Abstract
The aim of this study was to investigate the involvement of endothelins (ET) in brain injury. The effect of ET was studied in the isolated basilar artery (BA) taken from control, sham-operated, and cold-lesioned rats. Cold lesion was induced by application of a precooled (−78°C) copper cylinder (outer diameter 5 mm) for 60 seconds to the intact dura over the parietal cortex. After precontraction with prostaglandin (PG) F2α, ET-3 (10−10 to 10−8 mol/L) dilated BA with a pD2 (negative log of the half-maximal concentration) of 9.06 ± 0.031 (mean ± SD) and a maximal effect (Emax) of 1.64 ± 1.0 mN at 3 × 10−9 mol/L in sham-operated animals. This dilation was reduced 24 and 48 hours after cold lesion by 33% and 73%, respectively, at 3 × 10−9 mol/L. The effects of acetylcholine (10−8 to 10−4 mol/L) and sodium nitroprusside (10−3 mol/L) were unaltered. Activation of the ETB receptor in thoracic aorta by the specific agonist IRL 1620 also resulted in a reduced dilation (51% by 48 hours after cold lesion). Reverse transcriptase-polymerase chain reaction of the BA showed unaltered expression of mRNA for the ETB receptor after cold lesion whereas ETB immunoreactivity in BA and in its intraparenchymal arteries was reduced at 24 and 48 hours. In contrast to the reduction of ET-3–induced dilation, the constrictor effects of ET-1 and ET-3 were retained after cold lesion. Endothelin-1 (10−12 to 10−6 mol/L) dose-dependently contracted segments of untreated control BA segments under resting conditions with a pD2 of 8.03 ± 0.22 and an Emax of 6.35 ± 0.70 mN. Further evidence that the constrictor ability of BA was not influenced by cold lesion is given by the unaltered response to 124 mmol/L K+ and 10−6 mol/L serotonin. We conclude that the ETB receptor of BA after cold lesion is downregulated specifically, apparently at the posttranscriptional level. Because the ETB-mediated dilation in thoracic aorta was also reduced, downregulation of the ETB receptor apparently is not restricted to cerebral arteries. The nitric oxide-cyclic guanosine monophosphate system in BA is, however, intact.
Keywords
Endothelin-1 (ET-1) was discovered by Yanagisawa et al. (1988) as a 21-amino acid vasoconstrictor polypeptide. ET-1, together with ET-2 and ET-3, is a member of a highly homologous peptide family (Doherty, 1992). The ET can exert both constrictor and dilator effects.
These opposite actions have been attributed to the existence of different ET receptors. The ETA receptor has high affinity for ET-1 and ET-2 but low affinity for ET-3, and the ETB receptor is nonselective in this respect (Douglas et al., 1994). Stimulation of ETA receptors, which are located on vascular smooth muscle, causes vasoconstriction (Adner et al., 1993; Salom et al., 1993, Feger et al., 1994). This vasomotor reaction is mediated by activation of phospholipase C and protein kinase C (Ferrer et al., 1992; Murray et al., 1992; Görlach et al., 1998). Endothelin-A receptors may also be present on brain capillary endothelial cells (Stanimirovic et al., 1994). Endothelin-B receptors on endothelial cells of arteries mediate vasodilation (De Nucci et al., 1988; Namiki et al., 1992; Karaki et al., 1993), although the presence of a contraction-mediating ETB2 receptor subtype on smooth muscle has been demonstrated (Sudjarwo et al., 1993; Fukuroda et al., 1994). The relaxant ETB receptor is coupled to nitric oxide (NO) production in cerebral endothelial cells (Schilling et al., 1995; Kitazono et al., 1995).
Endothelin-1 applied to the adventitial side of cerebral arteries evokes vasospasm, resulting in cerebral ischemia and neuronal damage (Sharkey et al., 1993; Macrae et al., 1993). Furthermore, the cerebral tissue concentration of ET-1 is increased after brain injury or ischemia (Barone et al., 1994; Yamada et al., 1995; McKenzie et al., 1995; Salzmann et al., 1996). Endothelin-1 might be a mediator of secondary brain damage because of its high potency and extremely long-lasting action in small and large brain vessels. Because Doppler sonography studies in humans after neurotrauma have demonstrated the most pronounced flow alterations in the vertebrobasilar circulation (Hadani et al., 1997), and because changes in the diameter of the basilar artery (BA) strongly influence cerebral blood flow in the rat (Fujii et al., 1991), we investigated the vasomotor response of the rat BA to ET. Because of the existence of the ET-NO network (Ehrenreich and Schilling, 1995), we also examined whether the NO system was functionally altered in BA, with a focus on ET-3–induced dilation. Because we found that neurotrauma reduces or abolishes the vasodilator response to ET-3 in BA, we investigated by reverse transcriptase-polymerase chain reaction (RT-PCR) and immunohistochemistry whether the expression of the ETB receptor mRNA or protein is changed after cold lesion.
MATERIALS AND METHODS
Animal preparation and experimental protocol
The investigation conformed with the “Guide for the Care and Use of Laboratory Animals” published by the National Institutes of Health (NIH publication no. 85-23, revised 1985). Three groups of animals were investigated: untreated control, sham-operated, and cold-lesioned animals. In the lesioned group, a cold lesion was induced to the left cortex after trepanation between the lambda suture and the bregma in male Wistar-Kyoto rats (280 to 320 g). Rats were anesthetized with 6.9 mg/kg xylazine (Rompun, Bayer, Leverkusen, Germany) and 30 mg/kg ketamine (Ketanest, Parke Davis, Berlin, Germany), except those used for immunohistochemistry. These rats were anesthetized with 1 mL/100 g 3.6% chloralhydrate solution. To produce a cold lesion, a copper cylinder (outer diameter 5 mm), precooled to –78°C by acetone and dry ice, was impressed to a depth of 1 mm onto the intact dura for 60 seconds with the aid of a micromanipulator. After suturing the skin, the wound was treated with lidocaine spray (Xylocaine, Astra, Wedel, Germany). The animals were killed under deep ether or xylazine—ketamine or chloralhydrate anesthesia at 16, 24, 40, or 48 hours after the cold lesion. The mode of anesthesia does not influence the data obtained. This follows from the demonstration of similar vasoreactivity of the vessels (see below). The sham-operated animals were treated as the lesioned ones, except that the copper cylinder was not cooled.
Ring segments obtained from 20 rats were prepared for measurement of isometric force as described by Feger et al. (1994). After removing the brain, the BA and the thoracic aorta were dissected under a binocular microscope. The circumference and the diameter (300 to 360 µm) are very similar in all portions of the BA. Each BA was cut into four ring segments of about 1.5-mm length and transferred into 5-mL organ baths filled with a modified Krebs-Högenstätt solution (in mmol/L: NaCl, 119; KCl, 4.6; NaH2PO4, 1.2; CaCl2-2H2O, 1.5; MgCl2-6H2O, 1.2; NaHCO3, 15; glucose, 10). The bath solution was bubbled continuously with humidified 90% O2 and 10% CO2 to give a pH of approximately 7.30 at 37°C. The ring segments were mounted on two L-shaped stainless steel wires (70-µm diameter), one of which was mounted rigidly to the bath and the second connected to a force transducer. Development of isometric force was measured by a transducer coupled to an amplifier (Q11 transducers and QT233 amplifier; Hottinger Baldwin, Darmstadt, Germany) and displayed on a pen chart recorder (BD 101, Kipp & Zonen, Solingen, Germany), The transducers were mounted on a microdrive (M33, Märzhäuser, Wetzlar, Germany) to set the resting tension at 2.5 to 3.5 mN in BA and at 6 to 7.5 mN in aorta.
After mounting, all segments were allowed a 90-minute accommodation period during which the segments were washed repeatedly every 15 minutes, the resting tension was set, and the bath temperature increased to 37°C. A reference contraction was obtained in all segments by exchanging the bath solution for a 124 mmol/L K+-Krebs' solution (NaCl replaced by KCl). The functional state of the endothelium was tested by cumulative application of acetylcholine (ACh, 10−8 to 10−4 mol/L) after precontraction with 10−6 mol/L 5-hydroxytryptamine (5-HT) in BA or 10−5 mol/L 5-HT in aorta. According to our previous classification (Schilling et al., 1995), relaxation to ACh exceeding 25% of precontraction was taken to indicate the presence of a functionally intact endothelial layer. Segments that did not meet this criterion or in BA that gave reference contractions less than 2.5 mN were discarded. To test the relaxant response independent of the endothelium, sodium nitroprusside (SNP, 10−3 mol/L) was applied.
The relaxant action of ET-3 was studied by cumulative application to BA after precontraction with prostaglandin F2α (PGF2α, 3 × 10−6 or 10−5 mol/L). In BA some tachyphylaxis has been described (Kitazono et al., 1995), but construction of one concentration-effect curve was possible. In some segments of aorta the relaxant action of the ETB receptor agonist IRL 1620 (10−8 mol/L) and ET-3 (10−8 mol/L) was investigated. Because of desensitization of the ETB receptor in aorta segments the vasomotor effect of only one individual concentration of the drug was tested in each segment (Moritoki et al., 1993).
The contractile action of ET-1 was studied under resting tension conditions. Only one concentration-effect curve was constructed in each segment of BA because of the tendency of BA to develop tachyphylaxis to ET-1 (Feger et al., 1994). To study whether the blood vessels are under tone from endogenously released endothelins, the specific ETA receptor antagonist BQ 123 (10−12 to 3 × 10−5 mol/L) was applied under resting conditions.
Total RNA isolation
For each sample (obtained at 48 hours from sham-operated and lesioned animals), BA from 6 rats were pooled for RNA isolation by the CsCl-gradient method (Sambrook et al., 1989).
Reverse transcriptase-polymerase chain reaction
First-strand cDNA synthesis was carried out in a reaction volume of 20 µL containing 1 µg of total RNA, 1 µg random hexanucleotides (pd(N)6) (Pharmacia Biotech Inc., Piscataway, NJ, U.S.A.), 4 µL 5 × first-strand buffer (delivered with Maloney murine leukemia virus reverse transcriptase [M-MLV RT] from Gibco Life Technologies, Gaithersburg, MD, U.S.A.), 1 µL dNTP (solution of 20 mmol/L each, prepared from the Pharmacia Ultrapure Kit (Pharmacia), 2 µL 100 mmol/L dithiothreitol (delivered with M-MLV RT), 1 µL dd H2O, 20 U of RNAsin (Promega, Madison, WI, U.S.A.), and 150 U of M-MLV RT (Gibco). After 10 minutes' incubation of the total RNA with the random hexanucleotides at 65°C, the reaction was allowed to proceed at 37°C for 55 minutes and was terminated by boiling for 5 minutes. After this, each tube was brought to a final volume of 100 µL by adding 80 µL dd H2O.
Polymerase chain reaction of the cDNA
A 5-µL aliquot of cDNA was added at 92°C (hot start) to the polymerase chain reaction (PCR) mixture in a total volume of 50 µL containing the following components: 5 µL 10×−100 Taq extender reaction buffer (200 mmol/L Tris-HCl, 100 mmol/L (NH4)2SO4, 20 mmol/L MgSO4 1% triton X-100, 1 mg/mL nuclease-free bovine serum albumin), 2.5 U Taq extender (Stratagene), 0.5 µL dNTP (20 mmol/L each, Pharmacia), 2.5 U Taq DNA polymerase (Gibco), and 20 pmol of each primer pair. The PCR was carried out using a Perkin Elmer thermal cycler programmed for 32 cycles. The initial template denaturation was conducted at 92°C for 4 minutes and was followed by primer annealing at 62°C for 1 minute, primer extension at 12°C for 1 minute, and denaturation at 92°C for 1 minute. At the end of the 32 cycles, the reaction was kept at 12°C for 5 minutes and then brought to room temperature. A 20-µL aliquot of the PCR product was electrophoretically analyzed on a 1.5% agarose gel containing ethidium bromide.
Primers
The primer sequences for ET-1 were as follows: CCC AGC ACA TCC TGG AGA (sense) and CTC CAG CAG CTG CTG ATA (antisense) yielding a product of 378 bp. For the ETA receptor the sequences were as follows: TAC AGA GCA GTG GCT TCC (sense) and TCT GTT GAG CAT CTC ACA (antisense) with a product size of 308 bp; for the ETB receptor the primer sequences were as follows: GAC AGA TAT CGA GCT GTT GCT TCT (sense) and TTC ACA CCT CTG AGG ATT GCT CTG (antisense) with a product size of 486 bp; and for lamin (as a housekeeping gene): ATT GAG TAT GAG TAC AAG CTG (sense) and CGC ATC TCT CTC TCT TTG TC (antisense) with a product size of 335 bp.
Immunohistochemistry
Animals (n = 15) were perfused in situ with 2% paraformaldehyde for 3 minutes followed by 3 minutes with 18% sucrose in phosphate-buffered saline (PBS). The brain was removed, quickly frozen, and stored at –70°C until it was sectioned. Coronal sections (18 µm) of the cerebellum and pons—medulla were cut in a cryostat (Jung CM3000, Leica Instruments, Nussloch, Germany). Four to six sections were mounted on an aminopropyltriethoxysilane-coated slide with about 450-µm distance between the sections. Immunohistochemistry was performed as described by Sairanen and coworkers (1997). In brief, the slides were postfixed for 30 minutes in 4% formaldehyde in PBS and rinsed quickly with PBS. To enhance the penetration of the antibodies, the sections were incubated in 0.3% Triton X-100–PBS for 20 minutes and rinsed in PBS. After blocking endogenous peroxidase with 30 minutes' incubation in 1% hydrogen peroxide (H2O2) in 0.3% Triton X-100–PBS, the slides were washed twice for 10 minutes in PBS, and blocked with 10% normal sera–0.3% Triton X-100–PBS for 1 hour at room temperature. Polyclonal sheep anti-ETB (Alexis Biochemicals, San Diego, CA, U.S.A.) was used as the primary antibody. The antibody was diluted with 0.3% Triton X-100−2% blocking sera—PBS to a concentration of 1: 1,000. The sections were incubated with the primary antibody 24 hours at 4°C and then washed twice for 10 minutes in PBS. Amplification of the signal was carried out using the avidin-biotin method with the appropriate peroxidase kit (Vector Laboratories, Inc., Burlingame, Ca, U.S.A.) and visualized by 4 minutes' incubation in 6 mg diaminobenzidine (Sigma Chemical Co, St. Louis, MO, U.S.A.) in 10 mL volume of 0.003% H2O2 in 0.1 mol/L Tris-HCl (pH 7.6). After a rinse with PBS the slides were mounted with Eukitt (O. Kindler GmbH & Co., Freiburg, Germany) and a coverslip was applied for investigation under a light microscope.
Compounds used in organ bath studies
Endothelin-1, ET-3, and IRL-1620 (suc[Glu9, Ala11,15]- endothelin 1(8–21)) were obtained from Alexis (Läufelfingen, Switzerland); SNP, ACh, and 5-HT (as the creatinine sulfate) from Sigma (Deisenhofen, Germany); and PGF2α from ICN (Eschwege, Germany). BQ 123 (cyclo[
Statistical analysis
For each experiment, the maximal relaxant or contractile effect (Emax) was determined and the pD2 value calculated as the –log10EC50 (i.e., that concentration at which the half-maximal effect occurred). The pD2 was determined for individual concentration—effect curves by linear regression after logarithmic transformation of the concentration less than and greater than the EC50. Data were analyzed by one-way analysis of variance (ANOVA) followed by Tukey's test for multiple comparisons of mean values or by Kruskal-Wallis analysis of variance followed by Dunn's test. P < 0.05 was considered significant. All values in the text and in the figures are given as mean ± SD with n indicating the number of observations.
RESULTS
Influence of the parietal cortical cold lesion on ET-3–induced vasomotor response
After precontraction with PGF2α, ET-3 relaxed BA at concentrations of 10−10 to 10−8 mol/L with a pD2 of 9.06 ± 0.31 and an Emax of 1.64 ± 1.0 mN at 3 × 10−9 mol/L in sham-operated animals. There was no difference in the response to ET-3 between sham-operated and control animals (not shown). At higher concentrations (5 × 10−8 and 10−7 mol/L) ET-3 induced contractions with an Emax of 2.01 ± 1.49 mN in sham-operated animals that was statistically not different from the lesioned animals (Fig. 1). The relaxation was reduced 24 hours after cold lesion by 33%, and 48 hours after cold lesion by 73% at 3 × 10−9 mol/L (Fig. 1). Acetylcholine was applied to test whether this resulted from a lack of ability of BA to release NO. Acetylcholine has been shown to elicit relaxation of the rat BA by release of NO from the vascular endothelium (Mackert et al., 1997). As seen in Fig. 2 and Table 1, the ACh (10−8 to 10−4 mol/L)-induced relaxation in ring segments of BA was not impaired at any time after cold lesion. This demonstrates an intact NO—cyclic guanosine monophosphate (cGMP) system after cold lesion. A further indication of an intact gua-nylate cyclase—cGMP system is the fact that the relaxation effect of SNP in BA was not altered at any time after cold lesion (Table 1).
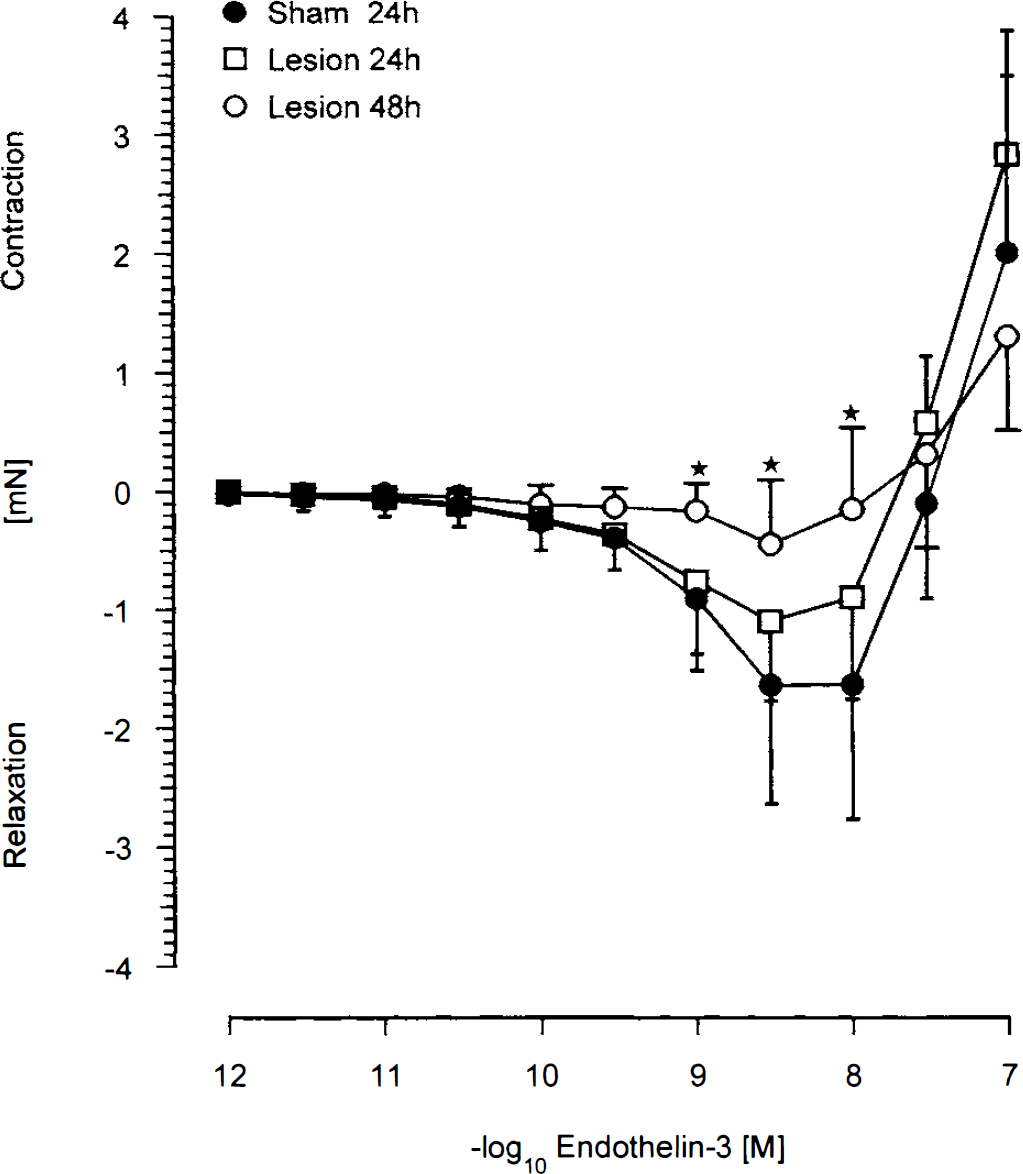
Concentration—effect curves for endothelin-3 in basilar artery (BA) segments obtained after 24 or 48 hours from sham-operated or cold-lesioned animals. Given are means ± SD of 11 to 13 observations. *, P < 0.05 versus sham.
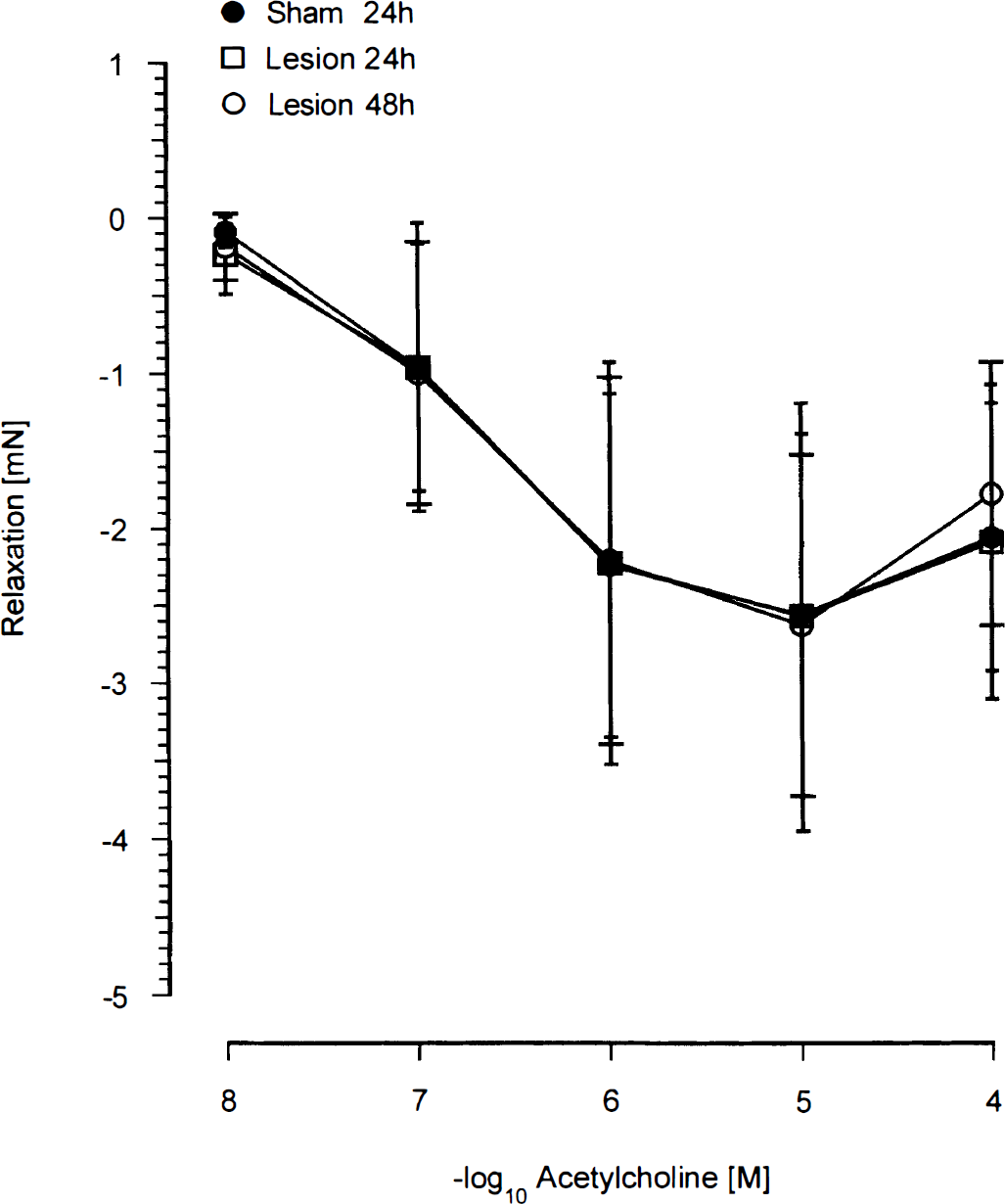
Concentration—effect curves for acetylcholine (ACh) in BA segments obtained after 24 or 48 hours from sham-operated or cold-lesioned animals. Given are means ± SD of 12 to 40 observations.
Vascular reactivity to various agonists
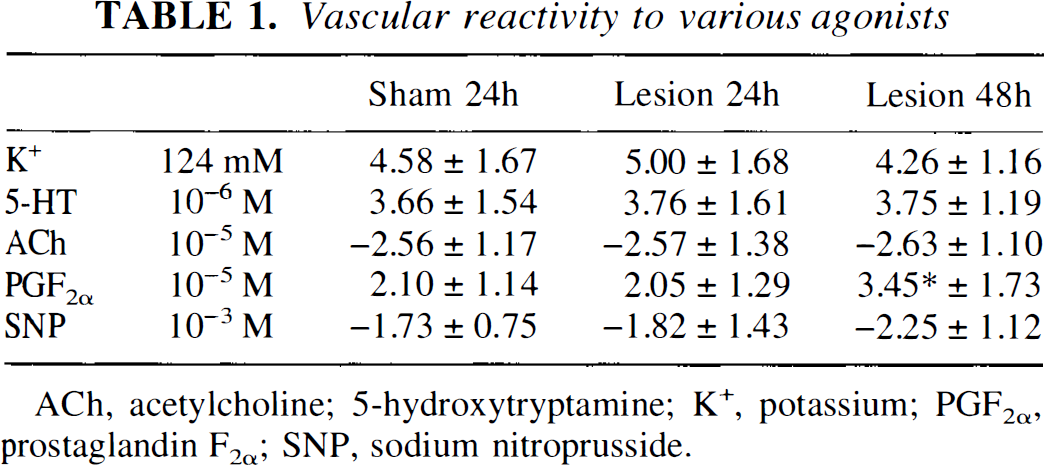
ACh, acetylcholine; 5-hydroxytryptamine; K+, potassium; PGF2α, prostaglandin F2α; SNP, sodium nitroprusside.
To test whether the loss of vasorelaxation by the ETB receptor after cold lesion was specific for BA we investigated the relaxing effect of IRL 1620, a specific ETB receptor agonist, in ring segments of rat thoracic aorta. Earlier studies from our laboratory have shown that, in the rat BA, IRL 1620 is a pure ETB receptor agonist that mimics only the relaxant, but not the contractile, response to ET-3 (Schilling et al., 1995). In PGF2α-precontracted rat thoracic aorta ring segments of sham-treated animals, IRL 1620 induced a relaxation of 2.91 ± 1.92 mN. Forty-eight hours after cold lesion, IRL 1620 (10−8 mol/L) elicited only a weak relaxation of 1.41 ± 1.27 mN in PGF2α-precontracted rat thoracic aorta (Fig. 3). Corresponding results (not shown) were obtained with ET-3 in rat thoracic aorta. As already shown for BA the relaxation caused by ACh (10−8 to 10−5 mol/L) was the same in thoracic aorta after cold lesion compared with the sham-operated status, demonstrating an intact NO—cGMP system. In addition, the contractile effect of 10−5 mol/L 5-HT was not altered in this vessel after cold lesion (data not shown).
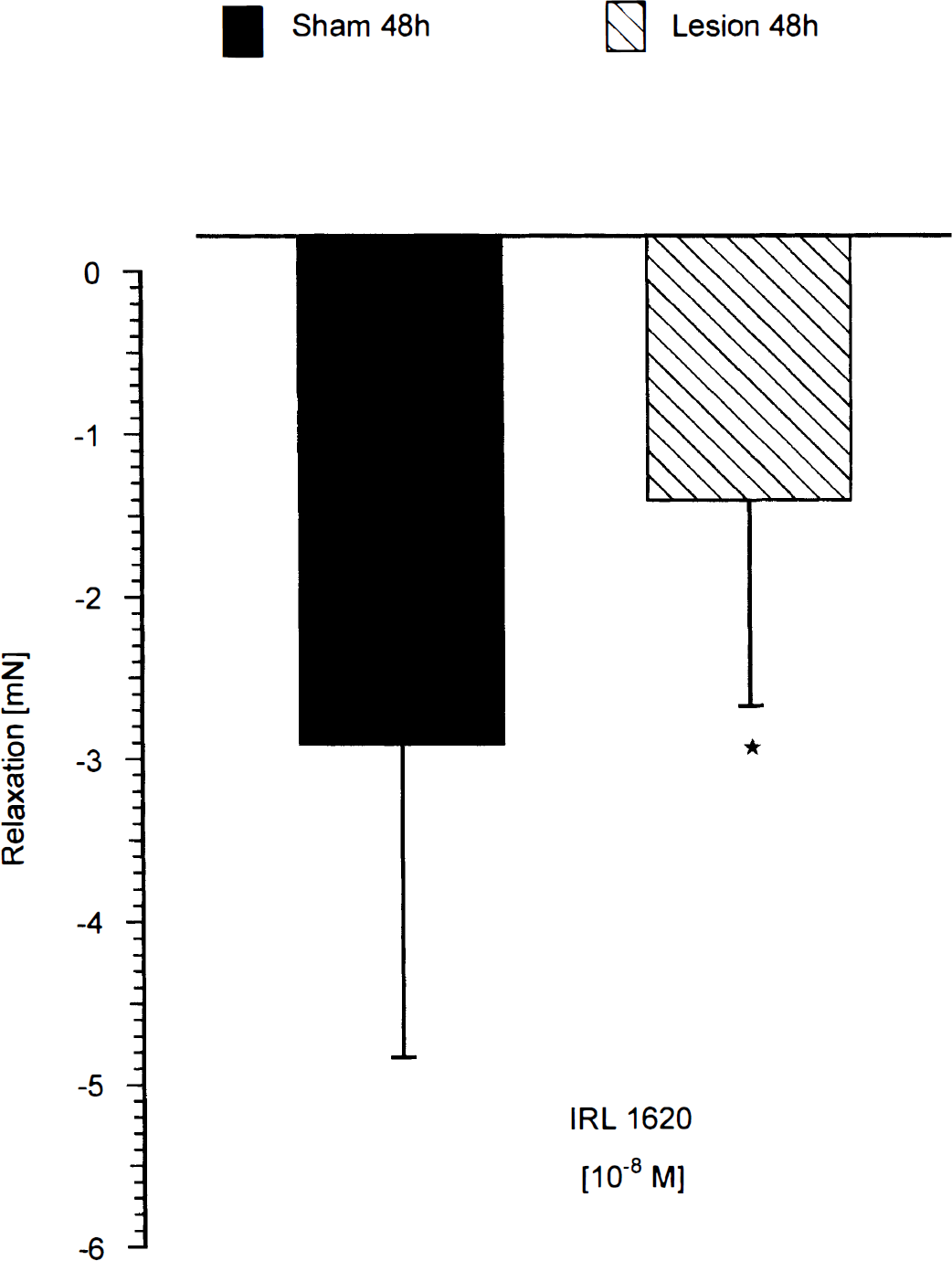
Effect of the ETB agonist IRL 1620 (10−8 mol/L) in segments of thoracic aorta obtained after 48 hours from sham-operated and cold-lesioned animals. Given are means ± SD of 13 to 27 observations. *, P < 0.05 versus sham.
Influence of parietal cortical cold lesion on ET-1–induced contraction
Administration of the selective ETA receptor antagonist BQ 123 (10−12 to 3 × 10−5 mol/L) to BA from control or lesioned animals did not induce any vasomotor effects, indicating that the tone of the vessels is not influenced by endogenously released ET acting on ETA receptors. Application of ET-1 (10−12 to 10−6 mol/L) resulted in a dose-dependent contraction under resting conditions in BA ring segments of untreated controls. The pD2 was 8.03 ± 0.22 and Emax was 6.35 ± 0.70 mN. In segments obtained 16 and 40 hours after cold lesion, ET-1 elicited the same contraction as in controls (Fig. 4). Moreover, cold lesion in the parietal cortex does not change the response to a variety of contractile agents. This is shown by the same effect caused by 124 mmol/L K+, 10−6 mol/L 5-HT, or 10−5 mol/L PGF2α as in segments of sham-operated animals (Table 1). However, the value for PGF2α obtained 48 hours after cold lesion was statistically different from control.
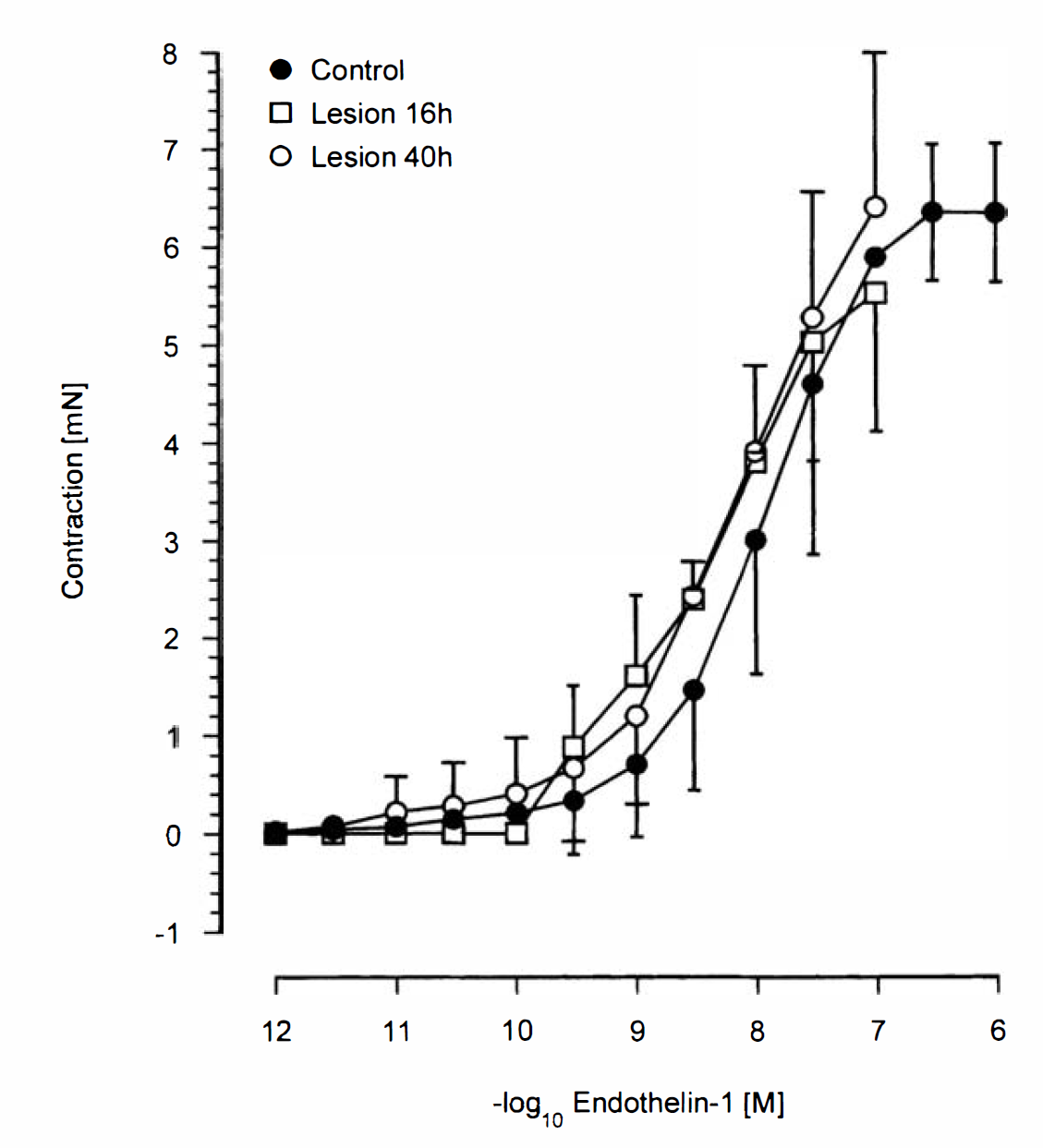
Concentration-effect curves for endothelin-1 in BA segments obtained from untreated controls or animals 16 and 40 hours after cold lesion. Given are means ± SD of 6 to 37 observations.
Influence of parietal cortical cold lesion on expression of ETB receptors in the BA
To explore further the impairment of the ETB receptor-mediated vasodilation after neurotrauma, RNA was isolated from the BA and analyzed by RT-PCR. As shown in Fig. 5, the mRNA for ET-1, ETA, and ETB are constitutively expressed in the BA of both sham-operated and lesioned animals, indicating that the alterations in ETB receptor expression were more likely to occur at the posttranscriptional level. Indeed, in the sham-operated animals, immunoreactivity for ETB receptor was found in the BA and in its intraparenchymal blood vessels of the brain stem, whereas 24 and 48 hours after cortical cold lesion ETB immunoreactivity in the BA and in the brain stem intraparenchymal vasculature was reduced (Fig. 6).
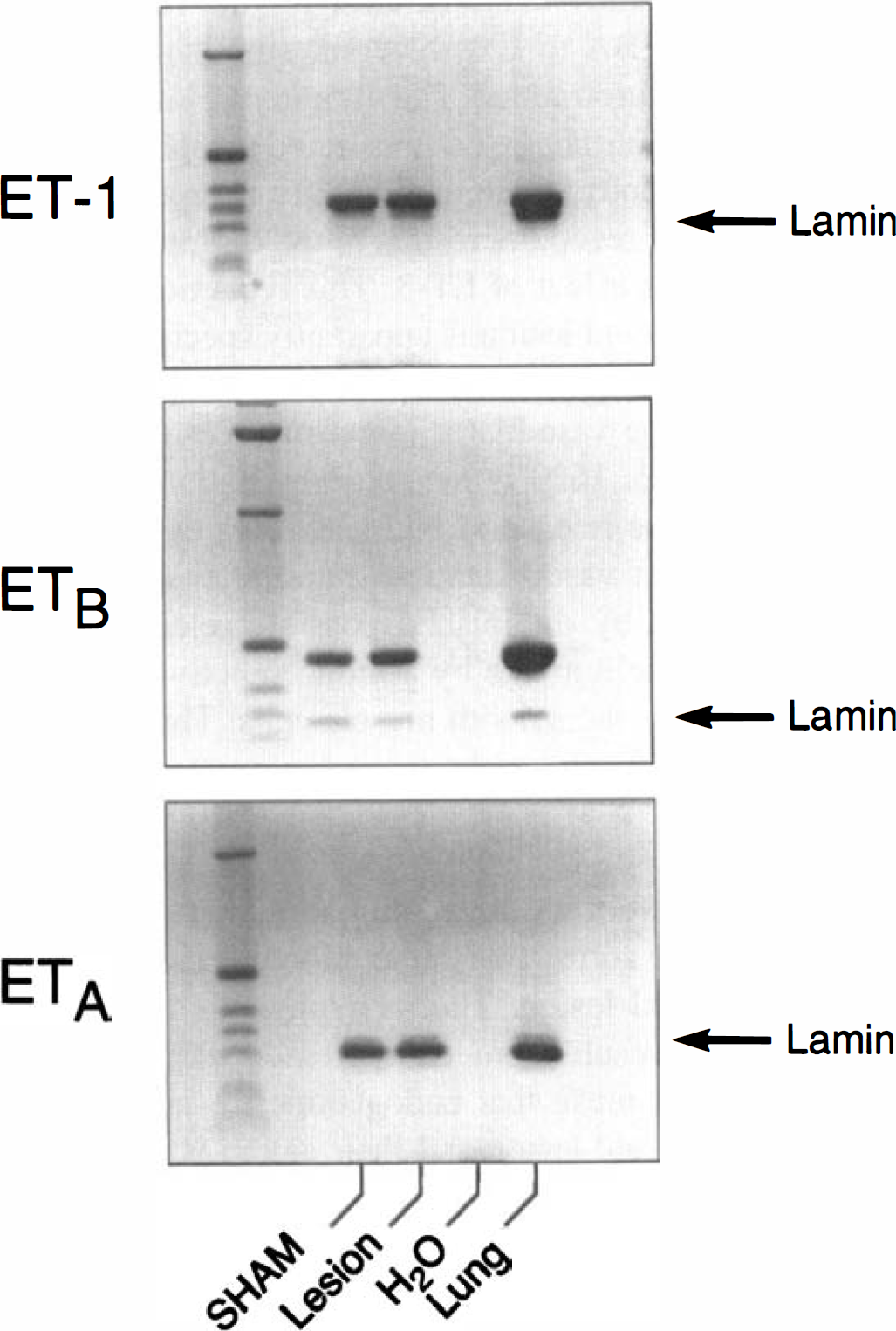
Reverse transcriptase-polymerase chain reaction analysis of gene expression of ET-1, ETB, and ETA receptors in BA of sham-treated and cold-lesioned rats after 48 hours.
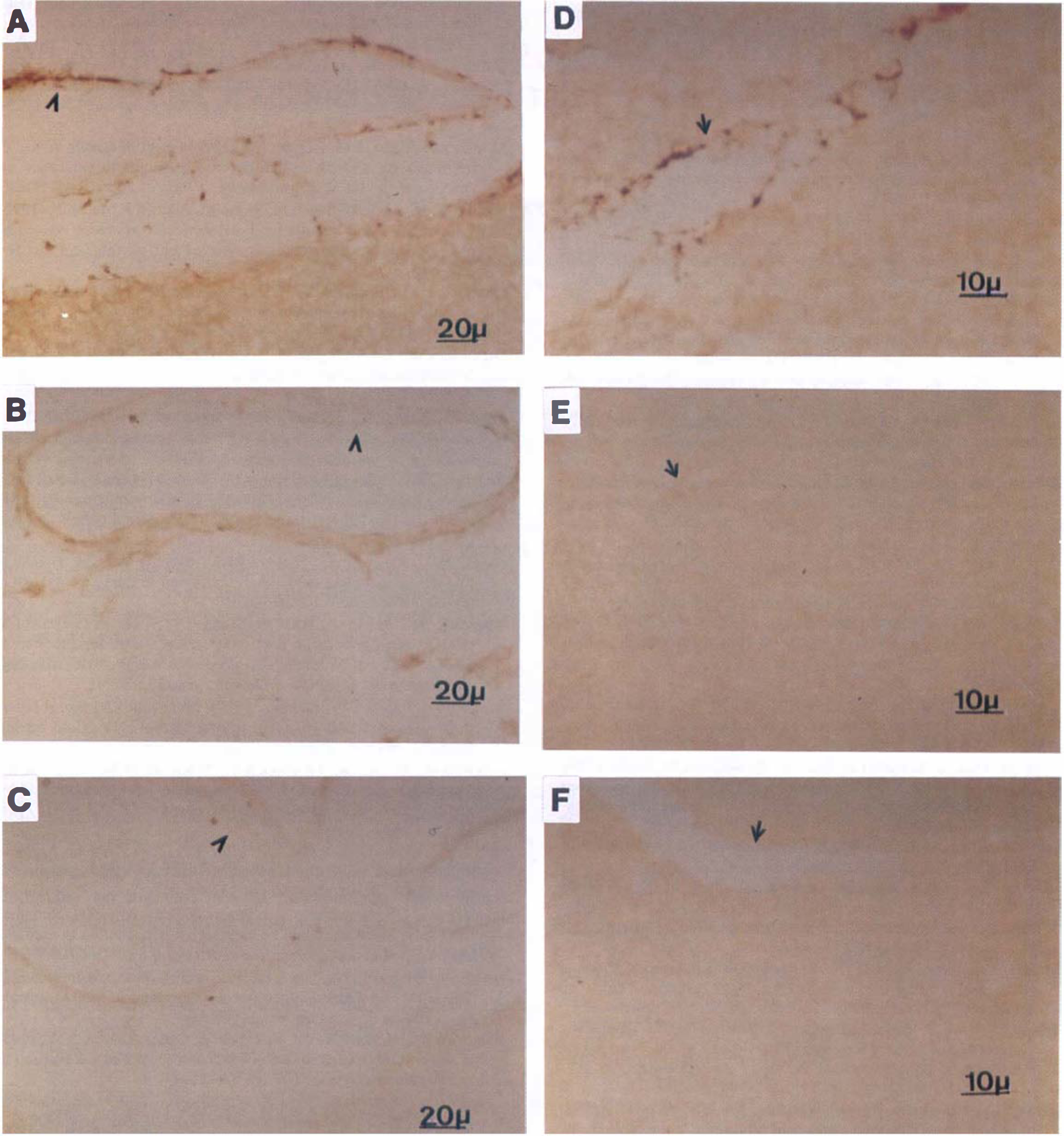
Expression of immunoreactivity (ir) for ETB receptors (brown reaction product) in sham-operated rats
DISCUSSION
The present study demonstrates, for the first time, a delayed loss of BA to ETB receptor stimulation after a standardized neurotrauma. The functional lack of ETB receptors could further be demonstrated morphologically by reduced antibody staining of the ETB receptor protein. The loss of ETB receptors results in the disappearance of the vasorelaxing effect of ET-3. This reduction of vasorelaxation after cold lesion is apparently specific for ETB receptors. Previous work from our laboratory has demonstrated that the vasodilator effect of ET-3 or the specific agonist IRL 1620 is mediated by endothelial ETB receptors and the release of NO (Schilling et al., 1995). The reduction of vasorelaxation found after cold lesion was not caused by diminished possible release of NO from the endothelium nor by a reduced activity of gua-nylate cyclase in the smooth muscle cells. This was also evident from the unaltered vasomotor response to ACh and SNP.
Development of tachyphylaxis may explain the delayed impairment of vasorelaxation to ET-3 or IRL 1620 in the BA and aorta, two large blood vessels remote from the cortical lesion. The tachyphylaxis of ETB receptors might result from an increased expression of ET. This would mean that endogenous ET are released during or after cold lesion and their action is weakened. Tissue ET-1 was increased 1 to 5 days after cold lesion in the rat (Yamada et al., 1995), and high plasma levels of ET were found in humans after stroke or subarachnoid hemorrhage (Ziv et al., 1992; Ehrenreich et al., 1992). Moreover, we have found that tachyphylaxis to the dilating component of the ET-3 response develops in the BA of normal animals during repeated application of cumulative concentration—effect curve (Feger, Schilling, Wahl; unpublished observations). Tachyphylaxis was also found in situ using microapplication of the same dose of the specific ETB agonist BQ 3020 to feline pial arteries. Furthermore, a loss of ETB receptor-mediated vasodilation also occurs after focal ischemia. In this study the microapplication of BQ 3020 elicited the same response as the artificial CSF, its solvent (Touzani et al., 1997).
In the aorta, the relaxing effect of IRL 1620 was reduced by 51% after cold lesion. The delayed reduction of ETB-mediated vasorelaxation in both BA and aorta after cold lesion might indicate a systemic reduction in the relaxant capacity of the ETB receptors, perhaps related to the multiorgan dysfunction that is characteristic of human neurotrauma (Ott et al., 1994; Nuytinck et al., 1987).
The ETB receptor mRNA expression in the BA was not altered 48 hours after the neurotrauma, at a time point when the functional impairment had reached its maximum, rendering the possibility of a downregulation of the ETB receptor at the transcriptional level very unlikely. In agreement with the loss of ETB-mediated vasorelaxation in BA 24 and 48 hours after cortical cold lesion, however, we found a delayed reduction of ETB receptor immunoreactivity in BA and in brain stem intraparenchymal vessels, which coincides with the functional alterations in BA reactivity to ET-3. These data demonstrate that neurotrauma induces delayed changes in the cerebrovascular ETB receptors that may be part of a generalized arterial dysfunction increasing the risk of late vasospasm and ischemia.
Cold injury to the exposed brain through the intact dura was used as the model for brain injury (Klatzo et al., 1958). Although the freezing injury is an artificial insult to the brain compared with the complexity of traumatic damage in humans, the model is highly standardized and yields reproducible focal disruption of the blood—brain barrier (Stoffel et al., 1997). The resulting brain edema leads to secondary brain damage, thus increasing the necrosis caused by the primary insult (Eriskat et al., 1994). In this respect, this experimental approach mimics the pathophysiology of human traumatic head injury (Klatzo et al., 1958; Wahl et al., 1988).
In this model of a cold lesion in the parietal cortex, the vascular tone of the BA was apparently not influenced by a component of the endogenous ET system. This is evident from the fact that BQ 123, a specific ETA antagonist, had no effect per se in the isolated BA after cold lesion and under control conditions. Similarly, BQ 788, a specific ETB antagonist, has no effect per se in the isolated BA under control conditions (Feger, Schilling, Wahl; unpublished observations), a finding also obtained by Touzani et al. (1997) in feline pial arteries. Because the ETB receptors were downregulated after cold lesion their contribution to the resting tone of BA could not be investigated. The data showing no effect of an ETA antagonist are in apparent contradiction to a study of Armstead (1996). This author used the model of a fluid percussion injury in the newborn pig and found that local application of BQ 123 diminished the constriction of pial arteries. Similarly, in the cat model of a focal cerebral ischemia, Patel et al. (1996) found that perivascular application of ET antagonists dilates pial arteries. The apparent discrepancy between these studies and the present one may be explained by the different time point of measurement, experimental design, species, and size and location of the artery under investigation: the BA, as a large cerebral artery, is remote from the primary cold lesion in the parietal cortex. We found not only an unaltered contractile response of the BA to ET-1 and ET-3, but also to other contractile agents, e.g., 124 mmol/L K+ and 5-HT, in lesioned and sham-operated animals. In addition, a normal relaxant response to ACh or SNP demonstrates an intact vasomotor responsiveness of this vessel except to ETB receptor stimulation.
In conclusion, after cold lesion we found reduced relaxation of the BA to ET-3, which is specific for the ETB receptor. The NO—cGMP system is intact. Because we noted an impairment of the ETB-mediated relaxation also in aorta, downregulation of ETB receptors appears to be a more general phenomenon. This downregulation of ETB receptors occurs obviously at the posttranscriptional level as immunoreactivity in BA was reduced whereas ETB receptor mRNA was expressed after cold lesion.
Footnotes
Acknowledgments
The authors thank Mrs. E. Held for expert secretarial help, and Mrs. J. Specker and Mrs. H. Kamrowski-Kruck for excellent technical assistance.
