Abstract
The reduction of the apparent diffusion coefficient (ADC) of brain tissue water in acute cerebral ischemia, as measured by diffusion-weighted magnetic resonance imaging, is generally associated with the development of cytotoxic edema. However, the underlying mechanism is still unknown. Our aim was to elucidate diffusion changes in the intracellular environment in cytotoxic edematous tissue. The ADC of intracellular metabolites was measured by use of diffusion-weighted 1H-magnetic resonance spectroscopy after (1) unilateral N-methyl-D-aspartate (NMDA) injection and (2) cardiac arrest-induced global ischemia in neonatal rat brain. The distinct water ADC drop early after global ischemia was accompanied by a significant reduction of the ADC of all measured metabolites (P < 0.01, n = 8). In the first hours after excitotoxic injury, the ADC of water and the metabolites taurine and N-acetylaspartate dropped significantly (P < 0.05, n = 8). At 24 and 72 hours after NMDA injection brain metabolite levels were diminished and metabolite ADC approached contralateral values. Administration of the NMDA-antagonist MK-801 1.5 hours after NMDA injection completely normalized the water ADC but not the metabolite ADC after 1 to 2 hours (n = 8). No damage was detected 72 hours later and, water and metabolite ADC had normal values (n = 8). The contribution of brain temperature changes (calculated from the chemical shift between the water and N-acetylaspartate signals) and tissue deoxygenation to ischemia-induced intracellular ADC changes was minor. These data lend support to previous suggestions that the ischemia-induced brain water ADC drop may partly be caused by reduced diffusional displacement of intracellular water, possibly involving early alterations in intracellular tortuosity, cytoplasmic streaming, or intracellular molecular interactions.
Keywords
In recent years, diffusion-weighted (DW) magnetic resonance imaging (MRI) has become an important tool in the assessment of stroke (see Hossmann and Hoehn-Berlage, 1995). Reduction of the apparent diffusion co-efficient (ADC) of brain tissue water is already detected in the very early stages of cerebral ischemia (Davis et al., 1994) and could potentially provide prognostic information on final tissue outcome (Dijkhuizen et al., 1998). These water diffusion changes are generally associated with the development of cytotoxic edema (i.e., cellular swelling) resulting from a rapid loss of ion homeostasis (Moseley et al., 1990; Verheul et al., 1994). The exact biophysical mechanisms responsible for the ADC drop, however, are still unknown. Correlations have been made between the water ADC decline and (1) a decreased ratio of the extracellular versus intracellular volume (Van der Toorn et al., 1996b), (2) a brain temperature decline (Hasegawa et al., 1994), (3) membrane permeability reductions (Helpern et al., 1992), and (4) an increased tortuosity of the extracellular space (Van der Toorn et al., 1996b).
Recently, Szafer et al. (1995) proposed a theoretical model for water diffusion in tissues. They postulated that the ischemia-induced tissue water ADC reduction is the combined result of an altered cellular volume fraction and changes in the intrinsic extracellular and intracellular diffusion. It was calculated that a significant decrease of the intracellular water diffusion is necessary to explain the large overall water ADC drop. Only a few studies have specifically addressed intracellular diffusion during ischemia (Wick et al., 1995; Neil et al., 1996; Van der Toorn et al., 1996a). Present DW MRI methodology does not allow a straightforward differentiation between extracellular and intracellular water diffusion (Niendorf et al., 1996). However, information on diffusion in the intracellular space in intact organs can be obtained from DW magnetic resonance spectroscopy (MRS) (for review see Nicolay et al., 1995) because the (water-suppressed) MRS signal is dominated by intracellular metabolites. By means of DW 1H-MRS it has been demonstrated that cerebral ischemia in fact results in a reduction of the apparent diffusion coefficient of intracellular metabolites in the early stages (Wick et al., 1995; Van der Toorn et al., 1996a). Several explanations for this remarkable finding have been proposed, including alterations in cytoplasmic viscosity and streaming. It remains unclear, however, what mechanisms are involved in ischemia-induced intracellular diffusion changes and to what extent an anticipated reduction of intracellular water diffusion would contribute to the large overall tissue water ADC drop.
The aims of this study were (1) to compare changes in intracellular metabolite diffusion between ischemic and nonischemic conditions, both of which are accompanied by cytotoxic edema and a water ADC drop, and (2) to assess intracellular diffusion in relation to recovery of the tissue water ADC on normalization of cellular volume. To that end, we measured the ADC of brain metabolites and water during reversible excitotoxic injury in neonatal rat brain and compared the results with measurements after cardiac arrest-induced global ischemia in neonatal rats. Neonatal rat brain is highly vulnerable to excitotoxicity, and intracerebral injection of excitotoxins results in large brain lesions (Ikonomidou et al., 1989). Intracerebral injection of the glutamate-agonist N-methyl-D-aspartate (NMDA) in neonatal rats is rapidly followed by dramatic cell swelling and a significant reduction of the brain water ADC (Verheul et al., 1994). These alterations can be readily reversed by timely administration of the noncompetitive NMDA-antagonist MK-801 (dizocilpine maleate). Importantly, although this type of brain injury bears strong similarities to ischemic brain damage, it is not accompanied by a cerebral blood flow decline (Taylor et al., 1995). These aspects make the neonatal model of NMDA-induced excitotoxicity ideally suited for the purpose of this study.
METHODS
Animal preparation
Excitotoxic injury was induced in neonatal Wistar rats (7 to 8 days old) by intrastriatal injection of NMDA (McDonald and Johnston, 1988). Rats were anesthetized with ether and placed in a form-fitting plaster mold in a stereotaxic frame. The skull was exposed and a hole was drilled at 2.5 mm left from bregma. A 5-μL Hamilton syringe was slowly lowered into the left striatum to a depth of 4.0 mm. Infusion of NMDA (20 nmol in 0.5 μL 50 mmol/L Tris-HCl buffer, pH 7.4) was performed at a rate of 0.25 μL/min by use of a microdrive. Control animals received 0.5 μL 50 mmol/L Tris-HCl buffer (pH 7.4). In one group of animals a catheter was placed subcutaneously for remote administration of the noncompetitive NMDA-antagonist MK-801 (1 mg/kg body weight administered in 0.05 mL) 1.5 hours after NMDA injection.
Global ischemia was elicited in neonatal Wistar rats (7 to 8 days old) by KCl-induced cardiac arrest. To that end, we placed an intraperitoneal catheter in ether-anesthetized animals to enable remote injection of 0.5 mL 3 mol/L KCl.
During the MR protocol (see below) animals were immobilized in a foam-rubber holder to minimize motion and anesthesia was maintained with 0.4% to 0.8% halothane in O2 and N2O (1:1) delivered through a face mask. Body temperature was maintained by means of a water-heated pad. Respiratory frequency was monitored by use of a small air-filled balloon connected to a pressure transducer, which was placed under the animal's chest.
MR experiments
Magnetic resonance experiments were performed on a 4.7-T Varian/SISCO spectrometer with a 220-mT/m gradient insert. Signal excitation and detection were accomplished by means of a Helmholtz volume coil (9 cm diameter) and a surface coil (2 cm diameter), respectively.
First, multislice DW MRI (spin echo sequence; repetition time 2 s; echo time 33 ms; 2 acquisitions; 2 or 3 b values [170 to 1,800 s/mm2]; 64 × 64 data matrix; field-of-view 3 × 3 cm2; eight contiguous 1.7-mm slices) was performed to enable accurate positioning of the volumes-of-interest for DW MRS.
Diffusion-weighted 1H-MRS was performed essentially as described by Van der Toorn et al. (1996a), except for some minor modifications. We used a PRESS sequence (repetition time 3 s; echo time 144 ms; 96 acquisitions; 5 b values [258 to 4094 s/mm2]; sine-shaped magnetic field gradients in three orthogonal directions simultaneously; δ 8 ms; Δ 26 ms; where b, δ, and Δ are the diffusion-weighting factor, the duration of the gradient pulses, and the time interval between the diffusion-sensitizing gradient pulses, respectively) in combination with CHESS water suppression. The diffusion time tdiff [(2/π)2 × (Δ − δ/4)] was 19.2 ms. Total acquisition time was 24 minutes. Sampling of the data was randomized by collecting DW MR spectra at each b value in three separate blocks of 32 acquisitions. The tissue water ADC was determined from DW MRS measurements without water suppression (four acquisitions; 5 b values, 316 to 2303 s/mm2, 1-minute acquisition time). For the global ischemia experiments we took a single 98-μL voxel which encompassed a major part of the brain. In the experiments that involved unilateral intracerebral NMDA- (or vehicle-) injection, a 32-μL voxel was positioned in the ipsilateral and in the contralateral hemisphere. In these cases, DW 1H-MRS was performed with 192 acquisitions (total experimental time 48 minutes) and data collection was divided in six separate blocks of 32 acquisitions. Experiments were done first on the ipsilateral and then on the contralateral side.
Experimental groups
NMDA-induced excitotoxicity. Three groups of animals were injected with NMDA only. Diffusion-weighted MRS experiments were done in the first group directly (i.e., between 0.5 and 3 hours) (n = 8), in the second group 24 hours (n = 8), and in the third group 72 hours (n = 8) after NMDA injection. In control neonates DW MRS was done 0.5 to 3 hours after Tris-HCl injection (n = 6).
MK-801 treatment. Two separate groups of animals were treated with MK-801, 1.5 hours after NMDA-injection. In the first group, animals underwent DW MRS of the ipsilateral voxel before and approximately 1.5 hours after MK-801 administration (n = 8); DW MRS of the contralateral voxel was performed in between these measurements (i.e., starting just after the MK-801 injection). Finally, in the second group DW MRS was done 72 hours after the administration of MK-801 (n = 8).
Cardiac arrest-induced global ischemia. First, DW MRS of water was done repeatedly starting 10 minutes before until 60 minutes after intraperitoneal KCl injection in neonatal rats (n = 6) to determine the dynamics of the global ischemia-induced tissue water ADC reduction.
Diffusion-weighted MRS with and without water suppression was performed before and after induction of global ischemia (from approximately 0.5 to 1 hour after intraperitoneal KCl injection; n = 8).
Data analysis
Water-suppressed MRS data were processed with 3-Hz line broadening. The areas of the resonances of choline-containing compounds (Cho, at 3.2 ppm), creatine/phosphocreatine (tCr, at 3.0 ppm), N-acetylaspartate (NAA, at 2.0 ppm), the inverted resonance of lactate (Lac, at 1.3 ppm), and the two resonances of taurine (Tau, fitted as two resonances at 3.27 and 3.44 ppm) were quantified with the VARPRO (variable projection method) nonlinear least-square time-domain fitting method. The area of the large water peak derived from the DW MRS experiments without water suppression was determined by simple integration. The areas were monoexponentially fitted to ln(S(b)/S0) = −b × ADC, where S(b) is the signal intensity at a certain b value, and S0 is the signal intensity at b = 0, to obtain ADC values. Fits with r < 0.9 were discarded.
The temperature in the brain was calculated from the chemical shift difference between the signals of NAA and the residual water, using the equation T (in °C) = 286.9 − 94.0 × (δwater − δNAA), where δwater and δNAA are the chemical shifts (in ppm) of water and NAA, respectively (Cady et al., 1995). This relationship, which was determined in newborn piglet brain, only holds for a certain protein concentration (Corbett et al., 1995). Because brain protein content in rats varies significantly during development, we used the above relationship only to estimate intraanimal temperature changes.
Statistical analysis
Values are presented as mean ± SD. Data were statistically evaluated by analysis of variance with post hoc Scheffé F test, and paired or unpaired Student's t tests where appropriate. A P value less than 0.05 was considered significant.
RESULTS
Global ischemia in neonatal brain
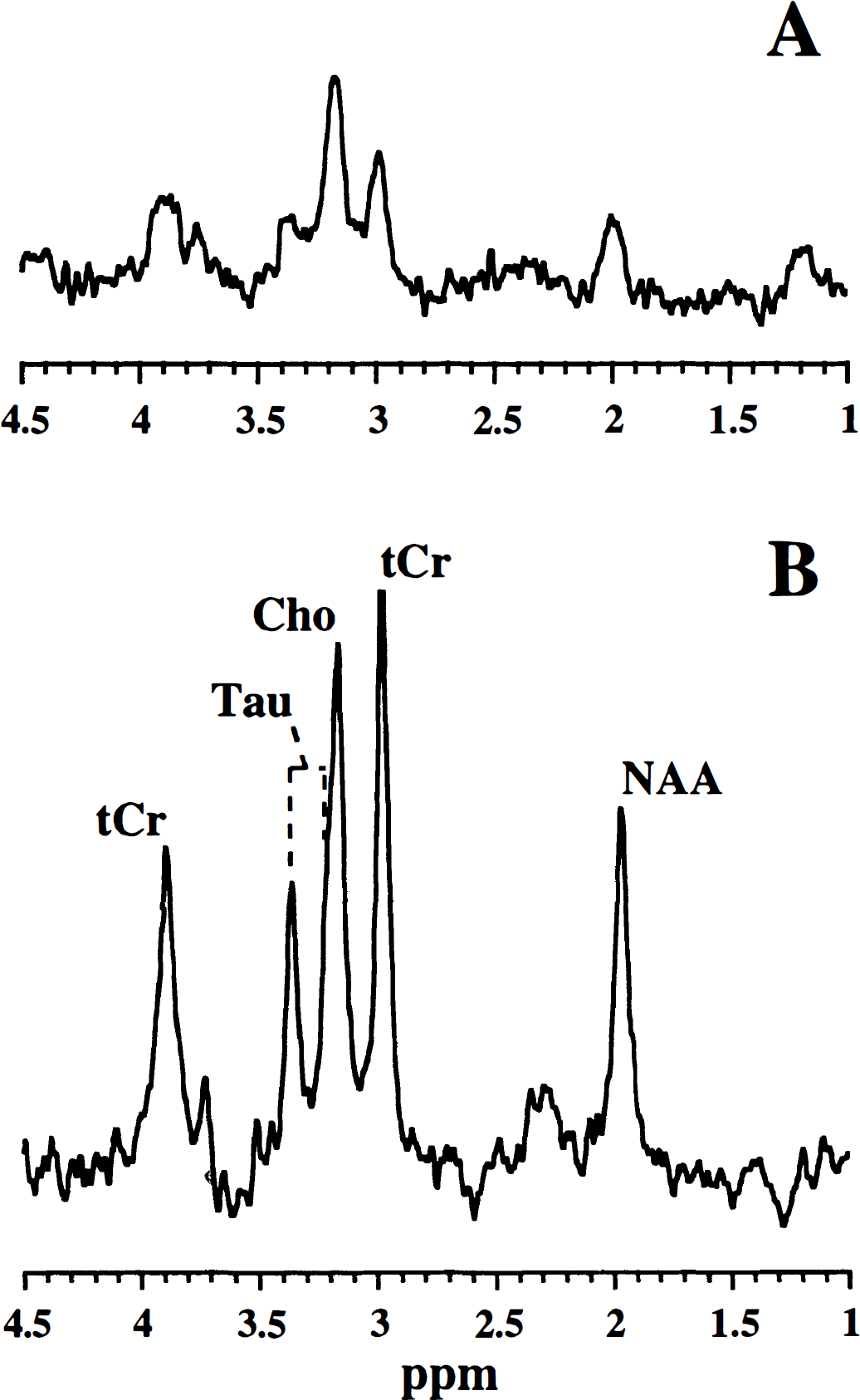
We first measured the ADC of brain water and metabolites in neonatal rats before and after cardiac arrest to establish the diffusion in control neonatal rat brain and the changes therein with ischemia. Fig. 1 shows representative 1H-MR spectra of neonatal rat brain before (Fig. 1A) and after cardiac arrest (Fig. 1B). Fig. 2 shows the ADC values of 1H-containing metabolites and water. In general, brain metabolites had considerably lower ADC values than brain tissue water, partly because of the differences in molecular mass. Diffusion-weighted MRS of neonatal brain after cardiac arrest-induced global ischemia was started approximately 30 to 40 minutes after intraperitoneal KCl injection, because at this time the rapid ischemia-induced water ADC drop had stabilized (Fig. 3). Cardiac arrest resulted in a dramatic reduction of the ADC of all metabolites and water (Fig. 2). Water ADC dropped to approximately 30%, and the ADC of brain metabolites to approximately 45% of their normal values. We also measured the ADC of lactate, which rapidly accumulated in the brain as a result of the severe ischemia. The relatively small lactate molecules, some of which supposedly are present in the extracellular space, exhibited a significantly higher apparent diffusion coefficient than the other metabolites (P < 0.01), except for Tau. From the change in chemical shift difference between the NAA and the residual water peak, it was estimated that global ischemia led to a significant reduction in brain temperature of 6.3°C ± 1.5°C (P < 0.01) after 0.5 to 1 hour.
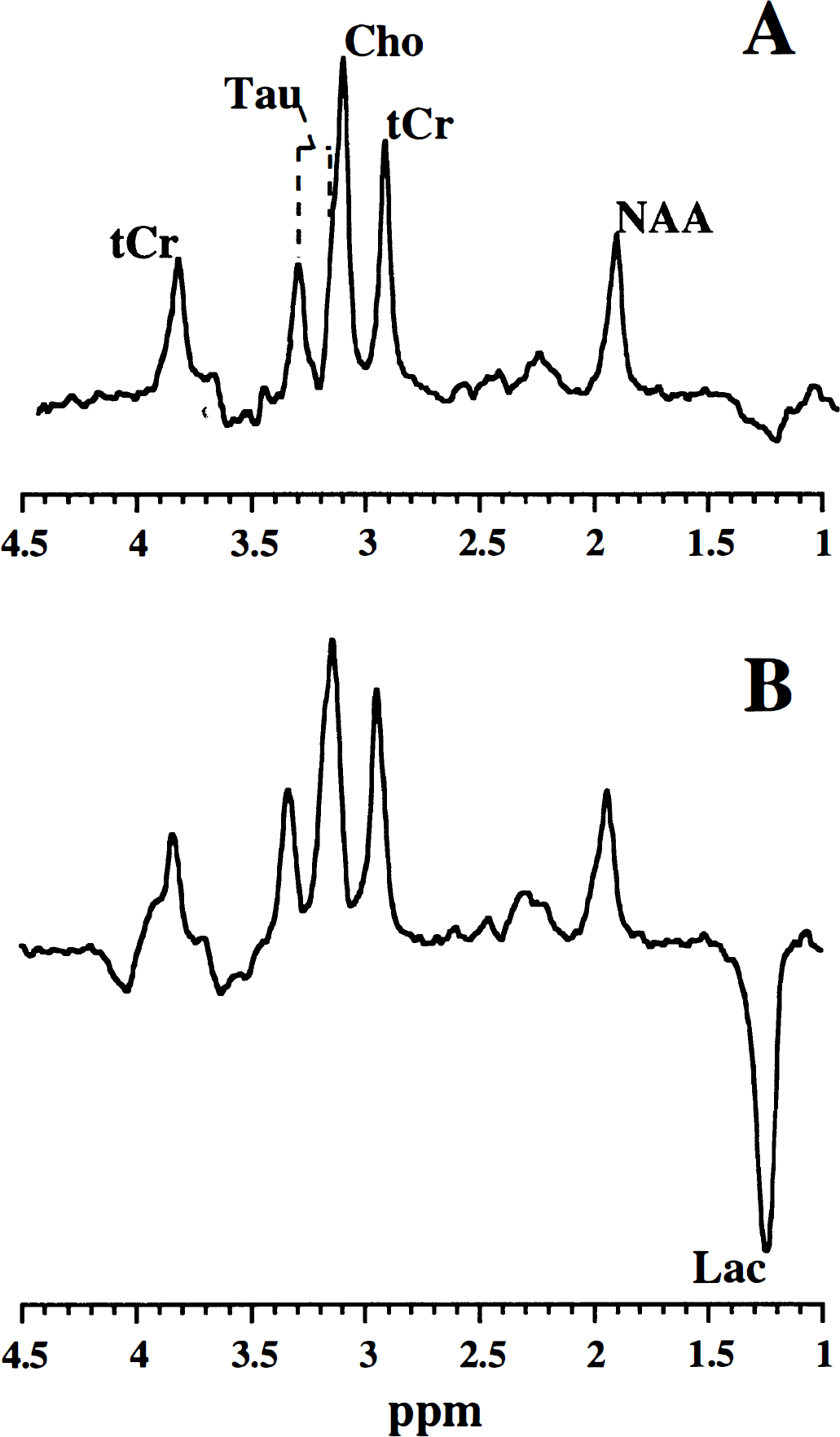
1H-magnetic resonance (–MR) spectra (obtained with a b value of 258 s/mm2) from neonatal rat brain before
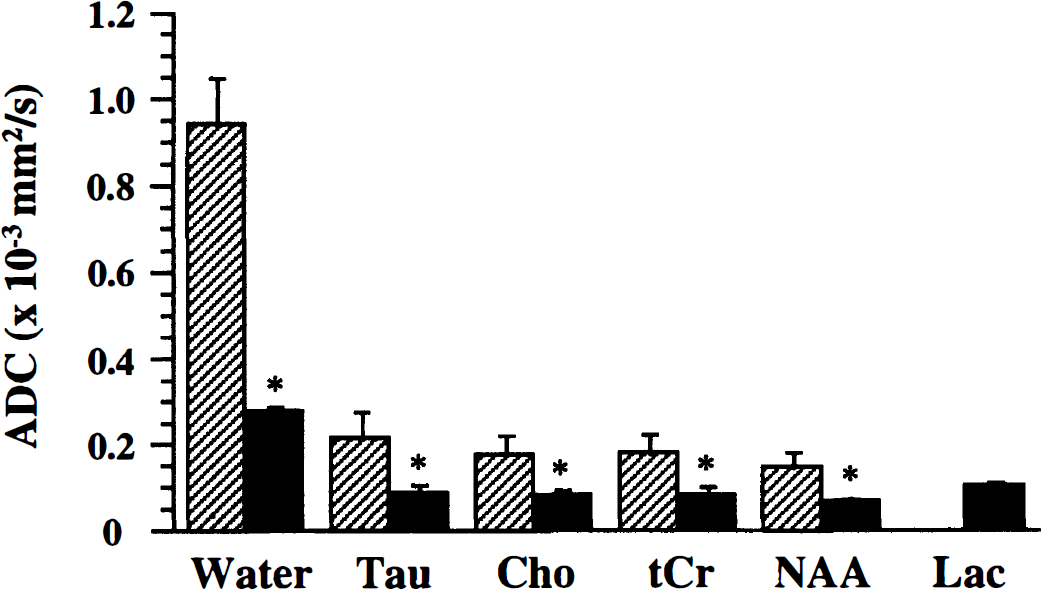
Apparent diffusion coefficients (ADC) (mean ± SD) of brain water and the brain metabolites Tau, Cho, tCr, NAA, and Lac before (shaded bars) and 0.5 to 1 hour after induction of global ischemia (black bars) in neonatal rat brain (*P < 0.01, versus before). Note that Lac was detected only in ischemic brain.
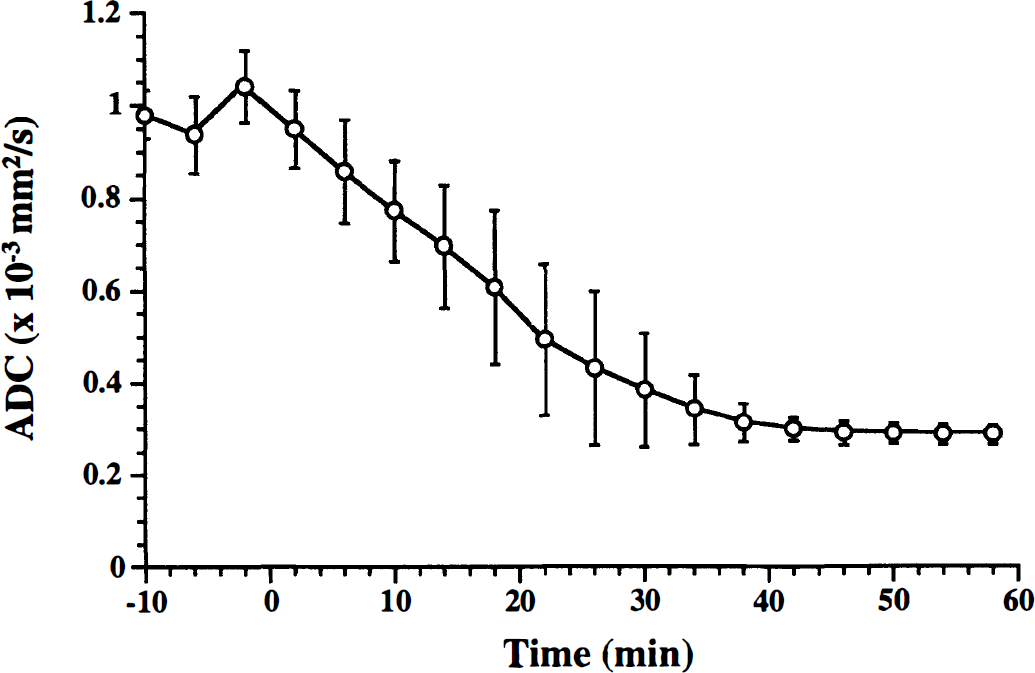
Time course of tissue water ADC in neonatal rat brain after KCl-induced cardiac arrest (KCl was injected at time 0).
Excitotoxic injury in neonatal rat brain
Intrastriatal NMDA injection resulted in a large ipsilateral lesion, characterized by a strongly reduced tissue water ADC in the first hours (Fig. 4). Brain metabolite levels were essentially unchanged (Fig. 4). Besides the highly significant (P < 0.01) drop of the ADC of water (to approximately 45% of contralateral), the ADC values of brain metabolites were also reduced (varying between 50% and 80% of contralateral values), which was statistically significant (P < 0.05) for Tau and NAA (Fig. 5A). Tissue temperatures were the same in both hemispheres (ipsilateral versus contralateral, ΔT = 0.3°C ± 2.3°C). Ipsilateral and contralateral ADC values in vehicle-injected animals were not significantly different (Fig. 5B). At 24 hours after NMDA injection, metabolite levels were clearly lower. A distinct lactate peak was not detected. Although the ipsilateral water ADC was higher as compared with the acute stages, it was still significantly decreased after 24 hours (Fig. 5C). The ipsilateral versus contralateral differences in ADC for all metabolites after 24 hours were significantly lower than the differences in the first hours (P < 0.05). Except for Tau, which still had lower diffusion values, the ADC of the metabolites had returned to baseline values (Fig. 5C). After 72 hours metabolites were hardly detectable in the ipsilateral voxel (Fig. 6A). The excitotoxic lesion was characterized as an area with a high ADC of water (Fig. 5D). In three animals metabolites were essentially undetectable, which excluded proper ADC calculation. The ADC values of brain metabolites in the ipsilateral hemisphere of the remaining animals were not significantly different from contralateral values (Fig. 5D). At 24 and 72 hours temperature differences between the ipsilateral and contralateral hemisphere were statistically insignificant (−1.8°C ± 1.1°C and −1.6°C ± 1.2°C, respectively).
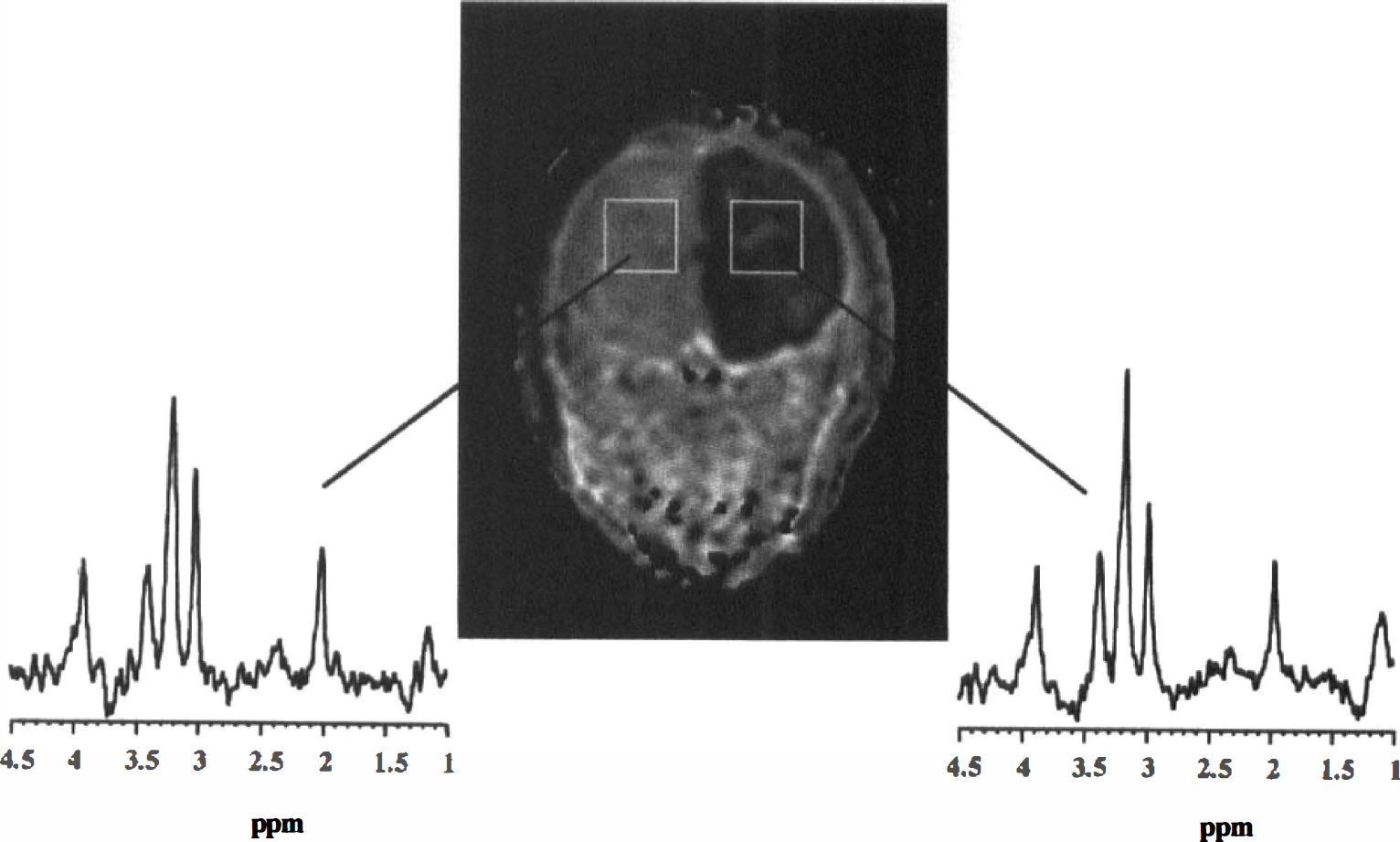
Tissue water ADC map of a coronal slice through neonatal rat head 0.5 hour after unilateral N-methyl-D-aspartate (NMDA) -injection. The lesion is characterized by a significant reduction of the tissue water ADC in the ipsilateral brain hemisphere. Typical voxel positioning for the diffusion-weighted (DW) magnetic resonance spectroscopy (MRS) experiments is indicated by white boxes. 1H-MR spectra (obtained with a b value of 258 s/mm2) from both hemispheres demonstrated comparable metabolite levels.
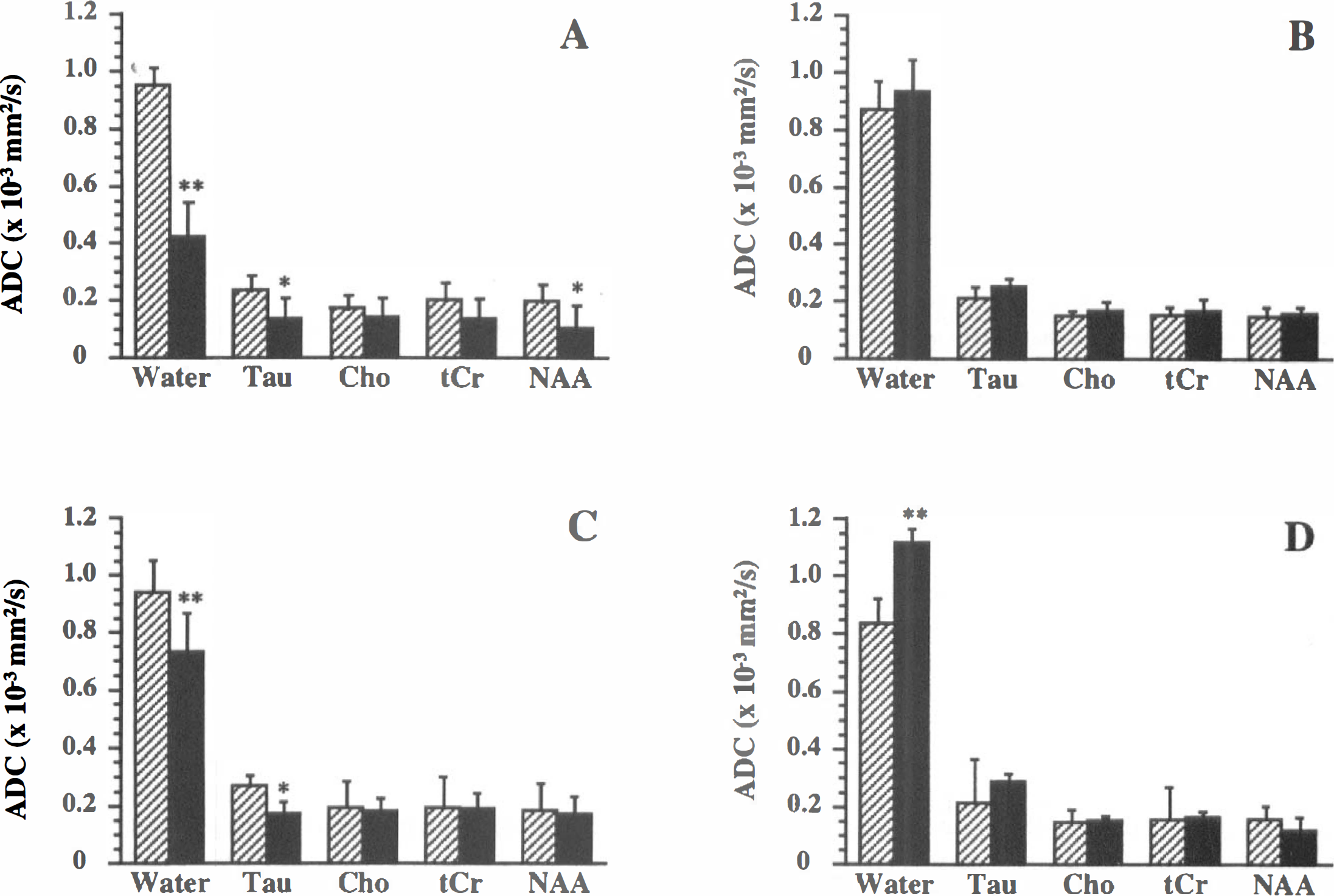
ADC (mean ± SD) of brain water and the brain metabolites Tau, Cho, tCr, and NAA in the ipsilateral (black bars) and contralateral hemisphere (shaded bars) at 0.5 to 3 hours after unilateral NMDA-
1H-MR spectra (obtained with a b value of 258 s/mm2) from the ipsilateral hemisphere in neonatal rat brain at 72 hours after unilateral NMDA injection, without
MK-801 treatment after excitotoxic injury in neonatal rat brain
MK-801 administration 1.5 hours after NMDA injection completely normalized the water ADC in the ipsilateral hemisphere (Fig. 7A). However, although ADC values of all metabolites showed a slight recovery 1.5 hours after MK-801 injection, the NMDA-induced ADC reduction was still apparent (Fig. 7A). At 72 hours after administration of MK-801 no damage was detected; the tissue water ADC was normal (Fig. 7B) and metabolite levels were maintained (Fig. 6B). In addition, metabolite ADCs were similar in both hemispheres (Fig. 7B). Temperature differences between the ipsilateral and contralateral hemispheres were not significant in MK-801-treated animals (0.7°C ± 1.5°C and −0.6°C ± 0.3°C at 1.5 hours and 72 hours after MK-801 administration, respectively).
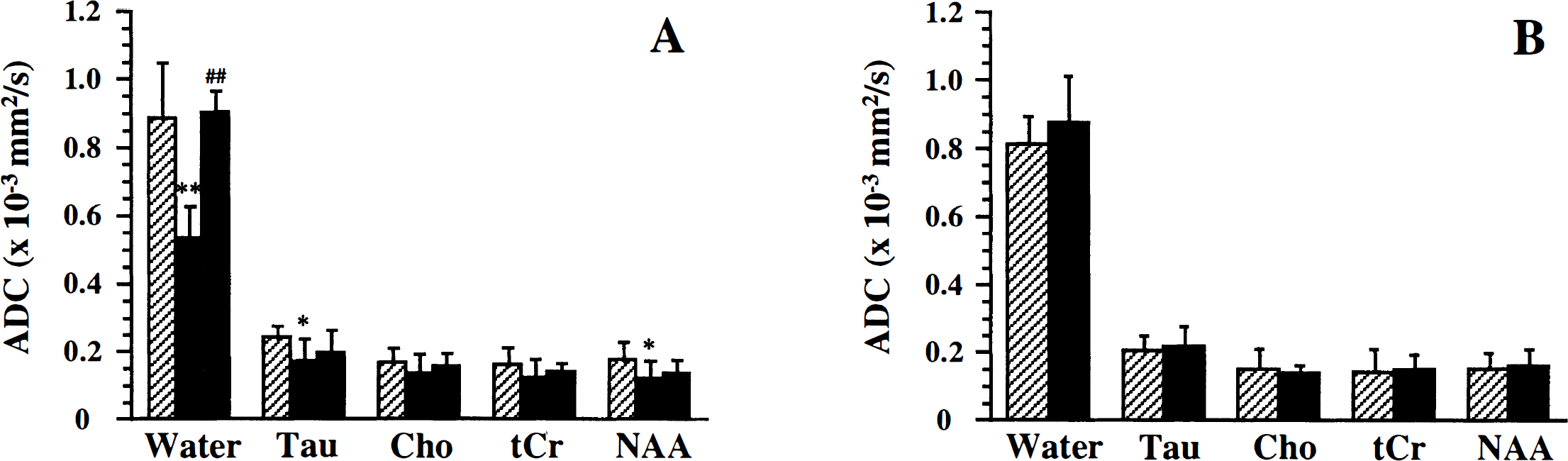
ADC (mean ± SD) of brain water and the brain metabolites Tau, Cho, tCr, and NAA.
DISCUSSION
In this study we used in vivo DW 1H-MRS to assess the diffusion of water and intracellular metabolites in relation to global ischemia and excitotoxic injury in neonatal rat brain to unravel a number of aspects of ischemic tissue damage. The experiments were performed in neonatal rats because (1) neonatal rat brain is highly susceptible to excitotoxicity, resulting in large and pronounced excitotoxic lesions, (2) excitotoxic injury is completely reversible in neonatal rat brain, which enables the assessment of recovery patterns, and (3) diffusion-related changes after both excitotoxic injury and global ischemia in neonatal rat brain have been analyzed extensively. The results are discussed in the light of biophysical alterations expected to occur in both neonatal and adult brain.
As expected, cardiac arrest-induced global ischemia resulted in a dramatic reduction of the tissue water ADC in neonatal rat brain. This dynamic ADC decline has been shown to correlate with a decrease of the extracellular space volume fraction and an increase of the extracellular tortuosity (Van der Toorn et al., 1996b), both of which are a manifestation of massive cell swelling (i.e., cytotoxic edema). These dynamic alterations stabilized after 30 to 40 minutes. Diffusion measurements performed between 0.5 and 1 hour after cardiac arrest demonstrated that not only had the water ADC dropped significantly, but that this was also the case for the metabolite ADC.
An ischemia-induced reduction of the diffusion of metabolites has been demonstrated previously in adult animals (Wick et al., 1995; Van der Toorn et al., 1996a). In addition, by use of cesium-133 MR techniques Neil and colleagues (1996) showed that the diffusion of the potassium analog cesium, which accumulates intracellularly after dietary intake, was retarded as a result of global ischemia in the rat.
The translational mobility of molecules in the intracellular space is determined by the cytoplasmic viscosity, macromolecular binding, collisions with intracellular obstacles, and bulk cytosolic motion. The calculated ADC is also related to the root mean square displacement √〈x2〉 according to the equation √〈x2〉 = √(2 × ADC × tdiff). The diffusion time tdiff in our DW MRS experiments (19.2 ms) would correspond with a √〈x2〉 of about 2.4 μm for the brain metabolites (assuming an average ADC of 0.15 × 10−3 mm2/s). Although this distance is of a subcellular dimension, restriction effects caused by, for example, the cell boundaries and intracellular organelles do supposedly affect the ADC.
Importantly, the cytotoxic edema-associated intracellular diffusion hindrance cannot be explained by enhanced restriction by cell boundaries, as the enlarged intracellular volume would be expected to increase molecular mobility. Furthermore, one would expect that the massive intracellular water accumulation would reduce cytoplasmic viscosity. In our study, the temperature drop and blood deoxygenation caused by global ischemia could potentially contribute to the observed metabolite ADC reduction (see also Van der Toorn et al., 1996a). We measured a brain temperature decline of approximately 6°C. Hasegawa et al. (1994) showed that the ADC of water in mature rat brain drops 0.013 × 10−3 mm2/s for every degree Centigrade temperature reduction. A temperature drop of 6°C would therefore account for an ADC decrease of approximately 0.078 × 10−3 mm2/s, which is far less than the actual mean water ADC drop of 0.665 × 10−3 mm2/s we measured in global ischemic neonatal brain. The loss of cerebral blood flow in ischemia is followed by tissue deoxygenation. An increase in the proportion of paramagnetic deoxyhemoglobin in the blood compartment affects the local magnetic susceptibility and could thereby potentially alter the effective b values (Van der Toorn et al., 1996a). Such misinterpretation of b values will result in an underestimation of the ADC values. Recently, Van der Toorn and coworkers (1996a) theorized that changes in brain oxygenation status and temperature are not expected to make large contributions to the decline of metabolite ADC in a model of focal cerebral ischemia in adult rats.
To exclude the combined effects of the ischemia-induced temperature decline and alterations in magnetic susceptibility gradients, we assessed the ADC of brain metabolites in cytotoxic edematous tissue under nonischemic conditions. We used a model of NMDA-induced excitotoxicity in the immature rat brain, which has a high vulnerability to the excitotoxin NMDA (Ikonomidou et al., 1989). Intrastriatal NMDA injection has been shown to rapidly result in extensive cell swelling (and consequent water ADC reduction) (Verheul et al., 1994) and is not accompanied by a decline in CBF (Taylor et al., 1995) and severe metabolic alterations (Dijkhuizen et al., 1996). Accordingly, 1H-MR spectra of the tissue in the excitotoxic lesion were essentially the same as the contralateral spectra shortly after NMDA injection and lactate was not detected. Yet, the ADC of the 1H-metabolites had declined. Because tissue deoxygenation- and temperature-related effects can be excluded, these intracellular diffusion changes strongly suggest that an intracellular transformation has occurred.
Previous histopathologic studies have reported various early intracellular alterations together with cellular swelling as a result of cerebral ischemia, which theoretically could lead to an increased hindrance for diffusion (e.g., see Yamamoto et al., 1986). Although there is less literature on excitotoxin-induced structural changes in the intracellular environment, similar findings have been described after intracerebral administration of NMDA (Van Lookeren Campagne et al., 1995). Swelling of cell organelles (e.g., mitochondria), disaggregation of polyribosomes, progressive cytoplasmic and karyoplasmic condensation, proliferation of intracellular membranes, and increase in the number of cytoplasmic fibrillary structures may contribute to an increased obstruction of the mobility of intracellular compounds. The typical disorganization of the cytoplasmic matrix (i.e., the microtrabecular lattice and the cytoskeleton) after ischemia (Molitoris, 1997) or excitotoxicity (Sanchez et al., 1997) could play a crucial role in the raising of intracellular obstacles. Early cytoarchitectural alterations include enhanced polymerization, disordering of microtubular arrangement, redistribution and aggregation of filaments, and release of cytoskeleton-associated surface membrane proteins (Molitoris, 1997). Furthermore, because all kinds of macromolecules are organized in the supramolecular structure of the cytoplasmic matrix, disruption of this structure could lead to dramatic changes in binding of low molecular weight molecules. In addition to the above mechanisms, overall displacements in the cytosol could be reduced as a result of diminished cytoplasmic streaming. It has been demonstrated that cytoplasmic streaming is halted at low ATP levels, low pH values, and elevated intracellular Ca2+ concentrations (Kamiya, 1984), all of which can be direct consequences of cerebral ischemia or excitotoxicity. Finally, it should be stressed that the involvement of the above-mentioned factors in the intracellular diffusion changes is only speculative, as these structural and functional cytosolic alterations were not actually measured in this study.
The NMDA-induced relative ADC reductions were smaller than after global ischemia. Interestingly, Van der Toorn et al (1996a) observed a larger decrease of metabolite ADC after global ischemia as compared with focal ischemia in adult rat brain. These findings could be explained by the less severe cellular pathophysiologic changes. In accordance, it has been demonstrated that acute excitotoxicity in this model is not accompanied by profound metabolic injury (Dijkhuizen et al., 1996). Other factors, such as the absence of significant temperature reductions and tissue deoxygenation, could contribute to the less severe ADC changes after intrastriatal NMDA injection.
At the more chronic stages of excitotoxicity, the decreased metabolite levels reflected serious tissue damage. The DW 1H-MRS experiments pointed out that, except for Tau at 24 hours, ADC levels of the remaining metabolites approached contralateral levels after 24 and 72 hours, which may be related to the degradation and loss of intracellular obstacles as a consequence of proteolytic activities. It has been demonstrated that the density of cytoskeletal filaments decreases at chronic stages of cerebral ischemia, but not in the early hours (Inuzuka et al., 1990). In addition, excitotoxic damage induced by intrahippocampal injection of the NMDA-agonist quinolinate led to loss of cytoskeletal fragments (Pang et al., 1996). Henriksen et al. (1995) reported an elevated diffusion of NAA in ischemic human brain, which they attributed to an increased intracellular water content. However, because most data were obtained several days after the stroke incident it could well be that degeneration of the intraneuronal environment was responsible for this ADC increase of NAA. In agreement with the latter, it was demonstrated in a recent study by Rosenbaum et al. (1997) that the initially reduced ADC of NAA increased at 6 hours after focal cerebral ischemia in the rat. At 72 hours after NMDA injection the excitotoxic lesion was characterized as an area with an elevated diffusion of water and almost complete loss of brain metabolites. These changes reflect severe neuronal degeneration and tissue cavitation (see Van Lookeren Campagne et al., 1994).
An important aspect of this model of NMDA-induced excitotoxicity is that the cellular swelling can be practically entirely reversed by administration of the noncompetitive NMDA-antagonist MK-801 within approximately 1.5 hours (Van Lookeren Campagne et al., 1994). As expected we found a complete normalization of the tissue water ADC. However, the reduced ADC of brain metabolites during excitotoxicity did not significantly recover 1 to 2 hours after MK-801 injection. This points toward a sustained modification of the intracellular environment. Similarly, Wick et al. (1995) found that reperfusion after global ischemia rapidly normalized the water ADC, but not the metabolite ADC values. Although the ADC of water and metabolites did not recover in parallel during the acute stages, we found that the ADC of both tissue water and brain metabolites was at control levels 72 hours after MK-801 administration and no signs of cerebral injury were detected. Clearly, in agreement with Van Lookeren Campagne et al. (1994), MK-801 had completely prevented the development of irreversible excitotoxic tissue damage.
In conclusion, this study has shown that in vivo intracellular diffusion measurements can provide important information on cellular pathophysiology. The data shed some light on the underlying mechanisms involved in the early ischemia-induced water ADC drop, which could aid in the improvement of MR-based diagnosis and prognosis in stroke patients. Ischemia- or excitotoxin-induced changes in the mobility of intracellular molecules may be the result of (1) alterations in the configuration of the cytoplasmic matrix, affecting intracellular tortuosity and molecular interactions, and (2) changes in cytoplasmic streaming. The contribution of brain temperature changes and tissue deoxygenation to ischemia-induced intracellular ADC changes was minor. Our data suggest that changes in the diffusion of water in the intracellular compartment are involved in the reduction of the diffusion of tissue water in cerebral ischemia. However, because the diffusion of metabolites may be affected by factors other than the diffusion of intracellular water, the extrapolation of our findings of reduced metabolite diffusivity to possible changes in intracellular water diffusion should be treated with caution and requires further study.
Footnotes
Acknowledgements
The authors thank Dr. Jan de Bruin and Paul Dijkhuizen for providing experimental requisites, Gerard van Vliet and Arnaud van Kranenburg for expert technical assistance, and Drs. Menno van Lookeren Campagne and Jaap Joles for critically reading the manuscript.
