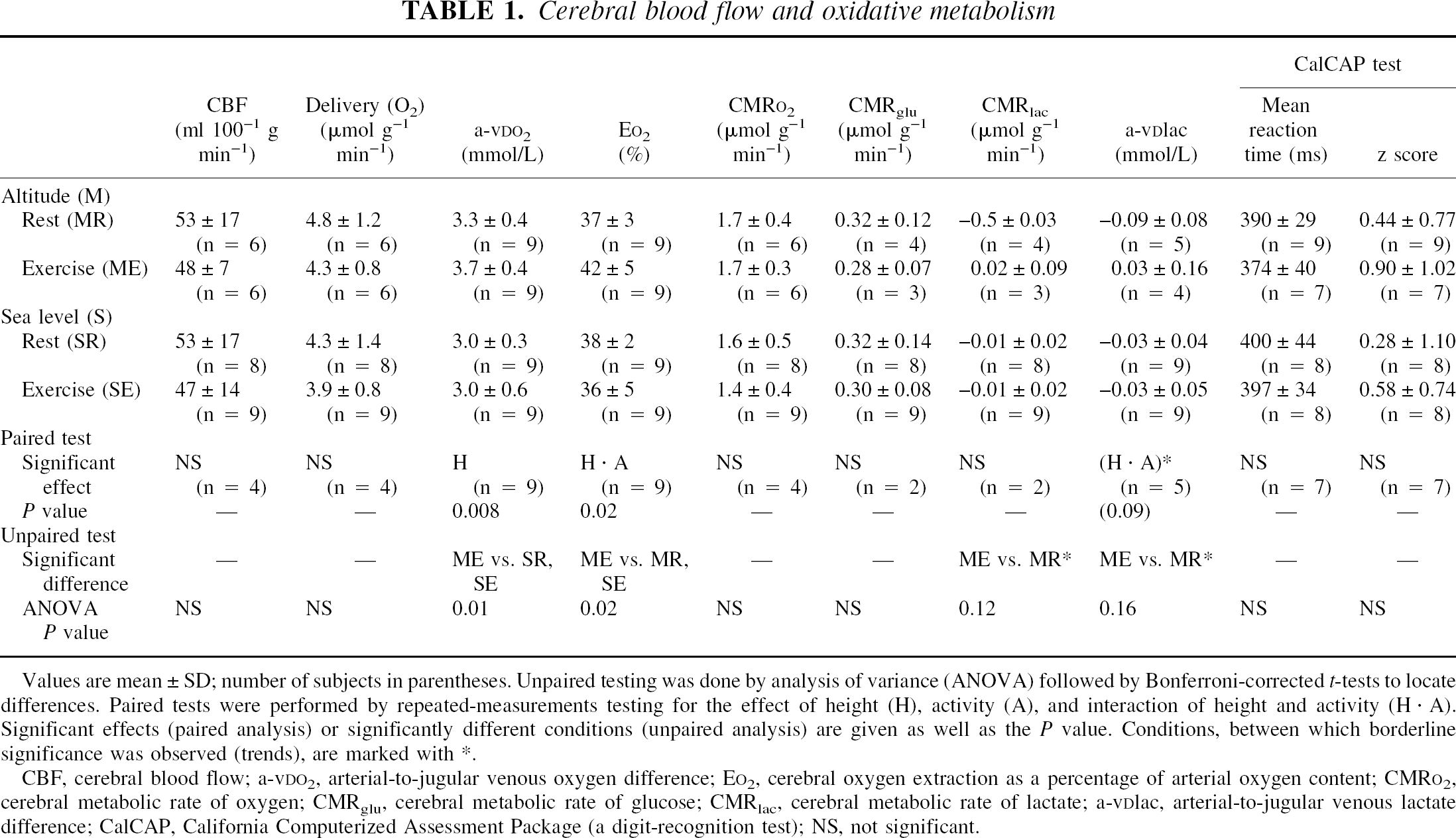
Values are mean ± SD; number of subjects in parentheses. Unpaired testing was done by analysis of variance (ANOVA) followed by Bonferroni-corrected t-tests to locate differences. Paired tests were performed by repeated-measurements testing for the effect of height (H), activity (A), and interaction of height and activity (H · A). Significant effects (paired analysis) or significantly different conditions (unpaired analysis) are given as well as the P value. Conditions, between which borderline significance was observed (trends), are marked with *.
CBF, cerebral blood flow; a-vDO2, arterial-to-jugular venous oxygen difference; EO2, cerebral oxygen extraction as a percentage of arterial oxygen content; CMRO2, cerebral metabolic rate of oxygen; CMRglu, cerebral metabolic rate of glucose; CMRlac, cerebral metabolic rate of lactate; a-vDlac, arterial-to-jugular venous lactate difference; CalCAP, California Computerized Assessment Package (a digit-recognition test); NS, not significant.
Cerebral blood flow measurements were conducted four times in all subjects; however, the quality of resulting curves was technically insufficient in some subjects, and these curves were excluded from further analysis. Therefore, the number of valid CBF measurements was six at rest at high altitude, six during exercise at high altitude, eight at rest at sea level, and nine during exercise at sea level. For paired analysis, four subjects had complete data sets (i.e., data from all four conditions).
Cerebral blood flow did not change significantly at high altitude compared with sea level or during exercise compared with rest (Table 1, Fig. 1). The cerebral oxygen delivery and the cerebral metabolic rate of oxygen were also unchanged. The paired analysis revealed a significant effect of height on the arterial-to-jugular venous oxygen difference (a-vDO2), indicating that a-vDO2 was increased at altitude compared with sea level; unpaired analysis indicated that a-vDO2 was significantly increased only during exercise at high altitude compared with rest and exercise at sea level. The cerebral oxygen extraction as a percentage of the arterial oxygen content was not affected by height only or activity only, but a significant interaction between the two was present, (i.e., the oxygen extraction increased during exercise at high altitude). This finding was corroborated by unpaired analysis (i.e., the oxygen extraction during exercise at high altitude was increased compared with rest at altitude and exercise at sea level).
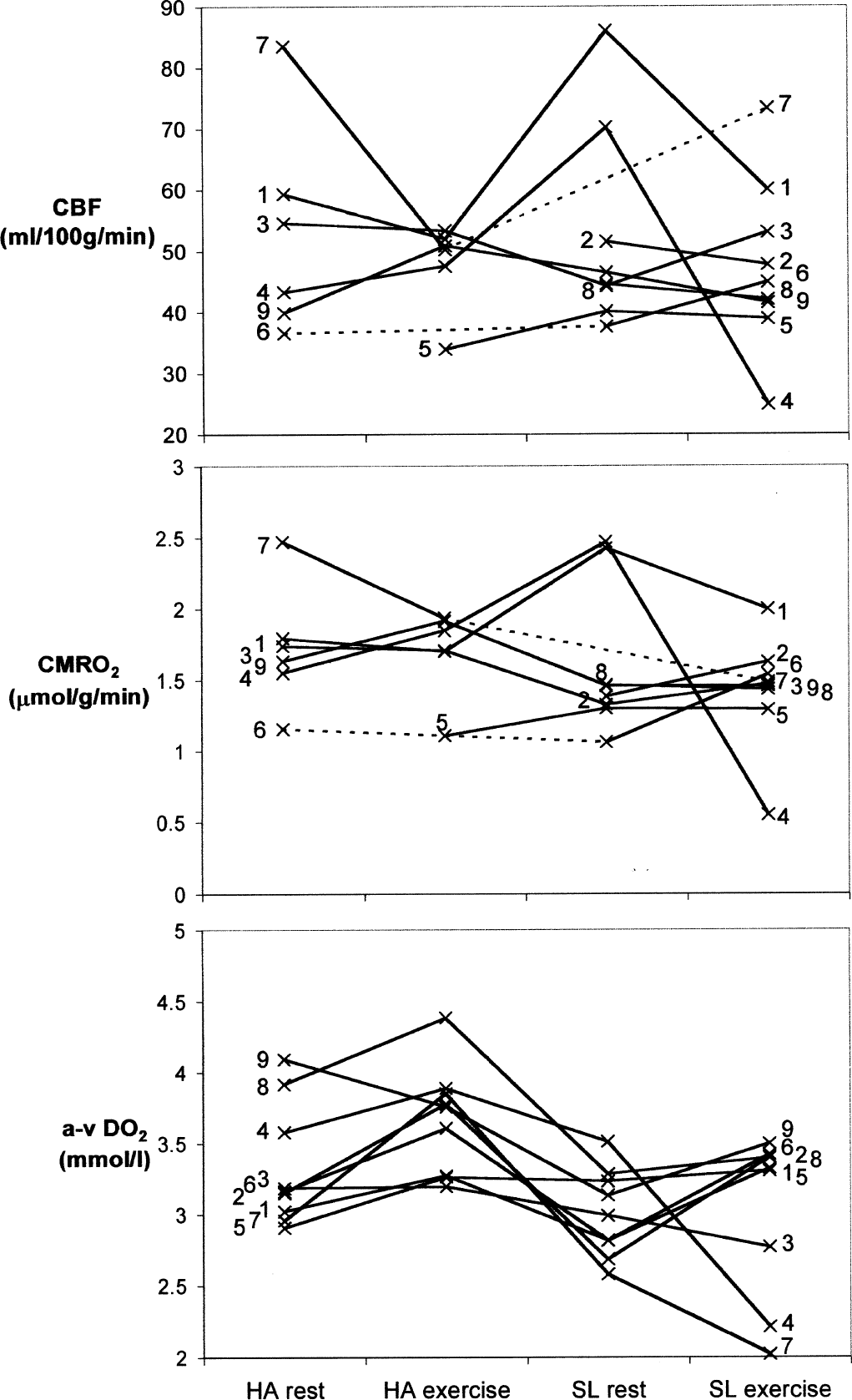
Cerebral blood flow (CBF), cerebral metabolic rate of oxygen (CMRO2), and arterial-to-jugular oxygen content difference (a-vDO2). Individual values of CBF, CMRO2, and a-vDO2 measured at rest and during exercise at high altitude (HA) and at sea level (SL), respectively. Values from each subject are linked by straight lines and are indicated by the subject's study number. A dotted line indicates that a value is missing between two measured values.
Lactate and glucose data were recorded in five subjects at altitude and in all nine subjects at sea level. The arteriovenous difference and the cerebral metabolic rate of glucose did not differ significantly between groups. A trend was observed toward a higher arteriovenous difference of lactate and cerebral metabolic rate of lactate during exercise compared with rest at altitude (i.e., a cerebral lactate efflux was observed at rest, but during exercise a cerebral uptake of lactate was present), though statistical significance was not reached during paired or unpaired testing. At sea level, values did not differ significantly between rest and exercise.
The indices of oxidative metabolism did not differ significantly between conditions, though trends were observed (Table 2).
Indices of cerebral oxidative metabolism
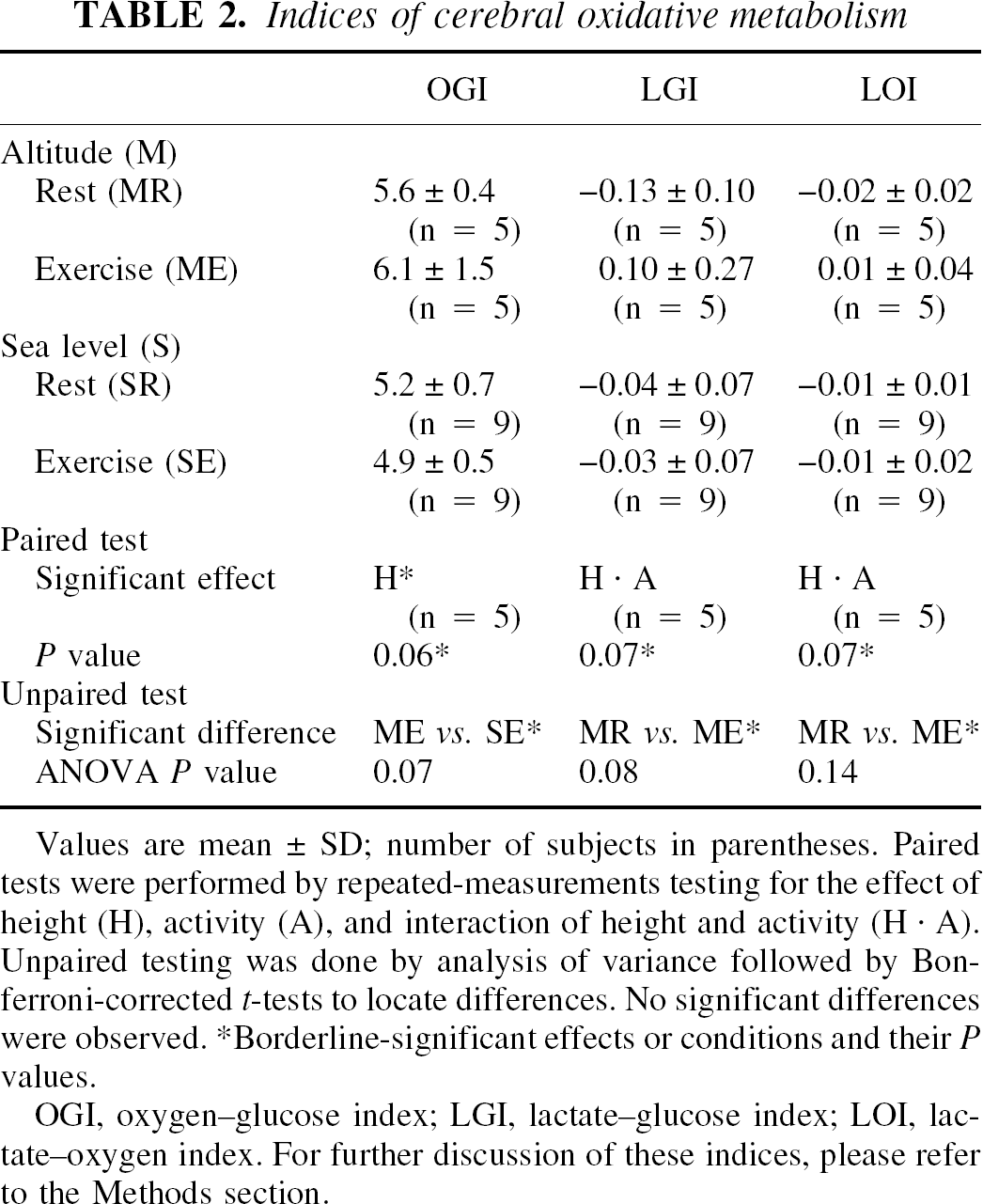
Values are mean ± SD; number of subjects in parentheses. Paired tests were performed by repeated-measurements testing for the effect of height (H), activity (A), and interaction of height and activity (H · A), Unpaired testing was done by analysis of variance followed by Bon ferroni-corrected t-tests to locate differences. No significant differences were observed.
Borderline-significant effects or conditions and their P values.
OGI, oxygen–glucose index; LGI, lactate–glucose index; LOI, lac- tate–oxygen index. For further discussion of these indices, please refer to the Methods section
Systemic parameters
At high altitude, both the arterial oxygen tension and the oxygen saturation were significantly lower than at sea level; however, this difference was fully offset by an increase in hemoglobin and hematocrit (Table 3). Thus, arterial oxygen content was significantly affected by height, (i.e., the arterial oxygen content was higher at altitude compared with sea level). In contrast, there was a significant interaction of height and activity, which acted in the opposite direction (i.e., the arterial oxygen content decreased during exercise compared with rest at high altitude, but not at sea level).
Systemic respiratory parameters
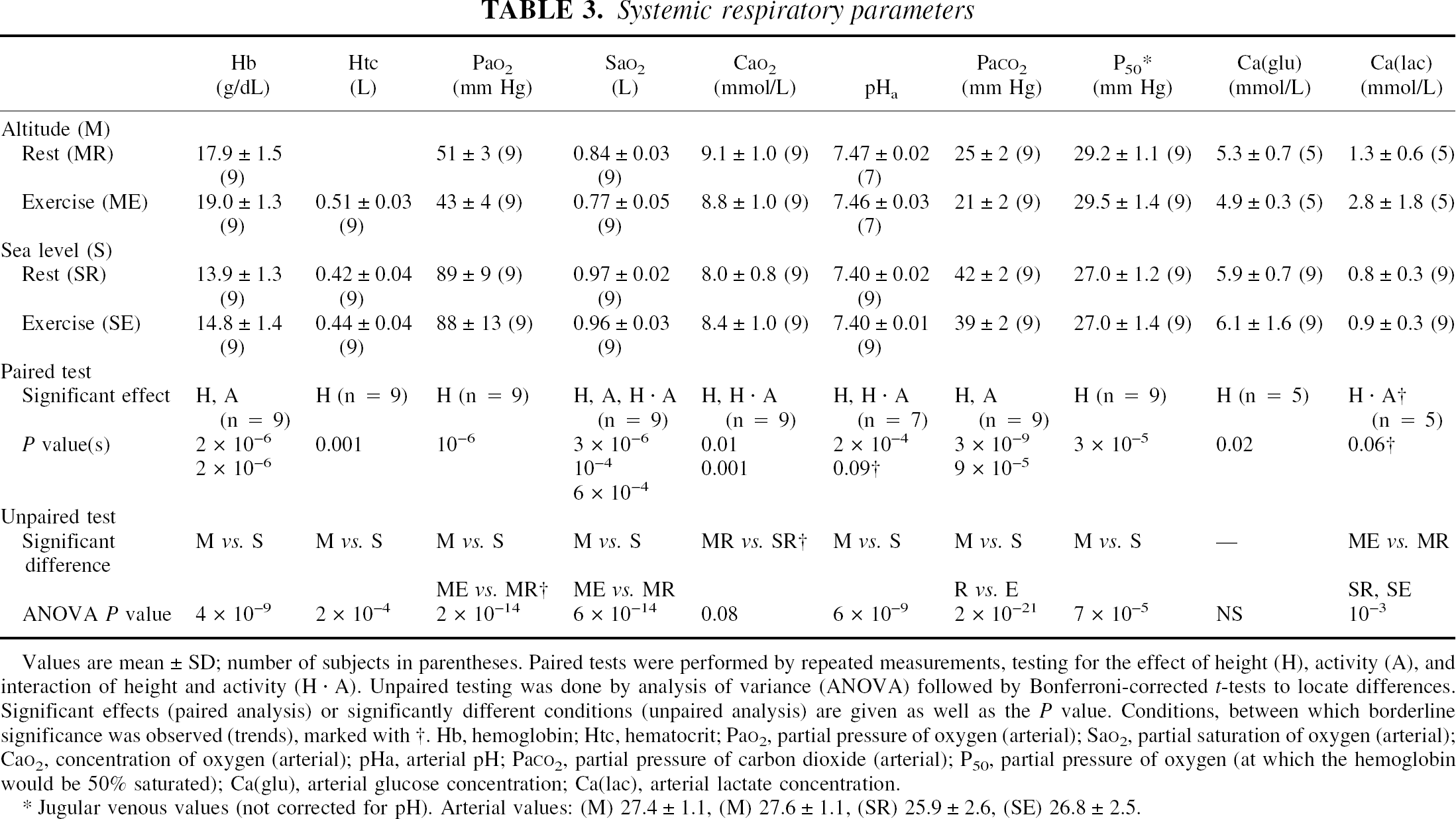
Values are mean ± SD; number of subjects in parentheses. Paired tests were performed by repeated measurements, testing for the effect of height (H), activity (A), and interaction of height and activity (H · A). Unpaired testing was done by analysis of variance (ANOVA) followed by Bonferroni- corrected t-tests to locate differences Significant effects (paired analysis) or significantly different conditions (unpaired analysis) are given as well as the P value. Conditions, between which borderline significance was observed (trends), marked with †. Hb, hemoglobin; Htc, hematocrit PaO2, partial pressure of oxygen (arterial); SaO2, partial saturation of oxygen (arterial); CaO2, concentration of oxygen (arterial); pHa, arterial pH; PaCO2, partial pressure of carbon dioxide (arterial); P50, partial pressure of oxygen (at which the hemoglobin would be 50% saturated); Ca(glu), arterial glucose concentration; Ca(lac), arterial lactate concentration.
Jugular venous values (not corrected for pH). Arterial values: (M) 27.4 ± 1.1, (M) 27.6 ± 1.1, (SR) 25.9 ± 2.6, (SE) 26.8 ± 2.5.
The arterial carbon dioxide tension was significantly lower at high altitude than at sea level, and the arterial pH remained slightly increased (incomplete pH adaptation). During exercise, a significant reduction occurred in arterial carbon dioxide tension compared with rest; pHa remained unchanged.
The calculated value of jugular venous P50 was significantly higher at high altitude than at sea level, whereas no significant difference was found for arterial P50. Exercise did not change P50 compared with the resting state.
The arterial glucose concentration was reduced at altitude compared with sea level, whereas the lactate concentration was unchanged. However, exercise at high altitude was associated with an increase in arterial lactate content compared with rest at high altitude. A similar phenomenon was not observed at sea level.
DISCUSSION
The present study is the first to measure CBF and metabolism on-site by the Kety-Schmidt method in sea-level dwellers who had resided for several weeks at high altitude. The major finding was the preservation of normal global CBF and oxidative metabolism, despite marked changes in arterial oxygen and carbon dioxide tensions. Cerebral oxygen delivery may be calculated as the total arterial oxygen content multiplied by the cerebral blood flow. This variable did not change between rest at sea level, exercise at sea level, rest at altitude, or exercise at altitude. However, as the arterial and jugular venous oxygen tensions at high altitude were reduced compared with sea level and tended to decline further during exercise, the oxygen tension in the brain tissue must have been reduced as well, assuming an unchanged capillary density. Even so, the cerebral metabolic rate of oxygen and cerebral function, as assessed by a simple neuropsychologic test, were not adversely affected by altitude or exercise.
At 5,260 m, the altitude of the present study, the barometric pressure was 410 mm Hg with a corresponding partial pressure of oxygen of 86 mm Hg, 54% of that at sea level. On arrival at altitude, the partial pressure, saturation, and content of oxygen in the arterial blood decrease. The acute physiologic responses to this hypobaric hypoxia include an increase in alveolar ventilation, which increases the alveolar oxygen pressure toward the inspired oxygen pressure and also leads to alveolar and arterial hypocapnia and acute respiratory alkalosis (Mosso, 1899). With prolonged stay, an increase in hematocrit (Douglas et al., 1913) is observed, leading to a normalization of the arterial oxygen content; hypocapnia persists (Douglas et al., 1913), and the alkalosis becomes almost fully compensated. Before the present study, the subjects had ascended from La Paz to progressively higher altitudes during 14 days and stayed in Chacaltaya (where the study was conducted) for an additional 21 days before the measurements were made. The increases in hematocrit and hemoglobin were similar to other studies of sea-level natives acclimatizing to altitudes higher than 5,000 m (Boutellier et al., 1990). Thus, our subjects were well acclimatized to the altitude under investigation.
Because no measurements were made during the acute exposure to altitude or during earlier phases of acclimatization, the present study does not imply anything regarding CBF and metabolism, neither during the acute phase of hypobaric hypoxia nor during early acclimatization. It does, however, suggest that acclimatization to altitude ultimately results in a CBF and oxidative metabolism similar to that at sea level, because CBF was unchanged during the four conditions. The unchanged CBF at altitude actually reflects an increase in flow compared with that at sea level because the hematocrit was increased at high altitude, which leads to a decrease in CBF (Paulson et al., 1973). This “flow increase” was probably due to a persistent effect of decreased arterial oxygen tension, leading to cerebral arteriolar dilation. Thus, we suggest that the “unchanged” CBF (in absolute values) after several weeks of acclimatization to altitude was the result of two opposite forces: one acting to increase flow because of the persistent low arterial oxygen tension, and the other acting to decrease flow as a consequence of the increase in hematocrit.
The standard deviations of the CBF measurements were large but resemble those found in previous altitude studies (Marc-Vergnes et al., 1974; Sorensen et al., 1974). The relation between CBF and metabolism, however, could be further evaluated by analyzing the measured values of a-vDO2. In the present study, a-vDO2 was increased at high altitude as compared with sea level. This finding may be explained in at least two ways.
First, assuming that the cerebral metabolic rate of oxygen (CMRO2) is constant, a-vDO2 changes in inverse proportion to CBF. Thus, an increase in a-vDO2 indicates that CBF has decreased, whereas a decrease in a-vDO2 suggests that CBF has increased. Therefore, it is possible that CBF was decreased at altitude in comparison with sea-level values, even though this decrease was not detected in the Kety-Schmidt measurements. Such a decline might be explained by the increase in hemoglobin or hematocrit, or by the vasoconstrictive effect of an incompletely compensated hypocapnia. However, the effect on CMRO2 at high altitude was one of the main questions of the study, and it may not necessarily be true that CMRO2 was unchanged. Therefore, the second possibility is that, assuming that CBF was constant, CMRO2 increased slightly at high altitude compared with sea level and further during exercise at altitude compared with rest at altitude. Nonetheless, the present findings appear not to be compatible with the hypothesis of a decrease in CMRO2 at altitude, as suggested by others (Hochachka et al., 1994).
Compared with sea-level values, a persistent respiratory alkalosis was observed. All other variables being equal, alkalosis induces a left shift of the oxygen-dissociation curve of hemoglobin (the Bohr effect) and, hence, a decrease in P50. In other words, at a given partial pressure of oxygen, the binding affinity of oxygen to hemoglobin increases with increasing pH. However, despite respiratory alkalosis, jugular venous P50 was significantly higher at high altitude compared with sea level, and arterial P50 was unchanged. This may be explained by an altitude-induced increase in the erythrocyte concentration of 2,3-diphosphoglycerate (Lenfant et al., 1968). The finding indicates that the oxygen-dissociation curve after acclimatization to high altitude was unchanged or shifted slightly to the right (i.e., that oxygen was bound with less or unchanged affinity to hemoglobin at altitude compared with sea level). Furthermore, the finding suggests that the release of oxygen from hemoglobin is not impeded at altitude compared with sea level; thus, tissue oxygen delivery is not adversely affected as a result of an increased binding affinity of oxygen to hemoglobin.
The Kety-Schmidt technique
The Kety-Schmidt technique is considered the gold standard for global measurements of CBF, primarily because it is model independent (i.e., it works independently of the number of compartments and the underlying kinetics of the perfused organ). The major limitation of the method relates to the duration of tracer saturation and washout. The theory behind the method assumes that the saturation and the washout phase are of infinite duration, whereas these phases are of finite duration during practical use. Performing a computer simulation, Madsen et al. (1993) estimated that using 30 minutes for saturation and 10 minutes for washout leads to reproducible results but overestimates global CBF by 10% to 15% in healthy volunteers, compared with the theoretical situation in which both the saturation and the washout phase are of infinite duration. They proposed a correction algorithm that applies to the desaturation mode for133Xe, which we used in the present study.
In the present study, ventilation was increased from sea level to altitude and from rest to exercise. Thus, the pulmonary clearance of133Xe would be increased at altitude and during exercise, and the arterial curve would be steeper during these conditions. In effect, a larger fraction of the total area was included in the sampling period from 0 to 10 minutes, and the overestimation of CBF would therefore be reduced. Thus, even if the CBF was unchanged, there was a risk of finding a spurious decrease at altitude or during exercise. To estimate the magnitude of this error, we conducted a two-compartment analysis of the arterial curves that were obtained under the different conditions. Thereafter, a computer simulation was performed according to the approach of Madsen et al. (1993). For the hypothetical situation of a “true” CBF (i.e., a CBF∞) of 56 mL · 100g−1 · minute−1, a variation in CBF30,10 of less than 1% was observed when the different arterial curves were used. Thus, the variation in ventilation between the different measurements was not a significant source of error in this study.
One important limitation of the Kety-Schmidt method is that it provides only a one-dimensional (i.e., global) estimate of CBF. Hence, this method is not feasible for the detection of regional disturbances in CBF and metabolism. Thus, whereas acute exposure to high altitude is associated with global changes in CBF, the neurologic deficits associated with prolonged sojourn at extreme altitude may be focal or global (Hornbein et al., 1989). The present study does not exclude the possibility that focal perfusion or metabolic changes occurred that were not reflected in the global measurements of CBF and metabolism.
Previous studies on cerebral blood flow and metabolism after acclimatization to high altitude
The number of studies of CBF and metabolism after acclimatization of moderate or long duration is limited, but are generally in accordance with the present study. Two previous studies, which both were conducted in Bolivian native-altitude residents, used85 Kr and the Kety-Schmidt technique (Marc-Vergnes et al., 1974; Sorensen et al., 1974) and found resting CBF values similar to those obtained in the present study. Cerebral blood flow increased during acute hypoxia (Sorensen et al., 1974) and during acute hypercapnia (Marc-Vergnes et al., 1974), decreased during acute hyperoxia (Sorensen et al., 1974), and was lower in persons with higher hematocrit (Sorensen et al., 1974). In accordance with the present study, Sorensen et al. (1974) concluded that CBF was not increased in high-altitude natives residing at altitude, and that this finding was satisfactorily explained by the increase in hematocrit. They also suggested that the cerebral hypoxic vasodilatory response was preserved in these subjects. The findings apparently indicate similar mechanisms of adaptation in sea-level natives and high-altitude natives, with cerebral oxygen delivery being a persistent important determinant of CBF.
In apparent conflict with the present study, Hochachka et al. (1994) studied six sea-level natives before and after 63 days of acclimatization to altitudes of 3,200 to 4,400 m, and found significant decreases in regional cerebral glucose metabolism after acclimatization as measured by fluorodeoxyglucose and positron emission tomography. They suggested that the acclimatization response was different in lowlanders compared with high-altitude natives because the natives exhibited systematic reductions in glucose metabolism in most brain regions. However, both studies were performed shortly after arrival at sea level, which may in itself have triggered changes in CBF and metabolism. Therefore, and because the study addressed regional changes in metabolism, that study is not directly comparable with the present study of global metabolism.
Rootwelt et al. (1986) used single-photon emission computed tomography and133Xe to study regional CBF in 8 climbers and 23 control subjects before, immediately after, and 1 year after an expedition to Mount Everest (8,848 m). Climbers had significantly lower CBF than controls even before ascent; measurements after 1 year revealed a nonsignificant reduction in CBF compared with preexpedition levels. The increase in CBF after acetazolamide was larger in climbers than in controls, indicating preserved functional capacity of cerebral vessels. All measurements were performed at sea level. The climbers' hematocrit levels were significantly increased before and immediately after the expedition comparison with controls. Cerebral blood flow is reduced during polycythemia and increases after hemodilution (Henriksen et al., 1981; Paulson et al., 1973), which may explain why CBF was reduced immediately after the expedition. A high hematocrit value may also explain the low CBF value during the preexpedition study, if the study was conducted during acclimatization to altitude.
Exercise
Exercise at altitude induces additional arterial hypoxemia at high altitude, even after acclimatization (Sutton et al., 1988). At this time, the compensatory mechanisms that act to maintain cerebral metabolism during chronic hypoxia will be further challenged compared with resting conditions. In other words, if the systemic oxygen delivery is close to the limits at rest at high altitude, then exercise may bring cerebral oxygen delivery even closer to the limits. In this case, exercise may lead to decompensation caused by a decrease in the arterial oxygen content. However, exercise did not appear to decrease oxidative metabolism of the whole brain in the present study. Thus, the decrease in oxygen saturation was largely compensated by an increase in fractional oxygen extraction, so that the cerebral metabolic rate of oxygen during exercise did not differ from that at rest. Also, a cerebral lactate influx was observed during exercise instead of the slight efflux noted at baseline. Although the global net uptake or excretion of lactate may not be a reliable indicator of lactate metabolism, we would have expected a lactate efflux rather than an influx if the oxygen supply to the whole brain was critically affected during exercise. Finally, one measure of cerebral function was not impaired during exercise as indicated by a test of attention and reaction time. Therefore, we conclude that global cerebral oxidative metabolism was unaffected even during hypoxemia induced by submaximal exercise. Furthermore, none of the present results indicates that cerebral metabolism or function limits exercise capacity after prolonged acclimatization to high altitude.
There was a trend toward an increase in the cerebral metabolic rate of lactate at altitude during exercise compared with at rest (i.e., the arteriovenous difference shifted from negative to positive, indicating that the brain metabolized lactate during exercise in contrast to the efflux observed at rest). This phenomenon, which was not observed at rest and during exercise at sea level, was probably the passive consequence of an increase in arterial lactate during exercise compared with resting levels at altitude. Thus, during cerebral activation in rats, a net influx of lactate into the brain is observed with an increase in arterial lactate levels (Madsen et al., 1998).
CONCLUSIONS
In nine healthy sea-level residents acclimatized to high altitude over a period of 5 weeks, systemic respiratory changes included an increase in hematocrit and hemoglobin, a decrease in arterial oxygen tension and oxygen saturation, and a respiratory alkalosis in combination with an increase in P50. Even though the arterial oxygen tension was reduced at altitude, the arterial oxygen content and cerebral oxygen delivery were not reduced from sea-level values. Furthermore, CBF and cerebral oxidative metabolism were unchanged at high altitude compared with sea level. During exercise at high altitude, a cerebral lactate influx occurred, which apparently reflected the increase in the arterial lactate content. We hypothesize that the successful acclimatization of sea-level residents to moderately high altitude (5,000 to 6,000 m) leaves cerebral function unaltered. We found no indication that cerebral oxidative metabolism or function was critically affected during submaximal exercise, and the results did not indicate that cerebral function or metabolism is a limiting factor for exercise capacity at high altitude. Furthermore, the mechanisms of acclimatization to high altitude appear not to differ between native sea-level and high-altitude residents when comparing the results of the present study to previous studies performed with similar methods in the high-altitude population.
Footnotes
Acknowledgments:
The authors particularly wish to acknowledge the invaluable help of Professor Carlos Aguirre (Academia Nacional de Ciencias de Bolivia) and Dr. Pedro Miranda (Director of the Laboratorio de Cósmica Física, Chacaltaya), where the experiments were conducted. We also wish to thank laboratory technician Birgitte Jessen (Copenhagen Muscle Research Center, Rigshospitalet) for handling and processing the blood samples for blood gas analysis, and neuropsychologist Anders W. Gade (Department of Neurology, Rigshospitalet) for providing the CalCAP program.