Abstract
The hypotheses that cerebral embolic events lead to repetitive episodes of cortical spreading depression (CSD) and that these propagating waves trigger the expression of c-fos, brain-derived neurotrophic factor (BDNF), glial fibrillary acidic protein (GFAP), and heat shock protein 70 (HSP70) mRNA were tested. Wistar rats underwent photochemically induced right common carotid artery thrombosis (CCAT) (n = 18) or sham (n = 8) procedures. In a subgroup of rats (n = 5), laser-Doppler flowmetry probes were placed overlying the right parietal cortex to record CSD-like changes in cortical blood flow during the initial 2-hour postinjury period. Rats were killed by decapitation at 2 or 24 hours after CCAT, and brains were processed for in situ localization of the gene expression. Two to five intermittent transient hyperemic episodes lasting 1 to 2 minutes were recorded ipsilaterally after CCAT. At 2 hours after CCAT, the widespread expression of c-fos and BDNF mRNAs was observed throughout the ipsilateral cerebral cortex. Pretreatment with the N-methyl-
Brain ischemia facilitates the expression of multiple genes and proteins that are hypothesized to participate in the pathogenesis of ischemic neuronal damage. After both global and focal ischemia, the induction of stress genes including heat shock protein 70 (HSP70) is commonly reported in areas of neuronal damage, including the CA1 hippocampus and middle cerebral artery territories (Sharp et al., 1991; Simon et al., 1991; Welsh et al., 1992; Kinouchi et al., 1993). The induction of immediate early genes including c-fos and c-jun has also been documented after cerebral ischemia (Jorgensen et al., 1992; Welsh et al., 1992; Lindsberg et al., 1996). These intermediate early genes can be induced in minutes and serve as transcription factors to regulate the expression of target genes. The induction of mRNAs for growth factors including nerve growth factor and brain-derived neurotrophic factor (BDNF) has also been documented after cerebral ischemia (Lindvall et al., 1992; Kokaia et al., 1993). The increased expression of neurotrophins may reflect a protective response to ischemic injury.
Periods of anoxic depolarization and repetitive episodes of cortical spreading depression (CSD) are recorded in models of focal ischemia (Strong et al., 1985; Back et al., 1994; Dietrich et al., 1994). Transient depolarizations are known to occur spontaneously in the periinfarct border zone (penumbra) of acute focal cerebral ischemia (Iijima et al., 1992; Back et al., 1994). In models of middle cerebral artery occlusion, repetitive episodes of CSD have been proposed to increase penumbral vulnerability (Back et al., 1996). Thus, inhibition of these episodes of CSD by treatment with glutamate antagonists or mild hypothermia leads to ischemic protection (Iijima et al., 1992; Gill et al., 1992; Chen et al., 1993).
Episodes of CSD have also been reported in brain regions remote from a region of focal ischemia. In a model of thrombotic cortical infarction, 10 to 15 episodes of CSD were recorded over hours using potassium-sensitive electrodes to monitor extracellular ionic changes in cortical regions distant from the injury site (Dietrich et al., 1994). In that study, the transient increase in extracellular potassium was associated with a transient hyperemic episode (THE) as recorded by a laser—Doppler probe placed over the pial surface. Transient increases in cerebral blood flow also occur when CSD is induced in normal brains (Hansen et al., 1980; Lauritzen, 1987; Kariko et al., 1998) and are believed to be reactive rather than a cause of CSD (Lauritzen, 1987).
Cortical spreading depression has been reported to induce the increased expression of various genes and proteins (Comelli et al., 1993; Herrera and Robertson, 1990; Herdegen et al., 1993; Kokaia et al., 1993). In intact animals, episodes of CSD induced by either electrical stimulation or topical application of potassium were shown to lead to the upregulation of glial fibrillary acidic protein (GFAP) mRNA and protein (Kraig et al., 1991). Kariko and colleagues (1998) reported that CSD caused rapid alterations in expression of c-fos mRNA and BDNF mRNA. Thus, in addition to potentially affecting the vulnerability of the ischemic penumbra, injury-induced episodes of CSD may induce the expression of genes and proteins that could affect the response of remote brain regions to secondary insults. In this regard, CSD has been reported to induce tolerance to subsequent periods of cerebral ischemia (Kobayashi et al., 1995; Matsushima et al., 1996). Although the mechanisms underlying the development of ischemic tolerance are unknown, the synthesis of stress proteins and neurotrophic factors has been suggested to play a role (Sharp et al., 1991; Kawagoe et al., 1992; Kokaia et al., 1993; Kariko et al., 1998).
To investigate the pathobiology of thromboembolic stroke, photochemical models have been developed (Watson et al., 1995). Nonocclusive common carotid artery thrombosis (CCAT) is a rat model of thromboembolic stroke that causes rapid damage to the carotid artery vascular endothelium and transient deposition of platelet emboli in the cerebral microvasculature (Dietrich et al., 1993a,b). In this model, the embolic process produces transient hemodynamic and behavioral abnormalities similar to those reported in patients with transient ischemic attacks (Alexis et al., 1995; Stagliano et al., 1998). Preliminary data indicated that embolic processes subsequent to CCAT led to the widespread expression of genes that may result from episodes of CSD. Thus, the purpose of this study was to determine whether evidence for CSD could be obtained in this experimental model of thromboembolic stroke and to assess the consequences of embolic processes on the expression of stress and neurotrophic genes. To this end, genes that have been reported to be expressed in the intact brain following the induction of CSD (c-fos, BDNF, GFAP) and/or genes associated with tissue injury (HSP70) were selected for investigation.
MATERIALS AND METHODS
Animal groups
Experiments were performed on 31 male Wistar rats weighing between 250 and 300 g, obtained from Charles River Breeders (Charles River, MA, U.S.A.). Rats were assigned to six groups. In group 1, rats (n = 5) underwent right CCAT and the determination of cortical blood flow with laser-Doppler flowmetry to obtain evidence for episodes of CSD. In group 2, rats (n = 4) underwent sham procedures and were killed at 2 hours. Group 3 rats (n = 4) underwent sham procedures and were killed at 24 hours. Group 4 rats (n = 7) underwent CCAT and were killed at 2 hours. Group 5 rats (n = 7) underwent CCAT and were killed 24 hours later. Group 6 rats (n = 4) were pretreated with the N-methyl-
Surgery
Surgical procedures for CCAT using the photochemical technique have previously been described (Dietrich et al., 1993a). In brief, animals were anesthetized with halothane, intubated, and artificially ventilated using a rodent respirator. A tunable argon/dye laser (wavelength 562 nm; continuous-wave power 325 mW) was focused to an intensity of ~25 W/cm2 onto the saline-immersed right CCA with a 61-cm focal length spherical lens. The photosensitizing dye rose bengal (15 mg/ml in 0.9% saline) was injected at a dose of 20 mg/kg and the CCA irradiated for 10 minutes. This photochemical procedure has been shown to produce 50 to 75% stenosis of the CCA. Body temperature was monitored throughout the procedure and maintained between 36.8 and 37.2°C via a heating pad. Sham-operated animals underwent all surgical procedures, including the injection of rose bengal, but were not irradiated.
Laser-Doppler flowmetry
To assess the hemodynamic consequences of thromboembolic stroke, a laser-Doppler probe (BPM 403A; TSI, St. Paul, MN, U.S.A.) of 0.8 mm in diameter attached to a stereotaxic holder was positioned over the right hemisphere in the region of the middle cerebral artery territory (4 mm lateral to midline and 2.3 mm posterior to bregma). In three of five rats, a second laser-Doppler probe was also positioned over the contralateral cerebral cortex to determine whether evidence for bilateral CSD could be demonstrated. Previous studies from various laboratories have shown that laser-Doppler flowmetry represents a useful approach to assessing the hemodynamic consequences of CSD (Dietrich et al., 1994; Kariko et al., 1998). Starting 2 hours before irradiation, baseline flow values were obtained. Large blood vessels were avoided under microscopic guidance. After CCAT or sham procedures, hemodynamic data were continuously monitored for a 2-hour period.
In situ hybridization
At 2 and 24 hours after CCAT or sham procedures (groups 2 to 6), brains were removed under RNase-free conditions and frozen by suspension and gradual immersion in dry ice-cooled 2-methylbutane. Brains were then transferred to a cryostat at −20°C and sectioned at 12 μm under RNase-free conditions. Fresh-frozen cryosections were labeled and stored at −80°C. Sections were thawed to room temperature and fixed for 5 minutes with 4% formaldehyde in phosphate-buffered saline sections. Sections were acetylated for 10 minutes at room temperature in 0.25% acid anhydride and 0.1 mol/L triethanolamine-HCl (pH 8). Then they were dehydrated through a series of graded ethanols, delipidized in 100% chloroform for 5 minutes, rinsed in 100% ethanol, and allowed to dry.
Sections were hybridized to 35S-labeled riboprobes generated by in vitro transcription of the antisense (for positive probe) and sense (for negative probe) strands of cDNA clones subcloned into transcription vectors using the Promega Riboprobe System. The human cDNA clone for GFAP was purchased from American Type Culture Collection (Rockville, MD, U.S.A.). The mouse genomic clone pc-fos mouse 3, also from ATCC, was subcloned into pGEM7zf. Brain-derived neurotrophic factor was detected using a 460-bp EcoR1-Hind3 fragment of the rat cDNA subclone into pGEM4z (kindly supplied by Dr. B. Fennie, Genentech, Cambridge, MA, U.S.A.). The rat-inducible HSP70 cDNA clone was supplied by Dr. Frank Sharp.
The denatured probe (2 × 107 dpm/mL) was added to a solution containing 100 μg/mL salmon testes DNA, 250 μg/mL each of yeast total RNA and tRNA, 50% formamide, 20mmol/L Tris-HCl (7.4), 1 mmol/L ethylenediaminetetraacetate, 300 mmol/L NaCl, 10% dextran sulfate, and 1× Denhardt's solution. The hybridization solution was added to the sections, covered with coverslips, and hybridized under humid conditions at 55°C for 20 hours. After removal of the coverslips, the sections were washed at room temperature in a series of decreasing amounts of standard saline-sodium citrate with 1 mmol/L dithiothreitol to a final concentration of 0.1 × saline-sodium citrate. The slides were treated with 20 μg/ml RNase A for 30 minutes at 37°C. The final high-stringency wash was carried out in 0.1 × saline—sodium citrate and 1 mmol/L dithio-theitol for 1 hour at 65°C. Sections were dehydrated through a series of ethanols containing 300 mmol/L ammonium acetate, ending with 100% ethanol. Sections were exposed to Amersham Hyperfilm at 4°C for various amounts of time, developed in Kodak D-19 for 4 minutes, and fixed with Kodak Rapidfix. Following film exposure, slides were dipped in Kodak NTB2 emulsion diluted 1:1 with water and exposed for various times at 4°C. Negative control (sense strand) probes showed no hybridization signals (data not shown).
Autoradiographic image analysis
Coronal autoradiographic images from individual animals were digitized by means of a charge-coupled device base camera (8 to 12 bit; Xillix Technologies Corp., Vancouver, BC, Canada) equipped with a Micro Nikon 55-mm lens (Nikon, Tokyo, Japan) interfaced to an advanced image analysis system (MCID model M2; Imaging Research, St. Catherines, Ontario, Canada). [14C]Methylmethacrylate standards placed on each film were also digitized. Standards, when read and plotted, confirmed that the hybridized signals being measured were in the linear range of the film. Image files were subsequently transferred to a computer cluster consisting of a DEC 3600 minicomputer and 3200 VAXStation (Digital Equipment Corp., Maynard, MA, U.S.A.).
Region-of-interest and statistical analyses
Region-of-interest analysis was conducted by an investigator blinded to the experimental groups using a drawing tool to obtain radioisotope activity from cortical regions ipsilateral and contralateral to right CCAT. Cortical regions of interest were analyzed based on the rat stereotaxic atlas of Zilles (1985) and included sections at 2.7, −2.3, and −5.3 mm to bregma. At least three readings per animal were taken for each structure, and an average reading was then calculated. Activity data were expressed as mean ± SD. Statistical assessments were performed using one-way analysis of variance followed by Dunnett's test to compare cortical laser-Doppler flow perfusion units and the expression levels of each probe at various time periods with sham levels.
RESULTS
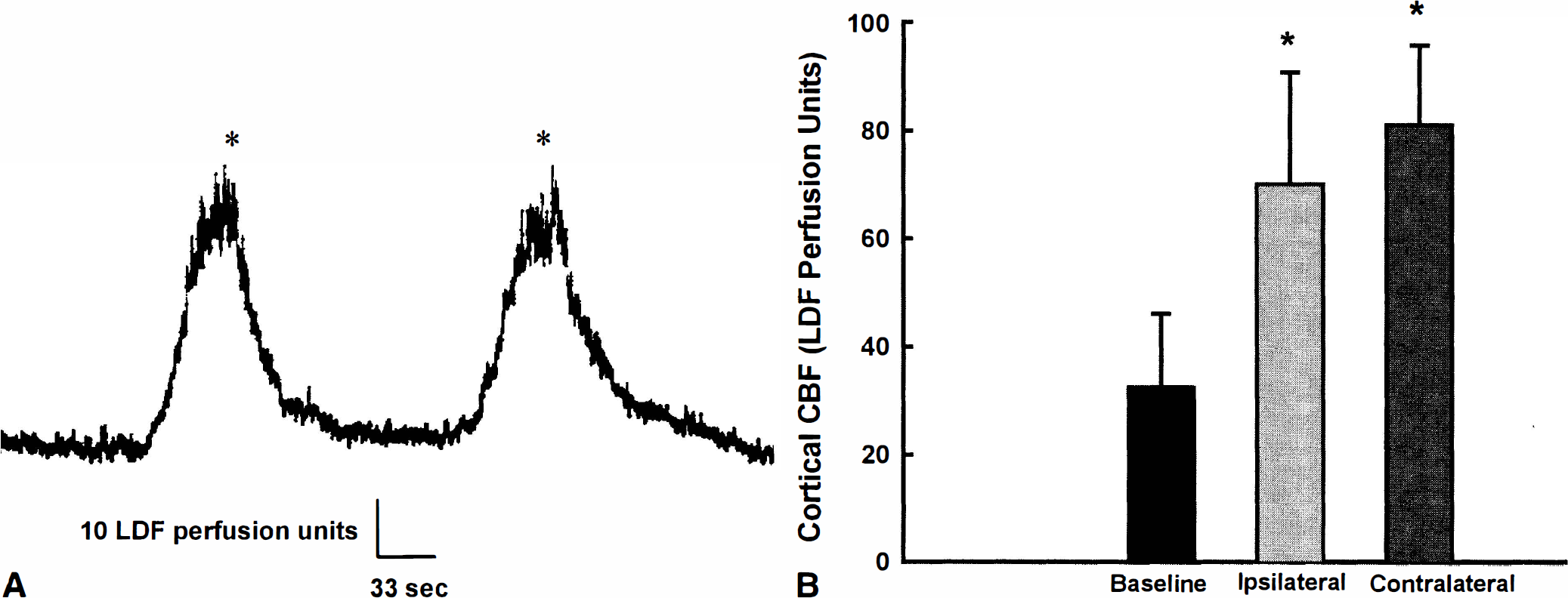
Physiologic and hemodynamic findings
The physiologic data obtained immediately before CCAT or sham procedures are summarized in Table 1. Physiologic variables were within normal ranges and did not vary among the experimental groups. Infusion of rose bengal dye in nonirradiated rats had little effect on arterial blood pressure or CBF measurements. In contrast, multiple THEs were observed in all rats after CCAT (group 1). Figure 1A shows two examples of these hemodynamic perturbations. The THEs were usually severe (~200% above baseline) and were ~2 minutes in duration. During the 2-hour study period, two to five THEs were observed in the right cerebral cortex of each thrombosed rat. Evidence for THEs was also obtained in the contralateral cortex in the three rats that underwent bilateral laser—Doppler probe placement. In these cases, one or two THEs were recorded during the 2-hour monitoring period and displayed similar degrees of hyperemia to those observed ipsilaterally (Fig. 1B). Thus, the frequency of THEs in the contralateral hemisphere appeared to be less than that observed ipsilateral to CCAT.
Physiologic variables
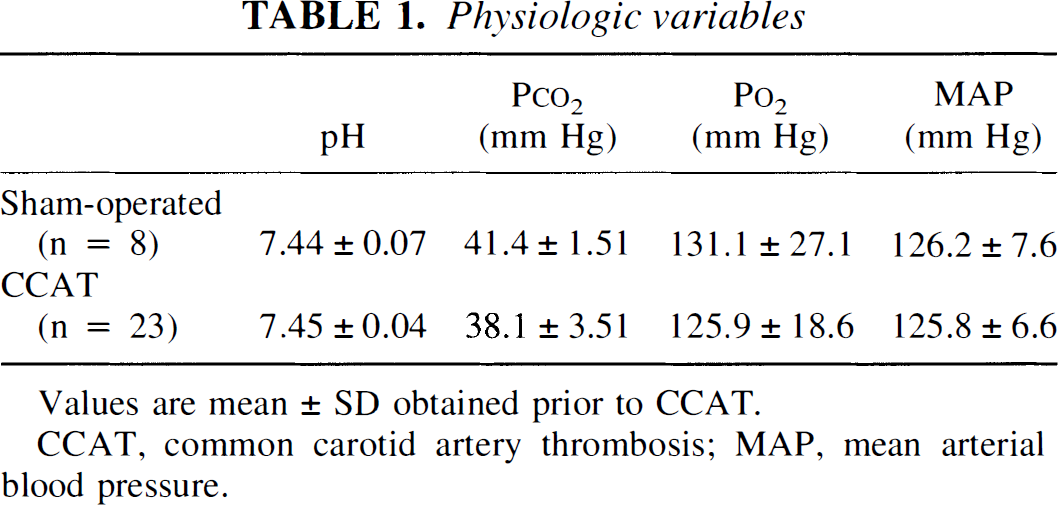
Values are mean ± SD obtained prior to CCAT.
CCAT, common carotid artery thrombosis; MAP, mean arterial blood pressure.
c-fos mRNA
The constitutive expression of c-fos mRNA in sham-operated control rats was present in the pyramidal and granular cell layers of the hippocampus and dentate gyrus. The expression was upregulated throughout the ipsilateral cerebral cortex at 2 hours after CCAT (Fig. 2). Areas of increased expression included the olfactory, frontal, parietal, insular, temporal, and occipital cortices. In contrast, increased expression did not extend into the granular retrosplenial cortex or presubiculum. In two of seven rats, increased c-fos expression was also present at 24 hours after CCAT. In the remaining rats, c-fos expression appeared as in sham-operated animals at 24 hours.
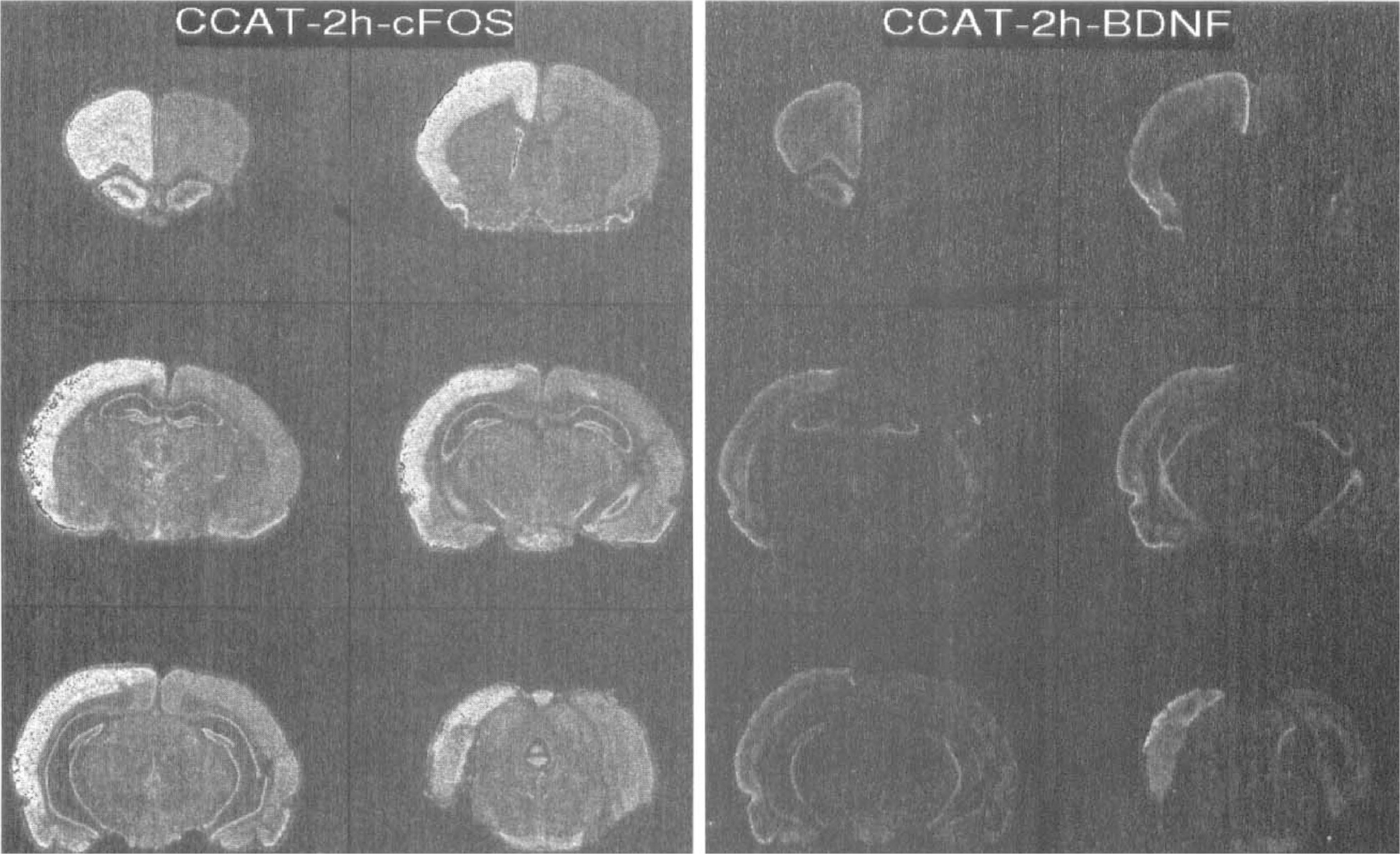
Representative in situ autoradiographs show c-fos and brain-derived neurotrophic factor (BDNF) mRNA expression at 2 hours after common carotid artery thrombosis. Note the increase of mRNA expression throughout the thrombosed cerebral cortex. The strongest expression of BDNF mRNA appears in the superficial cortical layer. In contrast, a lack of increased expression is seen in subcortical structures for both genes.
Brain-derived neurotrophic factor mRNA
The BDNF expression in sham-operated animals showed low levels of constitutive mRNA in the neocortex and entorhinal cortex and in the CA3 region and granular cells of the hippocampus. At 2 hours after CCAT, the level of expression was increased throughout the ipsilateral cerebral cortex (Fig. 2). Increased expression appeared to be most apparent within the superficial cortical layer. In one rat, mild expression of BDNF was also observed within the contralateral cerebral cortex. In contrast, increased BDNF mRNA expression was not observed at 24 hours after CCAT.
Glial fibrillary acidic protein mRNA
The GFAP expression in sham-operated and 2-hour CCAT rats was low, and no side-to-side patterns of increased expression were observed. However, at 24 hours after CCAT, both diffuse and focal increases in GFAP mRNA were observed (Fig. 3). Increased expression was seen focally in cortical and subcortical regions associated with sites of presumed structural damage. In these instances, a focal region of depressed mRNA expression was commonly present. Additionally, a more diffuse pattern of increased expression was seen throughout the cerebral cortex. This pattern was similar to that observed with c-fos mRNA expression at 2 hours. In three rats, GFAP expression was also seen within the contralateral hemisphere (cortex, striatum, hippocampus, and thalamus), appearing as focal regions of increased GFAP mRNA.
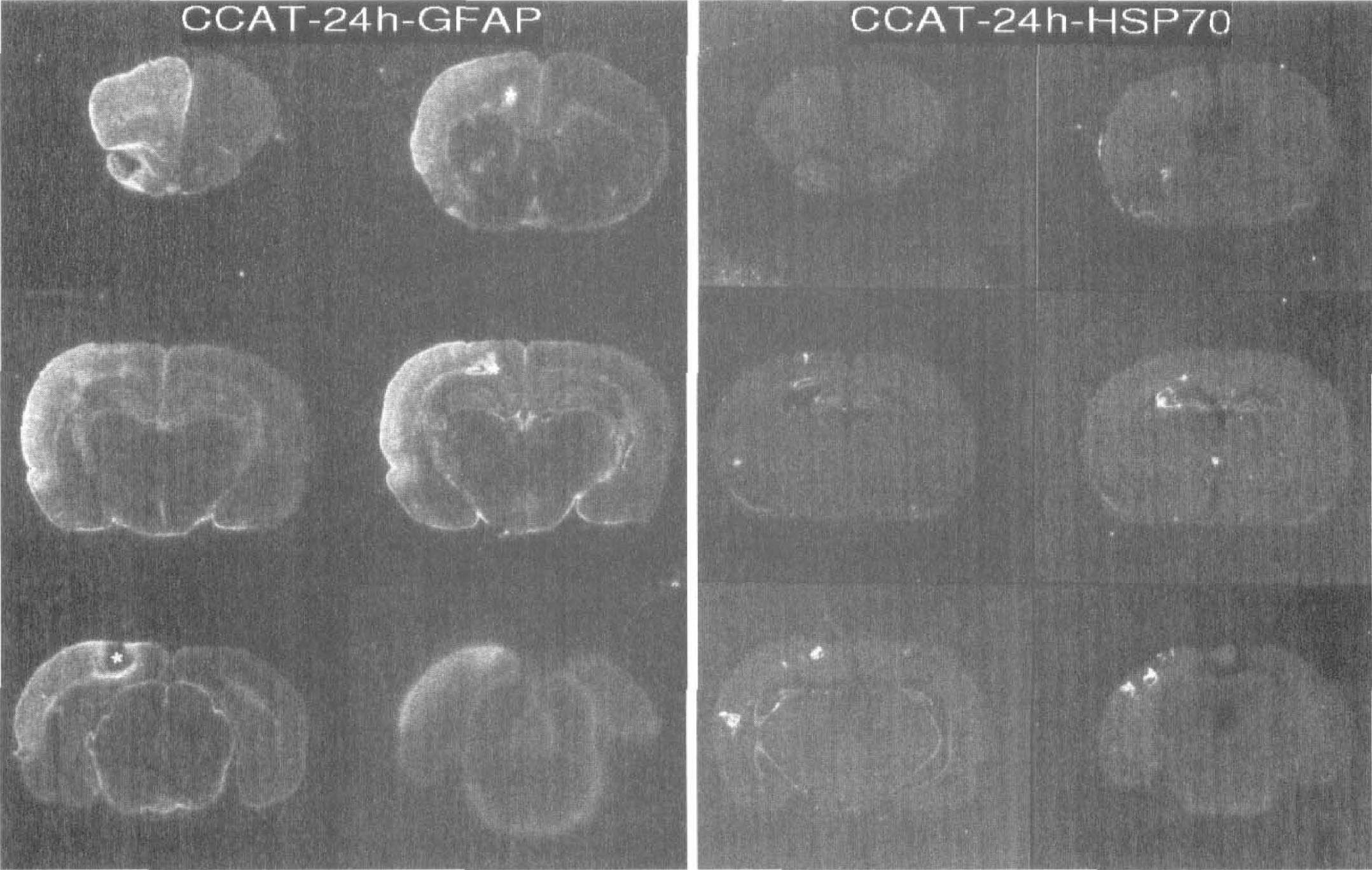
In situ autoradiographs show increased expression of glial fibrillary acidic protein (GFAP) and heat shock protein 70 (HSP70) mRNAs 24 hours after common carotid artery thrombosis. The GFAP mRNA is expressed throughout the thrombosed cerebral cortex. In addition, focal areas of enhanced and depressed (*) mRNA are seen. In contrast, HSP70 mRNA expression is multifocal and present in cortical and subcortical regions throughout the thrombosed hemisphere.
Heat shock protein 70
Abnormal expression of HSP70 was not observed in sham-operated control rats. The consequences of CCAT on HSP70 expression were seen at 2 and 24 hours after injury (Fig. 3). Multifocal regions of increased mRNA expression were commonly seen in ipsilateral cortical regions, striatum, hippocampus, and thalamus. In three rats, one to two areas of HSP70 expression were also present contralaterally but with less frequency as observed ipsilaterally. Serial frozen sections stained with hematoxylin and eosin commonly revealed evidence for local histopathological damage at sites of focal HSP70 expression (data not shown).
Quantitative findings
Figure 4 summarizes region-of-interest analysis of mRNA expression for c-fos, BDNF, and GFAP. Radioisotopic activity readings from the 2- and 24-hour shamoperated control rats were not significantly different (P > 0.05) and were therefore combined for subsequent analysis. Activity readings showed moderate increases in c-fos in several cortical regions at 2 hours after CCAT compared with control. These included the ipsilateral frontal cortex (bregma 2.7 mm), parietal cortex area 1, and parietal cortex area 2 (bregma −2.3 mm) and occipital cortex (bregma − 5.3 mm). A significant increase (P < 0.05) in c-fos activity was demonstrated within the occipital cortex. Moderate increases in BDNF mRNA expression were also observed in multiple cortical regions at 2 hours after CCAT. However, due to relatively large variations in activity within structures, no cortical region demonstrated activity readings that were significantly different from control. Pretreatment with MK-801 1 hour before CCAT depressed the expression of BDNF mRNA at 2 hours compared with that in nontreated CCAT rats (Fig. 4). By 24 hours after CCAT, activity readings for c-fos and BDNF mRNA were depressed compared with 2-hour readings.
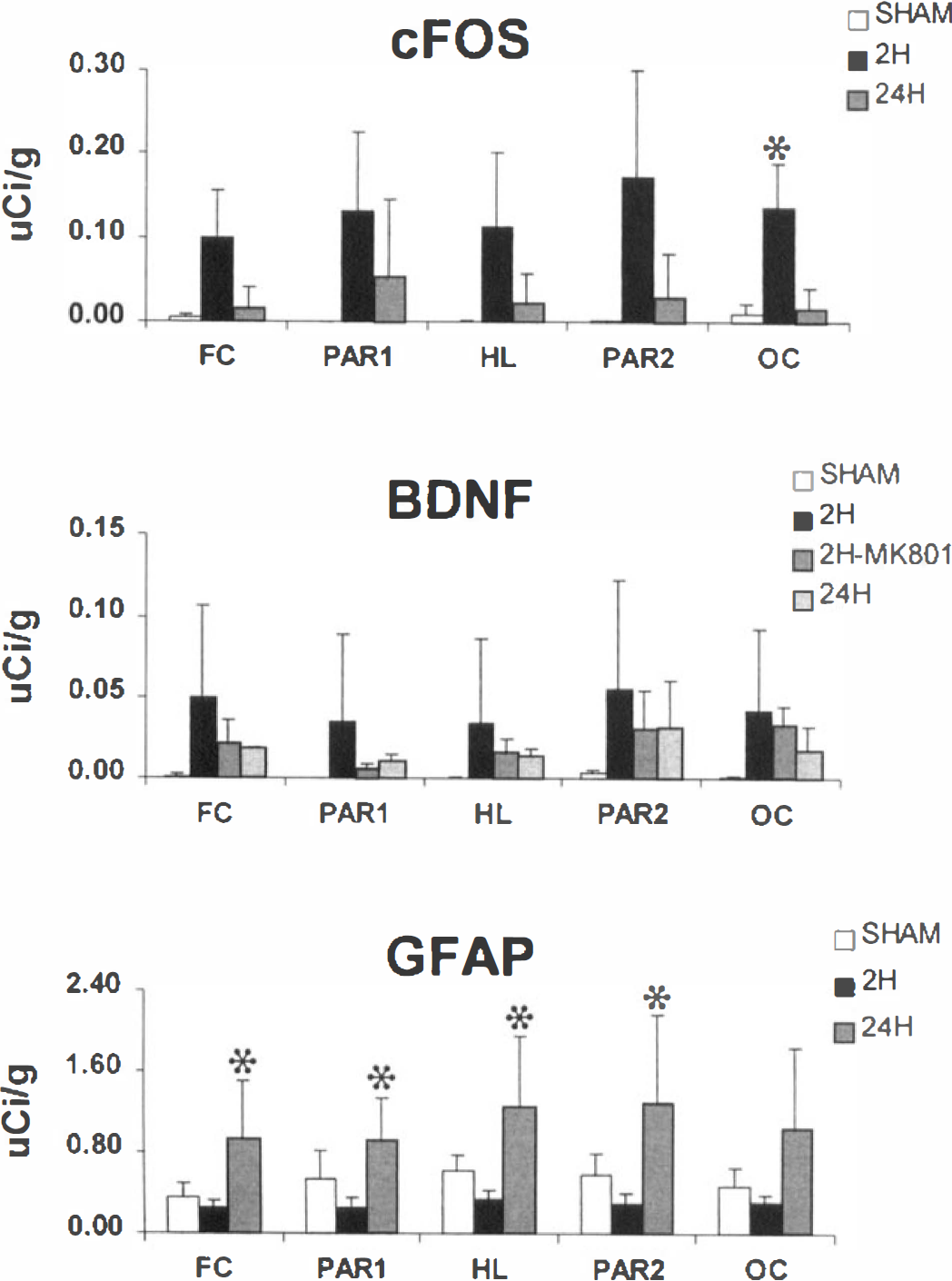
Quantitative assessment of altered c-fos, brain-derived neurotrophic factor, and glial fibrillary acidic protein mRNA activity within cortical brain regions ipsilateral to common carotid artery thrombosis. Film autoradiographs were measured for the increase in hybridization (μCi/g of 14C equivalents). Each mean value was obtained from readings taken from at least four rats. *Significantly different from control (P < 0.05).
Scattered areas of HSP70 mRNA expression were multifocal and therefore difficult to quantitate using the present regional approach. However, focal areas of increased expression were apparent at both 2 and 24 hours after CCAT. Quantitative assessment of GFAP mRNA expression demonstrated significant increases in activity at 24 hours within the frontal, parietal, and hindlimb cortical regions. Finally, significant differences in c-fos, BDNF, or GFAP mRNA expression were not documented in any contralateral regions compared with sham-operated animals (data not shown).
DISCUSSION
The major findings of the present study are that CCAT leading to platelet embolization to the brain produces repetitive THEs and a complex pattern of abnormal gene expression throughout the thrombosed hemisphere. During the first 2 hours after CCAT, two to five THEs were recorded within the thrombosed cerebral cortex by laser-Doppler flowmetry. Based on previous data, these THEs are interpreted as being stimulated by physiologic events resembling CSD (Lauritzen, 1987; Dietrich et al., 1994). These data support published studies showing that models of focal cerebral ischemia such as transient and permanent middle cerebral artery occlusion trigger episodes of CSD and abnormal gene expression. The present findings therefore extend published findings and demonstrate that embolic events leading to small infarcts can lead to similar consequences.
Cortical spreading depression is a transient, reversible phenomenon with characteristic ionic, metabolic, and hemodynamic changes (Hansen et al., 1980). Although CSD is known to be noninjurious in the intact brain, transient depolarizations that occur spontaneously in the periinfarct border zone (penumbra) of acute focal ischemia aggravate ischemic damage (Back et al., 1996; Chen et al., 1993; Iijima et al., 1992; Mies et al., 1994). In the present thromboembolic model, platelet emboli accumulate mainly within the thrombosed hemisphere immediately after CCAT (Dietrich et al., 1993a,b). A study using 111In-labeled platelets reported that the majority of platelet emboli are spontaneously removed by 24 hours after injury, with the remaining emboli leading to the generation of severe hemodynamic deficits and embolic infarcts (Dietrich et al., 1993b). Thus, in contrast to focal ischemia models that produce a single large focus of damaged tissue, the present model leads to transient embolic events and multiple small infarcts.
It is of interest that this thromboembolic model also leads to the generation of repetitive episodes of CSD that likely originate at sites of severe focal ischemia. Previous histopathological studies have demonstrated that this model produces multiple embolic infarcts. In this study, histopatholgical assessment of serial sections also demonstrated that some sites of focal HSP70 expression were associated with regions of ischemic damage. It therefore appears that embolic events within the ipsilateral and contralateral hemispheres may initiate CSD that may be associated with specific gene responses. The explanation for the differences in gene expression between the two hemispheres may involve the frequency of CSDs and/or other embolic consequences including hemodynamic and histopathological events that occur predominantly within the ipsilateral hemisphere. For example, although platelet emboli and focal lesions are occasionally observed contralaterally (Dietrich et al., 1993b), the incidence and severity of these events are significantly reduced compared with the ipsilateral hemisphere.
The importance of CSD in the pathophysiology of thromboembolic stroke remains to be clarified. Based on previous work, CSD could enhance ischemic damage within the border zones of embolic infarcts. However, unlike ischemic lesions produced by large-vessel occlusion that result in well-described penumbral zones (Jacewicz et al., 1992), microvascular occlusion leading to embolic infarcts results in well-demarcated ischemic foci with limited periinfarct border zones of moderate ischemia (Stagliano et al., 1998). Because embolic events can lead to selective neuronal necrosis in addition to infarction (Dietrich et al., 1993a), repetitive CSDs might enhance the vulnerability of selective neuronal populations associated with an ischemic focus.
Recent data indicate that CCAT results in severe but spontaneously reversible sensorimotor and cognitive behavioral deficits (Alexis et al., 1995). Although these behavioral changes may result from structural and/or hemodynamic abnormalities (Dietrich et al., 1993a; Stagliano et al., 1998), the role of CSD must also be considered. Experimentally induced SDs have been used to inhibit normal limb function in animals (Bures and Bracha, 1990). In a recent study, the behavioral consequences of induced SD in intact rats were assessed (Alexis et al., 1996). Intact rats in which SD was repetitively evoked showed severe behavioral deficits at 2 and 6 hours but no significant deficits at 24 hours. Repetitive episodes of CSD combined with other embolic events may therefore participate in the functional consequences of thromboembolic stroke.
The present data indicate that episodes of CSD precede widespread alterations in gene expression. These findings are supported by previous studies that reported similar patterns of c-fos, BDNF, and GFAP expression in intact rats in which CSD was induced (Herrera and Robertson, 1990; Kraig et al., 1991; Nowak et al., 1991; Herdegen et al., 1993; Kokaia et al., 1993; Kariko et al., 1998). The noncompetitive N-methyl-
Episodes of CSD and altered gene expression after CCAT may influence the susceptibility of the postthrombotic brain to subsequent insults. For example, CSD has been reported to induce tolerance to subsequent periods of cerebral ischemia (Kobayashi et al., 1995; Matsushima et al., 1996). Mechanisms underlying ischemic tolerance are unknown but have been suggested to include the synthesis of heat shock proteins and neurotrophic factors (Sharp et al., 1991; Kawagoe et al., 1992; Lindvall et al., 1992; Kokaia et al., 1993). Brain-derived neurotrophic factor mRNA expression is coupled to neuronal excitation (Ghosh et al., 1994) and is considered to be a neuroprotective mechanism in models of focal brain injury (Thanos et al., 1988; Comelli et al., 1993). Although the significance of GFAP mRNA expression is unclear, astrocytes are reported to be a major source of nerve growth factor (Goss et al., 1998). Embolic events or cerebral insults that produce microvascular damage may induce endothelial alterations including HSP72 expression (Lindsberg et al., 1996) that may also influence the cerebrovascular response to secondary injury. Because CCAT leads to CSD and the expression of both stress and neurotrophic genes, one might question whether CCAT would induce ischemic tolerance.
In this regard, recent data indicate that rats that undergo CCAT followed 24 hours later by a brief global ischemic insult have worse histopathological damage than postischemic rats without prior CCAT (Dietrich et al., 1999). Thus, thromboembolic events appear to be a risk factor for widespread cerebral infarction when combined 24 hours later with a delayed ischemic insult. Although transient embolic events induce molecular responses in neurons and glia that could potentially regulate the brain's ability to resist injury, other processes including microvascular or neuronal damage may override these protective responses, especially in the early postembolic stages. In this regard, Koh and colleagues (1995) have reported that prolonged exposure of cortical neurons to neurotrophins (BDNF, neurotrophin-3, and neurotrophin-4/5) potentiated the necrotic death induced by exposure to oxygen-glucose deprivation or N-methyl-
In summary, we report that transient embolic events following CCAT lead to multiple episodes of CSD and abnormal expression of glial and neuronal genes. As discussed, these hemodynamic, ionic, and molecular events may alter the response of noninjured brain regions to secondary injury and/or influence recovery mechanisms. A greater understanding of the importance of these consequences of thromboembolic stroke should help with the development of neuroprotective strategies to be applied to patients following transient embolic events to prevent the initiation of stroke and enhance functional recovery.
Footnotes
Acknowledgments
The superb technical assistance of Judith Y. Pita-Loor is gratefully acknowledged. The authors also thank Helen Valkowitz for typing the manuscript.
