Abstract
Surgical flow augmentation for treatment of cerebral hemodynamic impairment remains controversial. Here, we investigated the benefit of endothelial progenitor cell (EPC) treatment in a rat model of chronic cerebral hypoperfusion. At repeated time points after 3-vessel occlusion (3-VO), animals were treated with 1 × 10
6
Dil-labeled (a)
Keywords
INTRODUCTION
Chronic impairment of blood supply to the brain due to arterial steno-occlusive disease contributes to ischemic stroke in ~10% of all cases. 1 Owing to the slow disease progression, patients typically present with transient ischemic symptoms before the manifestation of brain infarction. Here, the outgrowth of preformed collateral vessels (arteriogenesis) is the most important endogenous rescue system to cope with the chronic cerebral hypoperfusion. However, the natural collateral network is often not efficient enough to compensate the progressive disease. 2
Arteriogenesis is regulated by molecular and cellular mechanisms, which can be manipulated to induce and accelerate the outgrowth of preexisting collaterals.
3
This concept of therapeutic stimulation of collateral vessel growth has recently been adapted to the cerebrovascular system.4,5 In contrast, postnatal vasculogenesis describes the
Against this background, several experimental and clinical studies have reported beneficial implications of EPC to stimulate postnatal neovascularization in peripheral6–8 and coronary artery disease.9,10 However, little is known about the contribution of vasculogenesis and its main effectors—EPC—to collateral vessel formation in the cerebrovascular system.
In this study, we investigated whether
MATERIALS AND METHODS
Ethics Statement
Experiments were permitted by the local ethics committees on animal research (Regierungspräsidium Karlsruhe, AZ: 35–9185.81/G-105/04, Karlsruhe, Germany and LaGeSo No. G 0262/07, Berlin, Germany) and in conformity with the German Law for Animal Protection and the National Institute of Health Guidelines for Care and Use of Laboratory Animals.
Cell Culture Experiments
In terms of cell therapy aimed at revascularization, one advantage of e-EPC over differentiated endothelial cells is the higher plasticity of e-EPC. Thus, it is important to provide evidence that the e-EPC used in this study acquire endothelial cell (human umbilical vein endothelial cells; HUVEC) characteristics after differentiation to e-EPDC and that e-EPDC have the same characteristics as differentiated endothelial cells (HUVEC). Before our animal experiments, we therefore studied e-EPDC and e-EPC in tube formation assays on Matrigel (BD Biosciences, Heidelberg, Germany) with and without coculture of HUVEC.
Isolation and Labeling of Human Umbilical Vein Endothelial Cells, Embryonic-Endothelial Progenitor Cells and Embryonic-Endothelial Progenitor-Derived Cells
Human umbilical vein endothelial cells were used for
The isolation and
In vitro Tube Formation Assays
Matrigel was thawed on ice at 2°C to 8°C and kept on ice during preparation. Matrigel was then added to the wells of a 96-well plate in a volume of 50 μL and allowed to solidify at 37°C for 30 minutes. Embryonic-endothelial progenitor cells (10 × 10
4
cells/mL;
For coculture experiments (
In an additional set of experiments (
Animals and Experimental Design
Forty-eight adult, male Sprague-Dawley rats (body weight 296 to 332 g) were randomized to the following groups:
(a) Embryonic-endothelial progenitor cells ( (b) Embryonic-endothelial progenitor-derived cells ( (c) Saline (
Intravenous injections via the tail vein were performed immediately after and on days 7 and 14 after induction of chronic cerebral hypoperfusion. Each procedure was performed under general anesthesia with intraperitoneally applied chloral hydrate (04 g/kg bw). Postoperatively, animals received a subcutaneous injection of 30 μg/kg bw buprenorphine (Temgesic, Essex Pharma, Munich, Germany) for postoperative analgesia. During surgery, body temperature was maintained at 37°C using a temperature-controlled heating pad. All data regarding CVRC assessment, latex perfusion, and histology were obtained and analyzed in a masked fashion.
3-Vessel Occlusion
Rats were immobilized in the prone position with a stereotactic head fixation device. First, the cervical spine and the atlanto-occipital junction were exposed via a paravertebral access and the vertebral arteries were bilaterally cauterized according to Pulsinelli and Brierley. 14 Next, the animals were turned to supine position and the right common carotid artery was exposed through a midline neck incision. The common carotid artery was carefully dissected from the vagal nerve and permanently ligated with an 8–0 silk suture. The skin was sutured with 6–0 nylon.
Measurement of the Cerebrovascular Reserve Capacity
Measurements of the CVRC were performed immediately before (day 0) and on days 7 and 21 after 3-VO (saline:
Assessment of Parenchymal Capillary Density and the Localization of Dil-Positive Cells
For histologic assessment of the parenchymal capillary density and the localization of Dil-positive cells on day 21, six animals of each group received 1.5 mL of Evans Blue (Sigma-Aldrich, 2% dissolved in saline) via the tail vein. After a 90-second perfusion period, the rats were killed and the brains were removed and frozen. From each brain, 10 μm coronal cryosections were obtained from bregma level −0.4 mm to −6.4 mm in 2 mm intervals. The sections were mounted and observed under a fluorescence-enhanced microscope (Axioplan 2, Zeiss, Oberkochem, Germany). For each section level, specific cortical and subcortical brain areas were analyzed (–0.4 mm: caudate putamen; −2.4 mm: primary sensory cortex (S1); −4.4 mm, hippocampus (Ca1); −6.4 mm, primary auditory cortex (Au1)). Capillaries were defined as a round shape or transverse line with a maximum diameter of 8 μm and capillary vessel density was assessed as number of vessels per field of view. Embryonic-endothelial progenitor cells and e-EPDC were identified according to Dil-positive fluorescence.
Assessment of the Cerebral Collateral Vessel Growth by Latex/Carbon Black Perfusion
In a separate set of animals (
Body weight, MAP, and arterial blood gases on day 21
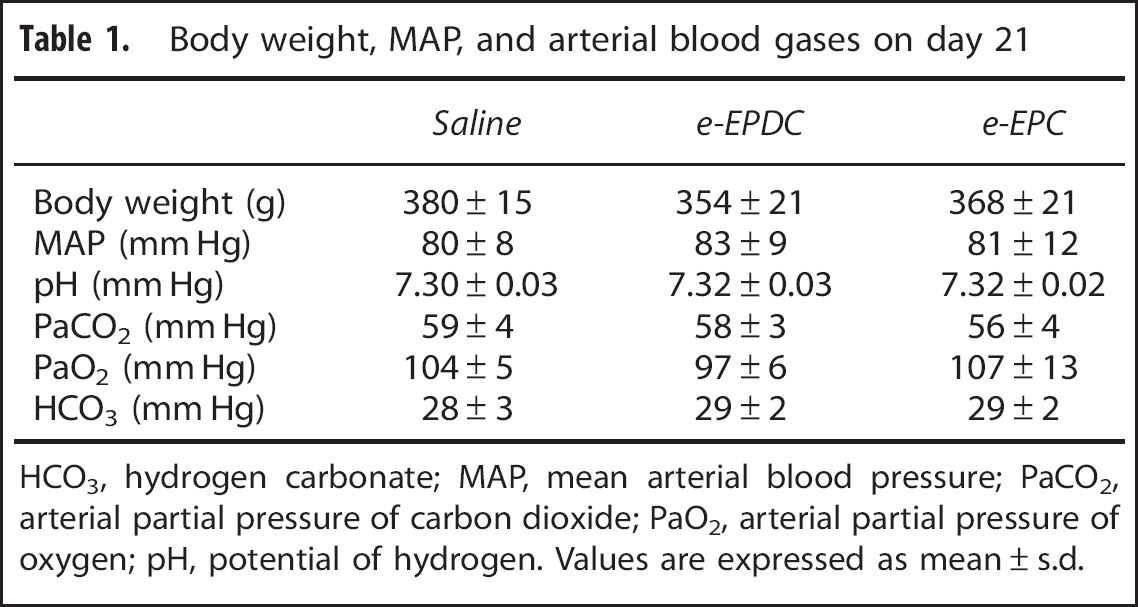
HCO3, hydrogen carbonate; MAP, mean arterial blood pressure; PaCO2, arterial partial pressure of carbon dioxide; PaO2, arterial partial pressure of oxygen; pH, potential of hydrogen. Values are expressed as mean ± s.d.
Measurements of the external diameters of the basal vasculature (anterior cerebral artery (ACA), middle cerebral artery (MCA), internal carotid artery (ICA), posterior cerebral artery (P1 segment), posterior communicating artery and basilar artery) and the diameter and number of leptomeningeal anastomoses of both hemispheres were performed with freeware image analysis software (lmageJ64, http://rsbweb.nih.gov/ij/) by a masked observer: The diameters of the ACA, MCA, posterior communicating artery and P1 segment were determined in the middle third of the vessel between its origin and next branching point or confluence (i.e., the ACA diameter was determined between its origin from the ICA and the just proximal point where ACA diverges the olfactory artery). The ICA diameter was determined just proximal to the ICA bifurcation. The basilar artery diameter was determined just proximal to the origin of the superior cerebellar arteries. In each vessel, three measurements were performed and the mean of these three values per vessel was compared between all groups. Leptomeningeal anastomoses were defined and counted at their point of confluence between the distal MCA and the distal ACA or between the distal MCA and the posterior cerebral artery. The mean of the total number of anastomoses was compared between all groups. The diameter of each anastomosis was obtained as the average of three measurements along the middle third of the collateral. The mean of these three values per vessel was used to compile relative frequency histograms of vessel diameters for each group.
Intravital Fluorescence Video Microscopy
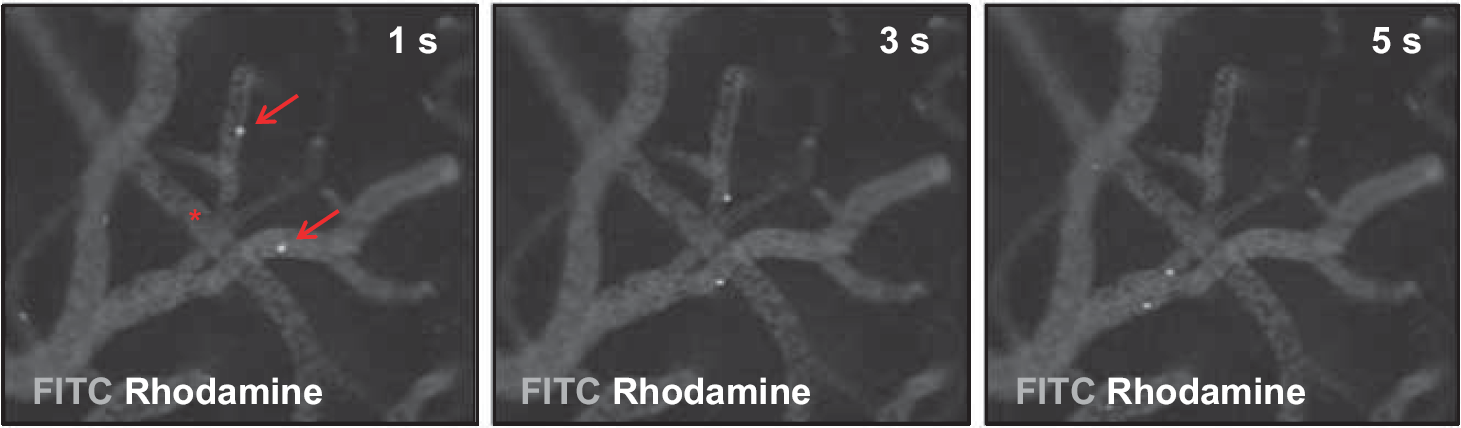
In a separate set of animals (
Statistical Analysis
Data are presented as mean ± s.d. For comparison of CVRC after 3-VO, a two-way analysis of variance procedure for repeated measures with subsequent pair-wise comparison of means by Fisher's least projected difference test was performed. Differences in physiologic parameters, basal vessel diameters, the total number of leptomeningeal anastomoses, and the parenchymal capillary density were tested by a one-way analysis of variance and subsequent Bonferroni correction (two-tailed) for multiple comparisons. The relative frequency of leptomeningeal vessel diameters was compared by a two-way analysis of variance and subsequent Bonferroni correction. All statistics were generated with GraphPad Prism for Mac (Version 5.0d, GraphPad Software, San Diego, CA, USA). Statistical significance was set at
RESULTS
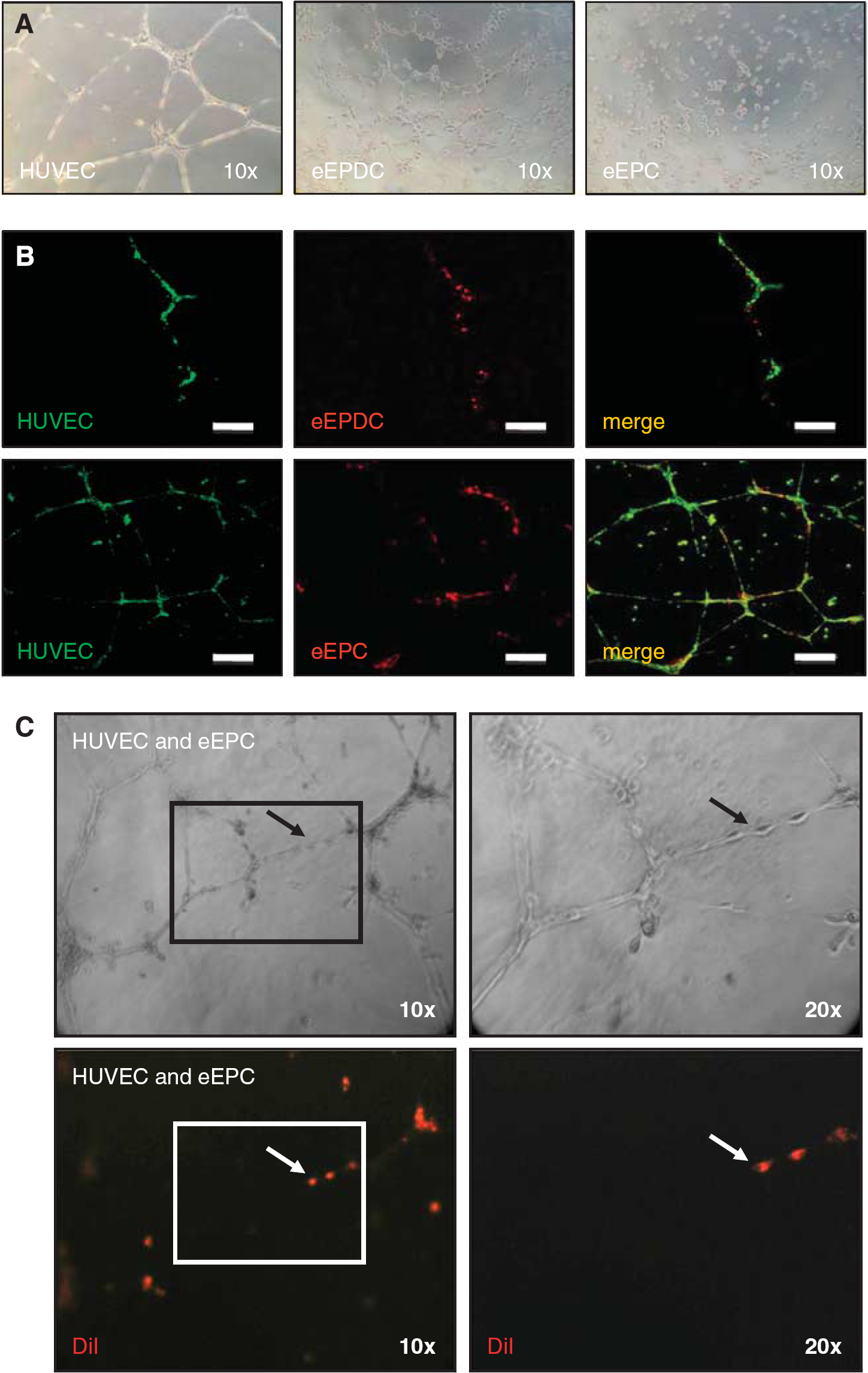
Embryonic-Endothelial Progenitor Cells Do not Form Cord-Like Structures In Vitro but Incorporate into a Vascular Network
Embryonic-endothelial progenitor-derived cells (e-EPC stimulated with cAMP and retinoic acid) spontaneously formed cord-like structures on Matrigel, similar to HUVEC (Figure 1A). Embryonic-endothelial progenitor-derived cells incorporation into a developing HUVEC network (Figure 1B, upper panels) further confirmed that e-EPDC behave as differentiated endothelial cells and may serve as a biologic control. In contrast, e-EPC cultured alone failed to form tubes (Figure 1A). However, when cocultured, immediately or 5 hours after HUVEC were taken into culture, e-EPC elongated and became an integral part of the developing (Figure 1B, lower panels) or preformed (Figure 1C) HUVEC network as a sign of high plasticity and that e-EPC acquire endothelial cell characteristics after differentiation.
Improved Functional Recovery of the Cerebrovascular Reactivity After Treatment with embryonic-endothelial progenitor cells
Graph traces of cerebral blood flow responses to acetazolamide stimulation on day 21 are shown in Figure 2A. On days 7 and 21 after 3-VO, CVRC was significantly higher after treatment with e-EPC compared with saline- and e-EPDC-treated animals (day
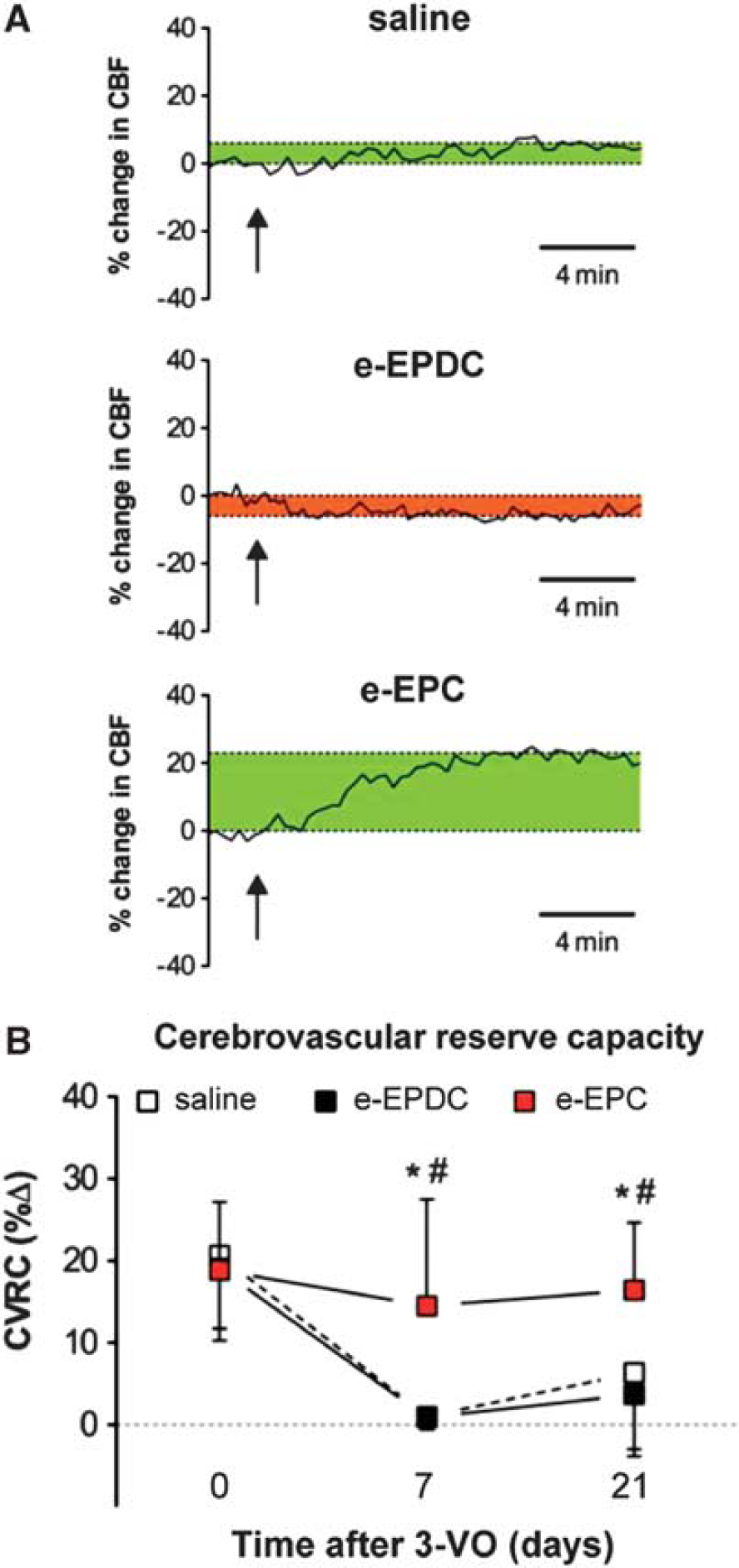
Functional recovery of the cerebrovascular reserve capacity (CVRC). (
Before 3-VO (day 0), CVRC did not differ between groups (saline: 20.7 ± 9%; e-EPDC: 19.1 ± 9%; e-EPC 18.9 ± 8%). Seven days after 3-VO, CVRC was significantly reduced in animals treated with saline (1.1 ± 2%,
Embryonic-Endothelial Progenitor Cells Augment Basal and Leptomeningeal Collateral Vessel Growth
On day 21, treatment with e-EPC resulted in a significant increase of the external vessel diameters of the MCA and ICA segments in the anterior circulation compared with animals treated with saline, whereas e-EPDC-treated animals only showed a significant increase of the external vessel diameter of the PI segment of the posterior cerebral artery (Figure 3; *
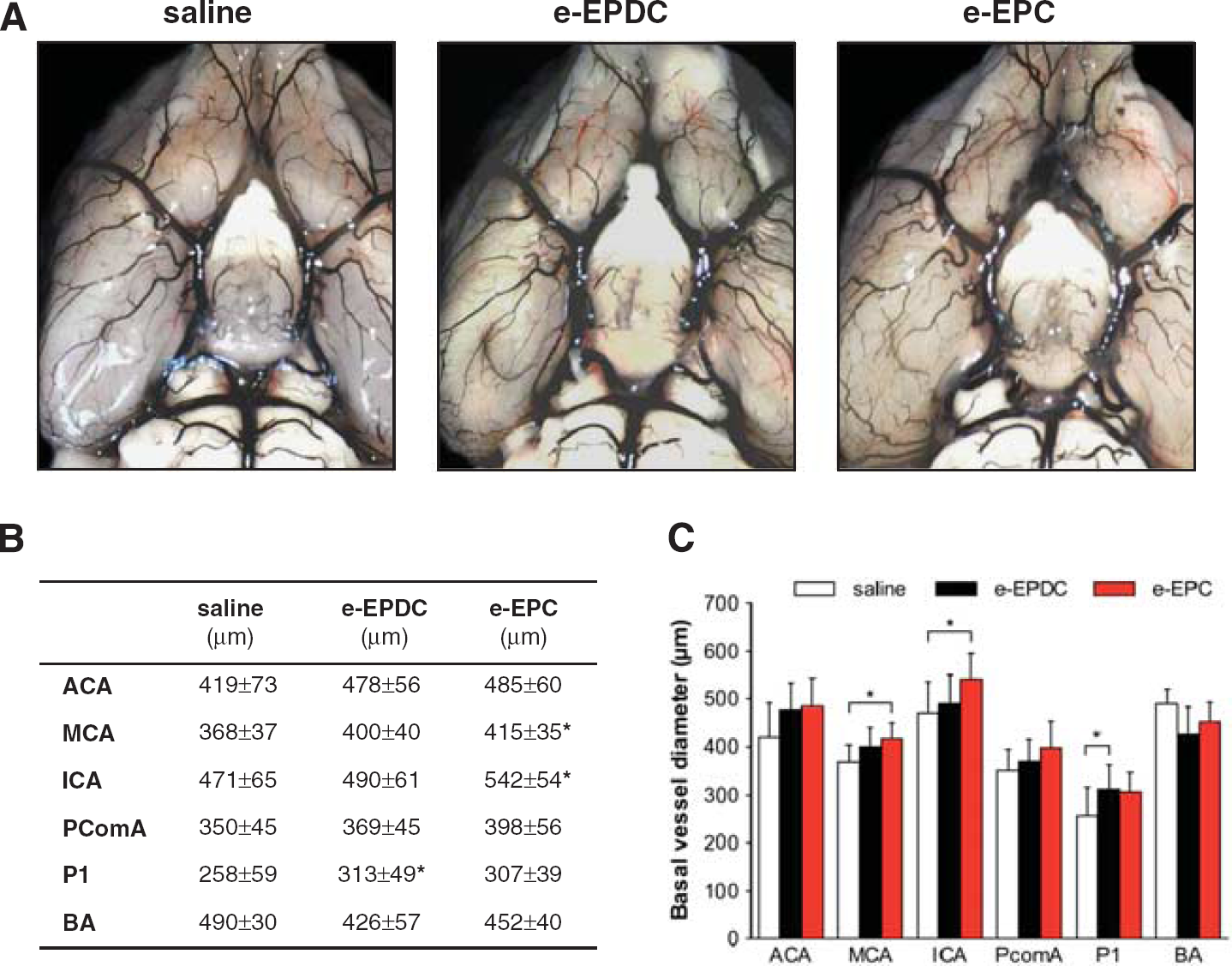
Collateral vessel growth—basal vasculature. (
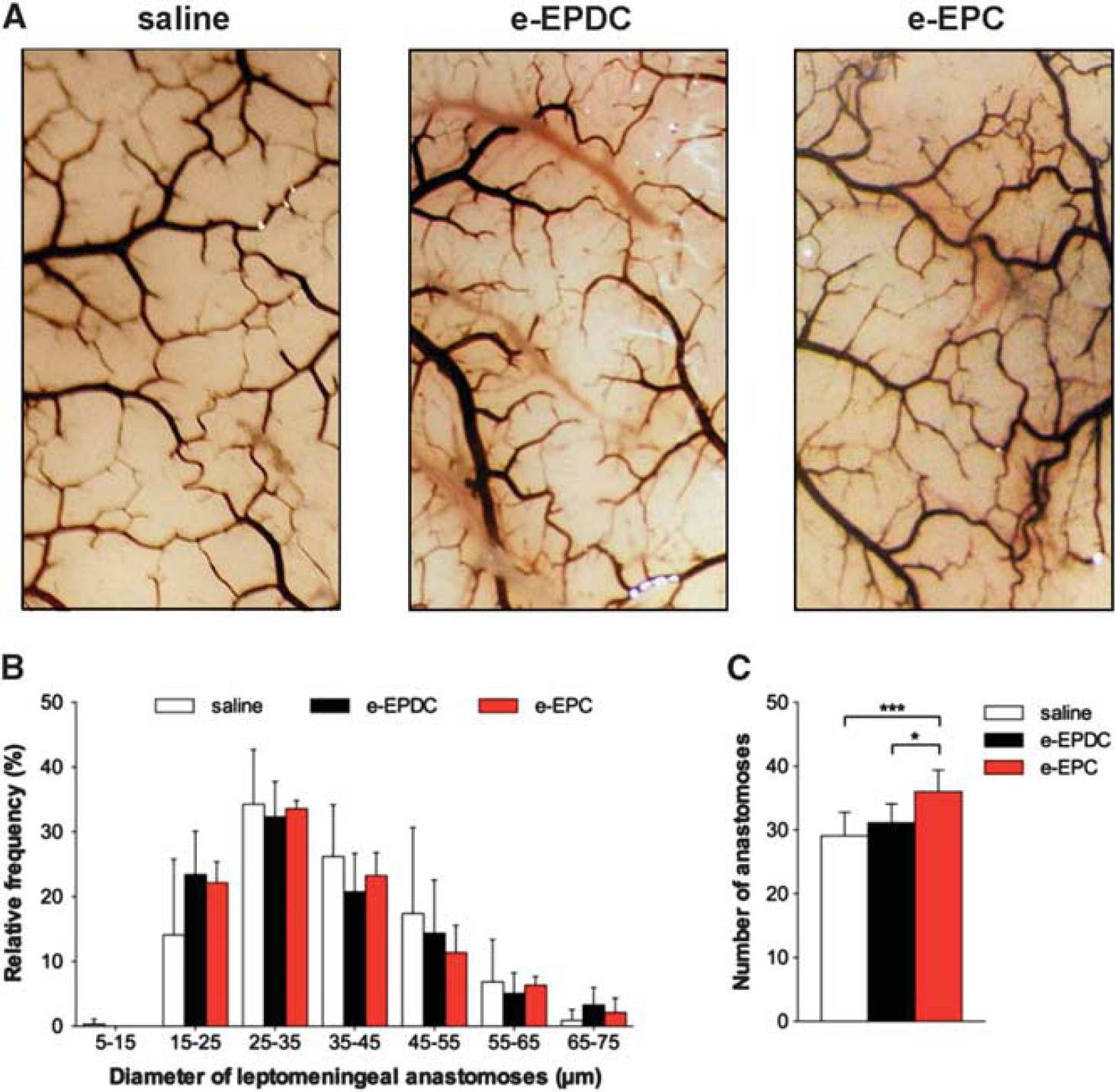
Collateral vessel growth—leptomeningeal anastomoses. (
Increased Cortical Parenchymal Capillary Density after Treatment with Embryonic-Endothelial Progenitor Cells
On day 21, treatment with e-EPC led to a significantly higher density of patent capillaries within the cortical S1 area compared with animals treated with e-EPDC or saline. In the subcortical CA1 region of the hippocampus, no difference in capillary density was observed (Figure 5).
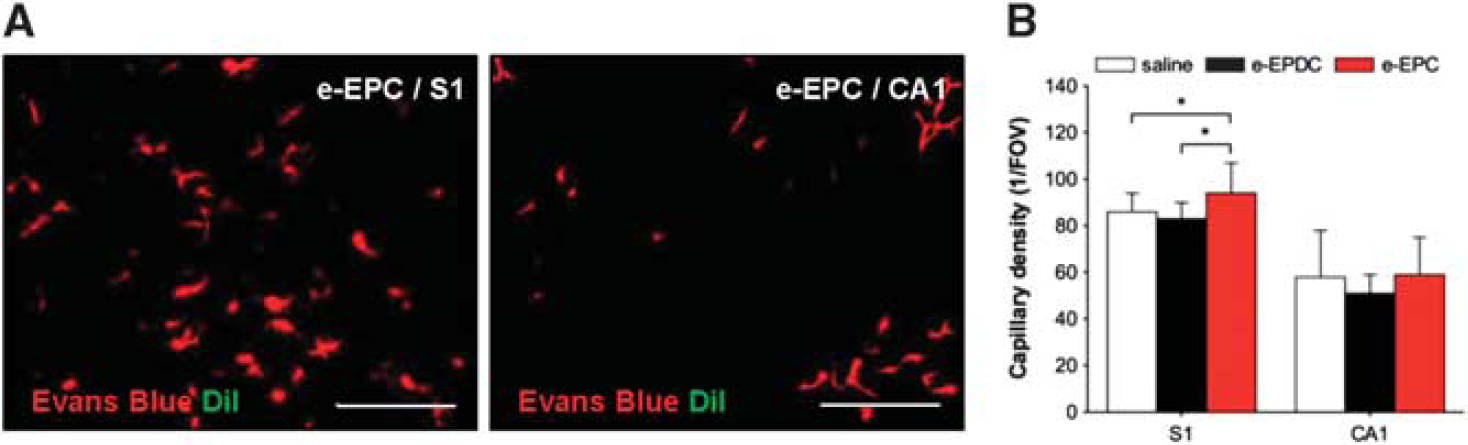
Parenchymal microvascular density and embryonic-endothelial progenitor cells (e-EPCs) incorporation on day 21. (
Embryonic-Endothelial Progenitor Cells do not Incorporate into the Cerebral Vasculature In Vivo in Chronic Cerebral Hypoperfusion
On day 21, histologic cryosections failed to show Dil-positive e-EPC within the cerebral vasculature or the perivascular niche (Figure 5). Interestingly, intravital fluorescence video microscopy detected a transit of circulating e-EPC in the cortical microvascular environment immediately after e-EPC injection after 3-VO on day 0 (Figure 6). Although interaction of the Dil-labeled cells with postcapillary venules was noted, e-EPC transmigration into the perivascular niche or incorporation into the existing arterial/arteriolar vasculature was not observed.
DISCUSSION
In the present study, we demonstrate that e-EPC have potential for therapeutic stimulation of collateral vessel growth in the cerebrovascular system. Compared with their differentiated biologic controls (e-EPDC) and saline-treated animals, e-EPC (I) restored hemodynamic impairment, (II) increased basal and leptomeningeal collateralization and parenchymal capillary density but (III), did not transmigrate and incorporate into existing arterial/arteriolar brain vessels despite positive
Therapeutic Stimulation of Collateral Vessel Growth in Chronic Cerebral Hypoperfusion
Hemodynamic impairment is mirrored in an abolished CVRC, which correlates with the risk of subsequent ischemic stroke. 16 Hemodynamic rescue is a result of augmented collateral flow and expressed as an improvement of CVRC. According to the literature, bypass grafting remains controversial and does not appear to be the way to effectively improve hemodynamic reserve in patients with typical arteriosclerotic symptomatic ICA occlusion.17,18 Thus, there is a need to develop safe and technically robust revascularization techniques.
The cerebrovascular system underlies permanent vascular remodeling to maintain adequate tissue perfusion under physiologic and pathologic conditions but the development of new blood vessels in the chronically hypoperfused brain is poorly understood. Adaptation via neovascularization may be achieved by different mechanisms. Sprouting of newly formed capillaries from preexisting ones (angiogenesis) is mainly driven by hypoxia and does not significantly contribute to the gross tissue perfusion. Arteriogenesis, however, is characterized as the positive outgrowth of preexisting collaterals at the level of the conductance or resistance vasculature, which coincides with an augmentation of collateral flow.4,5,19 Against this background, we investigated EPC as a novel treatment modality for chronic cerebral hypoperfusion because EPC secrete various proangiogenic growth factors,20–22 which may be beneficial in chronic steno-occlusive cerebrovascular disease where neovascularization in terms of collateral vessel growth is required in regions apart from the ischemic tissue. Moreover, EPC are characterized by high plasticity and promigratory activity with the potential for active incorporation into a developing vasculature.9,23,24 In an experimental model, this hypothesis must be tested against differentiated endothelial cells; although saline may serve as a sufficient control from a purely therapeutic point of view, it does not from a biologic/mechanistic one. In the present study, we therefore used e-EPC because they harbor the advantage of easy proliferation and maintenance in culture, possible genetic manipulation and the hallmark of
Functional Recovery of Hemodynamic Impairment after Treatment with Embryonic-Endothelial Progenitor Cells
The perfusion response (CVRC) after application of a vasodilatory stimulus has previously been used as a functional readout by others and our group with comparable results.4,26 Occlusion of three of the four major brain-feeding arteries leads to a completely suppressed CVRC, which continued for the observation period of 3 weeks. Intravenous application of e-EPC, however, improved CVRC within 1 week of treatment. This time interval might seem short, taking into account that for functional recovery structural remodeling is necessary. Some previous studies evaluated arteriogenic effects after longer time intervals—for example, after systemic administration of growth factors—suggesting a long-term recovery effect with the requirement of a longer time interval to permit sufficient structural remodeling.4,5 Other studies, however, also demonstrated structural changes as soon as 1 week after conductance vessel occlusion27,28 and recent findings from our group on vascular remodeling in a mouse model of chronic cerebral hypoperfusion are in line with these results. 19 Next to the structural remodeling, functional improvement of the vasodilation response could additionally contribute to the higher CVRC after treatment with e-EPC.29,30
Augmentation of Basal and Leptomeningeal Collateralization after Treatment with Embryonic-Endothelial Progenitor Cells
In an experimental model of peripheral artery disease, rabbits with chronic hind limb ischemia treated with the mouse e-EPC from the present study showed significantly better collateralization, higher capillary density and better perfusion.
20
In the brain, therapeutic stimulation of collateral vessel growth at the level of the basal vasculature was initially reported by Buschmann
Leptomeningeal anastomoses are the main collaterals to compensate for restricted blood flow distal to the Circle of Willis. Beyond their function of regulating vascular resistance,32,33 these vessel segments also improve functional recovery of an impaired CVRC after granulocyte macrophage colony-stimulating factor treatment in rat and mouse models of chronic cerebral hypoperfusion.5,34,35 In our study, e-EPC resulted in a significant 20% increase of the total number of anastomoses, whereas the diameter of these vessel segments remained unchanged. This increase of the number of anastomoses leads to an increase of the total cross-sectional vessel area and thus, may improve the total conductance of the brain vasculature. Although this finding is in line with our previous report, 5 it differs from published mouse model studies,34,35 where a primary increase in the diameter but not the number of anastomoses was detected. Possibly, this difference in leptomeningeal outgrowth may be due to differences in the animal strain but more importantly, different vessel occlusion techniques in these studies may influence collateral outgrowth due to different perfusion pressure gradients across the leptomeningeal vasculature.
Next, we investigated the cerebral angioarchitecture beyond the basal and leptomeningeal vasculature and detected a higher density of patent capillaries in the cortical S1 area of the MCA territory. Although the functional contribution of parenchymal microcirculatory flow redistribution remains unknown, parenchymal angiogenesis was also observed in our mouse model of chronic cerebral hypoperfusion. 19 Regardless, the present findings should be interpreted with caution because an increased microvascular density could also be a sign of post-ischemic angiogenesis. However, hypoxic tissue injury after 3-VO was previously excluded and thus, the increased microvascular density more likely suggests an activation of parenchymal angiogenesis.
In Vivo and In Vitro Recruitment of Embryonic-Endothelial Progenitor Cells
After intravenous injection, EPCs mainly accumulate in the liver, lung, spleen, and kidney but not in the brain. 36 To rule out an influence of the injection site on this e-EPC distribution pattern, we compared intravenous (tail vein, jugular vein) and intraarterial (carotid artery) e-EPC injections in preliminary experiments in animals without 3-VO. In all cases, we found similar e-EPC distribution patterns demonstrating positive e-EPC cell clusters in the liver and the lung but not in the brain (data not shown). In this regard, a number of publications has provided evidence that EPC exert their effect via paracrine/endocrine signaling mechanisms and that direct EPC incorporation into sites of neovascularization and collateral outgrowth is dependent on the underlying pathology and may not be required in all cases.20–22 Our observed functional and morphologic improvement despite negative e-EPC incorporation into the cerebral vasculature or the perivascular niche supports this hypothesis.
Still, we were somewhat surprised that we detected no sign of e-EPC incorporation, because experimental stroke studies reported EPC incorporation into sites of neovascularization as a sign of postnatal vasculogenesis.37–39 However, this effect was observed in the ischemic zone and within the penumbra of the infarct, an area with ischemic/hypoxic damage and altered blood–brain barrier function. The blood–brain barrier might be one explanation for this phenomenon, because an intact blood–brain barrier could hamper e-EPC host vessel incorporation and vascular transmigration. The fact that we were able to confirm e-EPC interaction with the endothelium of a preformed vascular bed
Together, our findings underline that treatment of chronic cerebral hypoperfusion with e-EPC appears to rely on mechanisms other than direct e-EPC integration alone. Although some interaction of e-EPC with the cerebral endothelium was observed
Footnotes
The authors declare no conflict of interest.
