Abstract
During development, neuropilin-1 is a receptor for semaphorin 3a-mediated axonal guidance and for vascular endothelial growth factor (VEGF) promotion of angiogenesis. The authors measured neuropilin-1 expression in the adult ischemic brain using Northern blot, in situ hybridization, and immunohistochemistry. Neuropilin-1 mRNA was significantly up-regulated as early as 2 hours and persisted at least 28 days after focal cerebral ischemia. Acute up-regulation of neuropilin-1 mRNA primarily localized to the ischemic neurons. A marked increase in both mRNA and protein of neuropilin-1 was detected in endothelial cells of cerebral blood vessels at the border and in the core of the ischemic lesion 7 days after ischemia, and neuropilin-1 gene expression persisted on these vessels for at least 28 days after ischemia. In these areas, neovascularization was detected using three-dimensional reconstructed images obtained from laser scanning confocal microscopy. Activated astrocytes also exhibited neuropilin-1 immunoreactivity during 7 to 28 days of ischemia. Double immunofluorescent staining showed colocalization of neuropilin-1 and VEGF to cerebral blood vessels and activated astrocytes. These data suggest that in addition to its role in axonal growth, up-regulation of neuropilin-1, in concert with VEGF and its receptors, may contribute to neovascular formation in the adult ischemic brain.
Neuropilin-1 is a transmembrane protein that binds to semaphorin 3a (He and Tessier-Lavigne, 1997; Kitsukawa et al., 1997). In vitro, semaphorin 3a causes collapse or repulsion of embryonic sensory ganglion growth cones, whereas antibodies against neuropilin-1 block the ability of semaphorin 3a to repel sensory axons and to induce collapse of their growth cones (Bagnard et al., 1998; Pasterkamp et al., 1999a). Target inaction of semaphorin 3a or neuropilin-1 gene in mice results in extensive inappropriate sprouting and defasciculation of nerve fibers during development (Kitsukawa et al., 1997). These data suggest that neuropilin-1 mediates axonal growth and guidance during development.
Neuropilin-1 is also a coreceptor for vascular endothelial growth factor 165 isoform (VEGF165) (Soker et al., 1998). Coexpression of neuropilin-1 with VEGF receptor 2 (KDR) in transfected cells enhances VEGF165 binding to KDR and KDR-mediated mitogenic and chemotactic activities of VEGF (Soker et al., 1998). Mice with overexpression of neuropilin-1 exhibit excess production of blood vessels and malformed hearts; neuropilin-1 mutant mouse embryos have vascular defects, including impairment in neural vascularization. These data indicate that neuropilin-1 plays an important role in vascular development during embryogenesis (Kawasaki et al., 1999; Kitsukawa et al., 1995).
Increases in vascular density and angiogenesis have been shown in ischemic brain of both human and experimental animals (Krupinski et al., 1993, 1994; Liu and Chen, 1994; Sbarbati et al., 1996). Transient and permanent middle cerebral artery occlusion in rat evokes expression of VEGF and its receptor Flt-1 and KDR in ischemic brain (Hayashi et al., 1997; Kovacs et al., 1996; Lennmyr et al., 1998), suggesting that after stroke VEGF may be involved in angiogenesis. The authors recently showed that administration of VEGF165 2 days after embolic stroke in rats enhances angiogenesis in ischemic brain and improves functional neurologic recovery (Zhang et al., 2000). Further, regeneration of neurites has been observed in ischemic brain (Stroemer et al., 1998).
Neuropilin-1 is expressed on neurons and endothelial cells (Kawakami et al., 1996). Because injured brain has characteristics of developmental brain (Cramer and Chopp, 2000) and neuropilin-1 guides nerve fibers and promotes vascular formation during development, the authors hypothesize that neuropilin-1 may contribute to the regulation of these processes after stroke. Therefore, in this study, the authors measured temporal and spatial profiles of neuropilin-1 expression after focal cerebral ischemia in rats.
MATERIALS AND METHODS
All experimental procedures were approved by the Care of Experimental Animals Committee of Henry Ford Hospital.
Animal model
Male Wistar rats (n = 50) weighing 300 to 350 g were anesthetized with halothane (1% to 3.5% in a mixture of 70% N2O and 30% O2) using a facemask. The middle cerebral artery was occluded by placing an embolus at its origin (Zhang et al., 1997). Briefly, a single intact fibrin-rich 24-hour-old homologous clot (approximately 1 μL) was placed at the origin of the middle cerebral artery using a 15-mm length of modified PE-50 catheter. After occlusion, all rats exhibited neurologic deficits, including turning to left (Zhang et al., 1997). Rats were killed at 2 hours to 28 days after occlusion. Nonoperated rats were used as a control group.
Tissue preparation
Rats were transcardially perfused with heparinized saline and 10% buffered formalin, and brains were removed. Using a rat brain matrix, each brain was cut into 2-mm-thick coronal blocks, for a total of seven blocks per animal. The brain tissue was processed and embedded, and 6-μm-thick paraffin coronal sections from each block were cut and stained with hematoxylin and eosin for histopathologic evaluation. A 6μm-thick paraffin coronal section from the center of the ischemic core (section D; bregma −0.8 mm) (Paxinos and Watson, 1986) was used for in situ hybridization and immunohistochemical staining.
Three-dimensional image acquisition
To examine neovascularization in ischemic brain, fluorescein isothiocyanate (FITC)-dextran (2 × 106 molecular weight; 0.1 mL 50 mg/mL; Sigma, St. Louis, MO, U.S.A.) was administered intravenously to the ischemic rats subjected to 14 days of middle cerebral artery occlusion. FITC-dextran remains dissolved and free in plasma (Zhang et al., 1999). This dye circulated for 1 minute, after which the anesthetized animals were killed by decapitation. Brains were rapidly removed from the severed heads and placed in 4% paraformaldehyde at 4°C for 48 hours. Coronal sections (100 μm) were cut on a vibratome. The vibratome sections were analyzed as previously described (Zhang et al., 1999a, 1999b) with a Bio-Rad MRC 1024 (argon and krypton) laser-scanning confocal imaging system mounted onto a Zeiss microscope (Bio-Rad, Cambridge, MA, U.S.A.).
Immunohistochemistry
A rabbit polyclonal antimouse neuropilin-1 antibody (gift from Dr. Fujisawa, Nagoya University, Japan) (Kawakami et al., 1996) was used at a titer of 1:400 to assess neuropilin-1. A goat polyclonal antibody against VEGF (Santa Cruz Biotechnology, Santa Cruz, CA, U.S.A.) was used at a titer of 1:200 to assess VEGF (Nag et al., 1997). A mouse antirat type IV collagen monoclonal antibody (M3F7, Developmental Studies Hybridoma Bank, University of Iowa) was used at a titer of 1:200 to detect type IV collagen (Foellmer et al., 1983). A rabbit polyclonal antibody against cow glial fibrillary acidic protein (GFAP) (1:400; DAKO, Carpinteria, CA, U.S.A.) was used to identify astrocytes (Zhang et al., 1999a).
Coronal sections were fixed in 4% paraformaldehyde overnight at 4°C and incubated with the antineuropilin-1 antibody for 1 hour at room temperature. The immunoreactivity was visualized with diaminobenzidine. Double immunofluorescence labeling for neuropilin-1 and type IV collagen or neuropilin-1 and VEGF was performed to evaluate neuropilin-1 and cerebral blood vessels, and neuropilin-1 and VEGF, respectively. Coronal sections were incubated with the antibody against neuropilin-1 for 3 days at 4°C, and sections were then incubated with the antirabbit immunoglobulin antibody conjugated to Cy5 (Vector, Burlingame, CA, U.S.A.). These sections were incubated with the antibody against type IV collagen or VEGF for 3 days at 4°C and then with the antimouse immunoglobulin antibody conjugated to FITC or antigoat immunoglobulin antibody conjugated to FITC. Control experiments consisted of staining brain coronal tissue sections as outlined above, but omitting the primary antibodies. Sections stained with single immunofluorescence (type IV collagen or VEGF) were used to compare the staining patterns with those obtained in the double-stained sections. Double-stained sections were analyzed with the laser-scanning confocal imaging system (Bio-Rad). Green and far-red fluorochromes were excited by a laser beam at 488 nm and 647 nm; emissions were simultaneously acquired with two separate photomultiplier tubes through 522-nm and 680-nm emission filters, respectively.
Reverse transcription–polymerase chain reaction and Northern blot analysis
Reverse transcription–polymerase chain reaction (RT-PCR) was carried out on total RNA according to the authors' previous method (Zhang et al., 1999b). Primers used to detect neuropilin-1 were synthesized according to mouse sequence (Kawakami et al., 1996): forward 5′ TGC CGT TGC TGT GCG CCA CGC TCG (nucleotides 361–375), reverse 3′ AGA CAA ATT TGA TGA AGA GAA AGG (nucleotide 696–720, GenBank accession No. NM 008737). Initially, reaction containing RNA (2 μg), RNase inhibitor (40 U), oligo-dT (1 μg), was made up to 20 μL with diethyl pyrocarbonate (DEPC) treated water and heated to 70°C for 10 minutes and then chilled on ice. Thereafter, final concentrations of dNTP (125 μg), RT-MMLV (40 U), and reverse transcriptase buffer (1×) were added in a total volume of 40 μL, and the reactions were incubated at 37°C for 60 minutes. Reverse transcriptase enzyme was inactivated by heating to 94°C for 5 minutes and chilling on ice. Aliquots (8 μL) of the cDNA reaction were used for PCR amplification. The samples were denatured at 95°C for 2 minutes and then amplified for 40 cycles. Each cycle consisted of denaturation at 95°C for 30 seconds, annealing at 52°C for 30 seconds, and extension at 72°C for 2 minutes. The samples (30 μL/well) were electrophoresed using 1× TAE buffer on 1.5% agarose containing ethidium bromide. To confirm expression of neuropilin-1, Northern blot was performed using a mouse cDNA of neuropilin-1 as a probe (gift from Dr. Fujisawa, Nagoya University) (Kawakami et al., 1996). The cDNA probe was labeled with [32P] dCTP using a Prime-a-Gene labeling system (Promega, Madison, WI, U.S.A.). The radioactive bands were quantified using an MCID image analysis system (Imaging Research, St. Catherine's, Canada).
In situ hybridization
The 562-bp mouse cDNA of neuropilin-1 was used as a probe for in situ hybridization (gift from Dr. Fujisawa, Nagoya University) (Kawakami et al., 1996). In situ hybridization was performed using a digoxigenin DNA labeling and detection kit (Boehringer Mannheim Biochemicals, Indianapolis, IN, U.S.A.) according to the manufacturer's protocol. Briefly, after deparaffinizing, coronal sections (6 μm) were fixed by 10% paraformaldehyde for 20 minutes at room temperature and were digested by proteinase K (100 μg/mL) for 15 minutes at 37°C. Prehybridization solution containing 4 × SSC, 50% deionized formamide, 1 × Denhardt's solution, 0.5 mg/mL salmon sperm DNA, 10% dextran sulfate, and 0.25 mg/mL yeast tRNA was applied for 1 hour at room temperature. The denatured digoxigenin-labeled cDNA probe concentration for hybridization was 750 ng/mL. Coronal sections were incubated under coverslips overnight at 42°C with hybridization solution in a humidified chamber. Posthybridization stringency washes at room temperature included 2 × SSC for 10 minutes and 60% formamide, 300 mmol/L NaCl, and 30 mmol/L sodium citrate for 30 minutes. After treatment with 2% normal sheep serum, hybridization probe was detected by an antidigoxigen antibody conjugated to alkaline phosphatase at a 1:500 dilution for 2 hours at room temperature. Nitro blue tetrazolium/5-bromo-4-chloro-3-indolyl phosphate solution was used as a color substrate. Color reaction time was 20 hours.
Measurement of vascular diameters
Each neuropilin-1 hybridized coronal section was digitized under a 20× or 40× objective (Olympus BX40; Olympus Optical, Japan) for measurement of diameters of neuropilin-1 mRNA vessels using a 3-CCD color video camera (Sony DXC-970MD; Sony, Japan) interfaced with the MCID image analysis system. The maximum diameter (the maximum internal distance perpendicular to the maximum curved chord) of vessels exhibiting neuropilin-1 mRNA was measured using the MCID system. Vessels were categorized by their diameters as capillary (<7.5 μm), precapillary arterioles and postcapillary venules (7.6 to 30 μm), or small arterioles and connecting vessels (31 to 50 μm) (Zhang et al., 1999b).
Statistical analysis
Analysis of variance followed by t-tests with Bonferroni correction was used. All data are presented as mean ± standard deviation. P < 0.05 was considered statistically significant.
RESULTS
In all rats subjected to middle cerebral artery occlusion, an ischemic lesion was detected in the ipsilateral hemisphere on coronal sections stained with hematoxylin and eosin. RT-PCR on total RNA isolated from the ischemic hemisphere showed a predicted 360-bp product from 2 hours to 7 days after ischemia, but not in the control rats (n = 3 per time point) (Fig. 1). To analyze neuropilin-1 expression further, a mouse cDNA of neuropilin-1 was used as a probe for Northern blot analysis. Neuropilin-1 mRNA was detected by Northern blot in nonoperated rats (n = 3), and its level was low, consistent with previous findings that the adult brain expresses much lower levels of neuropilin-1 than the embryonic brain (Kawakami et al., 1996). In contrast, significant increases in neuropilin-1 mRNA were detected in the ipsilateral hemisphere at 2 hours; they persisted at least to 7 days after occlusion (n = 3 per time point). In situ hybridization was performed to identify the cells expressing neuropilin-1. In situ hybridization analysis did not reveal neuropilin-1 mRNA in the cortex and the subcortex of nonoperated rats (n = 4) or in the contralateral cortex and subcortex of ischemic rats (n = 4 per time point). Neuropilin-1 mRNA was detected in triangular cells in the ischemic lesion at 4 to 24 hours and within the border of the ischemic lesion at 48 hours after occlusion (Fig. 2). Morphometric analysis from an adjacent section stained with hematoxylin and eosin confirmed that these cells were ischemically injured neurons. However, immunostaining for neuropilin-1 showed no neuropilin-1 immunoreactive neurons in the ischemic lesion. In contrast, neuropilin-1 immunoreactivity was detected on hypertrophic astrocytes within the border of the ischemic lesion 7 to 28 days after ischemia. In addition, neuropilin-1 mRNA was localized to large cerebral blood vessels and capillaries within and around the ischemic lesion 7 days after ischemia (Fig. 3). Higher magnification revealed that endothelial cells of cerebral blood vessels in the ipsilateral hemisphere were neuropilin-1 immunoreactive.
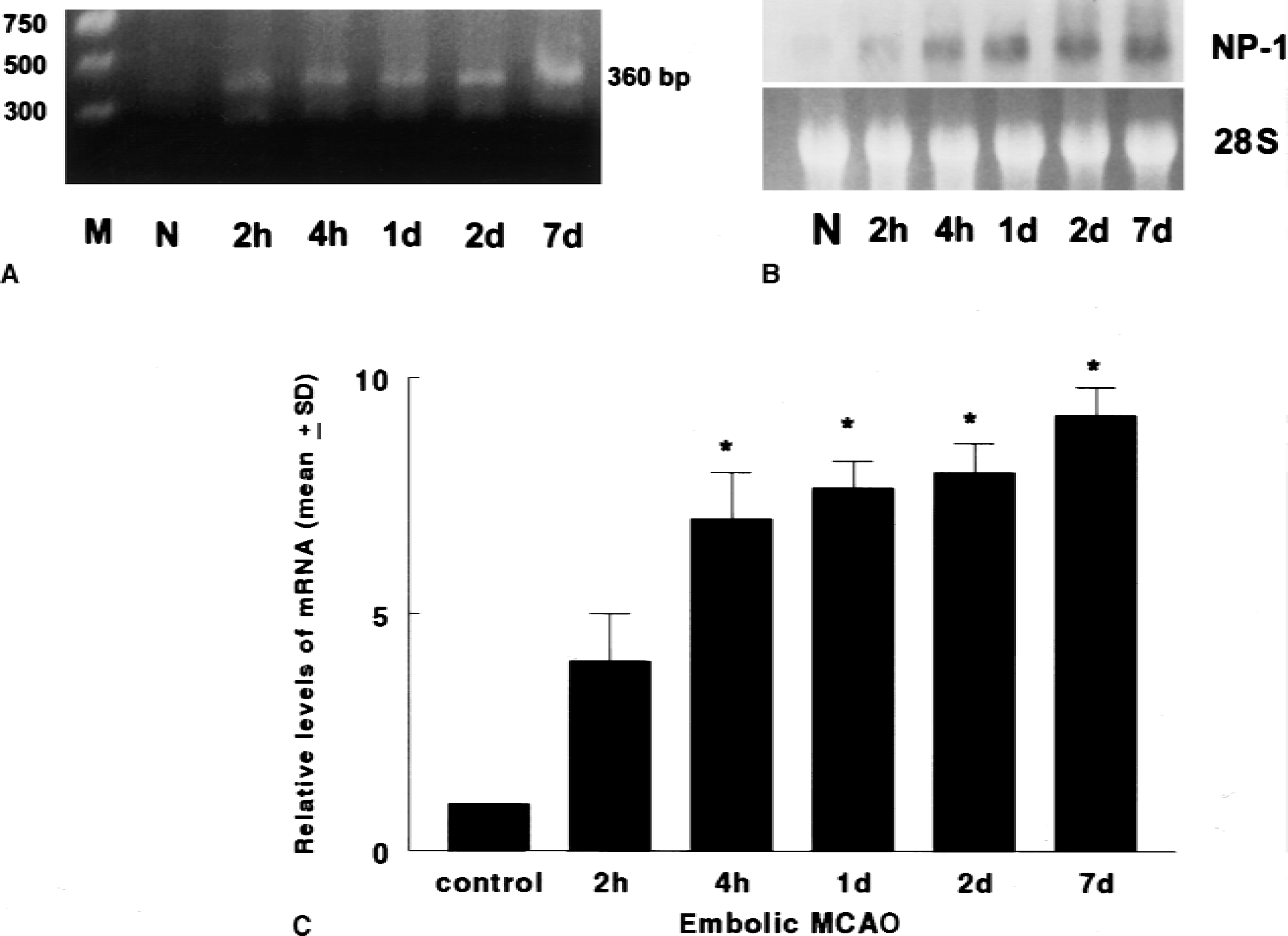
Reverse transcriptase–polymerase chain reaction (RT-PCR)
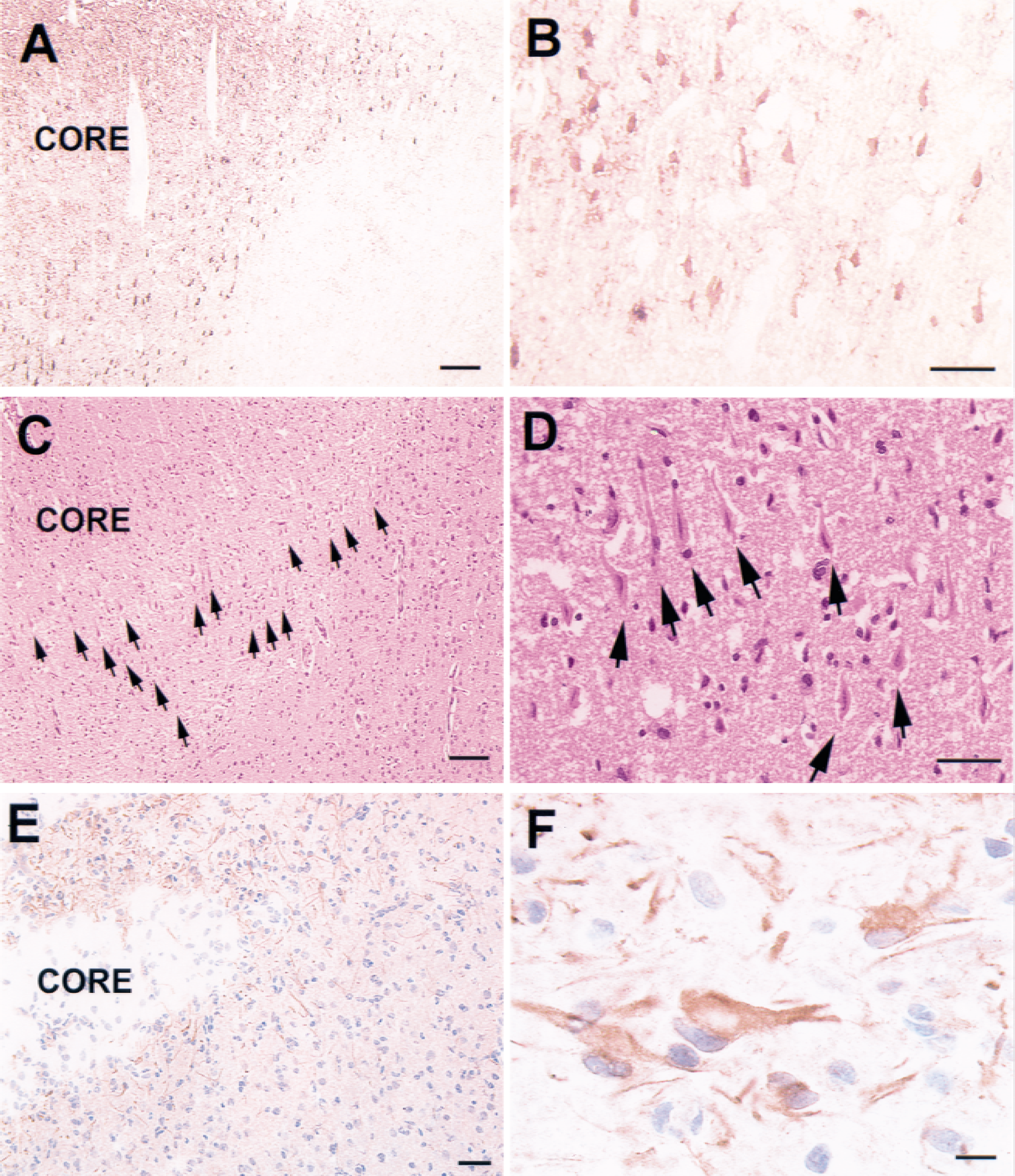
In situ hybridization
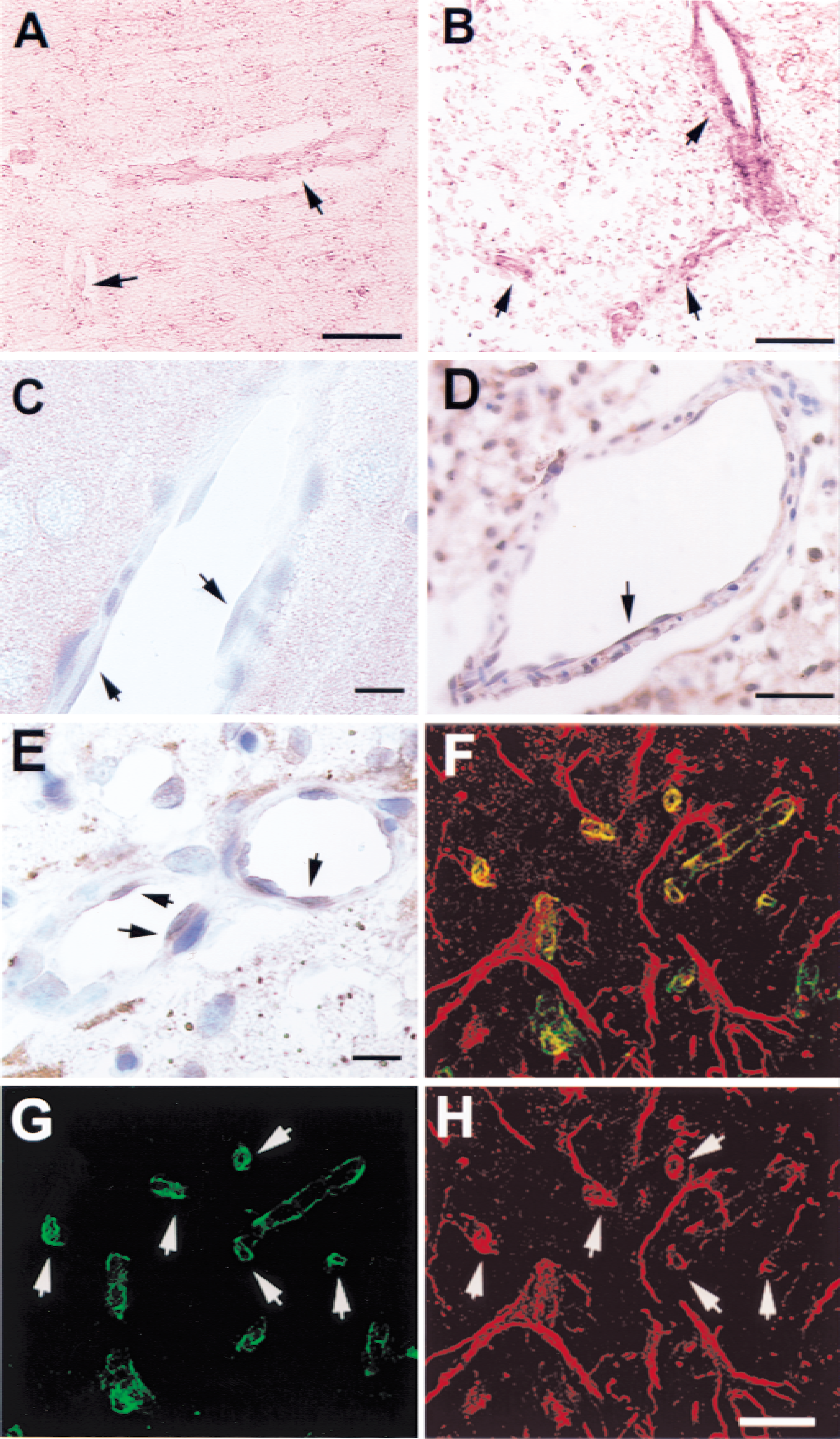
In situ hybridization of neuropilin-1
Quantitative analysis revealed that 70% of vessels with neuropilin-1 mRNA had diameters <7.5 μm, indicating that they were capillaries (Zhang et al., 1999b). Double immunofluorescent staining for neuropilin-1 and type IV collagen showed that some type IV collagen immunoreactive vessels exhibited neuropilin-1 immunoreactivity, indicating that vessels express neuropilin-1.
Three-dimensional images obtained from coronal sections perfused with FITC-dextran and immunofluorescently stained with GFAP showed strong GFAP immunoreactivity within the border of the infarct and an irregular pattern of tortuous cerebral blood vessels at the border of the infarct and the infarct areas 14 days after ischemia, suggesting gliosis and neovascularization in these areas (Fig. 4). Neuropilin-1 mRNA and protein were present in cerebral microvessels and astrocytes 28 days after ischemia. Double immunofluorescence labeling for VEGF and neuropilin-1 revealed that VEGF immunoreactive vessels and astrocytes were neuropilin-1 immunoreactive in the ipsilateral hemisphere, indicating colocalization of VEGF and neuropilin-1.
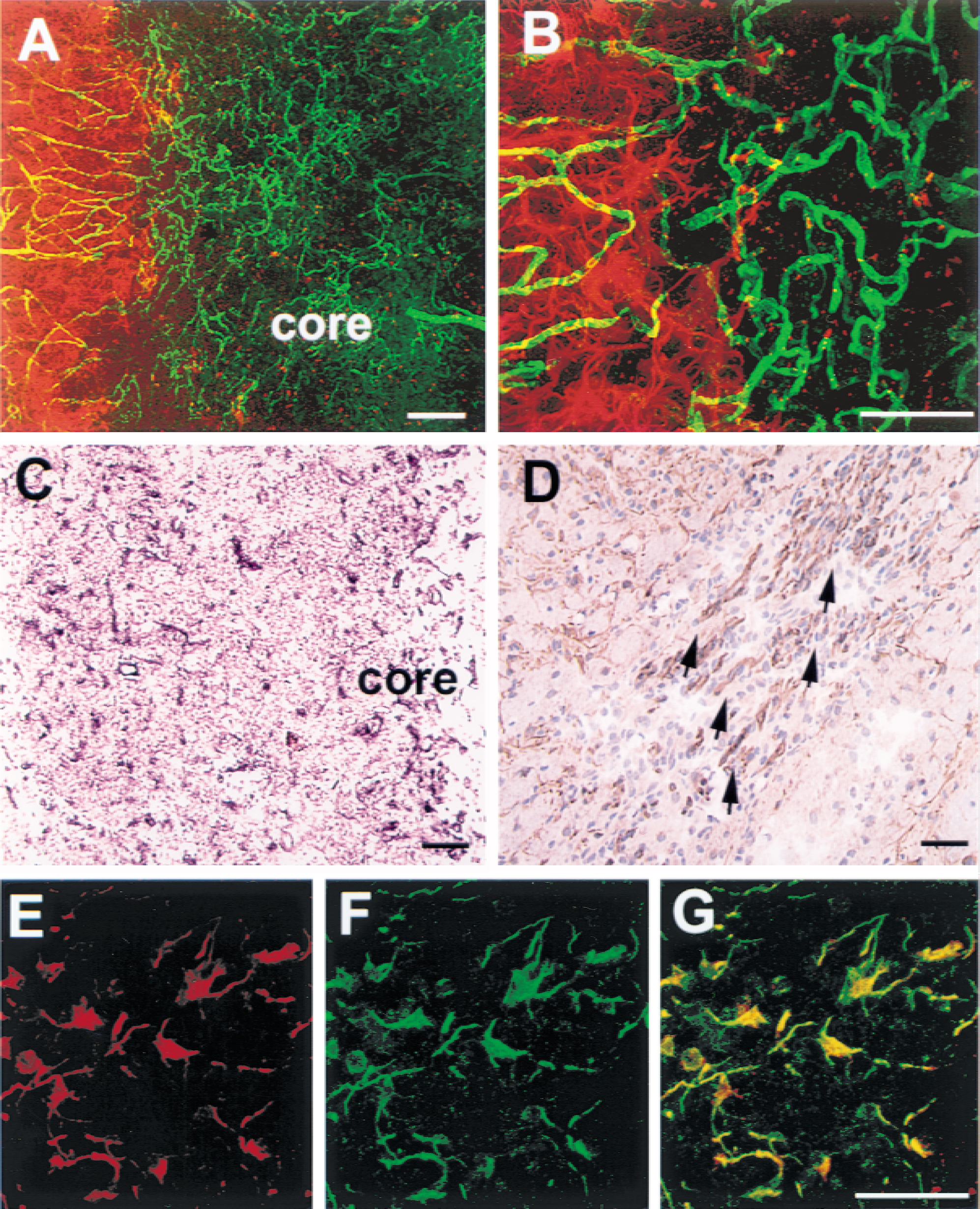
Laser scanning confocal microscope images from coronal sections with fluorescein isothiocyanate–dextran-perfused cerebral blood vessels (
DISCUSSION
The results of the current study show up-regulation of neuropilin-1 in the adult ischemic brain. A marked increase in the levels of expression of the neuropilin-1 was observed in endothelial cells and astrocytes at the border and within the ischemic lesion where neovascularization was present, initially at 2 days and extending through 28 days after ischemia. Expression of neuropilin-1 in endothelial cells is consistent with the results of previous studies that neuropilin-1 is expressed in endothelial cells in normal mouse embryos (Kawasaki et al., 1999; Kitsukawa et al., 1995). Studies on transgenic mice show that mouse embryos with overexpression of neuropilin-1 have excess blood vessels and dilation of vessels (Kitsukawa et al., 1995). In contrast, neuropilin-1 knockout mouse embryos show insufficient and delayed neural vascularization (Kawasaki et al., 1999). Both mutations of neuropilin-1 in mouse embryos are lethal (Kawasaki et al., 1999; Kitsukawa et al., 1995). Therefore, neuropilin-1 is essential for vascular development. Neovascularization in ischemic brain has been shown after focal cerebral ischemia in this rat model and others (Lin et al., 1993; Zhang et al., 2000). Regenerating capillaries are initiated at the border of the infarct areas between 2 and 7 days after ischemia (Garcia et al., 1971; Hills, 1964; Yoshida et al., 1989). An increase in neuropilin-1 mRNA and protein in the endothelial cells at the border and within the infarct at 7 days after ischemia is coincident with the time in which neovascularization becomes evident in ischemic brain. Further, most cerebral vessels with neuropilin-1 mRNA and protein were capillaries, in accordance with findings that mouse embryos with overexpression of neuropilin-1 exhibit excess capillaries (Kitsukawa et al., 1995). These data suggest that up-regulation of neuropilin-1 in the endothelial cells observed in the current study contributes to neovascularization in postischemic brain.
Interactions between neuropilin-1 and VEGF are demonstrated by in vitro studies that neuropilin-1 is a coreceptor for VEGF165 and enhances VEGF binding to its receptor KDR (Fuh et al., 2000; Soker et al., 1998). Increases in the density of neuropilin-1 on cells also enhance VEGF165 binding to neuropilin-1 (Fuh et al., 2000). Focal cerebral ischemia induces up-regulation of VEGF and its receptors, KDR and Flt-1, in endothelial cells (Cobbs et al., 1998; Hayashi et al., 1997; Lennmyr et al., 1998; Plate et al., 1999). Administration of VEGF165 at 48 hours after middle cerebral artery occlusion enhances angiogenesis within the penumbra in rats (Zhang et al., 2000). The results of the current study show that neuropilin-1 and VEGF were simultaneously expressed in endothelial cells in ischemic brain, suggesting that in addition to VEGF receptors KDR and Flt-1, neuropilin-1 may also regulate VEGF activity and thereby regulate new vessel formation in ischemic brain.
The role that neuropilin-1 plays in neovascularization of ischemic brain remains unclear, but its function during development of embryos indicates that it is required for the maturation and remodeling of the primary vascular network into the definitive embryonic vasculature (Kawasaki et al., 1999). Persistence of neuropilin-1 expression in endothelial cells beyond the initial period of angiogenesis in the first postischemic week suggests that neuropilin-1 may function in the maturation and remodeling of neovascularization in ischemic brain.
Astroglial end-feet and brain capillary endothelium have a common basement membrane (Paspalas and Papadopoulos, 1998; Risau et al., 1998). During normal development of brain and retinal vasculature, astrocytes play an important role in the formation of new vessels by secreting VEGF (Risau et al., 1998; Stone et al., 1995). After ischemia, hypertrophied GFAP immunoreactive astrocytes surround the ischemic lesion as early as 2 days after ischemia (Garcia et al., 1994; Zhang et al., 1999a). The progressive increase of neuropilin-1 immunoreactive astrocytes that began by 48 hours after ischemia and the colocalization of VEGF and neuropilin-1 in activated astrocytes suggest that activated astrocytes may also have a regulatory role for VEGF-mediated angiogenesis in the adult ischemic brain. However, in view of fact that neuropilin-1 also binds to semaphorin 3a, a chemorepulsive molecule of axonal growth, it is possible that increases in neuropilin-1 immunoreactive astrocytes are responsible for semaphorin 3a-mediated inhibition of axonal growth in the ischemic lesion (Bagnard et al., 1998; He and Tessier-Lavigne, 1997; Pasterkamp et al., 1999b).
In the current study, neuropilin-1 protein was immunolocalized to activated astrocytes in which neuropilin-1 mRNA expression was not detected by in situ hybridization. This result suggests that activated astrocytes may take up neuropilin-1 from the extracellular space in the same manner as they phagocytose serum proteins and fibronectin in areas of vasogenic edema (Nag et al., 1997). Alternatively, the method of in situ hybridization used in the current study is not sufficiently sensitive to detect neuropilin-1 mRNA in the activated astrocytes. Up-regulation of neuropilin-1 mRNA has been detected in vimentin immunoreactive cells after stab wound in the brain (Pasterkamp et al., 1999b), suggesting that astrocytes are capable of expressing neuropilin-1 mRNA in response to brain injury.
During normal development, neurons in the intermediate zone of the neocortex express neuropilin-1, and the expression of neuropilin-1 persists through the time when axons are actively growing to form neuronal connection, suggesting a role for neuropilin-1 in axonal growth and guidance (Kawakami et al., 1996). In the adult brain, neuropilin-1 expression is not detected in the cerebral cortex (Kawakami et al., 1996). Expression of neuropilin-1 mRNA in the ischemic neurons in the adult cortex indicates that ischemic brain reverts to a prenatal pattern of neuronal neuropilin-1 expression (Kawakami et al., 1996). However, these neurons did not exhibit neuropilin-1 immunoreactivity and seem destined for cell death (Garcia et al., 1993).
The results of the current study show up-regulation of neuropilin-1 in endothelial cells, astrocytes, and neurons in the adult brain after embolic focal cerebral ischemia. The temporal and spatial profiles of neuropilin-1 expression in endothelial cells in the areas with neovascularization suggest that neuropilin-1 may also play an important role in angiogenesis in concert with VEGF and its receptors. Because neuropilin-1 is a receptor for semaphorin 3a and VEGF, further study of the interactions between neuropilin-1 and VEGF as well as neuropilin-1 and semaphorin 3a may lead to new understanding of angiogenesis and neuronal plasticity in the adult ischemic brain.
Footnotes
Acknowledgments:
The authors thank Dr. H. Fujisawa for providing the antineuropilin-1 antibody and cDNA of mouse neuropilin-1, and Rita Tobey for manuscript preparation.
