Abstract
In patients with thrombotic stroke, the occluded artery often reopens over time. This results through a natural dissolution of the occluding material, and fragments of the material may move downstream to obstruct distal arteries. The current study was undertaken to investigate the patency of brain microvessels at varying time intervals after injection of a preformed clot into the right internal carotid artery of rats. Cerebral microvessels in brain sections were visualized using immunohistochemistry for fibronectin (detecting existing microvessels) and Evans blue (visualizing perfused microvessels). The percentage of patent microvessels was calculated as the number of Evans blue-positive microvessels divided by the number of fibronectin-positive microvessels. In normal control animals, results showed that 98% ± 3% (mean ± SD) of microvessels in the cortex and 94% ± 14% in the striatum were patent. In the ischemic animals, immediately after clot injection, microvessels in the cortex and striatum were occluded, mainly in the territory irrigated by the middle cerebral artery. One hour after clot injection, microvessels had reopened in most of the cortex but remained occluded in some portions of the striatum, possibly as a result of downstream movement of fragments formed from the original clot. By 3 hours after clot injection, microvessels in the cortex were patent in all animals, whereas in the striatum microvessels were patent in 50% of the animals. In the other 50%, small striatal perfusion deficits persisted. At 24 hours after clot injection, microvessels were patent in both the cortex and striatum of all animals except one. These findings suggest that intracerebral clots dissolve spontaneously in a relatively short period of time, but that fragments formed from the clot may obstruct more distal blood vessels. It is likely that clot fragments lodge in arteries with lower blood flow and poor collateral perfusion, where they continue to cause ischemia for a longer duration. These results may in part explain the resistance of the striatum to neuroprotective strategies used for the treatment of focal cerebral ischemia.
In patients with stroke, most ischemic episodes occur as a result of occlusion of the middle cerebral artery (MCA), and the duration of blood flow interruption is the most important determinant of infarct size. Thrombolysis results in an improvement in clinical outcome when used in patients and in experimental models of focal cerebral ischemia. After it was reported that tissue plasminogen activator (t-PA) was effective in opening coronary arteries, its usefulness in restoring cerebral blood flow was tested. Administration of t-PA 15 minutes after ischemia to rats with embolic occlusion produced a highly significant reduction in infarct volume and an improvement in functional recovery. This effect was also observed when treatment was delayed to 120 minutes after ischemia, but no further improvement was observed in rats when treatment was delayed for more than 240 minutes (Overgaard et al., 1994). Further, only a fraction of stroke patients benefit from t-PA treatment (Gress, 1994). In these patients, reperfusion in most instances is only partial (Baird et al., 1994). One likely explanation for the partial reperfusion is the downstream displacement of the clot material or its fragments.
The results of angiography studies also support this hypothesis. Previous studies have demonstrated that within the first hour after the onset of symptoms, 80% of patients with an acute ischemic stroke showed arterial occlusion. These rates decreased to 20% by 24 hours after stroke (Gress, 1994), suggesting that cerebral thrombi do fragment and dissolve naturally.
Moreover, in a recent animal study of embolic stroke, t-PA treatment quickly improved blood flow, followed by a continuous increase in perfusion for 40 minutes throughout the MCA territory (Busch et al., 1998). A secondary decrease of perfusion in the center of the lesion occurred thereafter, whereas perfusion in the border zone improved further. The results of that study suggested that the incomplete recovery in the lesion core is due to restricted reperfusion, induced by downstream displacement of the clot (Busch et al., 1998). Understanding the mechanisms of clot dissolution may elucidate factors to improve patient outcome after ischemic injury.
In the current study, our model exploits the innate mechanism of intravascular clot dissolution to test the hypothesis that the fragments formed from the clot obstruct downstream blood vessels and to characterize microvessel patency in the ischemic territory. Observations were made in two areas of the brain, the cortex and striatum, because the microvessels in these two areas were expected to occlude by the clot injected.
MATERIALS AND METHODS
Male Wistar rats, weighing 300 to 350 g, were purchased from Charles River (St. Constant, Canada). The rats were housed in a 12-hour light/dark cycle and had free access to water and food. Animal care and the general protocols for animal use were approved by the Animal Ethics Committee of the University of Alberta.
Animal model
Embolic focal cerebral ischemia was induced as previously reported (Wang et al., 2000; Zhang et al., 1997). In brief, rats were initially anesthetized with 3.0% halothane and then maintained with 1.5% halothane in a mixture of O2 and NO2 during surgery. A longitudinal incision 1.5 cm long was made in the midline of the ventral cervical skin. The right common carotid artery (CCA), right internal carotid artery (ICA), and right external carotid artery (ECA) were exposed. The distal portion of the ECA was ligated and cut. A modified PE-10 catheter (Becton Dickinson, Sparks, NJ, U.S.A.) filled with bovine thrombin (Thrombostat; Warner-Lambert, Scarborough, Canada) was introduced into the lumen of the right ECA through a small puncture. Five microliters of blood was withdrawn into the catheter and retained for 15 minutes to allow clot formation. Once the clot formed, the catheter was advanced 17 mm in the ICA until its tip was 1 to 2 mm away from the MCA origin. The preformed clot in the catheter was then injected, the wound closed, and the animal returned to its cage. The surgery was typically completed in 30 minutes. Figure 1 shows the clot in the ICA and MCA immediately after clot injection.
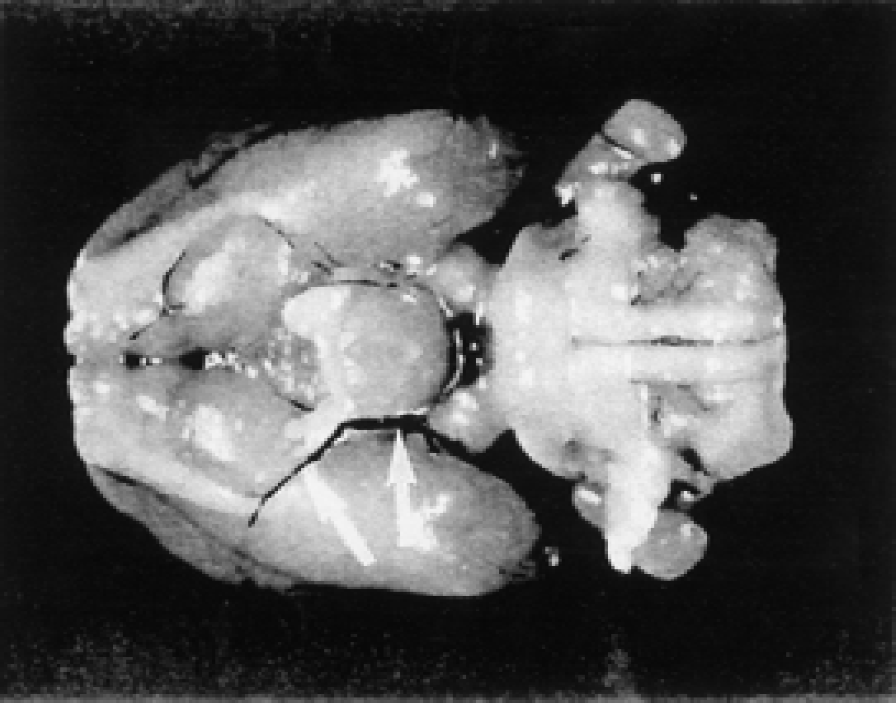
Immediately after clot injection, clot material is observed in the internal carotid artery and middle carotid artery stem (arrows).
Behavioral tests
Behavioral deficits and seizure activities were recorded at 2 hours after the clot injection in two groups of rats killed at 3 and 24 hours after clot injection.
Neurologic deficits were determined using a modified Bederson's scoring system (Bederson et al., 1986; Yang et al., 2000): 0, no observable deficit; 1, forelimb flexion; 2, forelimb flexion plus decreased resistance to lateral push; 3, unidirectional circling; 4, unidirectional circling plus decreased level of consciousness. Seizure activity was scored with a modified classification of the Racine score (Morimoto et al., 1997): 0, no seizure was observed; 1, rhythmic mouth and facial movement; 2, rhythmic head nodding; 3, forelimb clonus; 4, rearing and bilateral forelimb clonus; 5, rearing and falling.
Immunohistochemistry
Perfused and existing microvessels were visualized with a double staining procedure using Evans blue dye and antifibronectin antibody (Gobel et al., 1990). In anesthetized animals, Evans blue (E-2129, Sigma, Oakville, ON, Canada; 2% solution in saline, 0.2 mL/100 g body weight) was injected into the right jugular vein and allowed to circulate for 10 seconds. The animals were then decapitated and their brains removed. The brains were frozen in 2-methylbutane kept in dry ice, embedded in optimal cutting temperature (OCT) medium, and stored at −70†C until analysis. The brains were sectioned at 10 μm in thickness with a cryomicrotome beginning 3.7 mm anterior to the bregma. For each rat, nine consecutive sections were collected at an interval of 1 mm and stored at −70†C. To visualize Evans blue staining, the slides were transferred to acetone at −20†C for 30 seconds and then rinsed in acetone at room temperature for 30 seconds. The perfused microvessels (intraluminal Evans blue) in each section were inspected with fluorescent microscopy. In each rat, the middle five sections, starting from 1.7 mm anterior to the bregma, were photographed with Image-Pro Plus system (Media Cybernetic, Silver Spring, MD, U.S.A.). Specifically, the section was observed under regular light and three sample locations in the cortex of each hemisphere (dorsal, lateral, and ventral) were selected and their x-y coordinates on the mechanical stage were recorded. Three sample locations were also selected in the striatum (caudate putamen) in a similar fashion, and then a microphotograph was taken under fluorescent light according to the coordinates recorded.
For each area investigated, the x-y coordinates on the mechanical stage were again documented, and these coordinates were used for taking a second photograph after immunohistochemistry. The sections were then transferred into a humid chamber for 48 hours. This causes a gentle hydration and a homogeneous distribution of the Evans blue throughout each section. The same sections in which perfused microvessels were inspected were then immunoreacted with polyclonal rabbit antihuman fibronectin antibody (1:100, Dako, Mississauga, ON, Canada). After three washes in phosphate-buffered saline, the sections were incubated with fluoroscein isothiocyanate (FITC)-conjugated swine antirabbit immunoglobulin (Sigma) as the secondary antibody (1:60) for 30 minutes. The slides were then washed in phosphate-buffered saline and photographed by fluorescent microscopy. The two photographs (Evans blue and fibronectin staining) were then overlaid, and the numbers of perfused and existing microvessels were counted (Fig. 2). The percentage of patent microvessels was taken to be the number of Evans blue-positive microvessels divided by the number of fibronectin-positive microvessels. The sample area for each site was 0.1 mm2, and vessels with a diameter larger than 15 μm were not counted.
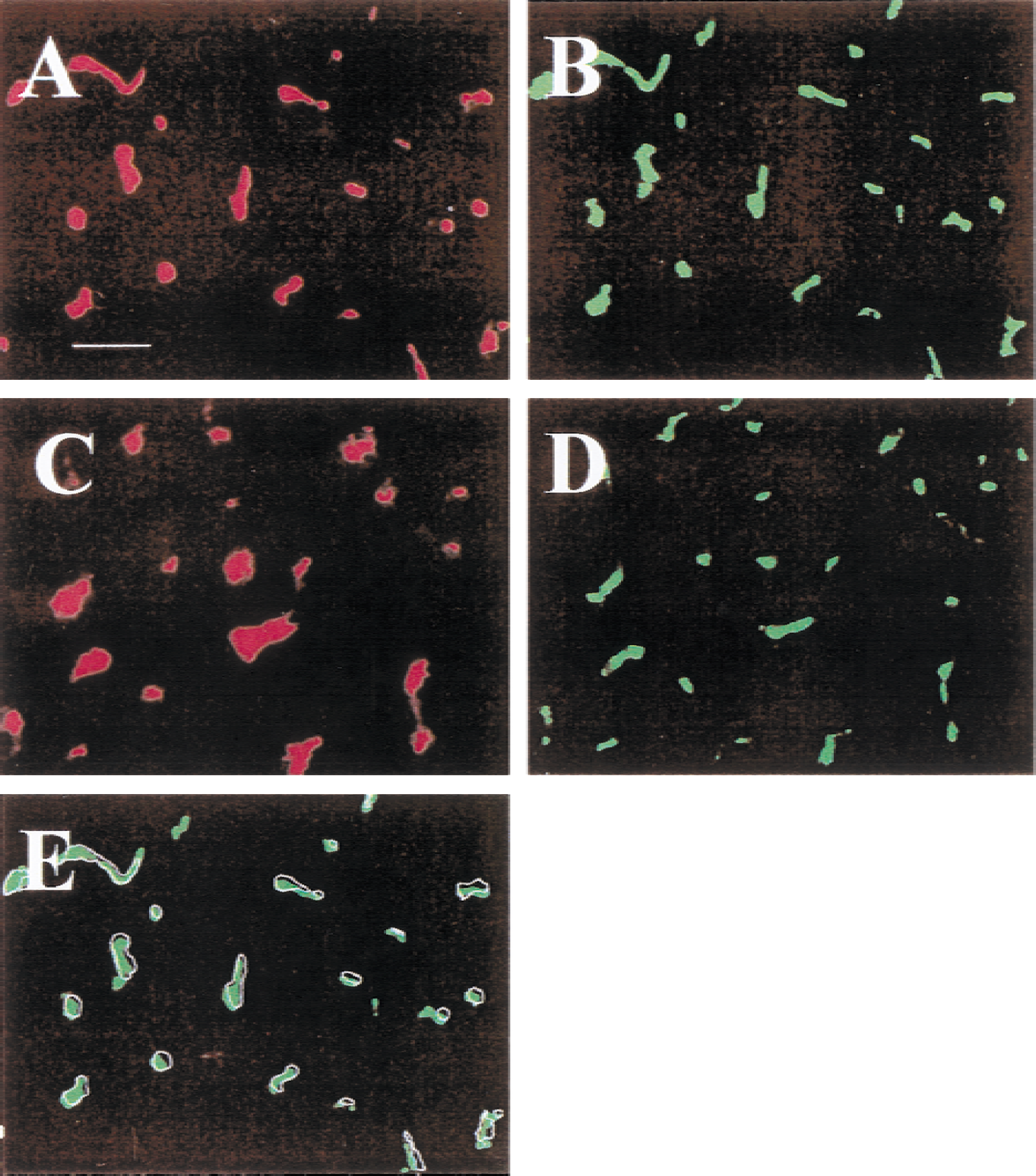
Microvascular patency in the cerebral cortex (
Experimental design
The study consisted of five groups, to which animals were randomly assigned. In the normal control group, no clot was injected (n = 3). In the ischemic groups, rats were killed at 15 minutes (n = 6), 1 hour (n = 6), 3 hours (n = 6), or 24 hours (n = 6) after clot injection.
Statistical analysis
The percentages of patent microvessels were ranked and analyzed with the Kruskal-Wallis test. Where main treatment effects were reached, comparisons between groups were run using the Mann-Whitney test (two-tailed). The rate of seizure occurrence was analyzed with the chi-square test. Differences were considered significant when P < 0.05.
RESULTS
Behavioral deficits and seizure activity were recorded 2 hours after clot injection in two groups of rats that were killed at 3 and 24 hours after clot injection. The behavioral deficit score was 3 ± 0.8 (mean ± SD) in the 3-hour group and 3 ± 1.2 in the 24-hour group. Seizures occurred in two rats in the 3-hour group (scores of 2 and 5) and three rats in the 24-hour group (scores of 2, 5, and 5). The rates of seizure occurrence between these two groups were not significantly different (P > 0.05).
Normal control and contralateral hemisphere to ischemia
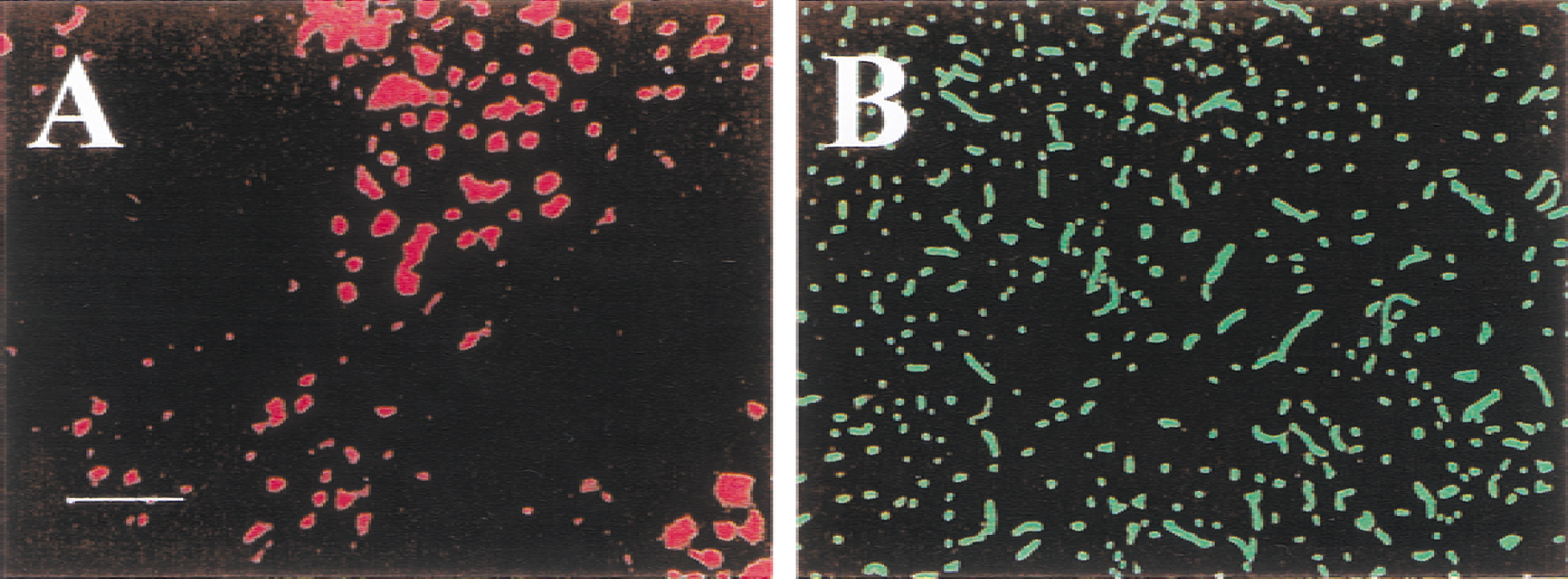
In normal rats, there were no significant differences in microvessel patency between the left and right hemispheres. Therefore, the data from the both hemispheres were pooled, and the resulting percentage of patent microvessels was 98% ± 3% in the cortex and 94% ± 14% in the striatum. Figure 2 shows the local microvessel pattern from a brain section after fluorescent double staining of the perfused and existing microvessels in a normal control rat. In the animals with embolic ischemic injury, no perfusion deficit was observed in the contralateral hemisphere. Statistical analysis showed that microvessel patency in the contralateral hemisphere in ischemic animals was not significantly different from that in the normal control rats at any time point considered.
Fifteen minutes after clot injection
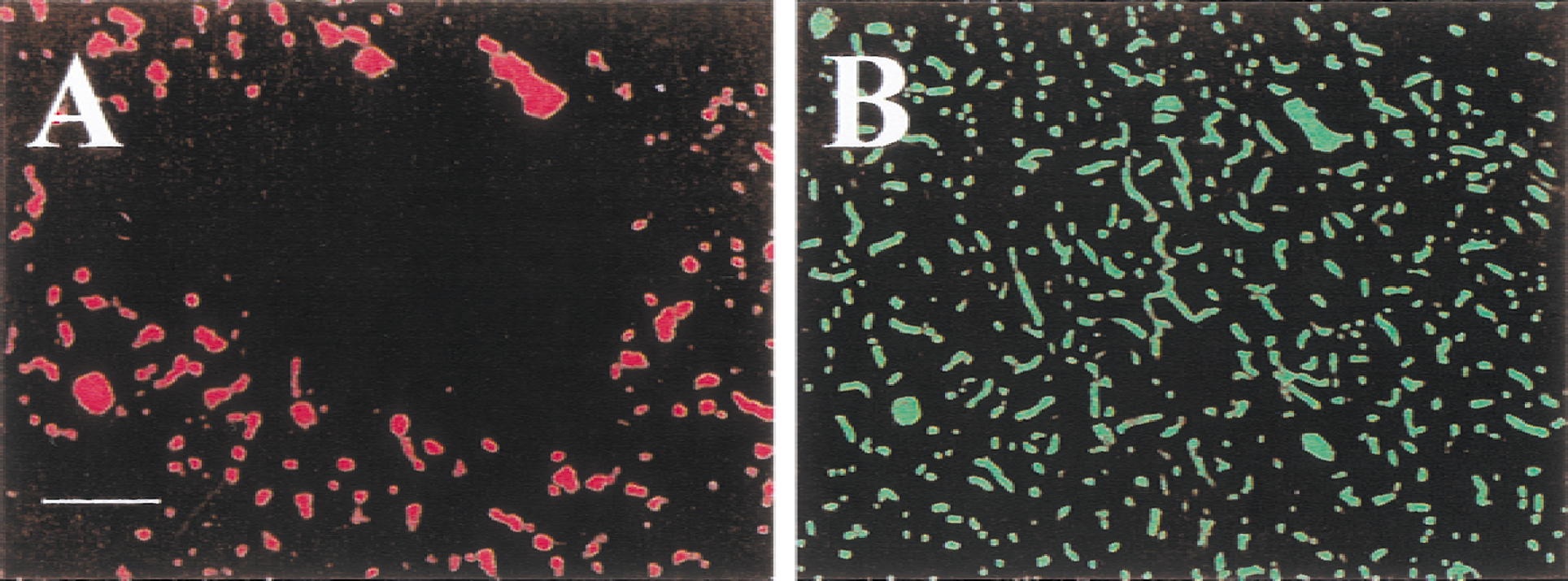
Perfusion deficits were observed in the areas supplied by the MCA in the ipsilateral hemisphere, mainly in the cortex and striatum (Fig. 3), and in areas supplied by the posterior striate arteries that also originate from the ICA. The shape of the perfusion deficit was irregular, and the size of the perfusion deficit varied from rat to rat. In the cortex, large perfusion deficits were evident in most of the ipsilateral cortex in three of six rats, and in the entire striatum.
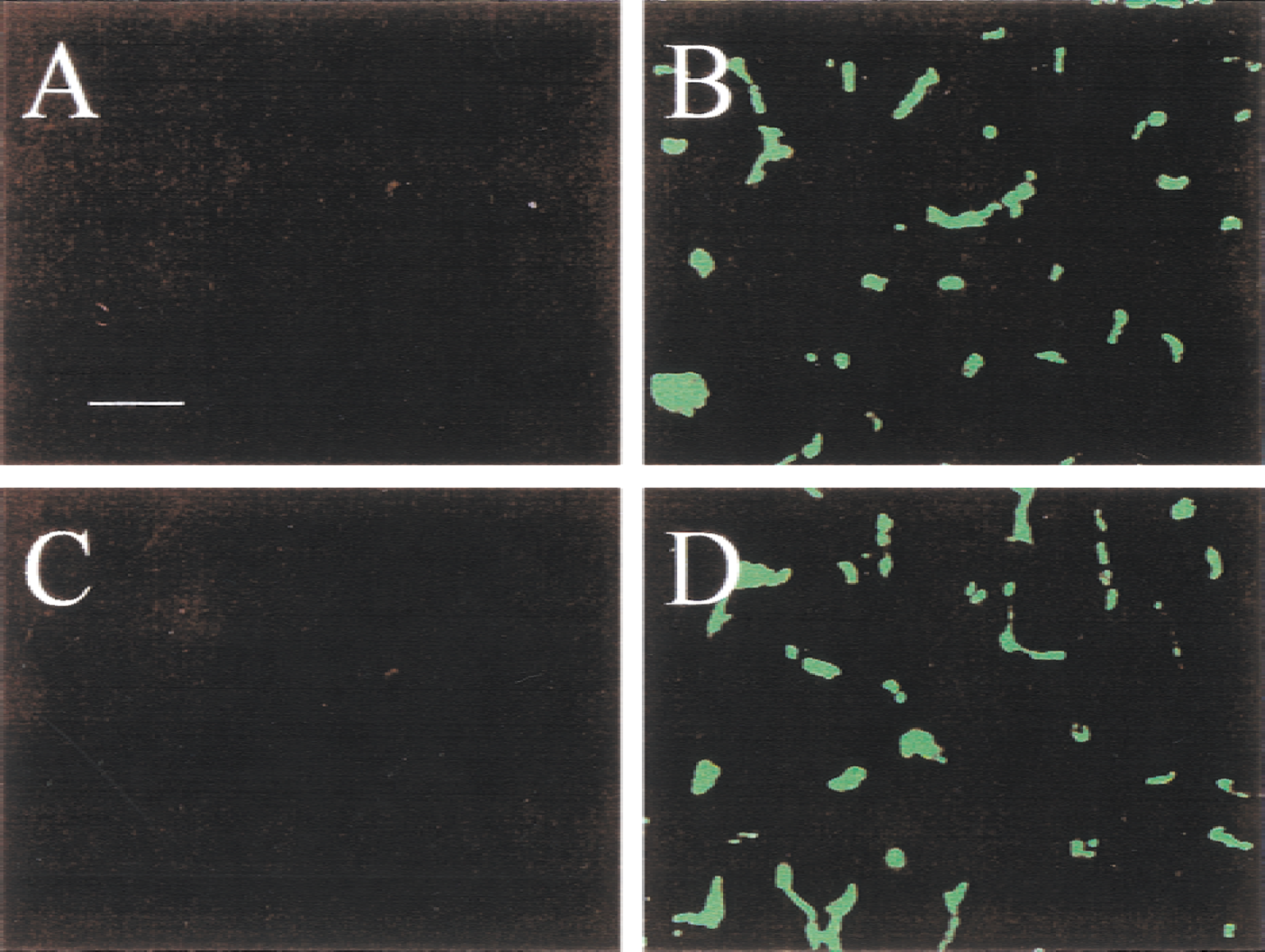
Microvascular patency in the cerebral cortex (
In these rats, the microvessels were perfused only in the most rostral and caudal cortical poles and in the medial part of the cortex. In one of six rats, a perfusion deficit was observed in the middle region of the cortex; this region occupied roughly half of the ipsilateral cortex. The perfusion deficit in this rat also occupied almost the entire striatum. In the remaining rats, perfusion deficits were observed in the middle-caudal aspect of the ipsilateral cortex, with only the ventral aspect affected. However, a perfusion deficit was observed in most of the striatum. The boundary of perfused and nonperfused regions usually was very clear (Fig. 4). In the center of the perfusion deficit, no perfused microvessels were detected. An area containing both perfused and occluded microvessels was observed, but only at the boundary of the territory supplied by the occluded artery.
Microvascular patency at the boundary of the perfusion deficit in the cerebral cortex immediately after clot injection. A region containing both perfused and nonperfused microvessels (arrows) is seen at the border of the perfusion deficit.
One hour after clot injection
In the ipsilateral hemisphere, a perfusion deficit was located in the caudal portion of the striatum (Fig. 5) and was also observed in the amygdala in four of six rats. Except for a small region surrounding the amygdala, most of the cortex was reperfused. In the region of perfusion deficit, small perfused strips were often observed, particularly in the border of the perfusion deficit (Fig. 6). In one rat a perfusion deficit shaped like a dumbbell was observed in the rostral-lateral aspect of the striatum, external capsule, and cortex. The body of the dumbbell was located in the external capsule. In one rat from this group, both the cortex and striatum were reperfused.
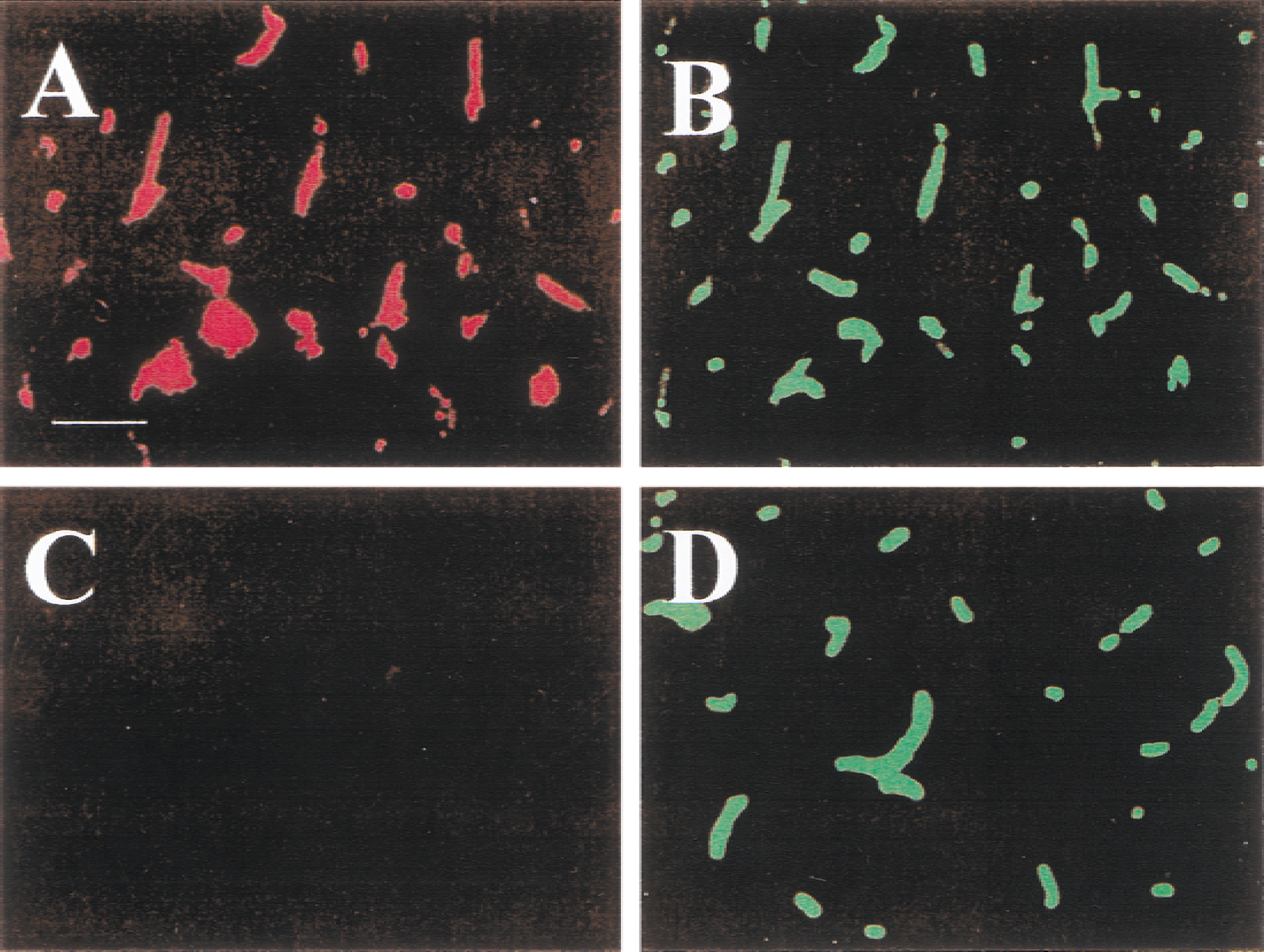
Microvascular patency in the cerebral cortex (
Microvessel patency at the border of the ischemic zone in the striatum 1 hour after clot injection. An area with perfused microvessels extends into the perfusion deficit.
Three hours after clot injection
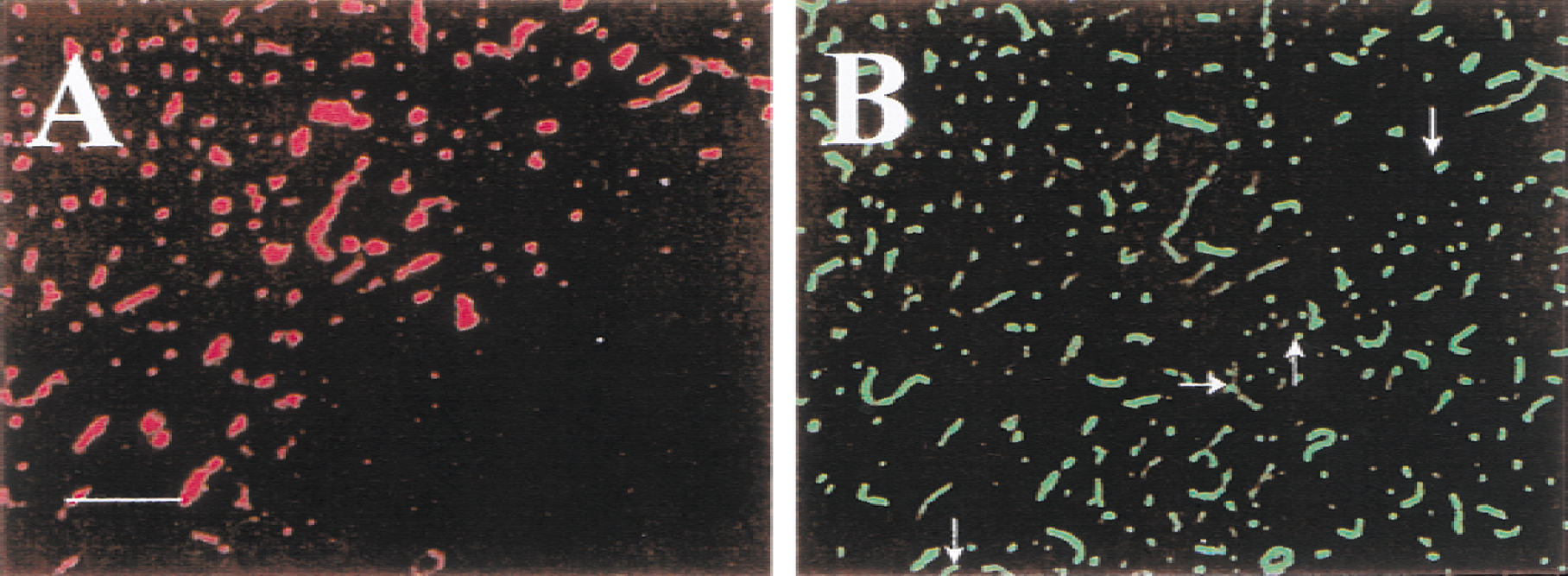
In the ipsilateral hemisphere, microvessels in both the cortex and striatum were patent in three of six rats. In the remaining three rats, the microvessels were reperfused in the cortex. However, small perfusion deficits were observed in the striatum. In one rat, two perfusion deficits appeared in the rostral portion of the striatum. One was located in the dorsal portion of the striatum (Fig. 7) and another in the ventral portion. In another rat, a perfusion deficit was observed in the medial-caudal portion of the striatum. In the third rat, the perfusion deficit was in the most caudal portion of the striatum; this perfusion deficit also affected the amygdala.
Three small perfusion deficits were observed in the striatum 3 hours after clot injection. One perfusion deficit is in the center of the photomicrograph; two others are seen in the upper right corner and the lower left corner of
Twenty-four hours after clot injection
Microvessels were patent and blood flow was reestablished in the cortex and striatum in five of six rats (Fig. 8). In one rat, a perfusion deficit was observed in the caudal portion of the striatum of the hemisphere ipsilateral to the occluded vessel. Intravascular blebs were observed in the brain sections examined. The blebs appeared as an Evans blue-void area surrounded by positive Evans blue staining. In normal animals, intravascular blebs were rarely observed in the microvessels. In contrast, blebs appeared in both the ischemic and nonischemic hemispheres of the rats receiving thrombotic clots. Intravascular blebs were observed only in the Evans blue section, but not in sections with antifibronectin staining. The intravascular blebs have been detailed previously (Chiang et al., 1968).
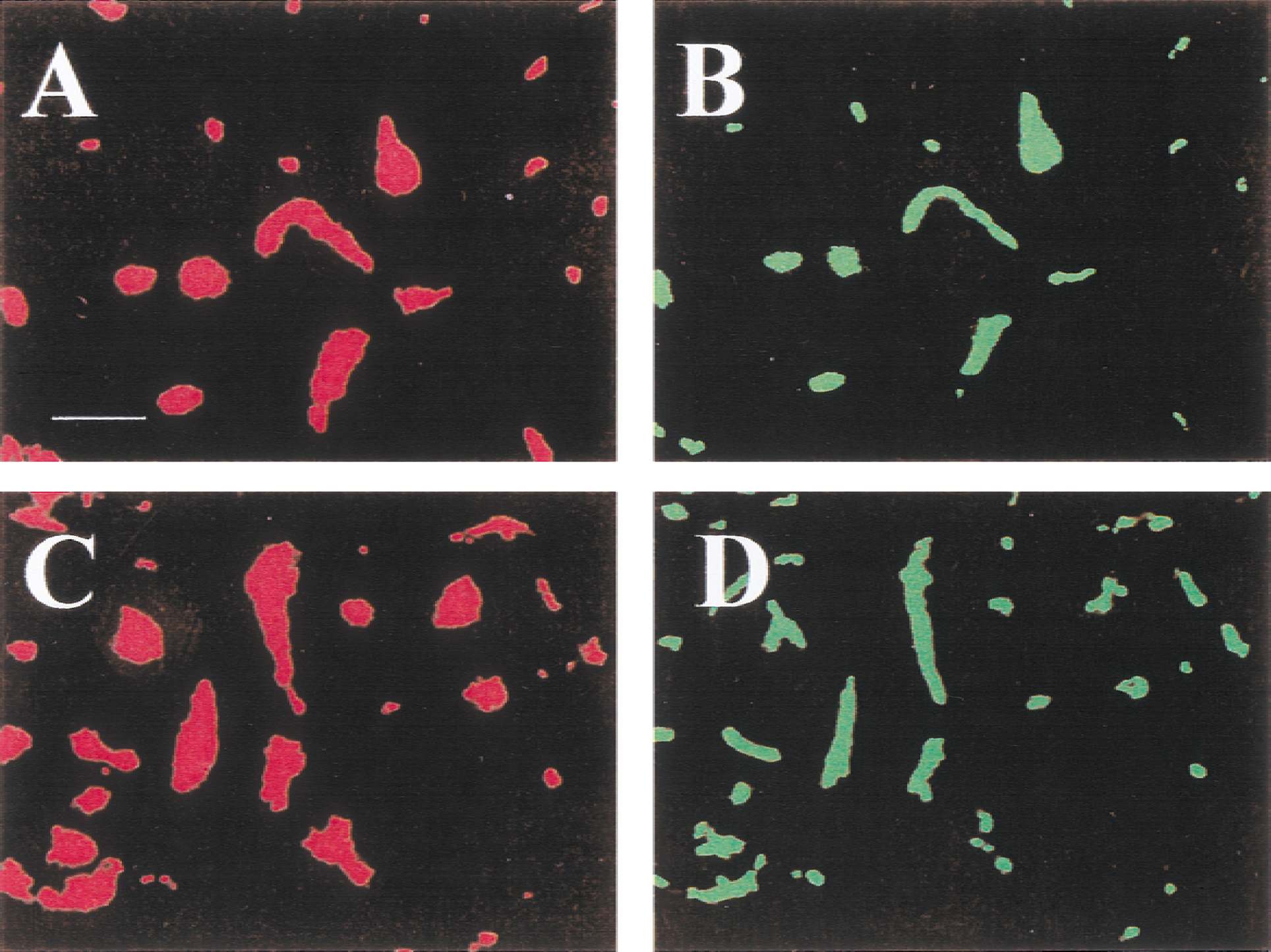
Microvascular patency in the cerebral cortex
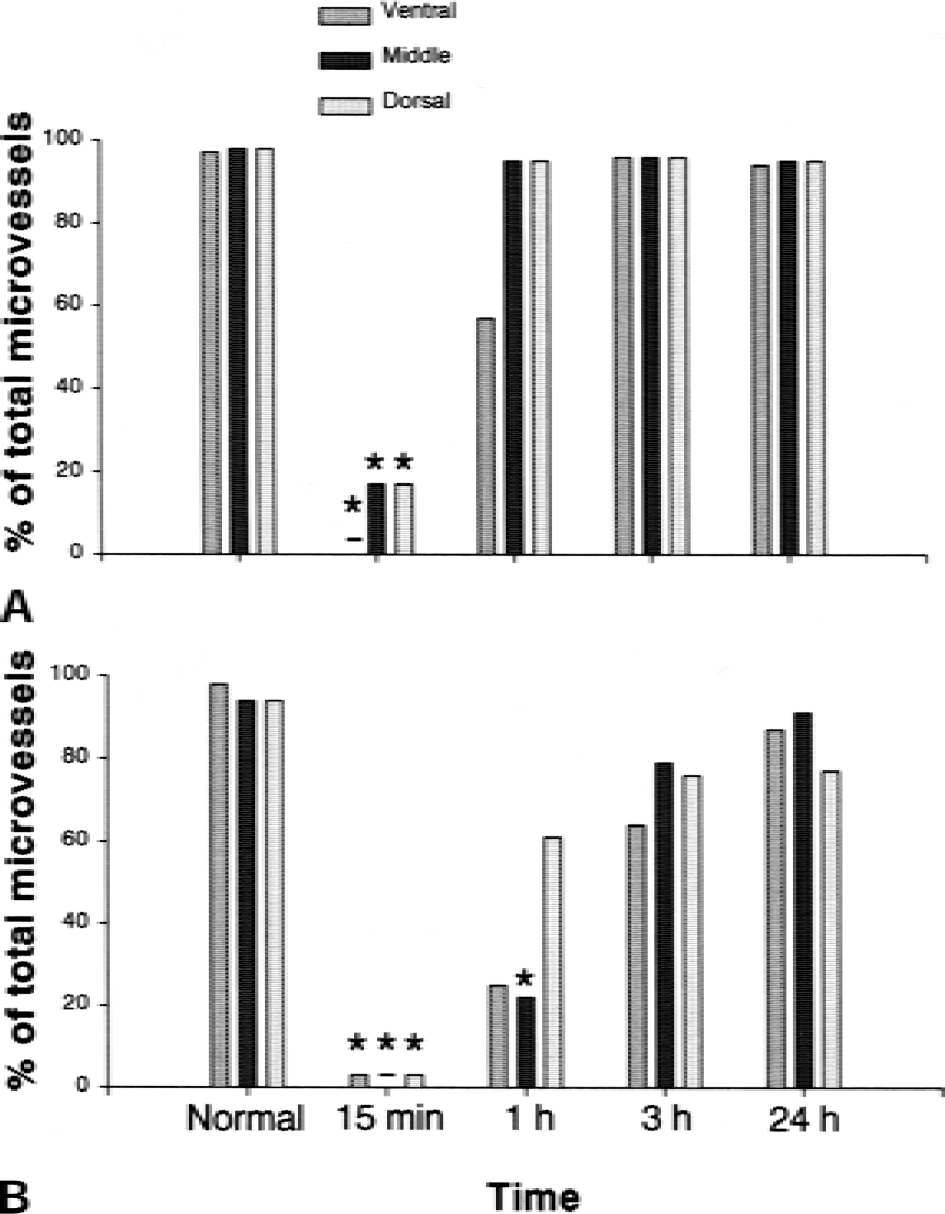
Figure 9A summarizes the microvessel patency in the cortex of the injured hemisphere. Fifteen minutes after clot injection, the microvessels were completely occluded in the ventral aspect, and only 15% and 20% of the microvessels were still open in the middle and dorsal regions, respectively. Microvessel patency was significantly lower in all three regions in the ischemic rats compared with normal controls (P < 0.05). At 1 hour after clot injection, 60% of the microvessels were reopened in the ventral region (not significantly different from the normal control group), and microvessels were reopened completely in both the middle and dorsal regions. Three and 24 hours after clot injection, differences of microvessel patency were not significant in all three regions of the cortex compared with those of the normal controls. Figure 9B shows microvessel patency in the striatum. Microvessels were completely occluded immediately after the clot injection in all three regions. One hour after clot injection, 22% of the microvessels were reopened in the middle striatum, significantly less than in the normal control group (P < 0.05). In the ventral and dorsal striatum, 25% and 60% of the microvessels were reopened, respectively, 1 hour after clot injection; these percentages were not significantly different from those in the normal controls. Three and 24 hours after clot injection, the microvessel patency did not differ significantly from that observed in the normal controls.
Summary of microvascular patency in the ischemic cerebral cortex
DISCUSSION
Our results show that shortly after injection of an autologous clot, there is microvessel occlusion located predominately in the territory supplied by the ipsilateral MCA. The clot injected is dissolved naturally, and this dissolution occurs in a relatively short period. Within a few minutes after clot injection, a large perfusion deficit appeared in the cortex and striatum. One hour after clot injection, microvessels in most of the cortex had been reopened, but there were partial perfusion deficits in the striatum. By 3 hours after clot injection, the microvessels were reperfused in the cortex. However, small perfusion deficits were still observed in the striatum in half the animals. Twenty-four hours after clot injection, all microvessels were reperfused and blood flow was reestablished in both the cortex and striatum in all rats except one. The rapid fragmentation and dissolution of the cerebral embolic materials after injection may be controlled mainly by fibrinolytic enzymes present within the embolic mass. Tissue plasminogen activator produced from endothelial cells may also contribute to the breakdown of the clot material.
Our results support the hypothesis that the debris generated from dissolution of the clot moved to peripheral arteries and occluded downstream blood vessels. It is possible that the clot fragments blocked the microvasculature directly. A second possibility is that instead of blocking the microvasculature, the clot fragments blocked feeding flow from more proximal arteries and resulted in perfusion deficits. At 1 hour after clot injection, the perfusion deficits disappeared from most of the cortex and the striatum was also partially reperfused. In the cortex, reperfusion was not homogenous: it occurred earlier in the dorsal aspect than in the ventral aspect. This indicates that some arteries were reopened but others remained occluded. Because the occluded vessels were downstream from the original site of clot formation, it is likely that clot fragments lodge in more distal striatal arteries with poor collateral flow, where they may remain undissolved for a longer time. This supposition agrees with previous findings that cortical arterial collaterals exist by means of leptomeningeal anastomoses and distal collaterals from three major cerebral arteries (Coyle and Jokelainen, 1982), whereas arterial collaterals in the basal ganglia are poorly developed. It may also be that the speed of blood flow in one region is greater than that in other regions of the brain as a result of anatomic differences.
Thus, the clot fragments move away more easily from some regions of the brain than from the others, as observed in the cortex and striatum. These reasons may also explain why the clot fragments were removed more rapidly in the dorsal aspect than in the ventral aspect of the cortex. The clots in the MCA and ICA were inspected visually only in two rats from the 15-minute and 3-hour groups and one rat from the 1-hour and 24-hour groups (data not shown). Clots were found in the rats from the 15-minute group, and no clots were found in all other rats, although perfusion deficits persisted in the 1-hour and 3-hour group rats. These results also support the idea that clot fragments may block the downstream arteries. If the hypothesis that the clot fragments occlude the downstream microvasculature was correct, observations from sequential brain sections should provide useful information. In the current protocol, we used Evans blue dye to evaluate vessel occlusion. This requires thin tissue sections for evaluation of vessel occlusion. Tracking of clot movement would, however, require sequential thick tissue sections; this was not possible because it would have made evaluations with Evans blue staining impossible.
In the reperfusion model of surgical occlusion of the common carotid artery, swelling of perivascular glia is the major cause of microvascular compression and the “no flow” phenomenon. In the embolic model used in this study, recanalization by natural thrombolysis is a slower process compared with the sudden onset of reperfusion after removal of the clamps from the arteries. The induction of low-pressure reperfusion during thrombolysis, together with the compression of the blood vessels by perivascular edema (Shigeno et al., 1985), and obstruction of vessel lumen by endothelial blebs may provide an excellent environment for the entrapment of debris formed from the thrombosed clot. The reperfusion may also result from collateral flow, particularly in the cortex. It is also possible that some secondary thrombi may not be detected with the procedures used in this study.
The occlusion of downstream branches by the clot fragment is supported by other studies. In animal models of thrombotic embolization, angiography has demonstrated occlusion in the ICA immediately after clot injection into the ECA. However, the occlusion moved to the MCA branches 3 hours after clot injection (Overgaard et al., 1994). Similar results were also found by Kaneko et al. (1985). Clinically, migration and fragmentation of microthrombi have been documented in the retinal circulation, which is implicated in the pathogenesis of transient ischemic attacks (Fisher, 1959). The disappearance of small vessel occlusions in patients with stroke has also been reported (Dalal et al., 1965). Measurements of cerebral blood flow after thrombolysis also support this idea. In an embolic stroke model, thrombolysis successfully recanalized the embolized MCA origin and led to gradual improvement of blood flow in the periphery of the ischemic territory, but only transient and partial improvement in the lesion center was observed (Busch et al., 1998). In patients with stroke, regional cerebral blood flow was measured in five clinical cases by single-photon emission computed tomography after thrombolysis (Herderschee et al., 1991). With the exception of one patient, measurements remained abnormal with further deterioration in two patients.
In our previous studies, injection of clot resulted in pathologic damage to the cortex and striatum (Wang et al., 2000). 2,3,5-triphenyltetrazolium chloride (TTC) staining showed that without treatment, the infarct volume occupied 23% of the ipsilateral hemisphere on day 3 after clot injection. In the current study, immediately after clot injection, the microvessels were closed in almost the entire hemisphere in three of six rats. A relatively large area of perfusion deficit also appeared in the rest of the rats. Thus, the area of perfusion deficit immediately after clot injection is much larger than that of infarction 3 days later. This indicates that the tissues in the periphery of the perfusion deficit were still functional, and that they recovered when blood flow was reestablished. In contrast, a previous study using TTC staining (Wang et al., 2000) showed that a central core lesion remained even though, as the current results suggest, recanalization occurs as early as 1 hour after clot injection. Together, these data imply that delayed recanalization does not necessarily improve the pathologic outcome. These findings are also consistent with those reported previously (Lee et al., 1989; Phillips et al., 1989).
A double staining procedure was used, with Evans blue dye used to detect perfused microvessels and fibronectin antibody used to examine existing microvessels in the cortex and striatum. This procedure can accurately locate the site and extent of the perfusion deficit and allows visualization of individual microvessels. However, there are limitations in the techniques we used. First, the clot was dissolved in a relatively short period after it was injected. One explanation is that a fresh clot was injected, and this fresh clot was easily dissolved. Another concern is that with the double staining procedures, the perfusion deficits induced by the blood vessel occlusion could be detected, but the occlusion material itself could not be visualized. Our protocol cannot exclude the possibility that endogenous microthrombi formed in the more distal arteries may contribute to the secondary occlusions. Further studies are underway to examine these issues and extend the current findings by injecting old and/or labeled clots.
Postmortem studies of patients with stroke have shown the presence of microthrombi in ischemic areas (Heye et al., 1992). The presence of fibrin in the thrombi indicates that they are formed after primary arterial occlusion. Opening, either spontaneous or through thrombolysis, of the originally occluded arteries restores blood flow to downstream arterioles and small arteries and may cause secondary emboli. The current data support this hypothesis, and it is likely that the clots, before complete dissolution, are fragmented by natural thrombolysis and flushed downstream. These secondary microthrombi cannot be easily reached by t-PA and may cause secondary tissue damage during reperfusion. Therefore, secondary ischemia is an important concern, and further investigation is needed to develop effective strategies for the treatment of thrombotic stroke.
Footnotes
Acknowledgments:
The authors thank Brian Tancowny for his technical support and Asim Raja for reading the manuscript.
