Abstract
The centrum semiovale may be susceptible to hypoperfusion as a result of carotid artery occlusion. Recent studies suggest that the cerebral hematocrit decreases with diminished cerebral perfusion pressure. To investigate whether the effect of carotid artery occlusion on the hematocrit in the centrum semiovale is different from that in the cerebral cortex, seven patients with unilateral carotid artery occlusion were studied with positron emission tomography. The distributions of the red blood cell and plasma volumes were assessed using carbon monoxide labeled with oxygen 15 and human serum albumin-dithiosemicarbazone tracers labeled with copper 62, respectively. The CBF and CMRO2 were also measured with the 15O steady-state technique. The calculated values for the hematocrit in the centrum semiovale ipsilateral to the arterial occlusion were significantly decreased compared with those in any of the other regions examined (the overlying cortical region and the contralateral cortex and centrum semiovale). This decrease in hematocrit, which resulted from a more pronounced increase in plasma volume than in red blood cell volume, was associated with a decrease in CBF and an increase in the oxygen extraction fraction. Hemodynamic disturbance caused by carotid artery occlusion may induce selective decrease of hematocrit limited to the centrum semiovale.
Keywords
The topographic pattern of cerebral infarcts is important as an indicator of the causes and pathophysiologic mechanisms of ischemic stroke (Bogousslavsky, 1991; Zülch and Hossmann, 1988). Stained or striated infarcts in the mid centrum semiovale have been associated with severe internal carotid artery (ICA) disease and considered to be of hemodynamic origin (Bogousslavsky and Regli, 1992; Nakano et al., 1995; Wodarz, 1980). The centrum semiovale lies in the distal field territory (terminal area) supplied by the ICA through the long medullary branches of the middle and anterior cerebral arteries, whereas the overlying cerebral cortex is supplied through the shorter branches of these arteries (Moody et al., 1990). Because of this difference of the pattern of vascular supply, the centrum semiovale may be more vulnerable than the overlying cerebral cortex to the reduction in cerebral perfusion pressure caused by ICA occlusions (Zülch and Hossmann, 1988). The association of lesions in the centrum semiovale with hemodynamic compromise of the overlying cerebral cortex in patients with ICA occlusion and no cortical infarction supports this hypothesis (Isaka et al., 1997; Moriwaki et al., 1997; Yamauchi et al., 1991). However, there is no direct evidence that this white matter region is more susceptible than the cortex to the reduction in perfusion pressure caused by ICA occlusion. No study has demonstrated specific alterations of the hemodynamic parameters confined to the mid centrum semiovale in patients with ICA occlusion.
The physiologic response to reduced cerebral perfusion pressure is compensatory vasodilation, which may be shown as an increased cerebral blood volume (CBV) in patients with ICA occlusion by using positron emission tomography (PET) (Gibbs et al., 1984; Powers et al., 1984). The difference between the values of CBV in the white matter and in the cortex of the normal brain makes it difficult to investigate the selective increase in CBV in the white matter, if any, compared with that in the cerebral cortex in the early stage of hemodynamic disturbance caused by ICA occlusion. In addition to the increase in total CBV, reduced perfusion may cause a change in the ratio of cerebral red blood cell volume (CRCV) to cerebral plasma volume (CPV), and thus in hematocrit (Hct). Recent experimental studies suggest that the diminished cerebral perfusion pressure decreases cerebral Hct, which might be caused by the more pronounced reduction of the velocity of plasma than of red blood cells (RBC) (Hudetz, 1997; Hudetz et al., 1995; Mchedlishvili and Varazashvili, 1987; Mchedlishvili et al., 1997). Thus, if the centrum semiovale is more susceptible to the reduction in perfusion pressure caused by ICA occlusion than are the adjacent cerebral cortical regions, it would show a lower Hct than these cortical regions. The decrease in Hct in the centrum semiovale with reduced CBF would lead to a reduction in oxygen delivery, which might be related to the high incidence of centrum semiovale infarct in ICA occlusion.
The CRCV, CPV, and Hct can be measured in vivo using the radiotracer method and PET or single photon emission computed tomography (Lammertsma et al., 1984; Sakai et al., 1985). Our preliminary study in seven patients with major cerebral arterial occlusion (Okazawa et al., 1996), including four of the patients with ICA occlusion included in this study, suggested a reduction in cerebral Hct distal to the major cerebral arterial occlusion site. However, it was unclear from the analysis of the data for the large regions of interest (ROI), including the whole hemisphere, whether the effect of major cerebral arterial occlusion on the regional cerebral Hct in cerebral cortex is different from that in white matter. The purpose of this study was to determine whether there is a difference between the effects of ICA occlusion on regional cerebral Hct in the centrum semiovale and in the cerebral cortex and, if so, whether ICA occlusion induces selective decrease of the regional cerebral Hct confined to the centrum semiovale.
PATIENTS AND METHODS
Patients
We studied seven patients with unilateral ICA occlusion in whom magnetic resonance imaging (MRI) demonstrated no cortical infarctions and at most only one infarction, less than 1.5 cm in diameter, in the centrum semiovale. These patients included five men and two women, aged 61 to 74 years (mean ± SD, 66 ± 5 years). The diagnosis of ICA occlusion was based on conventional angiography, which disclosed no significant arterial disease contralateral to the ICA occlusion in any patient. One patient had no symptoms, two had transient ischemic attacks, and four had minor hemispheric stroke with mild disability. All symptoms were related to the affected carotid distribution. In the asymptomatic patient, ICA occlusion was suspected because of the observation of flow void loss on MRI performed because of hoarseness. In each of the six symptomatic patients, T1-weighted MRI disclosed only one minor subcortical infarction, defined as a well-demarcated hypointense area in the middle cerebral artery (MCA) territory or watershed area of the hemisphere with ICA occlusion, including the centrum semiovale in one patient. The size of the infarct ranged from 50 to 240 mm2. On T2-weighted MRI images, punctate or patchy high-intensity areas were observed in the cerebral white matter with ICA occlusion in all patients. Three patients showed a confluent high-intensity lesion in the centrum semiovale ipsilateral to the ICA occlusion. Punctate or patchy high-intensity areas of lesser degree were found on the nonaffected side in two patients. The interval between the latest ischemic event and the evaluations with PET and MRI ranged from 2 to 41 months (mean ± SD, 20 ± 15 months). In the asymptomatic patient, ICA occlusion was confirmed on angiography 28 months before the PET study. All patients and their relatives gave informed consent to the conventional angiographic and PET studies.
Positron emission tomography
All patients were scanned with a commercially available PET system (PCT-3600W, Hitachi Medical Co., Tokyo, Japan) (Sadato et al., 1993). This system simultaneously acquires 15 slices with a center-to-center distance of 7 mm. All scans were obtained at a resolution of 7.5 mm full width half-maximum in the transaxial direction and of 6.5 mm in the axial direction in the wobbling mode. Patients were positioned with the orbitomeatal line parallel to the detector rings.
After a transmission scan, the CBF was determined while the subject continuously inhaled 300 MBq of CO15O per minute through a mask, and the CMRO2 and oxygen extraction fraction (OEF) were measured during continuous inhalation of 500 MBq of O15O per minute. Data were collected for 5 minutes. For measurement of the CRCV, 1.20 GBq of C15O was inhaled, and the PET scanning was started 30 seconds or more after the appearance of the peak count of the brain tissue adequate for data collection and continued for 3 minutes. We calculated CBF, CMRO2, and OEF based on the steady-state method (Frackowiak et al., 1980). The CBV was calculated from the data of the C15O scan and was incorporated into the correction of the CMRO2 and OEF (Lammertsma and Jones, 1983; Lammertsma et al., 1987). In the calculation of the CBV, a conventional Hct ratio of 0.85 was used. Functional images were reconstructed with 128 × 128 pixels, with each pixel representing an area of 2.0 mm × 2.0 mm.
After the completion of the 15O-gas study, 296 to 740 MBq of human serum albumin-dithiosemicarbazone labeled with copper 62 (62Cu-HSA-DTS) was injected intravenously over 15 seconds in a total volume of 8 mL to obtain CPV images (Fujibayashi et al., 1990). The PET data acquisition was started 3 minutes after administration of 62Cu-HSA-DTS and continued for 8 minutes. Blood samples were obtained at 1, 5, and 7 minutes after injection of 62Cu-HSA-DTS, and both the whole blood radioactivity and plasma radioactivity were counted (Okazawa et al., 1996). Regional CRCV and CPV were calculated using the PET images acquired in the C15O and 62Cu-HSA-DTS studies according to the following equations: CRCV = Cco/(Aco/AHct) (mL/g) and CPV = Chsa/Phsa (mL/g), where Cco and Chsa are the cerebral tissue radioactivities of C15O and of 62Cu-HSA-DTS, respectively, Aco is the whole-blood radioactivity of C15O, Phsa is the plasma count of 62Cu-HSA-DTS, and AHct is the large vessel arterial Hct measured in the blood sampled from the radial artery. The regional cerebral Hct (CHct) was calculated from the CRCV and CPV for each patient as CHct = CRCV/(CRCV + CPV). We also calculated the mean transit times of blood (Tb), red blood cells (Tr), and plasma (Tp), defined as follows: Tb = (CRCV + CPV)/CBF, Tr = Tb × (CHct/AHct), and Tp = Tb × 1 − CHct)/(1 − AHct), respectively (Pries et al., 1986).
We analyzed a tomographic plane 78 mm above and parallel to the orbitomeatal line, which corresponded to the level of the centrum semiovale. Each image was examined by placing a total of 15 circular ROI 12 mm in diameter compactly over the gray matter of the cortex and three circular ROI 16 mm in diameter compactly along the anteroposterior direction in the mid centrum semiovale. We used a method correlating PET with MRI described elsewhere (Yamauchi et al., 1990a). The cortical ROI were included in the distribution of the MCA, as well as the watershed areas between the anterior cerebral artery and MCA (anterior watershed) and between the MCA and posterior cerebral artery (posterior watershed) (Yamauchi et al., 1990b). The mean values for the cerebral cortex and the centrum semiovale in each hemisphere were calculated as the average for all the regions including the cerebral cortex and the centrum semiovale, respectively. In the patient with infarcts in the centrum semiovale, the data for the ROI including the infarct were excluded from the analysis.
Statistical analysis
The values of the PET variables were compared by two-way analysis of variance (ANOVA) with respect to sides, i.e., the hemispheres ipsilateral to and contralateral to the ICA occlusion, and with respect to regions, i.e., the cerebral cortex and the centrum semiovale. Because the two-way ANOVA disclosed significant effects of both sides and regions with a significant interaction for the cerebral Hct, regional analysis was performed using one-way ANOVA and Scheffé F test for post hoc comparisons. A P value of < 0.05 was considered statistically significant. No significant interaction for the other PET variables was found. Thus, Wilcoxon signed-rank test was performed to compare the effect of ICA occlusion on these variables in each region separately. When Bonferroni's correction for multiple comparisons was used, P < 0.025 (0.05/2) was regarded as indicating statistical significance.
RESULTS
The value of large vessel AHct in the patients ranged from 0.35 to 0.41 (mean ± SD, 0.38 ± 0.02). No patient showed a significant change in Pao2 or Paco2 during PET scanning.
In the centrum semiovale ipsilateral to the ICA occlusion, the value of CHct was decreased compared with that in any of the other regions examined (the contralateral centrum semiovale and the overlying cortical regions in the two hemispheres) (Table 1). In the hemisphere contralateral to the ICA occlusion, no difference was found between the values of CHct in the centrum semiovale and in the overlying cerebral cortex.
Physiologic values for the cerebral cortex and centrum semiovale ipsilateral and contralateral to the internal carotid artery occlusion
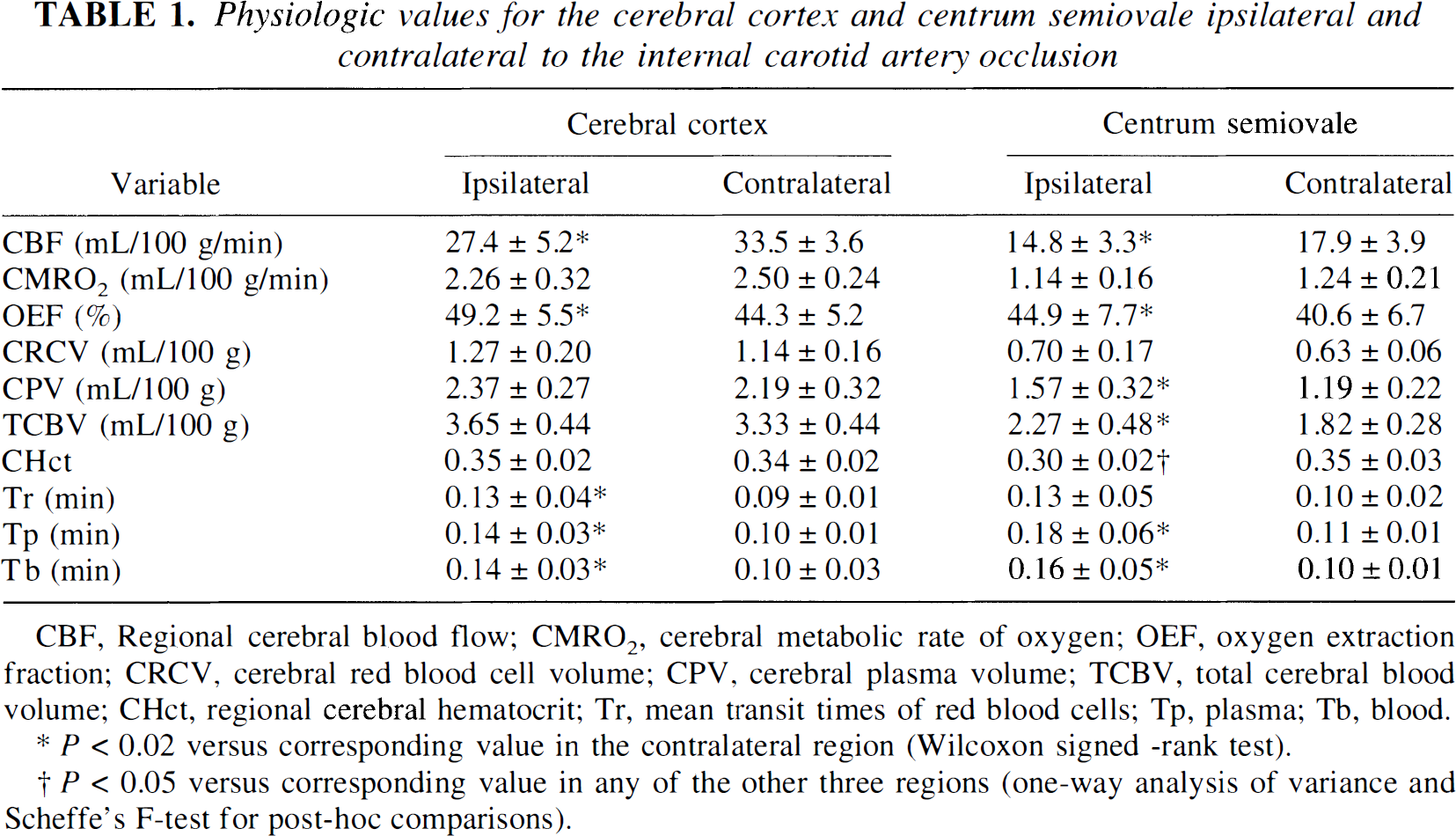
CBF, Regional cerebral blood flow; CMRO2, cerebral metabolic rate of oxygen; OEF, oxygen extraction fraction; CRCV, cerebral red blood cell volume; CPV, cerebral plasma volume; TCBV, total cerebral blood volume; CHct, regional cerebral hematocrit; Tr, mean transit times of red blood cells; Tp, plasma; Tb, blood.
P < 0.02 versus corresponding value in the contralateral region (Wilcoxon signed -rank test).
P < 0.05 versus corresponding value in any of the other three regions (one-way analysis of variance and Scheffe's F-test for post-hoc comparisons).
Compared with the contralateral centrum semiovale, the ipsilateral centrum semiovale showed a significantly decreased CBF with increased OEF. The total blood volume was increased in this region, and the more pronounced increase in CPV than in CRCV caused the decrease in Hct. The calculated Tb was increased, with greater increase in Tp than Tr.
Fig. 1 shows the images of CHct in the patient with transient ischemic attacks in whom no high-intensity lesions were depicted in the centrum semiovale on T2-weighted MRI images. A selective decrease in the Hct was found in the centrum semiovale ipsilateral to the ICA occlusion.
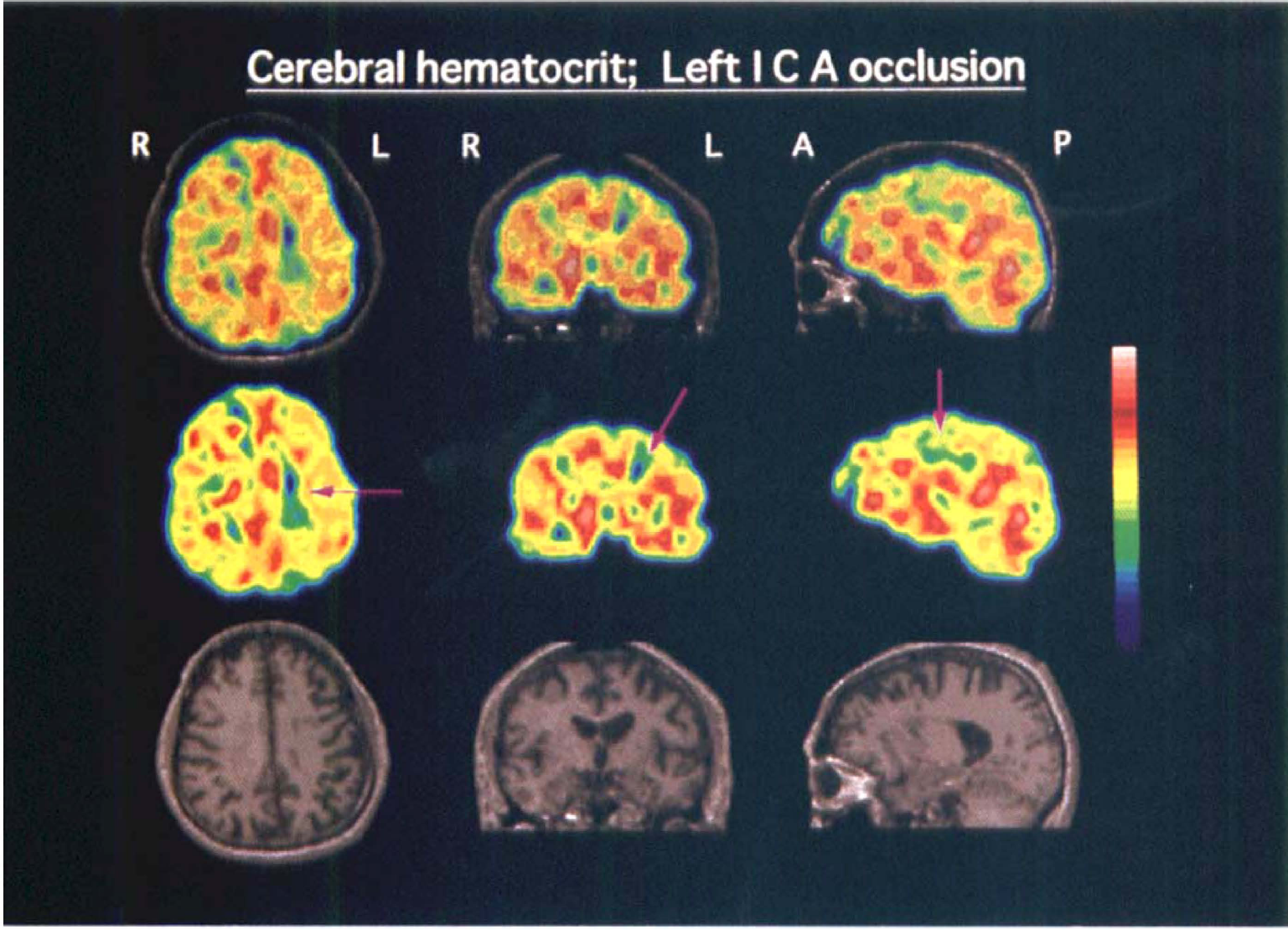
Examples of the PET images of cerebral hematocrit in a patient with left (L) internal carotid artery (ICA) occlusion. Upper row: The three-dimensional PET images (Middle row) were superimposed on the three-dimensional, spoiled gradient echo MRI images (Lower row) (TR = 40 ms, TE = 5 ms, flip angle = 40°, FOV = 24 cm) that were obtained with a 1.5 T device (Signa Advantage, GE Medical Systems, Milwaukee, Wisconsin, U.S.A.). From left to right (R): axial, coronal, and sagittal sections through the centrum semiovale. Sagittal sections are the images of the left hemisphere. Note the decrease in cerebral Hct in the centrum semiovale of the left hemisphere with arterial occlusion (arrows). A, anterior direction; P, posterior direction.
DISCUSSION
This study revealed that there is a difference between the effects of ICA occlusion on the regional CHct in the centrum semiovale and in the cerebral cortex, and that ICA occlusion induces selective decrease of the regional CHct confined to the centrum semiovale. We found that in these patients with ICA occlusion in the chronic stage, the value of the regional CHct in the centrum semiovale ipsilateral to the ICA occlusion was decreased compared with any of the other regions examined (the overlying cortical region and the contralateral cortex and centrum semiovale). In the hemisphere contralateral to the ICA occlusion, no difference was found between the values of CHct in the centrum semiovale and those in the overlying cerebral cortex, as in the normal control subjects previously studied (Okazawa et al., 1996). Therefore, the decrease in Hct in the centrum semiovale was related to the occlusion of the ICA.
The decrease in Hct in the centrum semiovale ipsilateral to the ICA occlusion may result from hemodynamic disturbance caused by the occlusion. This was supported by the finding that significant decrease of the CBF with increase of the OEF was found in the centrum semiovale ipsilateral to the ICA occlusion, compared with the contralateral centrum semiovale (Baron et al., 1981; Powers et al., 1984). The total blood volume was increased in this region, and the more pronounced increase in CPV than in CRCV caused the decrease in Hct. Because CBF was decreased in this region, the calculated Tb was increased, with greater increase in the Tp than in the Tr. Reductions in the perfusion pressure caused by ICA occlusion cause vasodilation and slowing of blood (Gibbs et al., 1984; Powers et al., 1984). Reduced cerebral perfusion may differentially influence RBC and plasma flow in the centrum semiovale, leading to the decrease in Hct.
The mechanism for the difference between the effects of ICA occlusion on the regional CHct in the centrum semiovale and in the cerebral cortex was not clarified by this study. Much greater changes in CPV and Tp in the centrum semiovale than in the overlying cortex caused this difference. The decrease in Hct with much greater increase in CPV than in CRCV was also present in the cerebral cortex, showing pronounced increase in OEF (Yamauchi et al., 1998), and the value of Hct was lower in the centrum semiovale than in the cortex in such patients as well. Therefore, the centrum semiovale may be more susceptible than the cortex to the hemodynamic disturbance caused by ICA occlusion, leading to the difference in Hct.
The stress on the importance of Hct in the cerebral ischemia applies primarily to capillary blood flow. Thus, the clinical importance of our results depends on the validity of the assumption that the Hct in this study, which was estimated for brain tissue overall, reflects the change in capillary Hct. Although this assumption cannot be verified from these results, recent experimental studies have suggested that the cerebral capillary Hct decreases after carotid artery occlusion (Hudetz, 1997; Mchedlishvili and Varazashvili, 1987; Mchedlishvili et al., 1997). In the rabbit carotid artery occlusion model causing about 50% reduction of CBF without infarct, a decrease in capillary Hct as well as in venous Hct was observed by direct measurement of Hct (Mchedlishvili and Varazashvili, 1987; Mchedlishvili et al., 1997). Studies of capillary flow with cranial window and video microscopy disclosed that RBC flux decreases more markedly than does RBC velocity with reduction in cerebral perfusion pressure caused by hemorrhagic hypotension, suggesting a decrease in capillary Hct (Hudetz, 1997; Hudetz et al., 1995). Thus, capillary Hct may decrease with reduction in cerebral perfusion pressure. These findings are in accordance with our results, although no experimental data have been obtained concerning the difference of the change in capillary Hct between the centrum semiovale and the cerebral cortex with changes in cerebral perfusion.
There are basic problems with respect to theoretical hemorheology, based on in vitro studies, that make our results intrinsically difficult to interpret in terms of either mechanisms or clinical implications (Gaehtgens and Marx, 1987). The relationship between blood flow properties observed in vitro and the functional behavior of blood in the living microcirculation remains unclear (Pries et al., 1996). Thus, we can only speculate on the following possibilities to explain the decrease in Hct in the centrum semiovale caused by hemodynamic disturbance. First, the lesser decrease in the velocity of RBC than that of plasma may result in concentration of RBC in the centers of vessels with an increase in the marginal cell-free plasma layer (Reinke et al., 1987). A study of cerebral microvessels (arteriole and venule) in the cat demonstrated that the thickness of the cell-free plasma layer was increased with a decrease in the pseudo-shear rate defined as the ratio of cell velocity to vessel radius, possibly because of aggregation of RBC (Yamaguchi et al., 1992). In the hemodynamically compromised region with increased blood volume and slow Tb, the thickness of the cell-free plasma layer of vessels may increase, resulting in the more pronounced increase in CPV than CRCV, and the decreased Hct. Second, an irregular distribution of RBC in arterial bifurcation may contribute to the decrease in Hct. It must be considered that volume flow and red cell flux partition at bifurcations modifies flow conditions in the complex branching network of microvessels (Pries et al., 1986). Direct correlation between the blood flow rate and Hct in arterial branches has been demonstrated (Gaehtgens et al., 1979). Such gradual separation of RBC and plasma in the arterial branching sequence leads to a decrease of RBC concentration in ischemic regions. During cerebral ischemia, a significant number of RBC-poor capillaries were demonstrated as a regular intermediate stage preceding closure of their lumen, contributing to the decrease in Hct in the ischemic region (Mchedlishvili et al., 1997; Theilen et al., 1994).
The decrease in Hct may also result from ischemic changes caused by ICA occlusion. The leakage of plasma tracer, 62Cu-HSA-DTS, into the cerebral parenchyma because of dysfunction of the blood-brain barrier may cause an artifactual increase in CPV that is disproportionate to any increase in CRCV. Lesions depicted on T2-weighted MRI as high-intensity areas were observed in the centrum semiovale in some patients. However, the selective decrease in Hct in the centrum semiovale ipsilateral to the occlusion was also found in the patient with no high-intensity lesions, and none of our patients showed any enhancement of the brain image on the MRI study, negative findings that do not support this mechanism.
The implications of the observed decrease in Hct in the centrum semiovale are also speculative, because the current understanding of oxygen delivery by the microcirculation and tissue oxygenation is not complete (Intaglietta et al., 1996). The homeostatic relationship between oxygen content and CBF maintains constant oxygen delivery under normal conditions (Harrison, 1989). The decrease in Hct in the centrum semiovale with reduced CBF indicates the exhaustion of this regulatory mechanism, which leads to a reduction in oxygen delivery (defined as the product of CBF and oxygen content, which is related to Hct). Increase in the OEF in this region reflects reduction in oxygen delivery relative to metabolic demand, which may cause structural changes because of oxygen deficiency if further reduction in cerebral perfusion pressure occurs. The decreased Hct even in the asymptomatic patient suggests that this change occurs at an early stage of hemodynamic disturbance in ICA occlusion. This white matter region may be more susceptible than the cortex to reduced oxygen delivery, which might be related to the high incidence of centrum semiovale infarct in ICA occlusion, although the appearance of ischemic changes may depend on the vulnerability of regions (the cerebral cortex or white matter) to hypoxia.
In conclusion, in these patients with chronic ICA occlusion, regional CHct was selectively decreased in the centrum semiovale ipsilateral to the occlusion. Although the significance of this decrease in Hct is unclear, it adds evidence for the difference between the effects of ICA occlusion on the regional cerebral hemodynamics in the centrum semiovale and in the cerebral cortex. The decrease in the Hct limited to the centrum semiovale with low CBF and increased OEF may reflect the susceptibility of this white matter region to reduction in perfusion pressure caused by ICA occlusion, which might be related to the high incidence of centrum semiovale infarct in ICA occlusion.
Footnotes
Acknowledgements
The authors thank Prof. Junji Konishi, MD, PhD, and his staff of the Department of Radiology and Nuclear Medicine, Faculty of Medicine, Kyoto University, Kyoto, Japan, for their support and technical help.
