Abstract
To investigate effect of assuming of upright posture on brain hemodynamics in patients with unilateral internal carotid or middle cerebral artery occlusion (MCAO), local tissue oxygen extraction fraction (OEF), and postural changes in regional cerebral blood flow (rCBF) during supine and sitting conditions were examined using positron emission tomography (PET) with 15O-gas steady-state method and H215O autoradiographic method. A total of 22 minor stroke patients at relatively early stages participated. The regions of interest method was used for analyzing levels of perfusion and oxygen metabolic parameters, and postural rCBF change within MCAO group was investigated using statistical parametric mapping. Region of interest analyses showed significant rCBF reduction in the cortical and subcortical regions distal to the artery occlusion in CAO patients during sitting. Regression analyses showed that magnitudes of rCBF reduction in those areas were correlated positively with OEF values and inversely with metabolic rates of oxygen (P < 0.05). Statistical parametric mapping for MCAO patients demonstrated further rCBF reduction by sitting in the occlusion-side MCA territory. The current study suggested that assumption of upright posture could exert an adverse effect on local perfusion in hemodynamically compromised patients with major cerebral vessel occlusion, possibly caused by impairment of local autoregulation.
Keywords
With advancing age, the assumption of the upright posture often accompanies neurologic symptoms such as dizziness, falls, or syncope (Graafmans et al., 1996; Lipsitz, 1989). These postural cerebral symptoms are considered to depend on the extent to which regional cerebral blood flow (rCBF) and oxygen delivery become impaired because of cerebral dysautoregulation (Paulson et al., 1990). A recent near-infrared spectroscopy study showed that upright posture decreased oxygen supply to the frontal cortex in humans with age (Jannet Mehagnoul-Schipper et al., 2000). The postural effect on the blood pressure was well accepted because postural hypotension predicted cerebrovascular mortality rates (Raiha et al., 1995) and because sudden declines in the blood pressure in patients with long-standing hypertension could induce cerebral ischemia (Meyer et al., 1973; Strandgaard and Paulson, 1989). A previous animal experiment showed that reduction of blood pressure to less than the lower limit of autoregulation caused heterogeneous blood flow with scattered ischemic regions (Hamar et al., 1979). In such ischemic regions distal to an arterial occlusion, the arteriolar pressure is considered to be less than the lower limit of autoregulation (Paulson, 1970; Symon et al., 1976). This might be true in patients with internal carotid artery occlusion (ICAO) because they exhibited regional changes in oxygen extraction fraction (OEF) gradually elevated in an axial direction on the occluded ICA territory (Yamauchi et al., 1990). Then, the question remains whether assuming of the upright posture would cause a negative effect on cerebral circulation in those hemodynamically compromised patients.
Previous imaging studies of cerebral perfusion during the orthostatic stress condition showed further reduction in accumulation of a lipophilic perfusion tracer 99mHM-PAO using single photon emission tomography in patients with cerebral ischemia (Hayashida et al., 1993). Because the frontal perfusion was reported to decrease to some extent during standing even in healthy subjects (Warkentin et al., 1992), it is likely that the ischemic brain areas might be so hemodynamically vulnerable to the orthostatic stress that they are lacking in common autoregulation. Although an application of the orthostatic stress test can estimate changes in relative changes in perfusion (Hayashida et al., 1993; Passant et al., 1993; Warkentin et al., 1992) or flow velocity (Levine et al., 1994; Schondorf et al., 1997), it does not allow measurement of absolute changes in rCBF during the upright posture condition.
The authors recently reported a new method of measuring rCBF during the assumption of upright posture in human subjects using positron emission tomography (PET) (Ouchi et al., 1999). To the authors' knowledge, there have been no reports about quantitative measurement of rCBF during sitting or standing in patients with cerebrovascular disease. Thus, the current study was designed to investigate absolute changes in hemodynamics in the ischemic brain region by assuming of the upright posture in patients with unilateral major cerebral artery occlusion.
MATERIALS AND METHODS
Patients
A total of 22 patients (13 men and 9 women), aged 41 to 75 years (mean ± SD, 61.2 ± 8.2 years), with angiographically proven unilateral CAO (12 with unilateral ICAO and 10 with unilateral middle cerebral artery occlusion [MCAO]) participated in 15O-gas/15O-H2O PET study, and 8 healthy volunteers (control group; 6 mean and 2 women, mean ± SD, 56.4 ± 9.6 years) participated in 15O-H2O PET study. Details about each patient's characteristics are described in Table 1. Angiographic studies revealed consistent presence of various amounts of collateral circulation through the circle of Willis in each case. Neurologic assessment showed that 8 patients were totally asymptomatic and the other 14 patients suffered from mild-to-moderate neurologic deficits in the appropriate cerebral artery territory at the time of PET measurement. All patients underwent blood analyses for determination of risk factors and magnetic resonance imaging (MRI). Magnetic resonance imaging showed mild-to-moderate subcortical abnormalities (high intensities on the T2-weighted images) in the territory of ICA on the occluded side, the volume of which were less than 2.4 cm3 measured by a DrView software (DrView, Asahi Kasei Co, Tokyo, Japan) (Ouchi et al., 1998). Medical treatment was temporarily ceased at the time of PET measurement. None of the participants in the control group had neurologic problems, medical treatment, or MRI abnormalities. The current study was approved by the Ethics Committee of the Hamamatsu Medical Center and all participants gave their written informed consent after the nature and possible risks of the experiment were explained.
Patient characteristics

ICAO, internal middle cerebral artery occlusion; MCAO, middle cerebral artery occlusion; DD, disease duration from onset to positron emission tomography measurement (months); TIA, transient ischemic attack; HIAs, high intensity areas; Cx, cortex; subCx, subcortex; WM, white matter; NS, nothing special; R, right; L, left; anti-HT, anti-hypertensive drug; anti-HMG, 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase inhibitor for treatment of hypercholesterolemia; anti-DM, oral drug for diabetes mellitus.
Postural condition and physiology
Because some patients with mild motor weakness had difficulties in assuming of voluntary upright position, each participant sat passively (reclined) with eyes open staring forward on the scanner bed tilted at an angle of 75°. Catheterization in the left brachial artery and electrocardiogram prepared in each subject allowed us to monitor continuously the arterial blood pressure (ABP) and the cardiac rhythm during the sitting condition. Other physiologic parameters such as Pao2, Paco2, and pH were measured periodically. The authors' preliminary study before PET measurement showed short-term elevation of systemic ABP after postural shift from the supine to the sitting position followed by gradual decline to the plateau level within approximately 3 minutes, agreeing with results from a recent ultrasonics study (Novak et al., 1998). The current orthostatic PET was performed during the plateau period of ABP.
Positron emission tomography and magnetic resonance imaging procedure
Specifications of the high resolution PET scanner (SHR2400, Hamamatsu Photonics, Hamakita, Japan) have been reported elsewhere (Yamashita et al., 1990). Briefly, the scanner with 5 detector rings yielding 9 slices simultaneously offers spatial resolution of 2.7 mm (full-width at half-maximum) transaxially and 5.5 mm axially and 80 mm axial field of view. This PET system had a mobile gantry that enabled vertical movement and tilting (−20° to +90°). Just before PET measurement, each participant underwent MRI for determination of the scanning brain area by using a static magnet with 3-dementional mode acquisition (0.3 T MRP7000AD; Hitachi, Japan) (Ouchi et al., 1997, 1999). By tilting the PET gantry parallel to intercommissural line determined on the MR image, the axial field of view could cover the area from the lower part of the superior frontal gyrus to the upper half of the cerebellum. After backprojection and filtering (Hanning filter; cut-off frequency 0.2 cycles/pixel), image resolution was 6.0 × 6.0 × 6.5 mm full-width at half-maximum. The voxel of each reconstructed image measured 1.45 × 1.45 × 8 mm.
After fixation of the head to the head holder using a specially made thermoplastic facemask, a 20-minute transmission scan was performed for attenuation correction with a 68Ge/68Ga source in the supine position. Then, based on the 15O-gas steady-state inhalation method (Frackowiak et al., 1980), 3 PET emission scans were performed consecutively while individuals inhaled 200 MBq C15O2/min for 5 minutes, 400 MBq 15O2 /min for 5 minutes, and a short period of 1500 MBq C15O flowing through a tightly fitted plastic oxygen face mask attached to the subject's face. The latter C15O data were used for measuring the cerebral blood volume (CBV) and correction for OEF values (Lammertsma et al., 1987; Lammertsma and Jones, 1983). During each of the three acquisition periods, three or two pairs of arterial blood samples were collected (Meyer and Yamamoto, 1984). Measurement of arterial blood gas, oxygen saturation (Sao2), arterial hemoglobin (Hb), and hematocrit values allowed the authors to calculate the arterial oxygen content (Cao2) using the following formula: Cao2 = 1.39 × Sao2 × Hb. The functional quantitative images for CBF and CBV-corrected OEF were reconstructed on a pixel-by-pixel basis. The pixel-based cerebral metabolic rate of oxygen (CMRO2) image was finally created using the following equation: CMRO2 = CBF × OEF × Cao2.
After completion of 15O-gas, 120-second dynamic emission scan (10 seconds × 12 frames) was performed while subjects in the supine position received a 600-MBq bolus of H215O through the right cubital vein by an automated injector. Simultaneously after injection, arterial blood was continuously withdrawn through the left brachial artery using the automated arterial blood γ-ray coincidence counter (BACC-2; Hamamatsu Photonics K.K., Hamamatsu, Japan) yielding arterial input data per second (Ouchi et al., 2000). After this supine H215O PET scan, the subject's head holder was temporarily removed from the receiver of scanner's gantry, and the subject reclined on the 75°-tilted scanner bed. Approximately 12 minutes later, the second quantitative H215O PET scan was performed in the sitting position. During this series of PET measurements, all physiologic parameters described above were monitored continuously. Estimation of quantitative rCBF was estimated using a 90-second H215O data accumulated after the tracer entered brain circulation by summating the dynamic frames based on the autoradiographic method (Herscovitch et al., 1983).
Data analysis
Region of interest analysis.
Multiple circular regions of interest (ROIs), each containing 96 pixels (1.58 cm2), were placed bilaterally over the cerebellar hemisphere, lower frontal area (the orbitofrontal and inferior frontal cortices; [Brodmann area or BA: 10/11]), upper frontal area (middle and superior frontal cortices; [BA: 6/8]), temporal (BA: 21/22), parietal (BA: 7), occipital (BA: 17/18) cortices, the striatum, the thalamus, and the centrum semiovale (CSO) on the MR images according to the human brain atlas (Mai et al., 1997) (Fig. 1). After completion of placing ROIs on the MR images parallel to the intercommissural plane, PET images were displayed side-by-side together with the MR images using an image processing system (DrView, Asahi Kasei, Tokyo, Japan) (Ouchi et al., 1997, 1999) on a SUN workstation (Hypersparc ss-20; SUN microsystems, CA, U.S.A.), which enabled placing those ROIs automatically on the same area on both the MR and the corresponding PET images (CMRO2, OEF, CBV, CBFsupine, CBFsitting). In the H215O PET study, to evaluate differences in arterial input functions under each condition (supine and sitting), dose-corrected integral areas calculated as percentage dose per liter from the time of H215O injection to the end of scan were compared between two groups using nonpaired Student's t-test. Regional CBF data in normal group were calculated by averaging bilateral values in each region. Statistical analysis for rCBF change was first performed using two-way analysis of variance (ANOVA) with post hoc Scheffe's F test with respect to type of group (CAO or normal) and type of location, that is, occluded or nonoccluded side of the brain regions. Because no cross-interactions were observed in the ANOVA between the two factors (P = 0.3295), repeated ANOVA was performed to compare rCBF change between CAO and normal groups in each hemisphere separately. As a correction for multiple comparisons was performed in each analysis, P < 0.05 was taken to indicate statistical significance. In addition, a simple linear regression analysis was performed to analyze the relation between metabolic or hemodynamic parameters (CMRO2, OEF, CBF/CBV) and magnitudes of postural rCBF reduction among CAO patients.
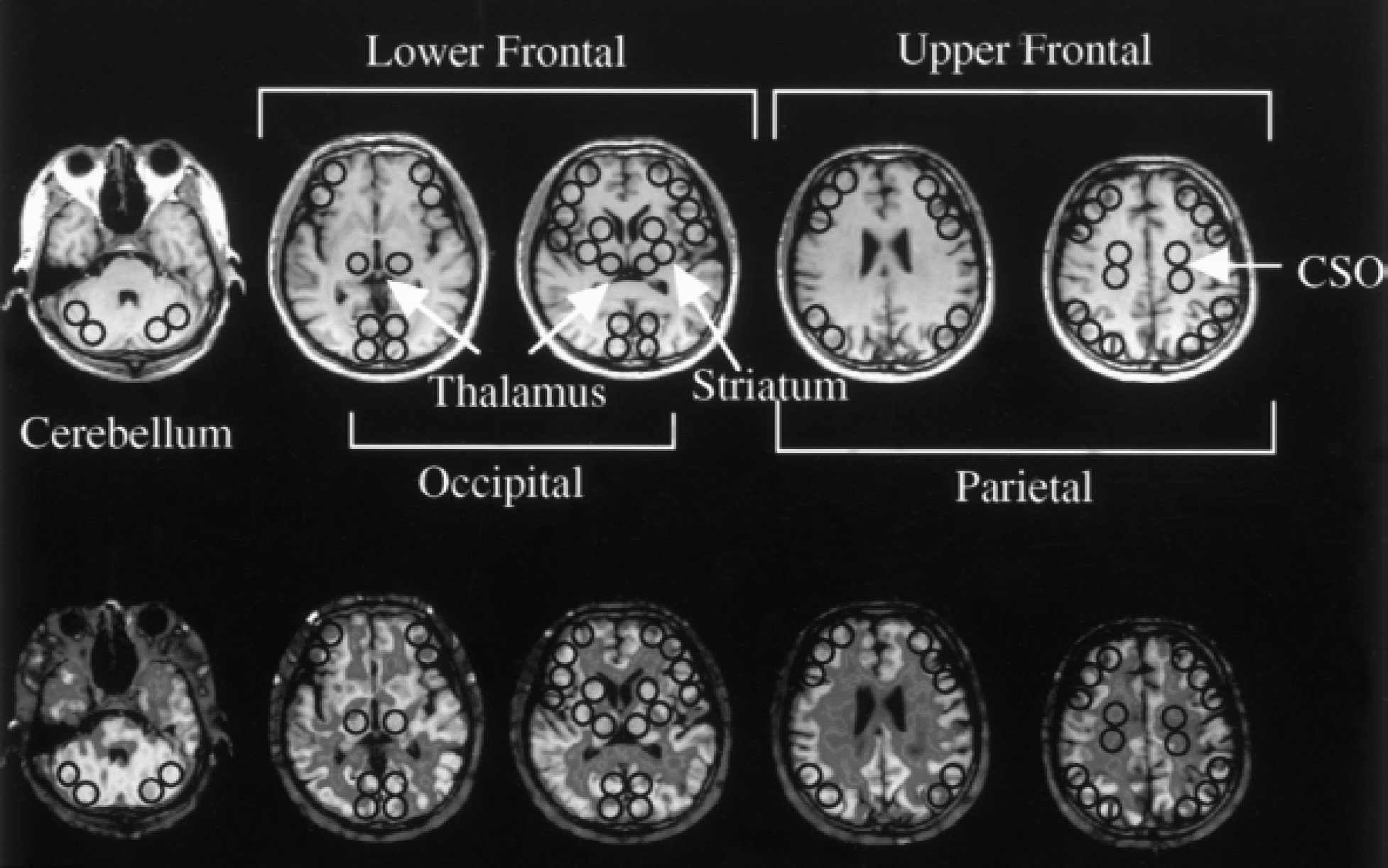
Region of interest (ROI) setting. Multiple circular ROIs consisting of 96 pixels (1.58 cm2) were placed on brain regions on the reformatted magnetic resonance images (top row) with the same pixel size as that of positron emission tomography (PET), which were automatically transferred onto the PET images (bottom row). CSO: centrum semiovale.
Statistical parametric mapping analysis.
To evaluate postural changes in rCBF more objectively, a voxel-based mapping analysis between sitting and supine was performed with statistical parametric mapping (SPM) software (SPM99; Wellcome Department of Cognitive Neurology, London, U.K.) for MCAO patients (n = 10). Before applying SPM, PET images of two patients (nos. 13 and 14) were flipped right to left to rearrange the damaged hemisphere to the same side (that is, the left being the occlusion side). The reason of selecting MCAO patients was no involvement of vascular abnormality in the cortex and similarity of T2 high intensity localization observed on patients' MR images. The effect of variance caused by global CBF was removed by using analysis of covariance with the global flow normalized to 50 mL 100 g−1 min−1, rather than using each quantitative rCBF image applied to the ROI analysis. This process generated normalized mean rCBF values on a voxel-by-voxel basis for each condition. Comparison of adjusted mean rCBF in the sitting condition with that in the supine position was performed on a voxel-by-voxel basis with t-statistics. The result set of voxel values for each contrast constituted a t-statistic SPM t, which was then transformed to a unit normal distribution (SPM z) map (Friston et al., 1995). The authors regarded statistically significant areas as clusters of voxels that had more than 40 voxels and a peak height P < 0.001 without a correction for multiple comparison.
RESULTS
Physiology
Although systolic ABP was significantly greater in CAO group than that in normal group, no significant differences were found in postural changes of mean ABP, pulse rate, Paco2, and arterial pH measured just before commencement of PET scan in the H215O study (P > 0.05, repeated ANOVA) (Table 2). These stable physiologic parameters during the upright condition in both groups were further supported by no differences in the calculated areas integrated under the arterial input between the two conditions. No clinical symptoms such as hyperventilation or reduced consciousness were observed during the upright condition in any of the participants.
Baseline physiologic data in patients with cerebral artery occlusion and normal controls
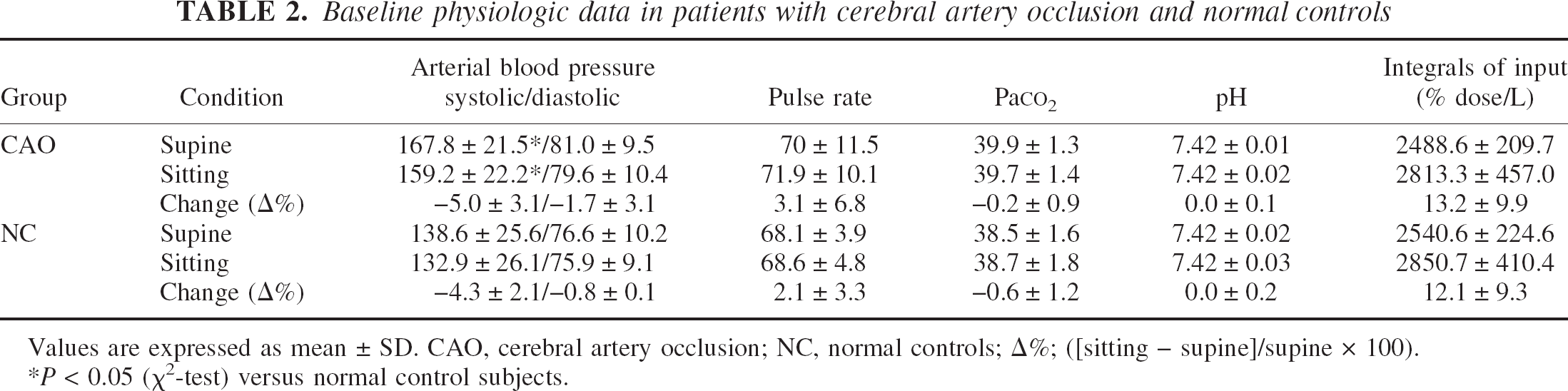
Values are expressed as mean ± SD. CAO, cerebral artery occlusion; NC, normal controls; Δ%; ([sitting – supine]/supine × 100).
P < 0.05 (χ2-test) versus normal control subjects.
Postural changes in rCBF in CAO patients: ROI analysis
Significant rCBF reductions were observed in the upper frontal and parietal cortices and CSO on the occluded side during the sitting position in CAO group, compared not only with data on the nonoccluded side in the same CAO group but also with data in normal group (Table 3, Fig. 2). This was also the case with the magnitude of the autoregulatory index calculated as change in rCBF divided by change in mean ABP (Table 3). The level of rCBF in the occluded-side striatum was significantly less than in the contralateral striatum in CAO group. Age-matched healthy subjects also revealed a mild rCBF reduction in the distal ICA territory (upper frontal area, parietal cortex, CSO).
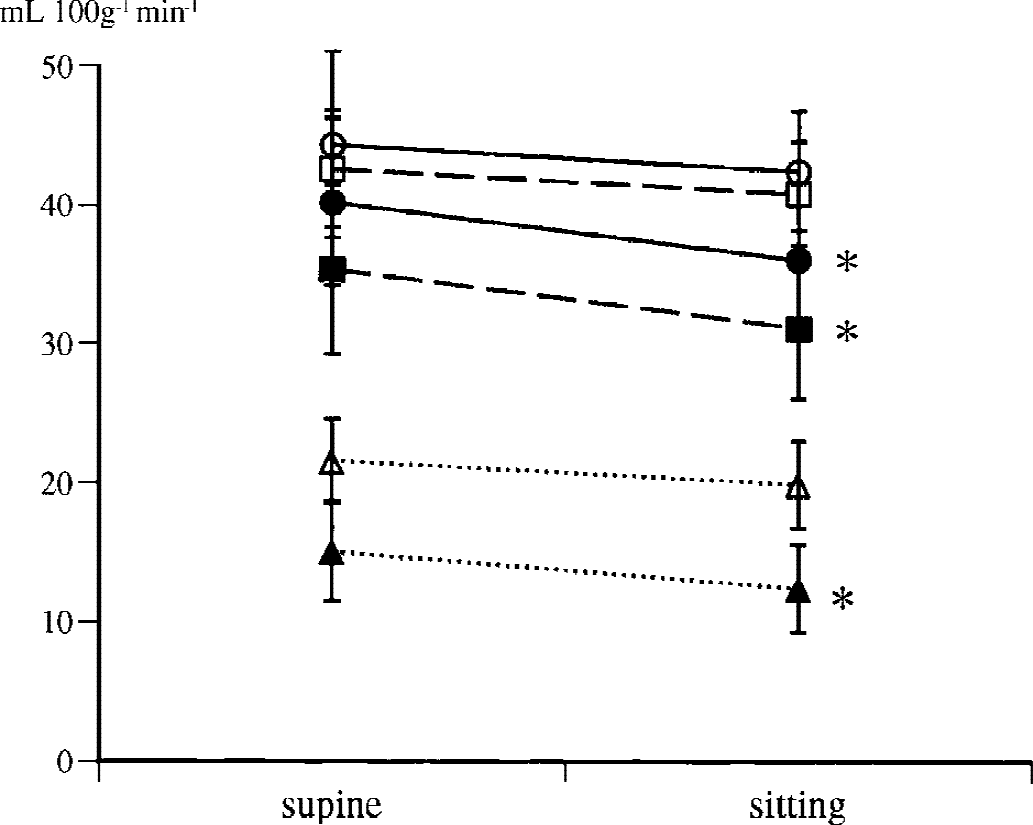
Significant orthostatic reductions in regional cerebral blood flow in the upper frontal area (•), parietal area (▪), and CSO (▴) on the occluded side in cerebral artery occlusion patients compared with those in normal subjects (upper frontal area (○), parietal (□), CSO (▵)). * P < 0.05 vs. normal control.
Hemodynamic parameters and rCBF changes by postural shift in patients with unilateral major cerebral artery occlusion and normal control subjects
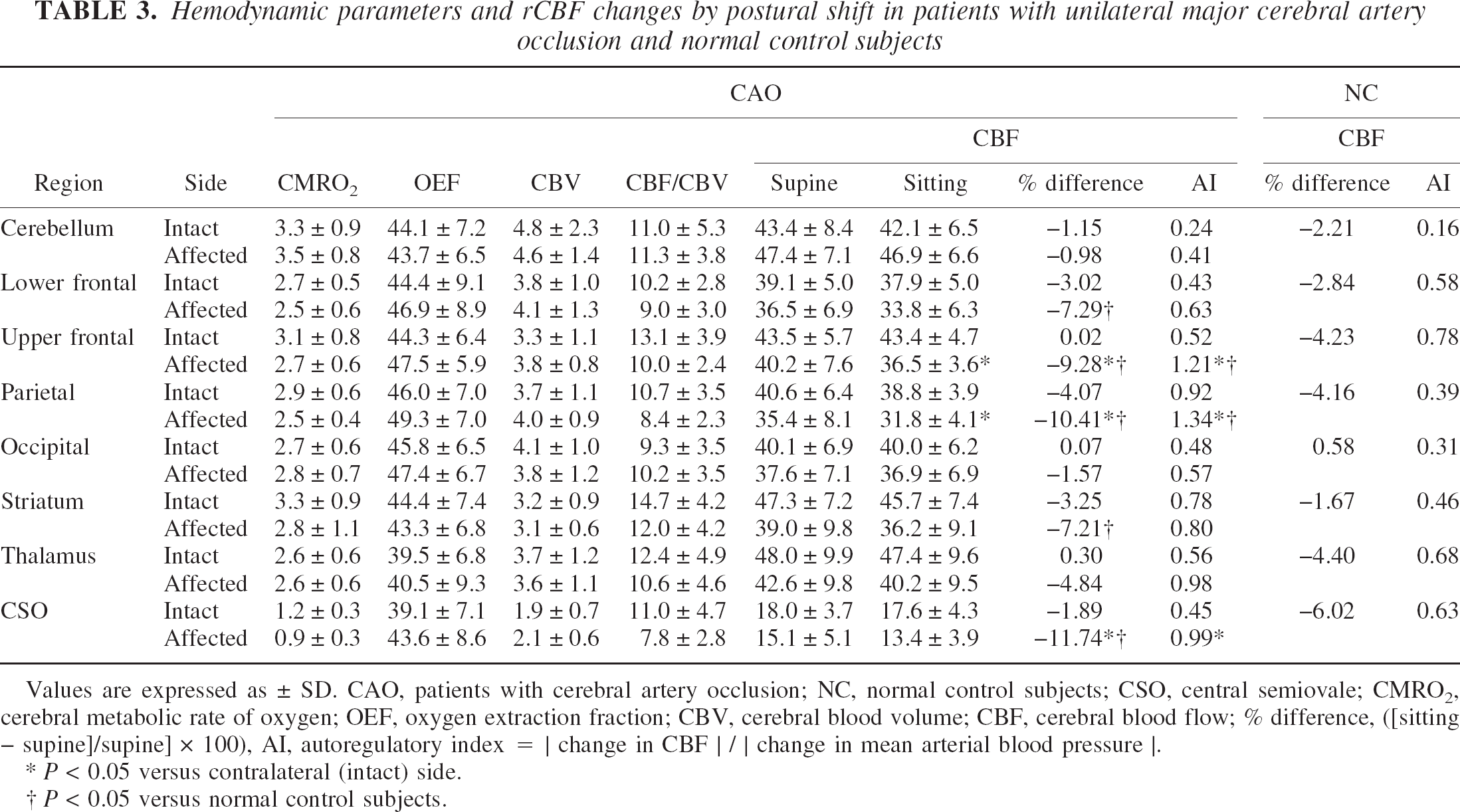
Values are expressed as ± SD. CAO, patients with cerebral artery occlusion; NC, normal control subjects; CSO, central semiovale; CMRO2, cerebral metabolic rate of oxygen; OEF, oxygen extraction fraction; CBV, cerebral blood volume; CBF, cerebral blood flow; % difference, ([sitting − supine]/supine] × 100), AI, autoregulatory index = |change in CBF |/|change in mean arterial blood pressure |.
P < 0.05 versus contralateral (intact) side.
P < 0.05 versus normal control subjects.
Relation between metabolic parameters and magnitude of postural rCBF change in CAO patients: ROI analysis
Although the values did not reach statistical significance, there were consistent tendencies of lower levels of CMRO2 and CBF/CBV (perfusion pressure index) and of moderate elevation in OEF on the occlusion side (Table 3). Regression analyzes showed that magnitudes of postural rCBF reduction were significantly correlated inversely with CMRO2 (Fig. 3A) in the upper frontal area (r = 0.717, P < 0.05, f(x) = −5.88x + 3.22), parietal area (r = 0.703, P < 0.05, f(x) = −4.84x + 3.00), and positively with OEF (Fig. 3B) in the upper frontal area (r = 0.716, P < 0.05, f(x) = 58.41x + 41.33), parietal area (r = 0.775, P < 0.05, f(x) = 86.32x + 39.34) in the CAO group. Levels of perfusion pressure index (CBF/CBV) in three regions (upper frontal area, parietal area, and CSO) tended to be inversely correlated with the magnitudes of rCBF reduction (Fig. 3D).
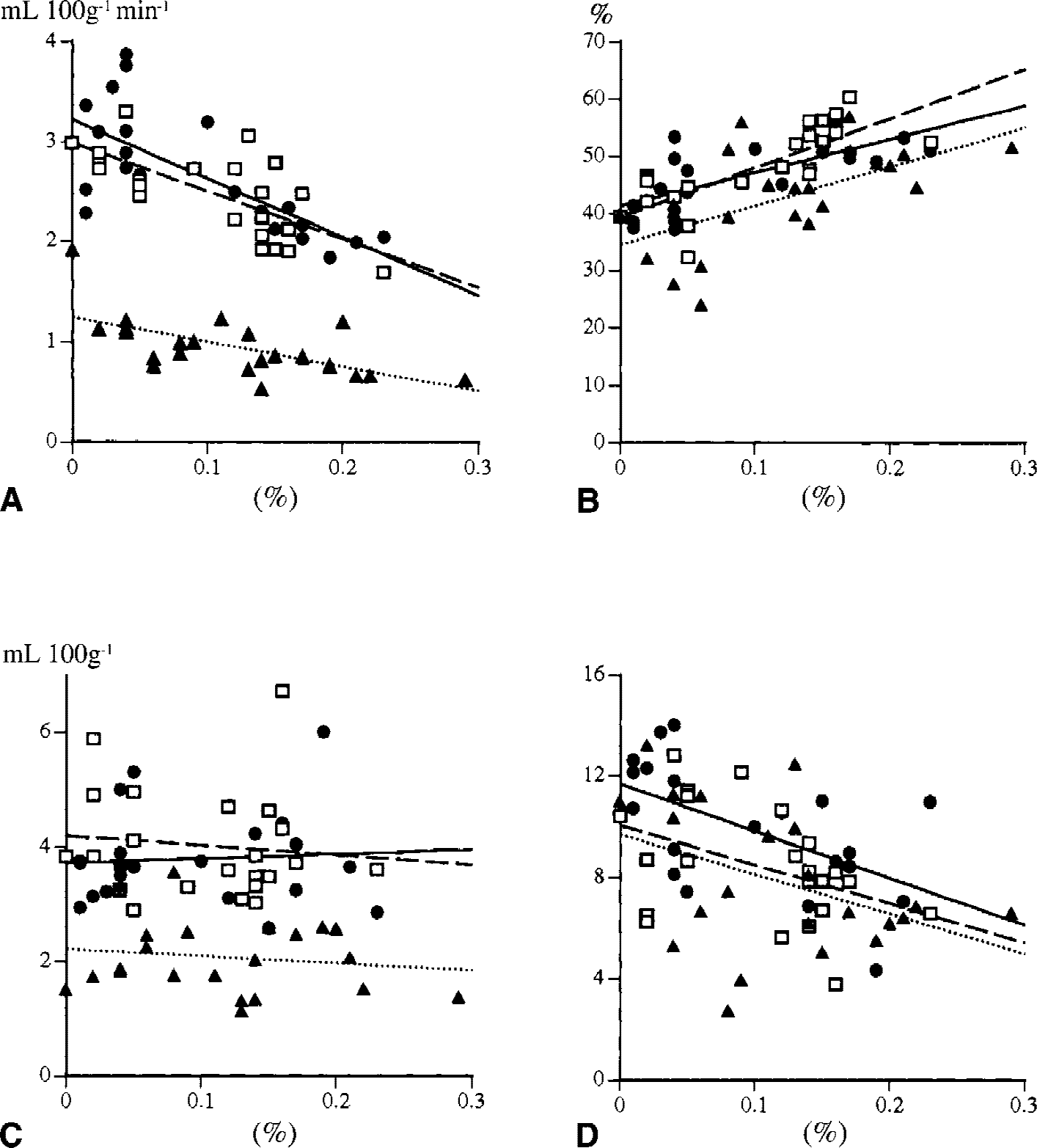
Correlations between metabolic or hemodynamic parameters and magnitudes of regional cerebral blood flow (rCBF) reductions in the distal internal carotid artery regions on the occluded side (upper frontal cortex (•), parietal cortex (□), centrum semiovale (▴)). The magnitudes of rCBF reduction were correlated inversely with levels of cerebral metabolic rate of oxygen
Relative rCBF reduction during the sitting position in MCAO patients: SPM analysis
Comparison of sitting with supine positions showed significant rCBF reduction in the parietal and frontal regions on the occluded side (uncorrected, P < 0.001;Table 4, Fig. 4). Conversely, there was a tendency that sitting posture increased rCBF in the amygdala region, occipital cortex, and cerebellar anterior vermis (uncorrected, P < 0.005; Fig. 5), compared with the supine posture. In contrast to the patient group, SPM results in normal group showed no significant increase and reduction in rCBF in the comparison of sitting with supine conditions (results not shown).
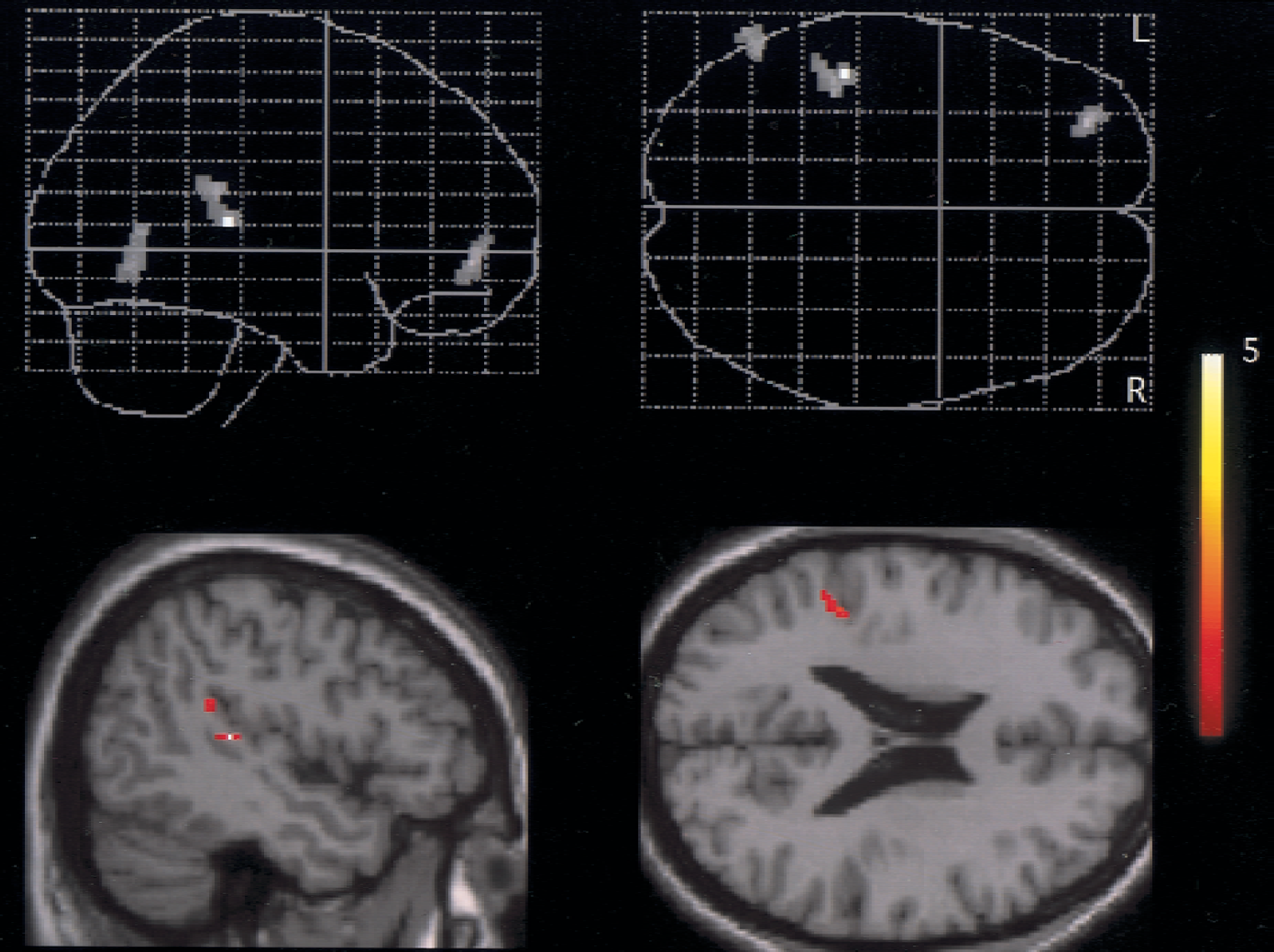
Significant reduction in relative regional cerebral blood flow was found in the middle cerebral artery regions on the occlusion side during sitting versus lying down (top: glass brain, bottom: watershed area superimposed on magnetic resonance imaging). Details of coordinates and Z-values are given in Table 4.
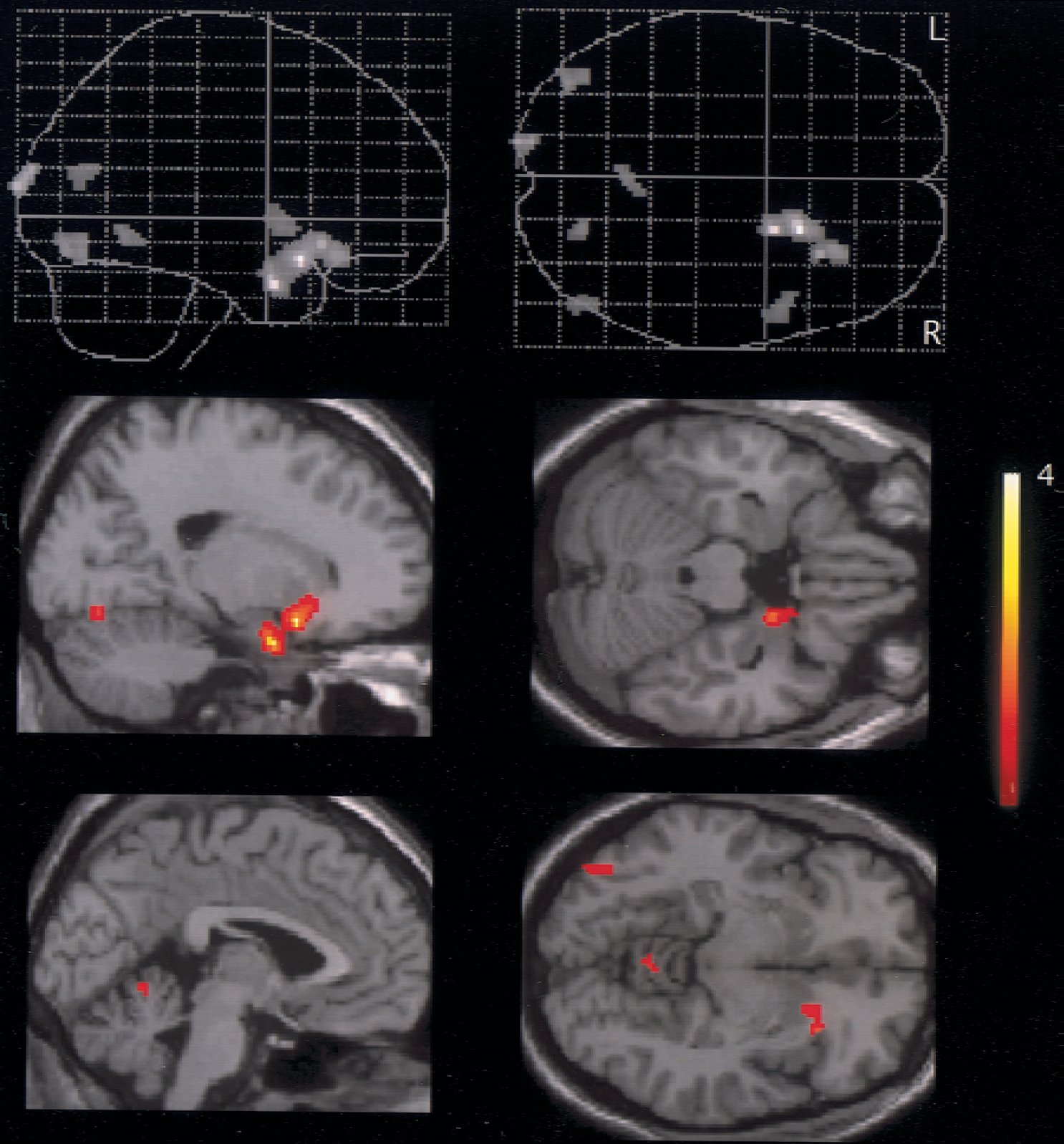
During sitting, regional cerebral blood flow tended to increase in the amygdala, visual cortex, and cerebellar anterior vermis (uncorrected, P < 0.005).
Coordinates and Z scores for significant rCBF change during sitting compared with lying in MCAO patients

rCBF, regional cerebral blood flow; MCAO, middle cerebral artery occlusion. L, left (occlusion side).
DISCUSSION
This study is the first to investigate absolute changes in local brain perfusion with the assuming of upright posture in hemodynamically compromised human subjects. Assuming of the sitting position along with a slight decline in blood pressure reduced absolute levels of rCBF in the cerebral region distal to the cerebral artery occlusion, supported by the present SPM result showing further reduction in relative rCBF in the ischemic territory by sitting. Presence of positive correlation between regional tissue demand of oxygen and rCBF reduction indicates impairment of local autoregulation in the hemodynamically vulnerable area. Although the value of rCBF measured during a stably seated condition did not decrease to less than 30 mL/100g/min in the cortical gray matter in the current study, there would be a possibility of a more severe reduction in rCBF in those regions during the course of head-up movement. Thus, upright posture per se may be an insidious risk for poststroke patients with a major cerebral artery occlusion at a subacute stage to provoke further ischemic attacks in the vulnerable region.
Before interpreting the current results, careful consideration was given to the methodologic limitations of this study. The current PET study was incapable of measuring rapid hemodynamic changes on a real time basis in response to postural shift, unlike transcranial Doppler studies (Daffertshofer et al., 1991; Novak et al., 1998; Schondorf et al., 1997). However, the Doppler method provides data of blood flow velocity in a major cerebral artery, not of rCBF. A recent orthostatic stress study using transcranial Doppler combined with near-infrared spectroscopy showed that the levels of physiologic parameters including heart rate, systemic blood pressure, and oxy/deoxy hemoglobin concentration plateaued 3 minutes after postural change from the supine to the standing position (Krakow et al., 2000). Because emission scan after head fixation in the sitting condition was performed over 5 minutes after postural change in the current study, the results reflected stable hemodynamics in the orthostatic condition. Second, as discussed in the authors' previous study (Ouchi et al., 1999), it was difficult to predict to what extent participants depended on fixation of the head during sitting, which might contribute to emotional activation resulting in alterations of physiologic parameters and/or CBF levels as probably illustrated in Fig. 5. However, the physiologic data in the current study showed that the basic tone of physiology was almost the same between the different postural conditions within each group. The stability of physiologic data during PET measurement was confirmed by the similarities in the patterns of arterial input functions in each condition.
The mechanism of postural rCBF reduction could be explained by impairment of local autoregulation as demonstrated by an increase in autoregulatory index in hemodynamically compromised regions in the current study. It has been reported that the arteriolar pressure is considered much less than normal and less than the lower limit of autoregulation in the ischemic areas distal to an arterial occlusion (Paulson, 1970; Shima et al., 1983; Symon et al., 1976). Thus, the correlation between OEF and postural CBF change during assuming of the sitting posture in the current subacute stroke CAO patients might indicate the presence of low perfusion pressure–autoregulatory vasodilation relation, in which CBF reduces relative to metabolic demand resulting in OEF elevation. This vascular pressure-flow relation hypothesis also was supported by other findings that perfusion pressure index (CBF/CBV) reduced with decreasing postural rCBF levels. Although the volume of major vessels cannot be affected by the postural condition in humans because orthostatic stress by simulation could not reduce the diameter of MCA (Serrador et al., 2000), rCBV can decrease with severe reductions in local perfusion pressure caused by passive collapse of vessels concurrent with decrease in rCBF. However, no correlation between rCBV and rCBF reduction in the current study did not support the speculation possibly because of large variation in rCBV values.
In the current study, a voxel-based analysis (SPM) confirmed significant postural rCBF reduction in the watershed area in MCA group (Fig. 4). These frontal and parietal areas (the distal ICA territory) were likely to be affected to some extent in normal healthy subjects as shown in Table 3. This observation was supported by previous 133Xe single photon emission computed tomography studies showing a significant reduction in relative rCBF values in the frontal area during 70° head-up tilt under normocapnic conditions (Passant et al., 1993; Warkentin et al., 1992) and by a near-infrared spectroscopy study showing significant reduction in frontal cortical oxygenation in the healthy elderly subjects (Jannet Mehagnoul-Schipper et al., 2000). Stable physiology in normal group in the current study could deny the concept of dysautoregulation as seen in diseases with orthostatic hypotension (Brooks et al., 1989; Meyer et al., 1973; Toyry et al., 1997). The result of correlation analysis in the authors' recent study showing a tendency of age-related reduction in frontal rCBF during standing suggested that vascular resistance, possibly caused by age-related arteriosclerotic changes, might affect the range of autoregulation in the distal ICA region (Ouchi et al., 2000). This hypothesis is supported by the previous report that the autoregulatory responses of arterioles, albeit not cerebral arterioles, was reduced in an axial gradient-dependent manner (Casellas et al., 1997).
The present orthostatic PET study may allow identification of a subgroup of patients with major cerebral artery occlusion who are at risk for subsequent stroke during assuming of the upright posture. Even in the supine condition, the boundary (watershed) area in ICA occlusion has more marked rCBF reduction (Mendelow et al., 1987), and “penumbra” (Astrup, 1982) in ischemic stroke suffers dysautoregulation (Olsen et al., 1983). Therefore, prevention of subsequent ischemic attack from assuming of the upright posture becomes important in acute and subacute stroke patients with major cerebral vessel occlusion.
Footnotes
Acknowledgments:
The authors thank Drs. Masanobu Sakamoto, Satoshi Tanaka, Masaru Honda, and Kenichi Yano for their advice on clinical assessment.
