Abstract
There has been an increasing interest in recent years in the evaluation of the neuronal and glial responses to ischemic insult. Some cytokines, including transforming growth factor-β (TGF-β), that are overexpressed after experimental stroke in rodents are thought to be implicated in the neuronal processes that lead to necrosis. Thus, such cytokines could predict tissue fate after stroke in humans, although data are currently sparse for gyrencephalic species. The current study addressed the expression pattern of TGF-β1 in a nonhuman primate model of middle cerebral artery occlusion. Focal permanent ischemia was induced for 1 or 7 days in 6 baboons and the following investigations were undertaken: cerebral oxygen metabolism (CMRO2) positron emission tomography studies, magnetic resonance imaging, postmortem histology, and reverse transcription-polymerase chain reaction. The aim of the current study was to correlate the expression of TGF-β1 to the underlying metabolic and histologic state of the threatened cerebral parenchyma. The authors evidenced increased TGF-β1 mRNA levels (up to 25-fold) in those regions displaying a moderate (20% to 49%) reduction in CMRO2. The current findings suggest that the greatly enhanced expression of TGF-β1 in the penumbral zones that surround tissue destined to infarction may represent a robust index of potentially salvageable brain. The current investigation, in the nonhuman primate, strengthens the authors' hypothesis, derived from rodent models, that TGF-β1 may be involved in the physiopathology of human stroke.
Hypoxia–ischemia insults to the central nervous system initiate various biologic processes that lead to neuronal and glial death. For therapeutic reasons, increasing interest exists to evaluate and mitigate the suffering of the cerebral parenchyma after an ischemic episode. In the last few years, various cytokines (interleukin-1β, interleukin-6, tumor necrosis factor-α, and transforming growth factor-β), the expression of which is enhanced in the ischemic rodent brain (DeGraba, 1998; Feuerstein et al., 1998), have been suggested to represent potential predictors of neurologic outcome after stroke. Among these cytokines, transforming growth factor-β (TGF-β) has elicited clinical interest, because Krupinski and collaborators (1996) have evidenced an increased immunoreactivity for TGF-β1 in the infarcted and penumbral brain areas of stroke patients.
Transforming growth factor-βs belong to a family of related peptides that play pivotal roles in intercellular communication (Massague, 1990; Bonewald, 1999). Transforming growth factor-β1 is the prototype of three different isoforms in mammalian species (TGF-β1, TGF-β2, and TGF-β3) that share a common set of serine and threonine kinase receptors (TßR-I and TßR-II) to transduce their biologic signal (Massague, 1996; ten Dijke et al., 2000). The cellular responses to these pleiotropic cytokines occur through the ligand-induced heteromeric complex formation of TßR-I and TßR-II as follows: the type II receptor binds TGF-β, then recruits and phosphorylates the type I receptor, allowing the transphosphorylation-mediated activation of this latter. The subsequent activation of the signaling mother against decapentaplegic peptide (Smad) transcription factor cascade thus regulates the transcription of key target genes involved in the control of the cell cycle and extracellular matrix metabolism (Attisano and Wrana, 2000; Massague and Wotton, 2000; Zimmerman and Padgett, 2000).
Transforming growth factor-β has been found to be abundantly expressed in brain biopsies from patients with chronic neurodegenerative or acute neurologic disorders, such as Parkinson's disease, Alzheimer's disease, or stroke (Pratt and McPherson, 1997). Consequently, this growth factor has been characterized as an injury-related peptide. Despite some opposing data, most of the in vitro studies have led to a consensual concept of a neuroprotective activity for TGF-β (Prehn et al., 1993; 1994; Prehn and Miller, 1996; Henrich-Noack et al., 1996), particularly with regard to the excitotoxic necrotic pathway, a major component of ischemic processes (Choi, 1996). In a model of primary murine cortical cultures, the authors have demonstrated previously that TGF-β1 exerts a selective neuroprotective activity against N-methyl-d-aspartate receptor-mediated neuronal death and that this neuroprotective activity is mediated by astrocytes (Buisson et al., 1998; Docagne et al., 1999). As previously reported (Wang et al., 1995; Lehrmann et al., 1998), the authors have found that in the rat brain the transcriptional activity of TGF-β1 is increased after middle cerebral artery occlusion (MCAO) (Ruocco et al., 1999). In the same study, the authors addressed the relevance of this endogenous production of TGF-β in the ischemic brain. The blockade of the biologic activity of TGF-β, induced by the local injection of a soluble TGF-β type II receptor (TGF-β antagonist), markedly exacerbated both excitotoxic and ischemic lesions. Overall, the latter studies suggest that the endogenous synthesis of TGF-β1 may play a key role in the pathophysiology of cerebral ischemia. Accordingly, to further investigate the potential and promising therapeutic interest of the TGF-β pathway, the goal of the current study was to determine the expression pattern of TGF-β1 as a function of the degree of ischemic injury estimated by both anatomo-pathologic and metabolic approaches. The authors thus combined magnetic resonance imagery (MRI), positron emission tomography (PET), histology, and semiquantitative reverse transcription-polymerase chain reaction (RT-PCR) to correlate gene expression with metabolic parameters after focal permanent cerebral ischemia in the nonhuman primate (baboon).
The current findings suggest that the expression of TGF-β1 may represent an endogenous adaptive response of the brain, and thus may be a pertinent index of neuronal stress and ischemic damage. Moreover, the current data strengthen, in the nonhuman primate, the hypothesis derived from rodent models that suggests that TGF-β is a promising therapeutic target for the treatment of cerebral ischemia in humans.
MATERIALS AND METHODS
Surgical procedures
Male baboons (Papio anubis, body weight ≈ 15 kg) were housed in individual cages maintained at 24°C with 50% relative humidity on a 12 hours light/12 hours dark cycle and were fed commercial chow supplemented with fresh fruits and water ad libitum. All experiments were performed according to the French national legislation that governs animal experimentation. The research protocols were approved by the Scientific Council of the authors' institution. A team of external veterinary surgeons was responsible for the animals' health and welfare.
As described previously (Young et al., 1996), the baboons were anesthetized, curarized, and ventilated with 67% nitrous oxide in oxygen under etomidate (0.3 mg/kg · h). Permanent MCAO was performed in six baboons. During surgical interventions, anesthesia was supplemented with isoflurane (0.5% to 1.5%). The transorbital approach to the right MCA was used. After enucleation, a small craniectomy was performed using a saline-cooled surgical drill to expose the MCA. The dura mater was opened and the arachnoid was dissected to allow the placement of two microvascular clips—on the proximal part of the main MCA trunk and on the orbitofrontal branch. The only variability encountered in the anatomic presentation lied in the more or less proximal origin of the orbitofrontal artery. This artery was unique in all cases and bifurcated after a short common trunk into two secondary branches—one lateral following the cortical orbital surface of the frontal lobe, and the other medial, recurrent towards the interhemispheric fissure and presumably giving rise to the lenticulostriate perforating arteries.
In all cases, the two aneurysm clips were placed in the following way: one small clip at the origin of the orbital frontal artery, proximal to both the lateral (cortical) and medial branches; a larger clip on the main MCA trunk (M1), just distal to the origin of the orbito-frontal artery and therefore, proximal to the origin of more lateral division branches (temporal and lateral frontal) of the MCA. Warmed saline was used to reduce the possibility of vascular spasm during these procedures.
Reconstruction of the orbit was performed under aseptic conditions and as follows: the craniectomy was covered by an absorbable collagen coated mesh (Vicryl Collagéne; Ethicon, Neuilly, France) and held in place with tissue adhesive (Histoacryl®; B. Braun Melsungen AG, Melsungen, Germany) applied to the sphenoid; the mesh then was covered with cement (GC Fuji I; GC Corporation, Tokyo, Japan). Thereafter, a silicon prosthesis (14 to 16 mm in diameter) was positioned in the orbit, and 1-cm squares of hemostatic collagen compresses (Panegen® Laboratoires Fournier, Dijon, France) were loosely applied around it. A local antibiotic (Rifocine® Merrell Dow, Levallois-Perret, France) then was applied to the orbit and a tarsorrhaphy was performed. These procedures allowed for a complete postoperative recovery in all baboons.
Physiologic (arterial pressure, heart rate, temperature) and biochemical (Paco2, Pao2, pH, hematocrit, hemoglobin, arterial glucose levels) parameters were monitored to maintain optimal conditions during and after the occlusion.
Positron emission tomography procedures
Approximately two weeks before the occlusion, a control MRI (T2) analysis was performed, as described previously (Young et al., 1996). Thereafter, cerebral oxygen metabolism (CMRO2) was measured during a baseline PET session with a 64-slice Siemens HR+ PET Camera (Siemens, Germany); these measurements were further repeated after occlusion (MCAO + 24 hours or MCAO + 7 days). To obtain reproducible head positioning (both inter-and intraanimal) the baboon's head was fixed in a specially designed frame with ear bars being placed in the bony portion of the external auditory canal and checked by a laser beam. Using the 15O2 steady-state technique (with measured attenuation correction), parametric imaging of CMRO2 corrected for blood volume was determined (Frackowiak et al., 1980; Sette et al., 1989).
After 24 hours (n = 3) or 7 days (n = 3), the baboons were deeply anesthetized with 2% to 3% isoflurane, curarized, and ventilated. Heparin (5000 IU) was administered intravenously. The baboons were placed in a supine position, the thorax was opened through a midline incision, and a cannula was inserted into the ascending aorta through the left ventricle. After incising the right atrium and clamping the descending aorta, heparinized saline was perfused at the baboon's mean arterial pressure until the perfusate from the right atrium was free of formed elements. After decapitation, brain tissues were harvested for both histologic and expression analyzes.
Histologic analysis
After euthanasia, brains were cut into 8 large sagittal slices (1 cm). Slices containing hypometabolic areas then were divided into MRI-related cubes (1 cm3). Tissue samples then were rapidly frozen in isopentane at −50°C and stored at −80°C. Approximately one quarter of each cube sample was cut (20 μm) in a microtome and stained by hematoxylin and eosin for standard histology.
Semiquantitative reverse transcriptase-polymerase chain reaction
Total RNA were harvested from each MRI-related cube by a phenol and chloroform extraction method using the RNaxel extraction kit (Eurobio, Paris, France). Samples (1 μg) then were transcribed into cDNA using poly-dT oligonucleotides. An aliquot of the cDNA was amplified using sense (5′-ccgaagcttgccctggacaccaactattgc) and antisense (5′-ccgtctagagctgcacttgcaggagcg) primers for TGF-β1 (Vivien et al., 1998). For the semiquantitative experiments, an aliquot of the same cDNA libraries was amplified by PCR with specific oligonucleotides for β-actin (sense 5′-gtgggccgctctaggcacaa and antisense 5′-ctctttgatgtcacgcacgatttc). β-Actin was chosen as a housekeeping gene because its transcription is not altered in rodents after experimental cerebral ischemia (Ruocco et al., 1999). Each set of oligonucleotides used gave the PCR product of the expected size (363 bps for TGF-β1 and 540 bps for β-actin). The specificity of the PCR products has been previously confirmed by using restriction digest control and sequencing (Vivien et al., 1998). Amplified products were separated by agarose gel electrophoresis and visualized by ethidium bromide staining. Then, PCR products were quantified by optical densitometry and a mean of three independent PCR experiments was calculated for the TGF-β/β-actin ratio for each tissue sample.
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis and Western blotting
Fragments of MRI-related cubes were lysed in Tris-NaCl-Triton buffer and centrifuged for 5 minutes (2500 g) to obtain whole cell extracts. Protein concentration was determined in supernatants, with the aid of the BCA protein assay kit (Pierce, Rockford, Illinois). Then, sodium dodecyl sulfate-polyacrylamide gel electrophoresis was performed on an equal amount of protein (40 μg), in the absence of reducing agents, before immobilization on a polyvinyl difluoride membrane. Western blotting was applied for 1 hour to the primary antibody (rabbit anti-TGF-β1; kindly provided by Dr. P. ten Dijke; dilution, 1:500), 1 hour to the appropriate secondary biotin-conjugated antibody (dilution, 1:1000), and for 1 hour to Extravidin (1:2000) before revelation using a chemiluminescence kit (NEN, Paris, France).
RESULTS
Twenty-four hours after recovery from MCAO, baboons exhibited variable neurologic deficits (stupor and deviation of the eye and head toward the ipsilateral side) associated with a contralateral hemiparesis. In those animals in which survival was permitted for one week, a near complete restoration of neurologic function was observed.
During all surgical procedures and PET sessions, physiologic parameters were monitored and controlled (Table 1). For each baboon, a control PET session was performed approximately two weeks before the middle cerebral artery occlusion (pre-MCAO CMRO2, Fig. 1 and 1D) and 24 hours (n = 3) or 7 days (n = 3) after ischemia (post-MCAO CMRO2, Fig. 1B and 1E). CMRO2 images presented in Fig. 1 are representative images obtained from animals that displayed an estimated infarct volume representative of that previously obtained in larger series (Touzani et al., 1995). The transposition of the ΔCMRO2 values (ΔCMRO2 = post-MCAO CMRO2 − pre-MCAO CMRO2) onto the corresponding MRI illustrates the increase in the area of severely hypometabolic tissues (<50% of contralateral mirror-image values) between day 1 and 7 after MCAO (Fig. 1C and 1F, respectively) that has been formerly described in the authors' laboratory (Touzani et al., 1995, 1997). Based on the literature (Touzani et al., 1997), the authors have defined 3 categories of brain tissue as a function of their residual CMRO2 normalized to their contralateral counterpart. Accordingly, regions with a large (>50%), moderate (20% to 49%), or slight (<20%) reduction in CMRO2 represented infarcted, ‘penumbral’ border zone, and healthy areas, respectively. The choice of these ‘metabolic thresholds’ was then further validated by histologic analysis based on the microscopic examination of hematoxylin and eosin–stained tissue sections. Tissue samples isolated from regions having a severe reduction of rCMRO2 exhibited a large eosinophilic area, with a general loss of cellular identity, a homogenous distribution of irreversible ischemic neurons (ghost neurons), and the absence of surviving parenchymal cells. This histologic pattern was consistent with necrotic tissue (Fig. 2C). This infarct core was separated from the areas of normal metabolism and histology (healthy tissue; Fig. 2A) by an intermediate metabolic area (called ‘border zone’) in which reversibly damaged neurons seem predominant (Fig. 2B). Based on the above criteria, the reduced CMRO2 was then binned and the histologic status of the brain (necrotic, border zone, or healthy) was analyzed as a function of the decrease in CMRO2 (Fig. 3A and 3B) at both 1 and 7 days after MCAO. These results are in agreement with those presented above in PET images (Fig. 1) showing an increase in the proportion of severely hypometabolic and necrotic tissue between 1 and 7 days after MCAO. These data also provide evidence that 24 hours after the onset of the ischemia, the hypometabolic tissue can be characterized as a penumbral and border zone region with essentially swollen but not dead neurons and only a few necrotic areas.
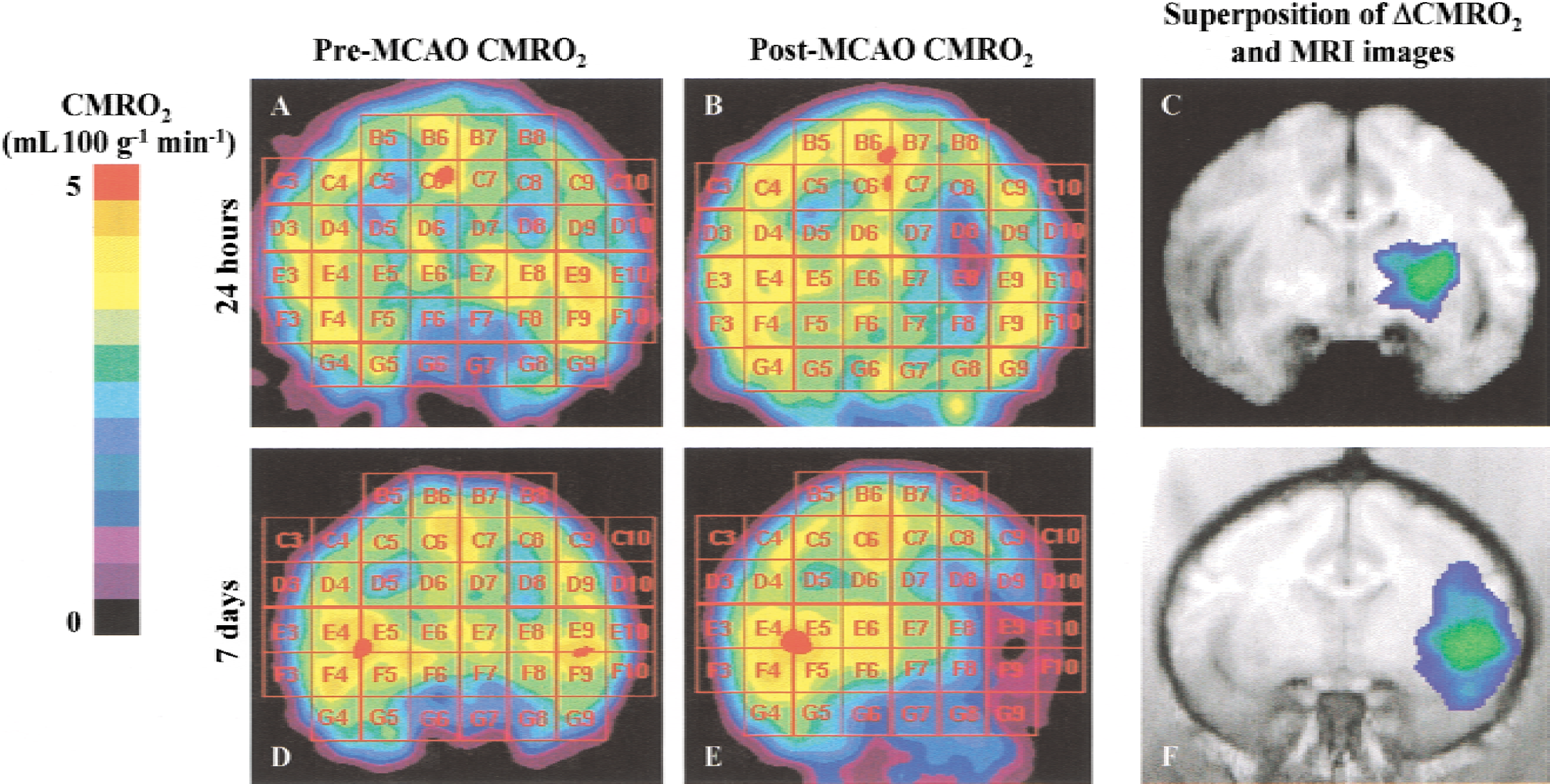
Parametric positron emission tomography (PET) images of cerebral oxygen metabolism (CMRO2) obtained from a single plane in one anesthetized baboon from each group, that is, before
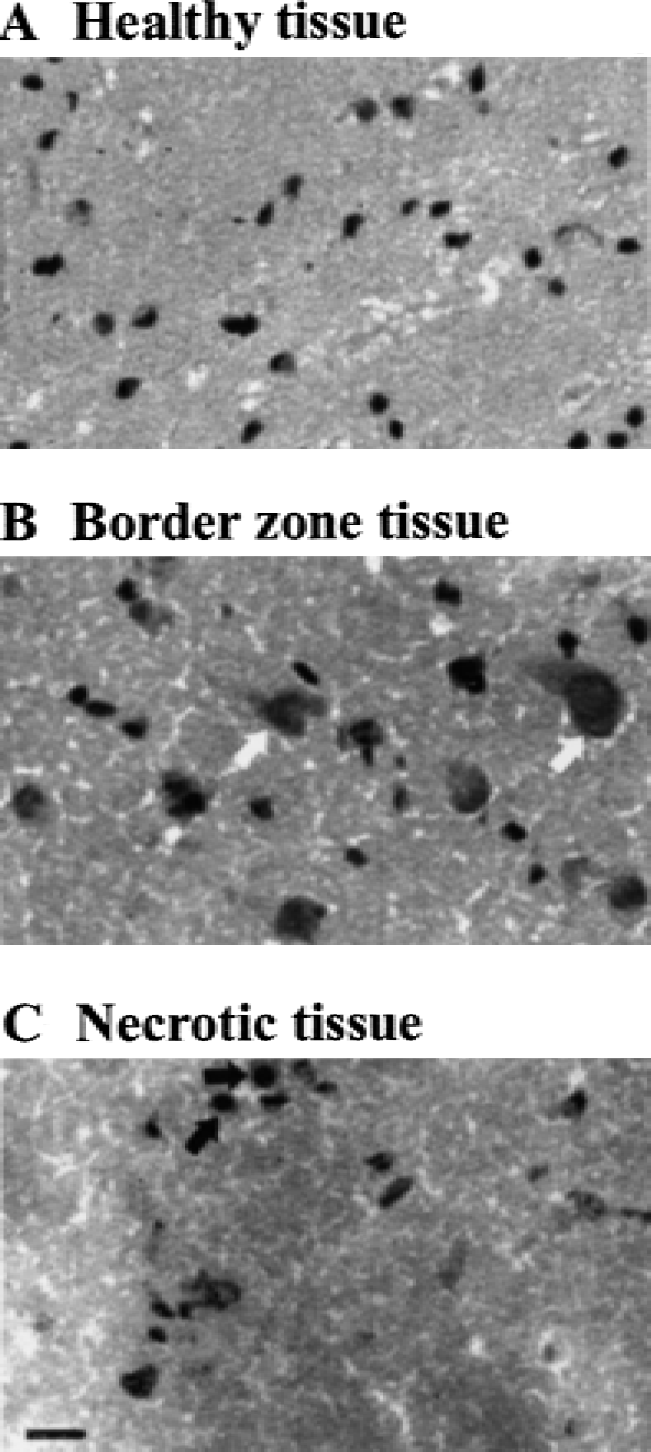
Representative histologic sections showing healthy
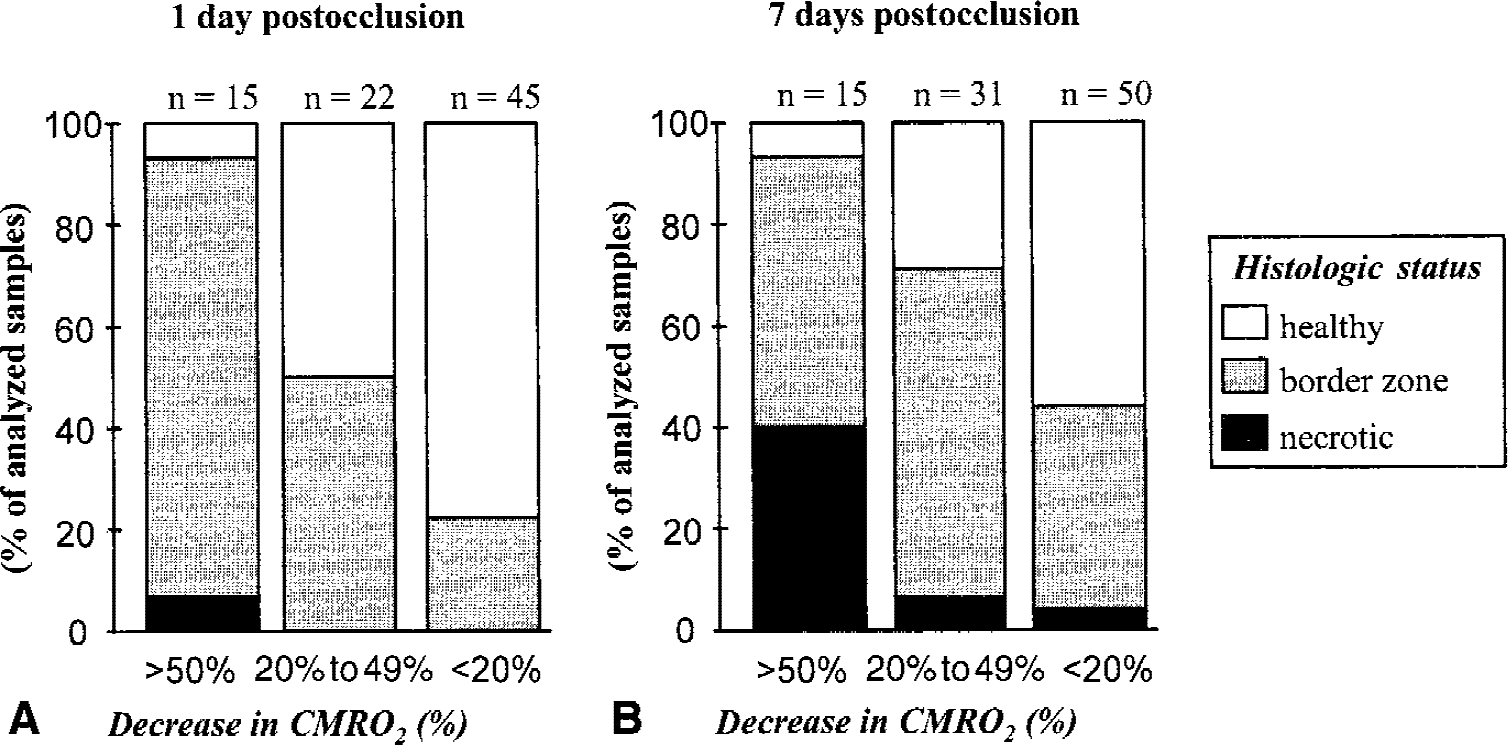
Evaluation of the histologic state of the cerebral parenchyma, as a function of the ischemia-induced hypometabolism. For each metabolic threshold (>50%, 20% to 49%, and <20% of reduction of the cerebral oxygen metabolism (CMRO2)) at 1 day
Biochemical and cardiovascular parameters measured approximately 2 weeks before (pre-MCAO) and 24 hours or 7 days after permanent ischemia (post-MCAO)
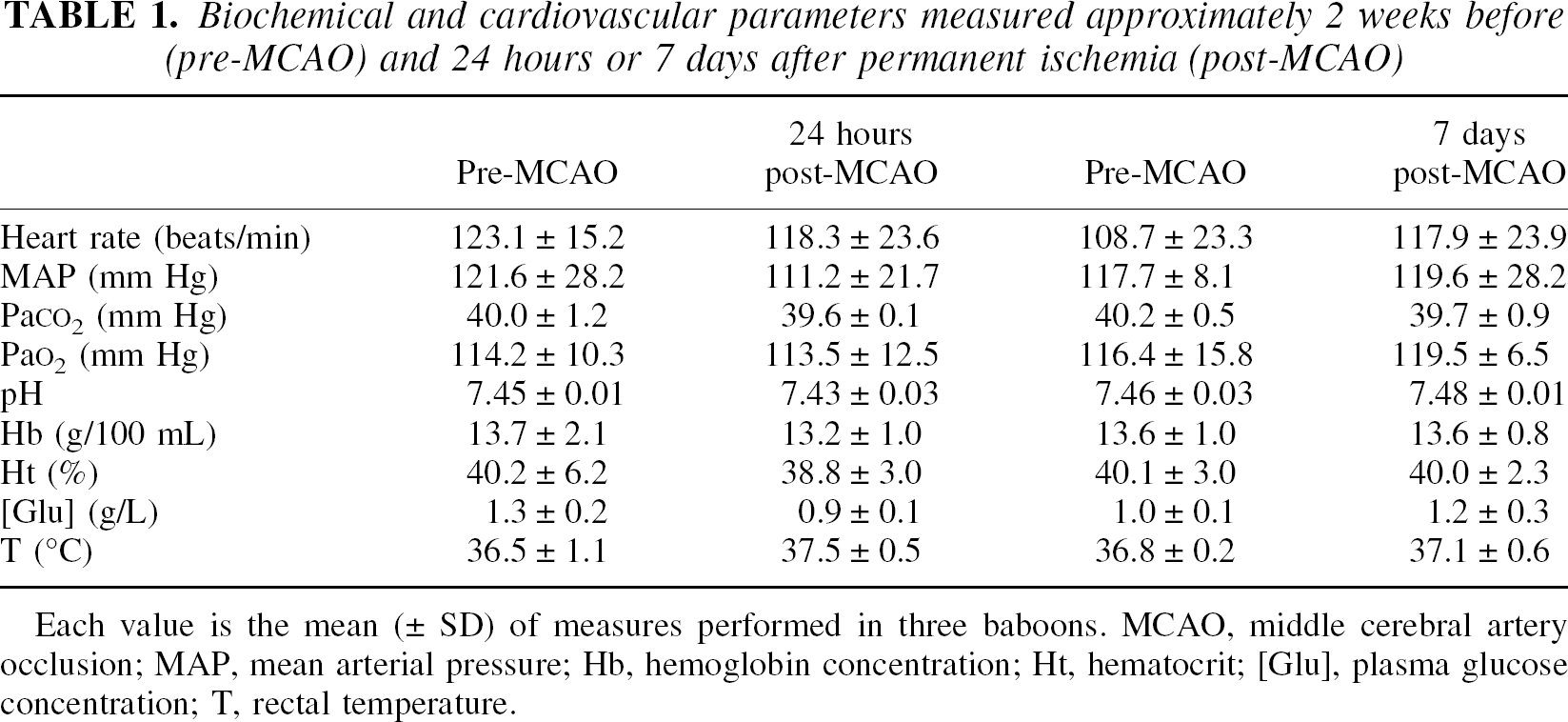
Each value is the mean (± SD) of measures performed in three baboons. MCAO, middle cerebral artery occlusion; MAP, mean arterial pressure; Hb, hemoglobin concentration; Ht, hematocrit; [Glu], plasma glucose concentration; T, rectal temperature.
Representative baboons—2 baboons at 24 hours and 1 baboon at 7 days postocclusion—were selected to construct a map of the expression of TGF-β1 mRNA after focal permanent ischemia (Touzani et al., 1997). The two slices that encompassed the majority of hypometabolic and ischemic cubes (as determined by the CMRO2 images;Fig. 1) were selected for the ensuing analysis. The extraction of total RNA was performed for each. Reverse transcription using poly-dT oligonucleotides (specific of poly-A+ mRNA) allowed the constitution of cDNA libraries that were then analyzed by PCR with specific oligonucleotides for β-actin or TGF-β1. An illustration of agarose gel electrophoresis obtained for the expression of TGF-β1 and β-actin mRNA in healthy, border zone and necrotic tissues of the lesioned hemisphere is given in Fig. 4. Contralateral tissue samples were taken from the mirror-image structures. The mean ratio of the optical intensity for the expression of TGF-β1 and β-actin mRNA from three different PCR experiments was calculated for each tissue sample analyzed. Then, the mean of each TGF-β/β-actin ratio for all the tissue samples analyzed were plotted as a function of the decrease in CMRO2 at both 1 and 7 days after MCAO (Fig. 5A and 5B, respectively). The authors, thus, were able to correlate the expression of TGF-β1 with the degree of ischemia-induced hypometabolism, as well as the probable tissue outcome. Semiquantitative RT-PCR analysis failed to reveal any modification in the expression of TGF-β1 in the nonlesioned contralateral hemisphere (data not shown). In contrast, the current data show in the moderately hypometabolic tissues (reduction of CMRO2 between 20% to 49%), an increased transcription of TGF-β1 one day post-occlusion (2.8-fold when compared with healthy tissue), that was further enhanced (24.3-fold) 7 days after occlusion. TGF-β1 expression thus was markedly elevated in regions in which the probability of ischemic or necrotic histology was great. This increased transcription of the gene encoding for TGF-β1 was accompanied by an increased level of TGF-β1 protein in moderately and severely hypometabolic tissues isolated in the ischemic side, as compared with contralateral regions (Fig. 6).
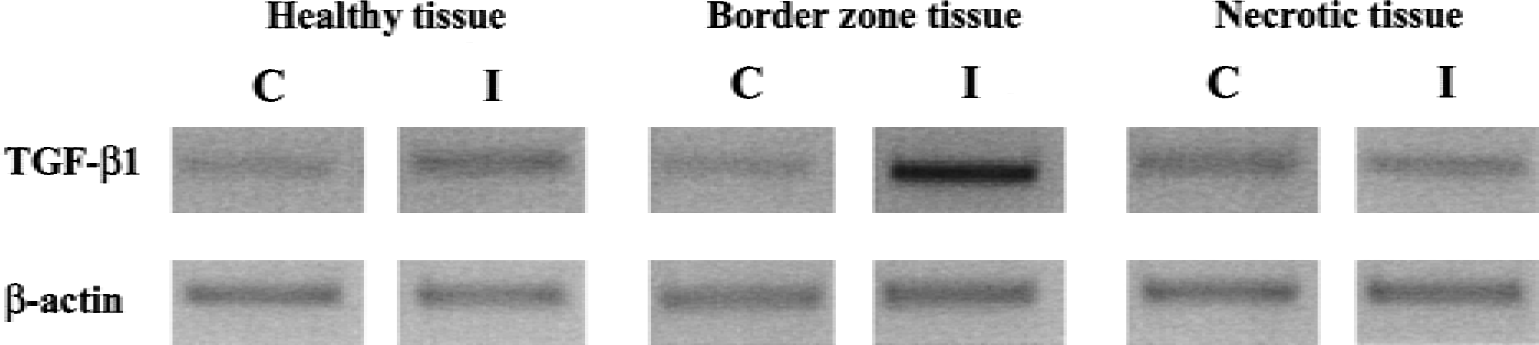
Representative agarose gel electrophoresis obtained in healthy, border zone, and necrotic tissues submitted to reverse transcription-polymerase chain reaction (RT-PCR) analysis of transforming growth factor-β1 (TGF-β1) and β-actin mRNA expression. Total RNA from healthy, border zone, and necrotic tissues of the lesioned hemisphere, ipsilateral (I) and contralateral (C) to the occluded middle cerebral artery, were isolated and analyzed by RT-PCR for TGF-β1 and β-actin mRNA expression, as described in the Materials and Methods.
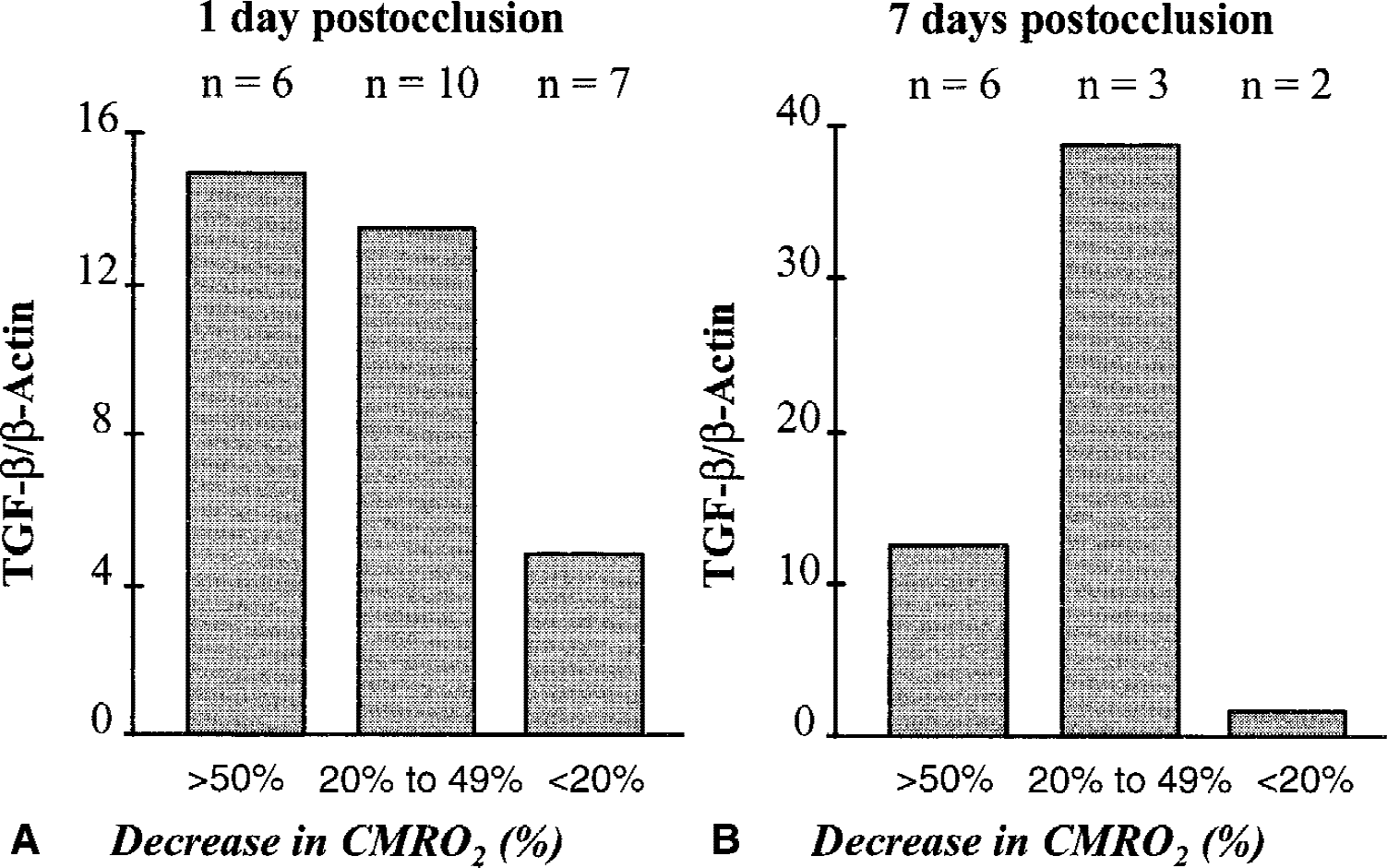
Semiquantitative analysis of the modulation of the cerebral expression of transforming growth factor-β1 (TGF-β1) mRNA as a function of the severity of hypometabolism induced by focal permanent ischemia in the baboon. Total RNA from brain tissue samples (magnetic resonance imaging-related cubes) were isolated at the time indicated after middle cerebral artery occlusion and analyzed by reverse transcription polymerase chain reaction (RT-PCR) for TGF-β1 and β-actin mRNA expression. Two individual animals were used at 1 day
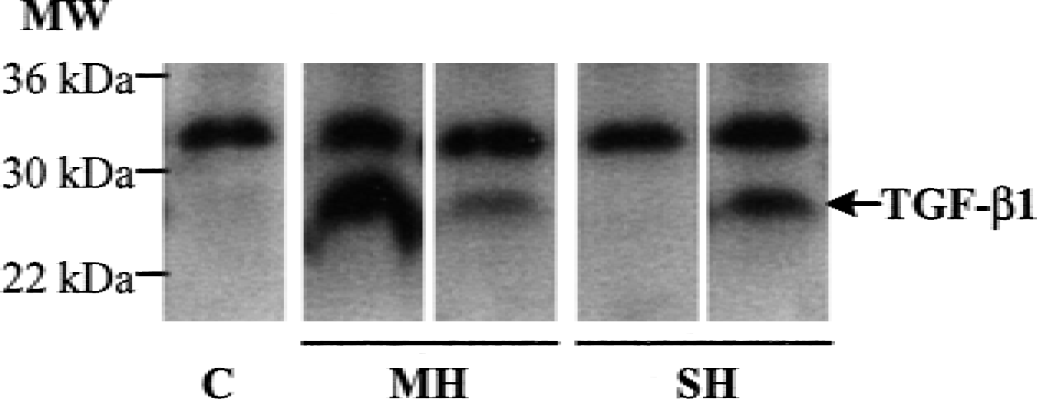
Increased expression of transforming growth factor-β1 (TGF-β1) in hypometabolic tissues induced by focal permanent ischemia in the baboon, as evidenced by Western blotting performed from contralateral (C), moderately hypometabolic (MH), or severely hypometabolic (SH) tissues, revealed with an antibody raised against TGF-β1. MW, molecular weight.
Taken together, these data provide for the first time a partial brain mapping of the expression of the previously characterized injury-related neuroprotective peptide, TGF-β1, in relation to the ischemia-induced disturbances of oxygen metabolism.
DISCUSSION
The noninvasive technology of PET has been developed to accurately evaluate and map regional hemodynamic and metabolic parameters. Positron emission tomography imaging thus represents a powerful tool to improve the pathophysiologic diagnosis of cerebrovascular disorders, such as stroke (Ueda et al., 1999). It is generally admitted that the metabolism of oxygen reflects an oxygen supply adapted to the metabolic needs of the tissue (Baron et al., 1984). The PET oxygen-15 steady-state method, which, in a single PET session, measures serially and at the regional level, cerebral blood flow, cerebral blood volume, oxygen extraction faction, and CMRO2, thus allows to delineate cerebral regions (ischemic core, penumbra, or healthy areas) as a function of the degree of hypoperfusion and hypometabolism. In the current study, the authors have thus attempted to correlate the extent of metabolic changes with the expression of the neuroprotective cytokine, TGF-β1. As such, the authors combined PET imaging and biomolecular and cellular technologies in the baboon brain 24 hours and 7 days after the onset of focal permanent ischemia.
The first finding of the current study suggests that it is possible to correlate the gene expression of the injury-related cytokine, TGF-β1, with the metabolic and histologic tissue outcome after an ischemic insult in the brain. Moreover, the current study supports the authors' previous reports derived from rodent models, which show that the endogenous production of TGF-β1 is a pivotal indicator of the extent of ischemic brain damage (Ruocco et al., 1999). To the authors' knowledge, no previous study in the nonhuman primate has evaluated, in a reliable model of focal ischemia, the metabolic and histologic parameters in comparison with the expression of a gene thought to play a role in infarct progression.
Various studies have related an increased expression of TGF-β1 in brain tissues after different types of acute brain injury, including cerebral ischemia, for which most of the available data have been obtained from rodent models (Wang et al., 1995; Knuckey et al., 1996; Lehrmann et al., 1998). In the current study, the authors confirmed that TGF-β1 is overexpressed after cerebral ischemia in the baboon. Previous studies conducted in rodent models of focal ischemia have suggested that the synthesis of TGF-β may originate from resident (astrocytes, microglia, neurons, and endothelial cells) and infiltrated (macrophages) cells (Lehrmann et al., 1995; Ata et al., 1999). A similar cellular pattern has been reported in humans after stroke (Krupinski et al., 1996). In addition, the current results evidenced that this induction of TGF-β1 is essentially located in those zones that display a moderate hypometabolism (as shown by a decrease in the CMRO2 ranging from 20% to 49%) and an histologic pattern mainly exhibiting neurons that displayed early ischemic damage with nevertheless some irreversibly damaged ischemic cells. This current observation is agrees with a previous report published by Krupinski and collaborators (1996), which demonstrated an increased immunoreactivity for TGF-β1 in the infarcted and penumbral brain areas after ischemic stroke in humans. However, in that study, the authors did not correlate this production of protein with either metabolic or histologic analyses.
Transforming growth factor-β1, endogenously produced by the cerebral parenchyma after ischemic injury, has been postulated to be of critical importance, but its role after brain damage remains to be clearly established. In vivo, the intracerebroventricular application of TGF-β decreases infarction volume after MCAO in the mouse (Prehn et al., 1993). The authors have recently assessed the relevance of the endogenous synthesis of TGF-β1 by the blockade of its biologic activity achieved by the injection of a soluble TGF-β type II receptor (Ruocco et al., 1999). Through this strategy, the authors found that TGF-β1 limits the extent of ischemic or excitotoxic lesions in the rat. Overall, the current data demonstrate that TGF-β1 exhibits an endogenous neuroprotective activity against ischemic brain insults. Nevertheless, the molecular and cellular mechanisms of TGF-β1 involvement during neurologic disorders such as stroke have not yet been fully resolved. Among the various possibilities, it has been suggested that TGF-β may exert neurotrophic effects (Blottner et al., 1996; Iwasaki et al., 1997; Krieglstein et al., 1998), display neuroprotective activity against excitotoxic paradigms (Prehn et al., 1993; Buisson et al., 1998; Ruocco et al., 1999; Ho et al., 2000), modulate astrocytic reactivity (Lindholm et al., 1992; Rich et al., 1999), regulate extracellular matrix metabolism (McCaffrey et al., 1995; Basile et al., 1998), and control thrombolysis (Fujii and Sobel, 1992; Thomas and Thibodaux, 1992).
Given the importance of the excitotoxic cascade in the development of cerebral ischemia, antiglutamatergic strategies were proposed as the first rational treatment of stroke; these therapeutic approaches have been unsuccessful largely because of secondary effects (De Keyser et al., 1999). Currently, the sole approved clinical therapy for stroke patients is in the administration of the thrombolytic agent, t-PA (tissue-type plasminogen activator). However, there is an ever-present interest in the development of alternative strategies that are primarily directed toward the modulation of the endogenous pathways involved in ischemic processes. One such strategy would be aimed to potentiate endogenous neuroprotective cytokines, such as TGF-β1. The approach used in the current investigation thus may represent a powerful tool in understanding the mechanisms of action of TGF-β1 during cerebral ischemia and to validate the authors' hypothesis that TGF-β1 may be a target for the development of therapeutic strategies against human stroke.
Footnotes
Acknowledgments:
The authors thank all of the members of the Cyclotron Unit at Cyceron for their technical expertise and Dr. P. Karamian for his kind and helpful comments.
