Abstract
The stroke-prone spontaneously hypertensive rat (SHRSP) is a strain with high incidence of cerebrovascular accidents increased by salt-rich diet and decreased by calcium-antagonist treatment. In the SHRSP rat basilar artery the authors have previously shown reduced contractility and altered structure including regions of smooth muscle cell (SMC) disorganization, The aims of this study have been to analyze (I) the morphology of these abnormal regions, (2) the structural modifications responsible for the reduced function, and (3) the effect of salt and calcium-antagonist treatment on vascular structure and function, Wistar Kyoto and SHRSP rats, untreated or treated from week 8 through 14 with 1% NaCl or 1% NaCl + 1 ·kg−1·d−1 lacidipine, were used, Function was studied with wire myography, Structure was analyzed in fixed intact arteries with confocal microscopy, Basilar arteries from SHRSP rat showed (1) reduced contractility, (2) discrete foci of SMC disarray with altered proportion of adventitia to SMC, and (3) decreased SMC and increased adventitial cell number. Arteries from salt-loaded SHRSP rats showed a higher degree of SMC disarray and further reduction in contractility, Lacidipine treatment of salt-loaded rats significantly improved structure and function, These data suggest that vascular remodeling can provide an explanation for the observed reduction in vascular contractility of SHRSP rat basilar arteries and might show light on the effects of salt load and calcium-channel blockers in life span and the incidence of cerebrovascular accidents in SHRSP rats.
The stroke-prone spontaneously hypertensive (SHRSP) rat constitutes an animal model of cerebrovascular disorders occurring in hypertension (Okamoto et al., 1974). In these rats, fibrinoid necrosis develops progressively in small intracerebral arterioles leading to vessel obstruction and stroke, In large cerebral arteries there is no fibrinoid necrosis. However, other abnormalities in these arteries, such as a decrease in the vascular compliance, could also participate in the development of stroke by contributing to the alterations in the downstream cerebral microcirculation (Smeda, 1992). In SHRSP rats there is evidence of an association between functional and mechanical impairment in middle cerebral arteries and stroke occurrence (Vacher et al., 1996a; Lee et al., 1996a). A reduction in contraction was also reported by us in the basilar artery from SHRSP rats (Arribas et al., 1996a; Salomone et al., 1997).
Alterations in vascular responsiveness can be caused by changes in vessel structure (Folkow, 1990). In hypertension, an increased wall to lumen ratio is usually associated with enhanced vascular contractility (Mulvany et al., 1978, 1980; Traub et al., 1995). However, in large cerebral arteries from SHRSP rats the increase in wall to lumen ratio is associated with a reduction in contractile responses (Arribas et al., 1996a; Vacher et al., 1996a). Specific cellular alterations such as number, distribution, or orientation can account for the above referred differences in vascular function. Changes in cell distribution or arrangement are difficult to study in intact arteries with conventional techniques. This information cannot be provided with wire or perfusion myography, traditionally used for the study of small vessel structure and function. On the other hand, histologic manipulation, such as tissue embedding and sectioning, alters vessel structure, and therefore, important information on vessel morphology can be lost. We have recently developed methods based on high-resolution laser scanning confocal microscopy (LSCM) and image analysis techniques, which allow for detailed analysis of vascular structure at the cellular level in intact small arteries (Arribas et al., 1994). With this new tool we were able to observe an alteration, not described before, in smooth muscle cell (SMC) orientation in basilar arteries from SHRSP rats (Arribas et al., 1996a). We have further developed methods, based on confocal microscopy and image analysis techniques, to study vascular structure at the cellular level in intact live or fixed small vessels (Arribas et al., 1997a).
In our previous study we described a reduced contractility, vascular remodeling, and altered SMC orientation in SHRSP rat basilar arteries (Arribas et al., 1996a). The aims of the present study were (1) to determine in more detail the wall structure at these points of SMC disorganization, and (2) to analyze other cellular changes in the vessel wall that could account for the reduction in contractility. In SHRSP rats a salt-rich diet increases the incidence of stroke and early death (Okamoto, et al., 1974; Ogata et al., 1980). On the other hand, calcium antagonists have been shown to have a protective effect on stroke occurrence and also to improve cerebrovascular function (Vacher et al., 1996b). The third goal of our study was to evaluate the effect of salt load and calcium-antagonist treatment on basilar artery structure and the functional consequences.
MATERIALS AND METHODS
Animals
Twenty SHRSP and 8 Wistar Kyoto (WKY) male rats (Iffa Credo, L'Arbresle, France) were used. At the age of 8 weeks SHRSP rats were divided at random into three groups: one was given salt-free water (SHRSP control, n = 8), a second group was given 1% NaCl in drinking water (S-SHRSP, n = 6). These two groups received standard rat chow (AO4, UAR Villemoison-sur-Orge, France; NaCl content, 1,900 mg/kg). A third group was given 1% NaCl drinking water and received the same food that contained lacidipine for a mean intake of 1 mg·kg−1·d−1 (SL-SHRSP, n = 6). Control and treated rats were kept in the same environment and received water and food ad libitum. Systolic blood pressure (SBP) was measured at 8 and 14 weeks of age by the tail-cuff method in conscious animals prewarmed to 35°C in thermostatic cages (Physiograph Narco, Houston, TX, U.S.A.). Considering that SHRSP rats show a maximal life span of 12 weeks after the beginning of a salt-rich diet and generally begin to die after 7 to 8 weeks of this treatment (Gaviraghi and Godfraind, 1993), rats were killed in this study by decapitation after 6 weeks of treatment, i.e., at 14 weeks of age. All experiments were performed according to institutional regulations regarding experiments in animals in Belgium.
The brain was removed and immersed in Krebs solution. The basilar artery was dissected from the brain under a binocular microscope (Leica Wild MZ8, Wetzlar, Germany). A 1-mm-long segment was used for the functional study. The rest of the tissue was fixed in 10% formal-saline solution and kept for analysis by LSCM.
Drugs and solutions
Krebs solution of the following composition in mmol/L was used: NaCl 122, KCl 5, CaCl2 1.25, KH2PO4 1.2, MgSO4 1.2, NaHCO3 15, glucose 11.1, pH 7.4 at 37°C. The 100 mmol/L KCl-depolarizing solution was obtained by isotonic replacement of NaCl with KCl in the Krebs solution (NaCl 27, KCl 100, CaCl2 1.25, KH2PO4 1.2, MgSO4 1.2, NaHCO3 15, glucose 11.1, pH 7.4 at 37°C). Drugs used included Hoechst 33342 trihydrochloride purchased from Sigma Chemical Co., Poole, U.K. Lacidipine was purchased from Glaxo, Verona, Italy.
Wire myography
Segments of the proximal area of the basilar artery (1-mm length) were mounted on a wire myograph (400A, JP Trading, Aarhus, Denmark). After mounting, each preparation was equilibrated, unstretched, for 20 minutes in Krebs solution, maintained at 37°C and aerated with a gas mixture of 95% O2 and 5% CO2, The vascular segments were then set to the normalized effective internal diameter, 0.91100' as previously described (Mulvany and Halpern, 1977). Contractile responses to 100 mmol/L KCl (osmotically corrected) were recorded into a computer, by using data acquisition hardware (MacLab) and data recording software (Chart v3.4.2, AD Instruments, Pty Ltd., Castle Hill, Australia). Vascular responses were calculated in active wall tension (millinewtons per millimeter).
Confocal microscopy
The fixed arteries were incubated for 15 minutes with 0.01 mg/mL of the nuclear dye Hoechst 33342 and washed for 5 minutes in Krebs solution. The arteries were then mounted on a slide with Krebs solution and their length measured with a grid under the dissecting microscope. Then, they were viewed with an Odyssey LSCM (Noran Instruments, Middleton, Wisconsin, U.S.A.) with a ×40 water objective (Nikon, NA 1.15; Kingston on Thames, U.K.), using the 364/400-nm line of the LSCM, with a pinhole aperture of 10 µm. Stacks of 1-µm-thick serial optical slices were taken in the z-axis from the top of the adventitia to the border between the media and intima. MetaMorph image analysis software (Universal Imaging Corporation, West Chester, PA, U.S.A.) was used for the image acquisition, measurements, and 3-dimensional reconstructions.
The medial-intimal border was determined by the internal elastic lamina, which is autofluorescent and was detected with the 488/515-nm line of the LSCM. In the basilar artery the external elastic lamina is not as clearly visible as in other muscular arteries, for example in mesenteric resistance arteries (Rhodin, 1980). With the aid of LSCM we have demonstrated that in this vascular bed a fibrous, clearly visible external elastic lamina separates oval-shape nuclei in the outer layer from elongate-shape SMC nuclei in the media (Arribas et al., 1994). Therefore the adventitial-medial border was determined by differences in the shapes of adventitial and SMC nuclei. Nuclei morphology was determined by computer analysis of shape factor (a value in the range 0 to 1 representing how closely an object resembles a circle; see results). The differences in shape factor determined by image analysis software also allowed us to measure in each stack of confocal images (all images = 95 × 95 µm) the following parameters: (1) number of cells in the adventitia, (2) number of cells in the media, and (3) angle of orientation of SMC in the media. This angle was measured with respect to the longitudinal axis of the vessel (shown by the orientation of the internal elastic lamina). We have previously shown that in the rat basilar artery the normal angle of orientation of the SMC is close to 90° (Arribas et al., 1996a). The cells located in different parts of the vessel wall are subject to different local influences (e.g., factors secreted from nerves or endothelium, circulating substances). We had previously shown that in old SHRSP rats disorganization of SMC was higher near the adventitial layer (Arribas et al., 1996a). We wanted to evaluate whether that was the case in our present experimental conditions and therefore evaluated the degree of disorientation in three layers of the media: near adventitia (outer), near endothelium (inner), and a middle layer (middle). Confocal microscopy allows for such division as it is possible to focus at different depths within the artery and all the cells located in a single plane of focus are clearly shown without interference of planes above and below.
Throughout the text the angle of SMC is expressed as deviation from 90°. Every artery was systematically checked for regions of SMC disorganization as previously described (Arribas et al., 1996a), and the number of these regions was counted. From each artery two stacks of serial optical sections were captured from regions with normal SMC orientation, and one additional stack was taken from a disorganized region (if found) for comparison.
In each vessel the thickness of adventitial and medial layers in the regions of SMC disorganization was measured and compared with the surrounding tissue. The comparison of wall thickness between SHRSP and WKY rat basilar arteries was not possible, as the vessels were not pressurized and the comparison between normotensive and hypertensive rats should be made at the transmural pressures equivalent to those experienced in vivo (Ashen et al., 1994).
Statistics and data analysis
Results are expressed as mean ± standard deviation (SD) and n denotes the number of animals used in each experiment. Statistical comparisons were made using Student's t–test for paired or unpaired experiments with Bonferroni correction for multiple comparisons. A P value of less than 0.05 was considered significant.
RESULTS
LSCM analysis of WKY and SHRSP rat basilar arteries
We have previously shown that the medial layer of WKY rat basilar arteries is composed of circularly arranged SMC. This homogeneous arrangement is disrupted at discrete points of the vessel media in SHRSP rats (Arribas et al., 1996a). The present results, performed in a different colony of SHRSP rat, confirms our previous finding. In these regions the angle of orientation of the SMC was significantly different when compared with the rest of the artery, and this disorganization affected the three layers of the media being more marked in the outer layer (Fig. 1). Morphology of WKY basilar arteries was homogeneous along vessel length and no regions of SMC geometric alteration were found. The angle of orientation of the SMC in WKY arteries was similar along the entire basilar artery with a very small deviation from 90° (outer layer, 8.4° ± 0.8°; middle layer, 8.5° ± 1.4°; inner layer, 7.9° ± 1.1°).
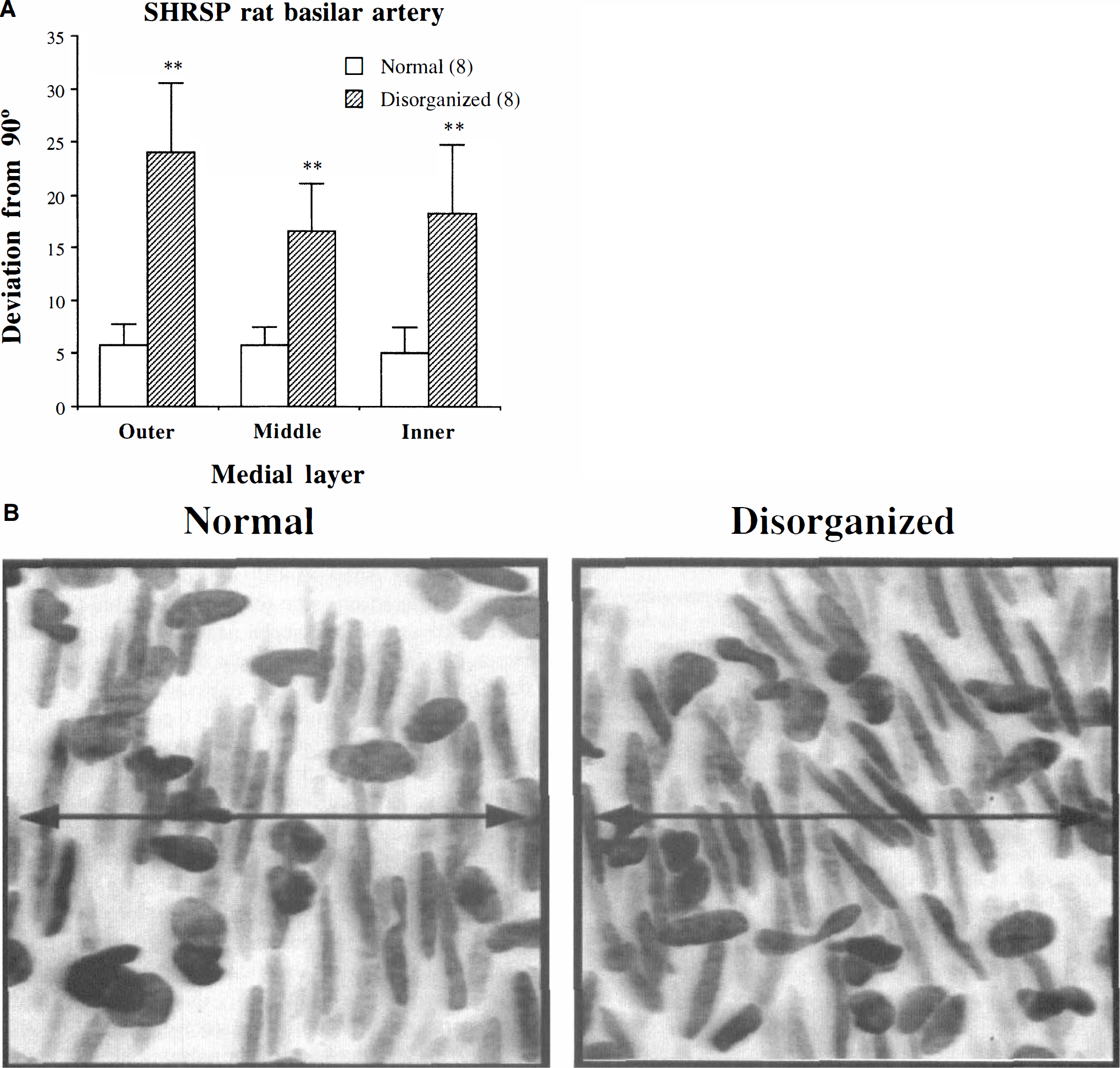
Orientation of the smooth muscle cells (SMC) in the three layers of the media of the basilar artery from stroke-prone spontaneously hypertensive (SHRSP) rats. The arteries were stained with the nuclear dye Hoescht 33342 (0.01 mg/mL), and the cell nuclei were visualized with a laser scanning confocal microscope (364/400-nm line). Stacks of images of 1-µm-thick optical slices were taken from adventitia to internal elastic lamina and stored for further analysis. MetaMorph image analysis software was used to measure the angle of orientation of the SMC with respect to the main axis of the vessel and for three-dimensional reconstructions. (
We further characterized the cell distribution and structure of these regions. The foci of SMC disorganization were present in all SHRSP rat basilar arteries analyzed. They were randomly located and two to three of these foci were found per basilar artery (density, 0.35 ± 0.06 foci/mm length). Each region of SMC disorganization covered an area of approximately 150 × 150 µm, involving 30 to 40 SMC. The structural characteristics of the adventitial and medial layers from these regions of SMC disorganization were compared with those of normal SMC orientation from the same SHRSP rat basilar arteries. These comparisons were made on the basis of the clear differences in the shape of adventitial and SMC nuclei. Adventitial cell nuclei had an oval shape (shape factor, 0.76 ± 0.01; length, 12.5 ± 0.4 µm; breadth, 7.7 ± 0.3 µm). Nuclei of SMC had an elongated shape (shape factor, 0.35 ± 0.01; length, 24.5 ± 0.6 µm; breadth, 4.5 ± 0.1 µm; P < 0.01 compared with adventitial cell nuclei). In SHRSP rat basilar arteries, cell number and thickness of each layer were different in the regions of SMC disorganization compared with the surrounding tissue with normal SMC orientation. In the foci of disarrayed SMC, adventitial cell number was significantly decreased whereas SMC number was significantly increased when compared with the rest of the artery (Fig. 2A). Adventitia thickness did not differ significantly between normal and disorganized regions, but media thickness was significantly larger in the regions of disarrayed SMC (Fig. 2B).
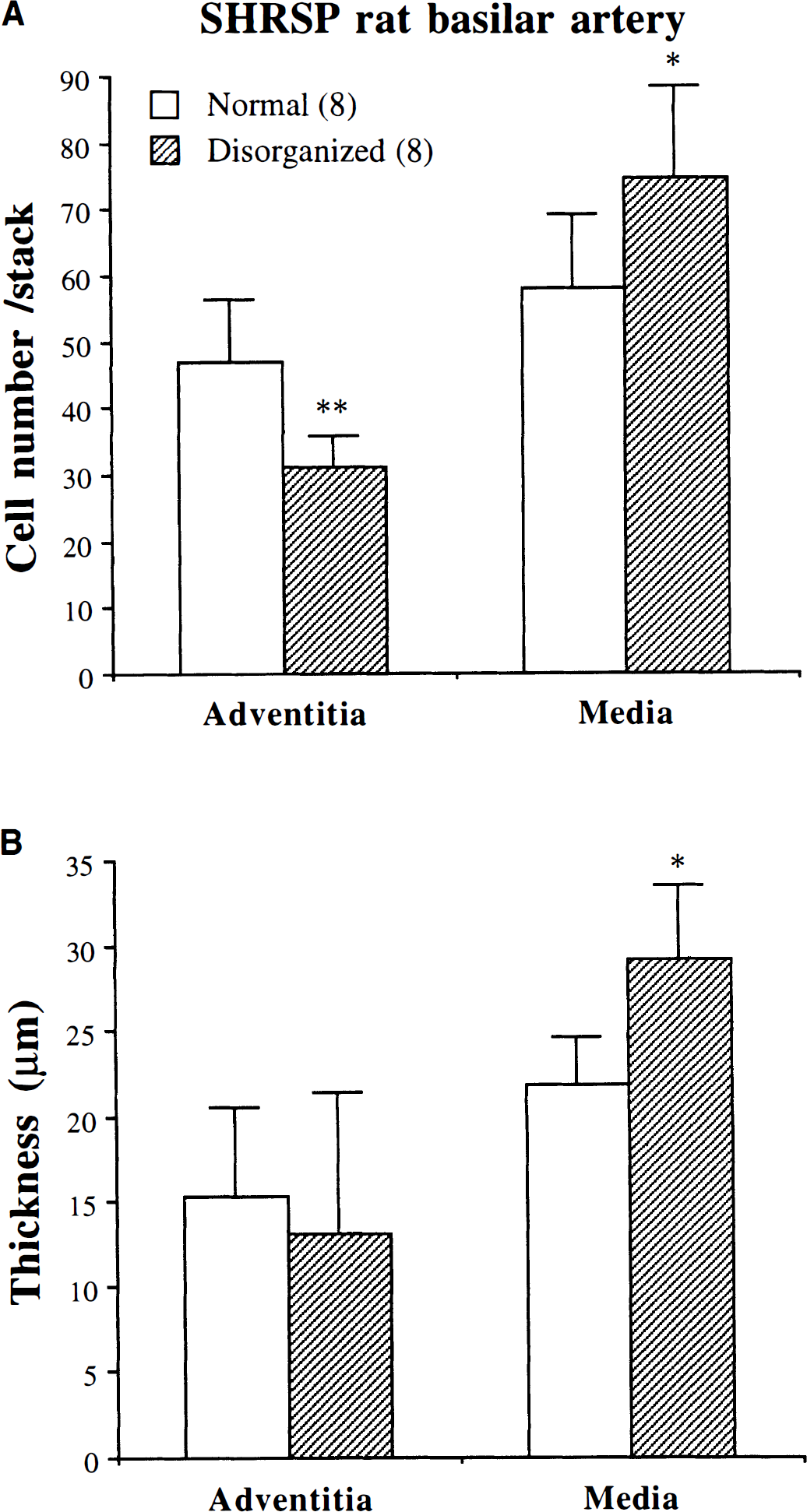
Comparison of the morphology of adventitia and media layers between regions of normal SMC orientation and foci of SMC disorganization in the basilar artery from SHRSP rats. Protocol is as in Fig. 1. MetaMorph image analysis software was used for measurements of layer thickness and cell number. Results are expressed as means ± SD; n denotes number of animals; ** P < 0.01, * P < 0.05 compared with normal regions (paired experiments). (
Cellular characteristics of the adventitial and medial layers from WKY and SHRSP rat basilar arteries were also compared. For this comparison only regions of normal orientation were analyzed. In WKY arteries the thickness of adventitia and medial layers was constant along the vessel length (adventitia, 8.8 ± 4.2 µm; media, 28.5 ± 3.1 µm). Cell number in each layer was also homogeneous. Total number of cells was similar in SHRSP and WKY rat basilar arteries (SHRSP, 105.5 ± 15.6 cells/stack; WKY, 102.8 ± 8.1 cells/stack). However, cell distribution and morphology were different in SHRSP and WKY. Basilar arteries from SHRSP rats showed a 34% increase in adventitial cell number and a 24% decrease in SMC number when compared with WKY (Fig. 3A). In WKY arteries, adventitia was only composed of oval-shape nuclei. In some SHRSP arteries elongated nuclei were observed in the outer part of the adventitial coat (Fig. 3B).
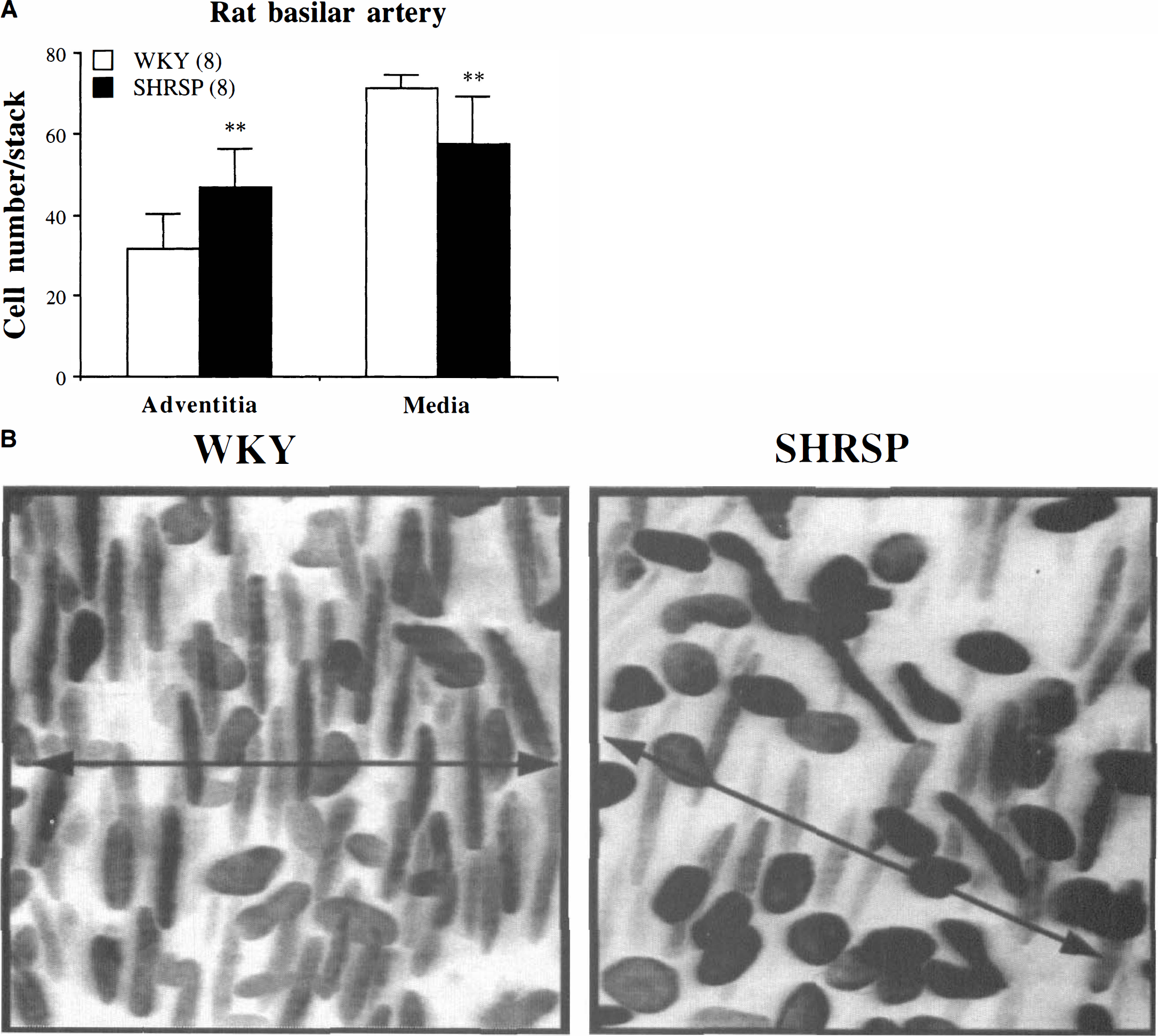
(
Effect of salt load and lacidipine treatment on SHRSP rat basilar arteries
At the age of 8 weeks SBP was already significantly increased in SHRSP rats when compared with WKY (WKY, 129.1 ± 6.5; SHRSP, 180.0 ± 12.2 mm Hg; P < 0.01). Systemic blood pressure was not stabilized at the age of 8 weeks. Between 8 and 14 weeks SBP increased by 10 mm Hg in WKY and more than 60 mm Hg in untreated SHRSP. High-salt diet from week 8 to week 14 tended to increase SBP in SHRSP rats. However, this increase did not reach significance. SHRSP rats treated with high-salt diet and 1 mg·kg−1·d−1 lacidipine during the same 6-week period showed a significant reduction in SBP when compared with both untreated salt-loaded and control SHRSP rats. Lacidipine treatment did not normalize SBP, which remained significantly different when compared with that of WKY rats (Table 1).
Basilar artery contractility and lumen diameters were determined in 14-week-old rats. At 14 weeks lumen diameters and contractions to 100 mmol/L KCl were significantly decreased in SHRSP rat basilar arteries compared with WKY arteries. High-salt diet induced a further and significant reduction of lumen diameter and contractile responses compared with SHRSP controls. Lacidipine treatment significantly increased lumen size and contractility to levels similar to control SHRSP rats, but did not restore normal structure and function (Table 1).
Effect of salt-load and lacidipine treatment on SHRSP rat
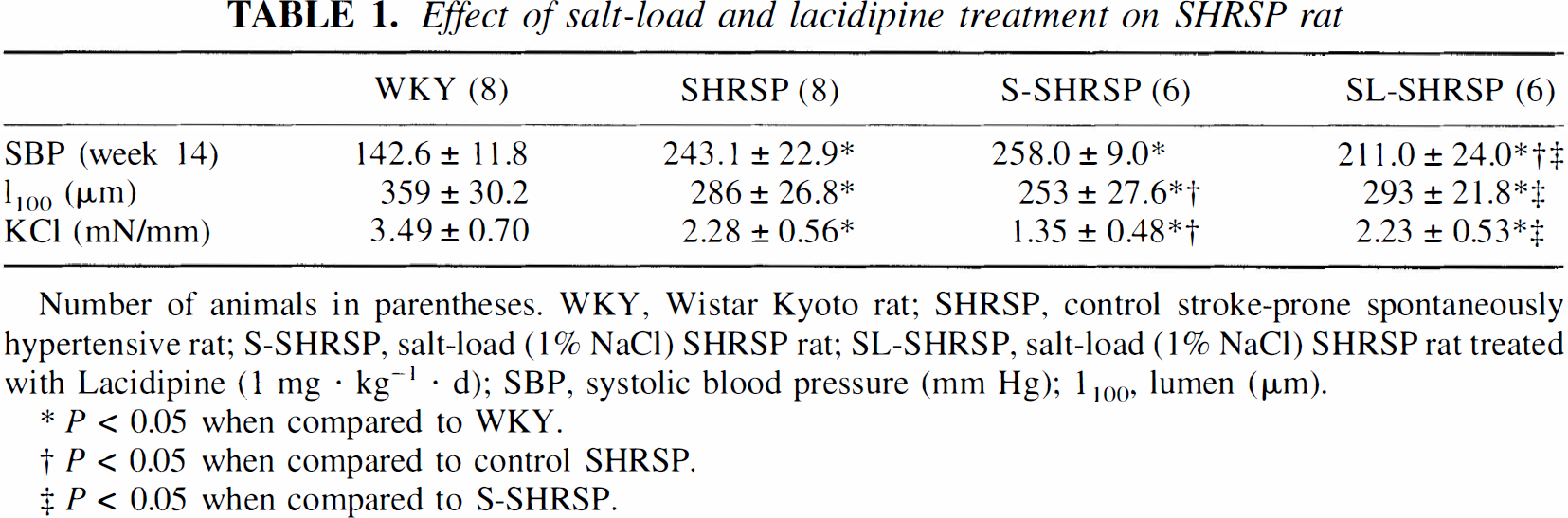
Number of animals in parentheses. WKY, Wistar Kyoto rat; SHRSP, control stroke-prone spontaneously hypertensive rat; S-SHRSP, salt-load (1% NaCl) SHRSP rat; SL-SHRSP, salt-load (1% NaCl) SHRSP rat treated with Lacidipine (1 mg · kg−1 · d); SBP, systolic blood pressure (mm Hg); 1100, lumen (µm).
P < 0.05 when compared to WKY.
P < 0.05 when compared to control SHRSP.
P < 0.05 when compared to S-SHRSP.
Basilar arteries from salt-loaded rats showed discrete regions of SMC disorganization, as found in the SHRSP controls. When compared with control SHRSP rats, disorganized regions from salt-loaded rats showed an increase in the degree of SMC disarray. The SMC in these regions had a significantly higher degree of deviation from 90°. Basilar arteries from salt-loaded rats treated with lacidipine also showed regions of SMC disorganization, which were found in all basilar arteries analyzed. However, there was a significant reduction in the degree of SMC disorganization in rats treated with lacidipine compared with untreated salt-loaded rats (Fig. 4A). Cell number was analyzed in normal regions from salt-loaded SHRSP rats with and without lacidipine treatment and compared with control SHRSP. In salt-loaded SHRSP rats adventitial cell number tended to increase compared with SHRSP controls, but this increase did not reach significance. Lacidipine treatment of salt-loaded rats significantly reduced adventitial cell number (Fig. 4B). The number of SMC was similar in untreated and salt-loaded SHRSP. Lacidipine treatment significantly increased SMC number (Fig. 4C).
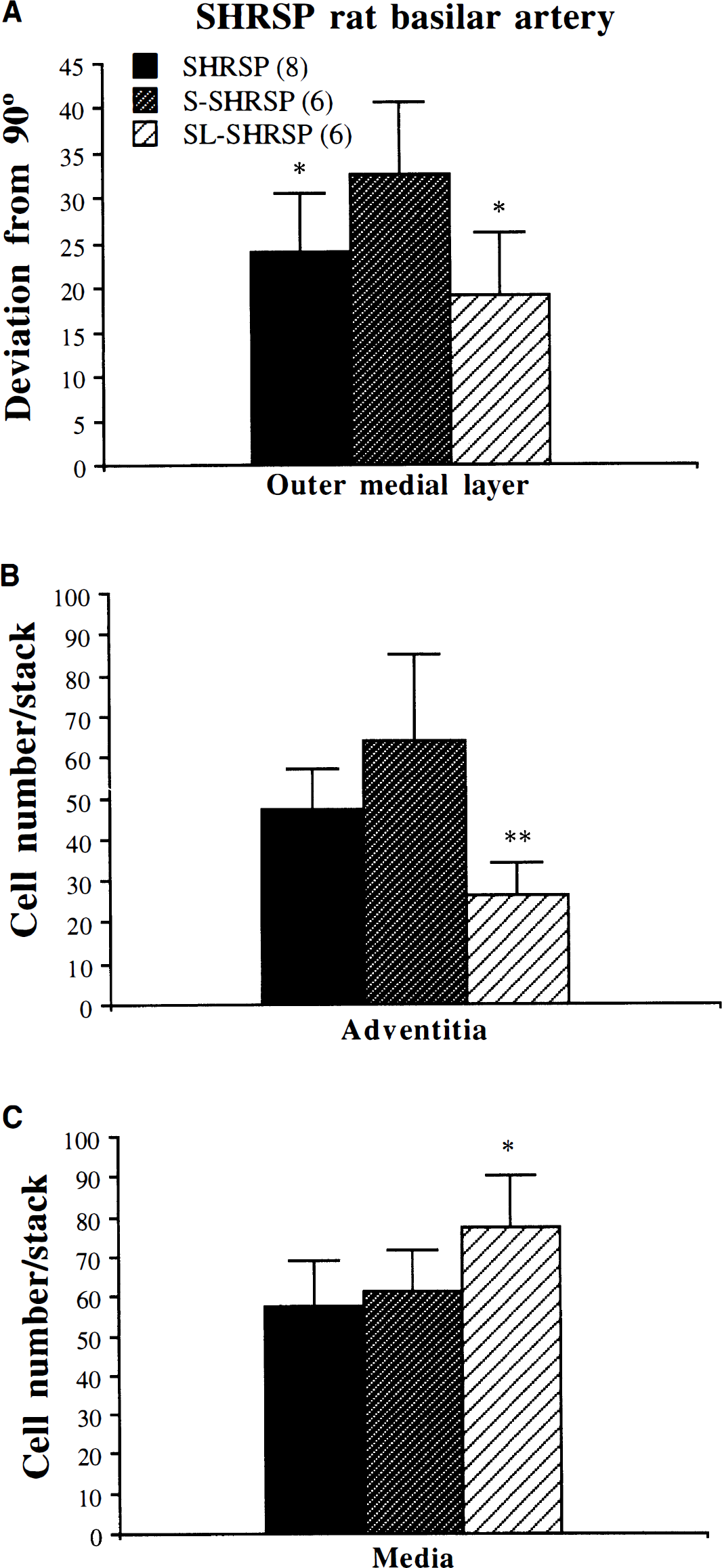
Effect of salt load (1% NaCl) and lacidipine treatment (1 mg·kg−1·d−1) on the structural alterations in SHRSP rat basilar arteries. Protocol is as in Fig. 1. MetaMorph image analysis software was used for measurement of angle of orientation of the SMC and to count cell number. (
As found in control SHRSP, in S-SHRSP and SL-SHRSP rat basilar arteries the pattern of cellular distribution was altered in the regions of SMC disorganization, showing a decreased adventitial cell number and an increased SMC number compared with the surrounding tissue (data not shown).
DISCUSSION
In our previous study we described a reduction in vascular contractility and vascular remodeling in SHRSP rat basilar arteries (Arribas et al., 1996a). The structural modification included decreased lumen size, increased wall thickness without net growth, and regions of disorganized SMC. A similar decrease in vascular responses was later reported by Salomone and coworkers in the same vessel (Salomone et al., 1997). The present study confirms our previous findings and demonstrates the existence of these regions of irregular SMC orientation in the SHRSP colony used in their study. These regions are characterized by an increased SMC to adventitial cell and media to adventitial thickness rate. In addition to these foci of SMC disorientation, SHRSP rat basilar arteries showed a decreased SMC number, increased adventitial cell number, and reduced lumen compared with WKY arteries. These alterations in SHRSP rat basilar structure are exaggerated in salt-loaded animals and reversed by treatment with the calcium antagonist lacidipine. In both cases the degree of structural abnormalities correlate with the degree of functional reduction. The present data suggest that vascular remodeling can provide an explanation for the observed reduction in vascular contractility of SHRSP rat basilar arteries.
Definition of vascular remodeling
It is well established that hypertension is associated with structural changes in small vessels such that the lumen is reduced and the wall to lumen ratio is increased. These changes may not be associated with net growth, and this mode of structural alteration was termed vascular remodeling by Baumbach and Heistad (1989). The term vascular remodeling means different things to different authors and is still question of debate (Mulvany et al., 1996; Lee et al., 1996b; Komer and Angus, 1997). We use this term to describe the structural changes found in SHRSP rat basilar artery; it is therefore important to clarify how we define vascular remodeling.
Vascular remodeling is used here in a broad sense to describe any change in vascular structure from a situation A to a situation B. This broad definition could include modifications of the structure of a given vessel through time, which are physiologic (i.e., developmental), pathologic (i.e., caused by hypertension, atherosclerosis, and so forth), or caused by intervention (i.e., after treatment). In the second case, this might be a comparison of two different vessels, for example in two different animals, where they might have been expected to be similar without the intervening factor. In the present study we are dealing with a pathologic situation, hypertension, imposed over a period when normal developmental remodeling is also taking place. Therefore we consider remodeling to be the structural differences observed in SHRSP rat basilar arteries compared with the normotensive reference strain WKY and concede that the term remodeling is etymologically inaccurate. However, this usage is used throughout the current literature. Overall we suggest that different remodeling might be better than remodeling in such cases. In our view vascular remodeling is related not only to gross structural differences, such as increase or reduction in lumen size and wall thickness, but also to modifications at the cellular level, such as cell number, distribution, and orientation. In the case of the interventions with salt load and lacidipine, there is more logic in using remodeling because a relative change has been imposed.
Vascular abnormalities in SHRSP rat basilar arteries
The present study shows a reduced vasoconstrictor response in SHRSP rat basilar arteries. Several studies have reported a decrease in function of cerebral arteries from SHRSP rats. Hajdu and Baumbach (1994) demonstrated a reduction in vascular distensibility in large cerebral vessels from this strain. In the middle cerebral artery, a loss of myogenic response, i.e., constriction in response to elevation of transmural pressure, has been described (Smeda, 1992; Lee et al., 1996). In the same artery, Vacher et al. (1996a) found a decrease in contractile responses to KCl and serotonin. Other studies suggest that responses to vasoconstrictors are normal in cerebral arteries from SHRSP. Volpe and coauthors (1996) found no difference in the responses to serotonin in basilar arteries from SHRSP compared with SHR. The difference might be related to the strains used in that study (SHRSP versus SHR). It is also possible that it is related to the contractile agent used, as reactivity to agonists could be differentially regulated compared with the reactivity to KCl solution. We have also observed a greater contractility to serotonin, but decreased responses to KCl in SHRSP compared with WKY (Salomone et al., 1997). This was related to a smaller endothelium-dependent release of nitric oxide in SHRSP rat basilar artery. Potassium chloride is an unspecific vasoconstrictor agent, and the responses to KCl mainly reflect the contractile ability of the tissue.
The response of small arteries to stimuli is critically dependent on vessel structure (Mulvany and Halpern, 1977). Hypertension is usually associated with a reduction in lumen size and an increase in wall to lumen ratio. This altered vascular structure has been proposed to play a role in the enhanced responsiveness observed in resistance and conduit arteries in hypertension (Schiffrin, 1992). However, in the rat basilar artery (Arribas et al., 1996a; present study), as well as in middle cerebral arteries (Vacher et al., 1996a) the opposite was found: the response was smaller despite a reduction in lumen size and increase in wall to lumen ratio. Based on these findings we postulated that modifications of vascular structure at the cellular level could be responsible for the functional reduction in SHRSP rat cerebral arteries.
Analysis of rat basilar arteries with LSCM showed a 24% reduction in SMC number in SHRSP rats compared with the normotensive reference strain WKY. We suggest that this reduction of SMC might contribute to the compromised contractile function. It is interesting to note that in the same vessels a concomitant increase in adventitial cell number was also found whereas the total number of cells was similar in SHRSP and WKY rat basilar arteries. We have previously demonstrated that in SHRSP rat basilar arteries there is no increase in the cross-sectional area (Arribas et al., 1996a). Therefore the amount of material is similar to that of WKY rats, but redistributed around a smaller lumen. The present results confirm that in this hypertensive strain, vascular remodeling of the basilar artery is characterized by a redistribution of cellular components.
In the present study the nature of the cells in the vascular wall was characterized by shape factor analysis. With the use of confocal microscopy and nuclear, intracellular, and extracellular dyes, we have previously demonstrated that the elongated nuclei observed in the media of mesenteric and cerebral arteries correspond to SMC (Arribas et al., 1994, 1996a). In the present study only cells with elongate-shape nuclei, significantly different from oval-shape nuclei present in the adventitial layer, were found in the media, and therefore we assume they are SMC. The nature of the cells in the adventitial layer is less clear. Histologic data demonstrate that fibroblasts are restricted to adventitial layer and that SMC is the only cellular component of the media (Rhodin 1980). However, in a pathologic situation this cellular composition and distribution may change. It is possible that a proportion of the nuclei in the outer layer of SHRSP basilar arteries corresponds to SMC with a changed phenotype and nuclear shape. There is clear evidence that arterial SMC occurs in at least two distinct states, usually referred to as a synthetic and a contractile phenotype. Synthetic-state cells have a fibroblast-like appearance, and their main functions are (like fibroblasts) to proliferate and produce extracellular matrix components. Contractile-state cells have a smooth muscle-like appearance, and their main function is contraction and thereby control of blood pressure (Thyberg et al., 1990). We do not have direct evidence of this alteration in SHRSP rat basilar arteries. However, several lines of evidence suggest this possibility. (1) The present results show that in SHRSP, but not in WKY, basilar arteries, elongated nuclei with a shape factor of SMC are distributed on the arterial surface. (2) Smooth muscle cells in the contractile phenotype are able to return to the synthetic phenotype, and this change is associated with pathologic situations such as atherosclerosis and hypertension (Thyberg et al., 1990; Korner and Angus, 1997). (3) Smooth muscle cells have the ability to change phenotype and migrate to the adventitia (De Mey et al., 1991; Shi et al., 1996). (4) In hypertensive rats during early life the synthetic phenotype is more proliferative than is the contractile phenotype (Scott-Burden et al., 1989). (5) In aortic and coronary SMC from SHRSP rats, a change in contractile phenotype has been demonstrated: these SMC present an accumulation of nonmuscular myosin and fibronectin, making them less efficient in the contractile process (Contard et al., 1993). Based on these findings, we believe that a change in SMC phenotype as part of the remodeling process and reduction in contractile function in SHRSP rat basilar artery is a possibility that requires further evaluation.
The second structural abnormality found in SHRSP rat basilar artery is the foci of SMC disorganization located randomly along the basilar artery. In our previous study we postulated the hypothesis that this alteration could contribute to the reduction in vascular function because the cells in these regions would be less efficient at developing force owing to the irregular geometric orientation. Under our experimental conditions it was not possible to test potential differences in contractility between segments with and without SMC disorganization. The manipulation of the artery to identify exact location of disorganized regions (i.e., mounting tissue on slide, laser from confocal microscope) damages the arterial function. This hypothesis still remains a possibility supported by the fact that in salt-loaded SHRSP functional responses of the basilar artery were further reduced compared with non-salt-loaded SHRSP rats, and this reduction was accompanied by a greater degree of SMC disorganization but no further decrease in SMC number. However, it is important to note that salt loading also induced a further reduction in lumen size. This gross structural alteration in vessel geometry could also contribute to the reduction in contractile responses.
We do not think high blood pressure or salt load are the initial driving forces for the disorganization of SMC found in this artery. However, they might contribute to the further development of these regions and might be responsible for some of the other structural changes observed in SHRSP rat basilar artery structure, such as decreased lumen and increased wall to lumen ratio. Several lines of evidence support this hypotheses. (1) We have demonstrated that antihypertensive treatment with perindopril or losartan, before hypertension is developed, does not prevent the occurrence of the disorganization although it is able to normalize other structural alterations (Arribas et al., 1996a, 1996b). (2) We have found that experimental hypertension in WKY rats (the control reference strain) does not induce this structural alteration in the basilar artery (Arribas et al., 1997b). (3) Studies of SMC disorganization in basilar arteries from the F2 generation of a cross between SHRSP and WKY rats show that these foci of SMC with abnormal orientation are inherited in 30% of the animals independent of hypertension (unpublished results). (4) The foci of SMC disorganization are found in salt-loaded as well as in control SHRSP rats, although salt loading seems to induce a greater degree of disorganization. Therefore, we suggest that initial embryonic development, rather than hypertension, is responsible for this abnormality.
Consequences of the structural and functional alterations in SHRSP rat basilar arteries
The current study presents evidence of several structural abnormalities in SHRSP rat basilar arteries: a reduction in SMC number, altered vessel geometry, and regions of SMC disorganization where there is an abnormal proportion of SMC and adventitial cells. How could they contribute to the increased incidence of stroke occurrence in this strain? The term stroke includes both infarcts and hemorrhages. In SHRSP rats infarcts are more prominent in regions of the brain supplied by branches of the carotid artery rather than in the posterior cerebral artery territory (Yamori et al., 1982). In SHRSP rats hemorrhagic infarcts, together with fibrinoid necrosis, have also been described (Fredriksson et al., 1985). The structural and functional alterations found in SHRSP rat basilar arteries in our study support the possibility of hemorrhages rather than infarcts. The present study shows foci of SMC disorientation in SHRSP rat basilar artery. In these regions, in addition to an altered SMC geometric orientation, adventitial to medial distribution of cells and thickness is altered in favor of the medial layer. The adventitia renders the vascular wall a fair amount of stability (Rhodin, 1980). A similar disarray of SMC has been recently described at early stages of a saccular aneurysm formation (Kondo et al., 1998). It is therefore possible that the vascular wall would be weaker at this point, leading to rupture on sudden increases in blood pressure.
The reduction in contractile ability of SHRSP rat basilar artery might also contribute to hemorrhages. As a result of the deficit in large vessel contractility, downstream arterioles would no longer be protected against episodes or bursts of blood pressure. This would promote overperfusion of more distal vasculature when autoregulation limits are exceeded and also lead to hemorrhage. There is evidence that the loss of responsiveness in large vessels contributes to blood-brain barrier damage and fibrinoid necrosis development in downstream arterioles (Smeda, 1992; Hajdu and Baumbach, 1994). In addition, a correlation between decreased contractility and stroke occurrence has also been found in SHRSP rats (Lee et al., 1996a; Vacher et al., 1996a).
Effect of salt load and lacidipine treatment on SHRSP rat basilar artery
Salt loading of SHRSP rats induced a further reduction in basilar artery contractile response. In addition to a reduced contractile function, basilar arteries from salt-loaded SHRSP rats also showed a greater degree of SMC disorganization and a further reduction in lumen size. This increase in the degree of structural and functional alterations could provide an explanation for the increased incidence of cerebrovascular accidents and early death in SHRSP rats after salt loading (Okamoto et al., 1974; Ogata et al., 1980).
The structural and functional alterations induced by a salt load were attenuated by concomitant treatment with lacidipine. Basilar arteries from SHRSP rat treated with lacidipine showed an increase in lumen size, SMC number, and vascular responses to KCl. We have previously shown that perindopril (Arribas et al., 1996a) and also losartan (Arribas et al., 1996b) prevented blood pressure increase and the structural abnormalities (increased wall to lumen ratio) of the basilar arteries. However, none of these treatments had an effect on the structural disorganization of SMC. The increase in SMC observed with lacidipine might be specific for this drug as it was not observed in losartan-treated animals (Arribas et al., 1998). Calcium-antagonist treatment has been shown to improve life span and reduce stroke occurrence in SHRSP rats (Gries et al., 1989). This improvement has been associated with an increase in contractility of large cerebral arteries (Vacher et al., 1996b). Our study supports this finding and provides a structural explanation for the improvement in contractile function after lacidipine treatment: an increase in the number of SMC and lumen size and a reduction in SMC disorientation compared with untreated salt-loaded SHRSP rats. It has been argued that the beneficial effects of calcium-channel blockers on stroke occurrence could be related to blood pressure reduction (Gries et al., 1989; Takakura et al., 1994). Other studies, however, have demonstrated a full protection against stroke with a nonhypertensive dose of calcium antagonists (Shinyama et al., 1995). In our study lacidipine treatment at the dose of 1 mg·kg−1·d−1 had a small antihypertensive effect, but did not restore normal blood pressure. We have also demonstrated that a lower dose of lacidipine with no antihypertensive effect significantly reduced ventricular hypertrophy in salt-loaded SHRSP rats (Feron et al., 1995). These findings suggest that factors other than limitation of high blood pressure also play a role in the drug-induced improvement on cardiovascular structure.
Using high-power LSCM we have characterized some features of vascular remodeling in intact SHRSP rat basilar arteries. The structural alterations include abnormal vessel geometry, SMC and adventitial cell number, and SMC orientation. These morphologic abnormalities and subsequent loss of contractile function are enhanced by salt loading and reduced by calcium-antagonist treatment. These results provide a structural explanation for the decreased contractility in SHRSP rat basilar artery and might show light on the effects of salt loading and calcium-channel blockers in the life span and incidence of cerebrovascular accidents in SHRSP rats.
Footnotes
Abbreviations used
Acknowledgments
The authors thank Mr. Simon McGrory for his technical assistance with image analysis software measurements. Lacidipine was provided by Glaxo Wellcome (Verona, Italy).
