Abstract
This study describes the dynamics of flow activation by reading and investigates the potential use of repeated flow velocity measurements for the lateralization of speech. Using simultaneous transcranial Doppler recordings from both middle cerebral arteries and averaging techniques in 25 healthy volunteers, we describe the changes in blood flow velocity caused by repetitive reading tasks of variable duration in comparison with a resting state. Reading aloud evoked a characteristic temporal flow pattern in both hemispheres, consisting of three relative maxima in flow velocity during and after activation. Flow velocities lower than baseline were common during longer lasting activation. The amplitudes of two of the observed peaks decreased depending on the duration of the task. Reading silently produced a markedly different temporal pattern of activation than reading aloud. There were individually reproducible significant side to side differences. Right-handed persons (n = 15) almost without exception showed a significantly higher increase in flow velocity on the left hemisphere (e.g., reading silently 8.7% versus 5.3%; P < 0.0001). Three out of ten left-handed individuals, however, exhibited no significant side to side difference or exhibited lateralization to the right during one or more of the tasks. These findings suggest that reading induces task-specific temporal patterns of regional neuronal activity, which show habituation with longer duration of activation. Additionally, the observed side to side differences could be useful to predict language dominance.
Activity of cortical areas associated with sensory, motor, or cognitive functions induces changes in regional CBF (Lassen et al., 1978). Such changes have been studied by xenon-CBF methods (Wallesch et al., 1985; Risberg, 1986), by functional magnetic resonance imaging (Binder et al., 1995) and—most extensively—by positron emission tomography (PET) (Petersen et al., 1988; Wise et al., 1991; Howard et al., 1992; Leblanc et al., 1992; Rumsey et al., 1992). Despite its excellent spatial resolution, PET, however, presently fails to describe fast-acting temporal dynamics of CBF-activation, because its temporal resolution is in the range close to 1 minute. In contrast, transcranial Doppler sonography (TCD) offers very good temporal resolution and has been used to describe visually or cognitively evoked flow changes (Aaslid, 1987; Conrad and Klingelhöfer, 1989; Droste et al., 1989; Thie et al., 1992; Kelley et al., 1992; Markus and Boland, 1992; Thomas and Harer, 1993). Previous TCD studies, using cognitive stimulation, however, did not reveal exact temporal characteristics of activation or individually reproducible side to side differences (Droste et al., 1989; Diehl et al., 1990; Njemanze, 1991; Kelley et al., 1992; Thomas and Harer, 1993). With the development of simultaneous bilateral recording and averaging techniques the sensitivity of TCD to detect evoked flow changes and hemispheric dominance has been improved (Rihs et al., 1995). Analogous to multimodally evoked electrical potentials, which are averaged from multiple stimulus-evoked responses in the electroencephalogram, TCD now allows analysis of a repetitive stimulus. Thus random changes in cerebral blood flow velocity (CBFV) caused by systemic influences (e.g., cardiac, respiratory, or vegetative) or transient unintended activation of other brain areas are filtered out.
The intention of this study was to describe the temporal dynamics of a cognitive task of variable duration and to determine whether intraindividually reproducible side to side differences can be obtained.
SUBJECTS AND METHODS
Twenty-five healthy volunteers, 12 men and 13 women, 29 ± 3 years of age, without a history of reading disabilities, were studied at the Cerebrovascular Laboratory at Harborview Medical Center, Seattle, Washington, U.S.A., after informed consent had been obtained. The individuals were classified as right- or left-handed according to their indices of handedness by the Edinburgh Inventory (Oldfield, 1971). Subjects with a lateralization quotient of 0 or less were considered left-handed. Additionally the subjects were asked for familiar sinistriality.
The volunteers were sitting comfortably in an inclined position in a quiet room with limited light. After identification of the middle cerebral arteries (MCA) according to standard criteria (Fujioka and Douville, 1992), the probes were fixed in place by a headband, and continuous simultaneous recordings of CBFV from both sides were obtained using a computerized Doppler unit (Multidop X, DWL, Sipplingen, Germany). This unit is equipped with a special software program (version 7.40, written by R. Aaslid) for averaging the CBFV data in response to a repeated stimulus of defined duration. The computer algorithm calculates for each side the relative percent changes during and after each task in relation to a control value. After a number of cycles has been completed, the average percentage change in mean CBFV with time is displayed for each MCA. The mean velocity is expressed with error bars of a width of one standard deviation (SD) of all cycles on each side. The percentage side to side difference with time ± SD is calculated as well and expressed as the percentage change of the ratio of the MCA velocities. The last 10 velocity samples before each new stimulus serve as the control value (baseline of Fig. 1).
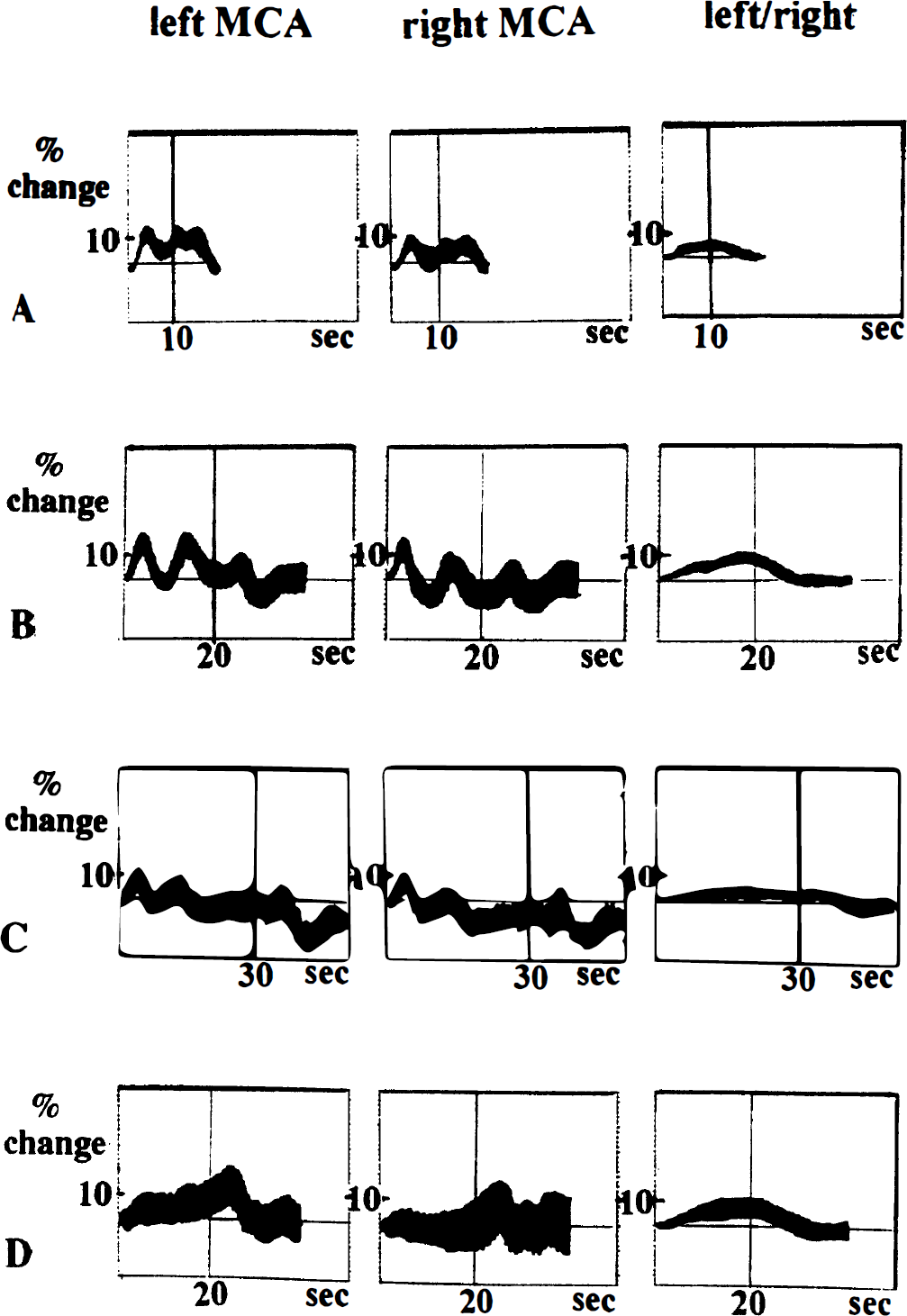
Effects of reading tasks of variable duration on cerebral blood flow velocity. (
After a stable baseline had been recorded for at least 5 minutes, reading sessions of variable duration were started in random order. The task consisted of repeated reading cycles (n = 10), during which the subjects read aloud (10, 20, or 30 seconds) or silently (20 seconds) from a prepared simple newspaper text. This text consisted of local politics from an area, which was not familiar to the individuals and was intended to have little emotional impact on the readers. The same text, which was not balanced regarding content or difficulty with respect to the duration of the reading task, was used in each trial. Each reading cycle was followed by a rest period of the same duration as the respective task, during which the volunteers kept their eyes closed and were asked to restrain from any other activity.
In one volunteer additional simultaneous recordings of arterial blood pressure (ABP) from a radial artery were obtained using a noninvasive continuous tonometric blood pressure monitor (Nellcor, N-Cat N-500, Hayward, CA, U.S.A.).
All calculations were performed off-line. In each subject the averaged changes in flow velocity ± SD during and after the stimulus as well as their averaged relative side to side difference ± SD (expressed as the change in the left to right ratio) were plotted for each MCA. Then for each task, the latency and amplitude of the relative maxima occurring during and after activity, the latency for return of the velocities back to baseline, and the side to side difference were noted. Further data analysis was performed, using paired Student's t test to compare the corresponding bilateral amplitudes of the velocity changes at each maximum and minimum. For each corresponding maximum and minimum and vessel a one-way analysis of variance was used to express significant differences in latency or amplitude between the different tasks (P < 0.05). The results of left-handed and right-handed individuals were compared by the Mann-Whitney test.
Simple regression analysis was performed to look for correlations between the induced velocity changes and age, sex, or lateralization quotient.
RESULTS
Fifteen individuals were considered predominantly right-handed, 10 were left-handed.
Reading aloud
Reading aloud, all subjects showed a characteristic temporal pattern of simultaneous bilateral changes in CBFV during and after stimulation.
Three relative maxima of CBFV (P1, P2, and P3) could be noted regardless of the duration of the task (Fig. 1). Each peak in velocity was followed by a marked decrease in CBFV. The amplitudes of the relative minima in CBFV after P1 were highly variable. After P2, CBFV fell below baseline on the less activated side in 12 of 25 subjects during the 20-second task and in all but 5 individuals during the 30-second task. After P3, CBFV typically had its minimum below baseline (Nm) and returned back to baseline after about 15 seconds.
Latencies and amplitudes of the relative maxima and minima of the CBFV changes on each side are shown in Tables 1 and 2 for the entire group. Because side to side differences in latency were hardly measurable and seemed negligible in comparison with the variations in latency of the individual cycles, the latencies of the minima and maxima of the left MCA were considered valid for both sides.
Latency (in seconds) of relative flow maxima/minima
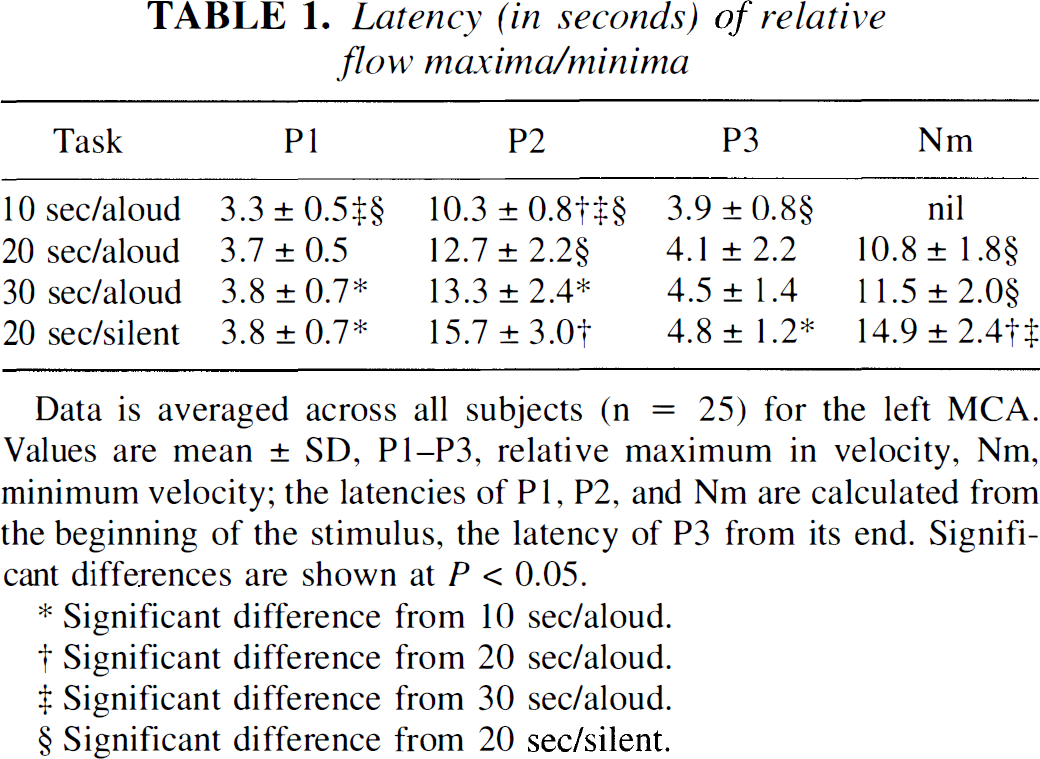
Data is averaged across all subjects (n = 25) for the left MCA. Values are mean ± SD, P1–P3, relative maximum in velocity, Nm, minimum velocity; the latencies of P1, P2, and Nm are calculated from the beginning of the stimulus, the latency of P3 from its end. Significant differences are shown at P < 0.05.
Significant difference from 10 sec/aloud.
Significant difference from 20 sec/aloud.
Significant difference from 30 sec/aloud.
Significant difference from 20 sec/silent.
Amplitudes (percent change) of relative flow maxima/minima and side to side differences (percent change)
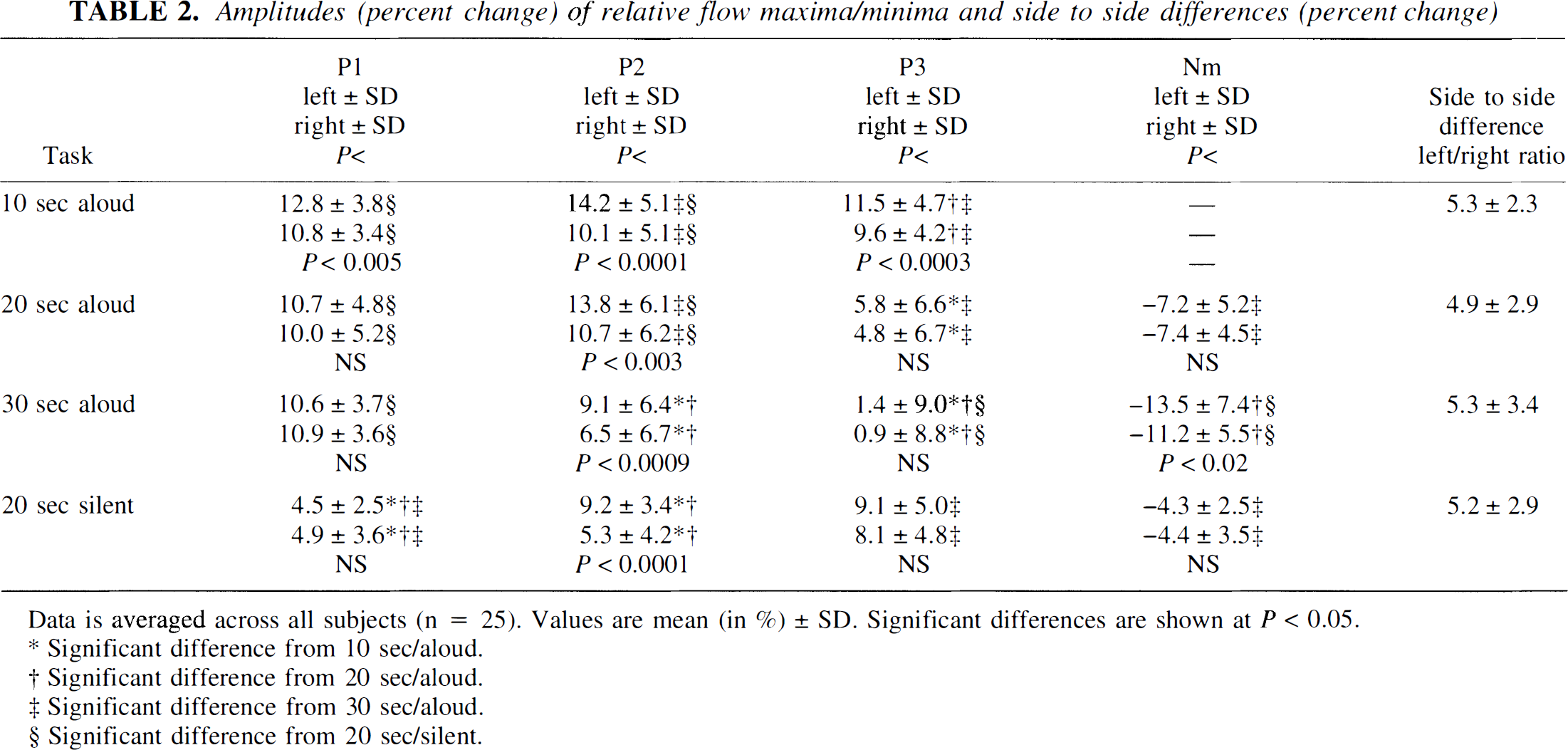
Data is averaged across all subjects (n = 25). Values are mean (in %) ± SD. Significant differences are shown at P < 0.05.
Significant difference from 10 sec/aloud.
Significant difference from 20 sec/aloud.
Significant difference from 30 sec/aloud.
Significant difference from 20 sec/silent.
The first maximum, P1, occurred about 3 seconds after the beginning of each task. The activation induced a relative rise in CBFV of about 10% to 13%. This maximum was observed slightly earlier and tended to be higher during the 10-second task than during the longer tasks. In contrast to P2 and P3, the P1 peak was accompanied by a concomitant increase in ABP, monitored in one subject simultaneously.
The second maximum, P2, was noted around the end of the 10-second task (10.3 ± 0.8 seconds). In contrast its mean latency was almost identical at about 13 seconds during the 20- and 30-second tasks. The mean amplitude of P2 was significantly higher during reading aloud for 10 or 20 seconds (about 14% increase on the left side, 11% on the right) than during reading aloud for 30 seconds (about 9% left, 7% right).
The third maximum, P3, was observed about 4 seconds after the end of each task and occurred sooner, on average, the shorter the activation was. Its amplitude diminished markedly from about 10% to 12% after reading aloud for 10 seconds to just above or even below baseline after reading aloud for 30 seconds.
The minimum in CBFV, Nm, occurred about 11 seconds after the end of a task and was therefore barely noticeable during the 10-second task. It tended to occur sooner during the 20-second task in comparison with the 30-second task. Its amplitude was significantly lower than baseline after reading aloud for 30 seconds (>11% reduction in flow), as compared with 20 seconds (about 7%).
Reading silently
Reading silently produced a distinctly different pattern of CBFV activation than reading aloud, which, however, shared some main features (Fig. 1D). The first peak was usually small and sometimes absent, whereas the second peak in velocity was occasionally indistinguishable within a longer lasting plateaulike increase in CBFV. Nevertheless it was possible to distinguish all peaks in the majority of the patients. The latency of all relative maxima and minima tended to be delayed in comparison with reading aloud (see Table 1). Comparing the two tasks of the same 20-second duration, this delay in latency was significant for P2 and Nm. Reading silently, P1 usually gained only less than half the amplitude of the respective maximum in reading aloud. However, P2 was significantly lower in reading silently than the corresponding maximum in reading 20 seconds aloud, and P3 tended to be more, and Nm less, marked than the corresponding maxima and minima (see Table 2).
Side to side differences
There were relevant side to side differences, as indicated by the error bars in at least one task in every subject (usually in all tasks). The mean individual side to side difference in CBFV activation for each task expressed as the change in the left to right ratio was close to 5%. When the time course of these side to side differences was plotted, a gentle continuous slope was observed, starting about 2 to 3 seconds after the beginning of the task. This increase then turned into a plateau during the task and tapered off gradually after the end of activation to reach baseline after about 5 to 10 seconds. The maximum side to side difference was usually not markedly distinguishable within the plateaulike increase. Its latency was close to that of P2 (Fig. 1, third column).
Statistical analysis for the entire group (right-handed and left-handed subjects together) and for the group of right-handed individuals revealed significant reproducible side to side differences of the amplitudes at P2 in favor of the left hemisphere for all tasks (see Table 2). When only the group of left-handed individuals was considered, we did not find significant mean side to side differences in amplitude at P2 in the reading aloud tasks because of exceptions in lateralization among the left-handed subjects (see below). No other significant differences in the velocity responses of right- and left-handed persons were detectable. Data analysis comparing the “dominant” MCA (exhibiting the greater increase in CBFV during the task) with the “nondominant” did not result in significant differences for latency or amplitude between left- and right-handed individuals.
Exceptions from lateralization to the left side and subgroups
There were, however, notable individual exceptions from lateralization to the left: reading silently, all right-handed subjects without exception demonstrated significantly higher percentage increases in CBFV on the left hemisphere. In contrast, reading aloud, one individual showed lateralization to the right hemisphere during all three tasks. This subject was truly right-handed (lateralization quotient 100%). Among left-handed individuals (n = 10), three subjects differed from the usual pattern. One subject always demonstrated significant flow lateralization to the right cerebral hemisphere, while two others showed no consistent significant side to side difference. For the longer reading aloud tasks this resulted in nonsignificant differences in amplitude between the MCA at P2 (e.g., reading 30 seconds aloud: 10.5% ± 5.8% for the left side versus 9.8% ± 6.4% for the right) because of the greater variance in this group. Correspondingly the left to right ratio was significantly smaller in the group of left-handed subjects in comparison with the right-handed group (e.g., reading 30 seconds aloud: 0.6 ± 8.3 for left-handed versus 4.2 ± 2.7 for right-handed).
We did not find any significant correlation between sex, age, educational status, or lateralization quotient and the relative amplitude of the maxima, their latencies, or side to side differences.
DISCUSSION
We have demonstrated in healthy subjects that reading evokes typical temporal patterns of CBFV in both MCA, which are accompanied by significant side to side differences. Because cerebral blood flow and metabolic demand are linked, the observed peaks in CBFV should reflect relative differences in activity, whereas non—task-related influences should be essentially diminished by the averaging process. Although TCD does not allow quantitative analysis of absolute CBF values, it is possible to interpret relative changes in CBFV as changes in CBF with one important assumption: using CBFV as an index of CBF requires that the diameter of the insonated vessel remains constant, because even small changes in vessel diameter could significantly affect data that are based on CBFV measurements (Kontos, 1989). There are no validation studies known to us that have proven that there are no changes in the cerebral conductance vessels during exercise or cognitive activity. Several studies, however, have been performed that suggested that there are no significant changes of the MCA diameter under a variety of rigorous conditions. These included CBF measurements by different established methods during spontaneous flow variations (Lindegaard et al., 1987), the administration of vasoactive drugs (Krapf et al., 1987), autoregulatory challenge (Newell et al., 1994; Larsen et al., 1994), and—within a wide range—changes of Pa
Interpretation of the relative maxima and minima
Using repetitive visual stimulation and averaging techniques in the posterior cerebral artery, Aaslid (1987) has shown that a fast increase in blood flow in the posterior cerebral artery can be noted after stimulation. The CBFV increase reached 50% of its maximum response after 2.3 seconds and 90% after 4.6 seconds. Changes in CBFV within a similar time span have been observed as a result of the autoregulatory response to a sudden fall in ABP (Aaslid et al., 1989). These results give evidence for the human brain's ability to adjust the diameter of its resistance vessels within a few seconds. In our study P1 seems to fit into this temporal frame, but it was significantly smaller or sometimes even absent during silent reading as compared with reading aloud. Early maxima of CBFV have been observed in other TCD studies in which the subjects were reading aloud (Droste et al., 1989; Diehl et al. 1990), and the suggestion had been made that the Valsalva maneuver accounted for some of the observed changes. Our own observations of CBFV and ABP during the Valsalva maneuver support this view. A strain of 20 mm Hg of intrathoracic pressure, e.g., produces a transient increase in CBFV of approximately 10% with a similar latency as P1 (Tiecks et al., 1995), while a single “ahem” may raise intrathoracic pressure and in its course ABP by 20 to 30 mm Hg (Sharpey-Schafer, 1953). Because whispering or silent tasks without any apparent Valsalva maneuver also may produce an initial peak in CBFV, it seems possible, however, that there is an additional nonspecific arousal with the beginning of the task.
Moreover, there is evidence for a transient, relatively small increase in MCA velocity caused by visual stimulation because of eye opening (Aaslid, 1987). This increase is smaller, however, almost by a factor of 10, in comparison with the respective increase in posterior cerebral artery velocity.
In contrast to its course during P1, ABP was not consistently increased during stimulation and did not reach a relative maximum at the time of P2. Additionally, cerebral autoregulation should have been able to restore constant cerebral blood flow within 13 seconds. (Aaslid et al., 1989; Newell et al., 1994). Thus it is most likely that the maximum in CBFV was specifically caused by the bilateral activation of different brain areas because of the respective task. Bilateral activation of distinctive cortical areas including the striate and prestriate cortex and primary and supplementary motor areas, and predominantly left-sided activation of the dorsolateral prefrontal cortex, the inferior frontal region (Broca's area), and different parts of the temporal and perisylvian parietal cortex has been demonstrated during linguistic tasks by PET (Wise et al., 1991; Howard et al., 1992; Leblanc et al., 1992; Rumsey et al., 1992). Activation of the cerebellum, the cingulum, and the basal ganglia including both caudate nuclei and a left thalamic-pallidal area has also been reported (Wallesch et al., 1985; Petersen et al., 1988; Howard et al., 1992). We presented a relatively simple task, which was designed to have minimal emotional or cognitive impact. Nevertheless, it was complex enough to include primary visual processes, pattern recognition, access to a visual input lexicon, and semantic processing, as well as word retrieval, word production, and finally articulation. Although we tried to avoid other influences it cannot be excluded that there was additional secondary activation of other brain areas caused by associative thoughts or emotions (Bryden et al., 1991; George et al., 1995) or modulation by varying degrees of attention (Meyer et al., 1991) or anxiety (Gur et al., 1987). These variables may also account for some of the variance between the subjects. Considering the size of those brain areas, which are potentially stimulated, however, it is not surprising that we found changes in CBFV of more than 10% in both MCA, which supply most of the above areas except for the visual cortex.
The amplitudes of the induced changes during the task, however, suggest that there is habituation to the stimulus: after P2, CBFV tended to decrease below baseline on both hemispheres when the stimulus lasted for 20 seconds or more. In addition, CBFV at P2, P3 and Nm was smaller, the longer the task lasted. During pilot trials (unpublished) CBFV also tended to be diminished with the number of repetitions. Habituation to a stimulus has been reported previously (Risberg, 1986; Droste et al., 1989), and other authors have found a relative drop in CBFV during tasks of longer duration (Droste et al., 1989; Diehl et al., 1990). Whether this is caused by a decrease in attention (Meyer et al., 1991) or motivation, maybe in part because of the repetition of the same text, or whether it may be a reflection of better adaptation to the respective stimulus is an open question to be addressed.
The side to side differences, however, do not show any marked decrease before the end of the respective task. In combination these findings can be explained by a short-lasting activation of major brain areas on both hemispheres, which is tapering off after about 13 seconds in reading aloud. Another theoretical explanation would be that the activation effect is counterbalanced by hypoperfusion of other regions in the same vascular territories. At the same time, centers that are specific for the task obviously remain activated and continue to receive relatively more blood, as expressed by the relatively small but very constant side to side difference of about 5%.
We were not able to confirm the sex- or handedness-related differences in blood flow activation reported in a previous PET study (Gur et al., 1982). This may be related to our limited number of cases and the better spatial resolution of PET, which allows better analysis of a localized region of interest.
Nevertheless, analysis of CBFV changes by the induction of mental activity may become a clinically helpful tool. It is likely that severe focal or general defects will affect the patterns or the latencies and amplitudes of cognitively evoked flow, because intact metabolic coupling in the respective brain areas is required to produce the characteristic “normal” response. Whether such possible abnormalities, as they have been described after auditory stimulation (Bruneau et al., 1992), may lead to new diagnostic or prognostic insights remains to be answered.
The diagnostic potential of the side to side differences
Almost all previous Doppler studies using nonsimultaneous insonation of the two hemispheres found it difficult to demonstrate individually reproducible side to side differences (Droste et al., 1989; Diehl et al., 1990; Njemanze, 1991; Kelley et al., 1992; Thomas and Harer, 1993). Markus and Boland (1992), however, also showed lateralization on an individual basis with this technology. Using bilateral TCD monitoring in right-handed persons, Rihs et al. (1995) found an unexceptional velocity shift to the left hemisphere during language tasks in comparison with visual-spatial tasks of 20 seconds' duration.
These authors demonstrated that 1 of 14 (right-handed) subjects failed to lateralize CBFV to the left side when a reading task (reading aloud) was compared with the baseline condition instead of with the visual-spatial task. Our results also show 1 of 15 right-handed subjects lateralize CBFV to the right during reading aloud. Because this finding was reproducible in all three conditions of reading aloud, it is most likely not an artifact. On the other hand, the same individual showed significant lateralization to the left when reading silently. A possible explanation would be that areas predominantly involved in motor aspects of speech are situated on the right hemisphere in this individual, whereas other linguistic areas are on the left side. No other subject, including the left-handed individuals, however, showed such dissociation. Three of ten left-handed subjects, however, showed no lateralization or a shift to the right, suggesting right-hemispheric localization of relevant linguistic areas. This percentage compares well with the results of the Wada procedure in left-handed individuals (Dinner, 1991).
In general all of our results were well reproducible, e.g., the subjects always showed lateralization to the same side in all tasks. Thus, our study adds further evidence that simultaneous bilateral TCD monitoring—backed by computerized data acquisition and averaging—is a promising tool to evaluate noninvasively and easily the lateralization of speech. Even a simple stimulus such as our repetitive reading tasks seems to allow reproducible lateralization of language in most subjects. Congruence of lateralization in reading aloud and reading silently may be a strong hint to the respective localization of the relevant areas and could be helpful in patient management, e.g., in preoperative decision making. It would be desirable to use the Wada amobarbital test as a gold standard to prove this hypothesis; however, this is impossible in volunteers for ethical reasons. Thus, a prospective comparison of similar TCD data and the results of the Wada procedure, e.g., in patients awaiting epilepsy surgery, should be performed to further evaluate the potential of this method.
