Abstract
Retrograde tracing and immunohistochemistry was used in rats to investigate whether the ganglia in the cavernous sinus contribute to cerebrovascular innervation. The cavernous sinus ganglia in rat include the cavernous part of the pterygopalatine ganglion (PGC) and small cavernous ganglia (CG). The tracers, fluorogold and fast blue, were applied to the middle cerebral artery in eight rats. After 1 to 4 days, the cavernous sinuses were dissected out and studied as whole mount preparations and sections. A moderate number of labeled neurons were visible in the ipsilateral PGC and CG. Furthermore, fibers in the cavernous nerve plexus and abducens nerve were labeled, suggesting that the pathway from the cavernous sinus ganglia to the cerebral arteries runs through the cavernous plexus and then retrogradely along the abducens nerve to the internal carotid artery. Selected sections were immunohistochemically stained for the cholinergic marker, vesicular acetylcholine transporter (VAChT). Most cells in the PGC and CG were VAChT-immunoreactive, some of which also contained tracer. It is concluded that in rat, the cavernous sinus ganglia, consisting of the PGC and small CG, contribute to parasympathetic cerebrovascular innervation and that the cavernous nerve plexus and abducens nerve are involved in the pathway from these ganglia to the cerebral arteries.
Keywords
Basal cerebral arteries receive sympathetic, parasympathetic, and sensory innervation (Edvinsson et al., 1987). Peripheral sources of cerebrovascular innervation have been determined to a large extent in the rat. Sympathetic fibers arise in the superior cervical and stellate ganglia (Arbab et al., 1988; Handa et al., 1990), parasympathetic fibers in the pterygopalatine, otic, and internal carotid ganglia (Suzuki et al., 1988; Edvinsson et al., 1989; Hara et al., 1993; Shimizu, 1994), and sensory fibers in trigeminal, internal carotid, and first and second cervical spinal ganglia (Arbab et al., 1986; Suzuki et al., 1989). The pterygopalatine ganglion is often termed sphenopalatine ganglion, although the first term is the official anatomic term (Federative Committee on Anatomical Terminology, 1998).
Neural pathways to the supratentorial arterial tree are distributed through the cavernous sinus and surrounding regions (Bleys et al., 1996). In the rat, the cavernous sinus contains a nerve plexus and several ganglia. One of these ganglia is a caudal extension of the pterygopalatine ganglion along the Vidian nerve in the floor of the cavernous sinus, which is named the cavernous part of the pterygopalatine ganglion (PGC) (Bleys et al., 1996). Furthermore, there are numerous small ganglia (cavernous ganglia) in the nerve plexus of the cavernous sinus, which mainly occupy its lateral wall (Bleys et al., 1996). Small ganglia also have been observed in the human cavernous sinus (Gellert, 1934; Carvalho, 1985; Suzuki and Hardebo, 1991a; Bleys et al., submitted).
The targets of the PGC and cavernous ganglia are unclear. They may, however, contribute to cerebrovascular innervation because it was found that in the rat there is a morphologic substrate for a pathway from these ganglia to the internal carotid artery through the cavernous plexus and then running retrogradely along the abducens nerve (Bleys et al., 1996). The PGC is continuous with the ‘classical’ pterygopalatine ganglion in the orbit (PGO: orbital part of the pterygopalatine ganglion) and the cavernous ganglia are closely associated and connected with the PGC. For this reason, these ganglia may all be parasympathetic in nature. This assumption is supported by the finding that most of the ganglion cells in the human cavernous sinus contain vasoactive intestinal polypeptide with additional presence of choline acetyltransferase or substance P and calcitonin-gene related peptide in several of these cells (Suzuki and Hardebo, 1991a).
The aim of the current study was to find out whether ganglia in the cavernous sinus, both the PGC and the small ganglia, contribute to cerebrovascular innervation in rat. For this purpose, retrograde tracing was performed from the middle cerebral artery (MCA). Staining for vesicular acetylcholine transporter (VAChT), a marker for cholinergic neurons (Arvidsson et al., 1997), was performed to investigate the presumed parasympathetic nature of these ganglia.
MATERIALS AND METHODS
Nine male Sprague–Dawley rats (12-weeks-old) were used. Because the authors' description of neural structures in the cavernous sinus region was based on acetylcholinesterase (AChE) staining in Wistar rats (Bleys et al., 1996), the authors used one rat to verify the arrangement of nerves and ganglia in the cavernous sinus by AChE histochemistry. Eight rats were used for retrograde tracing.
AChE histochemistry
One rat was killed by an overdose of carbon dioxide and was perfused through the heart with phosphate-buffered saline (PBS). Then 500 mL 1% paraformaldehyde and 1.25% glutaraldehyde in PBS was perfused at 4°C, followed by perfusion of 400 mL PBS containing 15% sucrose at 4°C. The head was halved in the median plane. The part of the skull base medial to the cavernous sinus and the medial orbital wall were removed. The cavernous sinus was opened in its medial wall. AChE histochemistry through immersion was performed according to the method described elsewhere (Bleys et al., 1994).
Retrograde tracing
Rats were anesthetized using a mixture of 2.5% halothane and oxygen, and the heads were securely clamped in a stereotaxic head frame. An incision of the skin covering the zygomatic arch was made and the skin retracted to expose the left temporalis muscle. The temporalis muscle was incised and retracted to expose the side of the skull. Using a small dental burr, a hole was drilled through the temporal bone, midway between the eye socket and the attachment of the zygomatic arch, to reveal the dura mater. Underlying the dura mater a large branch of the MCA could easily be identified. A small incision was made with a fine Beaver blade (Becton Dickinson, Rochelle, U.K.) and small pieces of gel foam, preabsorbed with 2 μL fluorogold (Fluorochrome, Denver, CO, U.S.A) and 2 μL fast blue (Sigma, Poole, U.K.), were placed over the most proximal part of the artery that was visible. A collagen fleece impregnated with gentamicin was placed over the exposed area and the temporal muscle and skin were closed. Buprenorfin (Temgesic, 0.06 mg/kg body weight) analgesia was administered intramuscularly to all animals postoperatively.
After 1 to 4 days, the animals were killed by an overdose of carbon dioxide and were perfused through the heart with PBS followed by 500 mL 4% paraformaldehyde in PBS and subsequently perfused with 200 mL PBS. The heads were halved in the median plane, and both ipsilateral and contralateral cavernous sinuses were carefully dissected out. The exact site of the tracer application was determined on the brain by visual inspection. Several cavernous sinuses were studied as whole mount preparations after being opened by a longitudinal incision and spread out. Most ipsilateral sinuses, however, were sectioned on a cryostat and viewed under a fluorescence microscope with appropriate filter settings to locate the fluorogold and fast blue positive neurons. Table 1 lists several details of the procedure.
Relevant details of the retrograde tracing procedure
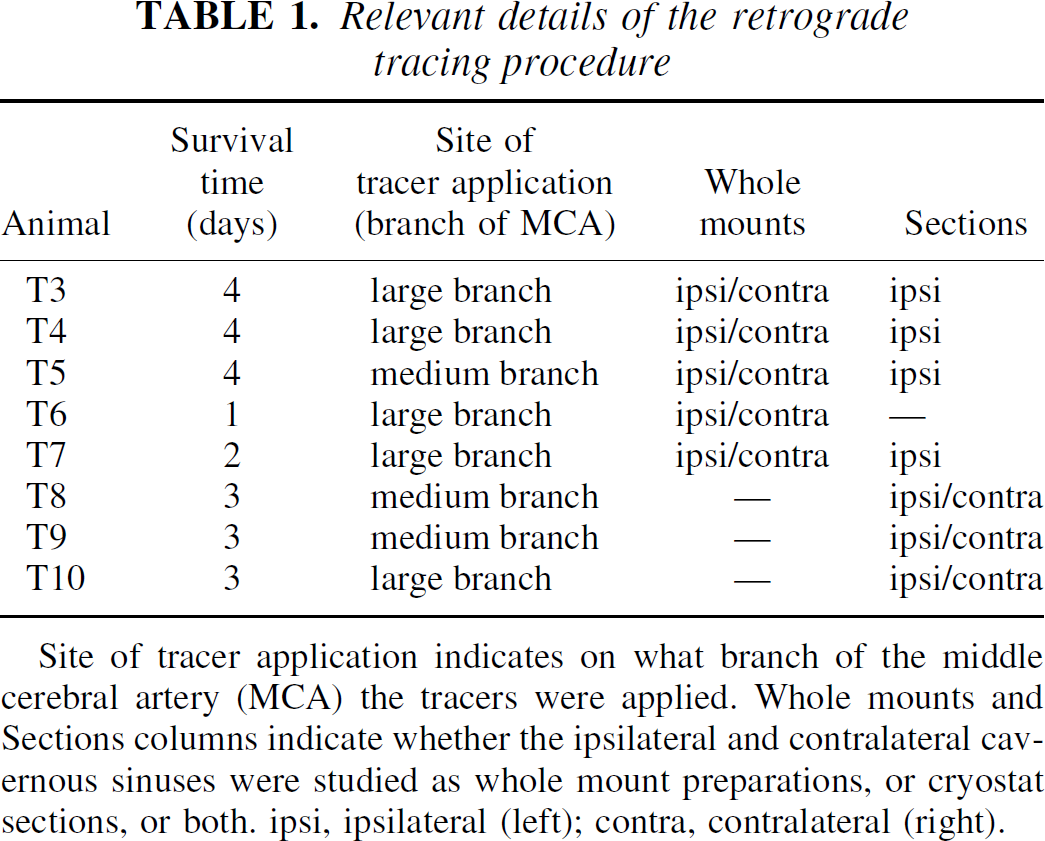
Site of tracer application indicates on what branch of the middle cerebral artery (MCA) the tracers were applied. Whole mounts and Sections columns indicate whether the ipsilateral and contralateral cavernous sinuses were studied as whole mount preparations, or cryostat sections, or both. ipsi, ipsilateral (left); contra, contralateral (right).
Immunohistochemistry
Selected sections with labeled neuron cell bodies were stained immunohistochemically for VAChT by the streptavidin-biotin method, at room temperature. Sections were washed in Hepes buffer containing 0.1% Triton X-100 for 40 minutes, followed by preincubation in 5% goat serum, 5% bovine serum albumin, and 0.1% Triton X-100 in Hepes buffer for 90 minutes. Subsequently, the sections were incubated overnight in anti-VAChT antibodies (Phoenix Pharmaceuticals, Mountain View, CA, U.S.A.) diluted 1:3000 in Hepes buffer, containing 1% goat serum, 0.1% Triton X-100, and 0.1% DL lysine. After washing in PBS, the specimens were incubated in biotinylated goat–anti-rabbit antiserum (Dako, Glostrup, Denmark) diluted 1:500 in PBS, containing 1% goat serum, 0.1% Triton X-100, and 0.1% DL lysine, for 90 minutes. Sections were washed in PBS, and subsequently incubated in fluorescein isothiocyanate-(FITC) conjugated streptavidin (Dako) diluted 1:100 in PBS, containing 1% goat serum, 0.1% Triton X-100, and 0.1% DL lysine, for 90 minutes. Sections were washed in PBS, stained with 0.05% pontamine sky blue in PBS for 10 minutes, and washed again in PBS. Sections were mounted in antifade mountant (Citifluor, London, U.K.).
RESULTS
Whole mount AChE staining of one rat demonstrated that the arrangement of nerves and ganglia was similar to that seen in Wistar rats (Fig. 1). Thus, there was a cavernous plexus proper in the lateral cavernous sinus wall and a lateral extension of the plexus above the ophthalmic and maxillary divisions of the trigeminal nerve. There were several small ganglia in the cavernous plexus as well as a PGC along the Vidian nerve. The cavernous plexus was connected to the PGC and the abducens nerve. The authors could not find any differences between Wistar rats and Sprague–Dawley rats with respect to the anatomic arrangement of nerves in the cavernous sinus region.
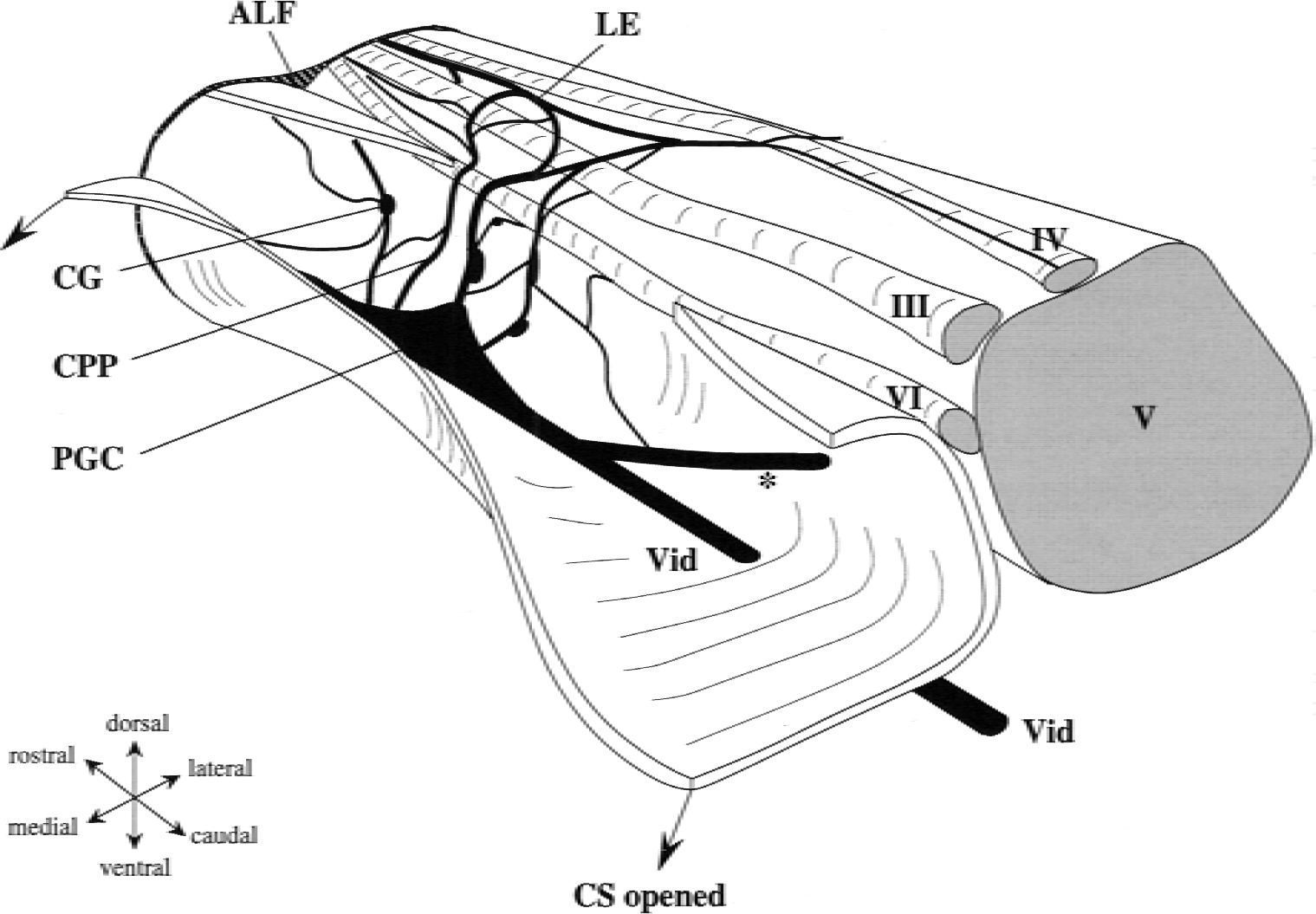
Drawing of the right cavernous plexus as present in Wistar and Sprague–Dawley rats. The cavernous sinus (CS) has been opened and part of the wall removed to demonstrate the cavernous plexus proper (CPP) in its lateral wall. The lateral extension (LE) lies above the ophthalmic and maxillary divisions of the trigeminal nerve. III, oculomotor nerve; IV, trochlear nerve; V, trigeminal nerve; VI, abducens nerve; ALF, anterior lacerated foramen that leads into the orbit; CG, cavernous ganglion; PGC, cavernous part of the pterygopalatine ganglion; Vid, Vidian nerve. *Nerve that runs to the trigeminal ganglion.
Evaluation of the site of tracer application showed that, in most cases, the tracer had been applied to a large branch of the MCA, that is, after the first bifurcation of the artery. In the remaining three animals, a smaller branch had been the site of tracer application (Table 1). Results of the retrograde tracing experiments demonstrated labeled neurons in the PGC and small cavernous ganglia on the ipsilateral side. The tracer was visible in the whole mount preparations, but the sections (Fig. 2) were definitely much clearer. The fluorogold tracer gave labeled neurons a granular appearance that was superposed on an even background of fast blue staining. The number of labeled cells varied between the animals. In general, more labeled cells were visible when the tracer had been applied to a larger branch of the MCA (Fig. 2). It was roughly estimated that in the “best” animals approximately 30% of the cells were labeled. In two animals, labeled neurons were also observed in small ganglia of the contralateral side. Especially (but not exclusively) in the animals with the shorter survival times of 1 to 2 days, fibers in the abducens nerve and the cavernous plexus proper were labeled on the ipsilateral side (Fig. 3). The tracing results are summarized in Table 2.
Relative number of labeled neural elements in the cavernous sinus (and surroundings) after retrograde tracing from the middle cerebral artery

PGC, cavernous part of the pterygopalatine ganglion; –, no elements observed; ±, occasional element observed in minority of animals; +, few elements observed; ++, moderate number of elements observed.
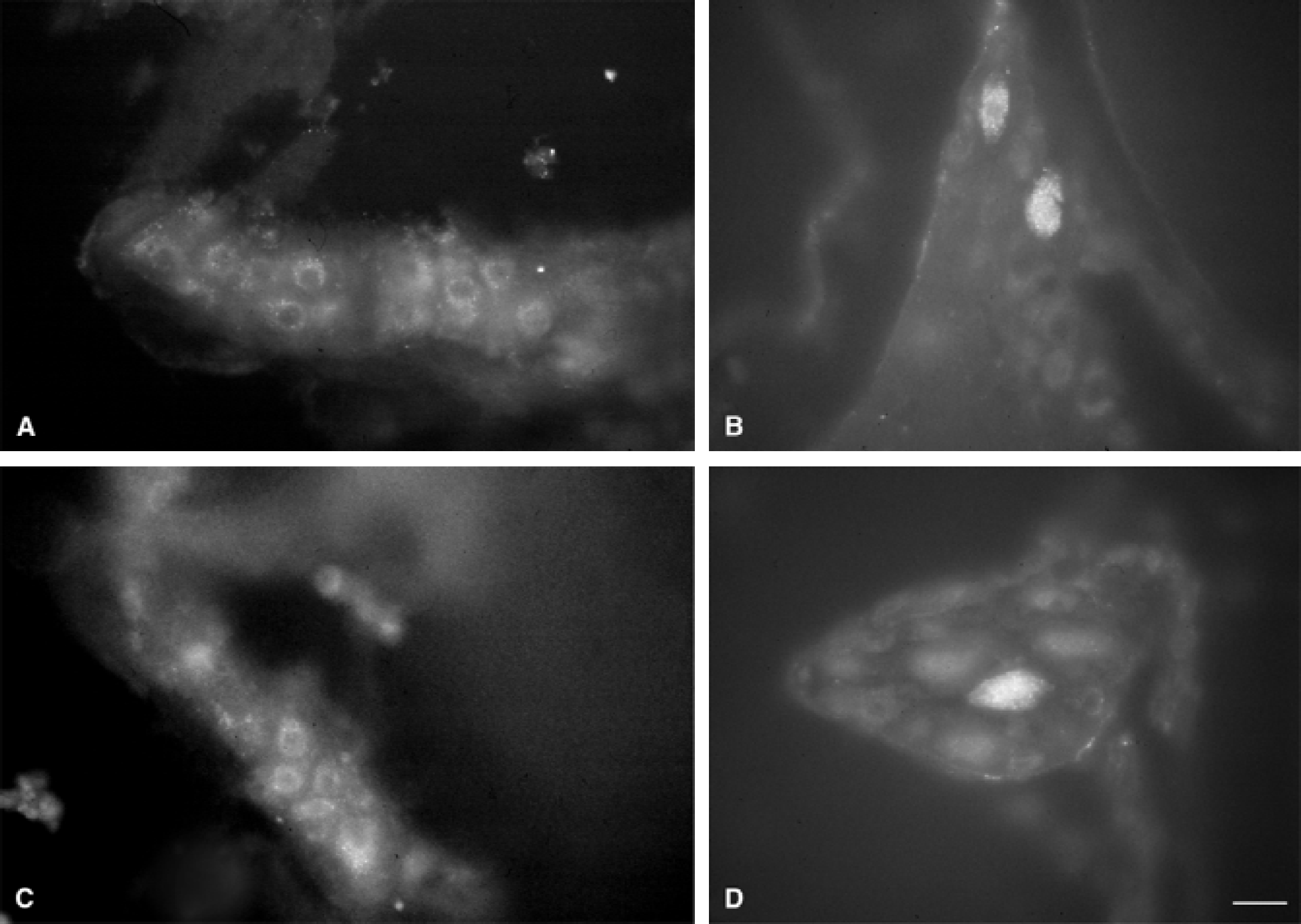
Accumulated fluorogold and fast blue in ganglia of the cavernous sinus of a rat with tracer application on a large branch of the middle cerebral artery
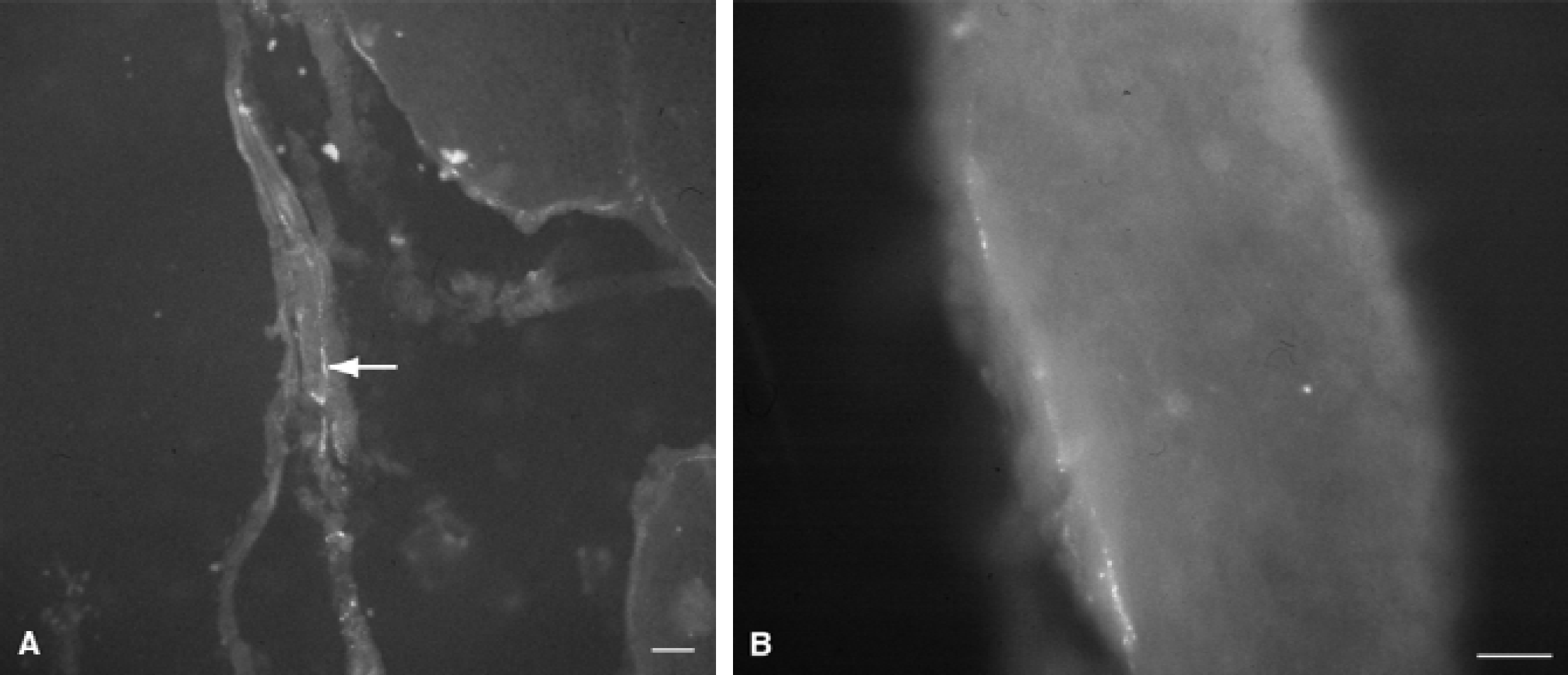
Tracer containing nerve fibers in
VAChT immunohistochemistry stained most cells in both the PGC and the small cavernous ganglia. Some of the VAChT+ cells also contained tracer (Fig. 4). The authors found only one tracer containing cell body that was not stained by VAChT immunohistochemistry (Fig. 4C to 4D).
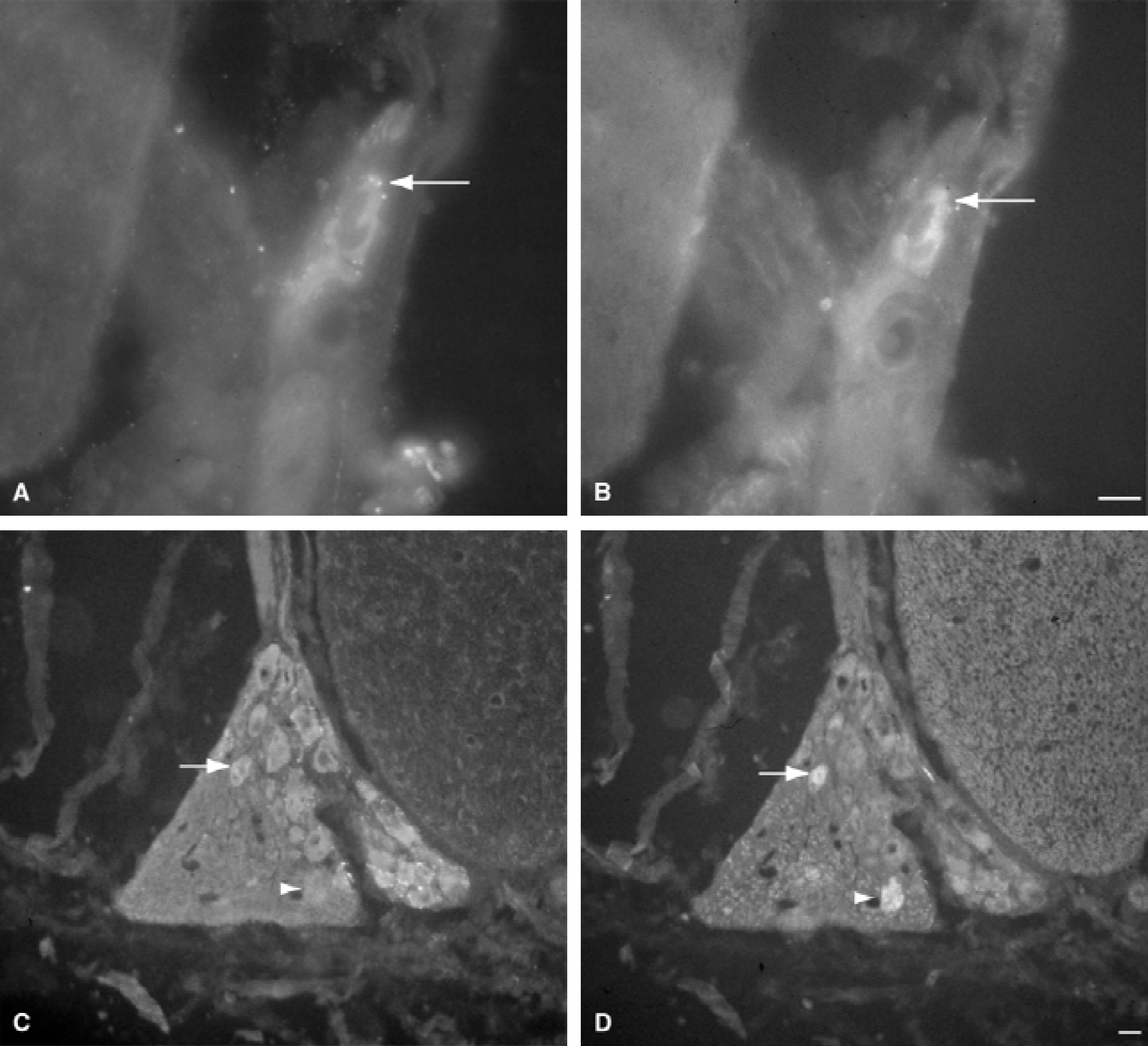
Cells labeled for vesicular acetylcholine transporter (VAChT) and for the tracers fluorogold and fast blue.
DISCUSSION
In the current study, the authors provided evidence for a role of the cavernous sinus ganglia in parasympathetic cerebrovascular innervation of the rat. Both the PGC and the small cavernous ganglia contribute considerably to innervation of the MCA. It is reasonable to assume that if the MCA is involved, other supratentorial basal cerebral arteries receive innervation from the same source (Branston, 1995). Currently, there is proof for parasympathetic innervation of the basal cerebral arteries by the pterygopalatine (that is, the “classical” orbital part: PGO), cavernous sinus (PGC and small cavernous), otic, and internal carotid ganglia. Retrograde tracing from the MCA resulted in labeling in all of these ganglia. This makes the parasympathetic cerebrovascular nerve supply a diffuse system and therefore difficult to approach in denervation or stimulation experiments. The authors have discussed previously that conflicting results after ganglionectomies could be explained by the presence of cavernous sinus ganglia as additional sources of parasympathetic innervation (Bleys et al., 1996).
Innervation of the MCA by the contralateral PGO and superior cervical and trigeminal ganglia has been reported (Edvinsson et al., 1989). Functional support for contralateral innervation from the PGO comes from a study by Seylaz et al. (1988) who, after stimulation of the PGO, found an increase in cerebral blood flow on the contralateral side besides an ipsilateral increase. Nevertheless, the authors have doubts regarding the few occasions in which a labeled cell was found in a cavernous ganglion of the contralateral side in the current study. This was not the case in the majority of animals, and therefore it may have been caused by leakage of the tracer in the subarachnoid space. Such leakage is a danger in experiments like this one. However, the authors believe that their results have not suffered from massive leakage because they are fairly reproducible. Furthermore, if tracer would have been picked up by other cerebral arteries and retrogradely transported to the cavernous sinus ganglia, this would still prove the contribution of these ganglia to cerebrovascular innervation. Leakage to the dura mater would probably not interfere severely with the tracing results because the parasympathetic nerve supply of the dura mater and its vessels is relatively sparse (Keller and Marfurt, 1991; Berger et al., 1994; Edvinsson et al., 1998).
VAChT is considered a reliable cholinergic marker in both the central and peripheral nervous system (Arvidsson et al., 1997; Schäfer et al., 1998). Although most cells in the PGC and in the small cavernous sinus ganglia were VAChT-immunoreactive, some cells were not, including a single tracer containing cell. Currently, the authors can only speculate about the nature of such cells. They may contain vasoactive intestinal polypeptide (VIP), another parasympathetic marker, because cerebral arteries receive a rich supply of VIPergic fibers from the PGO (Suzuki et al., 1988; Edvinsson et al., 1989), otic ganglion (Edvinsson et al., 1989; Suzuki and Hardebo, 1991b), and internal carotid ganglion (Suzuki et al., 1988). Only approximately 50% of the VIP+ cells in these ganglia also contain choline acetyltransferase (ChAT) (Suzuki et al., 1990). It should be noted, however, that ChAT immunohistochemistry in the peripheral nervous system is difficult and results, depending on the antibodies used, have been variable (Schäfer et al., 1998; Cowen, unpublished). Because ChAT and VAChT expression appear to be tightly linked (Weihe et al., 1996), the 50% reported by Suzuki et al. (1990) could be an underestimation. Indeed, there are other findings in the porcine pterygopalatine ganglion: 75% of the ganglionic cells contained ChAT and 42% VIP (Yu et al., 1998), but species differences may also play a role here. Another possibility is that the non-VAChT containing cells in the cavernous sinus are sensory. In that case, the cavernous sinus ganglia would be similar to the internal carotid ganglion in having parasympathetic and sensory cell groups (Suzuki et al., 1988, 1989).
The majority of neurons in the cavernous sinus ganglia were not labeled after tracer application to the MCA; therefore, there must be other targets. Besides other supratentorial cerebral arteries, possible targets are glands in the orbit, nasal cavity, and palate, which are known projection sites for the PGO.
The current results provide evidence for the involvement of the cavernous plexus proper and the abducens nerve in the pathway from the cavernous sinus ganglia to the cerebral arteries. The authors had speculated about such a pathway after discovery of the cavernous plexus and nerves that connected the abducens nerve and nerves around the internal carotid artery (Bleys et al., 1996). Of these connecting branches, one was known as a sympathetic nerve (Tamamaki and Nojyo, 1987; Handa et al., 1990), but the nature of another large and several small connecting nerves was unknown. The current findings suggest that parasympathetic fibers reach the internal carotid artery through these connecting nerves. The authors cannot exclude additional pathways from the cavernous sinus ganglia to cerebral arteries. For example, it is possible that axons that originate in the PGC run rostrally in the Vidian nerve, traverse the PGO, reach the internal ethmoidal artery through the ethmoidal nerve, and run from there to the cerebral arteries (Fig. 5). This is the classical pathway from the PGO to the cerebral arteries (Suzuki et al., 1988; Bleys et al., 1996). Tracer studies combined with transections of the ethmoidal, or the abducens nerve, or both, could solve this problem. An alternative approach would be the use of anterograde tracing that would also give information about the distribution of fibers over the cerebral arterial tree and to other targets. However, anterograde tracing from the dispersed and variably located small cavernous ganglia may be impossible and anterograde tracing from the PGC will be very difficult. A surgical approach through the cavernous sinus floor would be necessary or, alternatively, micro-injection using stereotactic coordinates may be used with the risk of missing the small ganglion and finding the tracer washed away in the circulation.
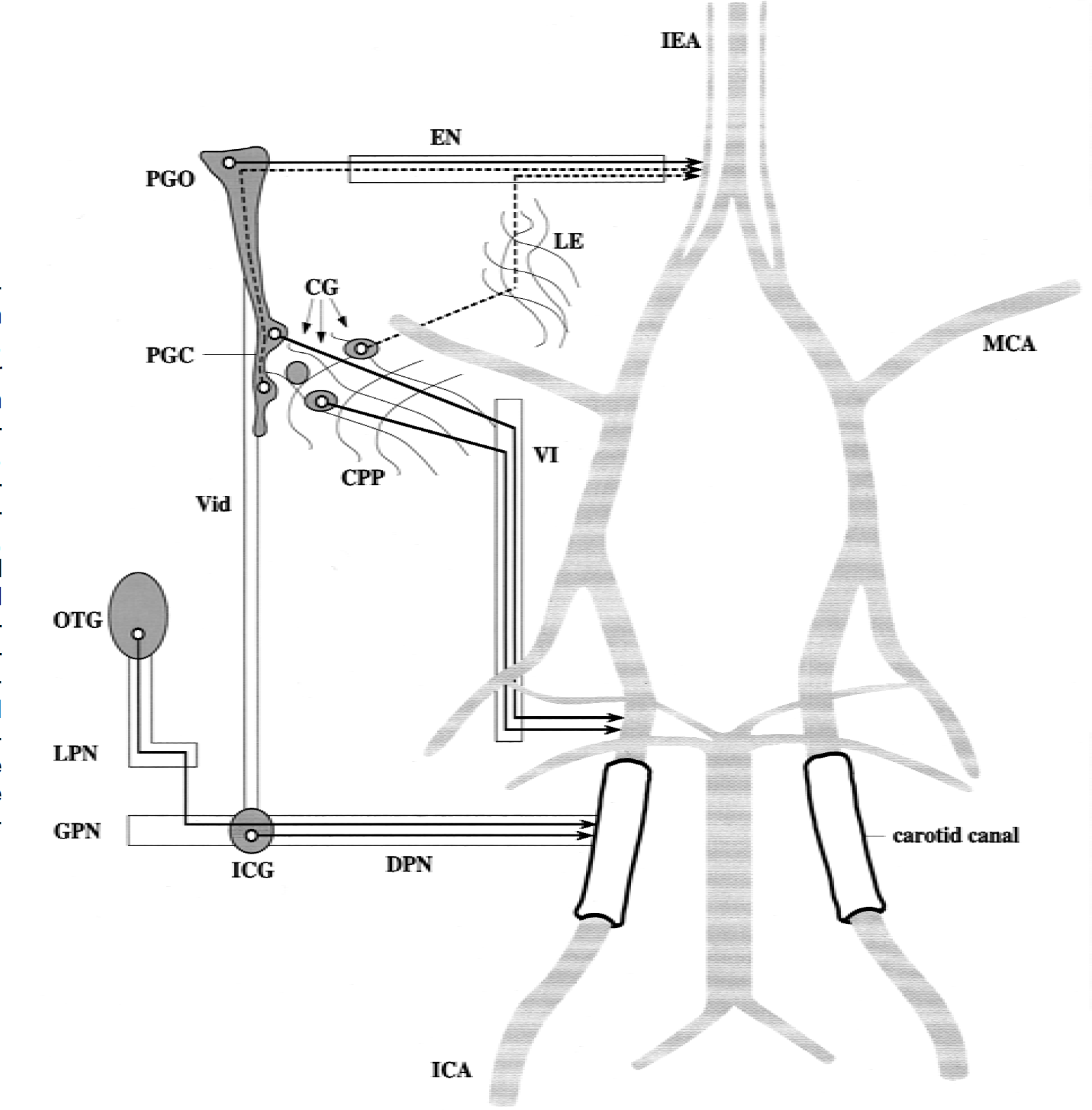
Sources of the parasympathetic cerebrovascular innervation and the pathways to the arteries. Solid lines represent pathways for which evidence has (partially) been obtained. Dashed lines represent alternative routes that may also exist. CG, cavernous ganglia; CPP, cavernous plexus proper; DPN, deep petrosal nerve; EN, ethmoidal nerve; GPN, greater petrosal nerve; ICA, internal carotid artery; ICG, internal carotid ganglion; IEA, internal ethmoidal artery; LE, lateral extension of the cavernous nerve plexus; LPN, lesser petrosal nerve; MCA, middle cerebral artery; OTG, otic ganglion; PGC, cavernous part of the pterygopalatine ganglion; PGO, orbital part of the pterygopalatine ganglion; VI, abducens nerve; Vid, Vidian nerve.
There have been recent advances in exploring the functional significance of the parasympathetic nerve supply of the cerebral arteries. It was found that in the porcine pterygopalatine ganglion all cholinergic and VIPergic neurons also contain nitric oxide synthase (NOS) (Yu et al., 1998). High numbers of NOS-immunoreactive cell bodies were also found in the PGO of the rat (Nozaki et al., 1993) and the pterygopalatine ganglion of the cat (Goadsby et al., 1996). Also, in humans, the majority of cell bodies in the pterygopalatine and otic ganglia contain VIP and NOS (Uddman et al., 1999). Nitric oxide (NO), coreleased with acetylcholine and VIP, induces relaxation in smooth muscle in cerebral arteries (Lee, 2000). Goadsby et al. (1996) investigated the role of NO in the cerebral vasodilator response in the cat and found that a NOS inhibitor attenuated the increase of cerebral blood flow after stimulation of the facial nerve. The facial nerve contains preganglionic nerve fibers that synapse in the pterygopalatine ganglion. Stimulation and destruction of the pterygopalatine ganglion in dogs made clear that release of NO by parasympathetic cerebrovascular nerves is important in maintaining cerebral arterial diameter under resting conditions (Toda et al., 2000).
Exact knowledge of the parasympathetic sources of cerebrovascular innervation is crucial for the performance and interpretation of experiments aimed at investigating functional aspects of the parasympathetic nerve supply to the cerebral arteries. An attempt to completely deprive these arteries of their parasympathetic innervation would require the destruction of a number of ganglia, otherwise it should be kept in mind that there are still active sources. The current knowledge of sources and pathways of parasympathetic cerebrovascular innervation in the rat is depicted in Fig. 5. The human cavernous sinus also contains small ganglia that are connected to the internal carotid artery (Bleys et al., submitted). Considering the current findings in rat and the authors' studies in humans, the authors suggest that the cavernous ganglia are among the parasympathetic sources for cerebrovascular innervation.
Footnotes
Acknowledgment:
The authors thank Mrs. G.W. van Wieringen for technical assistance.
