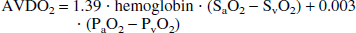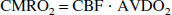Abstract
Studies documenting the cerebral hemodynamic consequences of selective brain hypothermia (SBH) have yielded conflicting data. Therefore, the authors have studied the effect of SBH on the relation of cerebral blood flow (CBF) and CMRO2 in the forebrain of pigs. Selective brain hypothermia was induced in seven juvenile pigs by bicarotid perfusion of the head with extracorporally cooled blood. Cooling and stepwise rewarming of the brain to a Tbrain of 38°C, 25°C, 30°C, and 38°C at normothermic Ttrunk (38°C) decreased CBF from 71 ± 12 mL 100 g−1 min−1 at normothermia to 26 ± 3 mL 100 g−1 min−1 and 40 ± 12 mL 100 g−1 min−1 at a Tbrain of 25°C and 30°C, respectively. The decrease of CMRO2 during cooling of the brain to a Tbrain of 25°C resulted in a mean Q10 of 2.8. The ratio between CBF and CMRO2 was increased at a Tbrain of 25°C indicating a change in coupling of flow and metabolism. Despite this change, regional perfusion remained coupled to regional temperatures during deep cerebral hypothermia. The data demonstrate that SBH decreases CBF and oxygen metabolism to a degree comparable with the cerebrovascular and metabolic effects of systemic hypothermia. The authors conclude that, irrespective of a change in coupling of blood flow and metabolism during deep cerebral hypothermia, cerebral metabolism is a main determinant of CBF during SBH.
There is considerable evidence that hypothermia is a useful intrainsult and postinsult treatment modality for brain injury caused by hypoxia—ischemia, trauma, or neonatal asphyxia (Dietrich, 1992, Gunn et al., 1998b; Wass et al., 1996). Moreover, systemic hypothermia is routinely used during cardiopulmonary bypass in cardiovascular surgery (McCullough et al., 1999; Jonas, 1998). Because systemic hypothermia causes potentially hazardous side effects (for review see Schubert, 1995), selective brain hypothermia (SBH) is being looked at more intensively with respect to feasibility and cerebroprotective effects (Gunn et al., 1997; Kuluz et al., 1992; Schwartz et al., 1996b).
Hypothermic cerebral protection is mediated in part by a reduction of cerebral metabolism (Lanier, 1995). Numerous animal and human studies have shown that systemic hypothermia decreases cerebral blood flow (CBF) and CMRO2 (Greeley et al., 1993; Michenfelder et al., 1991). However, very few studies have addressed the cerebrovascular and cerebral metabolic responses to SBH. Currently there is no study documenting the physiologic effects of SBH on CMRO2 and, consequently, on the coupling of CBF and CMRO2. Furthermore, investigations documenting the hemodynamic consequences of SBH have reported seemingly conflicting data on the cerebrovascular effects of SBH. A single study in rats has shown that external head cooling results in an increase of CBF (Kuluz et al., 1993). Based on this study it has been concluded that the method of cooling (systemic vs. selective) may be a key factor in determining the cerebral hemodynamic consequences of hypothermia (Dietrich, 1996). However, a more recent study in baboons reported a decreased CBF in response to hypothermic perfusion of the brain with extracorporally cooled blood through one carotid artery (Schwartz et al., 1996a). In consideration of these different findings it seems questionable that SBH by itself could increase CBF.
It has also been suggested that periods of mismatch between CBF and metabolism causing cerebral venous desaturation during topical cooling and rewarming of the brain may explain postoperative cerebral dysfunction after deep hypothermic procedures (van der Linden et al., 1989). Furthermore, it has been shown that even small temperature differences affect the extent of cerebral injury (Wass et al., 1995) Selective brain hypothermia results in temperature gradients within the brain (Gelman et al., 1997; Kuhnen et al., 1999; Schwartz et al., 1996a). However, the effect of regional temperature differences on regional CBF under conditions of SBH had not been studied until now. Therefore, the aim of the current study was to assess the coupling of CMRO2 and CBF in response to SBH and to investigate the effect of brain temperature on the regional cerebrovasculature during moderate and deep SBH.
METHODS AND MATERIALS
Animals and surgery
Using a protocol approved by the Committee of Animal Care and Use of the Thuringian State Government seven mixed breed female pigs (6-weeks-old, 12.9 ± 1.6 kg BW) were anesthetized with 1.5% isoflurane in nitrous oxide and oxygen (FiO2 = 0.35 − 0.40). In addition, all incision sites were infiltrated with 1% lidocaine. During surgery rectal temperature was maintained at 38°C by a heating pad and a temperature controlled heating lamp. Electrocardiogram recordings were made from standard limb leads using stainless steel needle electrodes. The urinary bladder was punctured and permanently drained (Cystofix, Minipaed, Braun Melsungen AG, Germany). A central venous catheter (PU, 1.4 mm OD) was inserted through a branch of the left external jugular vein to infuse physiologic saline containing glucose (3%) and heparin (2 IE/mL) with a rate of 5 mL kg BW−1 h−1. All pigs underwent a tracheotomy, were paralyzed with pancuronium bromide (0.2 mg kg−1 h−1), and were mechanically ventilated. The ventilator was set with a positive inspiratory pressure of 20 to 25 mbar and a positive end expiratory pressure of 2 to 4 mbar. Respiratory rate and inspired oxygen fraction were titrated to maintain a PaCO2 of 35 to 40 mm Hg and PaO2 of 120 to 150 mm Hg. A left saphenous artery catheter (PU, 1.4 mm OD) was inserted for blood pressure and blood gas monitoring and withdrawal of arterial blood samples. For measurement of cardiac output by thermodilution, a thermocouple was inserted through the right saphenous artery and positioned in the aorta at the height of the diaphragm. A left thoracotomy was then performed through the third intercostal space, and a catheter was inserted into a pulmonary vein and advanced towards the left atrium for injection of colored microspheres (CMS). During surgery 80 mL of arterial blood was withdrawn and substituted with 80 mL of a plasma expander (Gelafusal-N in Ringeracetat solution). Blood was stored in heparinized syringes and used to compensate the volume loss into the heat exchangers and for substitution of blood samples withdrawn during the experiment.
Instrumentation of the head
The animal was placed in sphinx position with the head fixed in a stereotaxic frame (D. Kopf Instruments, Tujunga, CA, U.S.A.). After exposing the skull, trepanations were made for insertion of a sagittal sinus catheter (2.5 Ch, PU; advanced to the confluens sinuum) and stereotaxic placement of the guide cannulas for insertion of thermodes—frontal cortex, parietal cortex, occipital cortex, thalamus, hypothalamus, nucleus caudatus of the left hemisphere, and one in the parietal cortex of the right hemisphere. According to Yoshikawa (1968), the stereotaxic coordinates of the guide cannulas have been predetermined in pigs of the same age. Each cannula was fixed with skull screws. All trepanation sites were closed with bone wax and dental acrylic. In addition, in two animals an epidural laser—Doppler flow probe (MBF 3D; Moore Instruments, Axminster, England) was placed above the right parietal cortex.
Vascular manipulations for temperature control of brain and trunk
After placement of the thermocouples in the guide cannulas the animal was turned on to the back and anticoagulated by a bolus injection of heparin (500 IU kg BW−1) followed by an infusion of 90 IE heparin kg BW−1 h−1 starting 2 hours after the bolus injection. The vascular manipulations necessary for independent control of Ttrunk and Tbrain have been described in detail elsewhere (Kuhnen, et al., 1999). Briefly, both common carotid arteries were cut and reconnected by silicone tubes (4 mm inner diameter). The right external jugular vein was cut and the distal end ligated. The proximal end of the vein was connected to a clamped silicone tube (4.8 mm ID). The systemic blood temperature (Ttrunk) was measured by a thermocouple that was positioned through the proximal carotid artery near the aortic arc. The thalamic temperature was used to guide the brain temperature manipulation.
Experimental protocol
After the surgical instrumentation was completed, the animal was allowed to recover from surgery for a minimum of 60 minutes. Isoflurane concentration was decreased to 0.5%. After a further 30 minutes of continuous temperature recordings, baseline values were measured. Then the proximal part of the left common carotid artery was connected through a Y-piece to both distal common carotid arteries. A heat exchanger was inserted between the left proximal common carotid artery and both distal carotid arteries for temperature control of the head and brain (head line). A second heat exchanger was inserted between the proximal right common carotid artery and the right external jugular vein for temperature control of the trunk (trunk line). The flow in the trunk line was throttled to approximately 150 mL min−1. The Tbrain was controlled by the heat exchanger of the head line at predetermined levels: stage 1, 38°C; stage 2, 25°C; stage 3, 30°C; stage 4, 38°C. The Ttrunk was maintained throughout the experiment at 38°C using the heat exchanger in the trunk line. Colored miscrospheres were injected at baseline and stages 1 to 4 when Tbrain was stable for at least 20 minutes. After injection of microspheres arterial, sagittal sinus, and mixed venous blood samples for measurement of oxygen saturation, blood hemoglobin, and blood gases were drawn (OSM2, ABL 50; Radiometer, Copenhagen, Denmark). Catecholamines—epinephrine, norepinephrine, dopamine—were measured in the arterial blood samples. The withdrawn blood was substituted. At the end of the experiment the animals were killed by intravenous injection of saturated potassium chloride solution. The whole head was fixed in 5% buffered formaldehyde. Correct positioning of all peripheral catheters and the guide cannulas of the thermodes was verified by autopsy.
Measurements of blood flow, oxygen metabolism, and blood values
Regional blood flow was measured with CMs using the reference sample method. The application of this method in piglets has been validated and described in detail elsewhere (Walter et al., 1997). Briefly, CMS (2–3 × 106 in 1 mL saline and 0.01% Tween 80) were injected through a pulmonary vein (white, yellow, red, violet, and blue in random order; Dye Trak; Triton Technology, San Diego, CA, U.S.A.). During each injection an arterial reference sample was withdrawn from the abdominal aorta at 3.53 mL min−1 over 120 seconds starting 15 seconds before the microsphere injection (syringe pump SP210iw; World Precision Instruments, Sarasota, FL, U.S.A.). A total number of 34 CBF measurements were used for statistical analysis. The baseline blood flow measurement in one animal could not be used because of impurities on the filtration membrane.
At the end of the experiment the lung, heart, stomach, intestine, liver, spleen, pancreas, kidneys, and adrenals organs as well as muscle and skin tissue were dissected into standardized tissue samples weighing 1 to 5 g. The fixed brain was removed and sectioned into 27 tissue samples weighing 0.5 to 2.5 g. All tissue and arterial reference blood samples were digested in 4 mol/L potassium hydroxide containing 4% Tween 80 and filtrated through a 10 μm pore-sized membrane. The dye was recovered from the CMS by adding 150 μL N,N-Dimethylformamide and the absorption spectra of the dye solution were measured with a UV/VIS spectrophotometer (Modell 7500; Beckman Instruments, Fullerton, CA, U.S.A.). Specific absorbances of the individual dyes were used to quantify the CMS (MISS software package; Triton) of each tissue sample.
Regional blood flows [mL 100 g−1 min−1] were calculated using the equation:
Assuming the oxygen capacity of swine hemoglobin to be 1.39 mL O2/g Hb (Busija et al., 1988), the AVDO2 was calculated according to the equation:
The CMRO2 was calculated:
The CBF represents the mean of all regions that were used for the calculation of CMRO2—frontal, basal, parietal, median, temporal and occipital cortex, hippocampus, basal ganglia, thalamus, and white matter of the forebrain. The cerebral vascular resistance (CVR) was calculated as mean arterial blood pressure divided by CBF. The Q10 values were calculated by linear extrapolation of the CMRO2 quotients of the respective experimental stages.
Cardiac output was measured by the thermodilution method with the temperature probe in the thoracic aorta (HZV BN 7206; August Fischer KG, Göttingen, Germany). A 2 mL cold saline bolus was injected into the superior caval vein near the right atrium at the end of expiration. The mean value of three consecutive measurements was considered to be representative.
Arterial blood pressure, pulmonary arterial pressure, central venous pressure, the electrocardiogram and, in two animals, the laser—Doppler flux signal were continuously recorded on a multichannel polygraph (MT95K2, Astromed, West Warwick, RI, U.S.A.), digitized, and stored on an external hard drive (sampling rate of 100 Hz). The flows in the silicone tubings of the trunk and head line were continuously measured by two electromagnetic flow probes and stored together with the temperature signals on hard disk (sampling rate of 0.1 Hz). The mean temperature was calculated as the electrical mean of 18 consecutive temperature recordings during each microsphere injection.
Statistics
All values are given as mean ± SD. The acute effect of the insertion of heat exchangers (stage 1) was tested for significant change in physiologic parameters compared with baseline parameters by the paired t-test. Changes within stage 1 to stage 4 were compared using the repeated measures one-way analysis of variance or, if the normality test failed, using the repeated measures one-way analysis of variance on ranks. Dunn's method was used for pairwise multiple comparison of stage 2, stage 3, and stage 4 values with stage 1 values. The dependency of regional CBF of regional brain temperature was tested by linear regression analysis. P < 0.05 was considered significant.
RESULTS
Cooling of the brain at normothermic trunk temperature
Physiologic variables are summarized in Table 1. The arterial catecholamine concentrations yielded no evidence of any thermoregulatory or otherwise stress responses to SBH.
Summary of physiologic parameters
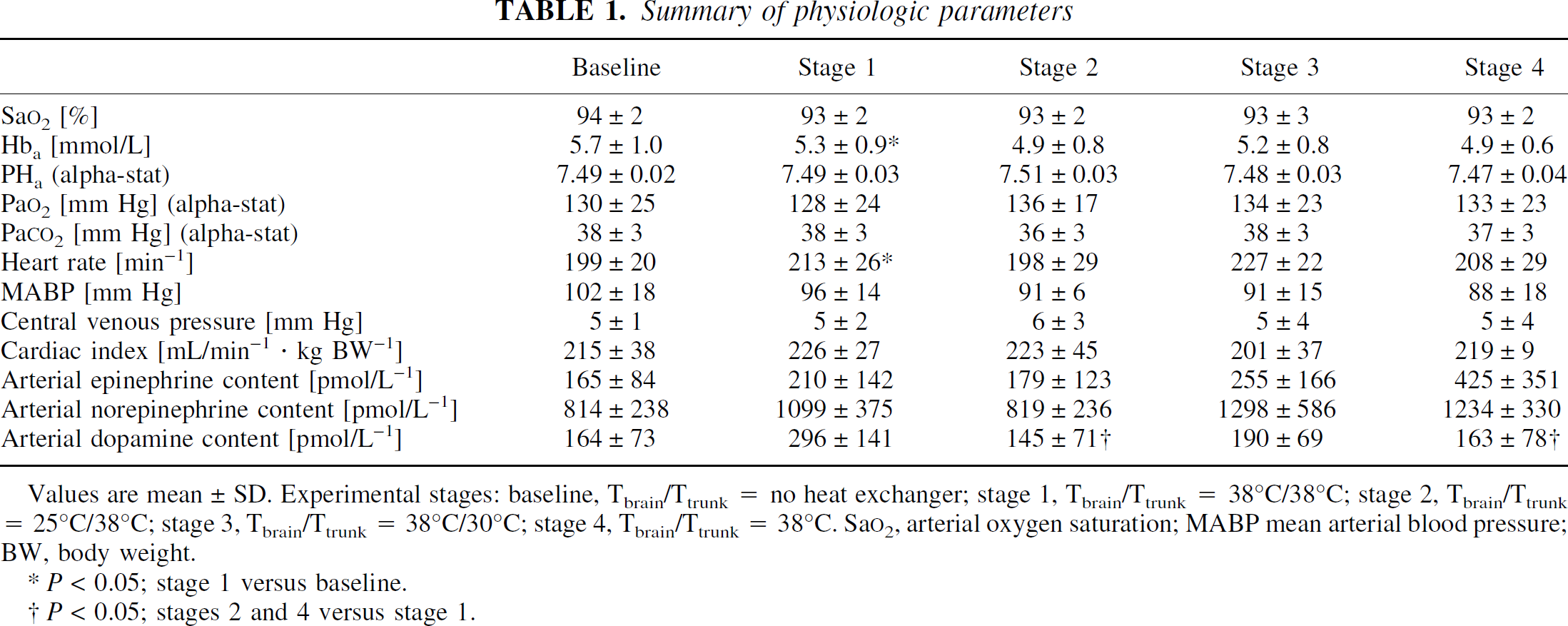
Values are mean ± SD. Experimental stages: baseline, Tbrain/Ttrunk = no heat exchanger; stage 1, Tbrain/Ttrunk = 38°C/38°C; stage 2, Tbrain/Ttrunk = 25°C/38°C; stage 3, Tbrain/Ttrunk = 38°C/30°C; stage 4, Tbrain/Ttrunk = 38°C. Sa
P < 0.05; stage 1 versus baseline.
P < 0.05; stages 2 and 4 versus stage 1.
Table 2 summarizes cerebral and trunk temperatures during CBF measurements at single experimental stages. Stabilization of the thalamic temperature at 38°C in each animal after the insertion of the heat exchangers resulted in a mean Tbrain of 37.9 ± 0.1°C. Ttrunk was kept normothermic throughout stages 1 to 4, which indicates a sufficient compensation of the cold venous return from the head by the heat exchanger of the trunk line. Cooling of the brain to a Tbrain of 25°C during stage 2 was achieved within 8.5 ± 4.2 minutes. Although Tbrain was kept within a narrow range during stage 2 (24.6 ± 0.4°C), the temperature in the frontal cortex was significantly lower than in the occipital cortex resulting in a fronto-occipital temperature gradient of −1.1 ± 0.3°C. Tbrain was changed from stage 2 to stage 3 within 4.1 ± 0.6 minutes and from stage 3 to stage 4 within 11.6 ± 4.9 minutes.
Brain and trunk temperatures during cerebral blood flow measurements (°C)
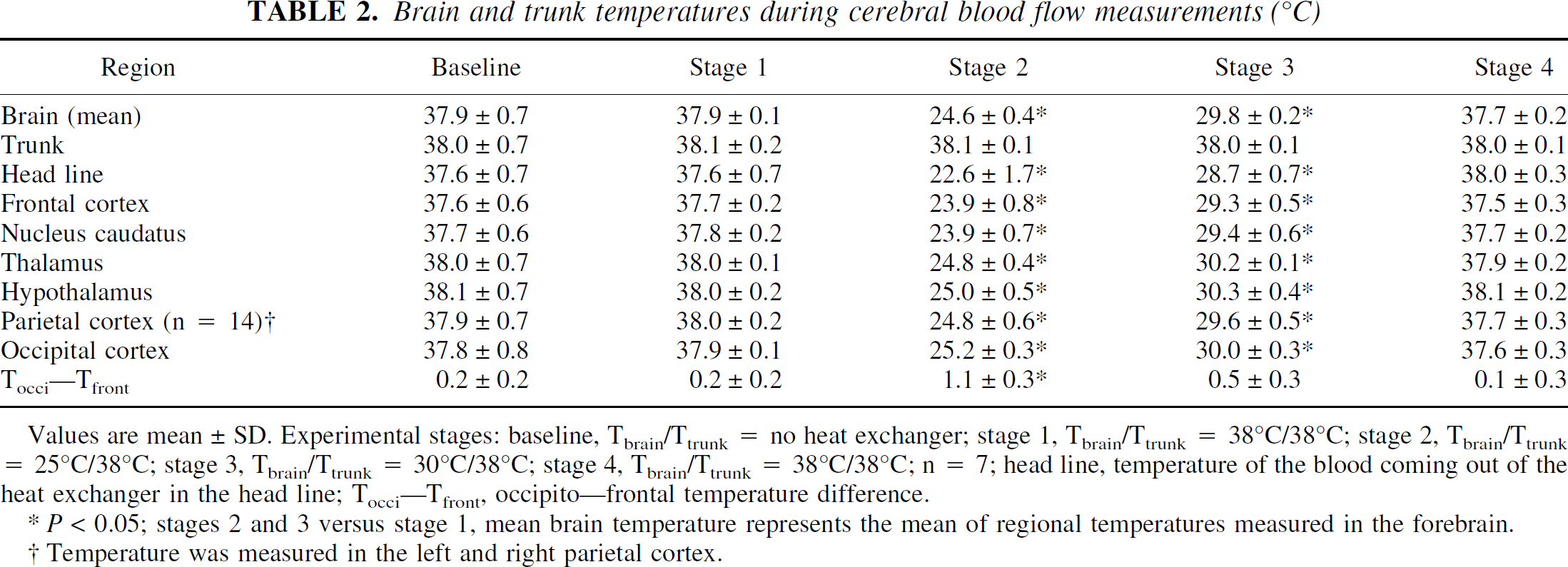
Values are mean ± SD. Experimental stages: baseline, Tbrain/Ttrunk = no heat exchanger; stage 1, Tbrain/Ttrunk = 38°C/38°C; stage 2, Tbrain/Ttrunk = 25°C/38°C; stage 3, Tbrain/Ttrunk = 30°C/38°C; stage 4, Tbrain/Ttrunk = 38°C/38°C; n = 7; head line, temperature of the blood coming out of the heat exchanger in the head line; Tocci—Tfront, occipito—frontal temperature difference.
P < 0.05; stages 2 and 3 versus stage 1, mean brain temperature represents the mean of regional temperatures measured in the forebrain.
Temperature was measured in the left and right parietal cortex.
Cerebral blood flow and metabolism
Figure 1 shows the global changes in CBF, CMRO2, cerebral vascular resistance (CVR), and AVDO2 throughout the experiment. The insertion of heat exchangers did not change these parameters. Selective cooling of the brain to 25°C and 30°C resulted in a temperature dependent decrease of CBF and CMRO2. During stages 2 and 3 the reduction of CBF was caused by an increase of CVR. Cooling of the brain to 25°C further decreased the cerebral AVDO2. Cerebral blood flow and oxygen metabolism returned to stage 1 values after rewarming of the brain to 38°C during stage 4. Table 3 summarizes the changes in regional CBFs. Despite ligation of one carotid artery and one external jugular vein, no difference between left and right hemispheric perfusion was found.
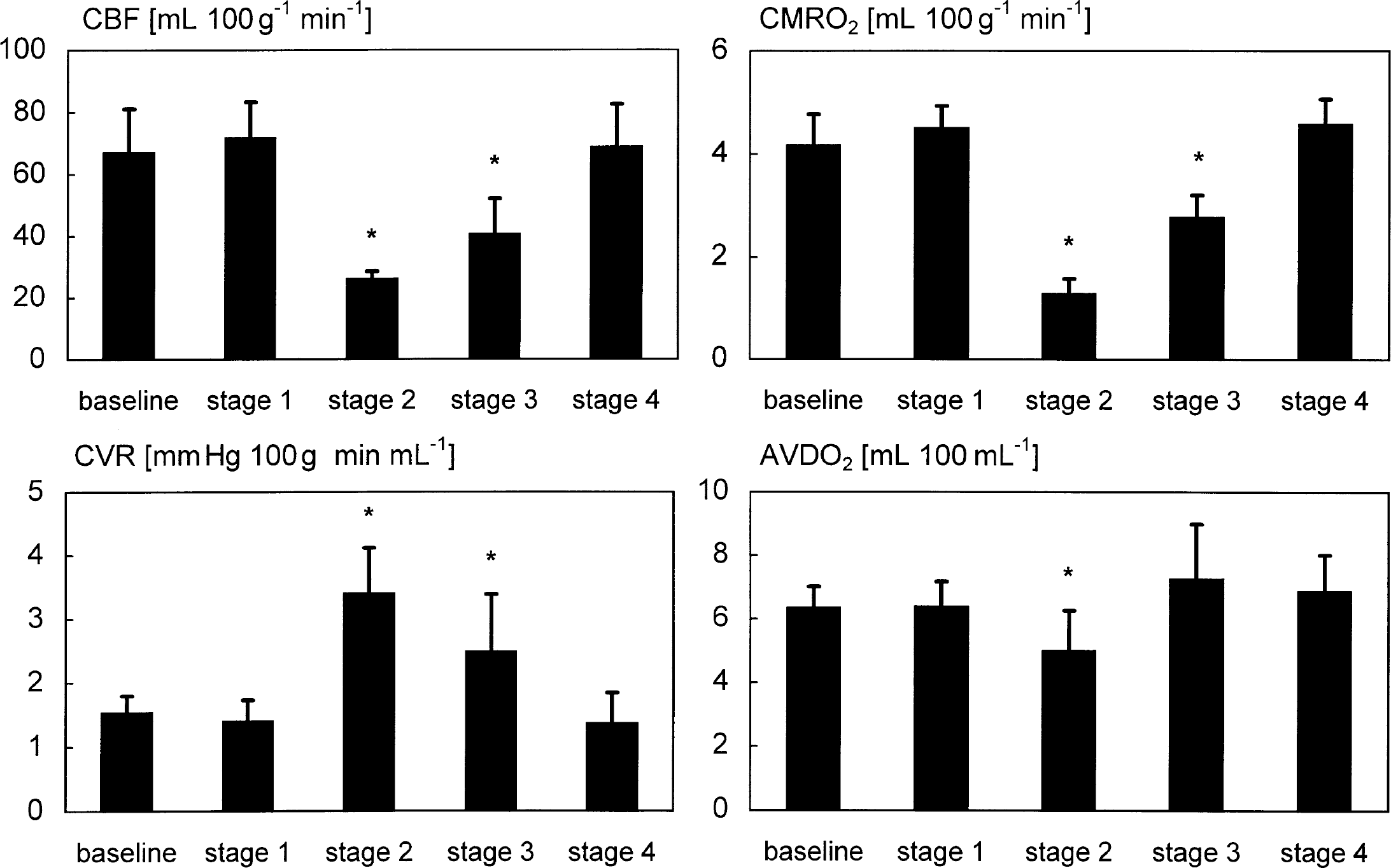
Changes of mean cerebral blood flow (CBF) (measured by colored microspheres), CMRO2, cerebral vascular resistance (CVR), and AVDO2 (mean ± SD, n = 7). Cerebral blood flow and CMRO2 remained unchanged after insertion of heat exchangers when brain temperature was kept at 38°C (stage 1). Convective cooling of the brain to a Tbrain of 25°C (stage 2) and 30°C (stage 3) resulted in a temperature dependent decrease of CBF and CMRO2 (*P < 0.05). Cerebral blood flow and CMRO2 showed no significant differences to stage 1 levels after rewarming of the brain to 38°C (S4). Reduction of CBF at stages 2 and 3 was caused by an increase of CVR. The cerebral AVDO2 was reduced only during stage 2.
Summary of cerebral blood flow data (mL 100 g−1 min−1)
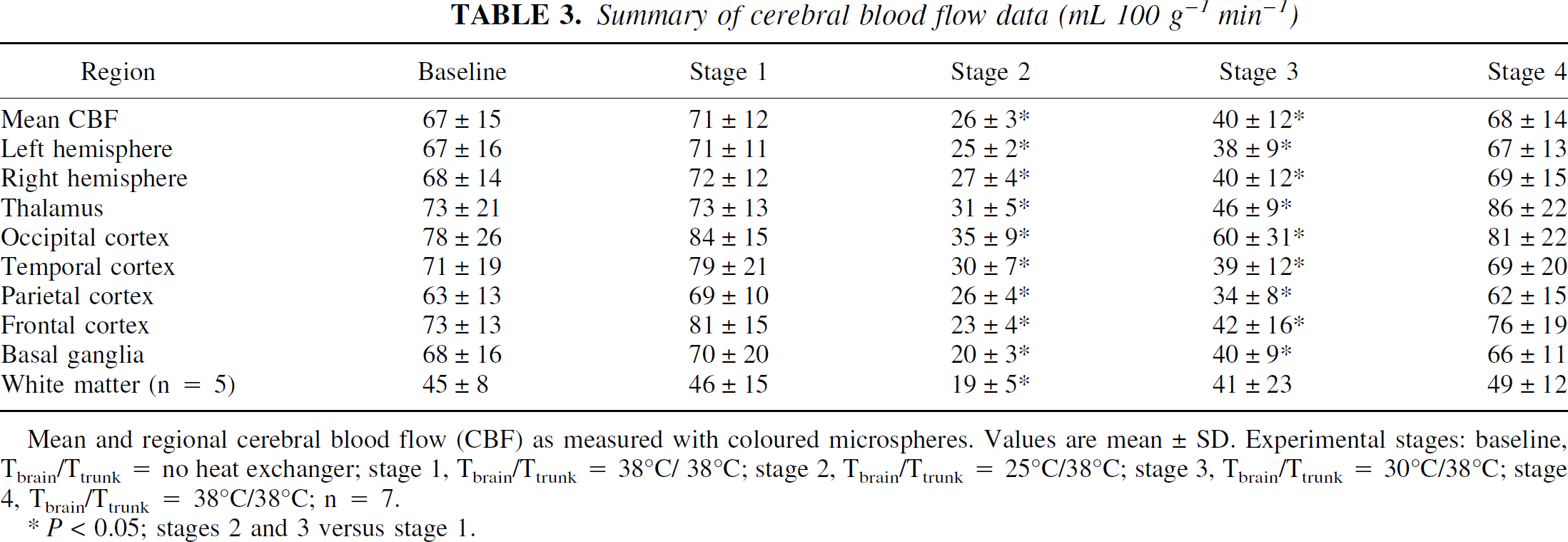
Mean and regional cerebral blood flow (CBF) as measured with coloured microspheres. Values are mean ± SD. Experimental stages: baseline, Tbrain/Ttrunk = no heat exchanger; stage 1, Tbrain/Ttrunk = 38°C/38°C; stage 2, Tbrain/Ttrunk = 25°C/38°C; stage 3, Tbrain/Ttrunk = 30°C/38°C; stage 4, Tbrain/Ttrunk = 38°C/38°C; n = 7.
P < 0.05; stages 2 and 3 versus stage 1.
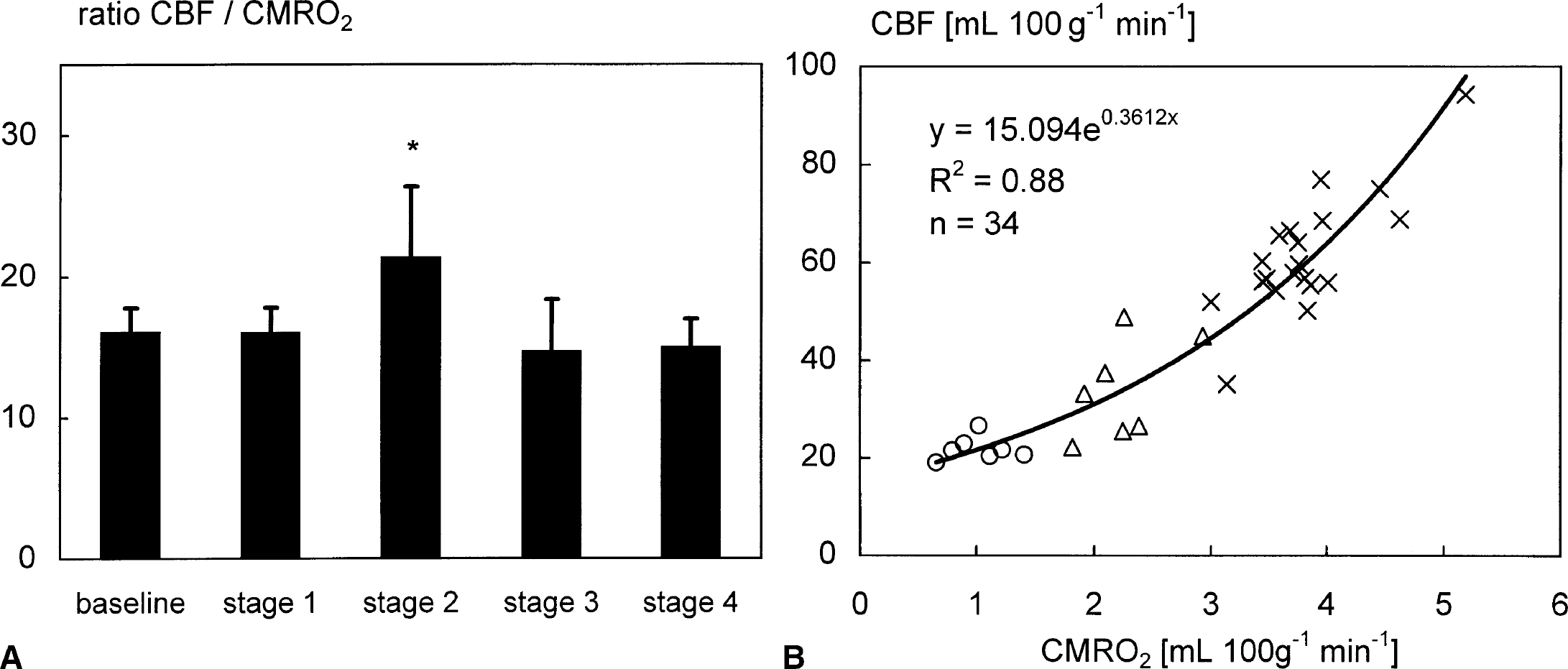
The CBF:CMRO2 ratio was used as an indicator of global flow—metabolism coupling (Fig. 2A). During moderate SBH (stage 3) the CBF:CMRO2 ratio did not change; however, the ratio increased during deep cerebral hypothermia (stage 2). The relation of CBF and CMRO2 can be described by an exponential function (Fig. 2B).
The fronto-occipital temperature gradient within the forebrain during stage 2 was used to study the temperature dependency of the regional cerebrovascular response during deep cerebral hypothermia (Fig. 3). The intrastage temperature differences of 1.1°C along the rostro-caudal axis of the forebrain during stage 2 correlated with the decrease in regional cerebral perfusion (P < 0.01), indicating a coupling of temperature and cerebrovascular response on a regional level.
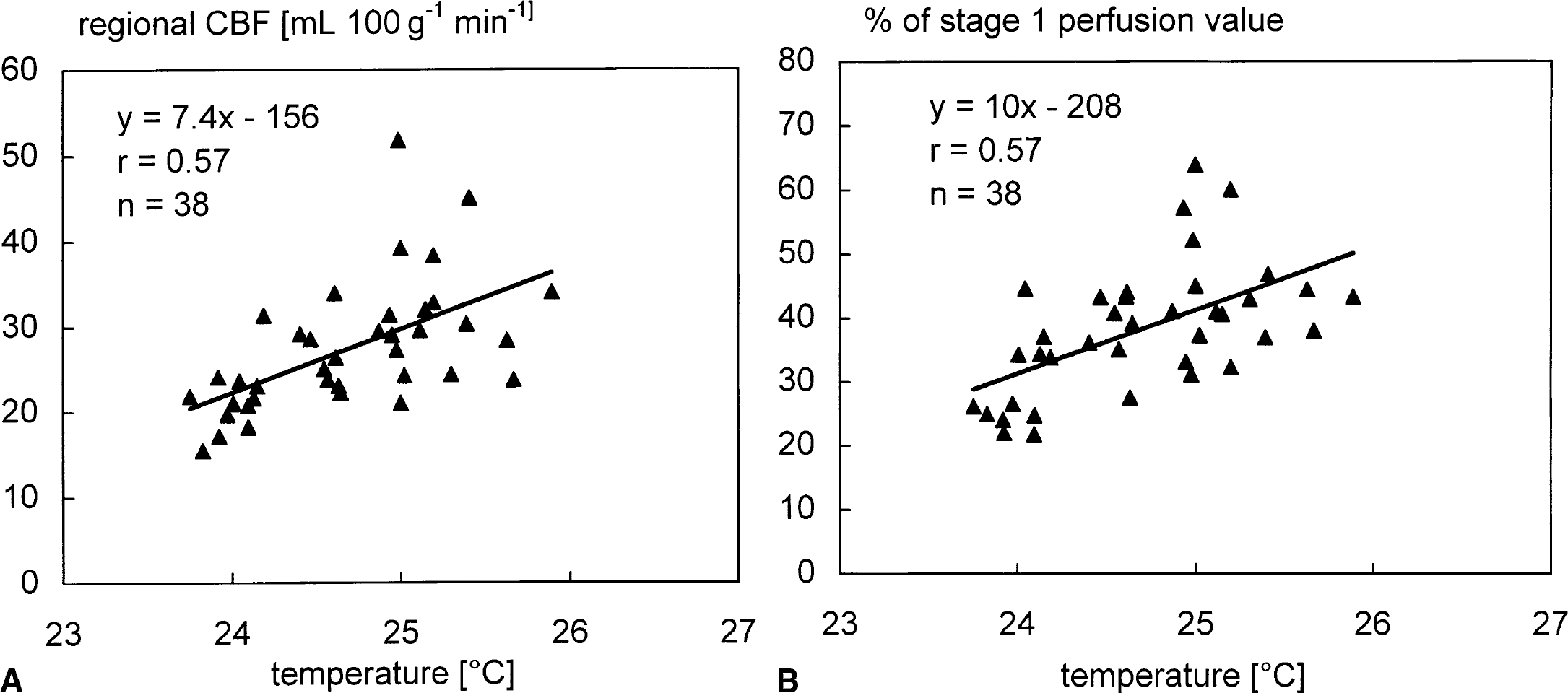
Correlation of regional cerebral blood flow (CBF) and local brain temperature (P < 0.01) during stage 2 indicates a coupling of temperature and blood flow on a regional level under conditions of deep cerebral hypothermia.
Additional monitoring of local CBF by laser—Doppler flowmetry in two animals indicated that the changes in local cortical perfusion were comparable with changes of regional CBF (Fig. 4). An example registration of changes of cortical blood flow and parietal temperature during cooling of the brain from 38°C to 25°C (stage 1 to stage 2) indicated that perfusion decreased with temperature. However, steady state values of cortical perfusion were measured several minutes after parietal temperature reached the desired temperature level (Fig. 5).
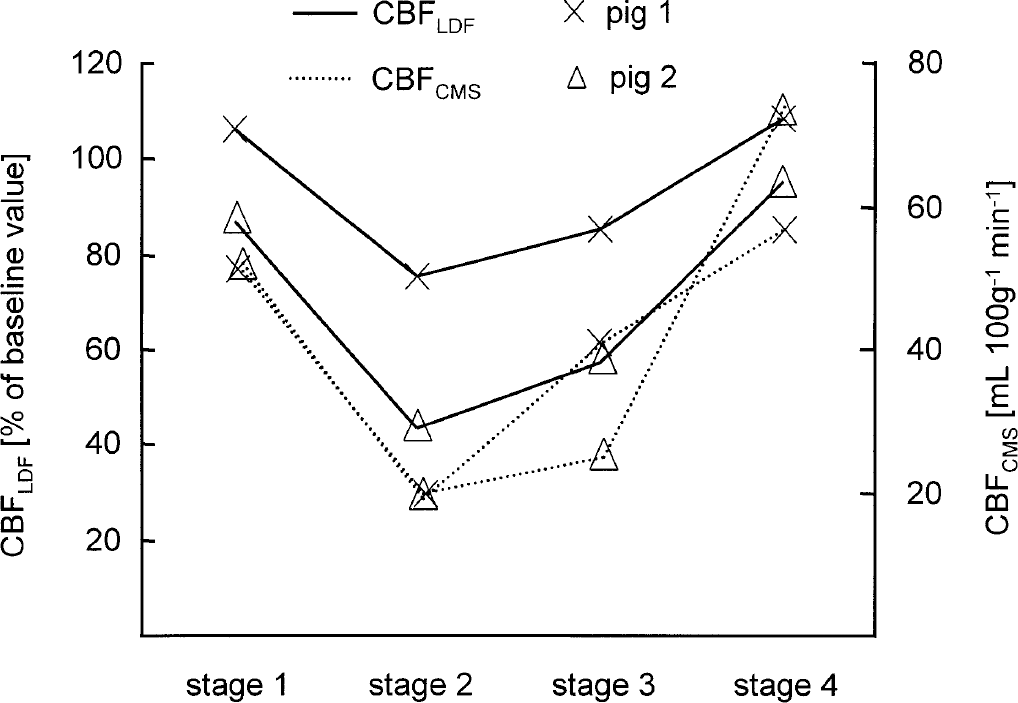
Simultaneous measurements of parietal cerebral blood flow (CBF), as measured with colored microspheres (CBFCMS), and local cortical perfusion of the parietal cortex, as measured with an epidural laser—Doppler flowprobe (CBFLDF), in two animals. Changes in local cortical perfusion were comparable with changes of regional CBF.
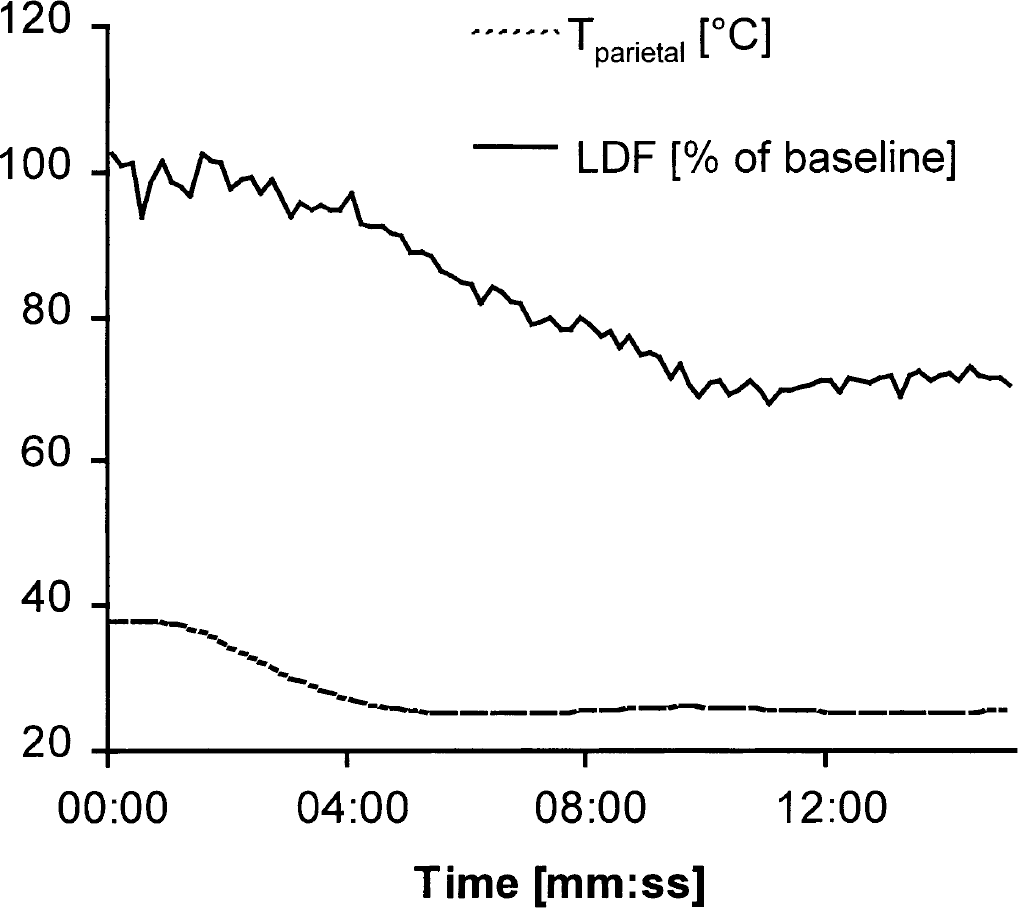
Example registration of changes in cortical blood flow as measured with laser—Doppler flowmetry (LDF), and parietal temperature during cooling of the brain from 38°C to 25°C (stage 1 to stage 2) in one animal (sampling rate: 0.1 Hz).
DISCUSSION
In the current study, the brain of juvenile pigs was cooled selectively in less than 10 minutes to a mean temperature of 25°C using heat exchangers interpositioned into the carotid circulation. Cooling and rewarming of the brain was achieved at normothermic core temperature and without systemic hemodynamic dearrangements. The measured temperature gradients within the forebrain during stages 2 and 3 should be attributed to the admixture of normothermic arterial blood through the vertebral arteries which were not ligated. Methodologic aspects of the experimental design used here and the resulting regional brain temperature profiles have been discussed in detail elsewhere (Kuhnen et al., 1999).
In two pilot experiments we found that routing a single carotid's blood flow through the heat exchanger into the distal segments of both carotid arteries decreased blood pressure as measured in the lingual artery by 2 to 3 mm Hg. This decrease of blood pressure is not considered to be of relevance for CBF regulation. Moreover, our results indicate that neither CBF nor CVR did change in response to the insertion of the heat exchanger into the carotid circulation.
The cerebral and extracerebral effects of systemic hypothermia, with or without cardiocirculatory support, have been extensively investigated. A temperature dependent decrease of CBF and cerebral oxygen metabolism during systemic hypothermia has been consistently reported in human and animal studies (Greeley et al., 1993; Kurth et al., 1998; Michenfelder et al., 1991). Changes in oxygen metabolism are expressed by Q10 values for total CMRO2 ranging between 2.1 and 4.6 (Michenfelder, 1988; van der Linden et al., 1991). However, little is known about the metabolic response of the brain to SBH. Previous studies have focused on the technical feasibility of the respective methods or neurologic outcomes (Ohta et al., 1996, Gunn et al., 1998a; Kuhnen et al., 1999; Wass et al., 1998; White et al., 1969). To our knowledge only one study has been published that documents the effect of conductive SBH on cerebral oxygen metabolism after resuscitation from six minutes of cardiac arrest in infant piglets (Gelman et al., 1996). In that study, cerebral oxygen uptake at 45 minutes of reperfusion was lower in selectively brain cooled animals than in normothermic control animals (44% vs. 69% of baseline, respectively). The decrease of cerebral oxygen uptake in the normothermic group indicated a suppression of cerebral oxygen metabolism obviously caused by the precedent global brain ischemia. A decrease of cerebral energy metabolites and mitochondrial respiration has been measured up to several hours after cerebral hypoxia—ischemia in piglets and in newborn and adult rats (Gilland et al., 1998; Rehncrona et al., 1979; Sutton et al., 1991). Therefore, the data of the interventional study of Gelman et al. (1996) do not allow the separation of the physiologic cerebral responses to SBH from effects of cerebral ischemia caused by cardiac arrest. Our results indicated that the decrease in CMRO2 between stage 1 and stage 2 (38°C/25°C) results in a mean Q10 of 2.8 (2.1 to 3.4) that is comparable to data drawn from studies using systemic hypothermia. During brain rewarming the calculated Q10 value was higher in the temperature range between 25°C and 30°C (Q10 = 4.7) than between 30°C and 38°C (Q10 = 2.1). In adult animals, the increase in Q10 associated with decreasing brain temperature has been explained by the temperature dependency of basal and functional brain metabolism (Klementavicius et al., 1996; Michenfelder et al., 1991). In a previous contribution, in which EEG was quantified by spectral analysis, it was clearly shown that there was a profound reduction of spontaneous brain activity at 25°C. However, a partial restoration of the EEG, predominantly that of the higher frequency band was found at moderate hypothermia of 30°C (Kuhnen et al., 1999). This may explain in part the higher Q10 value at lower brain temperatures measured in the current study. Greeley et al. (1993) reported an increased metabolic suppression in neonates, infants, and children with a mean Q10 of 3.65 perhaps reflecting a greater susceptibility of immature neurons and glial elements to hypothermia. Therefore, maturational differences effecting a change in the relation between functional and basal cerebral metabolism may further account for the higher Q10 value between 25°C and 30°C during rewarming of the brain in our animal model.
The presumption that changes in CBF and CMRO2 values reported between 25°C and 30°C were secondary to deterioration of the preparation with time or to changes in the tissue concentration of isoflurane can be clearly rejected. Cerebral blood flow and CMRO2 returned to baseline levels after rewarming of the brain and the mean Q10 between 38°C and 25°C did not differ for brain cooling (Q10 = 2.8 ± 0.6) and rewarming (Q10 = 2.9 ± 0.7). Hypothermia increases the solubility of isoflurane in blood and brain tissue leading to an increase of anesthetic potency and changes in CBF and CMRO2 (Allott et al., 1973). However, in our experimental model, the blood—gas exchange in the lungs always occurred under normothermic conditions. Therefore, the quantity of isoflurane, which solubilized in the blood and was transported to the brain, did not change throughout the experiment. According to the HENRY—DALTON law, the temperature effect on solubility of isoflurane is equalized by a decrease of partial pressure. Therefore, the number of isoflurane molecules acting at the lipid membranes of the brain tissue should not change during SBH.
CBF and cerebrovascular reactions
The limited number of studies documenting the cerebral hemodynamic consequences of SBH have yielded contradictory results. Experiments using convective brain cooling reported a decrease of CBF with decreasing temperature. Opposite effects have been observed when conductive cooling was applied to cool the brain selectively. Using conductive head cooling, Kuluz et al. (1993) reported an increase in cortical CBF in rats, as measured by laser—Doppler flowmetry (LDF), to 215% of baseline values during selective brain hypothermia (30.9°C). Based on these results, it has been concluded that the method of cooling (whole body hypothermia vs. SBH) may be a key factor in determining the cerebrovascular effects of hypothermia (Dietrich, 1996). In contrast, Schwartz et al. (1996a) reported a decrease of right hemispheric blood flow in baboons to 26 mL min−1 100 g−1 (vs. 69 mL min−1 100 g−1 at baseline) when isolated brain hypothermia (<25°C) was induced by perfusion through a single carotid artery with extracorporally cooled blood. In these experiments the core temperature of the trunk decreased moderately to 34°C during stable deep cerebral hypothermia.
In our study, convective brain cooling decreased CBF. Cerebral hypothermia was achieved without the need for circulatory support or concomitant systemic hypothermia. Additional monitoring of the local cortical perfusion by epidural laser—Doppler flowmetry in two animals clearly showed that the observed changes in local cortical perfusion were comparable with regional CBF changes measured with CMs (Fig. 4). Continuous monitoring of cortical perfusion during cooling of the brain from 38°C to 25°C provided no evidence for temporary increases of cortical blood flow during induction of SBH (Fig. 5). Therefore, the differences in the cerebrovascular response shown in this study compared with the results reported by Kuluz et al. (1993) obviously are not caused by the method of CBF measurement.
The currently available data together with the results presented here suggest that the cerebrovascular response to SBH to a much greater extent may depend on the method of selective cooling than on accompanying changes in core temperature, as long as systemic hemodynamics remain stable.
What may explain the differences in the cerebrovascular response to different methods of SBH? Cooling of the head from the outside produces a centripetal temperature gradient within the brain that results in temperature differences between superficial and deep cerebral structures (Gelman et al., 1997; Wass et al., 1998). It is conceivable that the cerebrospinal fluid of the subarachnoidal space and the outermost layers of the cortex could be cooled to profound hypothermic levels. Therefore, the pial vessels at the surface of the brain could be exposed to much lower temperatures than the vessels inside the brain tissue during external head cooling. Profound levels of hypothermia abolish cerebral autoregulation by producing cerebral vasoparalysis (Greeley et al., 1993; Taylor et al., 1992). Thus, an increase of cortical blood flow in response to conductive (external) head cooling could be caused by centripetal temperature gradients and would be limited to the outer layers of the cerebral cortex. Based on the spatial resolution of temperature and blood flow measurements in our experiments, we were able to demonstrate a remarkable homogeneity of brain cooling (Table 2) and a strong correlation of blood flow and temperature within the forebrain (Fig. 3). Therefore, we conclude that convective brain cooling causes a temperature dependent decrease of CBF during moderate and deep isolated cerebral hypothermia.
More recently, Frietsch et al. (2000) reported that mean CBF was unchanged during moderate systemic hypothermia (32°C) in adult rats under pH-stat ventilatory management. In a recent study, we found that moderate systemic hypothermia (32°C) decreased mean CBF under pH-stat management in infant piglets to 74±20% of baseline values (Bauer et al., 2000). Based on the different findings of these two studies we can not exclude that the cerebrovascular response to brain hypothermia also depends on species or age differences.
Another question we postulated was to which degree CBF is metabolically regulated during SBH. Hypothermia changes flow—metabolism coupling in the brain that results in a mismatch of flow and metabolism favoring perfusion during moderate and deep cerebral hypothermia. This has been demonstrated under cardiopulmonary bypass and nonbypass conditions (Cook et al., 1995; Frietsch et al., 2000; Greeley et al., 1991). In the current study, the ratio of CBF and CMRO2 was unchanged at a Tbrain of 30°C, indicating a coupling of flow and metabolism at this temperature level. The CBF:CMRO2 ratio was increased at a Tbrain of 25°C. The extent of the flow—metabolism mismatch can be calculated when assuming that the flow—metabolism ratio would not change during cooling of the brain to 25°C. Following this theoretical assumption, the flow—metabolism mismatch during stage 2 is caused by an increase of CBF relative to oxygen metabolism amounting 6 mL min−1 100 g−1, that is, 12% of the theoretically expected reduction in CBF. Based on our own data we can not decide if the increase in CBF relative to metabolism represents a luxury perfusion of the brain or if it is a change in coupling of flow and metabolism. A recent study clearly demonstrated that a change in the relation between CBF and glucose metabolism during moderate and mild hypothermia does not necessarily indicate uncoupling because a strong linear relationship between CBF and glucose metabolism is observed on a local level (Frietsch et al., 2000). The data of this study indicate that the physiologic response to cerebral hypothermia is a new equilibrium of CBF and metabolism.
Alternatively, the increase of CBF may be caused by other factors than metabolic regulation. Moderate and deep hypothermic levels may exert a direct effect on cerebral resistance vessels, that is, independent of metabolic autoregulation. Moderate hypothermia dilates cerebral arterioles in vitro (Ogura et al., 1991). This effect may in vivo counteract the metabolically mediated vasoconstriction during moderate and deep cerebral hypothermia.
Hypothermia is associated with increased blood viscosity (Hershenson et al., 1989). Therefore, changes in blood rheology may contribute to the observed decrease of CBF but would not cause critical reductions of CBF.
However, independent of these considerations our data indicated a strong coupling of CBF and CMRO2 on a global level (Fig. 3). Therefore, metabolic regulation is a main determinant of CBF decrease during SBH.
From the data obtained by analysis of regional temperature changes and corresponding changes in regional perfusion we have shown that temperature and perfusion remained coupled on a regional level during deep cerebral hypothermia (Fig. 3). Assuming that a flow—metabolism coupling also accounts for a mean brain temperature of 25°C, we can argue that changes in regional blood flow are directly related to changes in regional metabolism and vice versa that temperature dependent changes in regional metabolism are a main determinant of the cerebrovasular response on a regional level.
In summary, we have shown that the forebrain of juvenile pigs can be cooled selectively by perfusion of both carotid arteries with extracorporally cooled blood. Deep levels of cerebral hypothermia can be induced by conductive SBH at normothermic trunk temperature and without hemodynamic derangements. Selective brain hypothermia decreases cerebral oxygen uptake and CBF to an extent that is comparable to the effects of systemic hypothermia. During moderate SBH, the CBF is metabolically regulated. Deep hypothermia results in a change in coupling of blood flow and metabolism favoring perfusion. Regardless of this change in flow—metabolism coupling, metabolic regulation remains a main determinant of CBF even during deep cerebral hypothermia. Moreover, brain temperature and perfusion are coupled within the forebrain on a regional level. Thus, metabolic regulation is also a main determinant of regional CBF during deep SBH.
Footnotes
Acknowledgments
The authors thank E.S. El Hallagh, I. Witte, U. Jäger, P. Volk, L. Wunder, Y. Mondorf, and M. Holzmayr for their technical assistance.