Abstract
Ubiquitin, an essential protein in nonlysosomal proteolytic system, is expressed after metabolic stress to the cell, The authors investigated stress response of ubiquitin in the hippocampus of the Mongolian gerbil after forebrain ischemia, The level of hippocampal ubiquitin was compared with that under ischemic tolerance induced by ischemic preconditioning, The authors also studied ubiquitin gene expression using in situ hybridization method, Transient ischemia resulted in consumption of free ubiquitin and an increase of multiubiquitin chains, These changes were transient in the hippocampus outside of the CA1 region where neurons survived, whereas it was persistent in the CA1 region where neurons were destined to die after ischemia. Under tolerant condition, subsequent ischemia provoked rapid recovery and further increase of free ubiquitin. The signal of ubiquitin messenger ribonucleic acid was continuously detected after ischemia, not only under tolerant conditions, but without tolerance induced by preconditioning. Thus, ubiquitin stress response takes place, at least at a transcriptional level, in dying CA1 neurons. Under tolerant conditions, however, subsequent ischemia in the CA1 region induces the stress response of ubiquitin up to the translational level, leading to the rapid restoration of protein synthesis and to eventual neuronal survival.
Keywords
Ubiquitin, a small 76-residue protein, is an essential factor for the ATP-dependent nonlysosomal protein degradation. Once ubiquitinated, target proteins are rapidly degraded by 26S proteasome (Rechsteiner et al., 1993; Peters, 1994). This proteolytic pathway not only degrades wasted proteins but contributes to the regulation of short-lived regulatory proteins, such as proteins involved in the cell cycle, class I antigen processing, signal transduction cascades, receptor-mediated endocytosis, and apoptosis (King et al., 1996; Hochstrasser, 1996; Weissman, 1997). Ubiquitin also is known as a member of the stress proteins. Facing sublethal metabolic stress, stress proteins are massively expressed in most cells. This stress response is believed to be a cellular reaction essential to counter the adverse environment. Ubiquitin, as one of the stress proteins, undoubtedly is required under stressful conditions for cell survival.
Brief, transient ischemia selectively kills hippocampal CA1 neurons. Since CA1 neurons die far later after ischemic insult, the process is referred to as delayed neuronal death (Kirino, 1982; Pulsinelli et al., 1982). Its precise mechanism is yet to be clearly understood. Magnusson and Wieloch (1989) found a selective and sustained loss of ubiquitin immunoreactivity in the hippocampal CA1 neurons after brief forebrain ischemia in the rat. Their results indicate that CA1 neurons fail to express stress response after ischemia. By immunohistochemical methods, we demonstrated that the loss of ubiquitin immunoreactivity in CA1 neurons after ischemia resulted from depletion of free ubiquitin and that the stainability of conjugated multiubiquitin chains (MUC) was unchanged (Morimoto et al., 1996). These findings suggest that lethal ischemia brings about persistent depletion of free ubiquitin, leading to a dysfunction of ubiquitin proteolytic system in CA1 neurons. The dysfunction may provoke an accumulation of denatured proteins and could significantly alter the function of various regulatory proteins. We speculate that the disturbance of ubiquitin proteolytic pathway may, at least partially, affect the neuronal survival after ischemia.
Takada et al. (1995, 1996) have established methods of immunoassay of intracellular free ubiquitin and MUC with negligible cross-reaction. Their free ubiquitin radioimmunoassay system detects only a few MUC consisting of more than 20 ubiquitin molecules (Takada et al., 1996). The MUC enzyme-linked immunosorbent assay system detects MUC consisting of more than six ubiquitin molecules conjugating to the proteins destined for degradation (Takada et al., 1995). We considered that the values of free ubiquitin by radioimmunoassay and MUC by enzyme-linked immunosorbent assay described by Takada and associates demonstrate well the amount of tissue free ubiquitin and conjugated MUC.
Here we report quantitative changes, measured by this method, of free ubiquitin and MUC in the hippocampus after transient ischemia. We studied the ubiquitin content of the CA1 regions, where neurons are destined to die, and that of the rest of the hippocampus, the CA3 and dentate gyrus (CA3 + DG), where neurons survive. We also analyzed ubiquitin gene expression using an in situ hybridization method. Moreover, we compared the transcriptional and translational levels of ubiquitin after ischemia under tolerant conditions induced by preconditioning (nonlethal 2-minute forebrain ischemia). Our data indicate that the impairment and recovery of ubiquitin turnover correlate well with the neuronal death and survival in the hippocampus after ischemia.
METHODS
Induction of ischemia and sample preparation
Male Mongolian gerbils (Meriones unguiculatus, weighing 60 to 80 g) were housed at the ambient temperature of 23 to 27°C. Two animal subgroups were investigated. In the 5-minute ischemia group (n = 25), gerbils were anesthetized with 3% halothane in 33% O2 and 67% N2, and the bilateral common carotid arteries were exposed. Five-minute forebrain ischemia was induced by occlusion of these arteries using Sugita aneurysm clips. Halothane anesthesia was discontinued immediately after reperfusion. During the operation, the body (rectal) and temporal muscle temperatures were monitored and maintained at 37.5° ± 0.2°C using a heat lamp and warming blanket. In the double ischemia group (n = 25), gerbils were subjected to 5-minute forebrain ischemia at the 48-hour reperfusion period after a preconditioning ischemia. The preconditioning ischemic challenge for 2 minutes was induced by Tone's method (Tone et al., 1987; Kirino et al., 1991). Briefly, a 4-0 nylon suture was pulled tight for 2 minutes, which had been looped around each common carotid artery on the previous day of the ischemia. At 48 hours after 2-minute ischemia, 5-minute forebrain ischemia was induced by clipping as described earlier. Unoperated gerbils (n=5 each) were controls in the 5-minute ischemia group and the double ischemia group, and the results of gerbils killed at 48 hours after 2 minutes of ischemia (n = 5) in the double ischemia group served as the results at 0 hours after 5 minutes of ischemia.
The animals were given overdoses of pentobarbital and decapitated at reperfusion intervals of 3, 6, 12, 24, and 48 hours after 5 minutes of forebrain ischemia in the 5-minute ischemia group or the double ischemia group (n = 5 each). After quickly removing and freezing the brains, all samples were stored at −80°C.
Quantitative analysis of free ubiquitin and multiubiquitin chains
Sample preparation. Cryostat sections (55 µm thick) containing the dorsal hippocampi were quickly dried at room temperature, and the hippocampal CA1 region and the CA3 + DG region were separately cut out using Futaba sterile surgical blade under an operating microscope (Fig. 1). Each sample was homogenized in 100 µL of Tris-buffered saline (TBS, 50 mmol/L Tris-HCl pH 7.6, 150 mmol/L NaCl) containing 1 mmol/L ethylene glycol bis (2-aminoethyl-ether) tetraacetic acid and proteinase inhibitors (0.5 mmol/L diisopropylfluoro-phosphate, 0.5 mmol/L phenylmethylsulfonyl fluoride, 1 µg/mL Nα-p-tosyl- Dissected areas from the CA1 and the CA3 and dentate gyrus (CA3 + DG) regions for the quantification of free ubiquitin and multiubiquitin chains (MUC). Cryostat sections (55-µm thick) containing the dorsal hippocampus were quickly dried, and the CA1 region and CA3 + DG regions were separately cut out.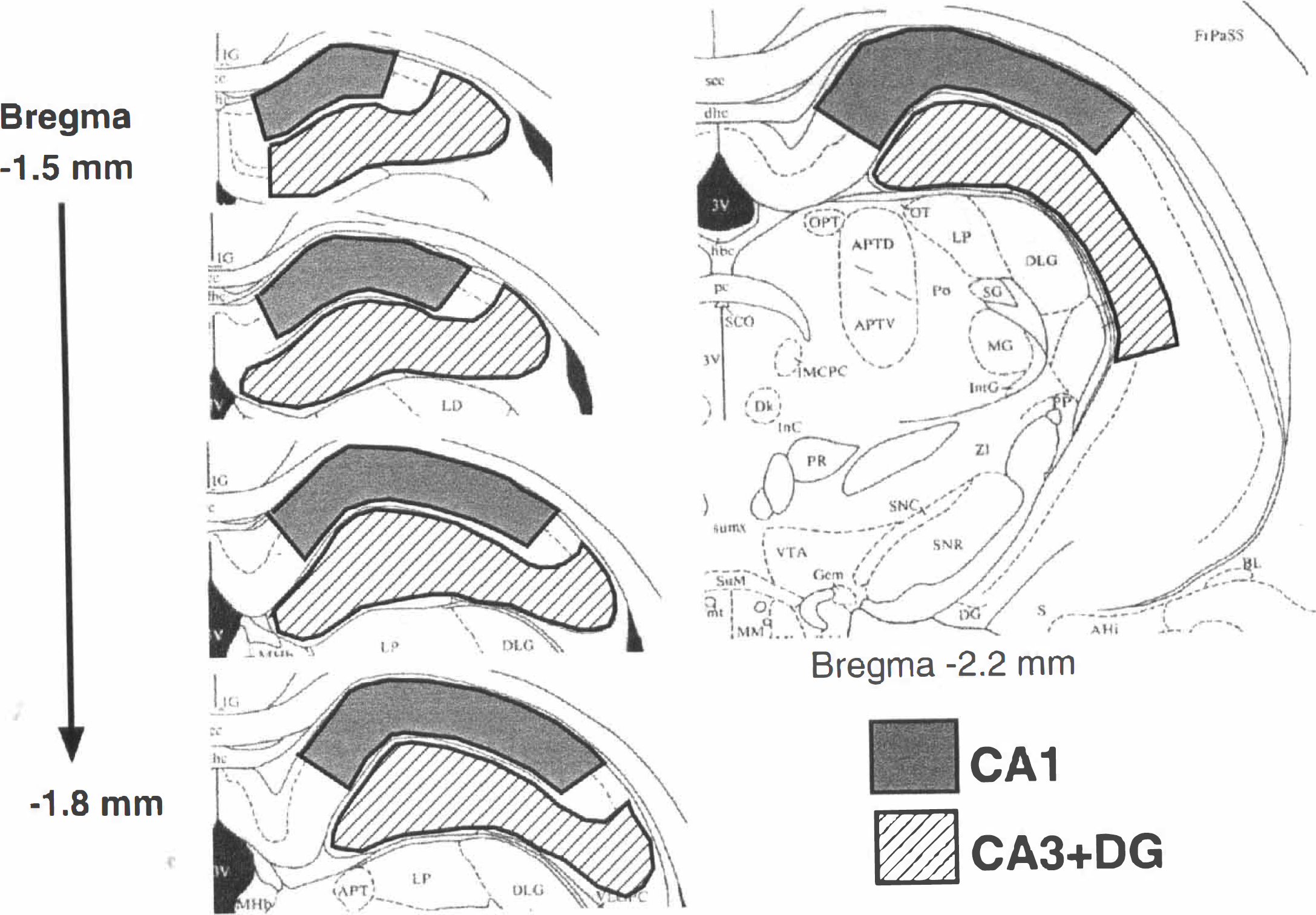
Free ubiquitin radioimmunoassay. We used a competitive radioimmunoassay system to measure the levels of free ubiquitin using the rabbit antisera US-1 against ubiquitin (detailed methods were described previously [Takada et al., 1996]). Briefly, 100 µL of each of the free ubiquitin (monoubiquitin) standards and the samples were transferred to each polystyrene tube (12 × 75 mm). Simultaneously, the antiserum (100 µL), free ubiquitin (100 µL) labeled with 125I by the chloramine T procedure (Hunter and Greenwood, 1962), and the assay buffer (300 µL) also were added to the tube. After incubation, 200 µL of goat antiserum to rabbit gamma-globulin and 3.5% polyethylene glycol 6000 was added, and the mixture was further incubated. Immune complex was obtained by centrifugation and counted in a gamma counter (Wallac Oy, Turku, Finland). A standard curve was plotted as counts per minute versus nanograms per milliliter of the free ubiquitin standard and fitted to a nonlinear regression curve. Concentrations of samples were determined by interpolation from the fitted curve.
Multiubiquitin chain enzyme-linked immunosorbent assay. To measure MUC, we performed a sandwich enzyme-linked immunosorbent assay (Takada et al., 1995) using the monoclonal antibody FK2 (Fujimuro et al., 1994), which recognizes conjugated ubiquitin, and MUC reference preparation 1 as the standard. Briefly, the samples (100 µL) and MUC standards (prepared according to Tamura and colleagues [1991] with some modifications) were transferred to each well of microtiter plates coated with the FK2 antibody and incubated. After washing each well, biotinylated FK2 antibody was added, followed by incubation with peroxidase-conjugated streptavidine (DAKO, Carpinteria, CA, U.S.A.). Color development was performed with 3, 3', 5, 5'-tetramethylbenzidine (0.15 mg/mL in 1 mmol/L sodium acetate, pH 5.5, containing 0.0025% H2O2), and the absorbance at 450 nm was determined. A standard curve then was plotted (absorbance at 450 nm versus nanograms per milliliter of the MUC standard), and concentrations of the samples were determined by interpolation from the fitted curve. Thus, the amount of MUC was expressed as that of standard MUC reference preparation 1.
Data analysis. We could not express quantitative data per total protein content in the dissected sample, since sample volume was so small that a sample loss was not negligible in homogenization of TBS-insoluble pellets by TBS containing 8 mol/L of urea. Therefore, we expressed free ubiquitin as micrograms per total milligrams of protein in TBS-soluble lysate, and MUC as nanograms per total milligrams of protein in urea-soluble lysate, respectively. Protein content of each sample (100 µL) was estimated by the modified Lowry method (Bensadoun and Weinstein, 1976). Thus, the levels of free ubiquitin were expressed as mean ± SD µµm/mg of protein of TBS-soluble fraction. Those of MUC were expressed as mean ± SD ng/mg of urea-soluble protein. Differences among groups in ubiquitin level were compared by two-factor factorial analysis of variance with Bonferroni/Dunn, and P < 0.05 was considered significant.
Probe preparation
The gerbil ubiquitin probe used in this study was the 186-bps cyclic ribonucleic acid (cRNA) probe. Partial ubiquitin cDNA was constructed by reverse transcription-polymerase chain reaction from total RNA of gerbil kidney using primers that were designed from conserved sequences in human and rat ubiquitin. The primers used were 5': GGGAATTCTGATCTTTGCAGGC AAGCAG and 3': GGGGATCCTGCCTTGACATTTTCAATGG (Einspanier et al., 1987; Hayashi et al., 1994). The polymerase chain reaction product was sequenced, and its homology was 89.7% with 4 repeats of the ubiquitin sequence of rat, 97.8% with 10 repeats of the ubiquitin sequence of rat, and 90.8% with 3 repeats of the human ubiquitin sequence (Einspanier et al., 1987; Hayashi et al., 1994). The cRNA probe of this fragment was labeled with α-35S-UTP using Riboprobe Kit (Promega, Madison, WI, U.S.A.). Sense probe findings were used as controls.
In situ hybridization
Five animals were evaluated at each time point for the in situ hybridization method. Fresh-frozen sections (12-µm thick) were fixed in 4% paraformaldehyde with phosphate-buffered saline for 10 minutes at room temperature. These sections were acetylated for 10 minutes. After dehydration through graded ethanol, sections were prehybridized in hybridization buffer for 2 hours and hybridized with the probe (1 × 106 cpm/50 µL) overnight at 55°C. Sections then were washed in 4 × SSC for 30 minutes, then in wash buffer (50% formamide, 10 mmol/L dithiothreitol, 5 mmol/L ethylenediamine tetraacetic acid, 10 mmol/L Tris pH 7.0, and 0.3 mmol/L NaCl) for 30 minutes at 55°C. After incubation with RNase A for 30 minutes at 37°C, the sections were washed in 2 × SSC for 20 minutes, then in 0.1 × SSC for 30 minutes. Autoradiograms were obtained by exposing the sections to BioMax MR film (Kodak, Rochester, NY, U.S.A.) for 36 hours.
RESULTS
Free ubiquitin
The TBS-soluble fraction was used to measure the free ubiquitin level. The MUC level in the TBS-soluble fraction also was studied. Since the MUC level in this fraction was low and was relatively stable after ischemia, only the result of free ubiquitin level is shown here. Free ubiquitin level in controls was slightly lower in the CA1 than the CA3 + DG region, but the difference was not statistically significant.
Five-minute ischemia group. Free ubiquitin levels in the CA1 and CA3 + DG regions were 7.2 ± 1.1 and 7.4 ± 1.4µg/mg of protein before ischemia and decreased to 58% (P < 0.001) and 65% (P < 0.001) of preischemic value, respectively, at 3 hours of reperfusion (Fig. 2A). The concentration of free ubiquitin in the CA1 region remained at a lower level until 48 hours of ischemia. However, that in the CA3 + DG region gradually recovered to the control level by the 48-hour reperfusion period. Thus, transient ischemia caused consumption of free ubiquitin in all regions of the hippocampus, and this reduction continued in the CA1 region until later death of pyramidal neurons.
Quantitative change of free ubiquitin in Tris-buffered saline-soluble fraction in the 5-minute ischemia group 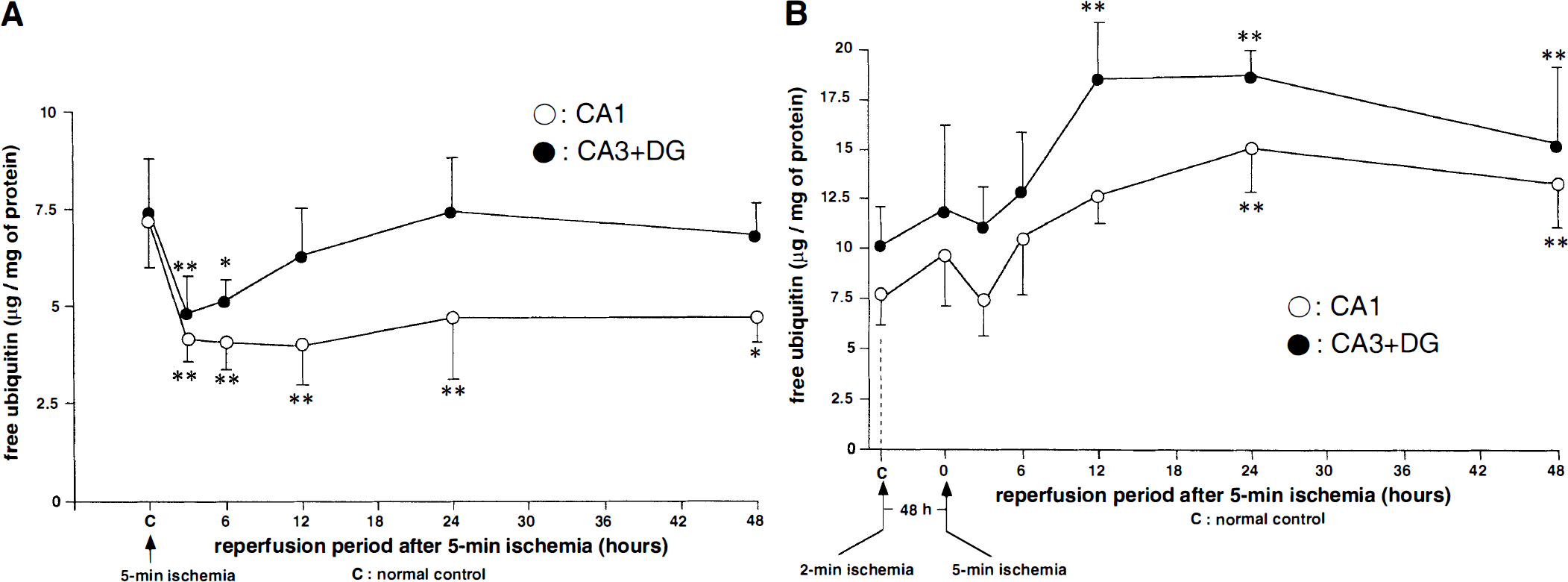
Double ischemia group. After sublethal ischemia, the baseline level of free ubiquitin did not change significantly (Fig. 2B). After the subsequent 5 minutes of ischemia, the free ubiquitin level only slightly decreased. However, it recovered promptly and further increased at 12 to 24 hours of reperfusion in both regions (P < 0.001). Thus, under tolerant conditions, ischemia-induced reduction of free ubiquitin is transient, and the level of free ubiquitin was promptly restored above the normal level, suggesting de novo ubiquitin synthesis.
Multiubiquitin chains
Multiubiquitin chain was measured with urea-soluble fraction. Since urea interferes with free ubiquitin radio immunoassay system, the level of free ubiquitin level in this fraction was not studied. The amount of free ubiquitin in urea-soluble fraction was considered to be small.
Five-minute ischemia group. Control levels of MUC in the urea-soluble fraction of the CA1 and CA3 + DG regions were 5.5 ± 3.9 and 8.6 ± 7.0 ng/mg of protein in terms of MUC reference preparation 1, respectively. The amount of MUC increased in the CA1 and CA3 + DG regions after ischemia (Fig. 3A). In the CA3 + DG region, values of MUC reached a peak at 12 hours and decreased to the control level at 24 hours. However, those of MUC in the CA1 region remained at an elevated level during the observation period for 48 hours after reperfusion.
Quantitative change of MUC in urea-soluble fraction in the 5-minute ischemia group 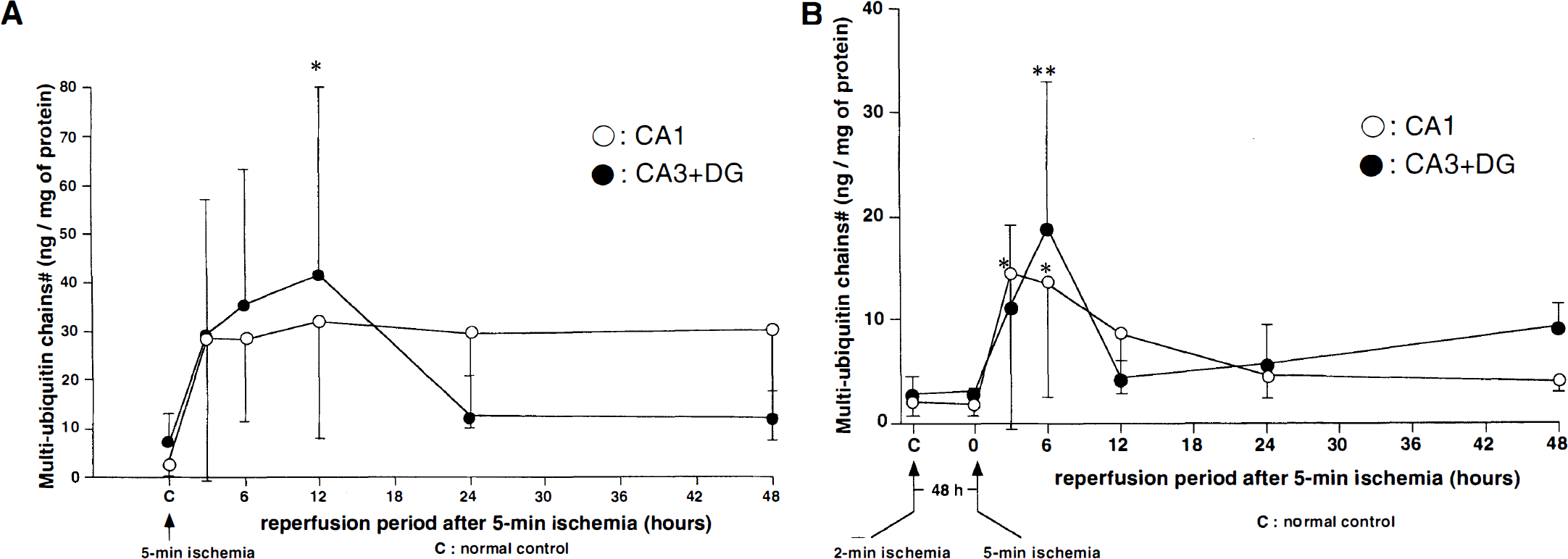
Double ischemia group. The amount of MUC in the urea-soluble fraction increased slightly at 3 to 6 hours after the second 5-minute ischemia and returned promptly to the control level at 24 to 48 hours in both regions (Fig. 3B). The peak value of increased MUC in the CA3 + DG region (12 hours after ischemia) in the 5-minute ischemia group was higher than that (6 hours after ischemia) in the double ischemia group (P < 0.05) (Fig. 3). The peak value of MUC in the CA1 region in the 5-minute ischemia group was higher than that in the CA1 region of the double ischemia group, but the difference was not statistically significant.
In situ hybridization
Five-minute ischemia group. Coronal sections at the level of the hippocampus in control animals clearly demonstrated constitutive expression of ubiquitin messenger ribonucleic acid (mRNA) (Fig. 4A). After 5 minutes of forebrain ischemia, the signals of ubiquitin mRNA in creased at 6 hours of reperfusion in all hippocampal neurons and in the cerebral cortex, which then decreased to the control level at 48 hours. Control sections hybridized with the sense probe did not show detectable signals.
Coronal sections at the level of the hippocampus showing ubiquitin messenger ribonucleic acid (mRNA) after 5 minutes of ischemia without 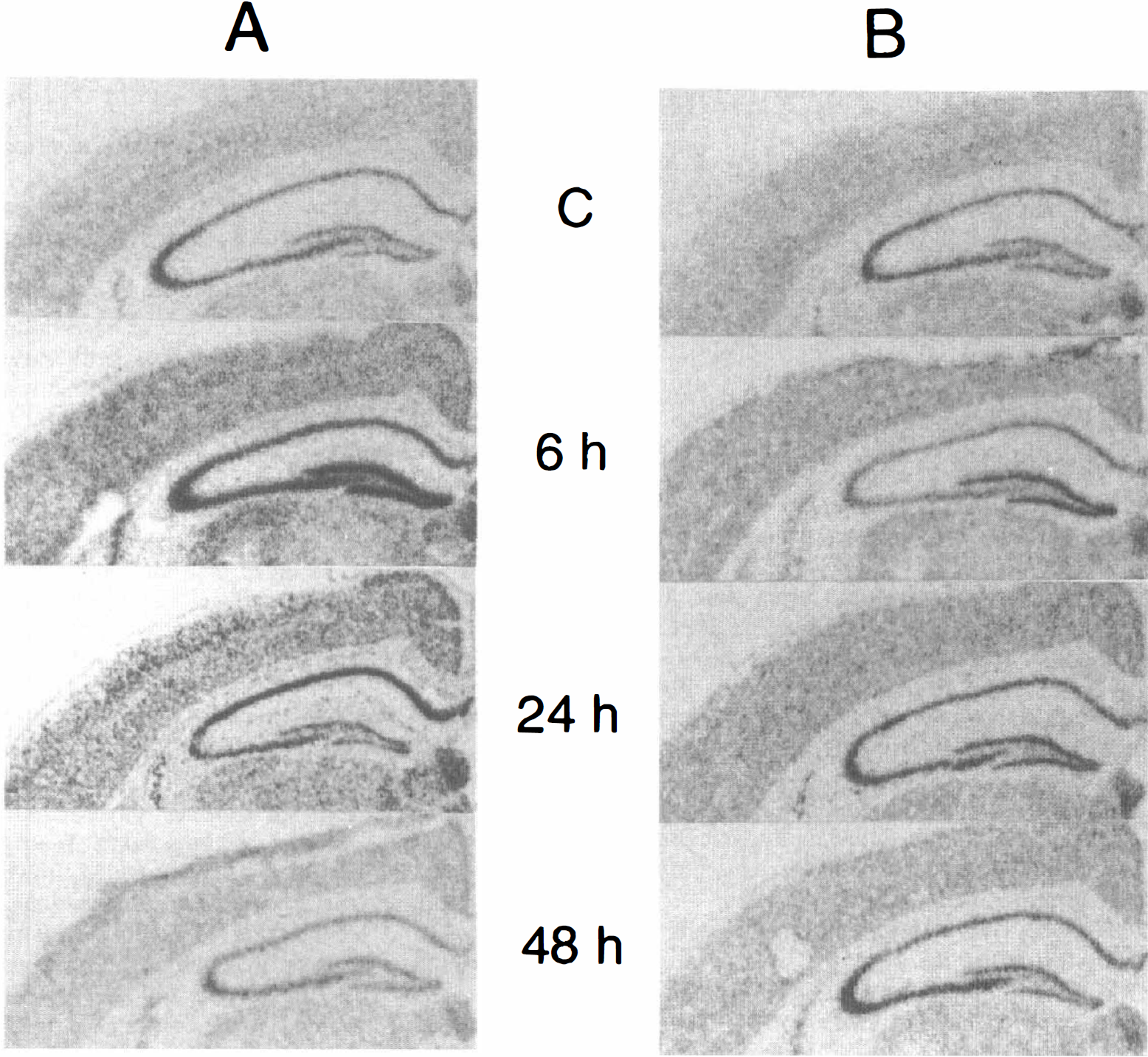
Double ischemia group. In the double ischemia group, ubiquitin mRNA did not change even after the second 5-minute ischemia (Fig. 4B).
DISCUSSION
These results indicate that the persistence or recovery of decreased free ubiquitin correlate well with the neuronal death or survival in the hippocampus after ischemia. Although transient forebrain ischemia resulted in consumption of free ubiquitin and an increase of MUC, these changes were transient in the CA3 + DG region, where neurons survive. On the other hand, the change persisted in the CA1 region, where neurons are destined to die after 5 minutes of ischemia. Ubiquitin mRNA was increased in CA1 neurons after ischemia by in situ hybridization. This means that ubiquitin stress response takes place, at least at a transcriptional level, in the dying CA1 region. It is unknown whether prolonged decrease of free ubiquitin means that ubiquitin turnover is inhibited, or that demands on the ubiquitin system are beyond the ubiquitin turnover. Under tolerant conditions, subsequent ischemia caused not only rapid recovery but further increase of free ubiquitin, and the peak values of increased MUC were lower than those after a single 5 minutes of ischemia. These findings suggest that ubiquitin stress response is quickly expressed, and turnover of ubiquitin is promptly restored and is accelerated in tolerant CA1 neurons.
Magnusson and Wieloch (1989) report continuous disappearance of ubiquitin immunoreactivity after lethal ischemia in the hippocampal CA1 neurons, Morimoto and others (1996) demonstrated by immunohistochemical methods that transient ischemia depletes free ubiquitin but not MUC in the CA1 pyramidal neurons of the gerbil. Also, Hayashi and coworkers (1992a, b, 1993) semiquantitatively showed a slight decrease of free ubiquitin and an increase of MUC in the CA1 region and the cortex after forebrain ischemia, The current data further demonstrate a significant decrease of free ubiquitin by quantitative methods.
After sublethal ischemia, the hippocampal CA1 neurons acquire transient tolerance to subsequent ischemia (Kitagawa et al., 1990; Kirino et al., 1991; Kato et al., 1991). Kato and associates (1993) showed a recovery of ubiquitin immunostaining in the CA1 neurons after ischemia under tolerant conditions. Our quantitative measurement demonstrates that under tolerant conditions, free ubiquitin decreased only transiently after sublethal ischemia, then recovered promptly, and further increased after subsequent ischemia. These results indicate that subsequent ischemia under tolerant conditions in the CA1 region induces stress response of ubiquitin up to the translational level and that an ATP-dependent protein degradation system, including 26S proteasome function, works successfully after ischemia, Although the mechanisms of ischemic tolerance remains unclear, the induction of heat shock proteins is believed to be one of the causative factors, Under tolerant conditions, 70-kd heat shock protein (hsp70) is strongly expressed immunohistochemically after subsequent ischemia (Kirino et al., 1991), although the role of enhanced hsp70 expression remains controversial. The situation is similar with ubiquitin. We speculate that ubiquitin stress response in the CA1 neurons under tolerant conditions may facilitate rapid turnover of ubiquitin and removal of denatured proteins, leading to the restoration of protein synthesis (Nakagomi et al. 1993) and to eventual neuronal survival.
A prolonged decrease of free ubiquitin level and probable inhibited ubiquitin proteolytic pathway are supposed to lead to impairment of degradation of target proteins. Not only are denatured or abnormal proteins degraded by this system, but also intracellular regulatory proteins. Regulatory proteins targeted by ubiquitin include, among others, p53, cyclins, and IkB (Ciechanover, 1994). Nuclear factor kappa B is a critical regulator of genes that function in inflammation, cell proliferation, and apoptosis (Baeuerle and Baltimore, 1996; Barnes and Karin, 1997; Baichwal and Baeuerle, 1997) and is activated by ubiquitination and degradation of IkB. Therefore, if turnover of these proteins is involved in apoptotic cascade, it would be directly related to ischemia-triggered neuronal apoptosis (Beiharz et al., 1995; Crumrine et al., 1994; Dragunow et al., 1995; Nitatori et al., 1995; Du et al., 1996). Moreover, the ubiquitin pathway might be directly involved in apoptosis (Delic et al., 1993; Drexler, 1997). Our current results warrant further study to clarify the relation between ubiquitin proteolytic pathway, proteasome function, and apoptosis in delayed neuronal death of CA1 neurons after ischemia.
Notice that MUC accumulation persisted in urea-soluble fraction in the CA1 region, where pyramidal neurons are destined to delayed neuronal death. Ubiquitin is known to be incorporated in cytoplasmic inclusion bodies in various diseases in the CNS. Such insoluble deposits are found not only in chronic neurodegenerative disorders but also in dementia pugilistica (punch-drunk syndrome) in ex-boxers (Tokuda et al., 1991). These findings, along with our current results, may indicate that metabolic stresses such as ischemia or trauma can trigger abnormal metabolism of ubiquitin, thereby contributing to an accumulation of insoluble ubiquitinated proteins.
In summary, we have shown that transient ischemia induced consumption of free ubiquitin and increase of MUC; these changes are transient in the CA3 + DG region, where neurons survive, and are persistent in the CA1 region, where neurons are destined to die after 5 minutes of ischemia. Under tolerant conditions, decrease of free ubiquitin and increase of MUC recovered promptly in the CA1 and CA3 + DG regions, and free ubiquitin showed further increase.
