Abstract
Two types of acid—base strategies are available for the blood gas management of patients during hypothermia: alpha-stat and pH-stat management However, the more suitable strategy for therapeutic hypothermia is unclear. We studied the effects of hypothermia (30°C) and acid—base management on reactivity to hypercapnia and hypotension in rat pial arterioles, using a closed cranial window, The baseline diameter during hypothermia decreased in the alpha-stat (Pa
Mild to moderate hypothermia is being clinically applied not only as an intraoperative means of preventing cerebral ischemia, but also as a therapeutic modality for treating severe head injury (Marion et al., 1993) and cerebral embolism (unpublished data), Studies have shown that decreasing brain temperature by only a few degrees markedly reduces infarction volume in experimental models (Busto et al., 1987; Minamisawa et al., 1990).
We believe that understanding the alterations in cerebrovascular reactivity induced by hypothermia is key to maximizing the benefits and minimizing the adverse effects of this treatment under various pathophysiologic conditions.
Two different strategies for acid—base management, pH-stat and alpha-stat management, during hypothermia have been proposed as a method for intraoperative management of cardiac surgery (Swain, 1988). In alpha-stat management, arterial CO2 tension is maintained at 40 mm Hg when measured at 37°C. The dissociation fraction of the imidazole moiety of histidine is kept constant, and the pH changes during hypothermia almost parallel the changes in the neutral pH of water. Enzymatic, cellular, and organ function are well preserved in the alpha-stat condition. In pH-stat management, arterial CO2 tension is maintained at 40 mm Hg when corrected to the patient's temperature. The pH-stat strategy requires CO2 addition to the inspired gas or controlled hypoventilation. Several studies have yielded contradictory conclusions about cerebral autoregulation and hypercapnic response in relation to acid—base management under hypothermic conditions (Lundar et al., 1985; Murkin et al., 1987; Greeley et al., 1989; Rogers et al., 1992; Cheng et al., 1995). However, all except one (Verhaegen et al., 1993) of these studies involved cardiopulmonary bypass (CPB) under moderate to deep hypothermia. The characteristics of cerebrovascular physiology and optimal management of the acid—base balance and Pa
We therefore investigated the influence of moderate hypothermia (30°C) managed by alpha-stat or pH-stat strategies on pial arteriolar responses to hypercapnia and hypotension in this study.
MATERIALS AND METHODS
Male Sprague-Dawley rats (350 to 400 g) were anesthetized by an intraperitoneal injection of α-chloralose (50 mg/kg) and urethane (750 mg/kg), both of which were supplemented as necessary during the procedure. The femoral arteries on both sides were cannulated to monitor blood pressure and to withdraw blood to induce hypotension. The rats were given a tracheostomy and mechanically ventilated. End-tidal CO2 was continuously monitored using a CO2 analyzer (Respina IH31, San-ei, Tokyo, Japan) and maintained at appropriate levels. Arterial blood pH and gases were measured with a pH/blood gas analyzer (168 pH blood gas system, Coming, Medfield, MA, U.S.A.). Rectal temperature was maintained at 37°C.
A closed cranial window was constructed as described previously (Morii et al., 1986) over the right parietal cortex. Pial microvessels were then observed in situ using a microscope (Nikon, Tokyo, Japan) fitted with a video camera system (Series CCD C2400 system, Hamamatsu Photonics, Hamamatsu, Japan). Briefly, the head of the rat was fixed in a frame and three polyethylene tubes were embedded in a ring of dental acrylic applied to the skull to suffuse the window with artificial CSF and to monitor intracranial pressure. A thermosensor was also embedded in the acrylic to monitor temperature in the window. Craniectomy (5 × 4 mm) was then performed over the right parietal cortex (1 to 6 mm posterior to the bregma and 1 to 5 mm lateral to the midline) in the center of the acrylic, and the dura—arachnoid complex was incised while the pial surface was superfused with artificial CSF. A glass coverslip was placed on the acrylic ring and sealed with cyanoacrylate. The pial arteriolar diameter was continuously measured using a video dimension analyzer (Model C1170, Hamamatsu Photonics).
Experimental protocol
After obtaining a stable baseline diameter, the pial arteriolar response to 5% CO2 inhalation for 5 minutes was recorded. The rats were then randomly divided into normothermic (n = 7), alpha-stat (n = 8), and pH-stat (n = 7) groups. Rats in the alpha-stat and pH-stat groups were cooled for about 30 minutes to reduce the temperature at both the rectum and the cranial window to 30°C. End-tidal CO2, monitored continuously, decreased as the temperature was reduced.
Alpha-stat management was achieved without changing the ventilation during cooling whereas in pH-stat management, tidal volume and respiratory rate had to be decreased to maintain constant end-tidal CO2, Both types of acid—base management were confirmed by blood gas analysis.
Hypothermia was maintained for 60 minutes and then the baseline diameter was measured again. A rectal temperature of 37°C was maintained for the same period in the normothermic group. Carbon dioxide reactivity was reexamined, and the femoral artery was opened into a saline-filled reservoir connected to a mercury manometer to maintain mean arterial pressure (MAP) at desired levels. The MAP was lowered in increments of 10 mm Hg by withdrawing blood into the reservoir, and MAP was maintained at each level for 2 to 4 minutes to record vessel diameter.
Statistical analysis
Statistical significance was tested using two-way repeated measures analysis of variance for comparison among the three groups, and the contrast method for comparison was used between two groups. P < 0.05 was considered significant.
RESULTS
Physiologic parameters and CO2 reactivities (dilation [%]/Pa
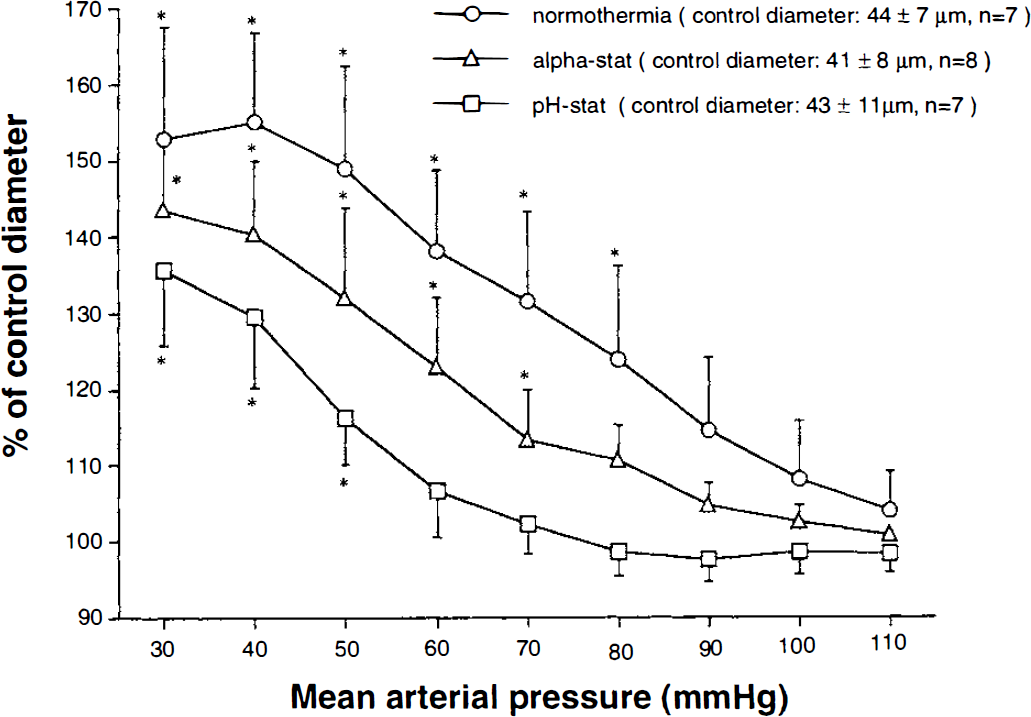
Changes in the pial arteriolar diameter were plotted as a function of changes in mean arterial pressure. Vasodilation associated with induced hypotension in hypothermic groups was reduced compared with that of the normothermic group (P < 0.01). The degree of attenuation in hypotensive vasodilation was greater under conditions of pH-stat than alpha-stat management (P < 0.01). Data represent the mean ± SD. *P < 0.05 compared with baseline diameter by one-way analysis of variance followed by Bonferroni/Dunn test.
Physiologic parameters and CO2 reactivity
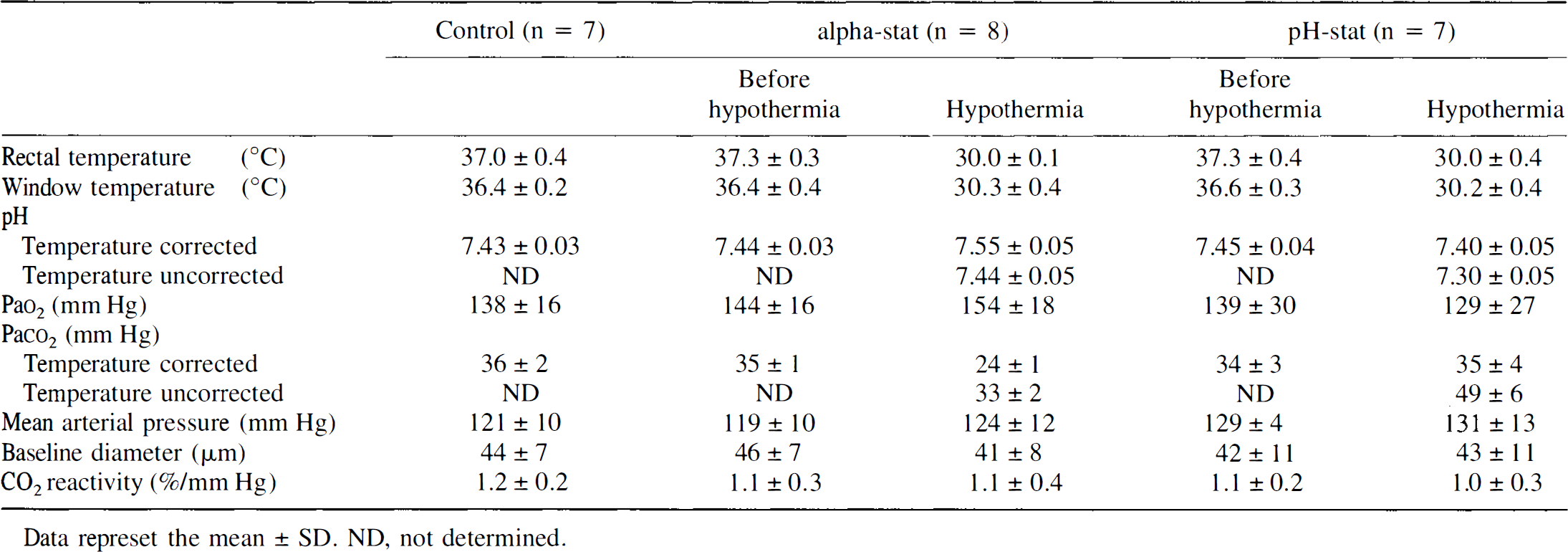
Data represet the mean ± SD. ND, not determined.
DISCUSSION
Autoregulation is the intrinsic ability to maintain constant blood flow despite changes in blood pressure. At the level of individual vessels, cerebral blood flow is autoregulated by arteriolar dilation in response to a fall in perfusion pressure. We examined autoregulation under moderate hypothermia using a closed cranial window that allowed direct observation of pial arterioles in situ under temperature-controlled conditions. We found that pial arteriolar dilation in the face of hypotension is significantly reduced under hypothermia.
The acid—base balance during hypothermia was maintained by alpha-stat and pH-stat management as described previously (Swain, 1988). There has been considerable controversy over which procedure is physiologically more suitable for nonhibernating homeotherms and more effectively prevents cerebral damage after cardiac surgery (Stephan et al., 1992; Kern and Greeley, 1995; Burrows, 1995). In the present study, although ventilation was decreased during cooling and temperature-uncorrected Pa
Pial arteriolar dilation in the face of hypotension was reduced under hypothermia although hypercapnic vasodilation was preserved in this study. Thus, we believe that impairment was specifically induced by faulty autoregulation, and not by nonspecific decline of cerebrovascular reactivity. In addition, the degree of impaired autoregulation was greater in the pH-stat, than in the alpha-stat, condition, These results were consistent with the findings from a study of the effects of moderate hypothermia and acid-base management on cerebral autoregulation under nonbypass conditions, using laser—Doppler flowmetry (Verhaegen et al., 1993).
However, when interpreting these results, the following points should be considered, First, hypotension was induced by withdrawing blood, which is associated with increases in vasoactive substances such as catecholamines, which may in turn affect pial arteriolar reactivity, Whether there is any difference in release of vasoactive compounds during induced hypotension between the normothermic and hypothermic conditions is unknown. Second, hypotensive vasodilation may have shifted toward the lower absolute levels of MAP in the hypothermic groups, However, it is unlikely that further dilation (maximal dilation) occurred below an MAP of 20 mm Hg in the hypothermic groups, which is beyond the MAP levels we examined, and thus we believe that hypotensive vasodilation was reduced, not simply shifted, in the hypothermic groups.
There are several possible mechanisms involved in altered autoregulation. The attenuation of cerebral metabolism induced by hypothermia, resulting in reduced tissue oxygen demands, may result in reduced hypotensive vasodilation, An oxygen-sensitive mechanism for cerebral blood flow autoregulation has been proposed in which tissue hypoxia would be responsible for arteriolar dilation through the liberation of vasodilator substances (Kontos and Wei, 1985). The stimulus for vasodilation (tissue hypoxia) may be relatively blunt in the hypothermic brain in which cerebral metabolic rates for oxygen are markedly decreased (approximately 65% of normal at 30°C according to Hägerdal et al., 1975), resulting in reduced production of mediators for vasodilation.
Another possible mechanism involves an increase in blood viscosity induced by hypothermia (Chen and Chien, 1978). Change in blood viscosity is an important factor for regulation of cerebral blood flow (Muizelaar et al., 1986) by changes in shear stress to the endothelium, with accompanying changes in release of endothelium-derived relaxing factor. This may contribute to alteration in cerebral blood flow autoregulation.
The involvement of other potential mediators of altered autoregulation, including adenosine (Berne et al., 1974; Kontos and Wei, 1985), calcitonin gene-related peptide (Lee et al., 1993), parasympathetic nerves (Koketsu et al., 1992), and nitric oxide (Tanaka et al., 1993), should also be considered. However, these mechanisms may not completely explain why autoregulation is more profoundly altered under pH-stat conditions as shown by this study and those of others (Murkin et al., 1987; Verhaegen et al., 1993). When considering this issue, two possibilities need to be taken into account, if the oxygen-dependent mechanism is assumed to play a major role in hypotensive vasodilation.
First, the baseline diameter changes seen in the present study (i.e., the pial arterioles constricted in the alpha-stat group whereas the baseline diameter did not change in the pH-stat group) suggest that the pH-stat condition is one of relative hyperemia for the reduced cerebral metabolism during hypothermia compared with the alpha-stat condition. We speculate that the increase in tissue oxygen demands in response to induced hypotension are relatively small in a brain with relative hyperemia under pH-stat conditions. Second, should the CMRO2 be lower in the pH-stat condition than in the alpha-stat condition? The finding that Pa
We demonstrated that moderate hypothermia and acid—base management alters the autoregulatory response, although the mechanism is not yet fully understood. The contribution of altered cerebrovascular reactivity to neuroprotection is currently under investigation.
Footnotes
Acknowledgment
The authors thank Shigeyoshi Maruyama for his technical assistance.
