Abstract
The ability to study multiple physiologic processes of the brain simultaneously within the same subject would provide a new means to explore the interactions between neurotransmitter systems in vivo. Currently, examination of two distinct neuropharmacologic measures with positron emission tomography (PET) necessitates performing two separate scans spaced in time to allow for radionuclide decay. The authors present results from a dual-tracer PET study protocol using a single dynamic-scan acquisition where the injections of two tracers are offset by several minutes. Kinetic analysis is used to estimate neuropharmacologic parameters for both tracers simultaneously using a combined compartmental model configuration. This approach results in a large reduction in total study time of nearly 2 hours for carbon-11–labeled tracers. As multiple neuropharmacologic measures are obtained at nearly the same time, interventional protocols involving a pair of dual-tracer scans become feasible in a single PET session. Both computer simulations and actual human PET studies were performed using combinations of three different tracers: [11C]flumazenil, N-[11C]methylpiperidinyl propionate, and [11C]dihydrotetrabenazine. Computer simulations of tracer-injection separations of 10 to 30 minutes showed the feasibility of the approach for separations down to 15 to 20 minutes or less. Dual-tracer PET studies were performed in 32 healthy volunteers using injection separations of 10, 15, or 20 minutes. Model parameter estimates for each tracer were similar to those obtained from previously performed single-injection studies. Voxel-by-voxel parametric images were of good quality for injections spaced by 20 minutes and were nearly as good for 15-minute separations, but were degraded noticeably for some model parameters when injections were spaced by only 10 minutes. The authors conclude that dual-tracer single-scan PET is feasible, yields accurate estimates of multiple neuropharmacologic measures, and can be implemented with a number of different radiotracer pairs.
The purpose of this study was to develop, implement, and validate positron emission tomography (PET) methodology that yields information about two different biochemical systems or two different neuropharmacologic aspects of the same system of the brain, at the same time, and during the same scanning session. Typically, studies of two distinct neuropharmacologic or biochemical measures with PET involve injecting the first radioligand and scanning for approximately 60 minutes, waiting up to 120 minutes for radionuclide decay, and then injecting the second radioligand and scanning for another 60 minutes. There are several obvious limitations of such a procedure. First, the long study duration (approximately 4 hours) makes it difficult to study certain patient groups. Second, these studies are expensive because they occupy the PET scanning room for an extended period. Third, when scans are separated by 2 hours, the biochemical or pharmacologic state of the subject might have changed. The dual-injection, single-scan approach developed here makes possible studies that were once entirely impractical. For example, if investigating how two neuropharmacologic systems interact in vivo, a research study involving a pharmacologic challenge and using traditional methods would necessitate four PET scans: two baseline scans (one for each ligand) and two challenge scans. Such a protocol would most likely take multiple days to perform, with the additional caveat that the physiologic or pharmacologic state may change during the course of a prolonged study. A dual-tracer protocol using a single PET acquisition with the two radioligands injected only 10 to 20 minutes apart offers a means of avoiding these problems. The scan time needed for a dual-tracer study would be reduced by approximately 2 hours, and interventional protocols involving a pair of dual-tracer studies (dual-baseline and dual-intervention scans) could be performed in a single morning or afternoon PET session. Furthermore, the neuropharmacologic measures estimated from the two radioligands would also be obtained over nearly the same time frame.
The rationale for dual-tracer PET stems from findings that a single neurochemical marker is often insufficient to fully characterize a neurologic disease. For example, parkinsonian dementia, multiple system atrophy, alcoholism, rapid eye movement sleep-behavior disorder, and diffuse Lewy-body disease have reported deficiencies in some aspect of the dopamine system. Many markers of dopamine function may be affected in Parkinson disease, but in other diseases only particular aspects of the dopamine system appear to be involved. Many PET and single-photon emission computed tomography radiotracers are available for studying different aspects of the dopaminergic system, including [11C]SCH-23390 for measuring D1-receptor density, [11C]raclopride for quantifying D2 receptors, [11C]WIN35,428 or [123I]β-CIT for measuring dopamine reuptake-site density, [11C]dihydrotetrabenazine (DTBZ) for measuring the vesicular monoamine transporter (VMAT2) binding-site density, and [18F]fluoroDOPA as an index of dopamine decarboxylase activity. In diseases involving both the number and the activity of dopaminergic nerve terminals, any or all of these markers have potential applications. We may be able to help better characterize these diseases by studying multiple aspects of the dopamine system.
An example of multitracer dopaminergic characterization is the investigation of altered presynaptic terminals and D2 receptors in unmedicated patients with Parkinson disease. It has been hypothesized that observations of reduced dopamine decarboxylase activity in Parkinson disease may be confounded by compensatory upregulation of enzymatic activity, whereas the density of synaptic vesicles, as reflected by VMAT2 binding, is a stable marker of presynaptic terminal integrity. This hypothesis could be directly investigated by performing [18F]fluoroDOPA, DTBZ, and raclopride studies in unmedicated patients at baseline, followed by repeat scans after administering a pharmacologic dose of a D2-agonist drug.
Multitracer characterizations also are possible with existing radioligands for studies of the cholinergic system with markers designed to measure presynaptic cholinergic vesicular binding ([123I]iodobenzovesamicol or [18F]fluoroethylbenzovesamicol), acetylcholinesterase (AChE) activity (N-[11C]methylpiperidinylpropionate [PMP]), butyrylcholinesterase activity (N-[11C]-methylpiperidinyl n-butyrate), muscarinic receptors (N-[11C]methylpiperidyl benzilate), and nicotinic receptors ([11C] or [18F]-labeled epibatine derivatives). An example of multitracer cholinergic characterization might involve the use of both [11C]PMP and N-[11C]-methylpiperidinyl n-butyrate to study the cholinergic deficits in Alzheimer's disease. In vitro studies (Davies, 1979; Perry et al., 1980) and, more recently, PET studies (Iyo et al., 1997; Kuhl et al., 1999) have shown acetylcholinesterase activity to be decreased in Alzheimer's disease, whereas butyrylcholinesterase activity has been reported to be increased by a factor of two to five or more in the hippocampus and temporal cortex (Perry et al., 1978; Mesulam and Geula, 1994).
Most other neurotransmitter systems can be studied with multiple markers examining different aspects of the system. For example, studies of multiple opioid-receptor types and of possible correlations or distinctions of agonist and antagonist recognition sites are possible through the use of [11C]carfentanil, [11C]diprenorphine, and [18F]cyclofoxy.
Besides studies designed to examine different aspects of one specific neurotransmitter system, the simultaneous examination of different neurotransmitter systems or examination of the interactions between two systems would be of great interest. For example, a project currently ongoing at our facility is designed to study both VMAT2 and benzodiazepine binding using [11C]DTBZ and [11C]flumazenil (FMZ) in healthy controls and in patients with Parkinson disease. This combination of tracers is one of the pairs reported in this work. The current acquisition protocol includes two separate 1-hour studies spaced in time by 2 hours, and thus requires a 4-hour time slot. With dual-tracer methodology, this time could be reduced by nearly a factor of two.
The present work focuses on the development, implementation, and evaluation of methods for the compartmental modeling and parameter estimation of dual-tracer, single-scan PET data. The specific goal of this work is to show the feasibility of such an approach. We view the potential utility of this methodology as one to aid in the study of the neurobiological bases of various central nervous system disorders. We begin with simulation studies that quantify the expected precision and biases of the methods, and then report results of the first dual-[11C]tracer PET studies performed in humans.
MATERIALS AND METHODS
Background
Typical PET neuropharmacologic studies have sufficient statistical quality to estimate from two to five kinetic parameters (Graham, 1985; Carson et al., 1986; Koeppe, 1990). There will be roughly double the number of parameters to be estimated for a dual-tracer procedure. This approach necessitates a parallel compartmental model configuration, which accounts for two distinct radiotracer input functions and two sets of rate constants. For a parallel model configuration, if the two tracers were injected simultaneously it would be impossible, without additional information, to interpret which set of parameter estimates corresponds to which tracer. Thus, the injection of one radiopharmaceutical must be offset in time from the other. Obviously, the farther in time the two injections are separated, the easier it will be to estimate and isolate individual parameters. However, to minimize the overall study duration and be able to make measurements of different biochemical systems nearly simultaneously, we need to inject the radiotracers as close together in time as possible.
Radiotracers
In this initial work, we chose to use radiotracers for which the kinetics had already been well characterized in single-injection PET studies: FMZ, a benzodiazepine receptor antagonist (Koeppe et al., 1991; Holthoff et al., 1991); DTBZ, a ligand for the VMAT2 binding site (Koeppe et al., 1996, 1997, 1999a); and PMP, a substrate for hydrolysis by the enzyme acetylcholinesterase (Kuhl et al., 1996; Koeppe et al., 1999b). Both FMZ and DTBZ can be classified as reversible tracers and have been analyzed successfully using both bolus and continuous infusion protocols; PMP can be classified as an irreversible tracer because the hydrolyzed product can not be converted back to authentic PMP and cannot cross the blood–brain barrier. We performed computer simulations for both injection orders for all three combinations of tracers: FMZ:DTBZ (FMZ injected first, followed by DTBZ), DTBZ:FMZ; FMZ:PMP, PMP:FMZ; and PMP:DTBZ, DTBZ:PMP. To date, we have performed human studies using both injection orders for two of these combinations: FMZ:DTBZ, DTBZ:FMZ; and FMZ:PMP, PMP:FMZ.
Simulation studies
Two types of computer simulations were performed: (1) those involving single computer-generated time–activity curves (TACs) based on kinetic model equations, and (2) those involving a computer-generated phantom based on a digitized Hoffman three-dimensional brain phantom warped into stereotactic brain atlas coordinates.
Single TAC simulations.
Noise-free TACs were generated for each tracer pair from model equations. For FMZ and DTBZ, noise-free curves were calculated using a model consisting of four rate constants: K1, k2, k3, and k4. For PMP, curves were calculated in the same manner, but with k4 = 0 because hydrolysis by AChE is irreversible. To examine the performance of the methods across a range of kinetic behaviors, TACs were generated using typical values for six different brain regions determined from actual human studies (cortex, cerebellum, hippocampus, pons, putamen, and thalamus). Noise-free dual-tracer data sets were calculated to simulate injection separations of 10, 15, 20, 25, and 30 minutes. Pseudo-Gaussian noise was added to each noise-free TAC, generating 1,024 sets of noisy data for each condition. The noise level was adjusted to that observed in actual PET volumes of interest (≈1 cm3) for three-dimensional scanning for injected doses of approximately 1, 3, 8, and 20 mCi per tracer. Results reported here are for 8-mCi simulations. A more complete description of these simulations has been reported previously (Koeppe et al., 1998).
Computer-generated three-dimensional brain phantom simulations.
Three-dimensional data sets of the brain simulating the isotope distribution of each of the three radiopharmaceuticals were produced from the transverse levels of a digitized version of the three-dimensional Hoffman brain phantom (Hoffman et al., 1991) warped into the stereotactic coordinate system of the Talairach and Tournoux atlas (1988). The brain was divided in 19 regions, identifying various cortical and subcortical structures, cerebellum and brainstem regions, white matter, and cerebrospinal fluid. Temporal sequences of three-dimensional data sets were generated by assigning each of the regions a temporally varying radionuclide concentration based on its unique set of kinetic parameters. Each three-dimensional data set was forward projected into sinogram space. Poisson noise was generated for each projection ray. Effects of detector response function, decay, scan duration, injected dose, attenuation, and random coincidences were included in the calculation of the noise level. After the generation of sinograms, the image volumes were reconstructed using filtered backprojection.
Human studies
The PET studies were performed on 32 healthy volunteers using either of two pairs of tracers: (1) FMZ and DTBZ, or (2) FMZ and PMP. Both injection orders were examined for each tracer pair (e.g., FMZ:DTBZ and DTBZ:FMZ), and the injected radioactivity was approximately the same for both tracers. Nine to fifteen mCi of each tracer was administered based on the subject's weight, and a dynamic sequence of 24 to 26 frames was acquired over a 70 to 80 minute period depending on separation between injections (10, 15, or 20 minutes). The scanning protocol for the 75-minute study was four scans × 0.5 minute, three scans × 1.0 minute, two scans × 2.5 minutes, one scan × 5.0 minutes, (second injection at 15 minutes after then injection), followed by four × 0.5 minutes, three × 1.0 minutes, two × 2.5 minutes, two × 5.0 minutes, and four × 10.0 minutes. Scans with 10- or 20-minute separations between tracer administrations have either one less or one more 5-minute scan before the injection of the second tracer. All studies were performed in three-dimensional mode on an ECAT EXACT-47 tomograph (CTI Pet Systems, Inc., Knoxville, TN, U.S.A.) Measured attenuation correction were performed from 12-minute duration two-dimensional transmission scans. Scatter correction was performed on all scans using the method of Ollinger (1993, 1995, 1996) as adapted by CTI PET Systems (Watson et al., 1995). Image sets were reconstructed using filtered back-projection (Kinihan and Rogers, 1989; Townsend et al., 1991) as implemented by CTI PET Systems using a Hanning filter with 0.5 cutoff for both in-plane and axial smoothing.
Radiochemical synthesis
Radiochemical synthesis of the two tracers was performed concurrently after single-target irradiation. No-carrier-added (+)-[11C]dihydrotetrabenazine, N-[11C]methylpiperidinyl propionate, and [11C]flumazenil were all synthesized by N-[11C]methylation of the corresponding desmethyl precursors. All radiotracers were obtained in specific activities exceeding 500 Ci/mmol at end of synthesis. Methods for simultaneous syntheses of two carbon-11 radiopharmaceuticals will be published separately.
Input function determination and metabolite analysis
The simultaneous kinetic modeling of two radiotracers presented the obvious challenge of determining separate input functions for both radiotracers. It was not sufficient to measure the total authentic radioactivity in arterial blood because we needed to determine the arterial concentration time course of authentic tracer for each radioligand separately from the radiolabeled metabolites. A total of 40 to 45 arterial blood samples were taken from the radial artery, plasma separated from erythrocytes and counted in a sodium iodine well counter. Standard counting and SEP-PAK (Waters, Milford, MA, U.S.A.)–based metabolite analysis were performed up until the time of the second injection. However, following the second injection the total radioactivity in arterial plasma was separated into three components, authentic ligand for the first tracer, authentic ligand for the second tracer, and labeled metabolites. It was not necessary to distinguish the metabolites of one tracer from those of the second as long as it has been verified that neither is able to cross the blood-brain barrier. Distinguishing between the authentic ligands required the use of two different SEP-PAK separations. One separation was designed to separate both authentic fractions from all labeled metabolites, whereas the second was designed to separate the authentic fraction of just one tracer from the authentic fraction of the second tracer plus the labeled metabolites. The difference between the two separations thus yielded the authentic component of the second tracer alone.
Standard methods for the separations for single tracers
For DTBZ, plasma samples were spiked with a trace amount of [3H]DTBZ, diluted with buffer, and passed through an activated C-18 SEP-PAK. All radiolabeled11C metabolites (there are no3H metabolites) and a fraction of the authentic [11C]DTBZ+[3H]DTBZ were eluted using a solvent mixture of 65/35 phosphate-buffered saline (pH 7.4)/ethanol. For DTBZ, there was approximately 20% breakthrough of authentic tracer into the “metabolite” fraction. The remainder of the authentic [11C]DTBZ+[3H]DTBZ was eluted using ethanol. Aliquots of both “metabolite” and “authentic” radiotracer elutions were then counted for both11C and3H. The fraction of3H in the “metabolite” elution was used to correct for11C breakthrough. For FMZ analysis everything was identical except that methanol was used instead of ethanol. [11C]flumazenil had approximately 50% breakthrough of authentic label into the “metabolite” elution (measured using [3H]FMZ). For PMP, samples were treated with physostigmine to stop radiotracer metabolism by blood cholinesterases, diluted with buffer, and passed through an activated C-18 SEP-PAK. All radiolabeled metabolites were eluted using a solvent mixture of 90/10 borate-buffered saline (pH 10)/ethanol. Authentic [11C]PMP was eluted using ethanol. Because there was consistently only 1% or 2% breakthrough of authentic PMP into the “metabolite” elution, the use of [3H]PMP corrections was unnecessary. Aliquots of both metabolite and authentic radiotracer elutions were then counted for11C.
At 10, 30, and 50 minutes after administration of the second tracer, at least three samples were analyzed with multiple SEP-PAK procedures to determine the concentration of authentic tracer for both ligands. For studies with [11C]FMZ followed by [11C]DTBZ, all samples collected before [11C]DTBZ injection were processed for the authentic FMZ fraction. All samples collected after [11C]DTBZ injection were processed using the DTBZ separation procedure described previously. Authentic [11C]FMZ stayed with the “metabolite” elution (< 0.3% breakthrough). Thus, this separation gave us the fraction of authentic [11C]DTBZ radioactivity in the plasma. The additional samples collected 10, 30, and 50 minutes after [11C]DTBZ administration also were processed using the FMZ separation procedure, during which all the authentic [11C]DTBZ and a portion of the authentic [11C]FMZ were separated from the remainder of the authentic [11C]FMZ and radiolabeled metabolites. The difference between these two measures, corrected by the [3H]FMZ data, gave the authentic [11C]FMZ fraction of the total [11C] plasma radioactivity. The same strategy was used for studies with [11C]DTBZ followed by [11C]FMZ. A slightly different calculation was needed for studies involving [11C]FMZ and [11C]PMP. The multiple samples were processed with both the FMZ and PMP separation procedures. Nearly 100% of both [11C]FMZ and [11C]PMP authentic fractions remained in the “authentic” elution of the PMP procedure giving the total authentic [11C] activity, whereas approximately 50% of the authentic [11C]FMZ (and [3H]FMZ) and 80% of the authentic [11C]PMP (and [3H]PMP) remained in the “authentic” elution of the FMZ procedure. These two equations were solved for the individual authentic fractions of [11C]FMZ and [11C]PMP.
Compartmental modeling and parameter estimation
Fig. 1 shows the combined dual-tracer parallel compartmental model configurations for [11C]FMZ plus [11C]DTBZ studies and [11C]FMZ plus [11C]PMP studies. Each model has two input functions and either four or five rate parameters. The combined model for FMZ and DTBZ consists of two parallel two-compartment two-parameter models, one for each tracer. This configuration is applicable for use with two reversible or rapidly equilibrating tracers. The combined model for FMZ and PMP consists of two parallel models, a two-compartment two-parameter model and a three-compartment three-parameter model. This configuration is applicable for use with one reversible and one irreversible tracer. The parameter k4 is fixed to zero because hydrolysis by AChE is irreversible. For two reversible tracers, four parameters were estimated: the transport rate constant K1 and the total distribution volume (DV) for each tracer. For the combined reversible/irreversible model, K1 and DV were estimated for the reversible tracer, whereas K1, k2, and k3, (or only K1 and k3 if K1 /k2 was fixed) were estimated for the irreversible tracer. Estimates were optimized by nonlinear least-squares analysis (Bevington, 1969). In addition to the four or five rate parameters, cerebral blood volume (CBV) and the time shift between brain and radial artery were considered. The time delay was estimated using a single large cortical region and the data before the second injection. In all subsequent fits for that subject, the time delay was fixed to the value determined for the large cortical region, whereas CBV was included as an additional model parameter. It was assumed that both the time delay and the fitted CBV value were the same during both the first and second tracer injections. Both voxel-by-voxel estimations, creation of parametric images of each of the rate constants, and parameter estimates for larger VOIs were performed. Images from all human studies were warped into the stereotactic coordinate system of the Talairach and Tournoux atlas (1988) as were the phantom studies of the digitized Hoffman brain. Thus, the predefined set of VOIs based on the stereotactic atlas was used to generate the regional time–activity curves for subsequent fitting.
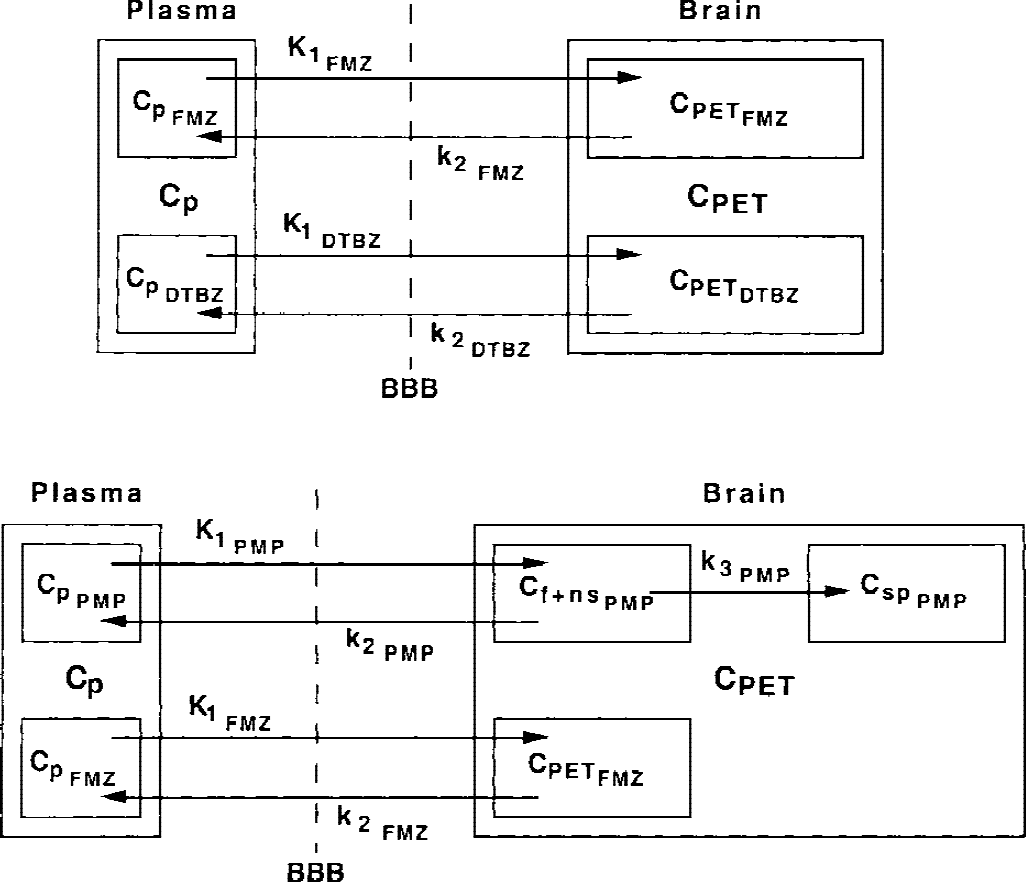
Compartmental model configurations for dual-tracer positron emission tomography studies. The top model was implemented for studies with FMZ and DTBZ, and this configuration is applicable to studies involving two rapidly reversible positron emission tomography radiotracers. Four parameters are estimated: K1 and DV(=K1 /k2) for both tracers. The bottom model was implemented for studies with PMP and either DTBZ or FMZ (shown here). This configuration is applicable to studies involving one reversible and one irreversible tracer. Four or five parameters are estimated: K1 and DV for the reversible tracer, and K1, k2, and k3, or only K1 and k3 (assuming as constant fixed value for K1 /k2) for the irreversible tracer. DTBZ, [11C]dihydrotetrabenazine; FMZ, [11C]flumazenil; PMP, N-[11C]-methylpiperidinylpropionate.
RESULTS AND DISCUSSION
Simulation studies
Single TAC simulations.
The studies for all six combinations of tracer pair plus injection order showed that both the precision and the bias in the estimates of model parameters were dependent on each of the following factors: (1) individual tracer (FMZ vs. DTBZ. vs. PMP), (2) specific pairing of tracers (e.g., FMZ and DTBZ vs. FMZ and PMP), (3) order of injection for a given pair of tracers (e.g., FMZ followed by PMP vs. PMP followed by FMZ), (4) brain region (e.g., cortex vs. putamen), (5) parameter (K1 vs. DV vs. k3), and (6) the separation time between the two injections.
Fig. 2 presents simulations of cortex and putamen for both injection orders: the tracer pair FMZ and PMP. Results for other regions and other tracer pairs led to similar conclusions, though the specific results of what regions and which parameters could be estimated with the greatest (or least) accuracy varied from condition to condition. The true parameter values for the simulations of Fig. 2 were K1FMZ = 0.30, DVFMZ = 7.5, K1PMP = 0.60, k2PMP = 0.15, k3PMP = 0.025 for cortex, and K1FMZ = 0.36, DVFMZ = 3.25, K1PMP = 0.72, k2PMP = 0.18, k3PMP = 0.875 for putamen. All K1 and DV values are given in mL·g−1min−1and mL/g, respectively. Both k2 and k3 values are given in min−1. The “true” DVFMZ values are calculated from the following combination of rate constants: (K1 /k2)(1 + k3 /k4), where these rate constants provide the values actually used to create the noise-free data TACs. The “true” input functions were used for the kinetic modeling. Results clearly show the effect of time between injections on the precision of parameter estimates. At the right of each panel, coefficients of variation (COVs) for single-injection studies of FMZ and PMP (i.e., nonoverlapping studies) are given for comparison. As expected, estimates of all parameters became less precise when injections were moved closer together, though some more than others; the magnitude of this effect varied considerably with region and injection order. For the FMZ and PMP pairing, there was not a particular injection order or separation time between tracer injections that could be considered optimal for all parameters and all regions. The K1 estimates for both tracers had little loss of precision as the injections were moved closer together, for either injection order. Injection offsets of as little as 10 to 15 minutes appear acceptable. The DV and k3 estimates, however, showed a definite interaction between the kinetics of the tracers for a given region and which tracer should be injected first. If one were interested primarily in cortical areas of the brain, then one would inject PMP first (lower left superior to upper left), whereas if the putamen were of greater interest (right panels), it does not appear to matter which tracer is injected first. Similar findings occurred for the FMZ and DTBZ pair (Koeppe et al., 1998), where injecting DTBZ first yielded better cortical estimates, whereas injecting FMZ first yielded better estimates in the putamen. For the DTBZ and PMP pairing (data not shown), both injection orders worked well in cortex, while neither order was very satisfactory in the putamen due to the relatively slow kinetics of both tracers in striatal regions. Given a specific region for any given tracer pair, estimates were more precise when the tracer that exhibits the more rapid clearance from tissue back to blood was injected first. In other words, because K1 values in gray matter structures are reasonably uniform across the brain, the tracer with the lower distribution volume (i.e., higher k2 value) or the tracer with the lower k3 value should be injected first.
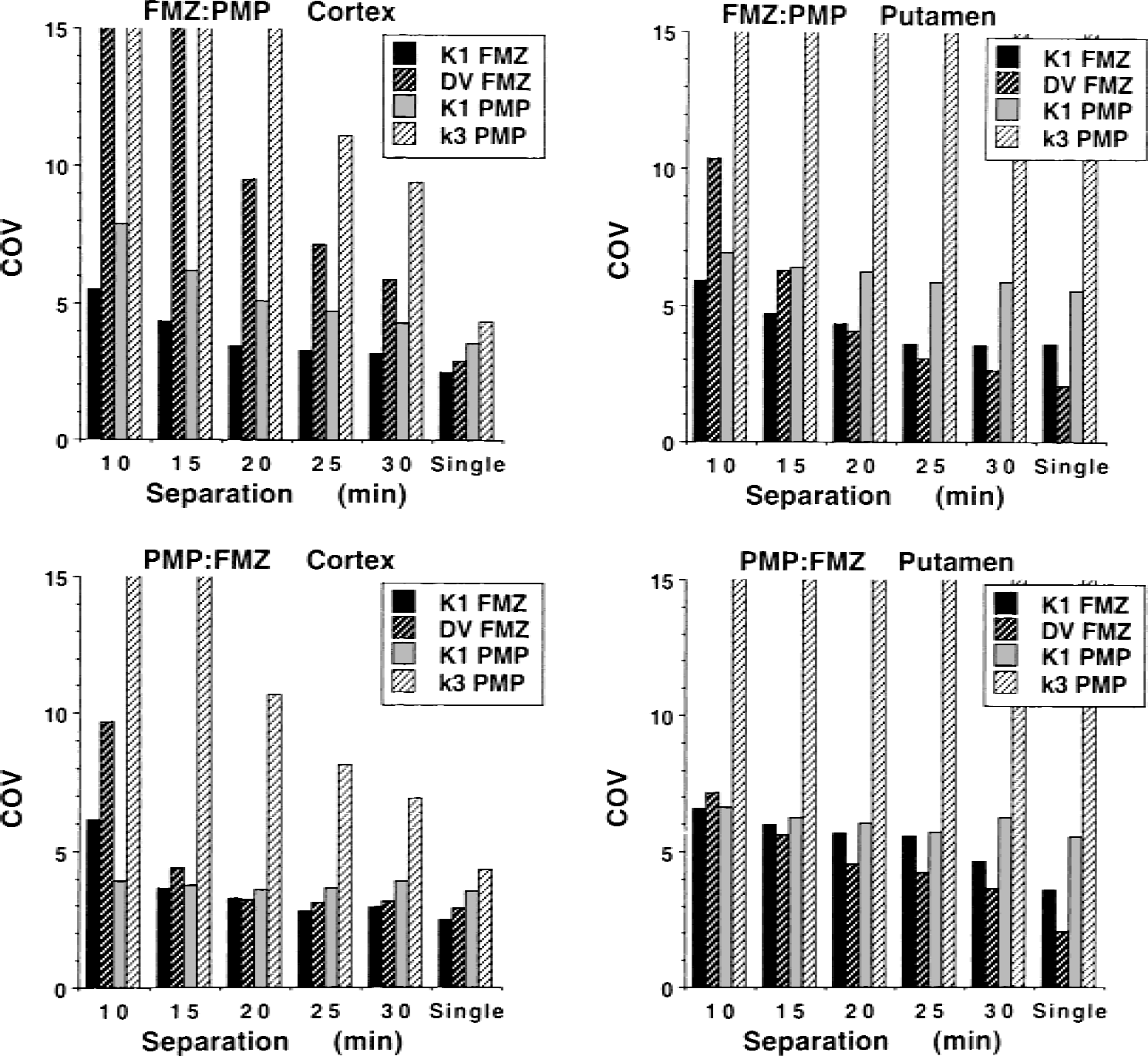
Coefficients of variation in K1 and DV estimates for FMZ and PMP from fits to 1,024 sets of noisy data. Noise levels approximate those for 8-mCi tracer injections and three-dimensional scanning. Each panel shows coefficients of variation for dual-tracer studies with injections separated by 10 to 30 minutes and coefficients of variation for single-injection studies. Results when FMZ is injected first are shown in the top panels, whereas results for PMP first are shown below. In general, estimates become less precise with decreasing tracer-injection separation. However, these data are highly region, injection-order, and parameter dependent. When PMP is injected first, cortical estimates are nearly as good as those derived from single scans, at a 15-minute separation. In contrast, estimates of PMP k3 in the putamen quickly become poorer as the separation between injections is reduced. DV, distribution volume; FMZ, [11C]flumazenil; PMP, N-[11C]methylpiperidinylpropionate.
Fig. 3 focuses only on injection separations of 20 minutes. We again can see the precision of the parameter estimates for tracer pairings of FMZ with PMP but also pairings of FMZ with DTBZ. In addition, the figure depicts systematic biases in and covariance between the various model parameters. The simulated values for FMZ and PMP are the same as in Fig. 2, whereas true DTBZ parameter values were K1FMZ = 0.40 and DVDTBZ = 3.8 for cortex, and K1FMZ = 0.48 and DVFMZ = 16.0 for putamen. As before, the “true” DV values for FMZ and DTBZ are calculated from the four rate constants, (K1 /k2)(1+k3 /k4). For FMZ with PMP, the differences in precision related to injection order noted in Fig. 2 are better elucidated. For cortex (top left), estimates are both precise and unbiased when PMP, the tracer with the more rapid kinetics, is injected first. There is only a slight negative correlation between PMP k3 and FMZ DV (r2= 0.156). However, injecting FMZ first results in a much stronger inverse coupling between these parameters (r2= 0.855). Because both high PMP k3 values and high FMZ DV values result in higher PET counts late in the study, the model, while able to correctly estimate the sum of the two components (i.e., the overall PET signal), cannot determine precisely how much of the overall uptake is due to each tracer. For putamen, the injection order again appears to make little difference. There is a slight negative bias (2%) in FMZ DV when FMZ in administered first, and only a 1% underestimation when PMP is injected first. N-[11C]methylpiperidinylpropionate k3 and FMZ DV are not strongly correlated for either order, with r2values of 0.001 (FMZ first) and 0.126 (PMP first). As seen in both Figs. 2 and 3, PMP k3 estimates in putamen have higher uncertainty than other parameters. As discussed in our previous work with [11C]PMP (Koeppe et al., 1999b), the high rate of hydrolysis by AChE in the basal ganglia causes a certain degree of flow-limited uptake, which causes a nonlinear relation between k3 and PET counts such that later PET scan results that are randomly high result in a greater overestimation in k3 than the corresponding underestimation in k3 caused if later scan results are randomly low. This finding can be seen in Fig. 3 (top right), where the majority of k3 estimates are fairly precise, yet those points that are inaccurate are all overestimated. There are, in fact, several estimates that are greater than the plot maximum of 1.75 min−1. These relatively few but inaccurate estimates result in the high k3 COVs for putamen seen in Fig. 2 (right). As with correlations between the two pharmacologic parameters, correlations between transport parameters (K1), or between a transport and a pharmacologic parameter (K1 and DV or K1 and k3) were dependent on the particular combination of radiotracers, the order of tracer administration, and the time between injections. Correlations between the transport parameter and the pharmacologic parameter for single-injection studies are given at the bottom of the table for comparison. It can be seen that correlations between parameters that approach ± 0.5 or 0.6 are common even in single-injection studies.
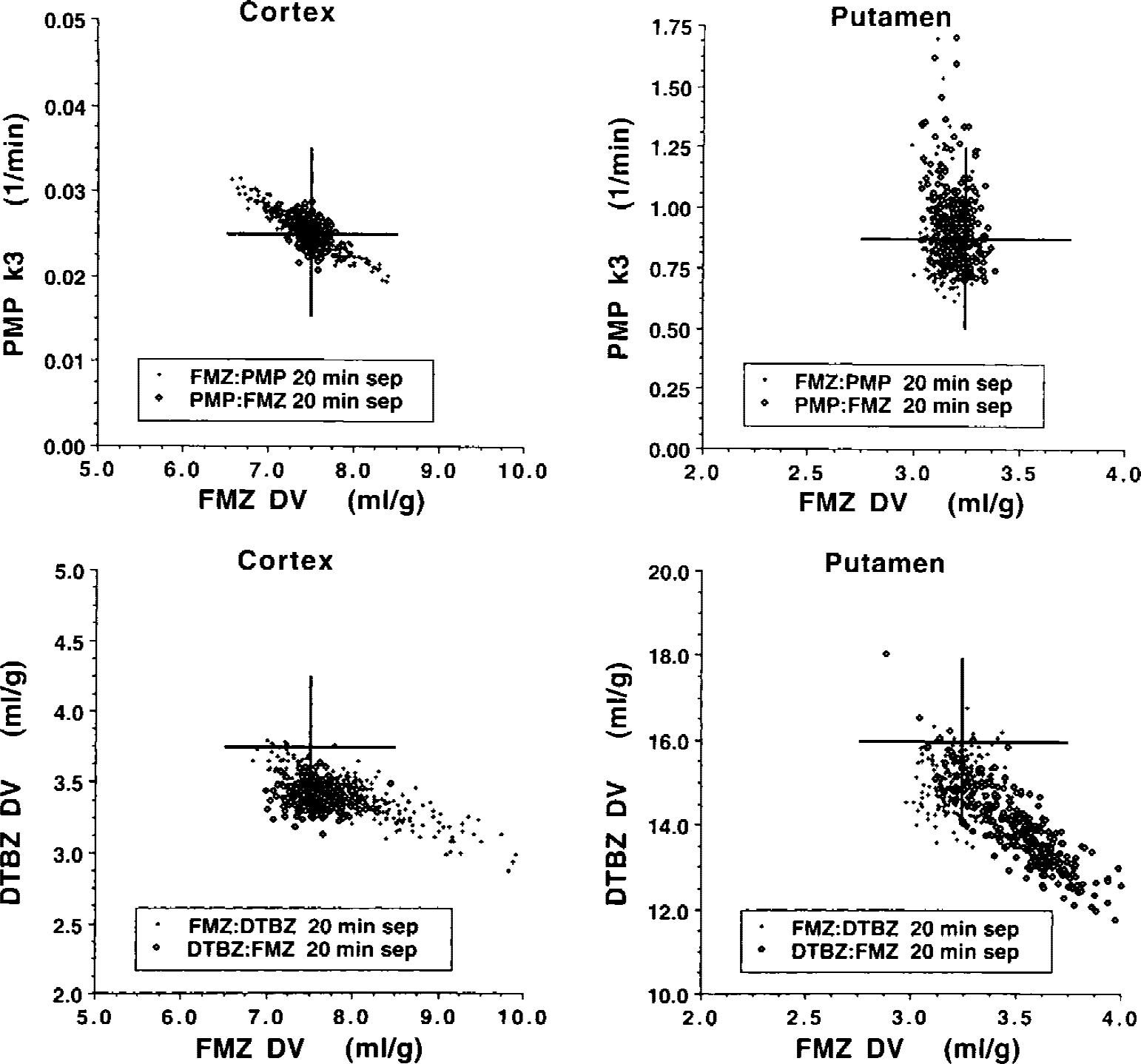
Bias and covariance in parameter estimates from dual-tracer positron emission tomography studies. Estimates are shown for simulations using FMZ and either PMP or DTBZ, both injection orders, and 20-minute separation between injections. The top pair of images shows estimates of PMP k3 and FMZ DV for simulations in the cortex (
Computer-generated three-dimensional brain phantom simulations:Fig. 4 shows voxel-by-voxel images at three different levels from the digitized Hoffman three-dimensional brain phantom for four model parameters: the transport rate parameter (K1) and the neuropharmacologic measure (k3 or DV) for each tracer. These results are for the PMP:FMZ protocol with a 20-minute injection separation. The quantitative scale of 0 to 100 applies across image sets differently as follows: 100 = 1.0 mL·g−1min−1for K1; 100 = 0.25 min−1for k3; and 100 = 10.0 mL g−1for DV. Noise levels corresponding to 8-mCi injections of each tracer, slightly lower than those used in the human studies, were used in creation of these data sets. The true input function for each tracer (i.e., the one used to create the noise-free time activity curves for each region) was used in the compartmental analysis. It is clearly evident that voxel-by-voxel estimation of parameters from multiple tracers is possible and, in fact, parameters are estimated with good precision and little bias other than those from partial volume effects. The ratio of K1 values between PMP and FMZ, which was always simulated to be exactly 2.0, (though absolute K1 values varied regionally) was estimated as 1.96 ± 0.08 across the 19 different regions. Absolute values tended to be underestimated because of partial volume, but the magnitude of this effect was nearly identical across tracers. Parametric images from the other combinations of tracers showed similar results, with trends that match the single-TAC simulations. For example, parametric images tended to be less noisy regionally when the tracer with the more rapid kinetics was injected first. Most combinations produce parametric maps with image quality that are comparable to single-tracer simulations. The one exception was for the DTBZ:PMP and PMP:DTBZ protocols that, though producing good results for cortex, yielded poor results in the basal ganglia, which was expected because the striatal kinetics for both tracers are relatively slow.
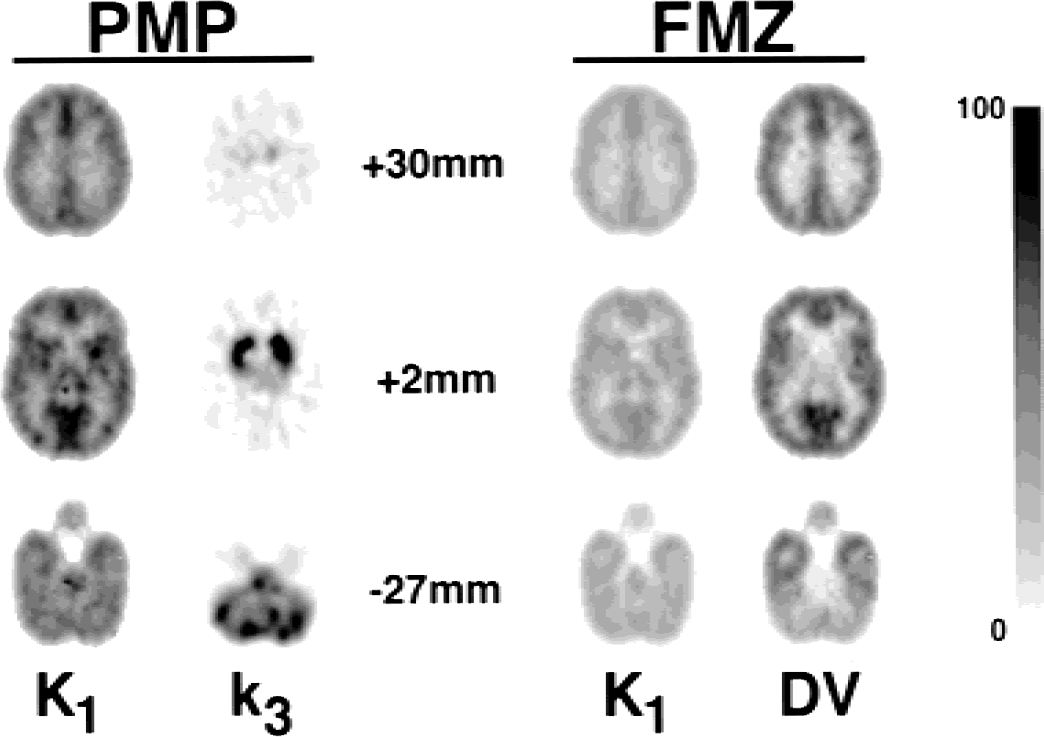
Parametric images from a PMP:FMZ simulation (20-minute injection separation) of the digital three-dimensional Hoffman brain phantom. Note the similarity of these images with a human study using the same dual-tracer protocol shown in the upper-left set of images in Fig. 7. FMZ, [11C]flumazenil; PMP, N-[11C]methylpiperidinylpropionate.
Human studies
Input function determination and metabolite analysis.
Arterial plasma input function measurements for two combinations of tracers are shown in Fig. 5. The top panel shows data from a FMZ:PMP study (20-minute injection separation), whereas the bottom panel shows comparable data from a FMZ:DTBZ study (20-minute separation). The filled circles show total radioactivity in plasma including authentic radioligand and radiolabeled metabolites for both tracers, and the open triangles show total authentic radioligand (sum of both tracers). In each study, FMZ was administered first and a standard metabolite analysis was performed up to the time of injection of the second tracer. After the second injection, samples drawn at 30, 50, and 70 minutes were analyzed by multiple SEP-PAK separations, allowing differentiation of the two authentic fractions. The inlays show the low count portions of the curves expanded for better viewing of the tails of the input functions. The authentic FMZ component for these samples is shown as the filled squares. The first 20 minutes of the curve plus the three authentic FMZ points with exponential fitting determined the calculated authentic FMZ input function. The authentic curves for the second tracer were calculated in roughly the same fashion, as described in Methods. All points are decay corrected, so the curves for the second tracer, injected nearly one half-life later, appear twice actual size. This fact is obscured because the input functions for FMZ tend to be approximately twice as large as the input functions for either DTBZ or PMP.
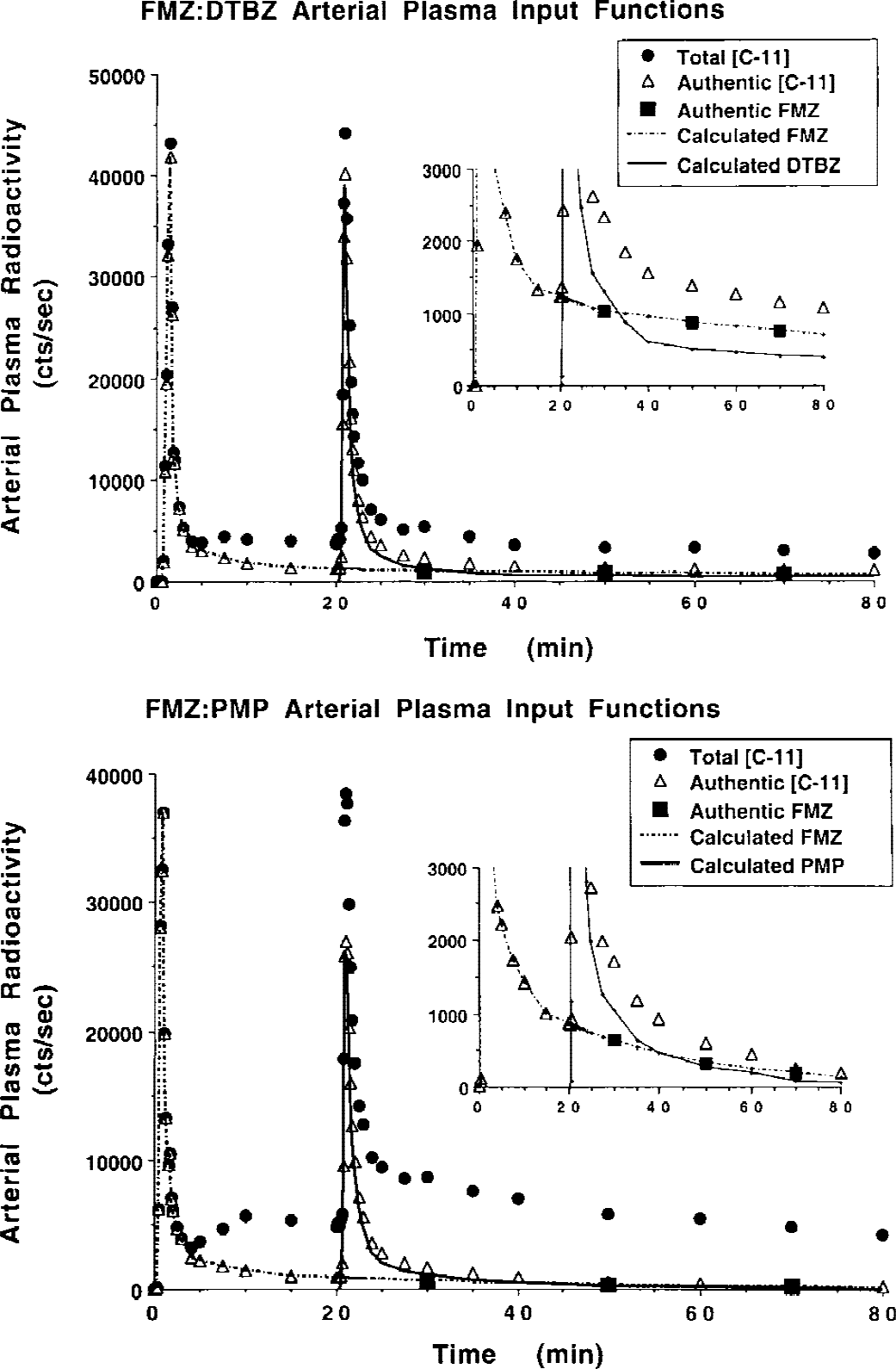
Arterial plasma input functions for dual-tracer positron emission tomography studies. The top panel shows blood data from a FMZ:PMP study, whereas the bottom panel shows comparable data from a FMZ:DTBZ study. The inlays show the tails for each set of curves on an expanded scale for better viewing. DTBZ, [11C]dihydrotetrabenazine; FMZ, [11C]flumazenil; PMP, N-[11C]methylpiperidinylpropionate.
The inlays show clearly that multiple SEP-PAK analyses are capable in separating not only authentic tracer from radiolabeled metabolites, but separating the two authentic input functions. In both examples, the estimated FMZ authentic components (filled squares) appear to fit well with the portion of the curve prior to injection of the second tracer. Because we do not distinguish between the metabolites of the two tracers, we can not compare the authentic fractions determined in dual-tracer studies directly with those from single-tracer studies.
Compartmental modeling and parameter estimation
Time–activity curves for various brain regions are plotted in Fig. 6. These data are from a PMP:FMZ study, also with 20 minutes between injections. As with the data in Fig. 5, curves are decay corrected and, thus, the relative contribution from the second tracer appears twice as high as the actual value. Data in the first 20 minutes predict the eventual pattern of PMP uptake; the putamen, cerebellum, and pons have the highest uptake (AChE activity), and the thalamus and cortical regions have a progressively lower uptake. After the FMZ injection at 20 minutes, the pattern reflects a mixture of the two tracers. The pons, with low benzodiazepine binding, clears most quickly. The radioactivity concentration in the putamen and cerebellum, with moderate benzodiazepine binding, remain higher than cortical regions because the high PMP retention, even though benzodiazepine binding is higher in the cortex. Therefore, although qualitative assessment of the uptake of the tracers is possible, kinetic modeling is absolutely essential for accurate quantification of the uptake and binding and retention of the two tracers.
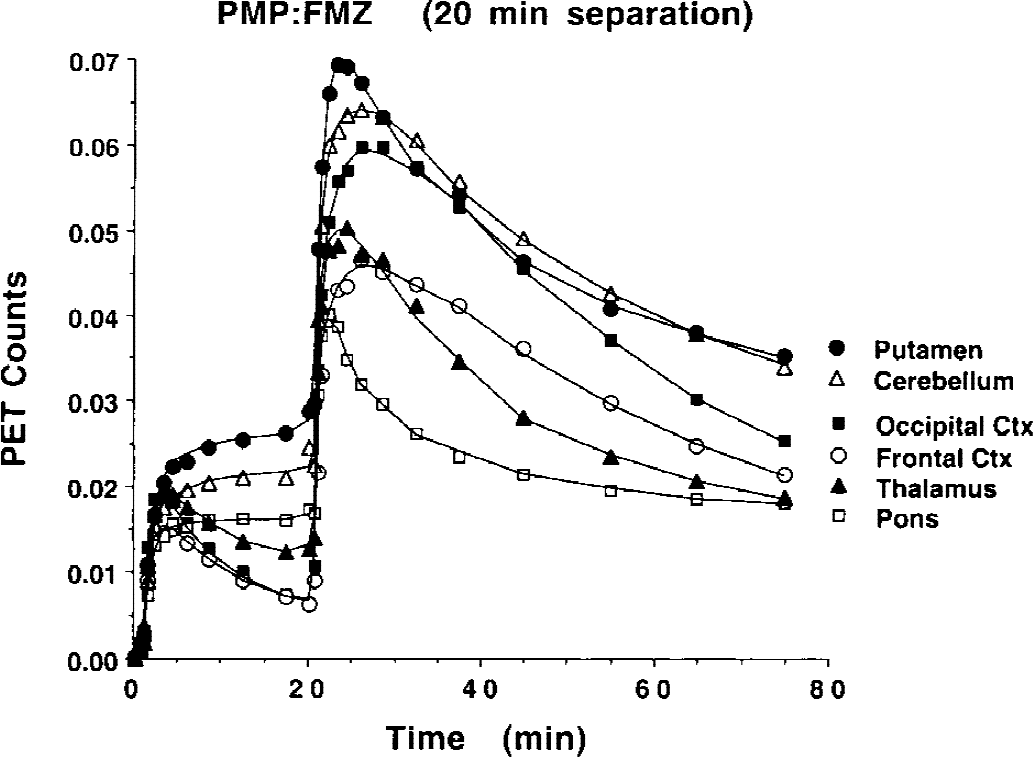
Brain time–activity curves for a dual-tracer positron emission tomography study. Data from 0 to 20 minutes shows the typical early pattern of uptake and retention for PMP. Injection of FMZ occurs at 20 minutes. The data is decay corrected, so the relative contribution of the FMZ is double that for PMP because the FMZ is injected at almost exactly one half-life into the study. FMZ, [11C]flumazenil; PMP, N-[11C]methylpiperidinylpropionate.
Compartmental analysis was performed both voxel-by-voxel and on the TACs from the larger VOIs. Because all data sets were coregistered and nonlinearly warped to match the Talairach atlas (Talairach and Tournoux, 1988), the set of 50 predefined regions based on the atlas were used both for generation of the TACs and for extraction of parameter estimates from the functional images. The parametric images from one study for each of the four dual-tracer protocols are shown in Fig. 7. All studies used a 20-minute injection separation. Although a few bad fits are detectable within each of the images, we are impressed with the overall image quality of each model parameter. The appearance of these images is nearly as good as is usually seen for studies derived from standard single-injection protocols. Some subtle differences can be observed that are consistent with the simulation studies. For example, with the combination of FMZ and DTBZ, parameter estimates in the cortex, particularly for DTBZ DV, appear less noisy when DTBZ was administered first (Fig. 7C), than in the study where FMZ was administered first (Fig. 7D).
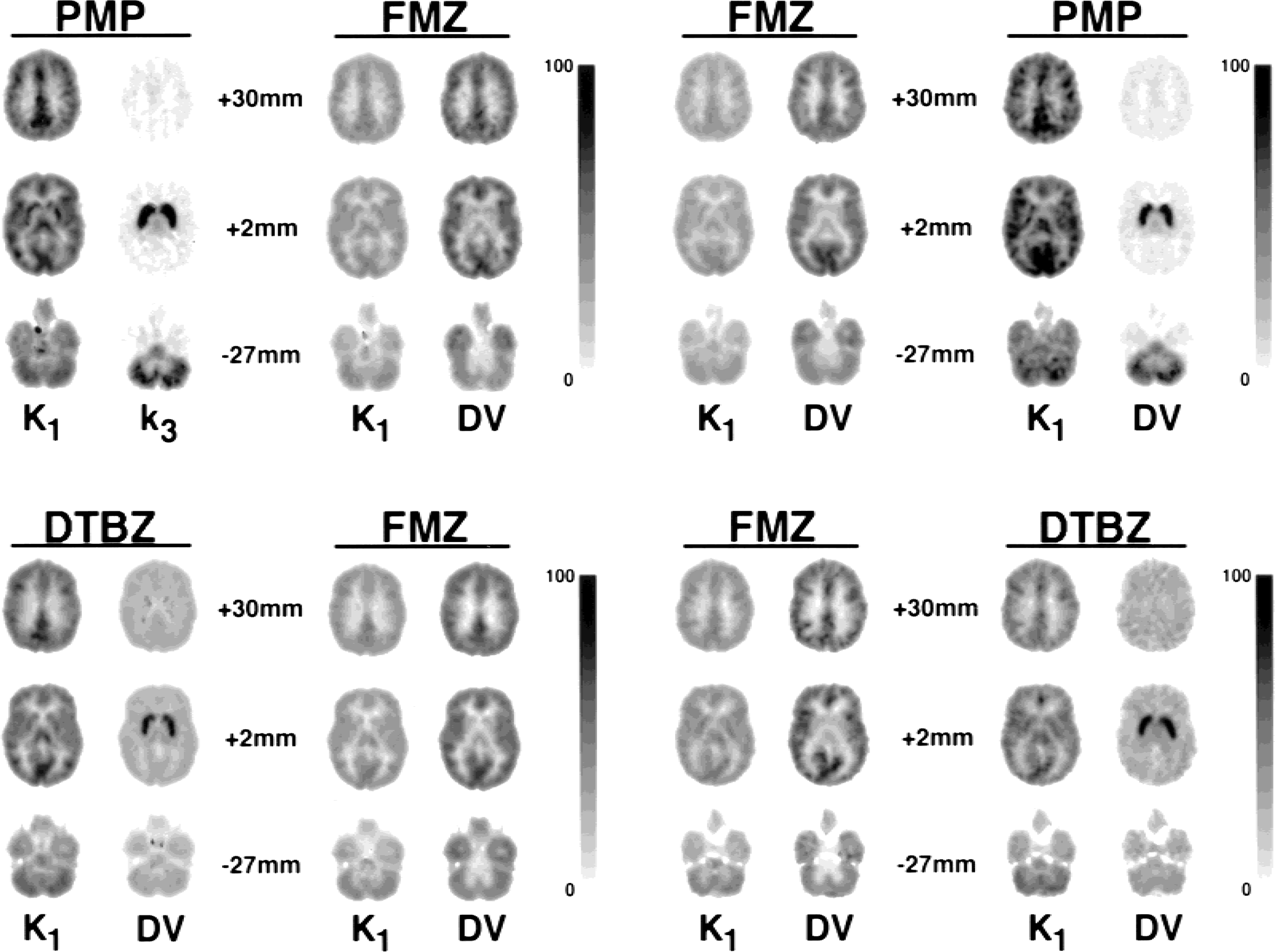
Parametric images for dual-tracer positron emission tomography studies. Voxel-by-voxel parameter estimates of four model parameters from one study of each of the four dual-tracer protocols are shown. For each panel, the first tracer injected is shown on the left, whereas images for the second tracer are shown on the right. All studies used an injection separation of 20 minutes. Each panel shows three different brain levels, where the numbers 30, 2, and −27 mm correspond to the distance of the image planes from the anterior commissure-posterior commissure plane of the Talairach atlas. The quantitative scale of 0 to 100 for each panel varies depending both on tracer combination and on the model parameter. The PMP:FMZ and FMZ:PMP panels have the following scales: 100 = 1.0 mL·g−1min−1for K1, 100 = 0.25 min−1for k3, and 100 = 10.0 mL/g for DV. Both DTBZ:FMZ and FMZ:DTBZ panels have these scales: 100 = 0.75 mL g−1min−1for K1, and 100 = 10.0 mL/g for DV. DV, distribution volume; DTBZ, [11C]dihydrotetrabenazine; FMZ, [11C]flumazenil; PMP, N-[11C]methylpiperidinylpropionate.
Parametric images for injection separation of 15 minutes looked nearly as good, and was slightly noisier on average, but occasionally showed larger covariance between model parameters. Parametric images from studies using only a 10-minute separation were noticeably noisier, and were even more likely have a greater degree of covariance between model parameters. This finding can be seen in Fig. 8, which shows images from one subject who received FMZ followed at 15 minutes by DTBZ. Note the relatively prominent basal ganglia in the middle DTBZ K1 image; this same pattern is not seen in the FMZ K1 image. Additionally, the DTBZ DV values in basal ganglia, though not obvious from Fig. 8, were lower than average, indicating an inability to accurately differentiate between parameters for this subject. Further investigation revealed that across the majority of voxels in the basal ganglia, the best-fit solution seemed to have ended in a local minimum of χ2space. Interestingly, fits to the larger volume TACs of the caudate and putamen for this subject did not exhibit this abnormal pattern, presumably because the fit did not get trapped in the same local χ2minimum, or that a local minimum did not exist in this same area within χ2space because of the improved statistical quality of the data afforded by a larger region.
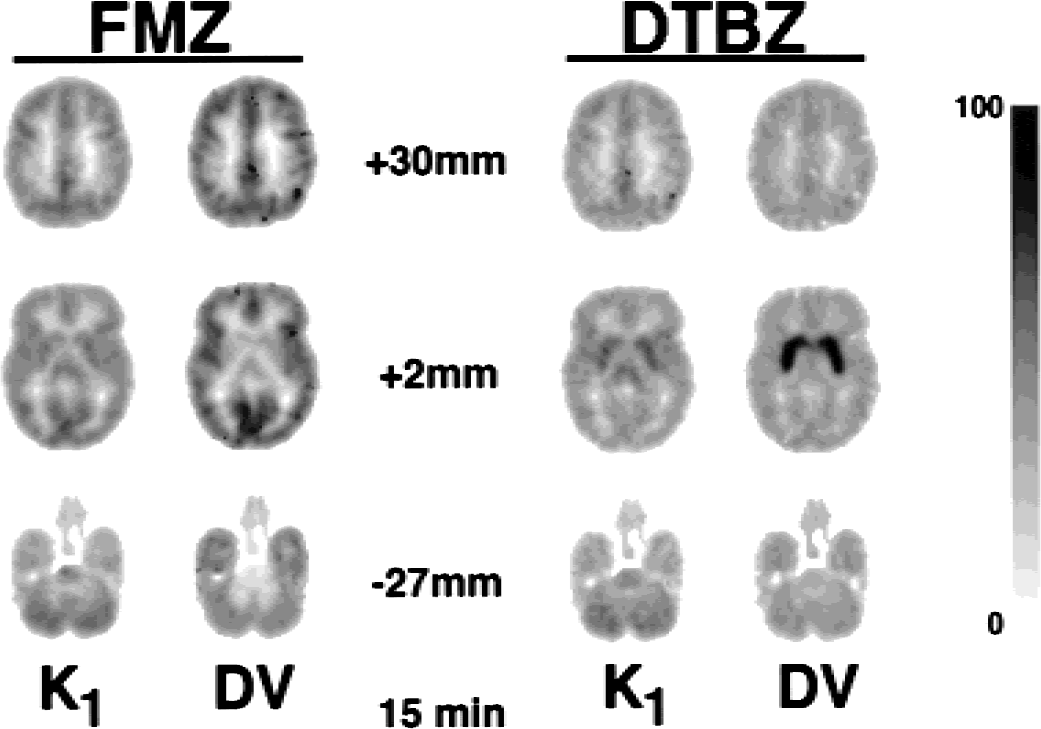
Parametric images from dual-tracer positron emission tomography studies with 15-minute injection separation. The layout is the same as for Fig. 7 panels. Although the overall image quality is good, note the increased DTBZ K1 estimates in the basal ganglia seen in the middle row of images. This uptake pattern is different than seen in the corresponding FMZ K1 image (
The absolute magnitudes and standard deviations of the estimated parameters from all dual-tracer studies with 20-minute separations between injections are shown in Fig. 9 and are compared with the corresponding values from single-injection studies for each of the three radiotracers. Fifteen healthy controls from a previous FMZ study in our facility, 12 from a previous DTBZ study, and 23 subjects from a previous PMP study were used as comparisons for the dual-tracer estimates. The FMZ DV estimates are shown at the top of the figure, the DTBZ DV estimates are shown in the middle, and PMP k3 estimates are shown at the bottom. In each panel, data are shown for 10 regions, with the single-tracer estimate shown as the right-most bar of each region. Each dual-tracer group consists of four subjects. For FMZ, there were 4 groups, 16 subjects in all: FMZ:DTBZ, DTBZ:FMZ, FMZ:PMP, and PMP:FMZ. For DTBZ and PMP, there were only two groups each: FMZ:DTBZ, DTBZ:FMZ, and FMZ:PMP, PMP:FMZ, respectively. There are some noticeable differences in the magnitude of the parameter estimates between dual-tracer and single-tracer studies. However, because of the small size of each dual-tracer group, it is difficult to determine with certainty whether any of these differences are real and likely to be reproducible. Unpaired t-tests were performed between each dual-tracer group and the corresponding single-injection studies for the same radiotracer. For FMZ, most comparisons were not statistically significant. However the following t-tests against the single-injection group yielded P values less than 0.05 uncorrected for multiple comparisons: DV in occipital cortex from the FMZ:DTBZ group, DV in parietal cortex from the FMZ:PMP group, and DV temporal cortex for both FMZ:DTBZ and FMZ:PMP groups. For DTBZ, the following tests against the single-injection group yielded P values <0.05: DV in cerebellum and in occipital cortex for the FMZ:DTBZ group. Other cortical regions for this group showed similar trends, with P values close to but greater than 0.05. Basal ganglia DV values in the DTBZ:FMZ group were marginally but not significantly lower (0.05 <P < 0.1) than the single-injection group. For PMP, no comparisons yielded P < 0.05.
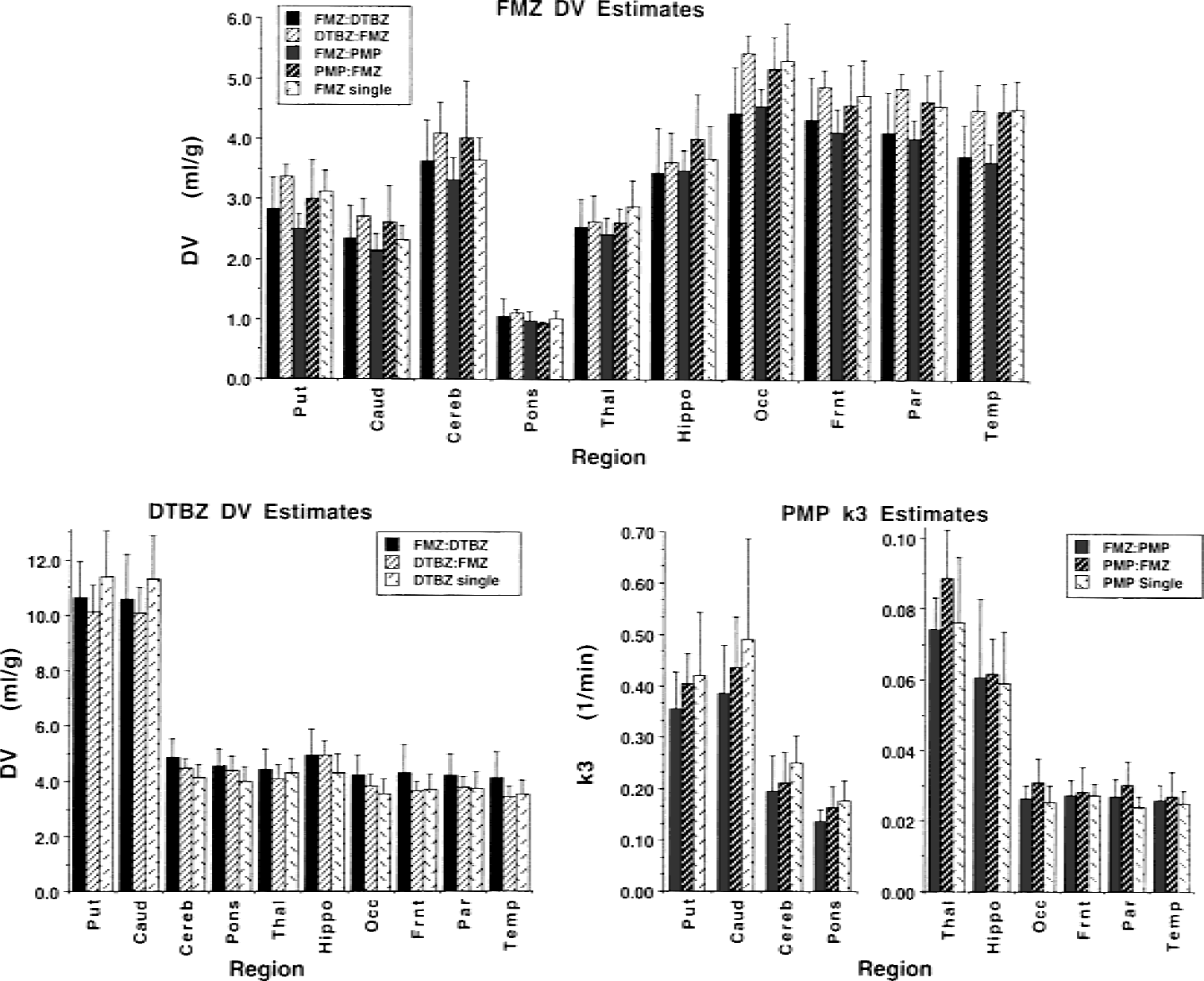
Comparsion of model parameter estimates for single- and dual-tracer positron emission tomography for various brain regions. The estimates for FMZ DV (
We compared the results from the human subjects with those predicted from the computer simulations and found both some definite consistencies and some inconsistencies. However, with only 32 dual-tracer subjects distributed across two different tracer combinations, each with two injection orders and with injection separations ranging from 10 to 20 minutes, one must be cautious in drawing firm conclusions. The general level of precision in parameter estimates was consistent with the single-TAC simulations. Simulations predicted relatively uniform coefficients of variation in the binding estimates across the brain for both FMZ and DTBZ in dual-tracer studies as well as in single-tracer studies. Precision of binding estimates in human studies across the brain (Fig. 9 top and middle) were indeed uniform. The 15 single-injection FMZ studies had coefficients of variation in DV ranging from 10% to 15% across the 10 brain regions shown in Fig. 9. The average COVs for the 4 groups of dual-tracer protocols using FMZ ranged from 10% to 17% across the same regions. For DTBZ, the 12 single-injection studies had COVs ranging from 12% to 16%. The average COVs for the two groups of dual-tracer studies using FMZ ranged from 11% to 17%. Precision in PMP k3 estimates was predicted to be better in regions of lower AChE activity (cortex), than regions with moderate activity (hippocampus, thalamus, pons, and cerebellum), and much better than in areas of high AChE activity (basal ganglia). For PMP in human subjects, the 23 single-injection studies had COVs ranging from 11% to 15% in cortical regions, 20% to 25% in regions with moderate AChE activity, and 29% to 40% in striatum. The COVs for the 2 groups of dual-tracer studies using FMZ ranged from 14% to 22% in the cortex, 16% to 32% in regions with moderate activity, and 20% to 30% in the striatum.
The predicted effects of injection order on precision and bias were less visible in the data. There were some consistencies; for example, the DV estimates in basal ganglia from the DTBZ:FMZ group were higher for FMZ (Fig. 9A) and lower for DTBZ (Fig. 9B) than those from the FMZ:DTBZ or the single-injection groups, as predicted by simulations (Fig. 3D). However, many of the more subtle results of the simulations were not observed. Cortical DV estimates for FMZ binding from the FMZ:DTBZ group, which showed positive biases in the simulations (Fig. 3C), were actually lower than estimates from single-injection studies or than those from any of the other dual-tracer groups (Fig. 9A). Also, the more subtle COV results from the simulations shown in Fig. 2 were not observed. For example, the prediction that COV would be higher for both FMZ DV and PMP k3 when FMZ was injected first (Fig. 2A vs. Fig. 2C) was not seen in the human studies. In fact, COVs were slightly lower for both parameters (Fig. 9A and C). Again, it should be pointed out that with groups of only four subjects, the reliability of the COV measures is limited. Furthermore, the simulations shown in Figs. 2 and 3 include only effects of noise in the PET data and not uncertainty in the measured input function. It is likely that a substantial portion of the intersubject error variability is related to the input function and metabolite measurement, and not just to noise in the PET data. Only when the variance related to the uncertainty in the PET measurement is high, as for PMP k3 in basal ganglia or cerebellum, does this source of error dominate. Therefore, variance due to true intersubject variability and to errors in the determination of the arterial input functions are likely masking the more subtle sources of bias and uncertainty illustrated in Figs. 2 and 3.
The results from the digitized Hoffman three-dimensional brain phantom simulations suggest an excellent degree of correlation with results from the human studies. The visual appearance of the parametric images from the PMP:FMZ phantom simulation shown in Fig. 4 reveals a striking similarity to the images from the human study using the same dual-tracer protocol (Fig. 9A), though warping of all image sets to a standard coordinate system helps in this regard. Parametric images from brain phantom studies simulating the other dual-tracer protocols shown in Fig. 9 also showed excellent agreement with the human studies. Digital Hoffman phantom simulations of new pairs of tracers will likely be useful in predicting their successful application to human studies.
Both the computer simulations and the scans in humans led us to conclude that even though dual-tracer studies are challenging to perform from both a radiochemistry and a blood sampling and metabolite analysis standpoint, they are definitely feasible and are capable of providing accurate measures of multiple neuropharmacologic quantities from a single PET scan. It may be surprising that parameter estimation accuracy was not compromised to any great extent. Although the number of fitted parameters has been doubled, the precision with which individual parameters were estimated did not worsen nearly to the same degree as occurs when a single-tracer data set is analyzed with additional parameters (e.g., [11C]FMZ analyzed with four instead of two rate constants). Parameter estimation using compartmental modeling is based on analyzing the shapes of the tissue TACs for various regions of the brain relative to the arterial plasma input function. Kinetic information pertinent to the estimation of individual rate parameters exists only when there is a net flux of tracer either entering or leaving the various model compartments, which occurs only when these compartments are not at equilibrium. With dual-tracer protocols, the approach toward equilibrium in the brain is disrupted by the administration of the second tracer and additional “kinetic” information becomes available that helps to differentiate the various model parameters of the two radiotracers. Therefore, although the number of fitted parameters is doubled, the “kinetic content” of the data increases dramatically. As the time between the injections is decreased, the total “kinetic content” is decreased and the ability to differentiate between model parameters also is reduced. This approach of increasing the kinetic information through multiple injections has been used in a more general way by several investigators (Bahn et al., 1989; Huang et al., 1989; Delforge et al., 1993, 1995; Wong et al., 1986a, 1986b; Morris et al., 1996a, 1996b). Common to each of these studies is the use of multiple administrations of the same radiotracer, often at different specific activities, either to estimate individual model parameters more precisely or to estimate additional parameters needed when using a more complex model (e.g., Bmax and KD instead of only Bmax /KD). In the present study, a second but different radiotracer is used to increase the kinetic content of the study and to allow the measurement of two different neurochemical parameters.Table 1
Correlations between estimated model parameters of dual-tracer and single-tracer studies
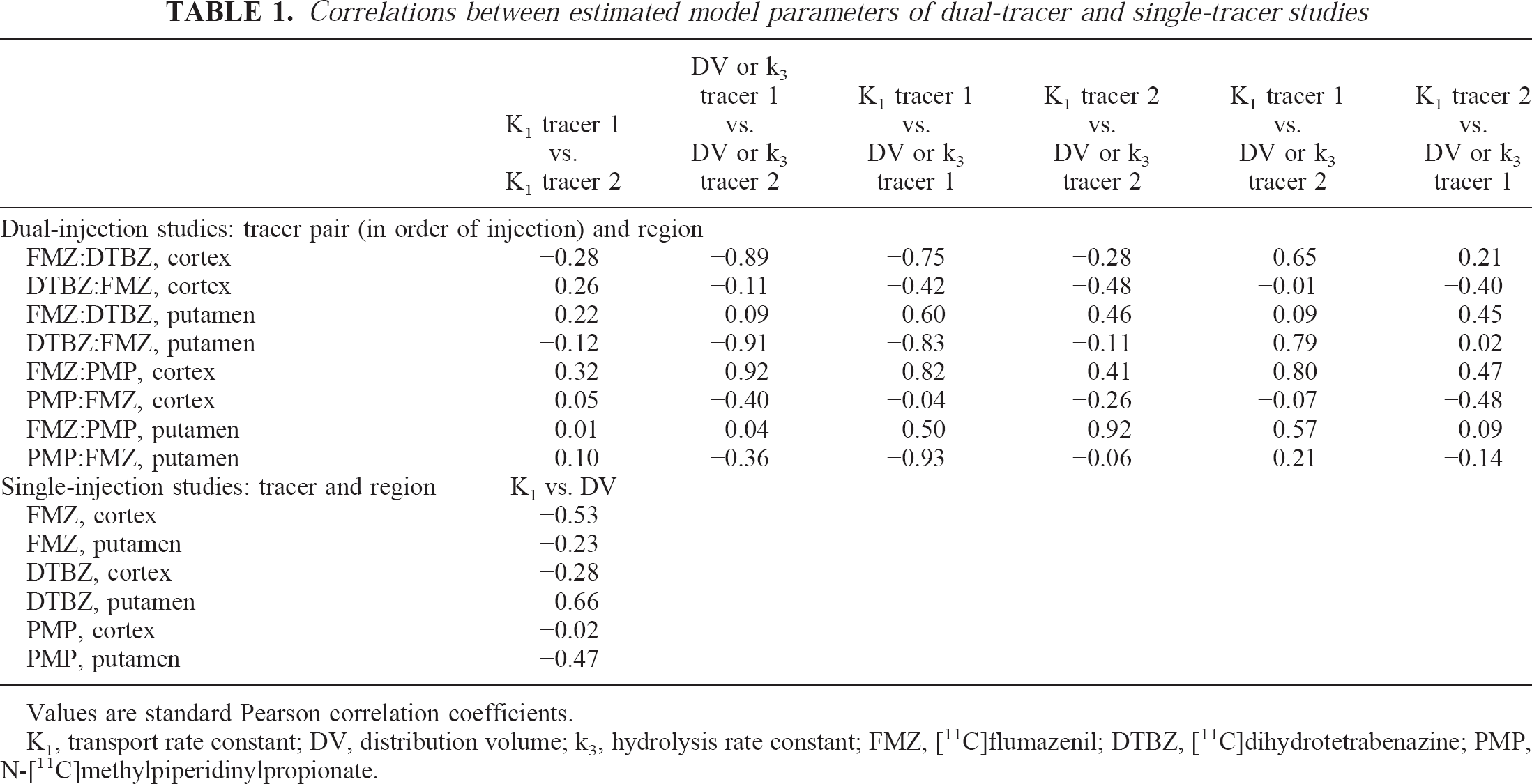
Values are standard Pearson correlation coefficients.
K1, transport rate constant; DV, distribution volume; k3, hydrolysis rate constant; FMZ, [11C]flumazenil; DTBZ, [11C]dihydrotetrabenazine; PMP, N-[11C]methylpiperidinylpropionate.
SUMMARY AND CONCLUSIONS
In summary, temporally overlapping dual-tracer PET using a single data-acquisition sequence is feasible. Under many conditions, the model parameters for two different [11C]-labeled radiotracers injected only 10 to 20 minutes apart can be estimated nearly as accurately as in standard single-injection studies. It may be difficult to translate the conclusions drawn from these present studies into general conclusions applicable to any tracer pair, so each new pair considered for use in dual-tracer designs must be evaluated independently. However, the first tracer injected should have sufficiently rapid kinetics so that the model parameters describing its in vivo behavior are reasonably well determined by the time of the second injection. Future work on dual-tracer scanning will continue with additional studies using the digital three-dimensional Hoffman brain phantom to explore more fully how tomograph and scanning properties affect these studies, and with the development of the methods needed to rapidly separate the authentic arterial plasma fractions of the two tracers. We are currently exploring dual-tracer studies that use two irreversible tracers, estimating a total of six kinetic parameters, and also are beginning dual-tracer validation studies using challenge protocols to determine sensitivity and specificity of model parameter estimates to specific interventions. The dual-tracer methods as described here, though complex in nature, offer new opportunities for obtaining neuropharmacologic information related to multiple biochemical systems or multiple aspects of the same biochemical system in a single PET scanning session.
