Abstract
The cerebral metabolism of lactate was investigated. Awake mice received [3-13C]lactate or [1-13C]glucose intravenously, and brain and blood extracts were analyzed by 13C nuclear magnetic resonance spectroscopy. The cerebral up-take and metabolism of [3-13C]lactate was 50% that of [1-13C]glucose. [3-13C]Lactate was almost exclusively metabolized by neurons and hardly at all by glia, as revealed by the 13C labeling of glutamate, γ-aminobutyric acid and glutamine. Injection of [3-13C]lactate led to extensive formation of [2-13C]lactate, which was not seen with [1-13C]glucose, nor has it been seen in previous studies with [2-13C]acetate. This formation probably reflected reversible carboxylation of [3-13C]pyruvate to malate and equilibration with fumarate, because inhibition of succinate dehydrogenase with nitropropionic acid did not block it. Of the [3-13C]lactate that reached the brain, 20% underwent this reaction, which probably involved neuronal mitochondrial malic enzyme. The activities of mitochondrial malic enzyme, fumarase, and lactate dehydrogenase were high enough to account for the formation of [2-13C]lactate in neurons. Neuronal pyruvate carboxylation was confirmed by the higher specific activity of glutamate than of glutamine after intrastriatal injection of [1-14C]pyruvate into anesthetized mice. This procedure also demonstrated equilibration of malate, formed through pyruvate carboxylation, with fumarate. The demonstration of neuronal pyruvate carboxylation demands reconsideration of the metabolic interrelationship between neurons and glia.
Lactate is of interest as an energy substrate for the brain. It accumulates in the brain during intravenous infusion (Dager et al., 1997) and is metabolized through the cerebral tricarboxylic acid (TCA) cycle (O'Neal and Koeppe, 1966). During hypoglycemia, lactate can sustain cerebral function in humans and animals (Thurston et al., 1983; Maran et al. 1994), and in vitro studies suggest that lactate is an important energy source for neurons after cerebral hypoxia (Schurr et al., 1997).
Astrocytes are believed to rely on glycolysis for the generation of ATP for Na+/K+-ATPase, and the lactate thus formed is thought to be released to the extracellular fluid to support neuronal oxidative metabolism (for review, see Tsacapoulos and Magistretti, 1996; Magistretti et al., 1999). Furthermore, glial cells may convert glutamate and TCA cycle intermediates into pyruvate and lactate (Cheng and Nakamura, 1972; Hassel and Sonnewald, 1995a; McKenna et al., 1996), which could be transferred to neurons for oxidative metabolism. In addition to the mentioned observations, a more detailed picture of the cerebral metabolism of lactate is needed.
The cerebral metabolism of 13C-labeled energy substrates can be studied quantitatively with 13C nuclear magnetic resonance (NMR) spectroscopy, by which the 13C enrichment of individual carbon positions in amino acids and other metabolites can be determined (for review, see Bachelard and Badar-Goffer, 1993). Because the labeling of the various carbon positions of amino acids depends on different enzymatic pathways, some of which are purely glial or neuronal, it is possible to deduce in great detail the metabolic fate of the 13C-labeled substrate (Cerdan et al., 1990; Fitzpatrick et al., 1990; Mason et al., 1992; Shank et al., 1993; Lapidot and Gopher, 1994; Hassel et al., 1995; 1997).
The current investigation studies the cerebral metabolism of lactate and compares it to that of glucose using [3-13C]lactate and [1-13C]glucose, both of which yield [3-13C]pyruvate, in conjunction with 13C NMR spectroscopy. From the 13C labeling of cerebral metabolites, inferences were made about which enzymatic pathways were operative in the metabolism of [3-13C]lactate. Therefore, the activities of certain enzymes (malic enzyme, fumarase, lactate dehydrogenase, 2-oxoglutarate dehydrogenase) were measured in cytosolic and mitochondrial/synaptosomal fractions from mouse brain. When it appeared likely that the metabolism of lactate involved pyruvate carboxylation in neurons, [1-14C]pyruvate, which labels aspartate, glutamate, and glutamine only if it is carboxylated to malate or oxaloacetate, was injected intrastriatally into anesthetized mice, and the radiolabeling of amino acids was determined. If [1-14C]pyruvate is metabolized primarily in the neuronal compartment, the specific activity of glutamate exceeds that of glutamine. If, on the other hand, the substrate is metabolized in glia, glutamine achieves the higher specific activity (O'Neal and Koeppe, 1966; Van den Berg et al., 1969; Hassel et al., 1992). This is so because glutamine synthase is expressed in glia and not in neurons (Martinez-Hernandez et al., 1977).
Notice that for [1-14C]pyruvate to label glutamate and glutamine, the [1-14C]malate formed through pyruvate carboxylation needs to equilibrate with fumarate, which leads to randomization of label between the two carboxylic groups. Without such randomization, the labeled carboxylic group in [1-14C]malate will be lost as CO2 in the formation of α-ketoglutarate from isocitrate. This was an advantage in the current study, since equilibration of malate with fumarate was assumed to be an important step in the metabolism of [3-13C]lactate.
MATERIALS AND METHODS
Materials
Female NMRI mice (Bomholt, Ry, DK), 25 to 30 g body weight, had free access to food and tap water. The air humidity was 50%, and the light-dark cycle was 12 hours. Some animals were fasted overnight before experiments but had free access to tap water.
Sodium [3-13C]lactate, 99% 13C enrichment, was from Isotec (Miamisburg, OH, U.S.A.), [1-13C]glucose, 99% 13C enrichment, was from Sigma (St. Louis, MO, U.S.A.). Before experiments, 0.5 mol/L solutions of [3-13C]lactate and [1-13C]glucose were made with double-distilled water, and pH was adjusted to 7.3. 3-Nitropropionic acid (NPA; Sigma) was dissolved in double-distilled water (20 mg/mL) and was neutralized with NaOH immediately before experiments. All chemicals for the enzyme assays were from Sigma.
Experimental protocols
Fed or fasted mice received 0.3 mL of the sodium [3-13C]lactate solution (150 μmol pyruvate equivalents) intravenously in a tail vein over approximately 10 seconds. This concentration and dose (per kg of body weight) of sodium lactate are the same as those used in humans for intravenous infusion over 20 minutes (Pitts and McClure, 1967; Knott and Lapierre, 1986). Care was taken to avoid injecting too rapidly, since in a preliminary experiment this had caused short-lasting muscle paralysis and respiratory arrest. To study the dose-labeling relationship for [3-13C]lactate, two mice received 0.3 mL of a 0.25-mol/L sodium [3-13C]lactate solution-half of the ordinary dose-and were killed at 5 minutes.
Fasted mice received 0.3 mL of the [1-13C]glucose solution (150 μmol) intravenously. This corresponds to 300 μmol pyruvate equivalents, of which 150 μmol are [3-13C]pyruvate. To study the effect of lactate on cerebral glucose metabolism, fasted mice received 0.6 mL unlabeled sodium lactate, 1 mol/L, subcutaneously. At 25 minutes, the animals received 0.3 mL of the [1-13C]glucose solution intravenously.
A group of fasted mice received NPA, 180 mg/kg subcutaneously to inhibit succinate dehydrogenase in the brain (Alston et al., 1977; Hassel and Sonnewald, 1995b). At 15 minutes, the animals received 0.3 mL (0.5 mol/L) [3-13C]lactate intravenously, and they were killed after another 5 minutes.
The animals (four to six in each group) were returned to their home cages after injection. At 5 or 15 minutes after injection of 13C-labeled substrates, they were killed by cervical dislocation and rapid decapitation so that the heads fell into liquid N2. Blood was collected from the severed vessels and kept on ice. The brains were homogenized in 4 mL of 3.5% (vol/vol) ice-cold perchloric acid. Ten brains were weighed in the frozen state before being homogenized and gave 404 ± 30 mg. This value was used to calculate the amount of 13C-labeled metabolites in the cerebral blood, which was present in the brain extracts (see Discussion). Protein was removed by centrifugation (13,000 × g for 20 minutes), resuspended in 30 mL of sucrose (0.32 mol/L), and assayed according to Lowry et al. (1951) using ovalbumin (Sigma) as standard. The perchloric acid was precipitated by KOH, 10 mol/L, and the KClO4 was removed by centrifugation (13,000 × g for 20 minutes). The volumes of the extract were measured accurately, and 450 μL were taken for the analysis of glucose, lactate, and amino acids by spectrophotometry or HPLC with fluorescence detection as described previously (Hassel et al., 1997). The rest was lyophilized to dryness and redissolved in 600 μL D2O with dioxan, 0.025%, as internal standard, for 13C NMR spectroscopy (see later). The blood samples were centrifuged at 3000 × g for 15 minutes, and serum was removed for analysis of glucose and lactate; then 1 mL perchloric acid, 7% (vol/vol), was added, and protein was removed by centrifugation. The perchloric acid was removed by KOH precipitation as described earlier. The volumes of the extracts were measured, and 200 μL were used to determine the total amount of glucose and lactate in the extracts; the rest was lyophilized to dryness and used for 13C NMR spectroscopy.
13C NMR spectroscopy
Inverse-gated 13C NMR spectroscopy was performed on a Bruker Avance DRX 500 (125 MHz). The number of scans per spectrum was 10,240, using a 30° pulse angle and 39.6 kHz spectral width with 64k data points. The acquisition time was 0.8258 second, and the relaxation time was 3 seconds. Thus, spectra were broad-band proton-decoupled only during acquisition to avoid nuclear Overhauser effects. Equimolar solutions of the metabolites of interest were run as external standard. The solutions contained unlabeled glutamate, γ-aminobutyric acid (GABA), aspartate, glutamine, alanine, lactate, glucose, 90.9 μmol of each compound, and 22.7 μmol dioxan, which gives 1 μmol 13C per peak in the 13C NMR spectrum. From the integrals of the peaks, correction factors with respect to dioxan were calculated for each carbon position. The validity of the correction factors has been confirmed by comparison of the 13C labeling of the carboxyl groups glutamate C-5, GABA C-1, and glutamine C-5 from [2-13C]glucose with the labeling of methylene groups glutamate C-4, GABA C-2, and glutamine C-4 from [1-13C]glucose. Using the correction factors, the percent enrichment of corresponding carboxyl and methene groups was the same (B. Hassel, unpublished data, 1998). All integrations of peaks were done blindly by one of us (A.B.) after expanding the spectra, using standard NMR integration procedures.
14C Labeling of amino acids from [1-14C]pyruvate
To study whether pyruvate carboxylation occurs in neurons in vivo, mice (N = 6) were anesthetized with fentanyl, fluanisone, and midazolam as described by Hassel et al. (1994), the skin over the scalp was removed, and a hole was drilled in the coronal suture, 2 mm lateral to the midline. [1-14C]Pyruvate, 1 μCi (specific activity 13.5 μCi/μmol; New England Nuclear, Boston, MA, U.S.A.) in 1 μL 0.9% NaCl, pH 7.3, was injected into the striatum with a 10-μL Hamilton syringe, which was inserted 2 mm dorsoventrally from the cortical surface. At 4 minutes, the animals were decapitated, and the neostriata were dissected out and homogenized in 1 mL of perchloric acid, 3.5%, with α-aminoadipate, 50 μmol/L, as the internal standard. The labeling of amino acids was determined as described (Hassel et al., 1992).
Enzyme assays
Brain homogenates were separated by a modification of the method of Gray and Whittaker (1962) into cytosol and mitochondria plus synaptosomes. The brains of four mice were homogenized separately to 10% homogenates in ice-cold isolation medium consisting of sucrose, 0.32 mol/L, sodium ethylenediamine tetraacetic acid, 1 mmol/L, dithiotreitol, 2 mmol/L, and Tris-HCl, 10 mmol/L, and they were subsequently centrifuged at 1000 × g for 10 minutes. The pellet was discarded, and 3 mL of the supernatants were centrifuged at 17,000 × g for 30 minutes. The supernatants and pellets obtained are termed S2 and P2, respectively. The S2 was recentrifuged to obtain a clear cytosolic fraction (100,000 × g for 1 hour). The P2, which contained free mitochondria and synaptosomes, was washed once by resuspension in 2.9 mL of isolation medium, centrifugation at 17,000 × g for 30 minutes, and resuspension in 2.9 mL of fresh isolation medium, after which Triton X-100 was added to 0.5%.
Malic enzyme was assayed by a modification of the method of Hsu and Lardy (1969), in both the carboxylating and the decarboxylating directions.
Decarboxylating. Into a 100-μL microcuvette (Starna, Hainault, Essex, UK), the following volumes were pipetted: 98 μL Tris-HCl, 40 mmol/L, pH 7.5; 5 μL MgCl2, 120 mmol/L; 2 μL sodium malate, 200 mmol/L; 2 μL KCl, 3 mol/L; and 2 μL NADP, 3 mmol/L. The reaction was started by adding 10 μL cytosol or 5 μL mitochondrial suspension, and the formation of NADPH was followed spectrophotometrically at 340 nm at 37°C.
Carboxylating. The following volumes were pipetted into a 100 μL microcuvette: 40 μL Tris-HCl, 40 mmol/L, pH 7.0; 40 μL NaHCO3, 200 mmol/L, pH 7.9; 20 μL sodium pyruvate, 200 mmol/L; 5 μL MgCl2, 120 mmol/L; 2 μL KCl, 3 mol/L; and 2 μL NADPH, 3 mmol/L. The reaction was started by adding 10 μL cytosol or mitochondrial suspension, and the disappearance of NADPH was followed spectrophotometrically at 340 nm at 37°C.
To measure lactate dehydrogenase activity in the direction of lactate, 100 μL of Tris-HCl, 40 mmol/L (pH 7.4) containing sodium pyruvate, 2 mmol/L, NADH, 0.2 mmol/L, and KCl, 60 mmol/L, were mixed with 5 μL of cytosol diluted 1:100 with sucrose, 0.32mol/L, containing 0.05% bovine serum albumin, or with 4 μL mitochondrial suspension diluted 1:32. The reaction was followed by the disappearance of NADH at 340 nm at 37°C. To measure lactate dehydrogenase activity in the direction of pyruvate, 100 μL of Tris-HCl, 40 mmol/L (pH 7.4) containing sodium lactate, 20 mmol/L, NAD, 0.34 mmol/L, and KCl, 60 mmol/L, were mixed with 3 μL cytosol diluted 1:10, or with 16 μL of the mitochondrial suspension diluted 1:32.
2-Oxoglutarate dehydrogenase (α-ketoglutarate dehydrogenase) activity was analyzed exactly as described by Mastrogiacomo et al. (1993). The reaction was started by adding 5 μL mitochondrial suspension or 10 μL cytosol to 100 μL of the reaction mixture, and the formation of NADH was followed at 37°C.
Fumarase was assayed spectrophotometrically by measuring the formation of fumarate from malate at 240 nm at 37°C (Hill and Bradshaw, 1969).
Statistics
Values are given as mean ± SD. Since the brain weights were similar, the amount of 13C is given per brain rather than per milligram of protein. Differences between groups are analyzed by Sigma Stat using one-way analysis of variance with Dunnett's method for multiple comparison. Differences in 13C or 14C enrichment between amino acids in the same animal or in activity between isozymes were analyzed by the paired t-test.
RESULTS
Labeling of cerebral amino acids from [3-13C]lactate in fasted and fed mice
During the first minute after injection of [3-13C]lactate, the animals sat motionless in a corner of the cage, not interacting with their litter mates, but they could be stimulated to walk normally about the cage. Thereafter, behavior was inconspicuous. This period of inactivity was not seen in mice that received [1-13C]glucose. The animals did not appear to be thirsty after the injections because they did not drink; gait was steady, and respiration was normal.
Injection of [3-13C]lactate led to labeling of cerebral alanine and amino acids associated with the TCA cycle (Table 1, Fig. 1). At 5 and 15 minutes after injection of [3-13C]lactate into fasted mice, the amount of 13C in the brain derived from [3-13C]lactate was 1630 ± 264 nmol and 2268 ± 248 nmol, respectively. This corresponds to 1.1% and 1.5% of the injected pyruvate equivalents, respectively, and it corresponds to an average cerebral uptake of 807 nmol [3-13C]lactate during the first 5 minutes. At 5 minutes after injection of 0.25 mol/L [3-13C]lactate, 0.3 mL, the amount of 13C in the brain was approximately half of that seen with 0.5 mol/L [3-13C]lactate, 870 ± 12 nmol. At 5 minutes after injection of [3-13C]lactate into fed mice, the amount of 13C in the whole brain was 1044 ± 66 nmol, or 36% lower than in fasted animals (p = 0.003); at 15 minutes, the amount was even lower, 763 ± 71 nmol.
Percent 13C enrichment in cerebral amino acids from [3-13C]lactate in fed and fasted mice
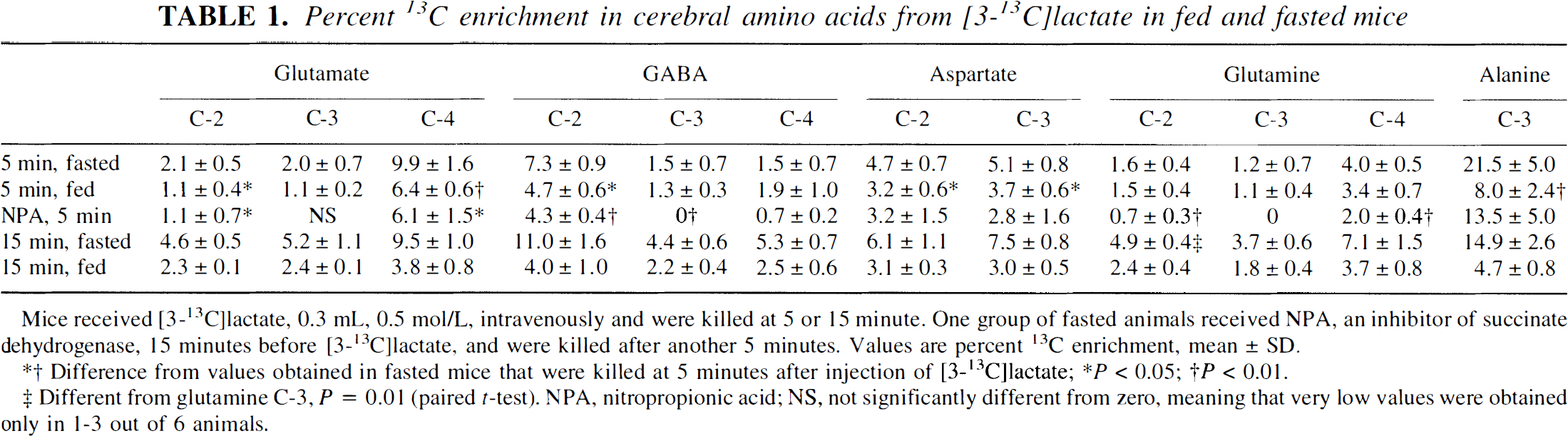
Mice received [3-13C]lactate, 0.3 mL, 0.5 mol/L, intravenously and were killed at 5 or 15 minute. One group of fasted animals received NPA, an inhibitor of succinate dehydrogenase, 15 minutes before [3-13C]lactate, and were killed after another 5 minutes. Values are percent 13C enrichment, mean ± SD.
Difference from values obtained in fasted mice that were killed at 5 minutes after injection of [3-13C]lactate; * P < 0.05; † P < 0.01.
Different from glutamine C-3, P = 0.01 (paired t-test). NPA, nitropropionic acid; NS, not significantly different from zero, meaning that very low values were obtained only in 1-3 out of 6 animals.
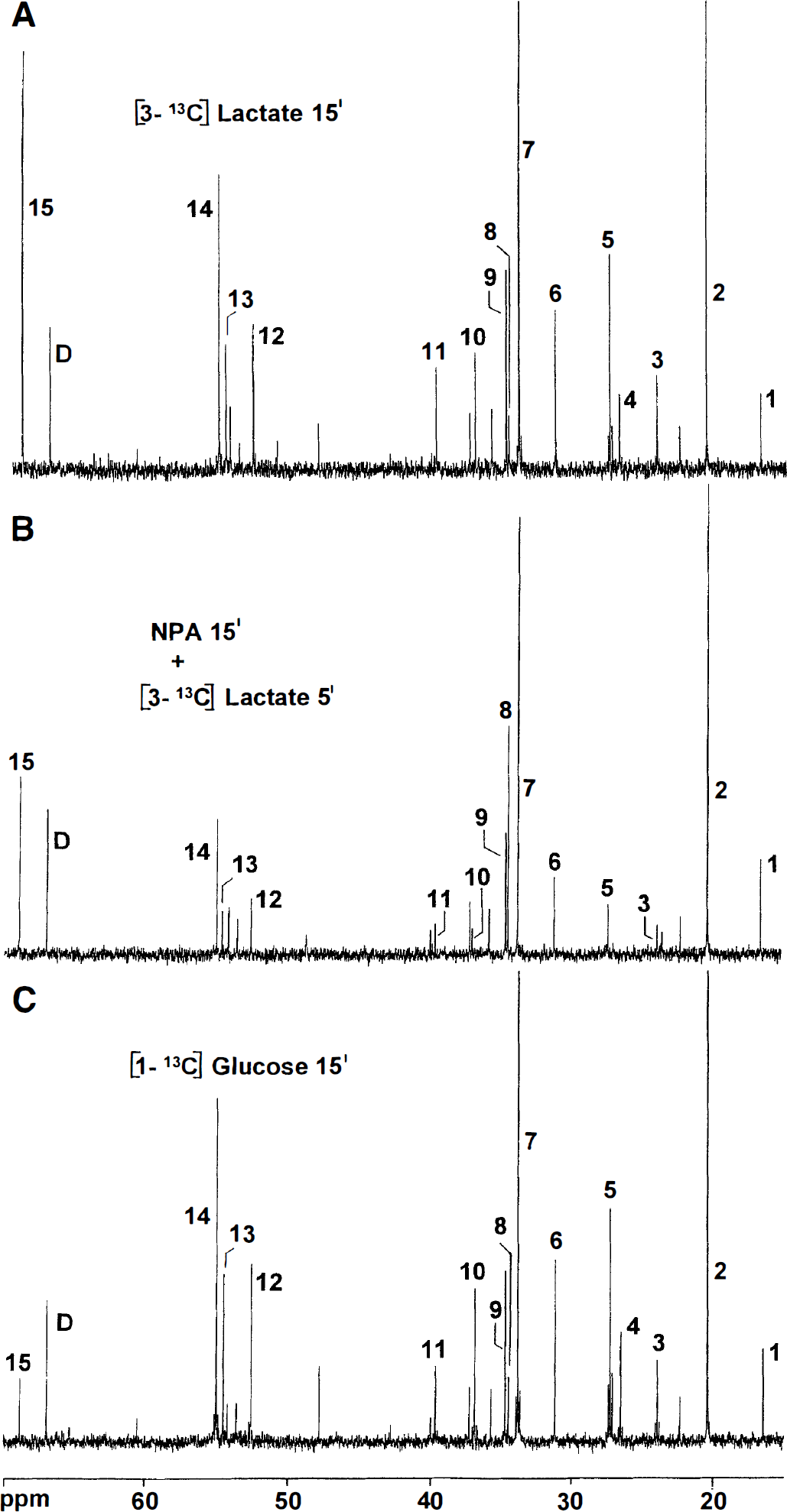
Typical 13C nuclear magnetic resonance spectra of brain extracts after injection of [3-13C]lactate or [1-13C]glucose.
At 5 minutes after injection of [3-13C] lactate, the percent enrichment of glutamate C-4 and GABA C-2 was higher than that of glutamine C-4 both in fed and fasted mice (Table 1), giving a glutamine-glutamate enrichment ratio of 0.4 ± 0.06 and 0.6 ± 0.14 in fasted and fed animals, respectively. At 15 minutes after injection of [3-13C]lactate, however, the enrichment of glutamine approached that of glutamate, giving a glutamine-glutamate enrichment ratio of 0.82 ± 0.04 and 0.98 ± 0.03 in fasted and fed animals, respectively. Only at 15 minutes after injection of [3-13C]lactate into fasted animals was glutamine C-2 better labeled than the C-3 position (Table 1), a sign that [3-13C]lactate was a poor substrate for glial pyruvate carboxylation (see Discussion). Cerebral [3-13C]lactate tended to equilibrate with alanine both in fasted and fed animals (Tables 1 and 2).
Percent 13C enrichment in cerebral lactate and glucose
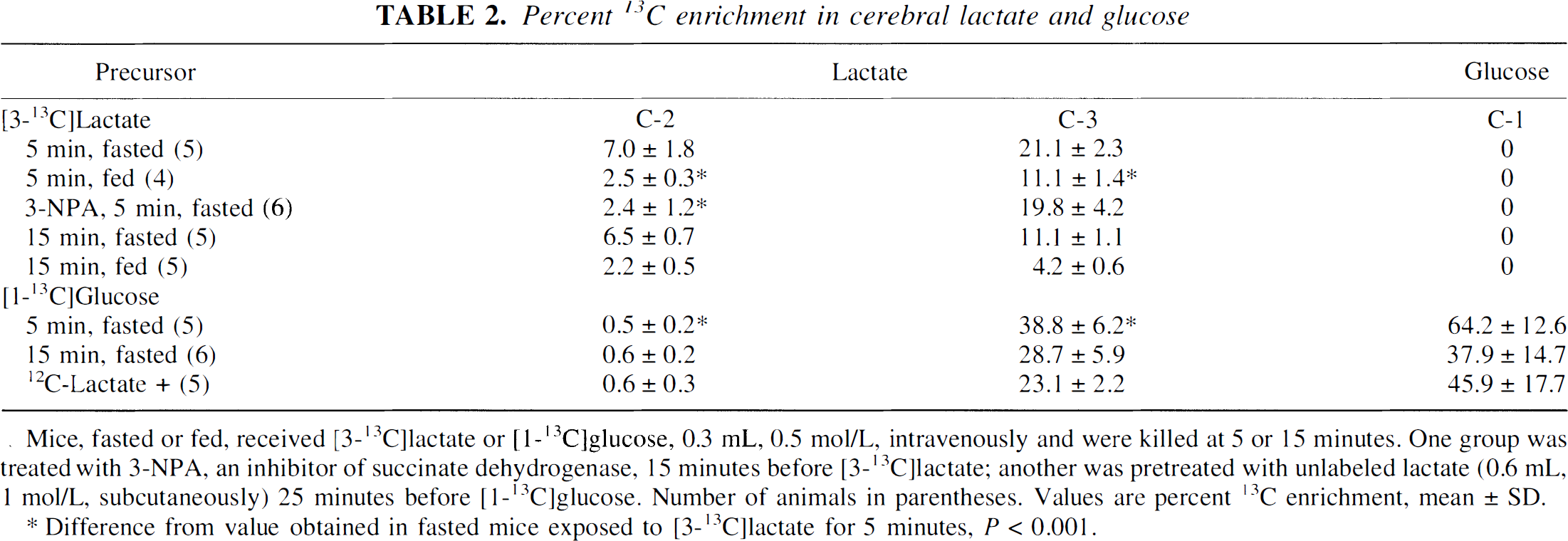
Mice, fasted or fed, received [3-13C]lactate or [1-13C]glucose, 0.3 mL, 0.5 mol/L, intravenously and were killed at 5 or 15 minutes. One group was treated with 3-NPA, an inhibitor of succinate dehydrogenase, 15 minutes before [3-13C]lactate; another was pretreated with unlabeled lactate (0.6 mL, 1 mol/L, subcutaneously) 25 minutes before [1-13C]glucose. Number of animals in parentheses. Values are percent 13C enrichment, mean ± SD.
Difference from value obtained in fasted mice exposed to [3-13C]lactate for 5 minutes, P < 0.001.
Both in fasted and fed animals, there was a striking labeling of lactate C-2 from [3-13C]lactate (Table 2), indicating randomization of label through equilibration with a symmetric intermediate of the TCA cycle (i.e., fumarate or succinate). At 5 minutes after injection of [3-13C]lactate, the total amount of [2-13C]lactate was 153 ± 36 nmol; at 15 minutes, it was 205 ± 4 nmol, or 9% of the total amount of label in the brain extracts at both 5 and 15 minutes. In mice that received half of the amount of [3-13C]lactate (0.3 mL, 0.25 mol/L), the amount of [2-13C]lactate was 10.5 ± 1.0% of the total amount of label in the brain. In fed animals, the amount of [2-13C]lactate was only 4.4 ± 1.2% of the total amount of label in the brain both at 5 and 15 minutes.
Pretreatment with the inhibitor of succinate dehydrogenase, NPA, caused sedation and inactivity, as previously described (Hassel and Sonnewald, 1995b). There was a massive buildup of [13C]succinate labeled from [3-13C]lactate (Fig. 1). The C-3 positions of glutamate, GABA, and glutamine were not labeled (Table 1), a sign that 13C had not passed the succinate dehydrogenase step of the TCA cycle. Even so, aspartate still was labeled at 68% and 55% of control values for the C-2 and C-3 positions, respectively, and the level of [2-13C]lactate remained at 34% of the control value (Table 2).
The carboxyl C-5 of glutamate, which is labeled from [2-13C]pyruvate, was significantly enriched both at 5 and 15 minutes after injection of [3-13C]lactate into fasted mice (0.4 ± 0.2% and 1.6 ± 0.4% enrichment, respectively). Both GABA C-1 and glutamine C-5 were labeled only at 15 minutes (2.3 ± 0.4% and 1.1 ± 0.6%, respectively). The percent enrichments of glutamate C-5 and GABA C-1 both were significantly higher than that of glutamine C-5 (p = 0.047 and 0.004, respectively, paired t-test).
Metabolism of [1-13C]glucose and the effect of pretreatment with 12C-lactate
Injection of [1-13C]glucose led to a labeling pattern in cerebral metabolites similar to that obtained with [3-13C]lactate (Table 3; Fig. 1) but with some notable differences. At 5 and 15 minutes after injection of [1-13C]glucose into control animals, the amount of 13C in the brain derived from [1-13C]glucose was 2009 ± 256 nmol and 2153 ± 642 nmol, corresponding to 2.7% and 2.9% of the injected pyruvate equivalents, respectively. In mice pretreated with unlabeled lactate, the amount of 13C was the same, 2123 ± 274 nmol, and the enrichment of the various carbon positions was the same as in controls (Table 3).
Percent 13C enrichment in cerebral amino acids from [1-13C]glucose in mice
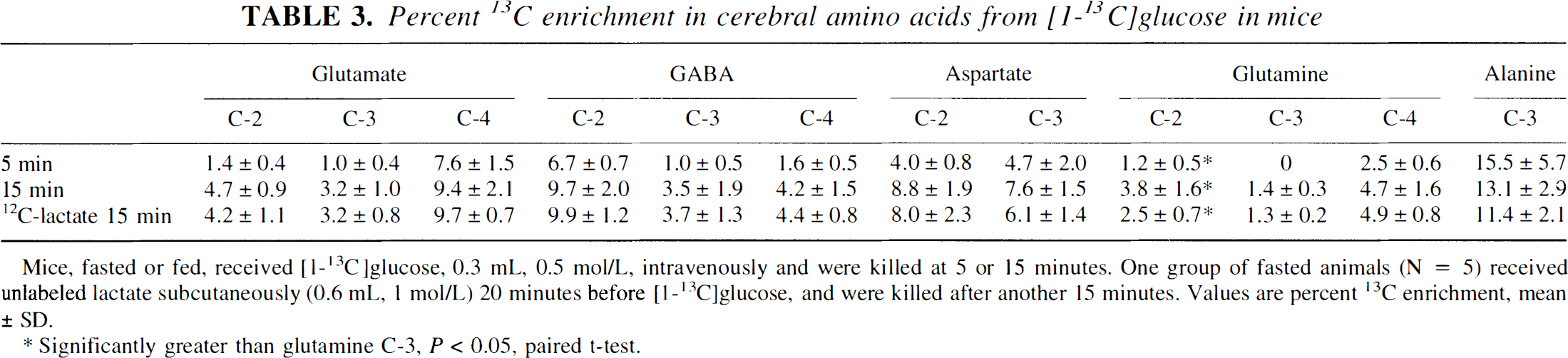
Mice, fasted or fed, received [1−13C]glucose, 0.3 mL, 0.5 mol/L, intravenously and were killed at 5 or 15 minutes. One group of fasted animals (N = 5) received unlabeled lactate subcutaneously (0.6 mL, 1 mol/L) 20 minutes before [1−13C]glucose, and were killed after another 15 minutes. Values are percent 13C enrichment, mean ± SD.
Significantly greater than glutamine C-3, P < 0.05, paired t-test.
Lactate was strongly labeled in the C-3 position from [1-13C]glucose but hardly at all in the C-2 position (Table 2). This was so, although the total amount of [3-13C]lactate in the brains of animals that received [1-13C]glucose was higher than in animals that received [3-13C]lactate (733 ± 160 nmol versus 432 ± 68 nmol at 5 minutes, p = 0.013). [3-13C]Lactate formed from [1-13C]glucose did not equilibrate with alanine in the way that the injected [3-13C]lactate did, suggesting that exogenous lactate does not completely equilibrate with endogenous lactate. There was no detectable 13C enrichment of glutamate C-5, GABA C-1, or glutamine C-5 in the [1-13C]glucose-treated animals.
Pool sizes of cerebral metabolites
Pool sizes were similar at 5 and 15 minutes for animals that received the same treatment. Therefore, only values obtained at 5 minutes are shown, as well as values obtained in animals pretreated with unlabeled lactate (Table 4). The levels of GABA, aspartate, lactate, and taurine were the same in all groups except for the NPA-treated animals. The levels of glutamate and glutamine, on the other hand, were lower in fasted than in fed animals and animals which were pretreated with unlabeled lactate. Treatment with NPA caused the level of GABA to increase by 38%, which is in agreement with the action of the toxin on GABAergic neurons (Hassel and Sonnewald, 1995b). The level of alanine increased with NPA treatment, and lactate tended to increase (by 44%), but this was not statistically significant.
Pool sizes of amino acid content (nmol/mg protein) in fed and fasted mice treated with [3-13C]lactate or [1-13C]glucose
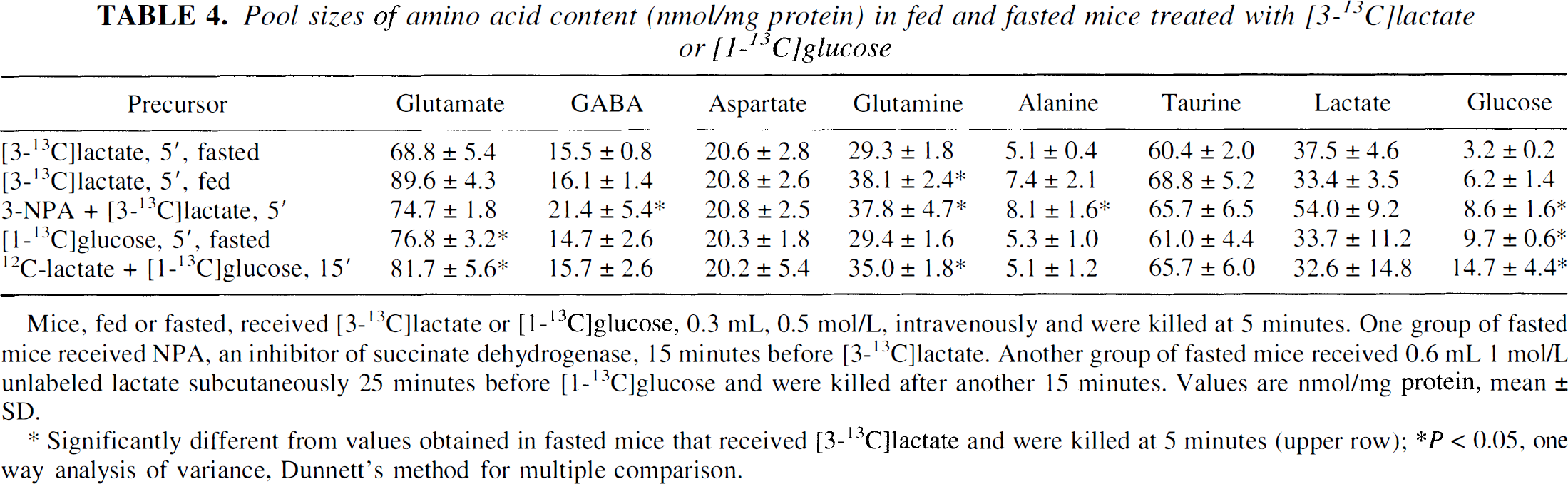
Mice, fed or fasted, received [3-13C]lactate or [1-13C]glucose, 0.3 mL, 0.5 mol/L, intravenously and were killed at 5 minutes. One group of fasted mice received NPA, an inhibitor of succinate dehydrogenase, 15 minutes before [3-13C]lactate. Another group of fasted mice received 0.6 mL 1 mol/L unlabeled lactate subcutaneously 25 minutes before [1-13C]glucose and were killed after another 15 minutes. Values are nmol/mg protein, mean ± SD.
Significantly different from values obtained in fasted mice that received [3-13C]lactate and were killed at 5 minutes (upper row); *P < 0.05, one way analysis of variance, Dunnett's method for multiple comparison.
Enzyme activities
Malic enzyme activity was measured because the data suggested an important role for the mitochondrial isozyme in the metabolism of [3-13C]lactate to [2-13C]lactate (see Discussion). In mitochondria, the maximal carboxylating activity of malic enzyme corresponded to 990 nmol/5 minutes in the whole brain (Table 5), far in excess of the 150 nmol of [2-13C]lactate formed in the first 5 minutes after injection of [3-13C]lactate. The maximal activity of 2-oxoglutarate dehydrogenase, a rate-limiting enzyme in the TCA cycle, was similar to the previously reported TCA cycle rate (Fitzpatrick et al., 1990). The activity of fumarase was 12 times higher than that of 2-oxoglutarate dehydrogenase (Table 5). The activity of lactate dehydrogenase was high in both fractions; in the cytosol it was only slightly more active in the direction of lactate than in the direction of pyruvate (p = 0.03), whereas in the mitochondrial/synaptosomal fraction it was 4.8 times more active in the direction of lactate (Table 5).
Enzyme activities (nmol/mg tissue × min−1) in mitochondrial and cytosolic fractions from mouse brain

Crude mitochondrial (including synaptosomes) and cytosolic fractions were analyzed for the activities of malic enzyme, fumarase, 2-oxoglutarate dehydrogenase and lactate dehydrogenase. Values are nmol/mg tissue × min−1, mean ± SD.
Difference from corresponding value for mitochondria, P < 0.001.
Difference from carboxylating activity, P < 0.001.
Concentrations and 13C enrichments of serum lactate and glucose
Serum lactate decreased by more than 50% from 5 to 15 minutes after injection of [3-13C]lactate into fasted and fed mice (Table 6). The percent enrichment of serum lactate C-3 was significantly higher in fasted than in fed animals at both 5 and 15 minutes after injection (p < 5 × 10−4; Table 7), indicating a more rapid clearance of [3-13C]lactate from the circulation in fed animals. In animals that received unlabeled lactate before [1-13C]glucose, serum lactate exceeded that seen in animals that received [3-13C]lactate only. Serum glucose was significantly higher in fed than in fasted animals both at 5 and 15 minutes after injection of [3-13C]lactate (Table 6).
Serum lactate and glucose (mmol/L) after intravenous injection of lactate and/or glucose

Fasted or fed mice received 0.3 mL [3-13C]lactate, 0.5 mol/L, intravenously and were killed at 5 and 15 minutes. One group of fasted mice received 3-NPA, an inhibitor of succinate dehydrogenase, 15 minutes before [3-13C]lactate. Another group of fasted mice received 0.6 mL 1 mol/L unlabeled lactate subcutaneously 25 minutes before [1-13C]glucose. Values are mmol/L, mean ± SD. N, Number of animals.
Percent enrichment of serum lactate and glucose, and the concentration (mmol/L) of [3-13C]pyruvate equivalents from lactate and glucose in fed and fasted mice administered [3-13C]lactate or [1-13C]glucose intravenously
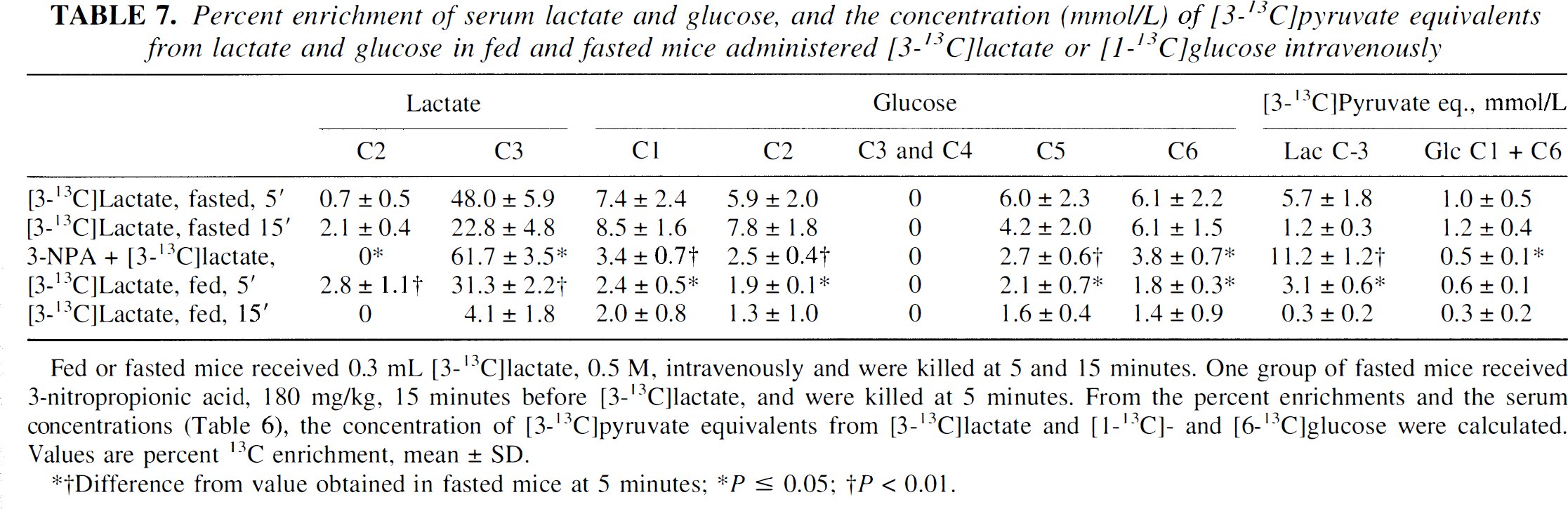
Fed or fasted mice received 0.3 mL [3-13C]lactate, 0.5 M, intravenously and were killed at 5 and 15 minutes. One group of fasted mice received 3-nitropropionic acid, 180 mg/kg, 15 minutes before [3-13C]lactate, and were killed at 5 minutes. From the percent enrichments and the serum concentrations (Table 6), the concentration of [3-13C]pyruvate equivalents from [3-13C]lactate and [1-13C]- and [6-13C]glucose were calculated. Values are percent 13C enrichment, mean ± SD.
Difference from value obtained in fasted mice at 5 minutes
P ≤ 0.05;
P < 0.01.
To assess gluconeogenesis from the injected [3-13C]lactate (Fig. 2), we calculated the serum concentration of [3-13C]pyruvate equivalents from [3-13C]lactate versus [1-13C]glucose and [6-13C]glucose (Table 7). At 5 minutes after injection of [3-13C]lactate, serum [3-13C]lactate corresponded to a serum concentration of [3-13C]pyruvate five times that obtained from the glucose isotopomers in both fed and fasted animals. At 15 minutes after injection of [3-13C]lactate, the concentration of [3-13C]pyruvate equivalents from [3-13C]lactate and 13C-labeled glucose was low but approximately the same. In animals pretreated with NPA, 13C-labeled glucose accounted for less than 4% of the total [3-13C]pyruvate equivalents in serum. Gluconeogenesis was even less pronounced in fed than in fasted animals, as can be seen from the 13C enrichment of serum glucose (Table 7).
Typical 13C nuclear magnetic resonance spectra of serum extracts. (
At 5 and 15 minutes after injection of [1-13C]glucose, the enrichment of serum glucose C-1 was 61.5 ± 15% and 19.2 ± 3.8%, respectively. No other carbon positions in glucose were labeled. The enrichment of lactate C-3 was 9.3 ± 4.3 and 5.5 ± 3.2 at 5 and 15 minutes, respectively, whereas lactate C-2 remained unlabeled.
Labeling of amino acids after intrastriatal injection of [1-14C]pyruvate
Injection of [1-14C]pyruvate led to a higher specific activity in glutamate than in glutamine (Table 8), giving a glutamine/glutamate relative specific activity that was significantly less than 1 (p < 0.01), indicating predominantly neuronal metabolism of [1-14C]pyruvate. The radiolabeling of aspartate was approximately four times higher than that of glutamate (Table 8), which is in agreement with entry of label into the TCA cycle at the level of malate and oxaloacetate (i.e., via pyruvate carboxylation).
Specific activities of cerebral amino acids after intrastriatal injection of [1-14C] pyruvate

Anesthetized mice received 1 μCi [1-14C]pyruvate in 1 μL of 0.9% saline by intrastriatal injection and were sacrificed at 4 minutes. Values are dpm/nmol in the three left columns and specific activity of glutamine relative to that of glutamate in the column to the right, mean ± SD.
DISCUSSION
The current study demonstrates that exogenous lactate is avidly metabolized by the brain in vivo. The labeling efficacy of [3-13C]lactate with respect to cerebral metabolites was approximately 50% of that of [1-13C]glucose after an intravenous bolus injection: 1.5% of the injected [3-13C]lactate and 2.9% of the injected [1-13C]glucose was found in cerebral metabolites of fasted animals at 15 minutes. Although significant cerebral metabolism of lactate has been shown previously in hypoglycemia (Thurston et al., 1983; Maran et al., 1994), the extent of lactate metabolism in our normoglycemic and hyperglycemic animals was surprising since normally there is a net release of lactate from the brain (Gibbs et al., 1942; Cremer et al. 1979). In fact, the average uptake of [3-13C]lactate during the first 5 minutes after injection was almost seven times higher than a previously calculated Vmax for uptake of lactate across the blood-brain barrier (Pardridge and Oldendorf, 1977). This agrees, however, with the finding by Cremer et al. (1979) that large amounts of lactate may enter the brain by nonsaturable diffusion.
[3-13C]Lactate predominantly entered the neuronal rather than the glial compartment, as can be seen from the higher percent enrichment of glutamate and GABA than of glutamine at 5 minutes after injection (Van den Berg et al., 1969; Hassel et al., 1992), which agrees with a previous study (O'Neal and Koeppe, 1966). [3-13C]Lactate was less of a glial substrate than [1-13C]glucose, since glial pyruvate carboxylation was much less evident with [3-13C]lactate, as shown by the similar labeling of glutamine C-2 and C-3 obtained with [3-13C]lactate. This makes isotopically labeled lactate a more specific marker for neuronal metabolism than labeled glucose.
An unexpected finding was the large formation of [2-13C]lactate from [3-13C]lactate. Such a conversion requires incorporation of label into a symmetric intermediate of the TCA cycle (i.e., fumarate or succinate) so that the label is randomized between the two middle carbon positions of these metabolites. Subsequent formation of malate with decarboxylation would yield both [2-13C]pyruvate and [3-13C]pyruvate and hence [3-13C]lactate (Fig. 3). In the current study, decarboxylation of malate formed through the ordinary operation of the neuronal TCA cycles could not account for the large formation of [2-13C]lactate, since [1-13C]glucose was a poor precursor of [2-13C]lactate. Nor could the massive formation of [2-13C]lactate have occurred through the ordinary operation of the glial TCA cycle, since administration of 13C-labeled acetate, an astrocyte-specific substrate, never leads to such dramatic labeling of lactate C-2 (Chapa et al. 1995; Hassel and Sonnewald, 1995a; Hassel et al., 1997). In animals pretreated with NPA, the label did not pass the succinate dehydrogenase step of the TCA cycle, and even so, formation of [2-13C]lactate still was active. Therefore, [3-13C]pyruvate must have been carboxylated to malate or oxaloacetate, with randomization of label in fumarate and subsequent decarboxylation of [2-13C]malate and [3-13C]malate to [2-13C]pyruvate and [3-13C]pyruvate (Fig. 3). This conclusion was supported by the finding that [1-14C]pyruvate labeled glutamate and glutamine, since such labeling requires both carboxylation of pyruvate and equilibration with fumarate (see earlier).
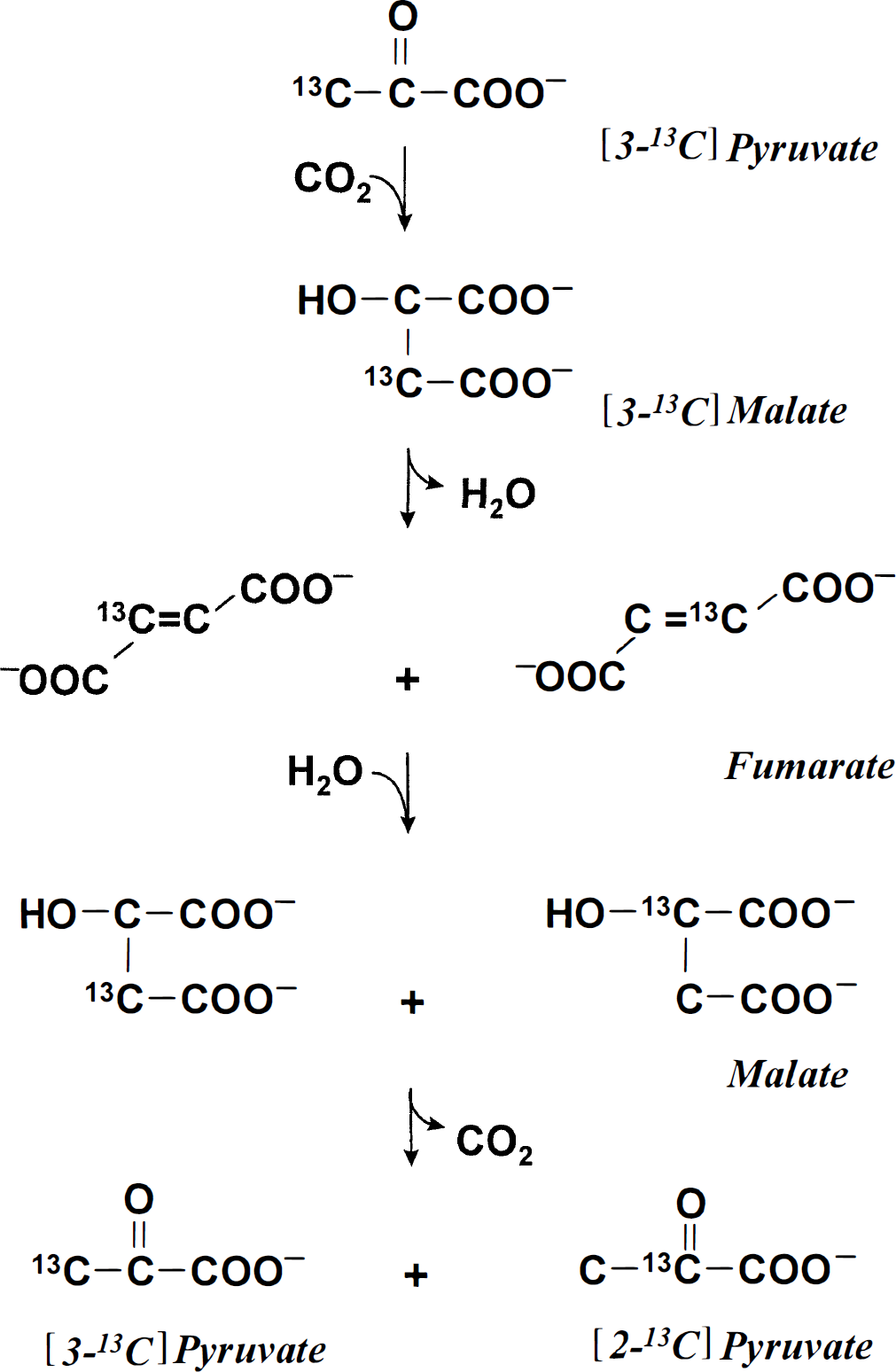
Carboxylation of [3-13C]pyruvate to [3-13C]malate and equilibration with fumarate with scrambling of label in the C-2 and C-3 positions of fumarate and, hence, in malate, which is decarboxylated to [2-13C]pyruvate and [3-13C]pyruvate.
The conversion of [3-13C]lactate to [2-13C]lactate probably took place mainly in neurons. In the current study, neurons were shown to be capable of pyruvate carboxylation, as could be seen from glutamate achieving a higher specific activity than glutamine when [1-14C]pyruvate was injected intracerebrally (O'Neal and Koeppe, 1966; Van den Berg et al., 1969; Hassel et al., 1992). This result also agrees with a recent study, which demonstrated avid pyruvate carboxylation in cultured neurons (Hassel and Bråthe, 2000). Mitochondrial malic enzyme, an enzyme with high pyruvate carboxylating activity (Salganicoff and Koeppe, 1968), has been found in neurons (Vogel et al. 1998). Regarding equilibration of malate with fumarate, Merle et al. (1996) found that malate equilibrates fully with fumarate in cultured neurons but not in astrocytes. In the current study, the stronger labeling of glutamine C-2 than of the C-3 with [1-13C]glucose as precursor, a sign of glial pyruvate carboxylation (Shank et al., 1993), indicates that equilibration with fumarate was not prominent in astrocytes. This suggests that cytosolic malic enzyme and pyruvate carboxylase, two pyruvate-carboxylating, astrocyte-specific enzymes (Kurz et al., 1993; Cesar and Hamprecht, 1995), were not quantitatively important for the formation of [2-13C]lactate from [3-13C]lactate. Taken together, these observations strongly suggest pyruvate carboxylation in neurons after injection of lactate.
The pyruvate carboxylating reaction was active: 9% of the label in the brain extracts was found in [2-13C]lactate in fasted animals. Subtraction of the 13C in the cerebral blood volume (66 nmol at 5 minutes) from the total amount of label in the brain extracts gives 9.8%. Since the same amount of [3-13C]lactate must be assumed to be formed through this reaction, this figure must be multiplied by two: 19.6%.
Formation of [2-13C]lactate from [3-13C]lactate depended on the nutritional state of the animal, since fed mice formed only half the amount of [2-13C]lactate, expressed as a percentage of the total amount of label in the brain. The formation of [2-13C]lactate from [3-13C]lactate could not be attributed to contamination with serum [2-13C]lactate, since it was observed in animals that had virtually no serum [2-13C]lactate. Further, the serum glucose that was labeled in the C-2 and C-5 positions (which may give rise to [2-13C]lactate) accounted for little of the [2-13C]lactate found in the brain extracts: at 5 minutes after injection of [3-13C]lactate into fasted mice, serum glucose corresponded to less than 1 mmol/L of [2-13C]pyruvate equivalents (Table 7) or less than 8 nmol of [2-13C]lactate. (The cerebral blood volume, of which approximately 50% is serum, is less than 4% [Edvinsson et al., 1973] of the total mouse brain of about 400 mg [see Methods], which gives approximately 8 μL cerebral serum.) This corresponds to only about 5% of the total cerebral amount of [2-13C]lactate, which was 153 nmol.
Formation of [2-13C]lactate from [3-13C]lactate through pyruvate carboxylation indicates exchange via malic enzyme, which is a reversible enzyme (Hsu and Lardy, 1969). However, the labeling of aspartate during inhibition of succinate dehydrogenase by NPA indicates that this reaction may also be truly anaplerotic; when [1-13C]glucose is administered to NPA-intoxicated animals, aspartate is not labeled at all (Hassel and Sonnewald, 1995b). Cerebral CO2 fixation previously has been attributed to astrocytes (Kaufman and Driscoll, 1992), where it subserves astrocytic formation of glutamine, a precursor for transmitter amino acids (Cotman and Hamberger, 1978; Paulsen et al., 1988; Hassel et al., 1997). The finding of pyruvate craboxylation in neurons indicates that neurons are metabolically more versatile and the glial-neuronal interrelatioship more flexible than has been assumed. Neuronal pyruvate carboxylation and malate decarboxylation may be of importance in regulating the level of TCA cycle intermediates in neurons, and pyruvate carboxylation could possibly subserve formation of transmitter amino acids.
Footnotes
Abbreviations used
Acknowledgements
The authors thank Dr. F. Rise for making the NMR facility available.
