Abstract
Technetium-99m-d,l-hexamethylpropylene amine oxime (99mTc-HMPAO) retention in the brain monitored by single photon emission computed tomography (SPECT) is currently used as a marker of cerebral blood flow. The purported mechanism by which 99mTc-HMPAO accumulates in the brain is through its intracellular conversion from a lipophilic form to more hydrophilic derivatives within the brain parenchyma. The issue of the contribution of different cell types on 99mTc-HMPAO retention was investigated in vitro by studying the accumulation of 99mTc-HMPAO in primary cultures of mouse cortical astrocytes and neurons. Results show that 99mTc-HMPAO retention predominates in astrocytes over neurons by a factor of ∼2.5 (0.26 ± 0.05 vs. 0.095 ± 0.042 fmol/mg protein after 120 minutes, respectively). Diethyl maleate (60 μmol/L), ethacrynic acid (1 mmol/L) and buthionine sulfoximine (1 mmol/L), 3 agents which significantly reduced glutathione levels also decreased 99mTc-HMPAO retention in both astrocytes (29%, 3%, and 46% of control, respectively) and neurons (69%, 11% and 63% of control). Decrease did not always correlate with glutathione levels, however, which suggests that other factors could be involved. The possibility that cell energy status determines 99mTc-HMPAO retention was also assessed. Agents that activate (glutamate, azide) or inhibit (cytochalasin B) glucose utilization in astrocytes, as measured by the 3H-2-deoxyglucose method, were without effect on 99mTc-HMPAO retention. In conclusion, the data presented indicate that astrocytes may constitute a prominent site of 99mTc-HMPAO retention and most likely contribute significantly to the SPECT signal. In addition, the data also suggest that specific alterations in glial cell metabolism could explain flow-independent changes in 99mTc-HMPAO retention in the brain as observed by SPECT in some pathologies.
Technetium-99m-d,l-hexamethylpropylene amine oxime (99mTc-HMPAO) has been developed as a tracer for measurement of regional cerebral blood flow (rCBF) using single photon emission computed tomography (SPECT) (Neirinckx et al., 1987). This lipophilic compound has the ability to cross the blood–brain barrier and accumulates in the brain proportionally to blood flow, as demonstrated by studies performed in humans (Leonard et al., 1986). However, linearity between retention of 99mTc-HMPAO and cerebral blood flow as determined by other methods (Andersen et al., 1988b; Inugami et al., 1988; Yonekura et al., 1988) was not always observed, especially at high flow rate. In addition, both hyper- and hypofixations relative to other CBF indicators have been reported in pathologic cases (Launes et al., 1995; Ogasawara et al., 2000), suggesting that in addition to blood flow, other factors could determine the levels of 99mTc-HMPAO accumulation within a specific region of the brain.
Previous reports have shown that 99mTc-HMPAO is converted from its lipophilic form to hydrophilic derivatives in the brain with the consequence that 99mTc-HMPAO remains trapped within the brain parenchyma (Neirinckx et al., 1988). A few in vitro studies have been performed to elucidate the molecular mechanism(s) responsible for 99mTc-HMPAO retention. Glutathione, an important reducing agent present in the central nervous system, has been directly implicated in the mechanism leading to 99mTc-HMPAO conversion (Neirinckx et al., 1988). Other factors, however, also have been proposed to play a role in flow-independent accumulation of 99mTc-HMPAO, including changes in oxido-reductive state (Jacquier-Sarlin et al., 1996), metabolic alterations (Ahn et al., 1994), and formation of a complex with proteins in subcellular organelles (Costa et al., 1989).
Very few data exist on the identity of the cells in which 99mTc-HMPAO conversion and its subsequent accumulation take place. Most in vitro studies have been performed either in brain slices (where cellular localization can not be determined) (Ahn et al., 1994; Colamussi et al., 1999) or in neuronally-enriched cultures (De Jong and Van Royen, 1989; Suess et al., 1992). Astrocytes, however, represent another important cell type in the central nervous system. They outnumber neurons by a factor of ten and occupy half of the brain's volume (Bignami, 1991). In addition, astrocytes possess specialized processes called end-feet, which cover almost entirely the surface of intraparenchymal capillaries (Peters et al., 1991). This feature suggests that astrocytes could represent a privileged site of 99mTc-HMPAO uptake and retention as it penetrates within the brain parenchyma. To investigate this possibility, the authors studied and compared 99mTc-HMPAO retention in both primary cultures of mouse cortical astrocytes and neurons. The current results suggest that astrocytes significantly contribute to 99mTc-HMPAO retention in the central nervous system.
MATERIALS AND METHODS
Media and reagents
Fetal calf serum was purchased from Fakola AG, Basel, Switzerland. Dulbecco's Modified Eagle Medium (DMEM), 5,5′- dithiobis (2-nitrobenzoic acid) (DTNB), 5-sulfosalicylic acid, GSH (reduced glutathione), GSSG (oxidized glutathione), as well as common chemicals, were obtained from Sigma, Buchs, Switzerland.
“Neurobasal” medium (without glutamine) and B–27 supplement were purchased from GIBCO BRL, Basel, Switzerland. NADPH and glutathione reductase were obtained from Boehringer, Mannheim, Germany. 3H-Deoxy-D-glucose ([3H]–2DG) was purchased from ANAWA Trading SA, Zurich, Switzerland. 2-Vinyl-pyridine (2VP; 97% pure) was obtained from Aldrich, Buchs, Switzerland.
Cell cultures
Primary cultures of cerebral cortical astrocytes were prepared from newborn Swiss Albino mice (1- to 2-days-old) as described by Sorg and Magistretti (1991). The cells were seeded in a final volume of 2.5 mL/dish of DMEM medium (D7777) containing 25 mmol/L glucose and supplemented with 10% fetal calf serum, 44 mmol/L NaHCO3, and 10 mL/L of an antibiotic-antimycotic solution (A-7292 Sigma), and then were incubated at 37†C in a water-saturated atmosphere containing 5% CO2:95% air. Five days after seeding, cells were washed once with DMEM–fetal calf serum, and subsequently, the medium was replaced every 2 to 3 days. Cultures were used for experiments at confluence, usually between 19 and 22 days after seeding. Cultures were stained immunocytochemically with the glial marker glial fibrillary acidic protein (GFAP) and were found to contain 90% to 95% of GFAP-positive cells as the authors previously described (Do et al., 1997).
Primary cultures of cerebral cortical neurons were prepared from 16- to 17-day-old Swiss Albino mice embryos as described by Stella et al. (1995) with some modifications. The cells were resuspended in a specifically designed neuronal culture medium made of “neurobasal” medium supplemented with B-27 at 2% and glutamine at 500 μmol/L (Brewer et al., 1993). Cells were seeded in a final volume of 2.5 mL/dish in Petri dishes that were precoated at least 2 hours with polyornithine. Cells were incubated at 37†C in a water-saturated atmosphere containing 5% CO2:95% air. Cultures were used for experiments at either 6 days or between 19 and 22 days after seeding. Cultures were stained immunocytochemically with the neuronal marker microtubule-associated protein 2 (MAP2) and were found to contain 90% to 95% of MAP2-positive cells.
Preparation of 99mTc-HMPAO
The 99mTc-HMPAO complex was produced by mixing 5 mL fresh 99mTc-pertechnetate obtained from a standard generator in daily use at the Department of Nuclear Medecine of the Center Hospitalier Universitaire Vaudois in Lausanne, Switzerland, with 0.5 mg of freeze-dried d,l-HMPAO in commercially supplied vials (Ceretec, Amersham Dübeudorf, Switzerland). The labeling procedure was performed following the manufacturer's recommendations immediately before use, and a radiochemical purity assessment by thin layer chromatography was performed for each preparation to define the percentage of total radioactivity being incorporated to the lipophilic form. The radiopurity of the preparation was always greater than 90%.
Uptake of 99mTc-HMPAO
Cells were preincubated where indicated in their respective culture medium with different substances for either 20 minutes, 2 hours, or 24 hours. In the case of time course studies, however, the medium was replaced with serum-free DMEM (D5030) supplemented with 5 mmol/L glucose, 44 mmol/L NaHCO, 0.045 mmol/L phenol red, and 10 mL/L of an antibiotic-antimycotic solution (A-7292, Sigma) 2 hours before incubation with the radioactive tracer. The medium then was replaced by 2 mL DMEM D5030 containing 99mTc-HMPAO at a concentration of 5 μCi/mL (1 μmol/L), and the cells were incubated for 20 minutes. The reaction was stopped by aspiration of the medium. Cells were rinsed three times with ice-cold phosphate-buffered saline. The cells were lysed by adding 2 mL of NaOH 10 mmol/L. Aliquots from the cell lysate were assayed for radioactivity by gamma-counting (2 × 500 μL), for glutathione by an enzymatic spectrophotometric assay (1 × 500 μL), and for protein measurement (2 × 50 μL) by the method of Bradford (1976). Results were also adjusted for the radioactive decay of 99mTc (T1/2 = 6 hours).
[3H]-2DG uptake
[3H]-2DG uptake was determined as previously described by Pellerin and Magistretti (1994). On the day of the experiment, the medium was replaced by 2 mL of the serum-free DMEM (D5030) supplemented with 5 mmol/L glucose, 44 mmol/L NaHCO3, 10 mL/L of an antibiotic-antimycotic solution (A-7292, Sigma), and 0.045 mmol/L phenol red containing [3H]-2DG at a concentration of 1 μCi/mL (∼33 nmol/L). Pharmacologic agents and inhibitors were coincubated with [3H]-2DG during 20 minutes. Reaction was stopped by aspiration of the medium, cells were rinsed 3 times with ice-cold phosphate-buffered saline, and 2 mL of NaOH 10 mmol/L was added to lyse the cells. Aliquots from the cell lysate were assayed for radioactivity by liquid scintillation beta-counting (2 × 500 μL), for glutathione by an enzymatic spectrophotometric assay (1 × 500 μL), and for protein measurement (2 × 50 μL) by the method of Bradford (1976).
Octanol extraction
Five hundred microliters of the cell lysate (or the incubation medium containing the 99mTc-HMPAO complex in certain cases) was extracted with 1 mL octanol (Andersen et al., 1988). After spontaneous separation of the 2 phases, 500 μL of the octanol fraction was assayed for radioactivity by gamma-counting. A 500-μL aliquot of the original cell lysate (or incubation medium) was also directly assayed to determine total counts. The percentage of lipophilic form present was calculated as described by Andersen et al. (1988a).
Glutathione assay
Total glutathione (GSH + GSSG) was assayed by an enzymatic recycling procedure as described by Baker et al. (1990) with some modifications made to measure glutathione in parallel to 99mTc-HMPAO retention in the same cells. The principle is based on the sequential oxidation of glutathione by DTNB (acid chromophore) and its reduction by NADPH in the presence of glutathione reductase. Standards containing from 1 μmol/L to 20 μmol/L of GSH, as well as blanks (mix solution without glutathione), were prepared in NaOH 10 mmol/L neutralized at pH 7 with HCL 1N and kept on ice until loading on the microtiter plate. Sample aliquots of 500 μL were also neutralized by HCL 1N. A reagent mix solution containing DTNB 0.15 mmol/L, NADPH 0.2 mmol/L, and glutathione reductase 1 U/mL in phosphate-buffered saline was freshly prepared at room temperature. A microtiter plate was first prepared by pipetting 50 μL of standards, samples, and blanks. Then 100 μL of the reagent mix solution was added into each well to start the reaction. A microtiter plate reader (Microtec produkte AG, Embrach-Embraporte, Switzerland) was used to monitor the rate of TNB formation at 405 nm for 4.5 minutes. Glutathione levels were determined by comparison with a standard curve of GSH.
To determine if treatments could affect the GSH:GSSG ratio, both GSH and GSSG contents in cell lysates were determined according to Baker et al. (1990). Reaction was stopped by aspiration of the medium, cells were rinsed twice with ice-cold phosphate-buffered saline, and 1 mL of a sulfosalicylic acid solution (1% in H2O) was added to lyse the cells. After 3 minutes on ice, cells were scraped off the plate and transferred to an eppendorf tube. After centrifugation (12,000 g, 5 minutes, 4†C), aliquots of the supernatant were analyzed for both total glutathione and GSSG contents. Ten microliters of the supernatant or GSSG standards (0 to 100 pmol of GSSG/10 μL) was transferred into microtiter plate wells and diluted with 90 μL H2O. In parallel, supernatant aliquots were treated with 2VP to derivatize GSH and allow GSSG determination. In brief, 130 μL of the supernatant was mixed with 5 μL 2VP and neutralized with 0.2 mol/L Tris to a pH between 5 and 7. A set of GSSG standards was treated the same way. After a 1-hour incubation at room temperature, 10 μL of the 2VP-treated samples and standards also was transferred into a microtiter plate and volume completed with 90 μL H2O. Then 100 μL of the same reagent mix solution, as described above, was added into each well to start the reaction. Each set of determination (total glutathione or GSSG) was obtained by comparison with its appropriate standard curve. GSH content was determined by subtracting GSSG values from total glutathione measurements.
Statistics
All results are presented as mean ± SD. Statistical analysis was performed with analysis of variance followed by Dunnett's test using the INSTAT software (Graphpad, San Diego, CA, U.S.A.).
RESULTS
Stability of the lipophilic form of 99mTc-HMPAO in the incubation medium was first determined for periods of time up to 4 hours (Fig. 1A), using the octanol extraction method which allows the separation of the lipophilic and hydrophilic forms of the 99mTc-HMPAO complex. Within the first few minutes after preparation, 91.8% ± 0.9% of the 99mTc-HMPAO was recovered as its lipophilic form. The 99mTc-HMPAO complex appeared stable up to 40 minutes, because 89.8% ± 1.9% of lipophilic form was still found after this time period. A linear decrease in the stability of the lipophilic form then was observed up to 240 minutes, the last time point measured, reaching a value of 36.8% ± 1.1%. A similar experiment was performed in the medium recommended by the commercial supplier (data not shown) that yielded comparable results (65.1% vs. 66.7% of the lipophilic form after 120-minute incubation).
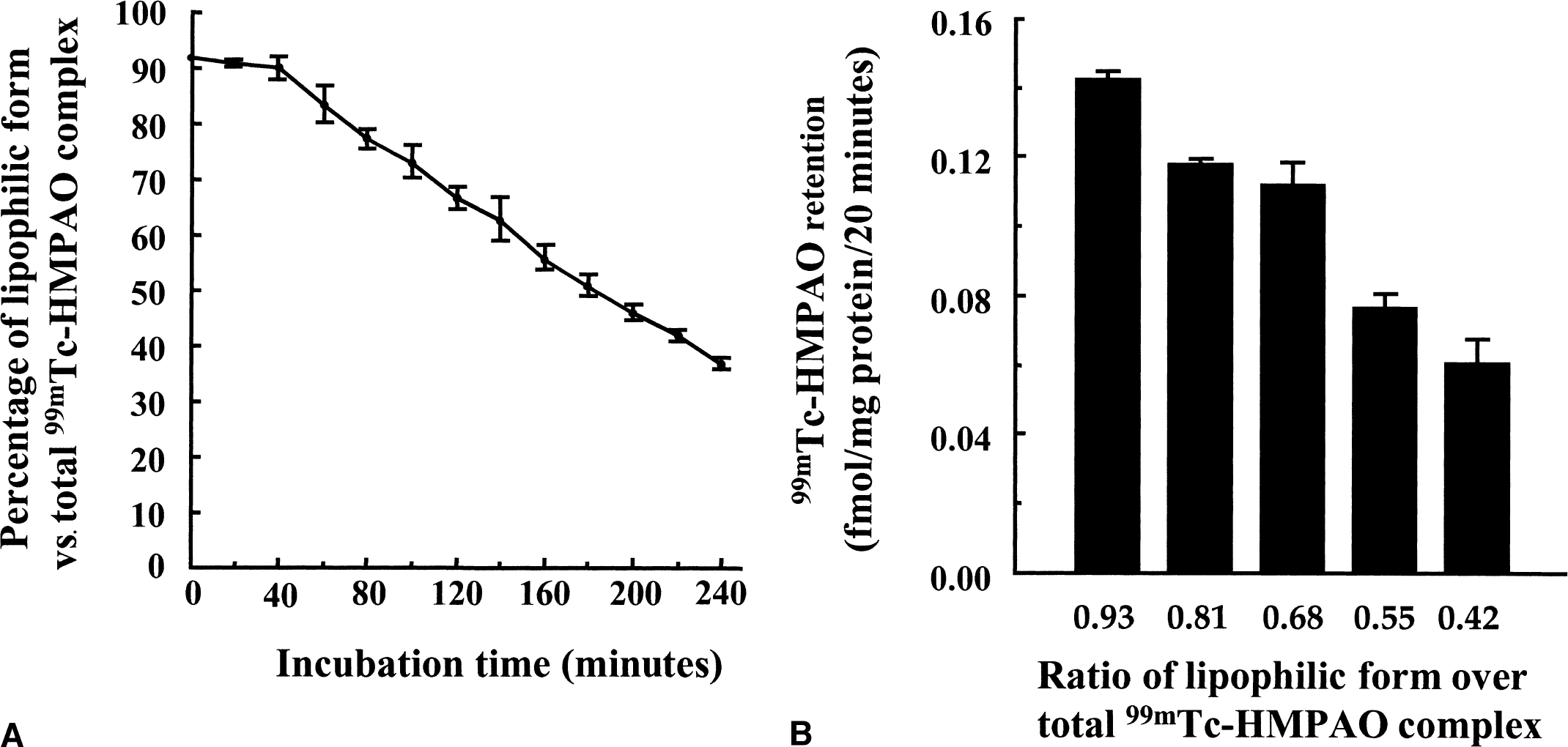
The influence of varying proportions of lipophilic versus hydrophilic forms of the 99mTc-HMPAO complex on its uptake in cultured mouse cortical astrocytes then was evaluated (Fig. 1B). Five different ratios of lipophilic form over total 99mTc-HMPAO complex corresponding to the ratios found after 0, 1, 2, 3, and 4 hours of incubation were selected and uptake of 99mTc-HMPAO complex was measured over a 20-minute period. A decrease of uptake was found in parallel with decreasing ratios, ranging from 0.142 ± 0.001 fmol/mg protein/20 min for the greatest ratio (0.97) to 0.060 ± 0.006 fmol/mg protein/20 min for the lowest ratio (0.43). For a ratio of 0.70, which corresponds to the ratio reached after 120 minutes, the uptake measured represents 78.9% of the initial uptake (0.112 ± 0.006 vs. 0.142 ± 0.001 fmol/mg protein/20 min). Based on this observation, the authors decided to perform all subsequent experiments within a 2-hour period after the complex preparation to minimize the effect of decreasing ratios on uptake values.
99mTc-HMPAO uptake was characterized in primary cultures of mouse cortical astrocytes and neurons (Fig. 2). In astrocytes, uptake was biphasic with a rapid accumulation within 60 minutes followed by a lower uptake rate between 60 and 120 minutes. Neurons displayed a rapid uptake up to 60 minutes followed by a plateau. Measurements performed at 4†C revealed a reduced uptake in both astrocytes and neurons. Maximal uptake after 120 minutes of incubation at 37†C reached an average value of 0.26 ± 0.05 fmol/mg protein in astrocytes (calculated from 12 separate determinations made in 4 independent experiments) as compared with 0.095 ± 0.042 fmol/mg protein in neurons (obtained from 18 separate determinations made in 6 independent experiments). Overall, uptake of 99mTc-HMPAO was approximately 2.5 times greater in astrocytes than in neurons after 120 minutes of incubation. Attempts were made to identify a saturable component for 99mTc-HMPAO uptake in both astrocytes and neurons. Uptake was determined with increasing concentrations up to 16 times the initial 99mTc-HMPAO concentration. The uptake increased linearly within that range of concentrations (162.5 nmol/L to 2.6 μmol/L) with no indication of saturation (data not shown).
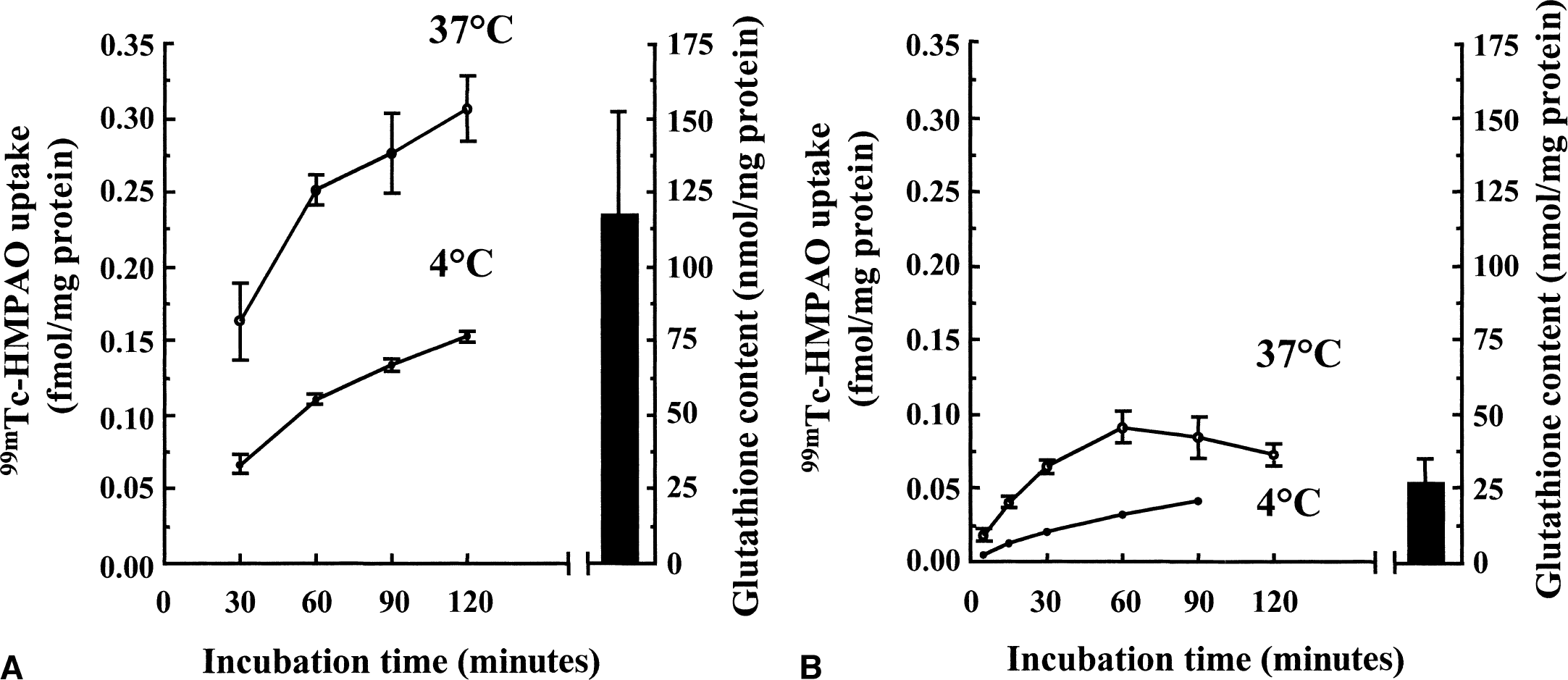
Time course of 99mTc-HMPAO uptake and total glutathione levels in mouse cortical astrocytes and neurons in culture. Astrocytes
The possibility that 99mTc-HMPAO uptake is reversible and that it can be released from astrocytes and neurons was then evaluated (Fig. 3). Cells were allowed to accumulate 99mTc-HMPAO for 120 minutes. At that time, the incubation medium containing the radioactive 99mTc-HMPAO complex was removed, and cells were extensively washed and placed in fresh incubation medium containing no radioactivity. At different time intervals after washout, the 99mTc-HMPAO cell content and the amount of 99mTc-HMPAO complex released in the medium were determined. 99mTc-HMPAO cell content initially decreased immediately after washout and then stabilized for up to 120 minutes (maximal decrease of 26.8% in astrocytes and 41% in neurons). Because the decrease ceased rapidly after washout, it appears that most of the 99mTc-HMPAO complex must be retained within cells. To determine under which form the 99mTc-HMPAO complex is retained, octanol extractions were performed. It was found that after 120 minutes of uptake (immediately before washout), most of the 99mTc-HMPAO complex is recovered as the hydrophilic form in both astrocytes (95% ± 3.5%; n = 30) and neurons (91.2% ± 2.1%; n = 12). This proportion then remained essentially the same throughout the incubation period after washout. In addition, the fraction that was released into the medium during the incubation period after the washout corresponded essentially to the hydrophilic form (92.9% ± 1.1%, n = 54 for astrocytes; 85.3% ± 4.7%, n = 18 for neurons).
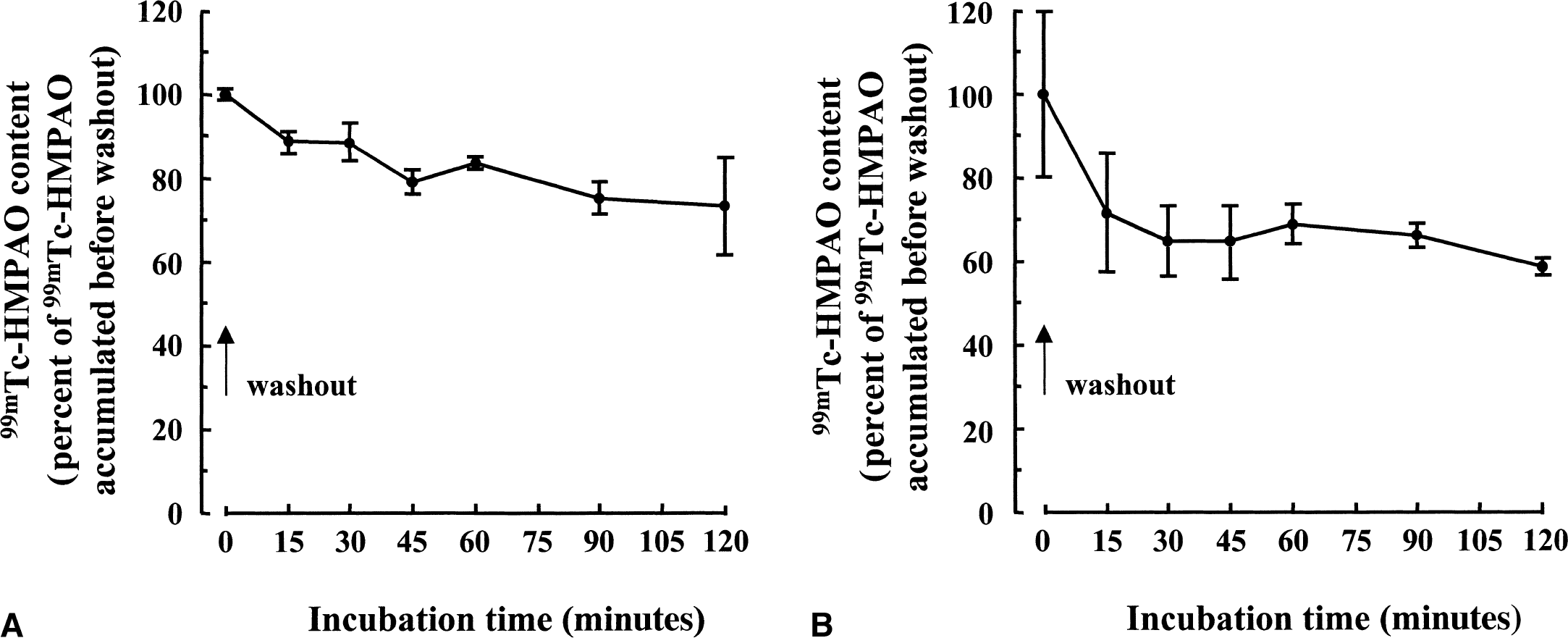
Retention of 99mTc-HMPAO complex within mouse cortical astrocytes and neurons. Astrocytes
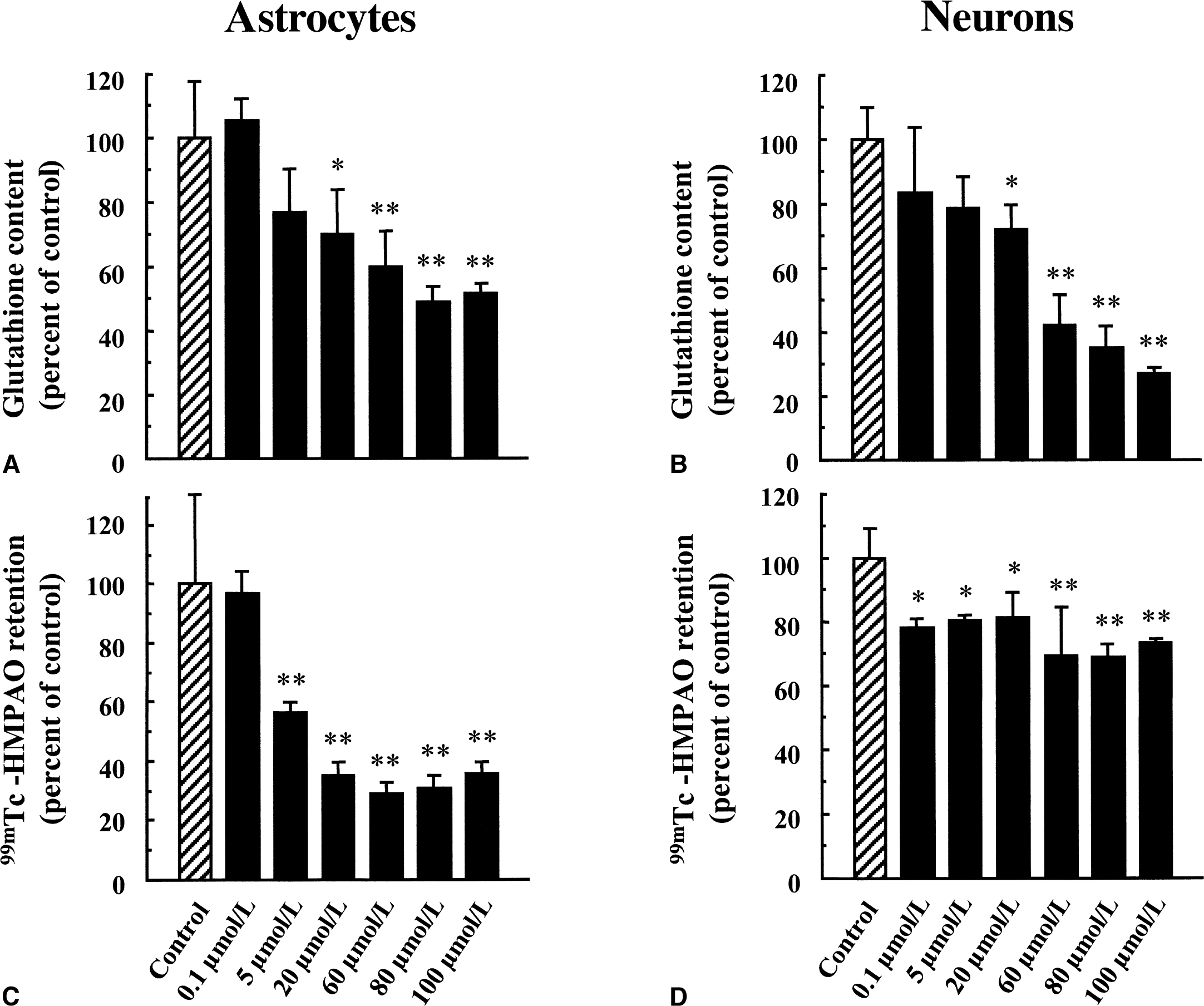
It was previously proposed that glutathione, by reacting with 99mTc-HMPAO, could account for the intracellular conversion of the 99mTc-HMPAO complex from its lipophilic to its hydrophilic form, thus causing its retention within cells. Total glutathione content was determined in primary cultures of mouse cortical astrocytes and neurons and found to be more important in astrocytes (117.11 ± 34.60 nmol/mg protein, n = 49 vs. 26.57 ± 7.45 nmol/mg protein, n = 75, respectively, see Fig. 2). The proportion of reduced versus oxidized glutathione also was determined, and more than 98% of total glutathione was found under its reduced form. The difference in total glutathione content correlated with the observed difference in 99mTc-HMPAO retention between astrocytes and neurons (Fig. 2), suggesting a possible role for glutathione in this process. To determine if glutathione levels were effectively linked to the extent of 99mTc-HMPAO retention within cells, the effect of substances known to alter cellular glutathione content was investigated in both astrocytes and neurons. As a preliminary control, the authors used the octanol extraction procedure to verify that none of the substances used interacted directly with 99mTc-HMPAO to convert it to hydrophilic derivatives (data not shown). Furthermore, the GSH:GSSG ratios even at concentrations in which they affect total glutathione levels were not altered (data not shown). In a first series of experiments, both astrocytes and neurons were preexposed to increasing concentrations of diethyl maleate (DEM), an agent known to conjugate with glutathione (Chasseaud, 1976). As expected, DEM caused a concentration-dependent decrease of the total glutathione content in both astrocytes and neurons with maximal reductions of 48.3% ± 2.6% and 72.9% ± 1.1%, respectively, observed at 100 μmol/L (Fig. 4A and 4B). In parallel, a concentration-dependent decrease in 99mTc-HMPAO retention was observed in astrocytes with a maximal reduction of 71.1% ± 3.4% obtained at 60 μmol/L (Fig. 4C). In contrast, the effect of DEM on 99mTc-HMPAO retention in neurons was small (30.8% ± 14.8% at 60 μmol/L) and was already reached at the lowest concentration tested (Fig. 4D). Ethacrynic acid (EA) is another substance that was shown to conjugate directly with glutathione (Awasthi et al., 1993). Thus, astrocytes and neurons were exposed for 2 hours to increasing concentrations of EA, and both total glutathione levels and 99mTc-HMPAO retention were assessed. Ethacrynic acid reduced glutathione levels in a concentration-dependent manner in both astrocytes and neurons, although the effect appeared to be more extensive in astrocytes (Fig. 5A and 5B). In parallel, 99mTc-HMPAO retention was reduced in a concentration-dependent manner to reach only 3% ± 0.1% of control levels at 1 mmol/L in astrocytes (Fig. 5C) and only 10.7% ± 0.1% in neurons (Fig. 5D). Finally, cells were exposed for 24 hours to buthionine sulfoximine (BSO), a specific inhibitor of the enzyme glutathione synthetase (Griffith and Meister, 1979), which is known to dramatically decrease glutathione content in various preparations. As shown in Fig. 6A and 6B, only 10 μmol/L BSO was sufficient to provoke a very important or even complete disappearance of glutathione content in both astrocytes and neurons. When the effect of BSO on 99mTc-HMPAO retention was determined, a distinct picture emerged. In astrocytes, 99mTc-HMPAO retention started to be significantly affected only at a concentration of 100 μmol/L and was maximally reduced to 46.1% ± 7% of the control at 1 mmol/L (Fig. 6C). In neurons, however, 99mTc-HMPAO retention was already decreased at the lowest concentration of BSO tested (62.7% ± 2.8% of control at 1 μmol/L) and did not significantly decrease further at greater concentrations (Fig. 6D).
Concentration-dependent effect of diethyl maleate (DEM) on total glutathione content and 99mTc-HMPAO retention in cultured astrocytes and neurons. Levels of total glutathione
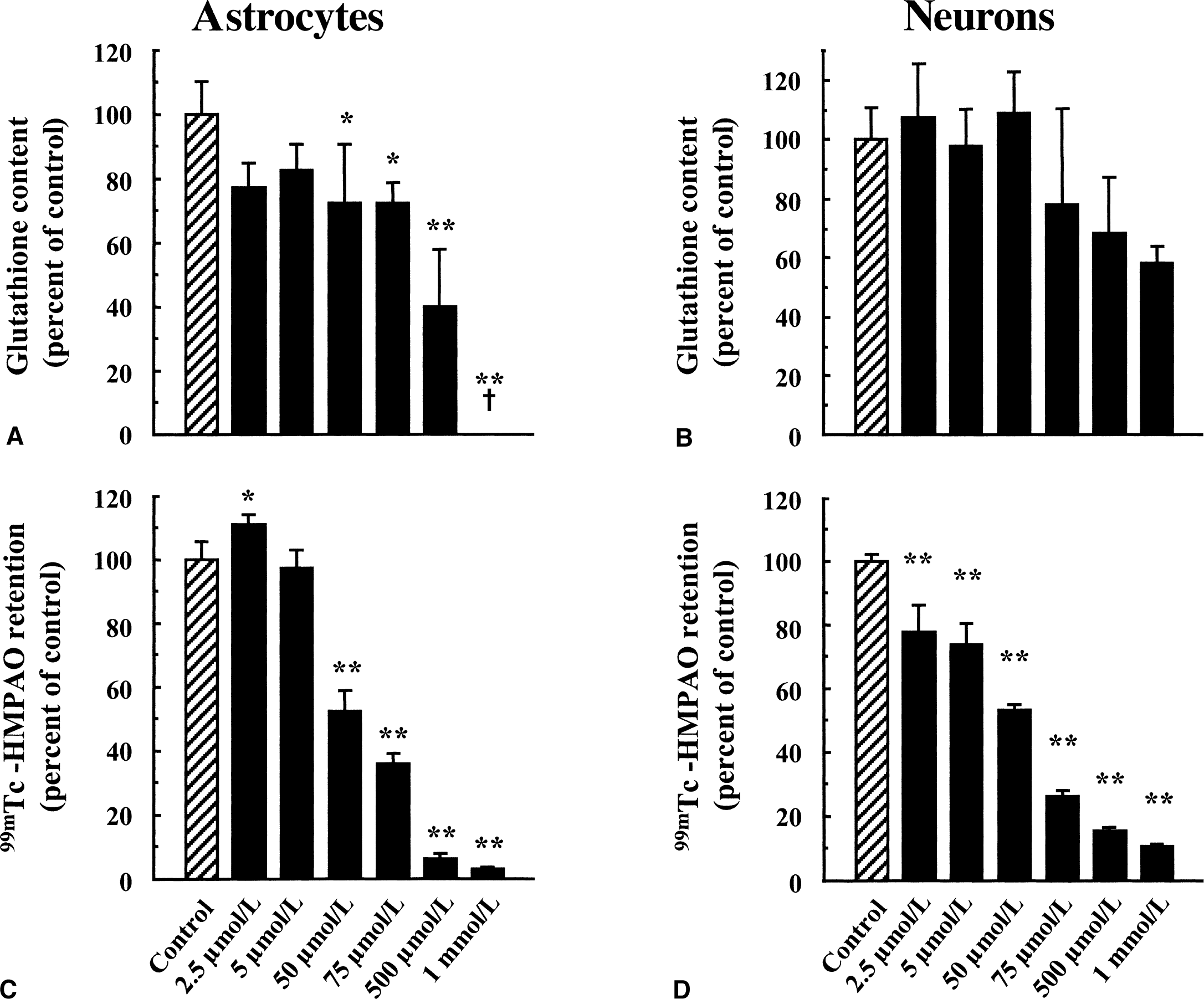
Concentration-dependent effect of ethacrynic acid (EA) on total glutathione content and 99mTc-HMPAO retention in cultured astrocytes and neurons. Levels of total glutathione
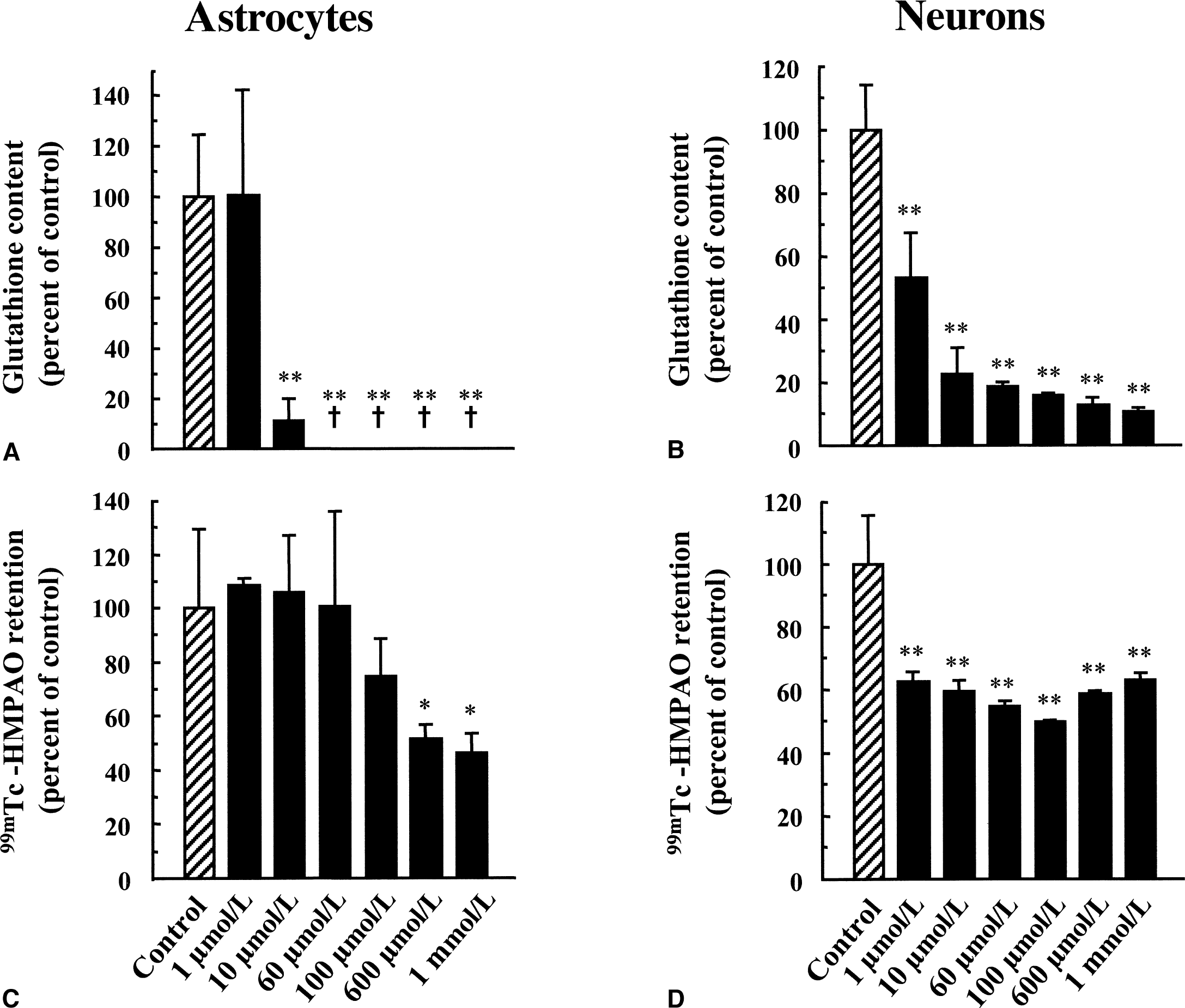
Concentration-dependent effect of buthionine sulfoximine (BSO) on total glutathione content and 99mTc-HMPAO retention in cultured astrocytes and neurons. Levels of total glutathione
Because it had been suggested in a previous study that metabolic alterations could affect 99mTc-HMPAO retention (Ahn et al., 1994), the effect of various agents known to alter energy metabolism in astrocytes was evaluated. It was first observed that both cytochalasin B, an inhibitor of glucose transporters, and iodoacetate, an inhibitor of glycolysis, reduced glucose utilization by astrocytes, as determined by measuring [3H]-2-deoxyglucose uptake (Fig. 7A). In contrast, the excitatory neurotransmitter glutamate and the cytochrome oxidase inhibitor, azide, enhanced glucose utilization by astrocytes (Fig. 7A). When 99mTc-HMPAO retention was determined under the same conditions, no effect was observed with either cytochalasin B, azide, or glutamate (Fig. 7B). Replacement of glucose in the medium by the unmetabolizable analog 2-deoxyglucose did not alter 99mTc-HMPAO retention (Fig. 7B). Iodoacetate was the only substance that led to a significant reduction in 99mTc-HMPAO retention (Fig. 7B). Iodoacetate, however, also was found to decrease glutathione levels in astrocytes (49.9% ± 10.5% of control level; P < 0.05 vs. control), whereas the other agents had no significant effect.
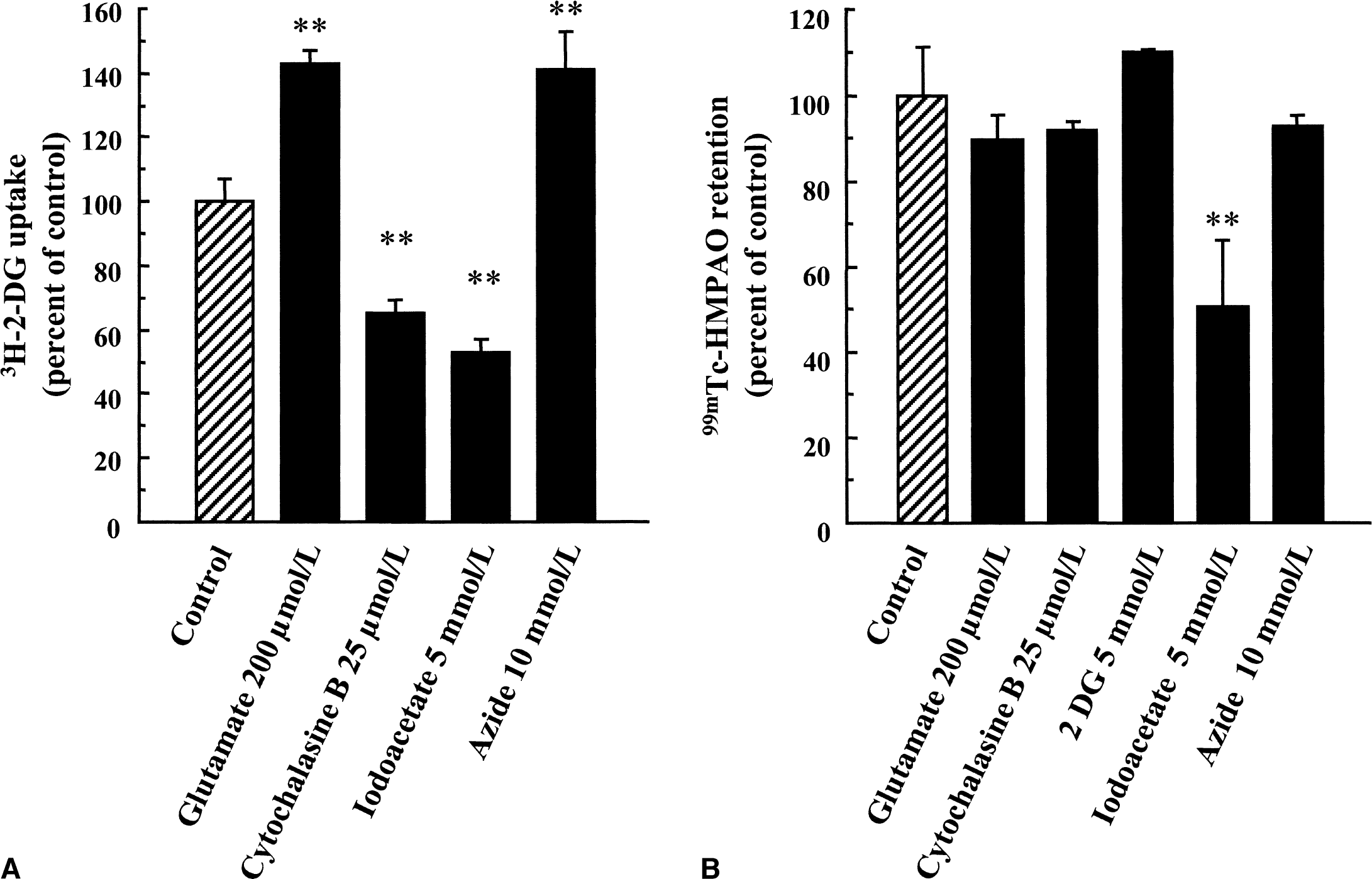
Effect of various agents on glucose utilization and 99mTc-HMPAO retention in cultured mouse cortical astrocytes. Astrocytes were exposed to each pharmacologic agent for 20 minutes during which [3H]–2-deoxyglucose uptake (2-DG)
DISCUSSION
99mTc-HMPAO is prepared as a neutral lipophilic complex that can be converted to hydrophilic derivatives consisting of a secondary complex, reduced hydrolyzed Tc99m, and pertechnetate (99mTcO4−) (Neirinckx et al., 1987). Although this conversion can take place spontaneously in vitro, its rate is low in aqueous media. Indeed, immediately after formulation, 85% to 93% of the complex is lipophilic, but more than 80% is still lipophilic after 1 hour in saline (Neirinckx et al., 1987; Hung et al., 1988). To verify the stability of the 99mTc-HMPAO complex under the authors' incubation conditions (37†C, 5% CO2) in culture medium (DMEM, pH 7.4), a rapid octanol extraction procedure was applied to evaluate the percentage of lipophilic form present after different times of incubation (Andersen et al., 1988a). Results show that the rate of degradation of the 99mTc-HMPAO complex is low within approximately the first half-hour, an observation consistent with previous results obtained in saline (Andersen et al., 1988a). Interestingly, the rate of degradation increases after 40 minutes, but the reduction of the lipophilic form observed at longer time periods does not differ appreciably from what has been described in a study in which the stability of the 99mTc-HMPAO complex was assessed in aqueous media for time periods longer than 30 minutes (Hung et al., 1988). Thus, the incubation conditions used in the current study do not appear to substantially affect the stability of the 99mTc-HMPAO complex as compared with standard conditions in aqueous media. This relative stability of the 99mTc-HMPAO complex in vitro needs to be contrasted with the rapid degradation occurring in vivo. Indeed, only 1% of the lipophilic complex remains 6 to 10 minutes after intravenous injection in human subjects (Nakamura et al., 1989). This extremely rapid conversion was explained by the passage of 99mTc-HMPAO into erythrocytes (60%) or its conversion in the plasma (40%) (Andersen et al., 1988a). Despite this instability in the circulation, appreciable amounts of 99mTc-HMPAO can be extracted by several organs including the brain. An average of 4% of 99mTc-HMPAO is trapped into human brain (Costa et al., 1985; Sharp et al., 1986), which is sufficient to allow image acquisition by SPECT. Similar results have been obtained in rats, where a mean of 2% of 99mTc-HMPAO is trapped in the brain (Nowotnik et al., 1985; Neirinckx et al., 1987). These observations suggest that despite the instability of the 99mTc-HMPAO complex observed in vivo in the blood, efficient uptake and retention mechanisms must be present within the brain parenchyma to account for the observed accumulation.
To gain insight about the contribution of the lipophilic form (vs. hydrophilic derivatives) to 99mTc-HMPAO uptake into brain cells, uptake in primary cultures of astrocytes was performed with decreasing proportions of the lipophilic form of 99mTc-HMPAO. Results show a parallel decrease in 99mTc-HMPAO uptake, thus suggesting that the lipophilic form is the major species taken up by cultured cells. This is consistent with the reported observation in brain slices that very little uptake was detected when no lipophilic form was present in the 99mTc-HMPAO complex (Ahn et al., 1994). In addition, Colamussi et al. (1999) have shown that in contrast with the 99mTc-HMPAO complex with a high proportion of the lipophilic form, the degradation product sodium pertechnetate (99mTcO4−) is not significantly taken up by cerebral cortical slices. These in vitro data on 99mTc-HMPAO uptake, which are also consistent with in vivo observations, support the use of in vitro systems, particularly brain cell cultures, as adequate models to investigate the cellular and molecular mechanisms involved in the retention of this purported cerebral blood flow marker.
Characterization of 99mTc-HMPAO uptake in primary cultures of mouse cortical astrocytes and neurons revealed that uptake is considerably greater into astrocytes than into neurons. Considering that astrocytes have processes that come in close contact with brain capillaries and cover almost their entire surface, it is not so surprising that these cells may constitute a prominent uptake site for a lipophilic marker penetrating the brain parenchyma through the circulation. A similar concept has been proposed, based on experimental data obtained in vitro and in vivo, for the uptake of deoxyglucose, another tracer commonly used in brain imaging (Magistretti and Pellerin, 1999). The uptake of 99mTc-HMPAO in brain cell cultures, which was rapid for the first 30 minutes, proceeded at a slower rate afterward and even reached a plateau in neuronal cultures. In addition, an important reduction in uptake was observed for both astrocytes and neurons when the experiment was performed at 4†C. A similar time and temperature dependency for 99mTc-HMPAO uptake has been reported for a preparation of rat brain astrocytes in culture (Costa et al., 1990). These observations suggest the involvement of a carrier-mediated process and/or an enzymatic reaction in the accumulation of 99mTc-HMPAO in brain cells. To investigate this aspect further, uptake with increasing concentrations of 99mTc-HMPAO was performed in cultured astrocytes but no saturation, indicative of the existence of a transporter and/or an enzyme-based accumulation, could be detected. Because of technical limitations, the concentrations tested were in the nanomolar range; therefore, the authors can not exclude the possibility of saturable sites at greater concentrations. This question thus remains unanswered.
In vivo, once 99mTc-HMPAO has been extracted by the brain, it was demonstrated that back diffusion into the circulation was very limited (Lassen et al., 1988). This suggested that 99mTc-HMPAO must be somehow retained within the brain parenchyma once it has crossed the blood-brain barrier. Washout experiments performed in astrocytes and neurons in culture demonstrated that a large proportion of 99mTc-HMPAO was retained within cells. These results are in agreement with previous studies showing that a majority of accumulated 99mTc-HMPAO is retained by cerebellar cultures of neurons (Suess et al., 1992) or by neocortical brain slice cultures (De Jong and Van Royen, 1989) after washout. As with the uptake, retention of 99mTc-HMPAO proved to be more important in astrocytes than in neurons. Over 95% of retained 99mTc-HMPAO complex in astrocytes and neurons was recovered as hydrophilic products. This is true not only in washout experiments but under all conditions studied. These data strongly suggest that an efficient process converts intracellularly the lipophilic form of 99mTc-HMPAO into its hydrophilic derivatives, thus providing an explanation for its retention not only in cultured cells, but also in vivo. In addition, the results indicate that the conversion process seems more efficient in astrocytes than in neurons.
On the basis of experiments performed in cell-free systems or in brain homogenates, it has been proposed that the mechanism responsible for 99mTc-HMPAO retention involves a direct interaction with glutathione (Neirinckx et al., 1988; Ballinger et al., 1988). Further supporting this hypothesis, it was reported that a correlation exists in vivo between the levels of glutathione in some brain tumors and their uptake of 99mTc-HMPAO measured with SPECT (Suess et al., 1991). It was thus interesting to examine the role of glutathione in the cell-specific retention of 99mTc-HMPAO. To this end, the effect of three different pharmacologic agents known to alter glutathione levels was studied. Results obtained with DEM, EA, and BSO on both glutathione levels and 99mTc-HMPAO retention in cultured astrocytes and neurons suggested a possible relation between intracellular levels of glutathione and the mechanism of retention. However, a closer analysis reveals some intriguing paradoxes. Indeed, in cultured astrocytes, DEM and EA caused a parallel and concentration-dependent decrease in both glutathione levels and 99mTc-HMPAO retention. In contrast, BSO, the inhibitor of glutathione synthetase, completely depleted glutathione at low concentrations (10 to 60 μmol/L) with no effect on 99mTc-HMPAO. In addition, only a moderate effect was observed at greater concentrations. The situation in neurons appeared even more complex. Both DEM and BSO decreased glutathione levels in a concentration-dependent manner. Their effect on 99mTc-HMPAO retention, however, was from small to moderate and did not vary within the concentration range chosen. On the other hand, ethacrynic acid induced a strong, concentration-dependent reduction of 99mTc-HMPAO retention in cultured neurons while having only a small effect on glutathione levels at the greatest concentrations. Such discrepancies also have been reported in vivo. DEM-treated mice and rats showed only a very small decrease in brain 99mTc-d,l-HMPAO retention as compared with controls, despite significant decrease in glutathione content (El-Shirbiny et al., 1989; Sasaki et al., 1996). Moreover, BSO-treated mice did not display a substantial reduction in 99mTc-d,l-HMPAO retention, whereas glutathione levels were significantly reduced (Sasaki and Senda, 1997). In view of these data, it appears unlikely that glutathione levels alone are the sole determinant of 99mTc-HMPAO retention within brain cells. Glutathione is known to act as a cofactor for different enzymes including glutathione peroxidase, an enzyme involved in free radicals detoxification (Dringen, 2000). As such, glutathione is involved in preventing oxidative damage and is one among many other factors that contributes to maintaining the oxidoreductive state of cells (Dringen, 2000). As suggested previously (Jacquier-Sarlin et al., 1996), other components influenced by the oxidoreductive state of the cells are likely to be involved in the retention of 99mTc-HMPAO. The current data are consistent with such an hypothesis, although they indicate that these components might differ between astrocytes and neurons.
It was proposed previously that metabolic alterations could influence 99mTc-HMPAO retention in brain cells (Ahn et al., 1994). Because astrocytes are known to play a crucial role in the regulation of brain energy metabolism (Tsacopoulos and Magistretti, 1996), experiments were conducted with a number of substances that activate or inhibit glucose metabolism in astrocytes, and 99mTc-HMPAO retention subsequently was evaluated. Despite significant effects on glucose utilization, results showed no significant effect on 99mTc-HMPAO retention. The only exception was iodoacetate, a classical inhibitor of glycolysis. However, in control experiments, the authors observed that iodoacetate also decreased glutathione levels. In view of the fact that 2-deoxyglucose, a compound that also blocks glycolysis, did not affect 99mTc-HMPAO retention, the authors conclude that the inhibitory effect of iodoacetate on 99mTc-HMPAO retention is because of its action on glutathione level. Thus, the current results are not consistent with two previous studies performed in rat brain cortical slices that suggested the involvement of the metabolic status of the cells in 99mTc-HMPAO retention (Ahn et al., 1994; Colamussi et al., 1999).
The use of 99mTc-HMPAO and SPECT to reveal cerebral blood flow abnormalities has been applied to a large number of pathologies including psychiatric disorders, neurodegenerative diseases, and brain tumors (Andersen, 1989). Either hyper- or hypoperfusion have been reported, for example, in stroke (Ogasawara et al., 2000), epilepsy (Paladin et al., 1999), ALS (Vercelletto et al., 1999), tumors (Rodrigues et al., 1993), AIDS (Christensson et al., 1999), or encephalitis (Launes et al., 1995). It has been assumed in most cases that alterations in 99mTc-HMPAO signal reflected blood flow changes. A few reports on specific pathologies—such as stroke, encephalitis, or brain tumors—have documented discrepancies between blood flow measured with the 99mTc-HMPAO SPECT technique and other imaging techniques, leading to the conclusion of either hypo- or hyperfixation of 99mTc-HMPAO in such selected cases (Rodrigues et al., 1993; Launes et al., 1995; Ogasawara et al., 2000). No satisfying explanation for these phenomena has been provided so far, but they have further suggested the existence of flow-independent mechanisms for 99mTc-HMPAO retention. Because the current data suggest that astrocytes might contribute significantly to 99mTc-HMPAO retention, it could be also possible that pathologic processes that affect glial cells could be responsible for hypo- or hyperfixation observed in such cases. For example, hypoperfusion detected with HMPAO SPECT in the prefrontal cortex has been reported previously in major depressive disorder patients (Galynker et al., 1998). In a recent study in which postmortem cell density counts were performed in the same area in patients also suffering from major depressive disorder, a reduction in the number of glial cells was demonstrated with no effect on neurons (Öngür et al., 1998). In addition, in the case of patients suffering from acute herpes encephalitis, an hyperfixation of 99mTc-HMPAO was reported in parallel with a neuronal loss, and the authors have concluded that the increased retention was not caused by increased neuronal activity but was rather linked to inflammation-related effects on glia (Launes et al., 1995). These observations thus point at a possible relation between glial cell alterations and flow-independent changes in 99mTc-HMPAO SPECT signal that could be relevant in certain pathologies.
In summary, the results reported here provide evidence that astrocytes could contribute significantly to 99mTc-HMPAO retention in the brain, as detected by SPECT. In addition, glutathione appears to be only one of the possible factors implicated in the mechanism responsible for 99mTc-HMPAO retention within brain cells, and other factors that are likely to be different between astrocytes and neurons remain to be identified. Finally, the current study proposes the possibility that glial cell alterations could provide an explanation for unusual hyper- or hypofixations of 99mTc-HMPAO occurring in certain pathologies.
Footnotes
Acknowledgments:
The authors thank Prof. Angelika Delaloye, Head of the Department of Nuclear Medecine, Centre Hospitalier Universitaire Vaudois, Lausanne, Switzerland for making available facilities for 99mTc-HMPAO preparation and Mrs. Monique Bezen¸on for technical assistance. The authors also thank Dr. Ralf Dringen, Tübingen, Germany for his advice with glutathione measurements.
