Abstract
Recent
Cytochrome c, a water-soluble peripheral membrane protein of the mitochondria, is known to be an essential component of the mitochondrial respiratory chain (Boyer et al., 1977). Its function is to transport electrons from the coenzyme QH2–cytochrome c reductase complex to the cytochrome c oxidase complex in the electron transport chain. Recently, it has been suggested that cytochrome c participates in apoptosis in a cell-free system (Liu et al., 1996). Mitochondria are assumed to be involved in apoptosis by releasing cytochrome c to the cytoplasm where it activates caspase 3 (CPP32), a molecule of the interleukin-1β-converting enzyme (ICE) family, which has been shown to then trigger apoptosis (Liu et al., 1996). Consistent with this finding, microinjection of cytochrome c has been shown to result in apoptosis (Li et al., 1997). More recent evidence shows that the expression of Bcl-2 acts to inhibit cytochrome c translocation, thereby blocking CPP32 activation and the apoptotic process, not only in a cell-free system but also in intact cells (Kluck et al., 1997; Yang et al., 1997). However, the involvement of cytochrome c in apoptosis is totally unknown
MATERIALS AND METHODS
Focal cerebral ischemia
Adult male Sprague-Dawley rats (250 to 280 g) were subjected to transient focal ischemia by intraluminal MCA blockade with a nylon suture as described (Yang et al., 1994). The rats were anesthetized with 2.0% isoflurane in 30% oxygen and 70% nitrous oxide using a face mask. The rectal temperature was controlled at 37°C with a homeothermic blanket. Cannulation of a femoral artery allowed the monitoring of blood pressure and arterial blood gases (Table 1), samples for analysis being taken immediately after cannulation, 10 minutes after occlusion, and 10 minutes after reperfusion. After a midline skin incision, the left external carotid artery was exposed, and its branches were electrocoagulated. A 22.0-mm 3-0 surgical monofilament nylon suture, blunted at the end, was introduced into the left internal carotid artery through the external carotid artery stump. After 90 minutes of MCA occlusion, blood flow was restored by the withdrawal of the nylon suture.
Physiologic variables during and after ischemia
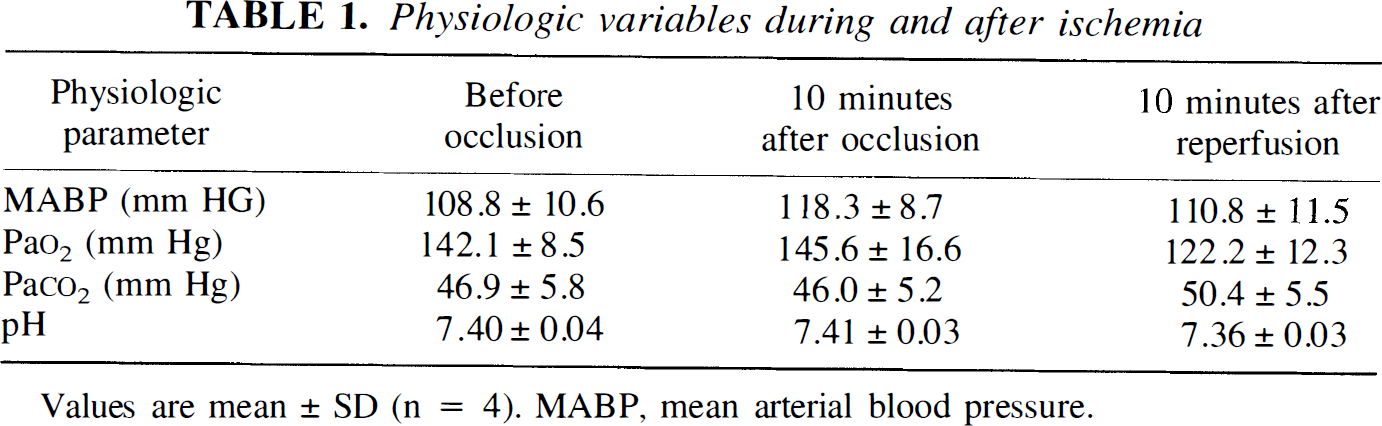
Values are mean ± SD (n = 4). MABP, mean arterial blood pressure.
In situ labeling of DNA fragmentation
At 4, 24, or 72 hours after 90 minutes of MCA occlusion (n = 4, each), the brains were removed and rapidly frozen. They were sectioned with a cryostat into a thickness of 20 µm from the anterior side to the posterior side. Brain sections at the level of the caudate putamen that showed typical infarction were stained using an
Immunohistochemistry
Anesthetized animals were perfused with 10 U/mL heparin and subsequently with 4% formaldehyde in 0.1 mol/L PBS (pH 7.4) after 4, 24, and 72 hours reperfusion after ischemia. Brains were removed, postfixed for 12 hours, sectioned at 50 µm on a vibratome, and processed for immunohistochemistry. The sections were reacted with mouse monoclonal antibody against rat cytochrome c (7H8.2C12, Pharmingen, San Diego, CA, U.S.A.) at a dilution of 1:500. Immunohistochemistry was performed using the avidin-biotin technique, and then the nuclei were counterstained with hematoxylin solution for 10 minutes. As a negative control, sections were incubated in the absence of primary antibodies. For histologic assessment, alternate slices from each brain section were stained with cresyl violet.
Western blot analysis
Protein extraction of both the mitochondrial and cytosolic fraction was performed as described (Liu et al., 1996), with modifications. Approximately 900 mg of tissue of both ischemic and nonischemic hemispheres, or alternatively, 150 mg of ischemic brain and nonischemic brain from the corresponding contralateral brain, were cut into pieces after 4, 24, and 72 hours reperfusion, homogenized gently by douncing 30 times in a glass tissue grinder (Wheaton, Millville, NJ, U.S.A.) in 7 volumes of cold suspension buffer (20 mmol/L HEPES-KOH [pH 7.5], 250 mmol/L sucrose, 10 mmol/L KCl, 1.5 mmol/L MgCl2, 1 mmol/L EDTA, 1 mmol/L EGTA, 1 mmol/L dithiothreitol, 0.1 mmol/L phenylmethylsulfonyl fluoride, 2 µg/mL aprotinin, 10 µg/mL leupeptin, 5 µg/mL pepstatin, and 12.5 µg/mL N-acetyl-leu-leu-norleucinal). The homogenates were first centrifuged at 750
RESULTS
Physiologic data
All rats survived the transient MCA occlusion. There were no significant differences in blood pressure, blood gas, and pH among groups (Table 1).
DNA fragmentation was detected after transient MCA occlusion
To elucidate whether DNA fragmentation is induced in 90 minutes of MCA occlusion and subsequent reperfusion, we examined
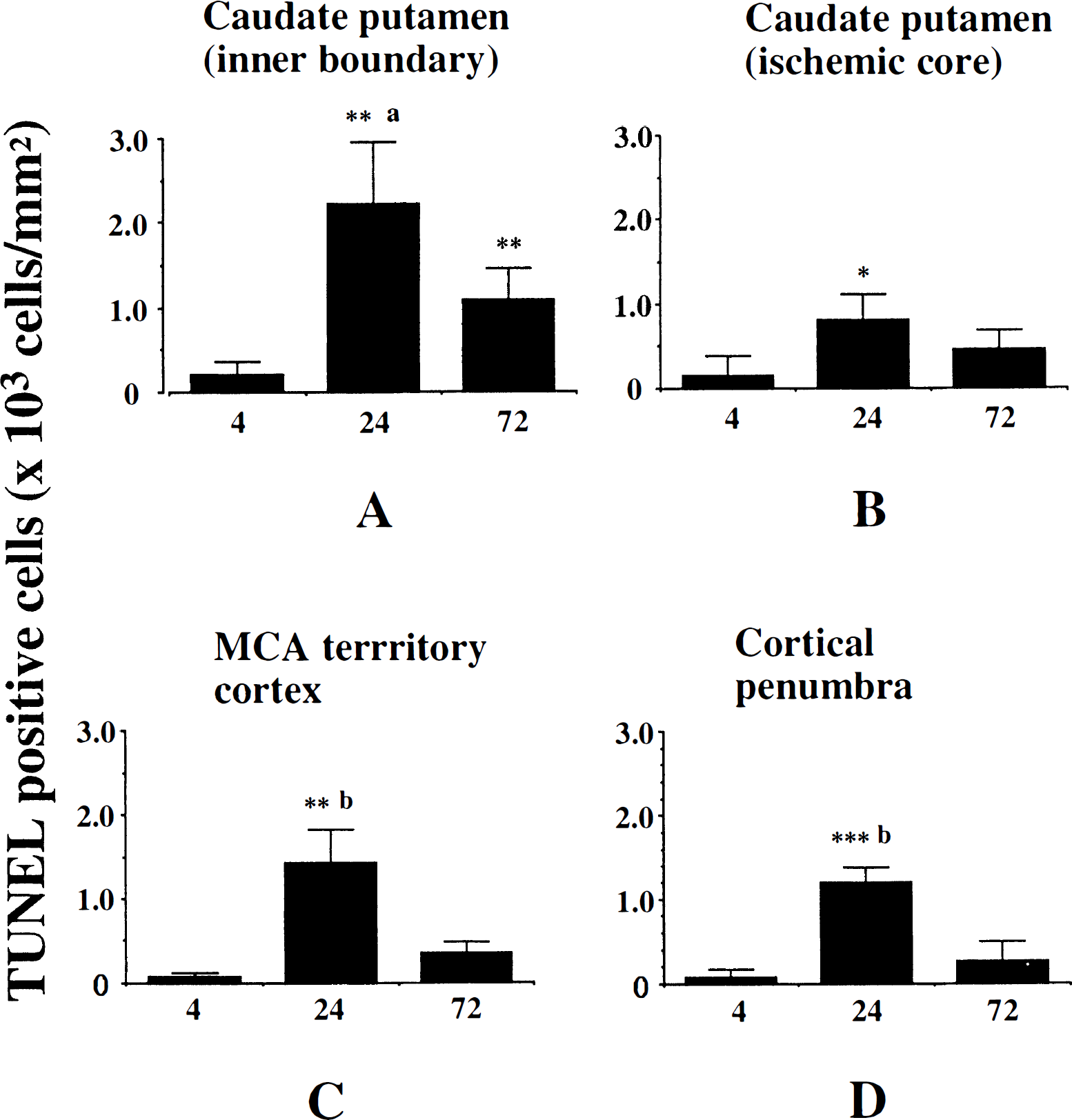
Temporal change of the number of TUNEL-positive cells in various brain regions.
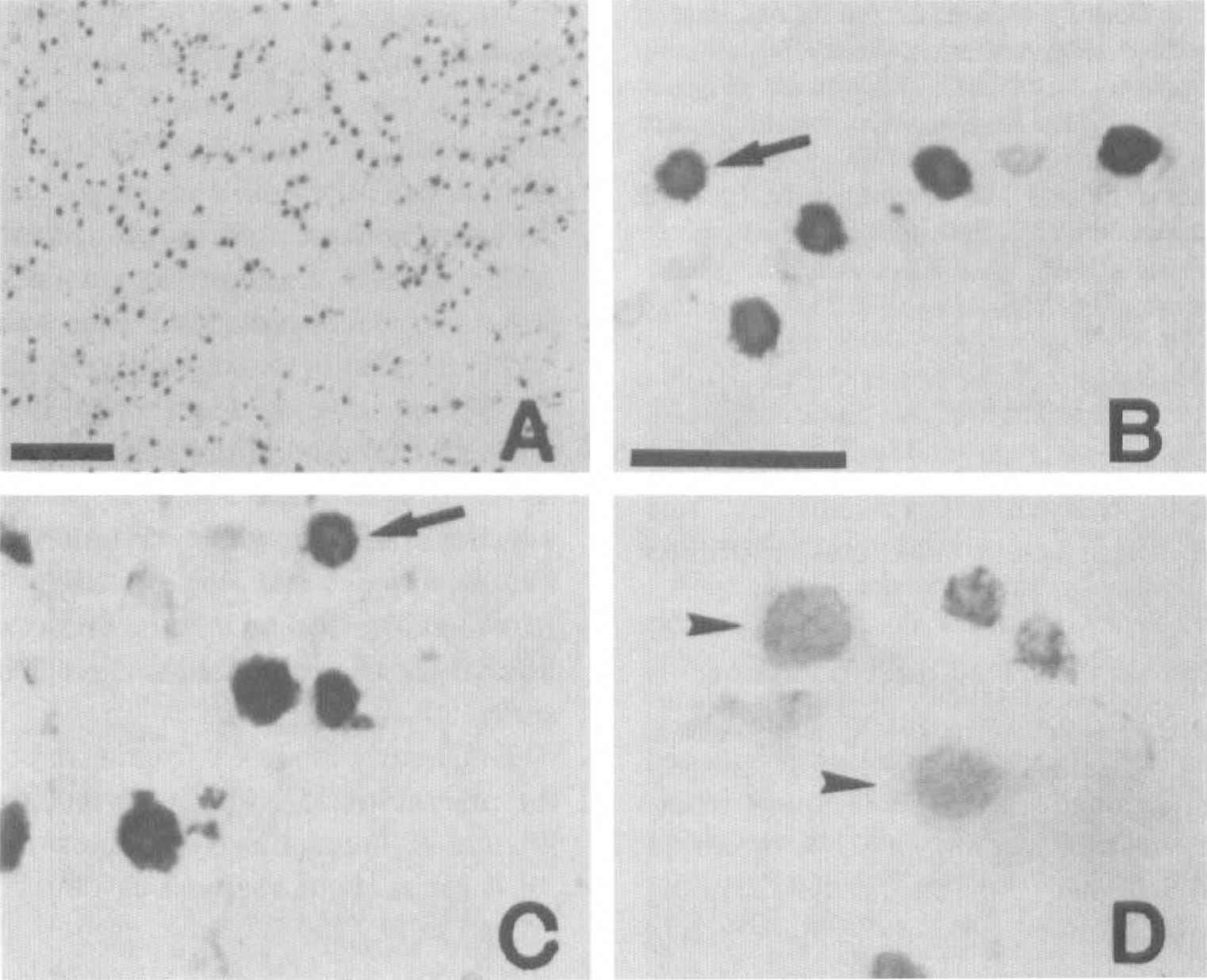
Photomicrographs showing TUNEL-positive cells in the inner boundary of the caudate putamen (
To quantify the DNA-fragmented cells after ischemia, the number of TUNEL-positive cells was counted at 4, 24, and 72 hours after 90 minutes of MCA occlusion (n = 4 each). Four regions, as defined previously (Murakami et al., 1997a), were used to assess the anatomic distribution of TUNEL-positive cells (inner boundary of caudate putamen, ischemic core of caudate putamen-lateral caudate putamen, MCA territory cortex, and cortical penumbra). The number of TUNEL-positive cells in each region was counted in a high-powered field (×400) by a investigator who was blinded to the studies, and expressed as number/mm2. Results were expressed as mean ± standard deviation. The statistical significance of differences between the time points in each group was evaluated by analysis of variance. A
Cytoplasmic expression of cytochrome c was detected in ischemic brain after transient MCA occlusion
The cresyl violet stain was used to determine the area of the infarcted tissue in the frozen coronal sections. The infarcted area apparently appeared at the caudate putamen and piriform cortex as early as 4 hours after reperfusion, and extended to the MCA territory cortex 24 to 72 hours after reperfusion (data not shown).
Cytochrome c protein expression after 90 minutes of transient focal ischemia and subsequent reperfusion was analyzed by immunohistochemistry. The data of the cytoplasmic cytochrome c expression are summarized in Table 2. Homogeneous cytoplasmic immunoreactivity of cytochrome c was visible as early as 4 hours after reperfusion in the whole ischemic area (Fig. 3B). After 24 hours of reperfusion, the population of immunoreactive cells was increased, and the intensity of cytoplasmic immunostaining was significantly enhanced with a slight background, compared with that observed after 4 hours of reperfusion (Fig. 3D). More immunoreactive cells were observed in the core of the caudate putamen and MCA territory cortex than in the inner caudate putamen and cortical penumbra. At 24 hours, we observed several cells presenting nuclear fragmentation, one of the characteristic features of apoptosis, as shown by hematoxylin counterstaining (Fig. 3E). However, no remarkable correlation between these cells and immunoreactive cells was shown in the present study. After 72 hours of reperfusion, most of the cells had been destroyed and the background immunoreactivity was increased (Fig. 3F). There was no immunoreactivity in the contralateral hemisphere (Fig. 3C) nor in the control specimens, which were treated without primary antibody (Fig. 3A). The absence of immunoreactivity in the contralateral hemisphere is considered to be caused by the thorough fixation of the brain by formaldehyde, which prevents the antibody from reaching the mitochondrial intermembrane space, but not the cytosol. In fact, immunohistochemistry with frozen sections results in dotted cytosolic immunoreactivity of cytochrome c in control brain (data not shown).
Summary of cytoplasmic cytochrome c protein expression after 90 minutes MCA occlusion
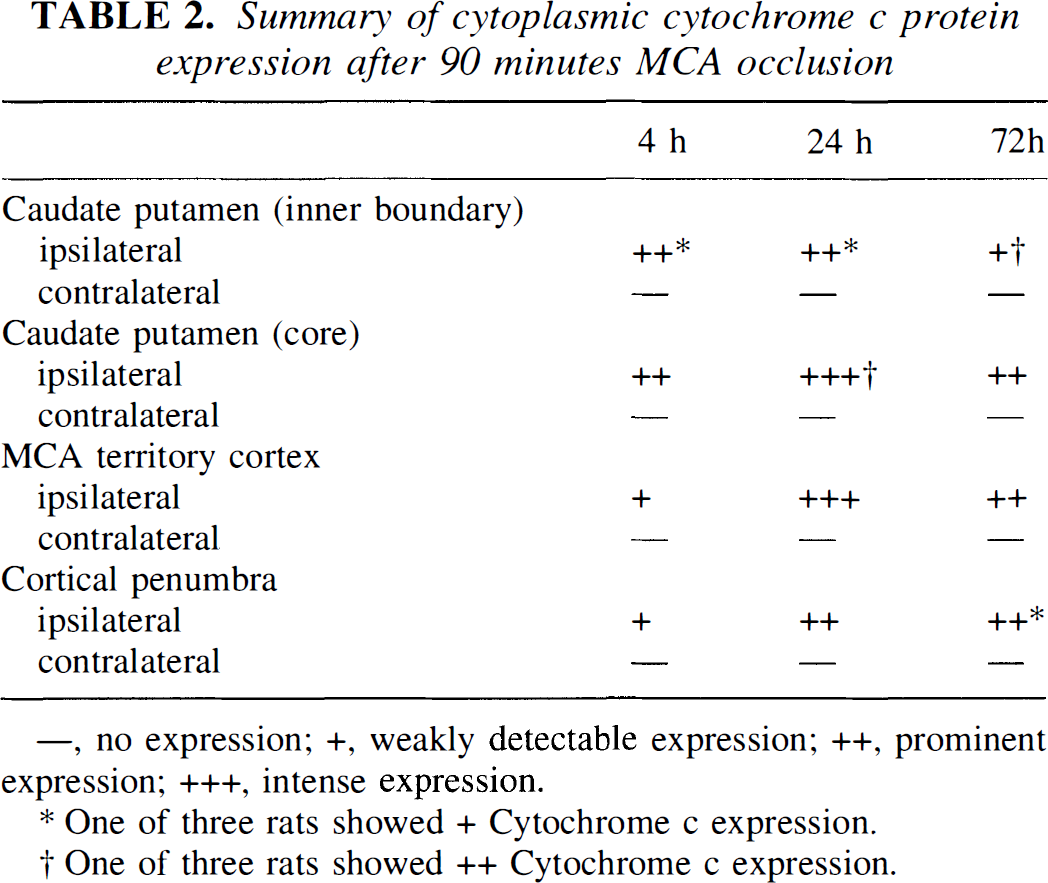
—, no expression; +, weakly detectable expression; ++, prominent expression; +++, intense expression.
One of three rats showed + Cytochrome c expression.
One of three rats showed ++ Cytochrome c expression.
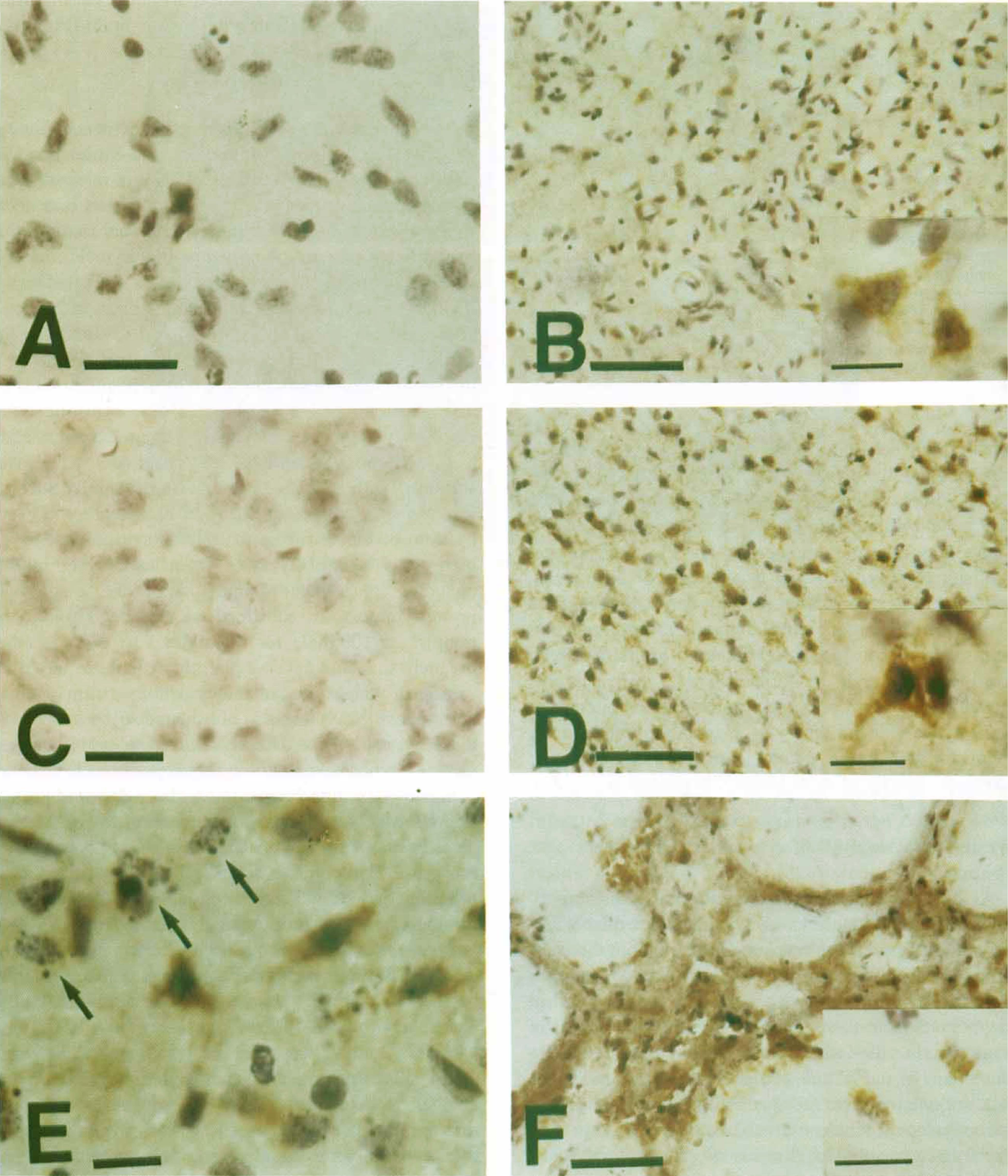
Cytochrome c immunostaining with hematoxylin counterstaining in coronal brain sections from rats after 4 and 24 hours of reperfusion after 90 minutes of ischemia.
Western blot analysis of cytosolic cytochrome c expression
As shown in Fig. 4A, cytochrome c immunoreactivity was evident as a single 15-kDa band, detected by Western blot analysis of the cytosolic fraction from the ischemic hemisphere, as early as 4 hours after 90 minutes MCA occlusion (lane 3), whereas there was no band in the nonischemic sample (lane 2). The characteristic single band in the ischemic sample was sustained at 24 hours (lane 5) and 72 hours (lane 7) after ischemia. These data not only confirm the specificity of the monoclonal antibody for cytochrome c used in this study, but also show that cytosolic localization of cytochrome c was significantly increased after transient ischemia. Furthermore, a significant amount of mitochondrial cytochrome c was detected by Western blot in nonischemic brain (mean optical density, 8.71; n = 2), which was decreased in ischemic brain after 24 hours ischemia (mean optical density, 5.83; n = 2). Correspondingly, the cytosolic fraction from the same sample showed a marked increase of cytochrome c in ischemic brain (Fig. 4B). However, cytochrome oxidase was strongly expressed in the mitochondrial fraction, but not in the cytosolic fraction of both ischemic and nonischemic brain (Fig. 4B).
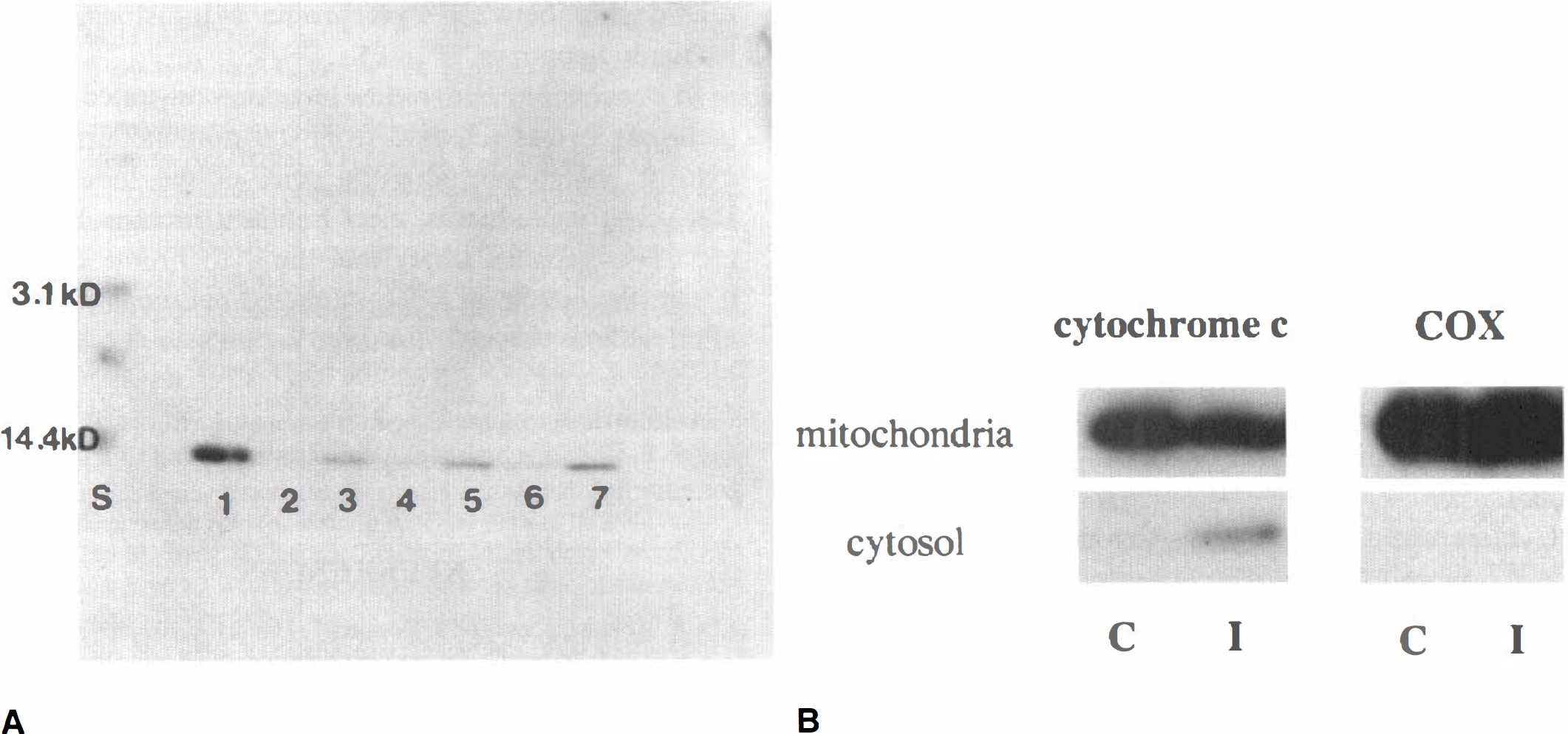
Western blot analysis of cytochrome c and cytochrome oxidase.
DISCUSSION
The current study provides the first evidence that cytochrome c, an essential component of the mitochondrial respiratory chain (Boyer et al., 1977), is released from mitochondria into the cytoplasm after transient focal ischemia. This observation derives from our findings that a significant amount of mitochondrial cytochrome c is detected in nonischemic brain (mean optical density, 8.71), which is decreased in ischemic brain at 24 hours after ischemia (mean optical density, 5.83), and that the cytosolic fraction from the same samples shows a marked increase of cytochrome c in ischemic brain (Fig. 4B). Also, by immunohistochemistry, cytosolic cytochrome c was detected only in the ischemic brain as early as 4 hours after ischemia (Figs. 3 and 4A), which was sustained until 24 and 72 hours after ischemia. Such an increase in cytosolic localization of cytochrome c appears to be derived from the mitochondrial inner membrane, and is unlikely to be from some other source, including injury-induced upregulation and a defect in protein import. In addition, we ultracentrifuged the samples at 100,000
Several authors have reported that cerebral ischemia could cause impairment of mitochondrial function (Abe et al., 1995; Almeida et al., 1995; Lee, 1995). Mitochondria are also known as the site where oxygen free radicals, especially superoxide anions, are produced during cerebral ischemia (Piantadosi and Zhang, 1996), and free radicals play a major role in the expansion of neuronal injury after both focal (Kinouchi et al., 1991; Yang et al., 1994; Chan, 1996; Kondo et al., 1997; Murakami et al., 1998) and global ischemia (Murakami et al., 1997b; Kawase et al., 1997). Thus, nonspecific mitochondrial degradation such as osmotic lysis and swelling may occur after ischemia/reperfusion injury and may therefore contribute to the increase of cytosolic cytochrome c. We do not exclude this possibility completely; however, we believe that not only mitochondrial degradation but also the translocation of cytochrome c contributes to the increase of cytosolic cytochrome c for the following reasons. First, as shown by Western blot analysis in Fig. 4B, cytochrome c was decreased in the mitochondria and correspondingly increased in the cytosol after ischemia, whereas cytochrome oxidase showed no alteration of the subcellular distribution after ischemia. This result suggests the intactness of the mitochondrial fraction isolated from the ischemic brain. From this observation, we believe the decrease of mitochondrial cytochrome c may indicate that cytochrome c release from the intact mitochondria occurred after ischemia. Second, immunohistochemistry showed cytoplasmic expression of cytochrome c only in the ischemic lesion, and cytoplasmic immunoreactivity was predominantly found in the cells presenting intact morphology at 4 hours after ischemia (Fig. 3B). Third, we have recently developed the
Neuronal cell death after focal cerebral ischemia has previously been attributed to passive necrotic processes. However, increasing evidence suggests that an active process similar to programmed cell death or apoptosis may contribute to the death of neurons after focal ischemia (Linnik et al., 1993; Tominaga et al., 1993; Li et al., 1995a,b,d; Charriaut-Marlangue et al., 1996; Choi, 1996; Chopp and Li, 1996; Gillardon et al., 1996; Asahi et al., 1997). Fragmented DNA as shown by agarose gel electrophoresis was present in rat brains subjected to permanent MCA occlusion (Linnik et al., 1993; Tominaga et al., 1993). The DNA fragmentation is known to be associated with increased intranucleosomal endonuclease activity (Tominaga et al., 1993), or to be reduced by the protein synthesis inhibitor cycloheximide (Linnik et al., 1993; Du et al., 1996). Recent morphologic studies with TUNEL staining demonstrate that the inner boundary zone of the caudate putamen is vulnerable to DNA fragmentation after focal cerebral ischemia (Li et al., 1995b; Charriaut-Marlangue et al., 1996), and that the number of DNA-damaged neurons is maximized at 24 to 48 hours after ischemia (Li et al., 1995c). In the current study, we used a transient focal ischemia model in rats, which has been shown to present some evidence of neuronal apoptosis such as TUNEL-positive cells and internucleosomal DNA fragmentation (Li et al., 1995a). In fact, a significant number of TUNEL-positive cells were detected in the current study, which were maximized at 24 hours after ischemia. More DNA-damaged cells were found in the inner boundary of the caudate putamen than in the core of caudate putamen, MCA territory cortex, and cortical penumbra. These data are consistent with previous reports, as shown above, and indicate that DNA-damaged neuronal death also contributes to the expansion of the lesion in our ischemia/reperfusion injury model.
Cytochrome c has drawn particular attention because of a recent cell-free study suggesting its critical role in apoptosis (Liu et al., 1996). This recent study showed that mitochondria are involved in apoptosis by releasing cytochrome c to the cytoplasm where it activates CPP32, a molecule of the ICE family that has been shown to trigger apoptosis when activated (Liu et al., 1996). However, the involvement of cytochrome c in apoptosis has not been demonstrated
Recent evidence shows that the expression of Bcl-2 process (Kluck et al., 1997; Yang et al., 1997) or Bcl-XL (Kharbanda et al., 1997; Kim et al., 1997) prevents cells from undergoing apoptosis and acts to inhibit cytochrome c translocation to cytoplasm, thereby blocking CPP32 activation and the apoptotic process. Martinou et al. (1994) have reported that overexpression of Bcl-2 in transgenic mice protects neurons from ischemic insult, and our previous report demonstrates that Bcl-2 protein is induced in neurons in the ischemic hemisphere that survive transient focal ischemia in rats (Chen et al., 1995). Although the relationship between Bcl-2 expression and ischemic apoptosis in these studies is not clear, it is conceivable that Bcl-2 also inhibits the neuronal apoptosis of cerebral ischemia by preventing cytochrome c translocation and subsequent activation of CPP32. A cytochrome c study using Bcl-2 transgenic or knockout animals will provide evidence necessary to establish the relationship between cytochrome c translocation and ischemic apoptosis.
In conclusion, our studies have demonstrated that cytochrome c was released from the mitochondria to the cytosol, which precedes the peak of the induction of DNA-fragmented cells, after transient focal ischemia in rats. These results imply that the cytochrome c release might play a role in DNA-damaged neuronal cell death after ischemia/reperfusion injury.
Footnotes
Acknowledgments
The authors thank L. Reola, B. Calagui, and S. F. Chen for their technical assistance, and C. Christensen for editorial assistance.
