Abstract
Although stroke in humans usually afflicts the elderly, most experimental studies on the nature of cerebral ischemia have used young animals. This is especially important when studying restorative processes that are age dependent. To explore the potential of older animals to initiate regenerative processes after cerebral ischemia, the authors studied the expression of the juvenile-specific cytoskeletal protein, microtubule-associated protein (MAP) 1B, and the adult-specific protein, MAP2, in male Sprague-Dawley rats at 3 months and 20 months of age. The levels of MAP1B and MAP2 transcripts and the corresponding proteins declined with increasing age in the hippocampus. In the cortex, the levels of the transcripts did not change significantly with age, but the morphologic features of immunostained fibers were clearly affected by age; that is, cortical MAP1B fibers became thicker, and MAP2 fibers, more diffuse, in aged rats. Focal cerebral ischemia, produced by reversible occlusion of the right middle cerebral artery, resulted in a large decrease in the expression of both MAP1B and MAP2 in the infarct core at the messenger ribonucleic acid and protein levels. However, at 1 week after the stroke, there was vigorous expression of MAP1B and its messenger ribonucleic acid, as well as MAP2 protein, in the border zone adjacent to the infarct of 3-month-old and 20 month-old male Sprague-Dawley rats. The upregulation of these key cytologic elements generally was diminished in aged rats compared with young animals, although the morphologic features of fibers in the infarct border zone were similar in both age groups. These results suggest that the regenerative potential of the aged rat brain appears to be competent, although attenuated, at least with respect to MAP1B and MAP2 expression up to 20 months of age.
Keywords
After reversible occlusion of the middle cerebral artery (MCA) in rats, an infarct surrounded by a border zone develops in the cortex. The border zone, or penumbra, is characterized by the sporadic loss of neurons, often in certain layers, and the development of glial changes. The immediate early genes, such as c-fos, c-jun, junB, and zif-268, are rapidly induced by neuronal injury after stroke (Uemura et al., 1991; Collaco-Moraes et al., 1994; Neumann-Haefelin et al., 1994; Herdegen and Zimmerman, 1995). Immediate early genes encode transcription factors that in turn regulate the expression of late genes, some of which may contribute to neuronal survival and recovery of function.
Although direct experimental evidence still is lacking, it is conceivable that the expression of late genes contributes to the recovery of motor and cognitive function after cerebral injury. Possible candidates are the genes coding for microtubule-associated proteins (MAP), which are key regulators of neural morphogenesis. During morphogenesis, “early” MAP (represented by MAP1B, juvenile tau, and MAP2c) are replaced with “late” MAP (MAP1A, MAP2, MAP2a+b, tau). Late MAP are assumed to contribute to structural stabilization, so that proper synaptic circuitry is maintained in the mature brain (Marsden et al., 1996; Hirokawa et al., 1996). Microtubule-associated protein 1B, which also is referred to as MAP5 and MAP1X (Tucker, 1990), is strongly associated with neuritic outgrowth during development (Riederer et al., 1985; Calvert et al., 1987; Schoenfeld et al., 1989). Moreover, MAP1B messenger ribonucleic acid (mRNA) and protein are upregulated in the rat hippocampus in response to seizure-induced synaptic reorganization (Fischer et al., 1995; Popa-Wagner et al., 1997).
Microtubule-associated protein 2 is abundant in the dendrites of neurons in the mature rat brain (Garner and Matus, 1988; Garner et al., 1988). Previous studies document that cerebral ischemia disrupts MAP2 expression in the hippocampus and cortex of experimental animals. For example, MAP2 immunoreactivity is reduced in the stratum radiatum of the ipsilateral hippocampus and ipsilateral cortex of neonatal (P7) rats a few hours after cerebral ischemia, reaching a minimum at 48 hours after ischemia (Malinak and Silverstein, 1996). Likewise, decreases in MAP2 immunoreactivity were detected as early as 1 hour after permanent occlusion of the MCA in rats (Dawson and Hallenbeck, 1996). In mature rats, the greatest loss of MAP2 immunostaining is in the CA1 region at 72 hours after trauma (Inuzuka et al., 1990). In Mongolian gerbils, the decline in MAP2 immunoreactivity in the CA1 region of the hippocampus and cortex occurs as early as 6 to 12 hours after a 5-minute occlusion of the common carotid arteries (Yoshimi et al., 1991; Matesic et al., 1994; Matesic and Lin, 1994) and persists even at 12 weeks after trauma (Kudo et al., 1992).
Although the immunoreactivities for MAP1B and MAP2 have been shown to be upregulated in hippocampal and cortical structures after traumatic injury to the brain (Kwak and Matus, 1988; Book et al., 1996), the participation of these proteins in the regenerative phase after cerebral ischemia has not been studied. Furthermore, although the incidence of ischemic stroke increases dramatically with advancing age (Palomaeki et al., 1993; Polak et al., 1993; Giovannoni and Fritz, 1993), few studies have been conducted on aged animals (Futrell et al., 1991; Millikan, 1992; Wang et al., 1995; Davis et al., 1995; Sutherland et al., 1996). We hypothesize that older rats would manifest a reduced capacity for neuronal regrowth after stroke compared with younger rats. To this end, we studied the cerebral expression of MAP1B and MAP2 during the regenerative phase after reversible occlusion of the MCA in young and aged rats. Preliminary data show that, of the time points examined, the expression of neuronal plasticity-associated MAP1B protein is first evident at 7 days after occlusion.
MATERIALS AND METHODS
The experiments reported in this study were conducted in accordance with the statement regarding the care and use of animals (Gärtner, 1991) and were approved by a federal animal care committee.
Eighteen hours before surgery, male Sprague-Dawley rats (n = 12 for each age group) were deprived of food to minimize variability in ischemic damage that can result from varying plasma glucose levels (Ginsberg and Busto, 1989); water remained available at all times. Surgery was performed between 8 AM and 1 PM.
Reversible occlusion of the middle cerebral artery
Blood flow through the MCA was temporarily interrupted using a modification of the method originally described by Brint and colleagues (1988), Dirnagl and associates (1989), and Zhang and Iadecola (1992). Throughout surgery, anesthesia was maintained by spontaneous inhalation of 1% to 1.5% halothane, in a mixture of 75% nitrous oxide and 25% oxygen, through a specially designed mask. Body temperature was controlled at 37 ± 0.2°C by a Homeothermic Blanket System (Harvard Apparatus, Edenbridge, Kent, U.K.). The anesthetized animals were immobilized in a supine position, and the tail artery was catheterized with PP50 tubing (internal diameter = 0.58 mm). This catheter enabled the continuous measurement of blood pressure and the withdrawal of blood samples for determination of pH and blood gasses (Blutgassystem IL 1620, Instrumentation Laboratory, Munich, Germany), as well as arterial glucose levels (Omnican7 Balance, B. Braun, Melsungen, Germany). Under a surgical microscope, the left and right common carotid arteries each were loosely encircled with a silicon thread to enable the temporary closure of these vessels (see later).
The animals were then placed in a prone position with the right side of the head exposed. The lateral skull surface was exposed and, using a microdrill (Fine Science Tools Heidelberg, Germany), a small segment of the skull above the MCA was removed 2 to 3 mm rostral to the juncture of the zygomatic arch and the pars squamosa of the temporal bone. To minimize local friction-induced warming, the bone was thinned using the drill at low speed and the site was superfused with physiologic saline. The bone flap was carefully removed with forceps and the underlying dura opened with a fine needle. The MCA then was slowly lifted with a tungsten hook attached to a micromanipulator (Maerzhaeuser Precision Micro-Manipulator Systems, Fine Science Tools) until blood flow through the artery was completely interrupted. Blood flow through the common carotid arteries then was stopped by tightening the prepositioned thread loops (see earlier). The surgical field was kept warm and moist with artificial cerebrospinal fluid (Liquicheck, Spinal Fluid Control, BioRad; Munich, Germany) for the duration of the procedure.
Three hours later, the tungsten hook was lowered and removed from the MCA and the common carotid arteries were reopened, allowing full reperfusion of the brain. The surgical wounds were sutured, the catheter withdrawn from the tail artery, and the animals returned to their cages. After a 7-day survival time, the rats were deeply anesthetized with 3% halothane in 75% nitrous oxide/25% oxygen and perfused with buffered saline followed by buffered, 4% freshly depolymerized paraformaldehyde. The brains were removed, postfixed in 4% buffered paraformaldehyde for 24 hours, cryoprotected in 20% sucrose prepared in 10 mmol/L phosphate-buffered saline (PBS), flash-frozen in isopentane, and stored at −70°C until sectioning.
Behavioral analysis
Twenty-four hours after surgery, the clinical status of the animals was assessed using the Bederson test (1986). At this time point, all animals that had undergone cerebral ischemia for 3 hours showed obvious neurologic deficits, including paresis of the contralateral extremities, particularly the hind legs, and rotation when lifted above the cage floor (grade 3 in the Bederson test). Control animals showed no neurologic deficits (grade 0).
Determination of the infarct volume
The area and partial volume of the infarct and ipsilateral cerebral hemisphere were determined in every 20th section. An integration of the resulting partial volumes gave the total volume of the ipsilateral hemisphere, along with the volume of the cortical infarct; infarct volume then was expressed as percent of the total hemispheric volume.
Histology
The brains were coronally sections at 25 μm on a freezing microtome, the sections were collected in 4% paraformaldehyde in 100 mmol/L PBS (pH 7.2), postfixed for 30 minutes, embedded in polyethylene glycol, and stored at −70°C until use. Every 20th section was stained using acid-vanadium fuchsin (Victorov et al., 1993).
Preparation of cRNA probes
Microtubule-associated protein 1B and microtubule-associated protein 2. Rat cDNA clones were kindly provided by Dr. C.C. Garner (Zentrum für Molekulare Neurobiologie, Hamburg) and subcloned into a pBluesscript [pBSSK(I)+] vector. These plasmids allowed the synthesis of antisense and sense RNA probes using digoxygenin-11-UTP. Finally, the RNA probes were purified by gel filtration.
Nonradioactive in situ hybridization
A 1.5-kb digoxigenin-11-UTP-labeled antisense RNA probe was synthesized using a kit supplied by Boehringer Mannheim (Mannheim, Germany) according to the manufacturer's specifications. RNA quality and quantity were evaluated by detection of the digoxigenin-labeled cRNA with anti-digoxigenin alkaline phosphatase, Fab fragment (Boehringer Mannheim) conjugate, followed by color development using the nitro-blue tetrazolium and 5-bromo-4-chloro-3-indolyl-phosphate system. A digoxigenin-labeled neurofilament-68 cRNA probe of known size and concentration also was used as a reference probe. Finally, the riboprobe was hydrolyzed to an average size of 300 base pairs by incubation in 1 mol/L NaHCO3/Na2CO3 buffer for 60 minutes at 60°C.
Sections (60 μm) were cut on a freezing microtome and processed for in situ hybridization as free-floating material. All steps were conducted in glass-covered Petri dishes with a diameter of 30 mm. Prehybridization was performed in a buffer containing 50% formamide, 5× SSC, 1% blocking reagent, and 500 μg/mL tRNA. Sections were hybridized in the same buffer containing 50 ng/mL cRNA probe for 3 hours at 65°C. After hybridization, the sections were washed 5 times in 2× SSC for 5 minutes each at room temperature and 10 times at 68°C in 0.1× SSC for 50 minutes. After high-stringency washings, the sections were postfixed in 4% paraformaldehyde in PBS for 30 minutes at room temperature. After posthybridization washes, slides were incubated for 2 hours at room temperature in a blocking buffer consisting of 0.1 mol/L maleate (pH 7.5), 150 mmol/L NaCl, 0.1% Tween 20, 0.2% Triton-X 100, and 1% blocking reagent (Boehringer Mannheim). Alkaline phosphatase-conjugated sheep anti-digoxigenin-Fab fragments, diluted 1:1000 in blocking buffer, then were applied. After 15 hours of incubation at 4°C in a humidity chamber, unbound conjugate was removed by three 15-minute washes in blocking buffer followed by one wash in alkaline phosphatase buffer (100 mmol/L Tris, pH 9.5; 100 mmol/L NaCl; 10 mmol/L MgCl2) for 10 minutes. For the detection of signal, slides were incubated at 37°C in darkness for 3 to 5 hours in a chromogen solution consisting of 330 μg/mL nitro-blue tetrazolium, 150 μg/mL of 5-bromo-4-chloro-3-indolyl-phosphate, and 250 μg/mL levamisole in alkaline phosphatase buffer. The reaction was stopped with 10 mmol/L Tris, pH 8.0, plus 1 mmol/L ethyl-enediamine tetra-acetic acid. Finally, the sections were mounted on slides and air dried, and coverslips were applied using a xylol-based mounting medium.
Controls. The specificity of the rat MAP1B and MAP2 antisense RNA probes was assessed by Northern blot analysis. Selected tissue sections also were hybridized with a hydrolyzed sense probe.
Immunocytochemistry
Free-floating sections were first treated with 0.3% hydrogen peroxide in PBS to inactivate endogenous peroxidase and then processed for immunocytochemical study. After blocking in 3% donkey serum/10 mmol/L PBS/0.3% Tween 20, sections were incubated overnight at 4°C with monoclonal mouse antibodies recognizing either an epitope on the MAP1B (clone 2 Boehringer Mannheim), or the MAP2-like proteins (clone AP20, Boehringer Mannheim) diluted 1:800 in PBS containing 3% normal donkey serum and 0.3% Tween. The primary antibody was detected using the ABC system (Vectastain Elite Kit, Vector, Burlingame, CA, U.S.A.). After extensive washing in PBS containing 0.3% Tween, sections were incubated for 1 hour at room temperature with biotinylated donkey anti-mouse IgG (Jackson ImmunoResearch Laboratories, West Grove, PA, U.S.A.) diluted 1:400 in PBS containing 1% normal donkey serum and 0.3% Tween 20, followed by overnight incubation at 4°C; after washing in PBS, sections were incubated for 2 hours at room temperature in ABC elite reagent diluted 1:100 in PBS containing 0.3% Tween. The antibody complex then was visualized with 0.025% 3′,3′ diaminobenzidine and 0.005% hydrogen peroxide in 100 mmol/L Tris buffer (pH 7.5) for 5 to 10 minutes. Finally, the sections were mounted onto slides and air dried, and coverslips were applied using a xylene-based mounting medium. The specificity of the antibodies was verified on Western blots and by omission of the primary antibodies in immunocytochemical experiments.
Semiquantitative analysis of tissue sections
Integrated optical densities were collected from the border zone adjacent to the infarct at three different levels for each rat using the National Institutes of Health software PC Image (Bethesda MD, U.S.A.). Statistical analysis was done using Student's t test for comparisons of means between the contralateral and ipsilateral sides using the SPSS software (SPSS Inc., Chicago, IL, U.S.A.). Finally, the changes in MAP1B and MAP2 mRNA and protein expression in the lesioned hemisphere were expressed as a percent of the contralateral levels for each age group, and the age differences were compared statistically using the t test.
RESULTS
In the first 24 hours after surgery, the animals were listless, probably partly because of the after-effects of the anesthesia. This condition improved in the ensuing days, although the animals tended to remain sensitive to tactile stimulation and noise. There was no evident difference in the clinical status of young and aged rats during the 7-day postsurgical survival time. Two young rats and three older rats died in the week after the infarct. Although the blood parameters showed some age-associated variation, notably blood pressure, the differences were not statistically significant (data not shown). Likewise, there was no significant difference in the volume of cortical infarcts between young and aged rats (42.7 ± 11.4% for the young rats and 43.9 ± 11.3% for the aged rats).
Microtubule-associated protein 1B expression in noninfarcted tissue of young and aged rats
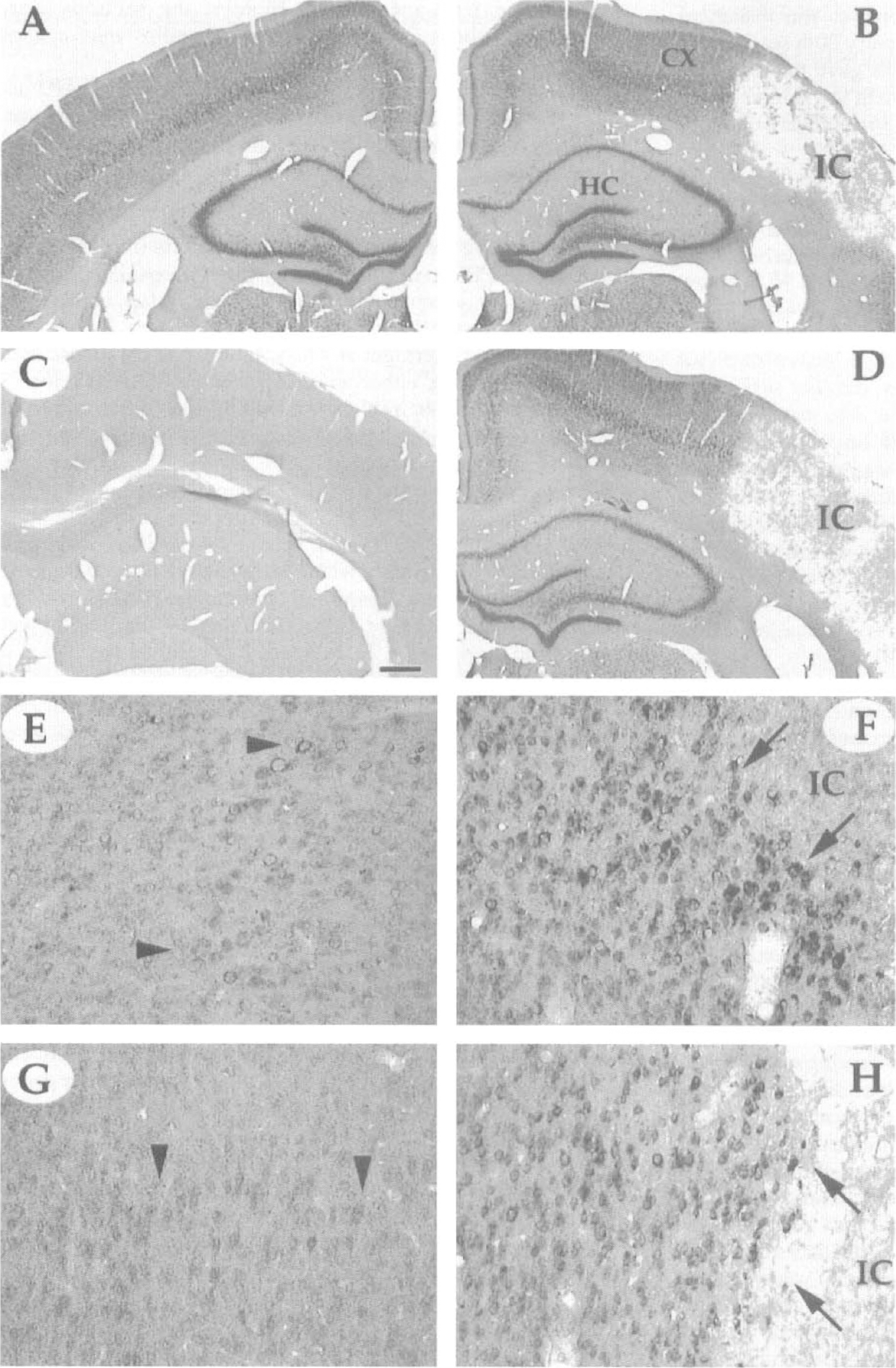
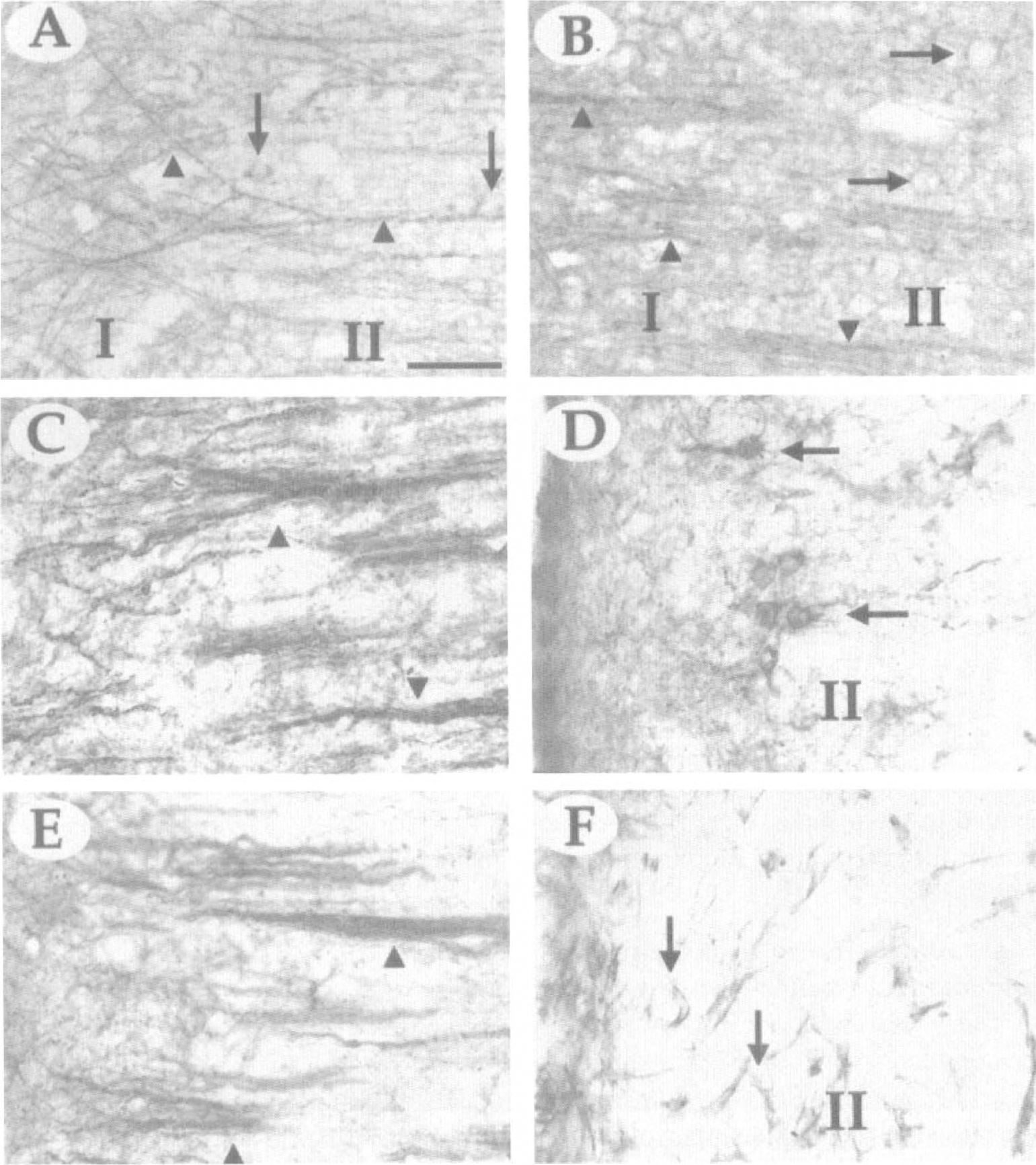
The levels and regional distribution of the MAP1B mRNA in the hippocampus and cortex are shown in Fig. 1. At 20 months of age, there was diminished expression of MAP1B mRNA in the hippocampus, but not cortex, of both ischemic and sham-operated control rats relative to levels in young animals (Fig. 1D versus Fig. 1B, respectively). The morphologic features of MAP1B immunostaining in the hippocampus and cortex of 3-month-old and 20-month-old rats are shown in Fig. 2. Generally, there was an age-dependent decline in MAP1B-like immunoreactivity in the hippocampus but not in cortex (not shown). However, MAP1B immunostaining in layers I and II (Fig. 2B) and layer V of the contralateral (noninfarcted) cortex of the 20-month-old rats showed age-associated structural changes, with the appearance of bundles of thick processes (Fig. 2A versus B) and neurons showing intense MAP1B immunoreactivity, most notably in the perinuclear cytoplasm (not shown).
In situ hybridization of microtubule-associated protein (MAP) 1B messenger ribonucleic acid (mRNA) shown 1 week after middle cerebral artery (MCA) occlusion. Notice that the infarct core is devoid of signal in both young
The MAP1B immunostaining of processes (arrowheads) and cell bodies (arrows) in the cortex at 1 week after MCA occlusion. Notice age-related changes in the morphologic features of MAP1B-immunopositive fibers of aged
Microtubule-associated protein 1B response to stroke in young and aged rats
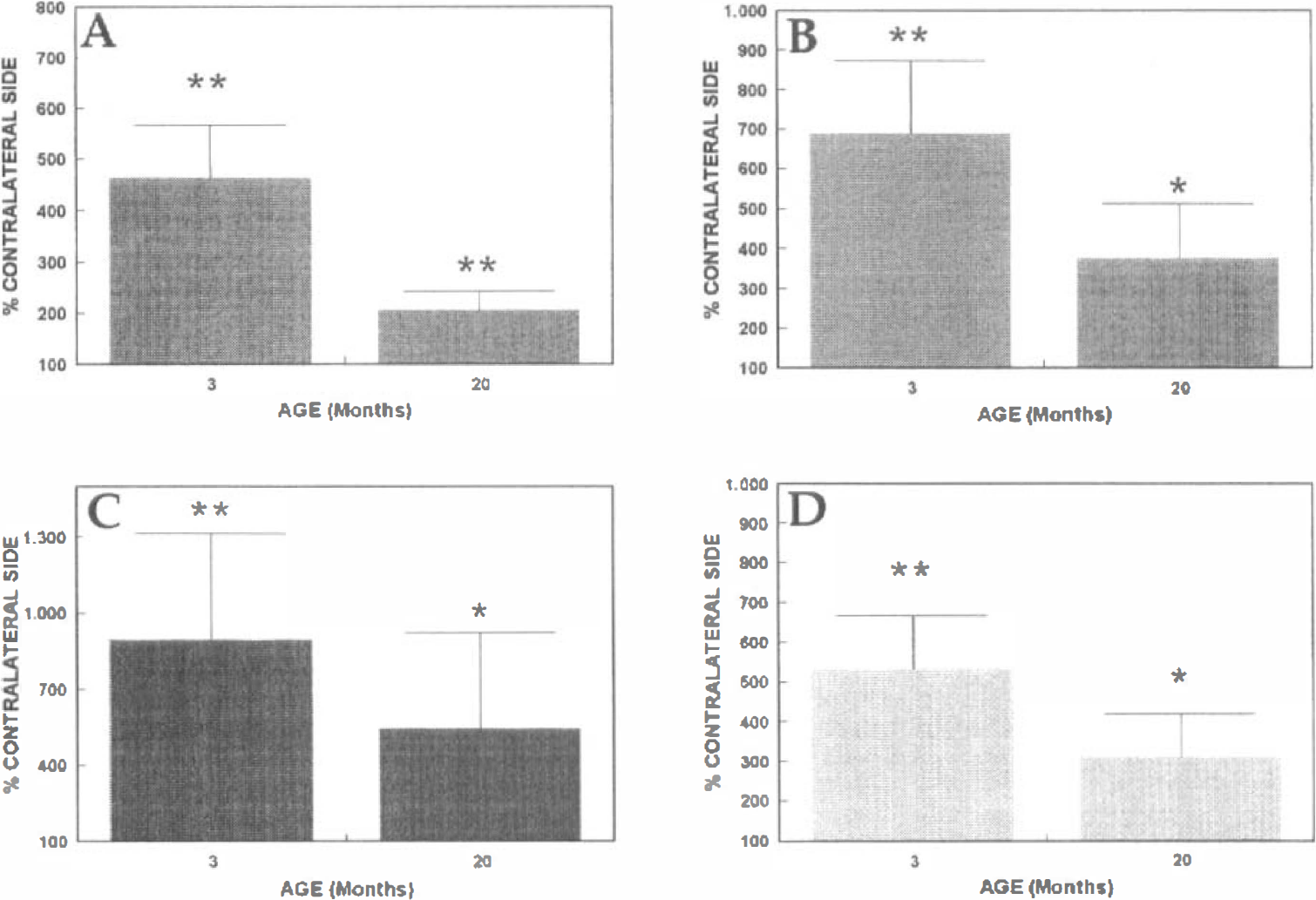
Microtubule-associated protein 1B messenger RNA. At 7 days after the cerebral infarct, MAP1B mRNA was well expressed in both the contralateral and ipsilateral hippocampus, as well as in the noninfarcted neocortex, of the 3-month-old rats (Fig. 1A and B); the infarct core, in contrast, was devoid of signal (Fig. 1B). In young rats, we noted intense mRNA staining of neurons (4.7-fold increase, P < 0.009, t test, compared with the corresponding noninfarcted region, Figs. 1E and 3A) in the area surrounding the infarct core (Fig. 1F), suggesting an increased synthesis of MAP1B mRNA by surviving neurons bordering the infarct core. However, in this same region of aged rats, the viable cells were less intensely labeled for MAP1B mRNA (Fig. 1H) (2.2-fold increase over the contralateral, noninfarcted side, P < 0.016, t test, Figs. 1G and 3A). The age difference between the MAP1B mRNA levels in the lesioned site also was significant, with the young rats having 2.1-fold higher MAP1B expression than the aged rats (P < 0.005, Fig. 3A).
Densitometric analysis of the levels of MAP1B and MAP2 in the hippocampus and cortex after cerebral ischemia. Integrated optical densities for MAP1B mRNA
Microtubule-associated protein 1B protein. Cerebral ischemia resulted in a substantial reduction of MAP1B immunostaining in the infarct core. However, MAP1B was strongly upregulated in neuronal processes along the interface between the infarct core and the healthy tissue of young rats (Fig. 2C) (6.8-fold increase, P < 0.002, Fig. 3B). There also was robust staining of neurons in the remnants of the former layer II of the cortex (Fig. 2D). Although the structure of MAP1B-immunoreactive fibers differed to some extent in noninfarcted tissue of young and aged animals, MAP1B immunostaining in the vicinity of the infarct core of the older rats was morphologically similar to that of the young rats (Fig. 2E versus C, respectively), albeit at lower intensity (54% of the integrated density shown by the young rats, P < 0.05, Fig. 3B). Major age-related differences also were observed in the former layer II of the cortex of older rats, where there was little immunostaining of neuronal somata (Fig. 2F).
Microtubule-associated protein 1B expression in noninfarcted tissue of young and aged rats
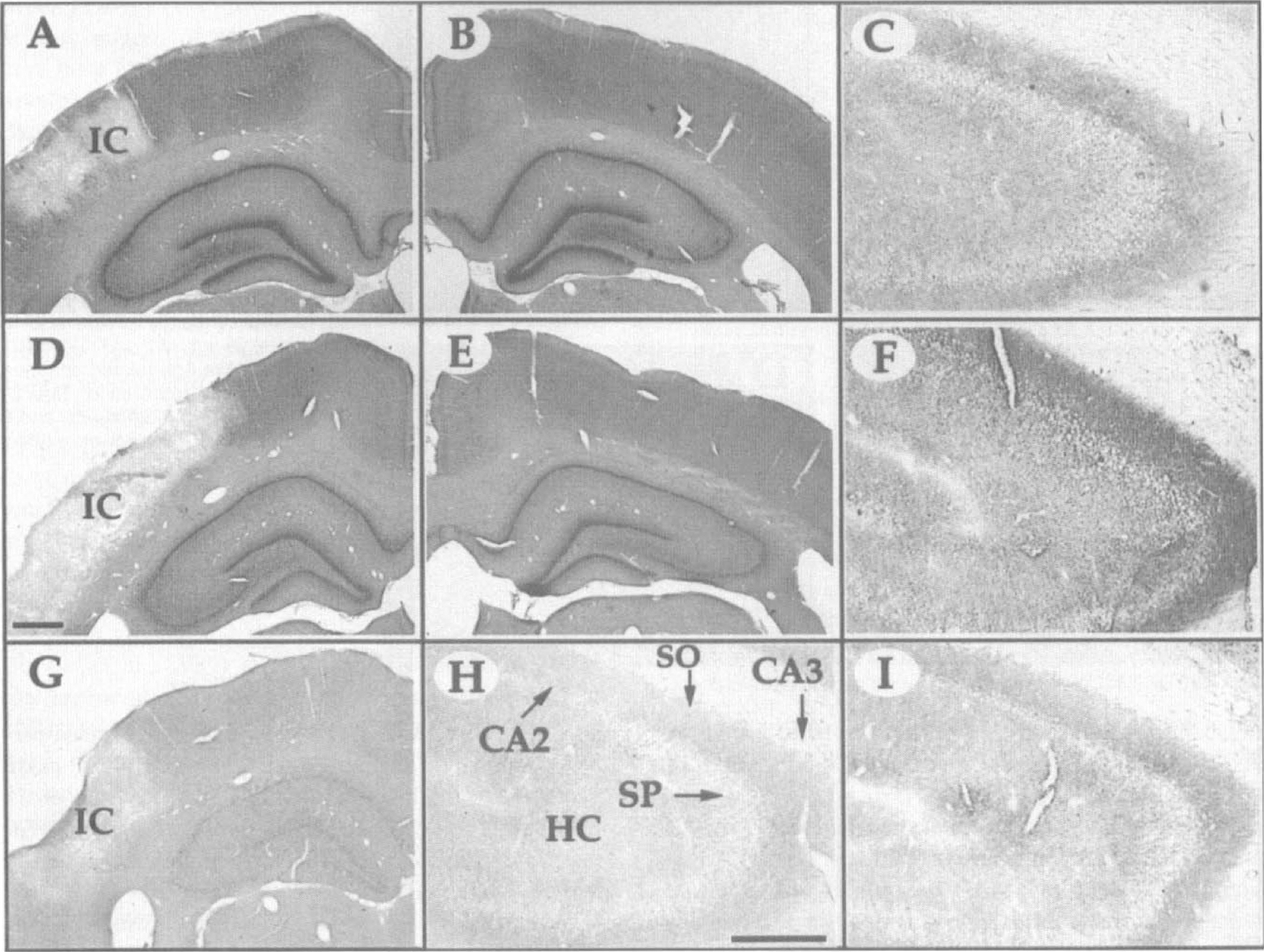
The levels of MAP2 mRNA were high in the hippocampus and layer II of the retrosplenial and motor cortex of young animals (Fig. 4B). The levels of MAP2 mRNA decreased in the hippocampus of 20-month-old (Fig. 4B versus E) compared with 3-month-old rats (1.9-fold reduction, P < 0.05). Lower levels of MAP2 transcripts were noted in layer V of the sensory cortex in both young and aged rats (not shown). With aging, immunostaining of dendrites with MAP2 in cortical layers I to III becomes diffuse (Fig. 5C) compared with the orderly staining in the young rats (Fig. 5A).
In situ hybridization of MAP2 mRNA and immunostaining of MAP2 in the hippocampal region. Generally, MAP2 mRNA levels and MAP2 immunoreactivity in the hippocampus decreased with age (
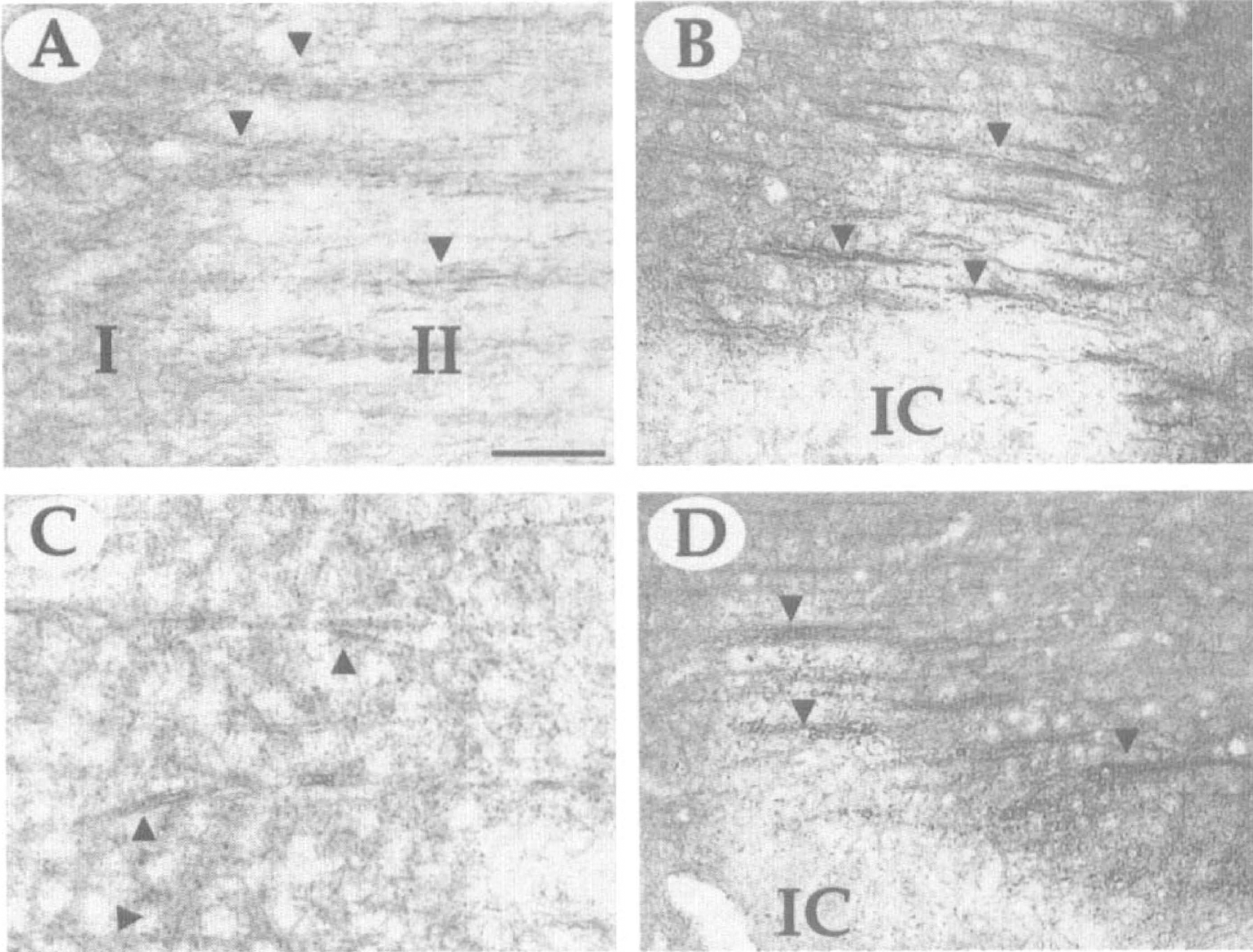
The MAP2 immunostaining of dendrites (arrowheads) shown 1 week after infarct around the infarct core of 3-month-old
Microtubule-associated protein 2 response to stroke in young and aged rats
Microtubule-associated protein 2 messenger RNA. The infarct did not alter significantly the levels of MAP2 mRNA expression in the ipsilateral hippocampus in either young or aged rats but resulted in a large decrease in the MAP2 mRNA levels in the infarct core at both ages. Neurons that were intensely stained for MAP2 mRNA were scattered around the infarct core of some of the 3-month-old but not the 20-month-old animals. However, the increase was not significant (not shown).
Microtubule-associated protein 2 protein. After a 3-hour occlusion, there was a large decrease in the immunostaining of MAP2 in the infarct core of both young and old rats (Fig. 5B and D). However, the young rats showed vigorous immunostaining of dendrites around the infarct core, particularly in layers I to III of cortex (9.2-fold higher than in the contralateral side, P < 0.01, Fig. 3C). The MAP2 immunoreactivity near the infarct of the 20-month-old rats also showed robust staining, much like that of young rats, but the increase over staining on the contralateral side was less (Fig. 5D) (4.5-fold increase, P < 0.05, Fig. 3C). The young rats had significantly greater MAP2 staining around the infarct core than did the aged rats (2.7-fold difference, P < 0.05).
Interestingly, stroke-related changes in MAP2 immunoreactivity also were noted in the hippocampus. One week after the occlusion, there was strong immunolabeling in the stratum oriens of the CA3 region of the infarcted hemisphere in the 3-month-old rats (Fig. 4C versus F) (5.4-fold increase, P < 0.001, Fig. 3D). Hippocampal MAP2 immunostaining also was increased in aged animals, but to a lesser degree (Fig. 4H versus I) (2.3-fold increase, P < 0.05, Fig. 3D). However, across the age groups, there was no significant difference between the hippocampal MAP2 levels.
Except for the age-related decline in the amount of MAP2 mRNA, as seen on the contralateral side (Fig. 4B versus E) in the hippocampus, changes in the levels of MAP2 mRNA on the ipsilateral side did not parallel the changes in the protein levels after stroke.
DISCUSSION
The results of this study show that the levels of two structural proteins, MAP1B and MAP2, are elevated in the infarct border zone of 3-month-old and 20-month-old rats at 1 week after ischemic damage to the cortex. However, the levels of MAP2 mRNA did not precisely parallel the increase in protein immunoreactivity; that is, in the older rats, the MAP2 mRNA around the lesion site was much lower than expected based on the amount of MAP2 immunoreactivity in the same region. The apparent dissociation between mRNA levels and protein levels also was evident for MAP2 mRNA and protein in the hippocampus at both ages. We attribute this phenomenon, among other factors, to a resolution problem: MAP1B protein is localized both in neurons and their processes, whereas MAP2 along with its mRNA have been reported to be localized mainly in dendrites (Garner and Matus, 1988; Garner et al., 1988; Steward et al., 1996). Our in situ hybridization technique on thick, floating sections did not permit the discrete localization of MAP2 mRNA in dendrites (i.e., in the neuropil). The limited resolution in thick sections also prevented us from determining if the more intense immunostaining in the vicinity of the infarct core resulted from an increased number of fibers or greater immunostaining within the processes. The amount of protein depends on a number of factors besides mRNA levels, such as protein turnover rate. For example, decreases in the turnover rate of MAP1B and MAP2 may lead to accumulation of protein and hence to similar immunoreactivity around the infarct core of both age groups, despite low levels of mRNA. A similar proposal has been made for MAP2, T-alpha-1 tubulin, and F1/growth-associated protein-43 after denervation of the dentate gyrus of adult rats (Stewart, 1995).
The MAP1B and MAP2 mRNA and protein levels were lower in the brains of older control rats than in young rats, especially in the hippocampus, which is in agreement with previous results (Popa-Wagner et al., 1992). The diminution of MAP is illustrated by the age-dependent decline in the intensity of the MAP1B immunostaining of fibers and cell bodies and of MAP2 immunolabeling of the dendrites in layers I to III of the cortex and the CA2 region of the contralateral hippocampus of the older group.
What could be the significance of increased MAP1B and MAP2 expression in the rat brain after stroke? A vast literature indicates that MAP1B expression is associated with growth and plasticity phenomena in the nervous system. For instance, neonatal rat brains have 10 times the amount of MAP1B found in adults (Riederer et al., 1985), and MAP1B increases dramatically in PC12 cells that have been induced to extend neurites. Moreover, attenuation of MAP1B mRNA expression by antisense oligodeoxynucleotides inhibits the initiation of neurite outgrowth in nerve growth factor-treated PC12 cells (Brugg et al., 1993). In vitro and in vivo experiments indicate that MAP1B is present almost exclusively in the growth cone of developing neurons (Gordon-Weeks, 1993; Black et al., 1994). The MAP1B expression also correlates with regenerative processes. For example, MAP1B is abundant in growing axons of neurons that have been transplanted into the adult rat hippocampus (Brook et al. 1993), as well as in axons, perikarya, and dendrites in explant cultures of mouse retinal segments at embryonic day 15 and in adults (Bates et al., 1993). Likewise, increased immunostaining of a phosphorylated isoform of MAP1B in cat neuronal somata and axons after peripheral and central lesions of the trochlear nerve has been reported (Book et al., 1996). In mature rodents, however, MAP1B normally is expressed in some brain areas. For instance, MAP1B is abundant in axons of the olfactory nerve and dendrites of mitral cells, the areas most involved in adult olfactory plasticity (Schoenfeld et al., 1989; Viereck et al., 1989). Furthermore, mice carrying a mutation in the Map1B gene exhibit an ataxic gait and spastic tremor of the hind limbs (Edelman et al., 1996), suggesting that MAP1B expression is required for the proper functioning of certain regions of the central nervous system in adult rodents.
Several studies have concentrated on the diminution of MAP2 immunostaining in dendrites after reduction of energy supply to the brain (Onizuka et al., 1996). However, a previous study has documented the re-expression of a plasticity-associated variant, MAP2c, in the CA1 region of the hippocampus and cortical regions after brief (10-minute) cardiac arrest (Saito et al., 1995). Generally, it appears that MAP2 immunoreactivity is increased at sites of brain injury. Peak MAP2 expression is highly dependent on the experimental conditions and ranges from hours (Saito et al., 1994; Dawson and Hallenbeck, 1996) to 1 week (Kwak and Matus, 1988). This time frame is in accordance with our observations; we found vigorous expression of MAP2 at 1 week after occlusion of the MCA. However, there was no apparent increase in the number of dendritic processes, suggesting rather that the increased immunostaining resulted from higher levels of protein within the existing processes. Given the strong immunolabeling of MAP1B and MAP2 fibers in the infarct penumbra after stroke, it could be inferred that MAP1B and MAP2 expression is associated with attempts by viable neurons to extend new processes. Interestingly, at 20 months of age, the regenerative potential of central neurons, as judged from the expression of MAP1B and MAP2, is not exhausted. However, the expression of the proteins retains the adult pattern, in which MAP2 is the prevailing form, rather than the embryonic pattern of MAP1B predominance that would have been expected if regeneration simply recapitulated developmental events (Tucker, 1990; Garner et al., 1990; Bates et al., 1993). A similar finding has been reported for MAP1B and MAP2 expression after injury of the sciatic nerve (Fawcett et al., 1994).
The phenomenon of plasticity in the brain of aged rats is well documented. Loss of synaptic connections as a consequence of age-related changes in the brain, such as loss of afferent supply and alterations in transmitters, receptors, and trophic factors, is followed by a compensatory regrowth of neuronal connections by surviving neurons to replace defective or lost ones (Coleman et al., 1990). Thus, it seems that the aged rodent brain retains the capacity, albeit at a slower rate, to maintain and repair its own circuitry (Geinisman et al., 1992; Hoff et al., 1982a, 1982b). Even in the absence of overt changes such as those occurring after stroke, there is continuous remodeling of the cerebral circuitry, as reflected in the persistent growth and regression of neuronal processes (Rafols et al., 1989). The available evidence suggests that regeneration in the nervous system takes longer in older individuals than in younger ones (Vaughan et al., 1992; Campbell and Pomeranz, 1993). This is especially evident in old rats, where the levels of growth-associated protein-43 and neurofilament-68 mRNA are much reduced in hilar and CA3 pyramidal neurons at various time points after the deafferentation of the hippocampus (Schauwecker et al., 1995). Likewise, old rats show reduced capacity to upregulate the structural protein β-tubulin in neurons of the locus ceruleus after a neurotoxic lesion (Unnerstall and Long, 1996). At the morphologic level, similar findings have been reported for regenerative processes in the molecular layer of the dentate gyrus of aged rats after deafferentation (Hoff et al., 1982a, 1982b). Taken together, these results suggest that the neurons of rats aged 20 months still have the ability to synthesize some of the components needed for structural repair and maintenance, whereas rats aged 28 months or more have lost most of this potential. Future studies of other molecules required for growth and repair processes are needed to evaluate the extent of functional recovery after cerebral ischemia in the aged rat.
