Abstract
The mechanism of spinal cord injury has been thought to be related to the vulnerability of spinal motor neuron cells against ischemia. However, the mechanisms of such vulnerability are not fully understood. We investigated a possible mechanism of neuronal death by immunohistochemical analysis for DJ-1, PINK1, and α-Synuclein. We used a 15-min rabbit spinal cord ischemia model, with use of a balloon catheter. Western blot analysis for DJ-1, PINK1, and α-Synuclein; temporal profiles of DJ-1, PINK1, and α-Synuclein immunoreactivity; and double-label fluorescence immunocytochemical studies were performed. Western blot analysis revealed scarce immunoreactivity for DJ-1, PINK1, and α-Synuclein in the sham-operated spinal cords. However, they became apparent at 8 h after transient ischemia, which returned to the baseline level at 1 day. Double-label fluorescence immunocytochemical study revealed that both DJ-1 and PINK1, and DJ-1 and α-Synuclein were positive at 8 h of reperfusion in the same motor neurons, which eventually die. The induction of DJ-1 and PINK1 proteins in the motor neurons at the early stage of reperfusion may indicate oxidative stress, and the induction of α-Synuclein may be implicated in the programmed cell death change after transient spinal cord ischemia.
Introduction
Spinal cord injury, after a successful operation of thoracic aorta, is a disastrous complication in humans. The reported incidences of paraplegia in such surgery range from 2.9% to 23% in operations on the thoracic aorta. The cause of acute spinal cord dysfunction is believed to be due to its ischemic damage during crossclamping. Ischemia can occur because of permanent exclusion of the essential intercostal arterial blood supply to the spinal cord, or from temporary interruption of blood flow to the spinal cord. However, patients undergoing thoracic aneurysm repair who awake with no neurologic deficit immediately after the operation may sometimes eventually develop paraplegia (Moore and Hollier, 1991); however, the exact mechanism of such delayed vulnerability is not fully understood. The development of delayed paraplegia is considered to be because of delayed and apoptotic neuronal death after transient ischemia. In rabbit spinal cord ischemia model, we have reported delayed and selective motor neuron death after transient ischemia (Sakurai et al, 1998, 2000, 2003a, b , 2005; Yamauchi et al, 2008). Furthermore in this model, we have reported that delayed and selective motor neuron death was greatly associated with an activated apoptotic signal (Sakurai et al, 1998, 2000, 2003a, b , 2005; Yamauchi et al, 2008).
Reactive oxygen species (ROS), free radicals that are normal by-products of oxygen metabolism, are produced in excess during the course of ischemia/reperfusion by a variety of mechanisms such as aberrant electron transport in injured mitochondria (Adam-Vizi, 2005), calcium influx (Starkov et al, 2004), and inflammatory reactions (Cho et al, 2005). ROS rapidly react with proteins, lipids, and DNA and cause damage and if severe, cell death. Equally important, they likely activate specific signaling pathways that initiate adaptive or death responses (Sugawara and Chan, 2003). This ability to control ROS is critical in neuronal ischemia, and neuronal damage occurs if the ‘oxidant-antioxidant’ balance is disturbed in favor of excess oxidative stress during ischemia/reperfusion (Islekel et al, 1999). Therefore, ROS management plays a central role in the pathogenesis of neuronal ischemia.
DJ-1 was originally as a novel oncogene in collaboration with activated small G-protein ras (Nagakubo et al, 1997), and it was later found to be the causative gene of familial Parkinson disease PARK7 (Bonifati et al, 2003). DJ-1 is a multifunction protein, which is involved in transcriptional regulation and acts as a molecular chaperon (Niki et al, 2003). It has been reported that the protein level of DJ-1 increased on exposure to oxidative stress (Mitsumoto et al, 2001). Furthermore, DJ-1 is directly oxidized by free radicals predominantly on cysteine residue at amino acid number 106 (Cys106), and it showed a sift in the isoelectric point from 6.2 to 5.8 (Canet-Aviles et al, 2004). However, recent studies using DJ-1 deficient mice have shown that loss of the DJ-1 resulted in abrogation of dopamine receptor activity and sensitization of cells against oxidative stress, suggesting that loss of function of DJ-1 is correlated with the pathogenesis of Parkinson disease (Kim et al, 2005a, b ). However, DJ-1 function on spinal cord ischemia-induced neurodegeneration is still unknown.
PTEN-induced kinase 1 (PINK1) was first identified as a gene upregulated by exogenous expression of PTEN, a negative regulator of Akt signaling (Unoki and Nakamura, 2001). Valente et al first mapped PARK6 in a Sicilian Parkinson family to chromosome 1p35-p36. Recent reports showed that putative kinase is localized to mitochondria and protects SH-SY5Y cells from proteasome inhibitor-induced apoptosis (Valente et al, 2004). Furthermore, overexpression of PINK1 also diminished both basal and staurosporine-induced activation of caspase3. These anti-apoptotic effects of PINK1 were abrogated by disease-causing mutations (Petit et al, 2005). However, PINK1 function on spinal cord ischemia-induced neurodegeneration is still unknown.
α-Synuclein (α-Syn) is a relatively small (140 amino acids), abundant protein found in cells throughout the nervous system and is particularly enriched in presynaptic nerve terminals. Although the functions of α-Syn are not well known, it has role at the presynaptic terminal (Chandra et al, 2004). α-Syn is a major structural component of Lewy bodies, but in most neurodegenerative disease, Lewy bodies are associated with accumulation of α-Syn (Goedert, 2001). In vitro, overexpression of α-Syn elevates the generation of intracellular ROS, and aggregation of α-Syn is associated with enhanced cell death (Giasson et al, 2002). Therefore, we hypothesize that motor neuron cells eventually dying in this model of acute spinal cord ischemia show prior induction of DJ-1, PINK1, and α-Syn. Thus, we examined the induction of DJ-1, PINK1, and α-Syn in motor neurons after transient spinal cord ischemia.
Materials and methods
Animal Models
During the experiment, the animals were treated in accordance with the declaration of Helsinki and the guiding principles in the care and use of animals. Also, the animal care committee of Sendai Medical Center approved the experimental and animal care protocol.
Fifteen Japanese domesticated white rabbits weighing 2 to 3 kg were used in this study, and they were divided into two groups: a 15-min ischemia group and a sham-control group. Anesthesia was induced with intramuscular administration of ketamine at a dose of 50 mg/kg and maintained with 2% halothane inhalation. A 5-Fr. pediatric thermodilution catheter (405, B. Braun Melsungen A.G., Germany) was inserted through a femoral artery and was advanced 15cm forward into the abdominal aorta. Preliminary investigations by laparotomy had confirmed that the balloon in the distal end of the thermodilution catheter was positioned 0.5–1.5cm just distal to the left renal artery. During the experiment, aortic pressures were continuously monitored both at the proximal and distal positions of the balloon. When the balloon of the thermodilution catheter was inflated in the abdominal aorta, systemic blood pressure of the rabbits did not change. The arterial pressure distal to the inflated balloon fell to near zero and no pulsation was recorded. On deflation of the balloon, systemic blood pressure of this portion decreased for 15 mins and then returned to the normal level (data not shown). Body temperature was monitored with a rectal thermister and was maintained at 37°C with a heating pad during surgery and subsequent ischemia. The animals were allowed to recover at ambient temperature and were killed with deep anesthesia of sodium pentobarbital (100 mg/kg intravenously) at 8 h, and 1 day after reperfusion (n=5 at each time point). In the sham control, the animals were killed at 7 days (n = 5) of reperfusion after insertion of the catheter into abdominal aorta without inflating the balloon. The spinal cords were quickly removed immediately after killing, using the plunger of a 1mL syringe. The tissue samples for Western blot analysis and immunohistochemical studies were frozen in powdered dry ice and stored at −80°C.
Western Blot Analysis
To investigate the changes of DJ-1, PINK1, and α-Syn expression, we performed Western blot analysis. The tissue samples were homogenized in a lysis buffer (0.1 mol/L NaCl, 0.01 mol/L Tris-HCl, pH 7.5, 1 mmol/L EDTA, and 1 µg/mL aprotinin), and then the homogenates were centrifuged at 7000 g for 15 mins at 4°C. The supernatants were used as protein samples. Assays to determine the protein concentration of the supernatants were subsequently performed by comparing to a known concentration of bovine serum albumin using a kit (BCA protein assay reagent kit #23225, Pierce, IL, USA). Sodium dodecyl sulfate polyacrylamide gel electrophoresis was performed in a 10% polyacrylamide gel under reducing conditions. In brief, protein samples were boiled at 100°C in 2.5% sodium dodecyl sulfate and 5% β-mercaptoethanol, and lysates equivalent to 20 µg of protein from each samples were run on the gel for 90mins at 20mA, together with a size marker (rainbow colored protein, Biorad). The electrophoresis running buffer contained 25mmol/L Tris base, 250mmol/L glycine, and 0.1% sodium dodecyl sulfate. The proteins on the gel were then transferred to a polyvinylidene fluoride membrane (Invitrogen, Carlsbad, CA, USA), with a transfer buffer consisting of 48mmol/L Trisbase, 39mmol/L glycine, 0.4% sodium dodecyl sulfate, and 20% methanol.
After the transfer, the membranes were placed in blocking solution of the kit (Western Breeze Chromogenic Western Blot immunodetection kit, WB 7103 or WB 7107, Invitrogen, CA, USA) to block nonspecific binding. Then they were incubated with primary antibodies at 1:1000 dilution for 20 h at 4°C. The primary antibodies used were as follows mouse monoclonal anti-DJ-1 antibody (SC-55572; Santa Cruz Biotechnology, Inc., California, USA), goat polyclonal anti-PINK1 antibody (SC-32584; Santa Cruz Biotechnology, Inc., California, CA, USA), and mouse monoclonal anti-α-Syn antibody (SC-12767; Santa Cruz Biotechnology, Inc., California, USA). After washing in phosphate-buffered saline (PBS), the membranes were incubated with alkaline phosphtase-conjungate anti-goat IgG (Western Breeze Chromogenic Western Blot immunodetection kit, WB 7107, Invitrogen, CA, USA) and alkaline phosphtase-conjungate anti-mouse IgG (Western Breeze Chromogenic Western Blot immunodetection kit, WB 7103, Invitrogen, CA, USA) at 1:1000 dilution in PBS for 90 mins, respectively. The blots were developed using Chromogenic detection method (Western Breeze Chromogenic Western Blot immunodetection kit, WB 7103, Invitrogen, CA, USA). To ascertain specific binding of the antibody for the protein, another membrane was stained in a similar way without the primary antibody. The images of Western blot study were quantified by plotting a two-dimensional densitogram using the image analysis program Image J, version 1.63 (Research Services Branch, NIMH, National Institute of Health).
DJ-1, PINK1, and α-Synuclein Immunocytochemistry
We also performed immunohistochemical study to investigate the changes of expression of DJ-1, PINK1, and α-Syn in five sections per animals (× 200). Spinal cord sections were rinsed in 0.1M PBS for 20 mins and blocked in 2% normal horse serum for 2 h at room temperature. Then, they were incubated with primary antibodies in 10% normal horse serum or 10% normal rabbit serum and 0.3% Triton-X 100 for 20 h at 4°C, respectively. The primary antibodies that were used were the same as those used for Western blot analysis noted above, and each dilution was as follows antibody against DJ-1 at 1:200, that against PINK1 at 1:200, and that against α-Syn at 1:200.
After quenching endogenous peroxidase activity by exposing slides to 0.3% H2O2 and 10% methanol for 20 mins, the slides were washed in PBS and incubated for 3 h with biotinylated anti-goat IgG (PK-6105; Vector Laboratories, Burlingame, CA, USA) and biotinylated anti-mouse IgG (PK-6102; Vector Laboratories, Burlingame, CA, USA) at 1:200 dilution in PBS containing 0.018% normal horse and rabbit serum, respectively. Subsequently, they were incubated with avidin-biotin-horseradish peroxidase complex (PK-6102; Vector Laboratories, Burlingame, CA, USA). The slices were colorized with DAB/H2O2 solution, and cytoplasm was counterstained with hematoxylin. To ascertain specific binding of antibody for the protein, a set of sections were stained in a similar way without the primary antibody.
Fluorescence Double-Labeling Study for DJ-1, PINK1, and α-Synuclein
Spinal cords sections were prepared as described earlier. A nonspecific-blocking procedure was performed using 10% horse serum before application of primary antibodies. Then, the sections were incubated with DJ-1 mouse monoclonal antibodies 1:100 (Santa Cruz Biotechnology) simultaneously with PINK1, and the sections were incubated with α-Syn mouse monoclonal antibodies 1:100 simultaneously with PINK1. These primary antibodies were incubated overnight at 4°C and were detected by using donkey anti-mouse lgG linked with Fluorescein (fluorescein isothiocyanate) I:50 (sc-3699, Santa Cruz Biotechnology, Inc., California, USA.) and donkey anti-goat lgG linked with Texas Red I:50 (sc-3856, Santa Cruz Biotechnology, Inc., California, USA). The slides were mounted in aqueous mounting media with DABCO and observed using fluorescein microscopy (× 200). We also examined the population of DJ-1, PINK1, and α-Syn expressing cells in five sections per animals.
Statistical Analysis
Quantitative analyses of the neurologic score, the cell numbers, and the optical density of Western blots were analyzed by ANOVA. P-value of less than 0.05 was considered to be statistically significant. Parametric data are present as mean±s.d.
Results
Western Blot Analysis
Representative results of Western blot analysis are shown in Figure 1. With antibody against DJ-1, a weak band was detectable in samples of sham control, but those at 8 h after blood flow restoration revealed a single band, with a molecular weight of 23 kDa (Figure 1). This band became scarcely detectable at 1 day after reperfusion. With antibody against PINK1, no band was detectable in samples of sham control, but those at 8 h after blood flow restoration revealed a single band, with a molecular weight of 66 kDa (Figure 1). With antibody against α-Syn, no band was detectable in samples of sham control, but those at 8 h after blood flow restoration revealed a single band, with a molecular weight of 19 kDa (Figure 1). The membrane without the primary antibody revealed no band (data not shown). With quantitative analysis, we found that DJ-1, PINK1, and α-Syn were significantly increased at 8 h of reperfusion (P > 0.0001, respectively).
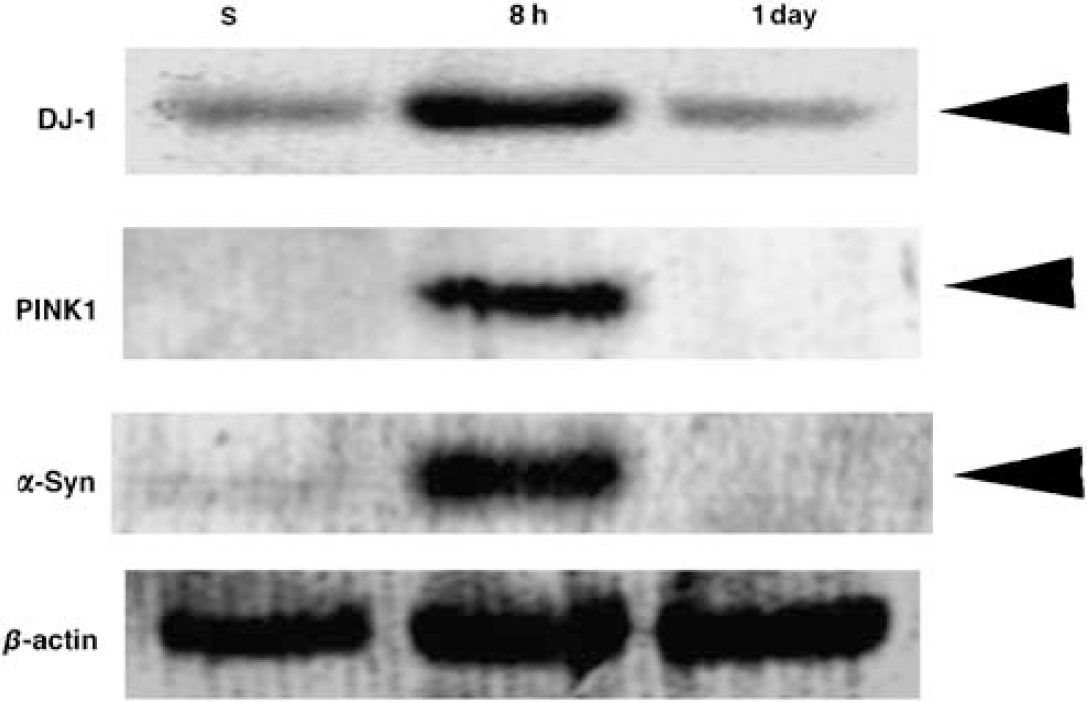
Representative Western blot for DJ-1, PINK1, and α-Syn. Transient ischemia affects expression profile of DJ-1, PINK1, and α-Syn at 8 h of reperfusion. 20 µg of protein from each samples were run on the gel for 90 mins at 20 mA. The proteins on the gel were then transferred to a PVDF membrane, they were then incubated with primary antibodies at 1:1000 dilution for 20 h at 4°C. The blots were developed using Chromogenic detection method.
Histochemical Study
Immunoreactive DJ-1, PINK1, and α-Syn of sections from spinals cords are shown in Figure 2. The spinal cords of sham-operated animals at 8 h and 7 days of reperfusion did not show DJ-1 (Figure 2A), PINK1 (Figure 2E), and α-Syn (Figure 2G) immunoreactivities in any cells. Motor neurons selectively showed strong immunoreactivity for DJ-1 (Figure 2B), PINK1 (Figure 2F), and α-Syn (Figure 2H) at 8 h of reperfusion. Immunoreactivity of motor neuron cells for DJ-1 (Figure 2C) and antigen was almost lost in the motor neurons at 1 day. Immunoreactivity of motor neuron cells for DJ-1, PINK1, and α-Syn decreased at 1 day (Figures 2C, 2F, and 2I).
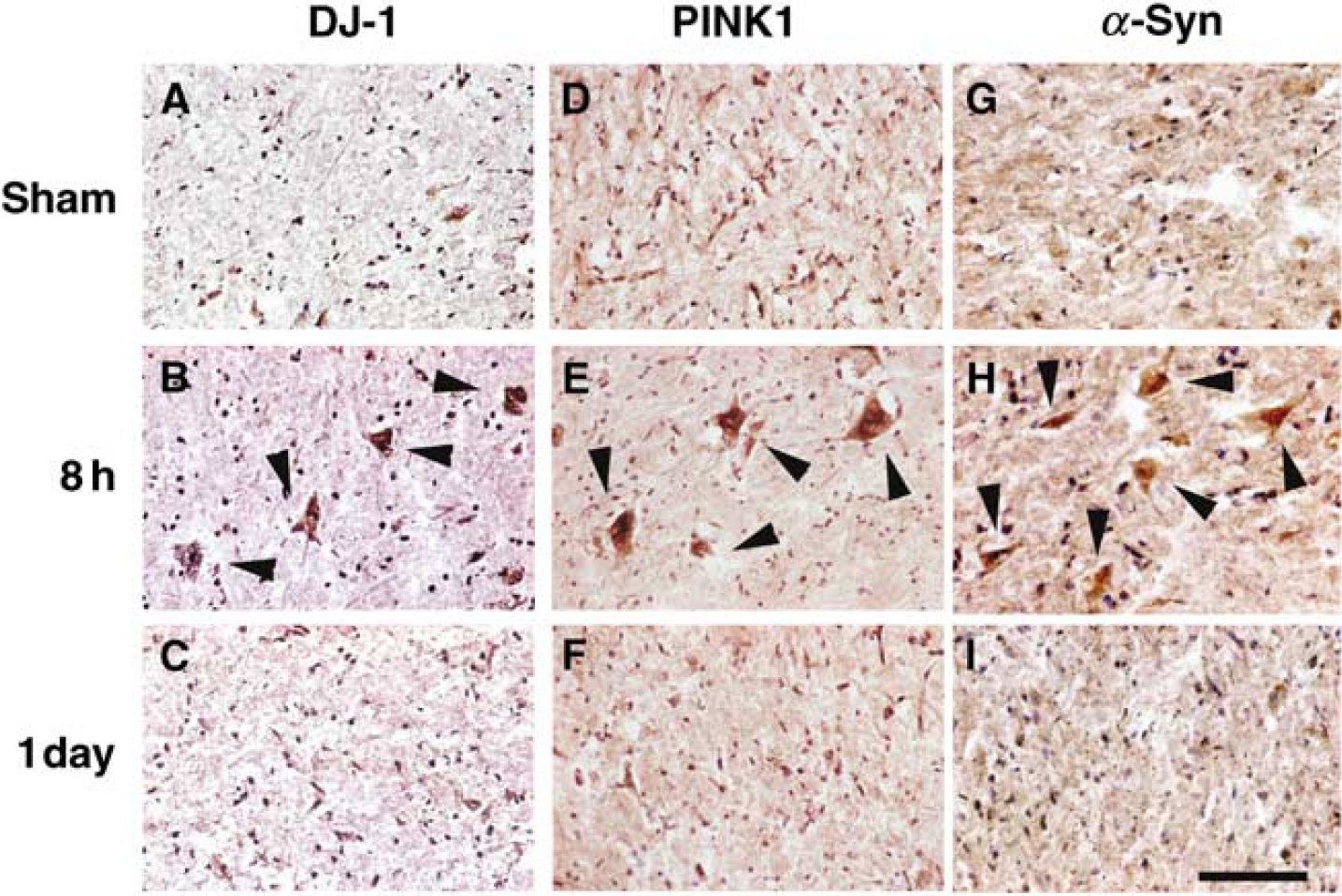
Immunostaining against DJ-1, PINK1, and α-Syn in motor neurons in a sham spinal cord (
Fluorescence Double-Labeling Study
The results of DJ-1 and PINK1 double-staining immunohistochemistry and α-Syn and PINK1 double-staining immunohistochemistry are shown in Figure 3. DJ-1 positive neurons (18.4±3.1/ventral horn, mean±s.d.) were strongly co-localized with PINK1 positive neurons (18.0±2.3/ventral horn) at only 8 h of reperfusion (Figures 3A and 3B), and α-Syn positive neurons (19.2±3.3/ventral horn, mean±s.d.) were strongly co-localized with PINK1 positive neurons (18.2±4.1/ventral horn) at only 8 h of reperfusion (Figures 3D and 3E). Furthermore, approximately 85% of motor neurons (15.7±1.6/ventral horn) expressed both DJ-1 and PINK1 (Figure 3C, arrows), and approximately 85% of motor neurons (16.8±3.8/ventral horn) expressed both α-Syn and PINK1 (Figure 3F, arrows).
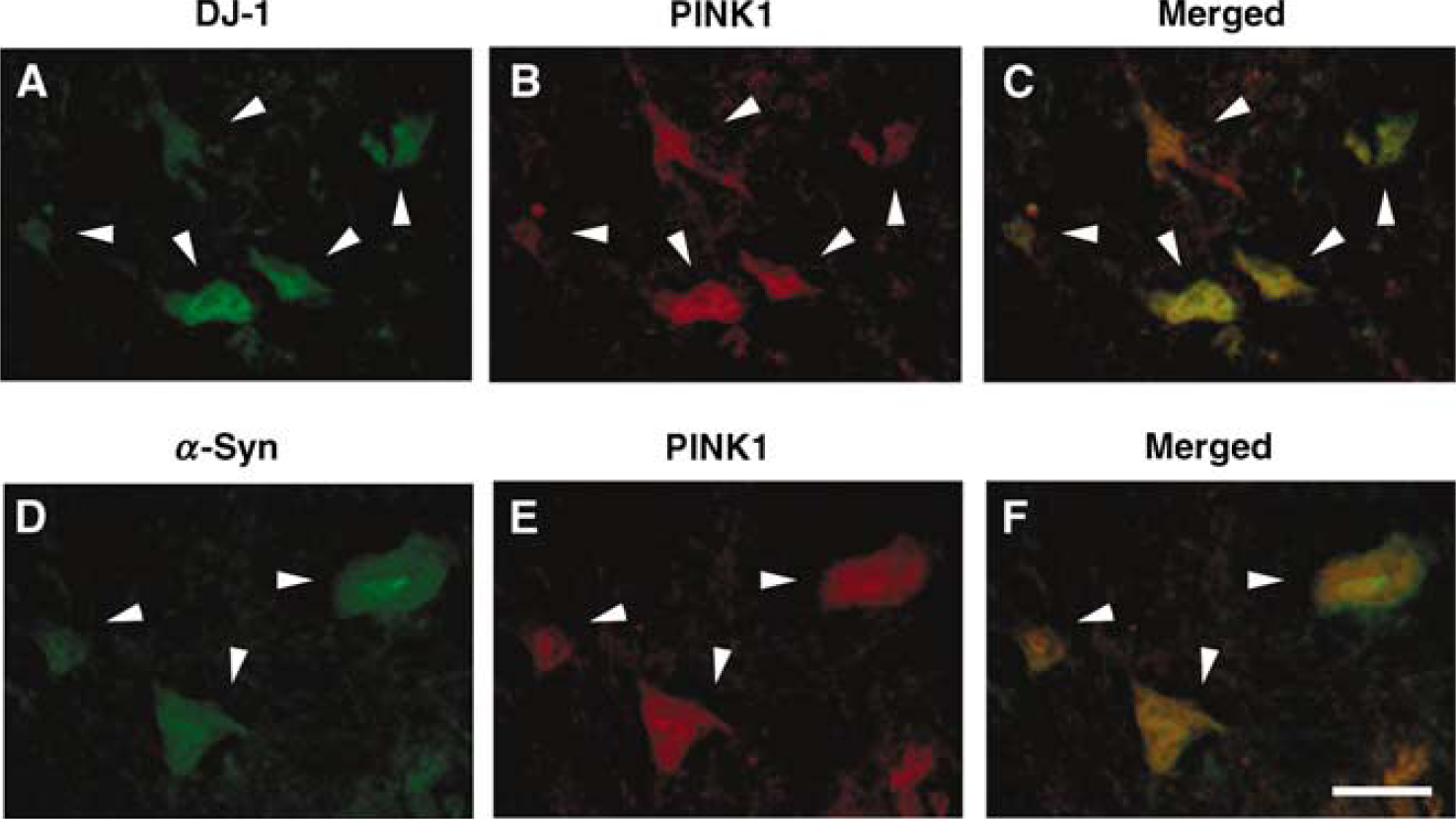
Co-localization of DJ-1 and PINK1 (
Discussion
Recent studies show that three genes, which cause autosomal recessive forms of Parkinsonism, Parkin, DJ-1, and PINK1, suggest that they have more initiate relationship with the mediators of programmed cell death and that loss-of-function mutations may result in an increased propensity for the neuron to die. Furthermore, the function of these genes have suggested that they may share roles in regulating survival signaling pathways, such as those mediated by the survival signaling kinase Akt (Kim et al, 2005a, b ).
Aleyasin et al reported that DJ-1 deficient mice showed larger cerebral infract size than wild-type mice after endothelin-1 induced brain ischemia (Aleyasin et al, 2007). Furthermore, Yanagisawa et al reported that the intrastriatal injection of recombinant glutathione S-transferase-tagged human DJ-1 markedly reduced infract size after MCAO model in rat (Yanagisawa et al, 2008). We have previously demonstrated the oxidative damage in motor neurons in this model, which showed apoptotic change and eventually die (Sakurai et al, 2003a, b ). In this study, DJ-1 was induced in motor neurons at 8 h of reperfusion, which returned to the baseline level at 1 day. These results suggest that the selective induction of DJ-1 in motor neuron cells may indicate oxidative stress that occurs in the spinal cord after 15 mins of ischemia, and finally, almost all motor neurons may be selectively damaged after 7 days of reperfusion. Therefore, the mechanism of induction of DJ-1 may be similar to the motor neuron cells in the spinal cord and rat brain.
A recent study showed that the PINK1 protein is increased in Parkinson disease brain (Muqit et al, 2006). This result suggests that PINK1 may be upregulated in response to Parkinson disease-related stress. This in vivo finding may support previous in vitro findings that PINK1 may have a neuroprotective role against cellular stress (Valente et al, 2004; Petit et al, 2005). Furthermore, we have previously reported that ubiquitin, Parkin, and UCH-LI were strongly induced in motor neuron at the early stage of reperfusion after spinal cord ischemia (Yamauchi et al, 2008). These results indicated that the vulnerability of motor neuron of the spinal cord might be partially attributed to the different response in ubiquitin-mediated stress response after transient ischemia. In this study, the increase in the immunoreactivity of PINK1 was showd selectively in ventral motor neuron cells in the spinal cord after 8 h of reperfusion. This finding suggests that the oxidative and ubiquitin-mediated stress could activate PINK1 induction as compensatory mechanism. Therefore, our results suggest that the mechanism of motor neuron death in the spinal cord after ischemia might have a similar feature with that of Parkinson disease.
α-Syn can protect neurons from apoptosis by inhibition of caspae3 (Alves da Costa et al, 2002). However, mutation in α-Syn abolish its inhibitory modulation of caspase3 (Alves da Costa et al, 2002). Aggregation of wt and Parkinson disease-related mutant α-Syn is associated with enhanced cell death (Giasson et al, 2002). This process could be caused by the intrinsic pro-apoptotic properties of α-Syn or the less of an anti-apoptotic function. The latter possibility is supported by evidence that α-Syn lowers the sensitivity of neurons to apoptosis driven by p53 (Alves da Costa et al, 2002) and activates mitogen-activated protein kinase pathway (Iwata et al, 2001). Kato et al, showd that in human amyotrophic lateral sclerosis, spinal motor neurons could display Lewy body-like filamentous inclusions (Kato et al, 1989), most of which are ubiquitinated (Kato et al, 1989), and significant abnormal α-Syn staining of spheroids and corticospinal axons (Dorerty et al, 2004). α-Syn is also upregulated in motor neurons in transgenic mice expressing mutant human Cu/Zn superoxide dismutase (Chung et al, 2003).
Recent study showd that motor neurons of Parkinson disease-related mutant α-Syn (A53T) transgenic mice were positive for TUNEL (terminal deoxynucleotidyl transferase-mediated 2‘-deoxyuridine 5‘-triphosphate-biotin nick end labeling) labeling, cleaved caspase3, and p53. Mitochondria in neurons had TUNEL positive matrices and p53 at the outer membrane. These results suggest that A53T mutant mice develop mitochondrial DNA and degeneration and apoptotic like death of motor neurons (Martin et al, 2006). We have previously reported that caspase3 were induced same motor neurons in this model (Sakurai et al, 2003a, b ), and in this study, α-Syn was induced at 8 h in motor neurons, which eventually die. Our results suggest that the mechanism of motor neuron death in the spinal cord after ischemia might have a similar feature with that of Parkinson disease.
This study shows that immunoreactivities for DJ-1, PINK1, and α-Syn were induced at 8 h in the same motor neurons, which eventually die. The induction of DJ-1 and PINK1 proteins at the early stage of reperfusion might indicate a stress response and be one factor responsible for the delay in neuronal death after spinal cord ischemia and that the induction of α-Syn may be implicated in the programmed cell death change after transient spinal cord ischemia in rabbits.
