Abstract
The induction and distribution of 3-
Keywords
Spinal cord injury after a successful operation on the thoracic aorta is a disastrous complication in humans. The reported incidences of paraplegia range from 0.9% to 40% (Crawford et al., 1981, 1986) in operations on the thoracic aorta. Acute spinal cord dysfunction is believed to be caused by ischemia from hypoperfusion during cross-clamping. Ischemia can occur because of permanent exclusion of the essential intercostal arterial blood supply to the spinal cord, or from temporary interruption of blood flow to the spinal cord (Kats et al., 1981; Livesay et al., 1985). However, patients undergoing thoracic aorta aneurysm repair who awake with no neurologic deficit immediately after the operation may sometimes eventually develop paraplegia (Moore and Hollier, 1991; Matsui et al., 1994). In the rabbit spinal cord ischemia model, we have reported selective motor neuron death after transient ischemia (Sakurai et al., 1996, 1997, 1998; Watanabe et al., 1996). However the exact mechanism of such vulnerability is not fully understood. To evaluate the mechanism of the selective vulnerability of motor neurons, we attempted to make a reproducible model of spinal cord ischemia and statistically analyzed cell damage.
Recent studies have suggested that nitric oxide (NO), a potent cerebrovasodilator and inhibitor of platelet aggregation (Moncada, 1992; Iadecola, 1993), may increase cerebral blood flow in the ischemic territory and ameliorate ischemic damage (Zhang and Iadecola, 1993). On the other hand, it is well established that NO has been implicated in the development of many neurologic disorders and brain function (Lafon-Cazal et al., 1993; Kumura et al., 1995; Bindikas et al., 1996; Patel et al., 1996). Nitric oxide reacts with superoxide (O2·_) to form a potent oxidant, peroxynitrite (ONOO−). In experiments using cultured cells, ONOO− was shown to be the major cytotoxic molecule in glutamate-induced normal cell injury and apoptotic cell death (Lipton et al., 1993). Although it is difficult to measure ONOO− in tissues or organs owing to its short half-life, 3-
MATERIALS AND METHODS
Animal models
Thirty-six Japanese domesticated white rabbits weighing 2 to 3 kg were used in this study, and were divided into two groups: a 15-minute ischemia group, and a sham-operated control group. Anesthesia was induced with intramuscular administration of ketamine at a dose of 50 mg/kg and maintained with 2% halothane inhalation. A SF pediatric thermodilution catheter (405, B. Braun Melsungen A.G., Melsungen, Germany) was inserted through a femoral artery and advanced 15 cm forward into the abdominal aorta. Preliminary investigations by laparotomy had confirmed that the balloon in the distal end of the thermodilution catheter was positioned 0.5 to 1.5 cm distal to the left renal artery. During the experiment, aortic pressures were continuously monitored both at the proximal and distal positions of the balloon. Body temperature was monitored with a rectal thermistor and maintained at 37°C with a heating pad during surgery and subsequent ischemia. Animals were allowed to recover at ambient temperature, and were killed with deep sodium pentobarbital anesthesia (100 mg/kg, intravenously) at 8 hours, and 1, 2, and 7 days after reperfusion (n = 6 at each time point). In the sham-operated control group, animals were killed after insertion of the catheter into the abdominal aorta without inflating the balloon. Spinal cords were quickly removed immediately after death, using the plunger of a 1 mL syringe (Zivin et al., 1975). The tissue samples for HPLC analysis and immunohistochemical studies were frozen in powdered dry ice and stored at −80°C. The samples for histology were fixed by immersing in 4% paraformaldehyde in 0.1 mol/L phosphate buffer, and then stored at 4°C for 1 week; they were then cut transversely at about the L2 or L3 level, and finally embedded in paraffin.
During the experiment, rabbits were treated in accordance with the Declaration of Helsinki and the Guiding Principles in the Care and Use of Animals. Also, the experimental and animal care protocol was approved by the Animal Care Committee of the Tohoku University School of Medicine.
Neurologic assessment
Neurologic function was observed at 2 days and at 7 days after the procedure. Animals were classified into a five-point scale according to the method of Johnson et al. (1993): 0, hind-limb paralysis; 1, severe paraparesis; 2, functional movement, no hop; 3, ataxia, disconjugate hop; 4, minimal ataxia; 5, normal function. Two individuals without knowledge of the treatment graded the neurologic function independently.
Histologic study
The sections were stained with hematoxylin-eosin and examined by light microscopy. An observer, unaware of animal group and neurologic outcome, examined each slide. With hematoxylin-eosin staining, the cells were considered “dead” if the cytoplasm was diffusely eosinophilic, and “viable” if the cells demonstrated basophilic stippling (that is, contained Nissl substance) (Mutch et al., 1993). To determine the ischemic change, a series of animals was allowed to recover for 7 days after the sham operation (n = 6) or the 15-minute ischemia (n = 6). The spinal cords were removed, fixed by immersing in 4% paraformaldehyde in 0.1 mol/L phosphate buffer for 7 days, and then embedded in paraffin. The number of intact large motor neuron cells in the ventral gray matter region was counted.
HPLC analysis of nitro-compound
Spinal cord samples were divided into two regions: ventral region and dorsal region, through which a line demarcated the central canal perpendicular to the vertical axis. Samples were homogenized with 500 µL of distilled water and hydrolyzed as the previous report with slight modifications (Tsugita et al., 1982). Briefly, homogenized tissue was incubated at 110°C for 24 hours in a vessel lack (JASCO Ltd., Tokyo, Japan) containing 6 N HCl and 0.1% phenol, which was added to rule out artifactual formation of NO2-Tyr during acid hydrolysis in the presence of nitrite or nitrate (Shigenaga et al., 1997). Separation of NO2-Tyr was achieved by HPLC on a Nucleocil 5-µm C-18 reverse-phase column (15 cm × 4.6 mm) with a guard column (JASCO Ltd., Japan).
The column was eluted with 50 mmol/L KH2P
Immunohistochemical study
Mouse monoclonal antibody against nitrotyrosine (1:250 dilution, Upstate Biotechnology, Lake Placid, NY, U.S.A.) was used for immunohistochemical studies. Specificity of the antibody has been noted elsewhere (Beckman et al., 1994; Ferrante et al., 1997). For immunohistochemical analysis, the sections were immersed in 0.5% periodic acid for 10 minutes and blocked with 2% horse serum for 20 minutes before exposure to mouse monoclonal antibodies for 20 hours at room temperature. The biotin-streptavidin system was adopted using the Elite Vectastain ABC kit (PK-6102; Vector Laboratories, Burlingame, CA, U.S.A.). Immunoreactivity was visualized using a 5-minute exposure with 3,3'-diaminobenzidine/H2O2 (Wako, Osaka, Japan). Hematoxylin was used for nuclear staining. To ascertain specific binding of antibody for the protein, a set of sections was stained in a similar way without the first antibody.
Statistical analysis
Statistical analyses of the neurologic score, the cell numbers, and level of NO2-Tyr were performed using the Mann-Whitney U test. A P value less than 0.05 was considered statistically significant. Parametric data are present as mean ± SD.
RESULTS
When the balloon of the thermodilution catheter was inflated in the abdominal aorta, systemic blood pressure of about 70/40 mm Hg did not change. The arterial pressure distal to the inflated balloon, on the other hand, fell to near 0 mm Hg, and no pulsation was recorded. On deflation of the balloon, systemic blood pressure of this portion decreased for 15 minutes and then returned to the normal level (data not shown). Spinal cord ischemia was achieved by inflating the balloon so as to obstruct blood flow to the spinal cord (Cheng et al., 1984; Herold et al., 1994). The results obtained with HPLC, histocytochemical, and histologic studies were very reproducible in animals at each time point.
Neurologic outcome
The results are summarized in Table 1. In the sham-operation group (n = 6), all rabbits were normal (grade 5). In the 15-minute ischemia group at 2 days after the procedure (n = 6), three rabbits (50%) were normal (grade 5), two rabbits (33%) had minimal ataxia (grade 4), and one rabbit (16%) had ataxia (grade 3). In the 15-minute ischemia group at 7 days after the procedure (n = 6), four rabbits (66%) did not hop (grade 2), and two rabbits (33%) had ataxia (grade 3). There was a significant difference between the Johnson's neurologic scores at 2 days and at 7 days after the procedure in the 15-minute ischemia group (P = 0.0074). This difference was similar between the sham-operated control group and the 15-minute ischemia group at 7 days after the procedure (P = 0.0017). Thus, 15 minutes of ischemia did affect neuronal function.
Neurologic score at 2 days and 7 days after procedure
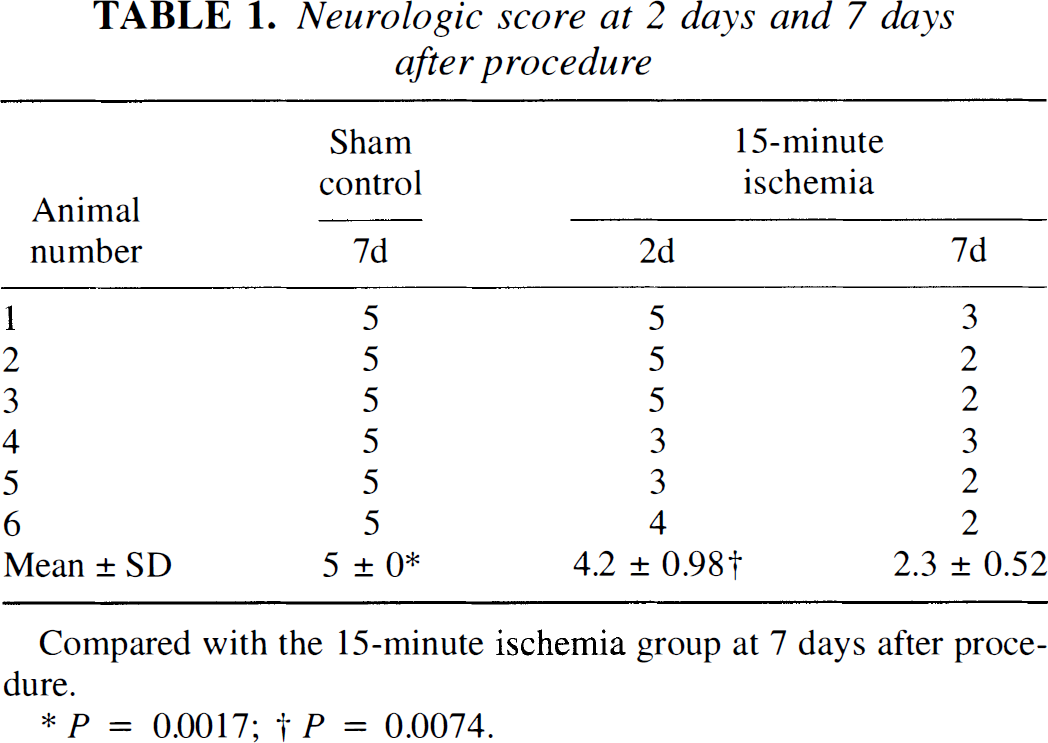
Compared with the 15-minute ischemia group at 7 days after procedure.
P = 0.0017;
P = 0.0074.
Histologic study
The results of cell counting in the ventral gray matter region on the paraffin sections obtained from this study are shown in Fig. 1. In the sham-operated control group, no significant change was seen in motor neurons (23 ± 3.2). On the seventh day of reperfusion after 15 minutes of ischemia, about 70% of motor neuron cells in the ventral gray matter were lost (7 ± 4.0, P = 0.0039), although most motor neuron cells had remained intact after 2 days of reperfusion (21 ± 4.8, P = 0.0039). Small motor neurons and intermediate neurons survived the ischemia. Dorsal horn neurons were intact after 15 minutes of ischemia (data not shown). The 15-minute ischemia affected the number of motor neuronal cells lost by 7 days after the procedure in contrast to the sham-operated control animals.
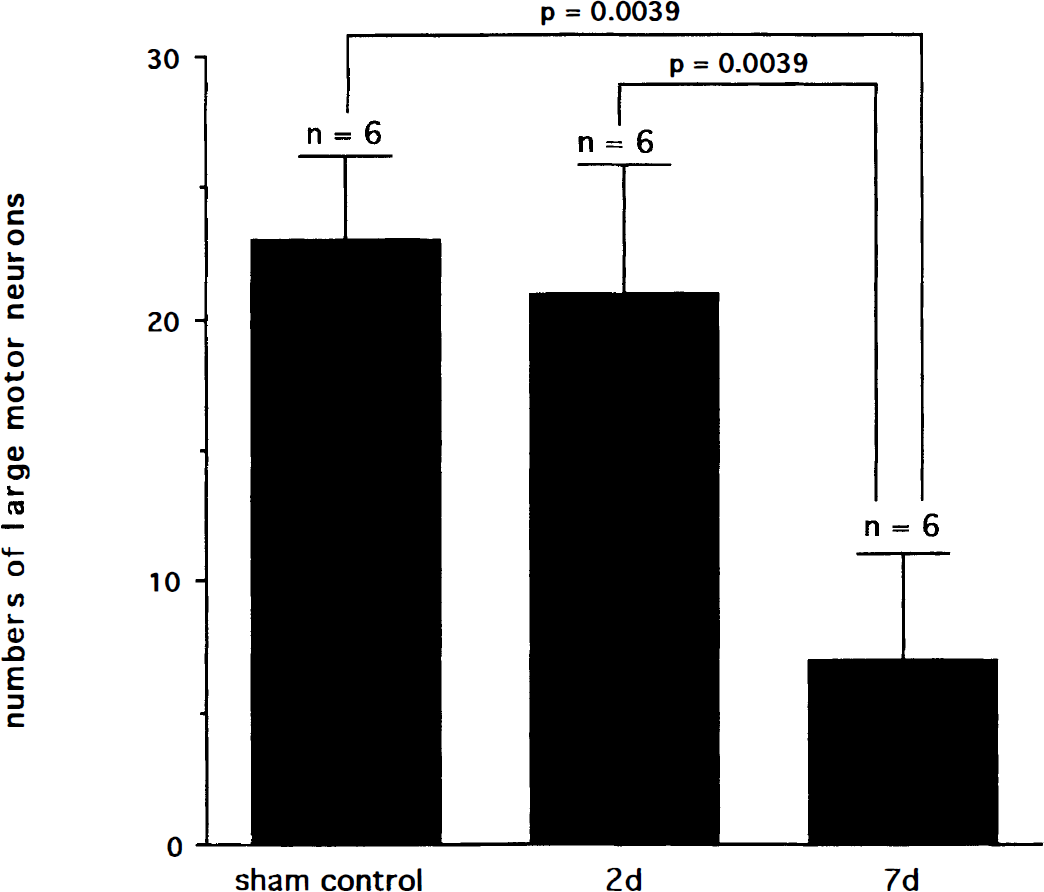
Change in numbers of large motor neurons in ventral gray matter at 2 and 7 days of reperfusion after 15 minutes of ischemia.
HPLC analysis of nitro-compound
Figure 2 shows the results of an HPLC chromatogram of the spinal cord from postischemic rabbits. In the sham-operated animals at 8 hours and 7 days after operation, a peak for NO2-Tyr was not detected (Fig. 2a, open arrow). In the 15-minute ischemia group, a peak for NO2-Tyr was detected in ventral sections of the spinal cord at 8 hours of reperfusion (Fig. 2b, filled arrow), but the amount of the peak for NO2-Tyr returned to the sham-operated control level by day 1 (data not shown). After addition of Na2S2O4, the peak for NO2-Tyr disappeared and a peak for sodium hydrosulfite appeared (Fig. 2c, open arrow). NO2-Tyr was not detected in the dorsal section of the spinal cord at any time of reperfusion. The quantitative results of NO2-Tyr values are shown in Table 2. NO2-Tyr was produced only at 8 hours of reperfusion (P = 0.0021) after 15 minutes of ischemia.
3-Nitrotyrosine in sham and postischemic spinal cord
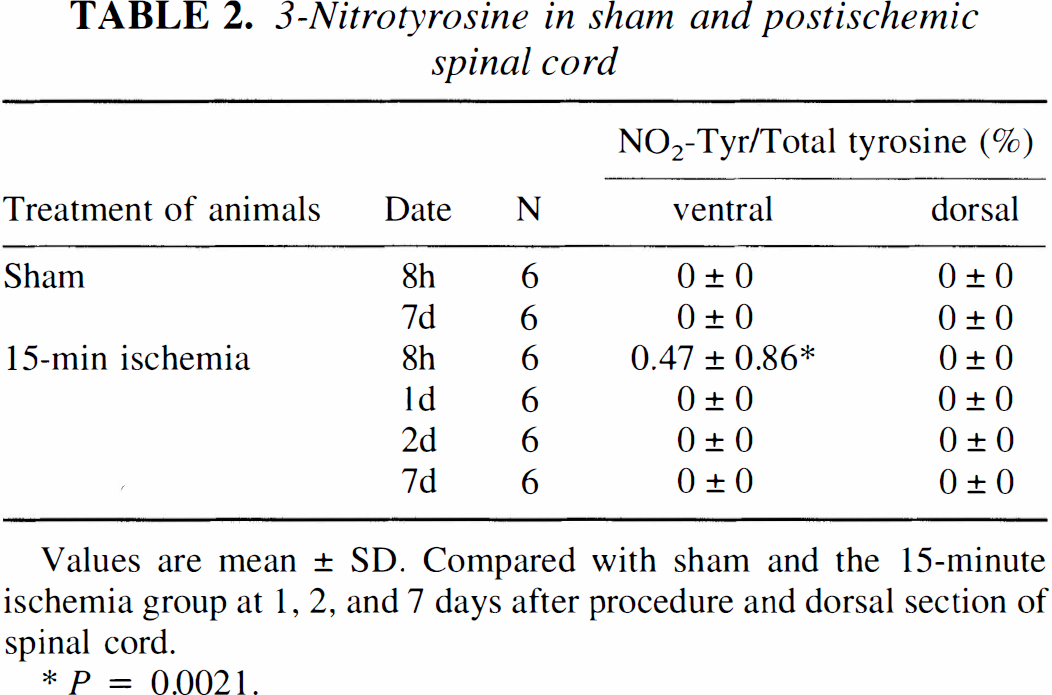
Values are mean ± SD. Compared with sham and the 15-minute ischemia group at 1, 2, and 7 days after procedure and dorsal section of spinal cord.
p = 0.0021.
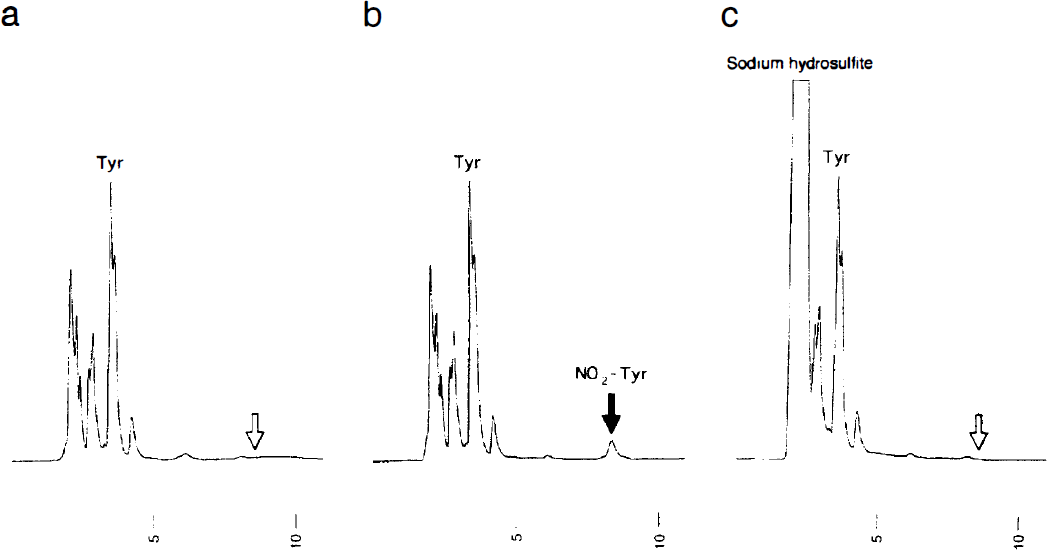
Representative HPLC chromatograms of spinal cord tissues. There is no NO2-Tyr peak in the sample from sham-operated control spinal cord (
Histochemical study
Photographs of sections from spinal cords to detect immunoreactive NO2-Tyr are shown in Fig. 3. The spinal cords of sham-operated animals did not show NO2-Tyr immunoreactivity in any cell (Fig. 3A). After 15 minutes of ischemia motor neurons selectively showed a strong immunoreactivity for NO2-Tyr at 8 hours of reperfusion (arrows in Fig. 3B). Other cells in the spinal cord did not express immunoreactive NO2-Tyr at 8 hours. Although 70% of motor neurons disappeared from the ventral gray matter, the surviving motor neuron cells were still positive for NO2-Tyr at 7 days after ischemic impact.
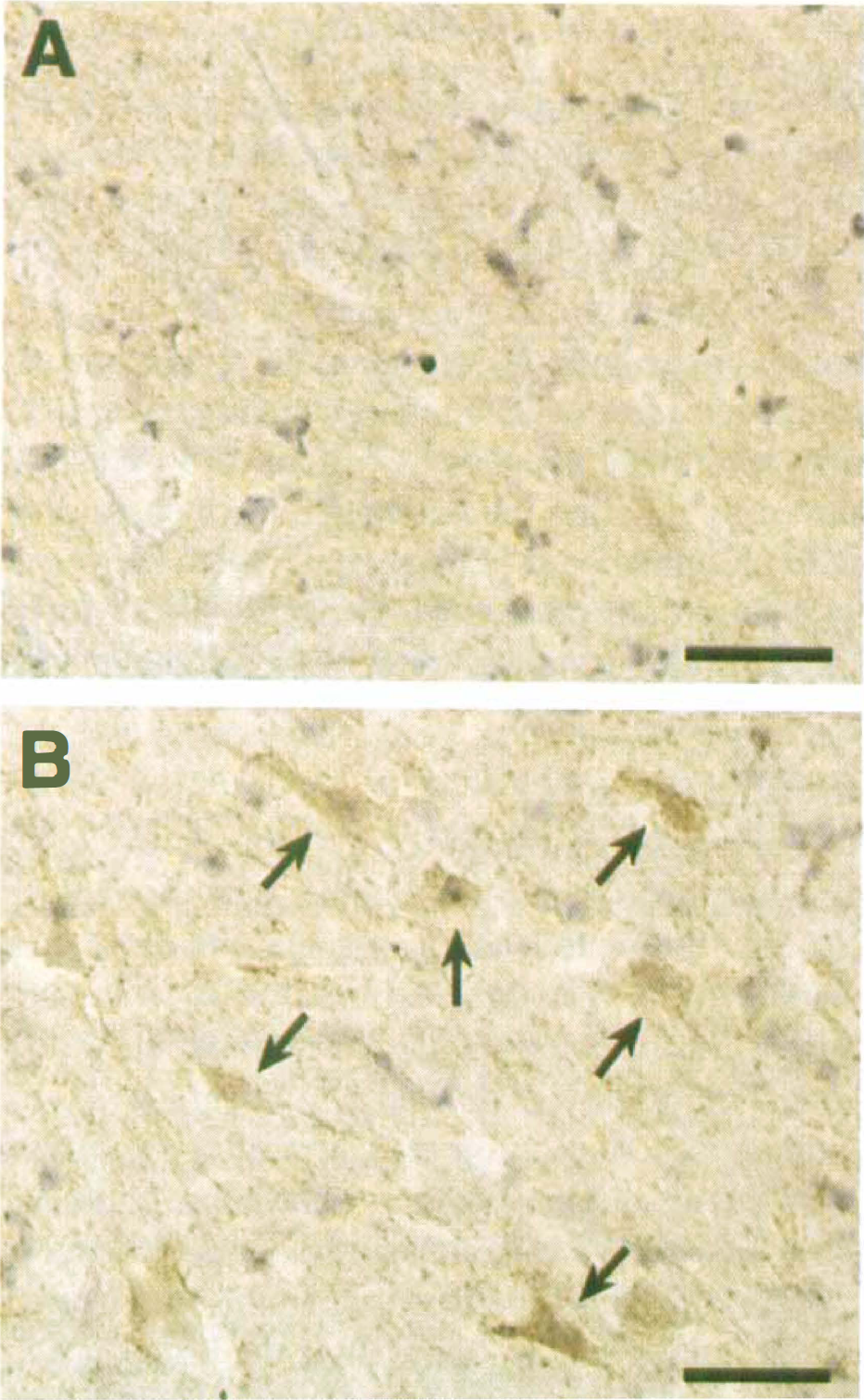
Immunostaining against NO2-Tyr in motor neuron cells in a sham-operated control spinal cord (
DISCUSSION
We have demonstrated delayed and selective motor neuron death in lumbar regions of the rabbit spinal cord with a reproducible model. The spinal cord ischemia model using balloon catheters has a number of advantages compared with other models of spinal cord ischemia (Cheng et al., 1984; Zivin and DeGirolami, 1980). First, the anatomy of the rabbit is simple in comparison with that of other animals such as dog or cat. Balloon occlusion of the abdominal aorta is simple, quickly accomplished, and less invasive compared with conventional methods of ligating the aorta by laparotomy. Therefore, postoperative complications were minimal, and the neurologic as well as histologic patterns of 15 minutes of ischemia in our models were very much reproducible. Analysis by HPLC was also reproducible at each time point (Table 2).
We have previously demonstrated delayed and selective motor neuron death in lumbar regions of the rabbit spinal cord with a reproducible model (Sakurai et al., 1996, 1997, 1998; Watanabe et al., 1996). Fifteen minutes of ischemia is a relatively short ischemic period in comparison to those of previously reported models (Cheng et al., 1984), and after the ischemia, delayed and selective motor neuron damages were observed only after 7 days of reperfusion in this model. This phenomenon is known as selective neuronal death in motor neuron cells after spinal cord ischemia (DeGirolami and Zivin, 1982). Despite recovery of blood flow (Jacobs et al., 1992), motor neurons, which initially appear to have survived the ischemic insult, go on to die days later. This result is compatible with delayed deterioration of neurologic function after spinal cord ischemia, and shows that motor neuron cells are selectively vulnerable to ischemic injury.
Several mechanisms have been proposed to explain the pathophysiology of ischemic neuronal death, including increased excitotoxicity, overload of intracellular calcium, inhibition of protein synthesis, and alterations in gene expression. In recent years, oxygen free radicals or oxidants have been proposed to be involved in central nervous system injury that is produced by cerebral ischemia and reperfusion (Siesjö et al., 1989; Chan, 1994).
The free radical gas NO performs many useful functions when produced in vivo by neurons and endothelial cells (Moncada et al., 1991). However, excess NO can exert cytotoxic and cytostatic effects (Stefanovic-Racic et al., 1993). These many involve both direct toxicity, such as by reaction of NO with mitochondrial iron-sulfur proteins (Hibbs et al., 1988), and the interaction of NO with other free radicals. Nitric oxide reacts with a very high rate constant with O2·− to give ONOO− (Huie and Padmaja, 1993; Koppenol et al., 1992). Peroxynitrite, a stronger pro-oxidant, can react with SOD to form a powerful nitrating agent, resulting in the nitration of tyrosine of cellular proteins and initiation of cellular dysfunction and death (Beckman et al., 1993). Our previous report described the induction of nNOS and SOD after spinal cord ischemia, with most motor neurons showing eventual death (Watanabe et al., 1996). This result suggested that induction of both nNOS and SOD may give rise to formation of NO2-Tyr. However, methods to measure ONOO− directly in biologic systems face the difficulty of nonspecific reactions (Crow, 1997; Gow et al., 1996). In contrast, the measurement of NO2-Tyr by HPLC is well established and reproducible (Kaur et al., 1994; Fukuyama et al., 1997). The specificity of the HPLC peak attributed to NO2-Tyr in the chromatogram was confirmed by elimination of the peak on treatment with excess Na2S2O4. Analysis by HPLC showed NO2-Tyr only after 8 hours. However, immunoreactivity for NO2-Tyr was still present in surviving motor neurons at 7 days of reperfusion. There may be a difference in sensitivity between the two techniques.
The cytotoxicity of NO2-Tyr is currently suspected in the mechanism of motor neuron death of amyotrophic lateral sclerosis (ALS) (Abe et al., 1995). Tyrosine is abundant in neurofilaments, and neurofilaments are rich in motor neuron cells. Therefore, NO2-Tyr formation may give rise to changes of filament function, leading to impaired axonal transport in motor neuron cells (Wu et al., 1994; Wu et al., 1995). Recent studies showed that NO2-Tyr had increased in transgenic mice with familial ALS mutations (Ferrante et al., 1997; Chou et al., 1996). In the present study, the increase of the immunoreactivity and level of NO2-Tyr were demonstrated selectively in ventral motor neuron cells in the spinal cord after transient ischemia, while dorsal neurons did not show such an induction of NO2-Tyr. This finding suggests that formation of NO2-Tyr may play an important role in ventral motor neuron cell death in the spinal cord. Therefore, our results suggest that the mechanism of motor neuron death in the spinal cord after ischemia might have a common feature with that of familial ALS.
In summary, the selective induction of NO2-Tyr in motor neuron cells at a stage without neuronal loss from 8 hours to 2 days may indicate that the lethal change occurs in the spinal cord after 15 minutes of ischemia, which finally results in about 70% of motor neurons being selectively damaged after 7 days of reperfusion. The induction of NO2-Tyr may be implicated in the activation of the death process after transient spinal cord ischemia in rabbits.
