Abstract
The major classes of glial cells, namely astrocytes, oligodendrocytes, and microglial cells were compared in parallel for their susceptibility to damage after combined hypoxia and hypoglycemia or hypoxia alone. The three glial cell types were isolated from neonatal rat brains, separated, and incubated in N2/CO2-gassed buffer-containing glucose or glucose substitutes, 2-deoxyglucose or mannitol (both nonmetabolizable sugars). The damage to the cells after 6 hours' exposure was determined at 0, 1, 3, 7 days based on release of lactate dehydrogenase and counting of ethidium bromide–stained dead cells, double-stained with cell-type specific markers. When 2-deoxyglucose replaced glucose during 6 hours of hypoxia, both oligodendrocytes and microglia rarely survived (18% and 12%, respectively). Astroglia initially increased the release of lactate dehydrogenase but maintained 98% to 99% viability. When mannitol, a radical scavenger and osmolarity stabilizer, replaced glucose during 6 hours of hypoxia, oligodendrocytes rarely survived (10%), astroglia survival remained at 99%, but microglia survival increased to 50%. After exposure to 6 and 42 hours, respectively, of hypoxic conditions alone, oligodendrocytes exhibited 10% survival whereas microglia and astroglia were only temporarily stressed and subsequently survived. In conclusion, oligodendrocytes, then microglia, are the most vulnerable glial cell types in response to hypoxia or hypoglycemia conditions, whereas astrocytes from the same preparations recover.
All brain cells absolutely require oxygen and glucose, among many other crucial factors necessary for survival and to perform normal functions. The pathologic conditions created after oxygen and glucose deprivation in focal and global forms of ischemia, such as stroke, heart attack, and hypoxia, indicate that subsets of neurons are the most sensitive elements and are destroyed on the front line of injury. Later, other neurons undergo delayed neuronal cell death mediated by glia (Giulian and Vaca, 1993). Glial cells are thought to resist ischemic death, based mainly on the studies of cultured astrocytes (Kelleher et al., 1993; Swanson et al., 1993) and immunostaining studies of astrocytes (Endoh et al., 1994) and microglia (Morioka et al., 1993) after cerebral stroke. However, all brain cells are affected even if they survive the initial damage. The in vivo events initiated after ischemic episodes involve edema, breakdown of the blood–brain barrier, invasion of leukocytes, depletion of ATP, production of nitric oxide, formation of oxygen radicals, and changes in ion concentrations affecting the buffering of potassium, calcium regulation, and pH. In addition, there is release of cytokines, proteases, neurotransmitters, and excitatory amino acids, primarily glutamate. Astrocytes and microglia cells are activated and known to contribute in these myriad processes.
Astrocytes that bridge between the vascular system, neurons, and themselves detect perturbations in the microenvironment and undergo changes involving morphologic features, proliferation and pathologic conditions such as gliosis (for review see Eddleston and Mucke, 1993). Endothelial cells at the blood–brain barrier lose the integrity of tight junctions, thus allowing blood-born cells to invade the brain tissue. The infiltrating blood-borne macrophages, in concert with the resident microglia, undergo morphologic and functional changes and migrate to the site of injury, which is described as “activation.” In addition, these immunocompetent cells, including macrophages and microglia, mount an inflammatory response (for review see Kreutzberg, 1996). The response of the third major glial cell type, the oligodendrocyte, has not been clearly defined. However, their vulnerability seems likely, since ischemia damages white matter as well as gray matter.
Defined populations of glial cells as a model to study selective vulnerability in ischemia
The direct effects of oxygen and glucose deprivation on individual cell survival can be best tested in vitro. It is nearly impossible to discern cell type and specific factor contributions after injury without attempts to simplify the brain environment. On the other hand, because of the complexity of the cellular interaction in the tissue, the information gleaned from cells in culture must relate back to the tissue and whole animal.
Many aspects of nervous system toxicity caused by ischemia have been analyzed in vitro with good correlation to in vivo processes. Cultured astrocytes and those in vivo exhibit the same input resistance during ischemic injury (Kraig and Chesler, 1990). Lactacidosis damage to astrocytes is similar in the time course and lowering of pH in both tissue culture and in vivo experiments (for review see Plum, 1993). Granulocyte-macrophage colony-stimulating factor, stimulated after brain injury, has been shown to expand microglia populations both in vivo and in vitro (Giulian and Ingeman, 1988). Nitric oxide alone is toxic to oligodendrocytes and neurons in culture but is only one of many factors producing deleterious effects in vivo (Mitrovic et al., 1995). The concept of excitotoxicity with glutamate as a major component of toxicity after ischemia was deduced after studying neurons in a defined environment, such as cultures of spinal cord or cortex (Regan and Choi, 1991). Since these cultures included glial cells, primarily astrocytes that survived the combined hypoxia and hypoglycemia, it was evident that they were more resistant to paradigms of ischemic damage than neurons (Goldberg and Choi, 1993). In contrast, the susceptibility of the other glial populations, particularly microglial and oligodendrocytes, has not been studied in detail.
Goal of the study
Cultures of purified glial cell types of the mammalian brain have not been simultaneously or rigorously investigated. In the current study, we made use of protocols to isolate the three major glial cell types of the brain: oligodendrocytes, astrocytes and microglia. The cells were purified from the same material of neonatal rat brains and cultured as highly enriched populations. We used cell-type specific markers to unequivocally identify the cells (see Methods). This approach allowed us to selectively test the individual glial cell types for vulnerability after exposure to combined hypoxia and hypoglycemia conditions. We are aware that such an approach is far from the in vivo situation. Such studies provide essential information at the single-cell level and may ultimately help to understand the complex interactions followed by ischemic damage in vivo.
METHODS
Glial culture preparations
Cultures were prepared from whole brains of neonatal Wistar rats (Charles River, Inc., Sulzfeld, Germany) in the manner of McCarthy and deVellis (1980). Briefly, meninges were removed, and the dissociated brain was trypsinized (1.0% trypsin, 0.05% DNase [Worthington Biochem. Corp., Freehold, N.J., U.S.A.]) at room temperature in Hank's balanced salt solution (HBSS) without Ca2+ and Mg2+. The tissue was washed twice with HBSS, resuspended in basal eagle's medium containing DNase (0.05% and 0.25% glucose) and triturated through Pasteur pipettes to yield single cells. The cell suspension was centrifuged at 800 rpm for 10 minutes at 4°C and washed twice with ice-cold HBSS. After resuspension in basal eagle's medium plus 10% heat-inactivated horse serum (Gibco, Gaithersburg, MD, U.S.A.), cells of approximately two brains were plated per poly-
Hypoxia and hypoglycemia protocol
All experiments were performed with the following assay buffer containing (in millimolar units) 124 NaCl, 4.9 KCl, 1.3 MgSO4–7H2O, 2.0 CaCl2–2H2O, 1.2 KH2PO4, 25.6 NaHCO3, 10 glucose at pH 7.4, and were filter-sterilized. The hypoglycemic buffer was the same, except that the glucose was replaced by 10 mmol/L 2-deoxyglucose or 10 mmol/L mannitol.
Cells exposed to three different experimental conditions are schematized in Fig. 1A, parts 1 through 4. Part 1: A combination of hypoxia and hypoglycemia is depicted. Glia deprived of both oxygen and glucose in the assay buffer represents the hypoxic–hypoglycemic state. Glucose was replaced by either 2-deoxyglucose or mannitol. Part 2: Cells deprived only of oxygen in the presence of glucose represent the hypoxic state. Part 3: Cells exposed to normal atmosphere but glucose-deprived by the substitution of 2-deoxyglucose or mannitol in the buffer represent the hypoglycemic state. Part 4: Cells incubated with glucose and normoxic atmosphere represent the normal control state. Cultured glia exposed to hypoxia were placed in the gas-controlled hypoxia chamber (Fig. 1B), which, along with the cells under normoxic atmosphere, were placed in the incubator for 6 or 42 hours.
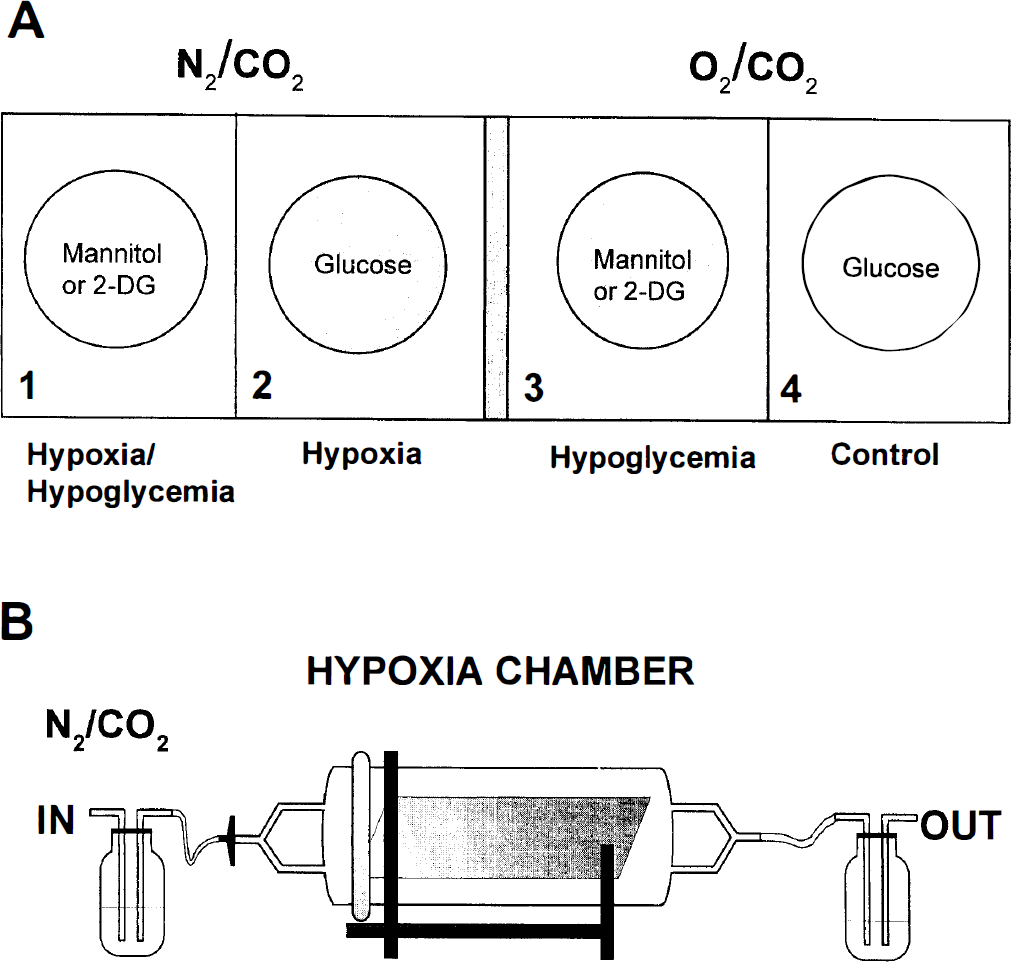
Experimental paradigm. Equal numbers of coverslips from each of oligodendroglia, microglia, and astrocytes were cultured from the same rat brain preparation. Cultures of these glia were chosen for matching density and exposed to combined hypoxia and hypoglycemia (
In each experiment, coverslips exposed to hypoxia or hypoglycemia always were compared with controls. Care was taken that the coverslips within one experiment started out with a similar cell density and purity as judged by morphologic criteria. After the experiment and at each time point, cells were stained with cell-type specific markers, described in section on immunostaining procedures. Employing live and dead labeling techniques, we were able to selectively count the populations of live and dead astrocytes, oligodendrocytes, and microglial cells. Additional experiments measuring lactate dehydrogenase (LDH) release yielded information about the stress that the cells had encountered.
The easily constructed noncommercially available hypoxia chamber was used for all hypoxia experiments (Fig. 1B). This closed system chamber fit into a standard cell culture incubator. A constant flow of N2/CO2 and 37°C were maintained throughout all experiments.
Hypoxia and hypoglycemia were induced under sterile conditions on cells in four-well plates that had been rinsed twice with buffer that had been gassed with 95% N2/5% CO2 for a minimum of 1 hour. One hour gassing was sufficient to remove 96% to 97% of the partial oxygen tension. Control buffer was gassed with 95% O2/5% CO2 for 1 hour. Under more stringent gassing for 4 hours or overnight, all but 0.3% oxygen tension could be removed, as indicated by a P
At the end of the experiment, cells were removed from the chamber and 2 × 100 μL supernatant removed from each well for colorimetric measurement of LDH activity. The remaining assay buffer was aspirated off and replaced by defined SATO medium with glucose but without serum, since this interferes with the LDH determination (recommendation of the LDH kit, Boehringer Mannheim, Mannheim, Germany). The remaining unstained coverslips were returned to normal atmosphere in the incubator, awaiting their time for live/dead determination.
For the hypoxia-alone experiments, glial cells were placed in the hypoxic chamber for 6 or 42 hours, removed for either immediate staining of live and dead cells, or returned to the normoxic incubator to assess the long-term effects of the hypoxia insult. Culture medium appropriate for each cell type containing glucose and serum was used for hypoxic experiments assessing cell survival, based on counting only. This ensured that observed changes would be from the effects of hypoxia alone. In other experiments for hypoxia alone, the cells were in the buffer described earlier to determine the LDH release without serum.
Immunostaining procedures
The purity of the cultures and identification of the glia were confirmed by staining with cell-type specific markers. The O4 surface antigen (Boehringer Mannheim) was used to identify oligodendrocytes. This antigen recognizes late precursor and mature oligodendrocytes of the oligodendrocyte lineage (for simplicity, these cells are referred to as oligodendrocytes). After incubation with secondary antibodies, dichlorotriazinyl amino fluorescein–conjugated or Cy3-conjugated μ chain–specific (both from Dianova, Hamburg, Germany) cells were fixed with 4% paraformaldehyde for 10 minutes before embedding in mowiol (Hoechst, Frankfurt, Germany). These oligodendrocyte cultures were 80% to 90% O4+.
Glial fibrillary acidic protein (GFAP; DAKO, Hamburg, Germany) was used to identify mature astrocytes. Cells were fixed with 4% paraformaldehyde for 10 minutes at room temperature. After rinsing, the primary polyclonal antibody against GFAP diluted in phosphate-buffered saline containing 0.1% Triton, 1% bovine serum albumin, and 1% normal goat serum was applied for 40 minutes. After washing, dichlorotriazinyl amino fluorescein–conjugated or Cy3-conjugated secondary antibodies were applied for 40 minutes and mounted on coverslides. About 80% of cells in the enriched astrocyte cultures were GFAP+, the rest being immature GFAP– cells, which were not counted.
Fluorescein isothiocynate–conjugated isolectin B4 from Griffonia simplicifonia (Boehringer, Mannheim) was used to identify microglia by direct labeling. Isolectin was incubated with microglia cultures for 15 minutes, rinsed, fixed, and mounted, as described earlier. The typical microglia morphologic feature was the rounded, amoeboid, and flat pancake-like shapes easily distinguished from the 1% and fewer of other cells present in culture.
Assessment of glia injury
A Live and Dead Kit (Molecular Probes, Leiden, Netherlands) was used to determine cell viability and cytotoxicity on glial cells after the hypoxia experiments in the presence of serum. Calcein AM was taken into live cells and remained as a marker for internal esterase activity and membrane integrity. At the same time, ethidium bromide homodimer-1 (EtBr), a nucleic acid stain, was taken into cells when the membrane was compromised. Each reagent was diluted together in the medium and applied to the cells immediately after exposure to hypoxia or specified time points for 5 minutes. The cells were fixed, mounted, and counted later without loss of fluorescence. The staining was stable for the times investigated.
A LDH cytotoxicity kit (Boehringer Mannheim) was used to quantify cell cytotoxicity based on membrane permeability. Permeabilized cells release a stable cytoplasmic enzyme, LDH, into the supernatant as an indicator of being damaged or lysed. One advantage of this assay was the quick determination of total cellular damage compared with time-intensive counting of individual cells. Another advantage was the ability to sample the medium during a 1-week time course from the same cells. Optimal cell density of each glial cell type was determined for detection of LDH under control conditions before plating the cells for the experiments. Aliquots of supernatant were removed from wells at specified times and frozen for LDH assay. The amount of LDH released then was compared with these controls for each of the experimental conditions, times, and cell types. Since there was a continual loss in cell numbers during the week, the typical normalization procedure was not practical for these experiments. To determine total LDH content, dimethyl sulfoxide can be applied to lyse the remaining cells, but this was impractical because dimethyl sulfoxide does not allow cells to be stained and counted at the end of the experiments.
RESULTS
Oligodendrocytes and microglia are susceptible to hypoxic damage in the presence of 2-deoxyglucose
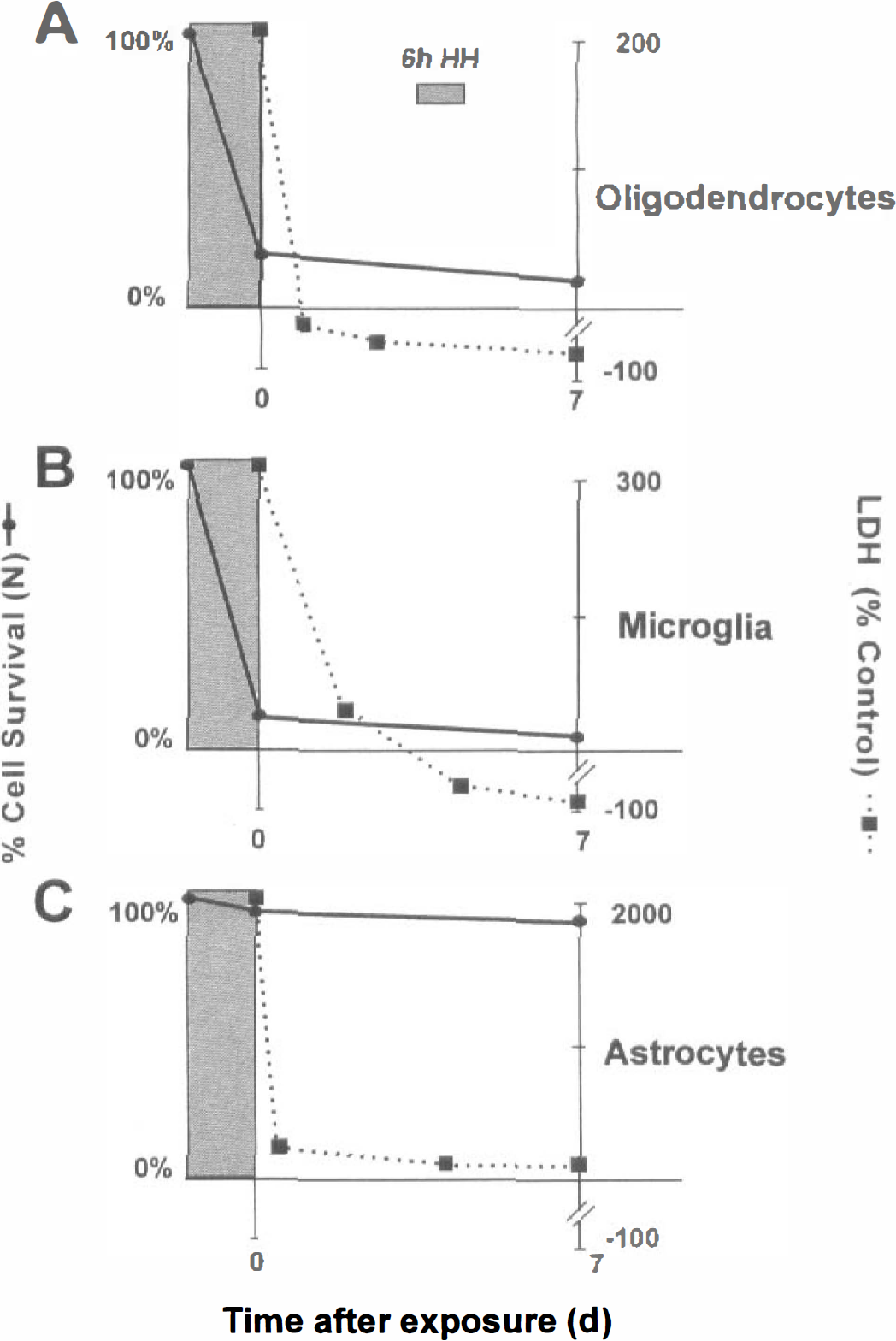
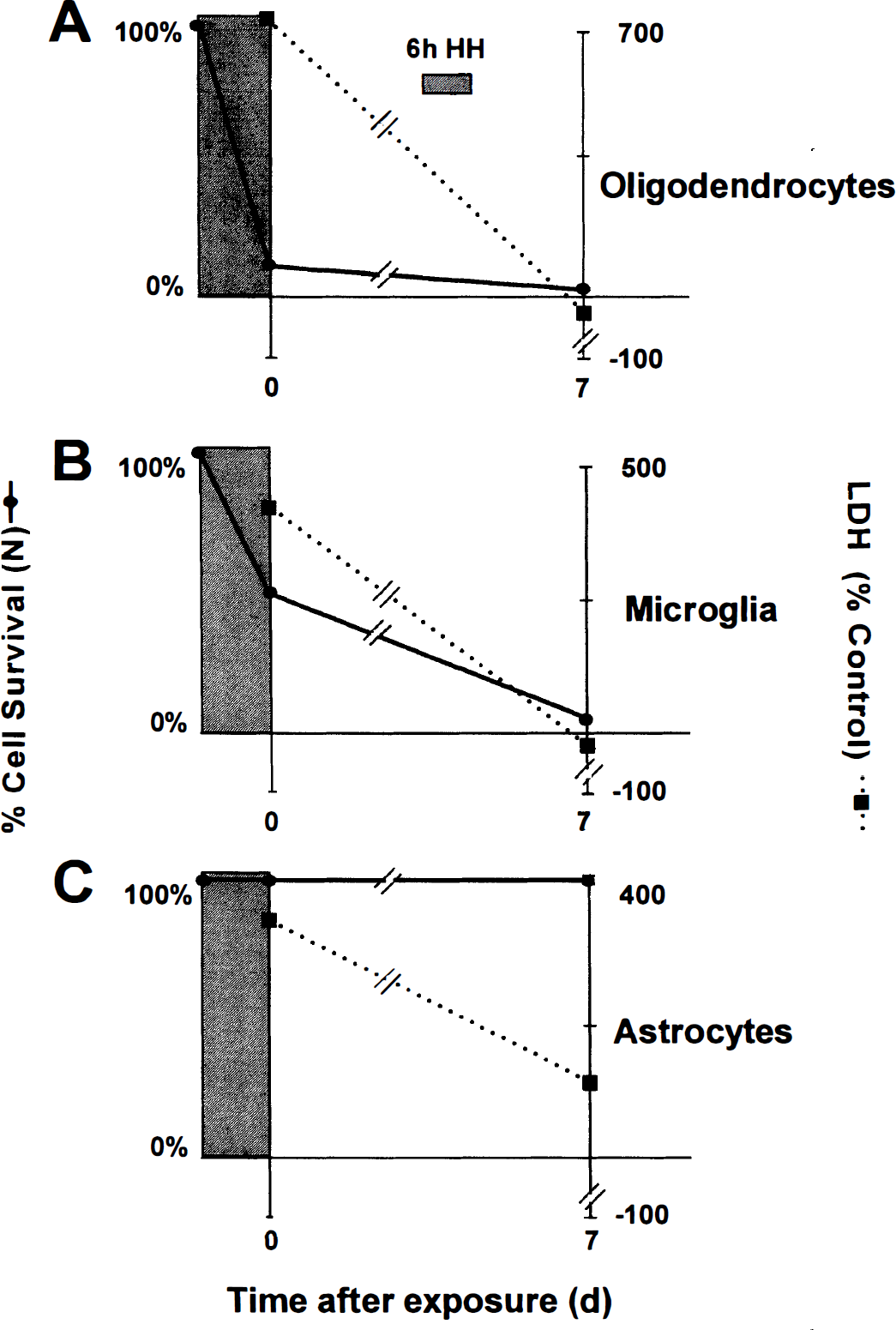
Oligodendrocytes were demonstratively damaged when exposed to 6 hours' combined hypoxia and hypoglycemia when glucose was replaced with 2-deoxyglucose. Immediately after the insult (t=0), nearly all cells (82% ± 5%, 390 of 476) were EtBr+, indicating that they were dead. Dead O4+/EtBr+ oligodendrocytes are shown (Fig. 2, open arrows) except for one live O4+/EtBr– cell (Fig. 2, solid arrows, top row). Even the few EtBr– cells underwent morphologic changes: the extensive arborization of the processes, typical for healthy oligodendrocytes, had deteriorated. The remaining coverslips were maintained for 7 days in the incubator in glucose-containing medium. At the end of 7 days, few -4–labeled cells were present, and of those, 90% ± 4% (185/207) did not survive (all EtBr+ cells; Fig. 2, second row). Many oligodendrocytes survived in the control cultures at t = 0 and 7 days (Fig. 2, solid arrows for O4+/EtBr– cells, bottom rows), although the total number of cells decreased with time in culture. Cell survival percentages based on all oligodendrocytes counted are shown in Fig. 3A. Release of LDH increased compared with the controls right after the insult. Monitoring the LDH release after 1, 3, and 7 days revealed LDH levels to be below control levels (Fig. 3A). This might reflect that only few cells survived on the coverslips as a potential source to release LDH.
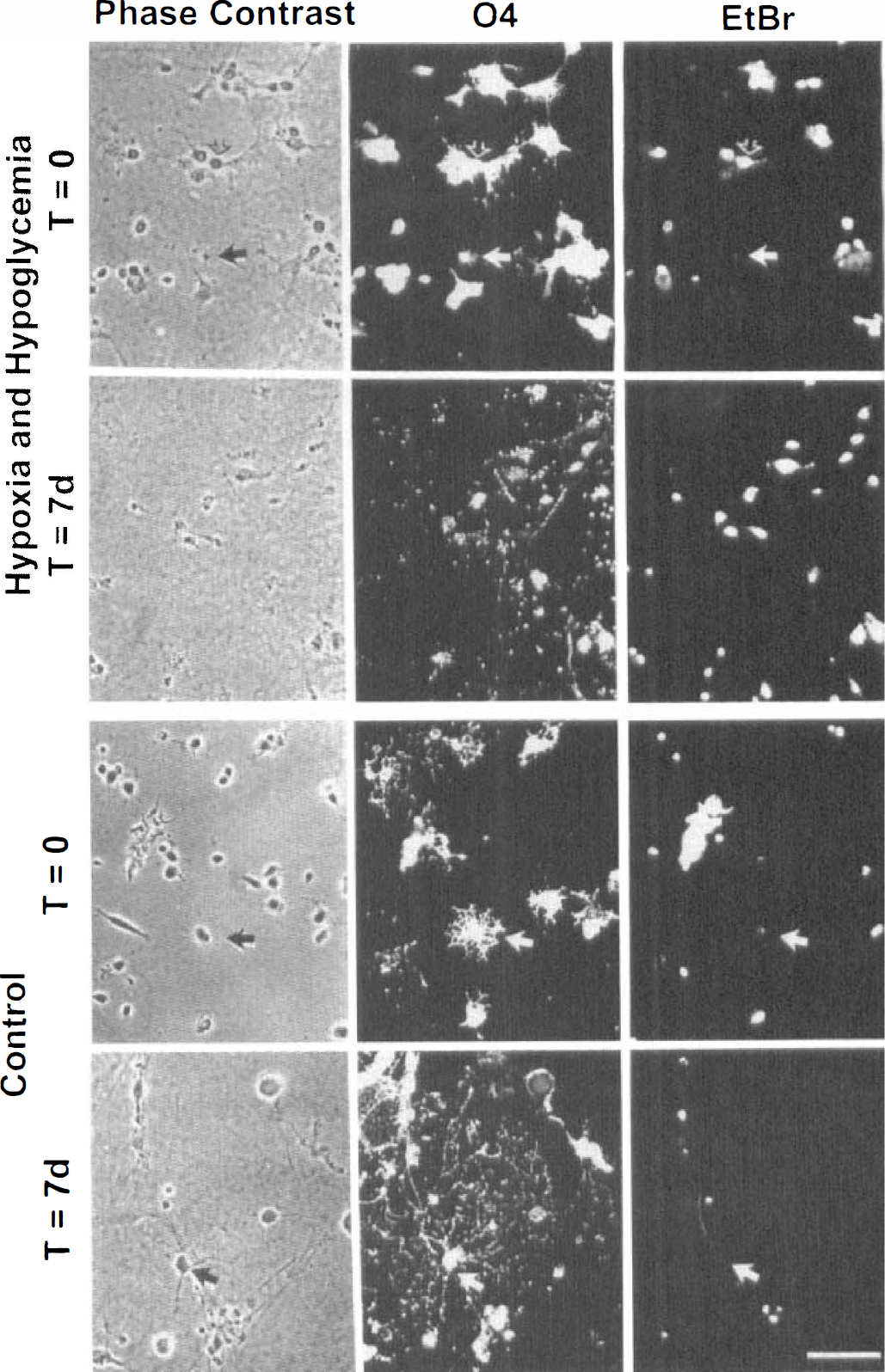
Cultured oligodendrocytes exhibit low survival after 6 hours' exposure to combined hypoxia and hypoglycemia in the presence of 2-deoxyglucose. The phase contrast, O4 immunostaining of oligodendrocytes and the EtBr staining of dead cells are shown in the top row of photomicrographs, immediately after 6 hours' exposure (t=0). Solid arrows point to live cells, and open arrows point to dead cells. The next row of photomicrographs shows oligodendrocytes from the same experiment but 7 days after the insult (t=7 days). The bottom two rows show control cultures of oligodendrocytes from the same experiment and preparation at 0 and 7 days after the 6 hours' exposure. All photomicrographs are at equal magnification. Scale bar = 60 μm.
Glial survival after 6 hours' combined hypoxia and hypoglycemia with 2-deoxyglucose substituted for glucose. (
Microglia also were susceptible to combined hypoxic and hypoglycemic damage in the presence of 2-deoxyglucose. The positive EtBr staining right after the insult indicated that 88% ± 6% (407/464) of the ILB4+ cells did not survive (Fig. 4B, all cells, third row). Seven days after the insult, the combined EtBr and ILB4 labeling indicated that the percentage of dead cells increased (93% ± 2%, 271/291; Fig. 4B, last row). The dead cells did not wash away, and the ILB4 staining on these cellular remains was weak and the cell boundaries were hard to discern (Fig. 4B, open arrow, last row). Photomicrographs of control cultures exhibited lack of EtBr label at t = 0 and 7 days (not shown, but similar to the controls for microglia under the mannitol conditions; Fig. 6). The small flecks seen in the photomicrographs are bubbles from mounting medium, not contamination of cells. As observed similarly for oligodendrocytes, microglia total survival and LDH release are shown in the graph (Fig. 3B). Release of LDH right after the insult increased threefold and fell below control levels after 3 days (Fig. 3B).
Cultured oligodendrocytes exhibit low survival after 6 hours' exposure to combined hypoxia and hypoglycemia with mannitol present. (
Astroglia recover after exposure to combined hypoxia and hypoglycemia in the presence of 2-deoxyglucose
Astroglia survived, as expected, after combined hypoxia and hypoglycemia treatment while in the presence of 2-deoxyglucose and in all other conditions (not shown). After 6 hours' exposure, few of the GFAP+ cells (5% ± 0.05%, 32/656) were EtBr+, indicating that most cells were alive (photomicrographs are not shown). Seven days after the 6-hour insult, cell numbers had not changed. The total number of surviving astrocytes did not change over the 1-week period (Fig. 3C). Surprisingly, the LDH release measured immediately after the insult strongly increased, showing similarities to the experiments with microglia and oligodendrocytes. A small difference in LDH release became apparent later after 1, 4, and 7 days in that the LDH levels remained higher than control levels (Fig. 3C). After longer (12-hour) exposure times, astrocytes were destroyed (data not shown).
Half of the microglial population survived in the presence of mannitol
The combined hypoxia and hypoglycemia experiments in the presence of 2-deoxyglucose (as described earlier) were repeated in a similar series of experiments where glucose was replaced by mannitol. Mannitol has properties as a radical scavenger and is thought to play a role in cell volume control, but these effects are not universal for every cell type (Gillbe et al., 1996).
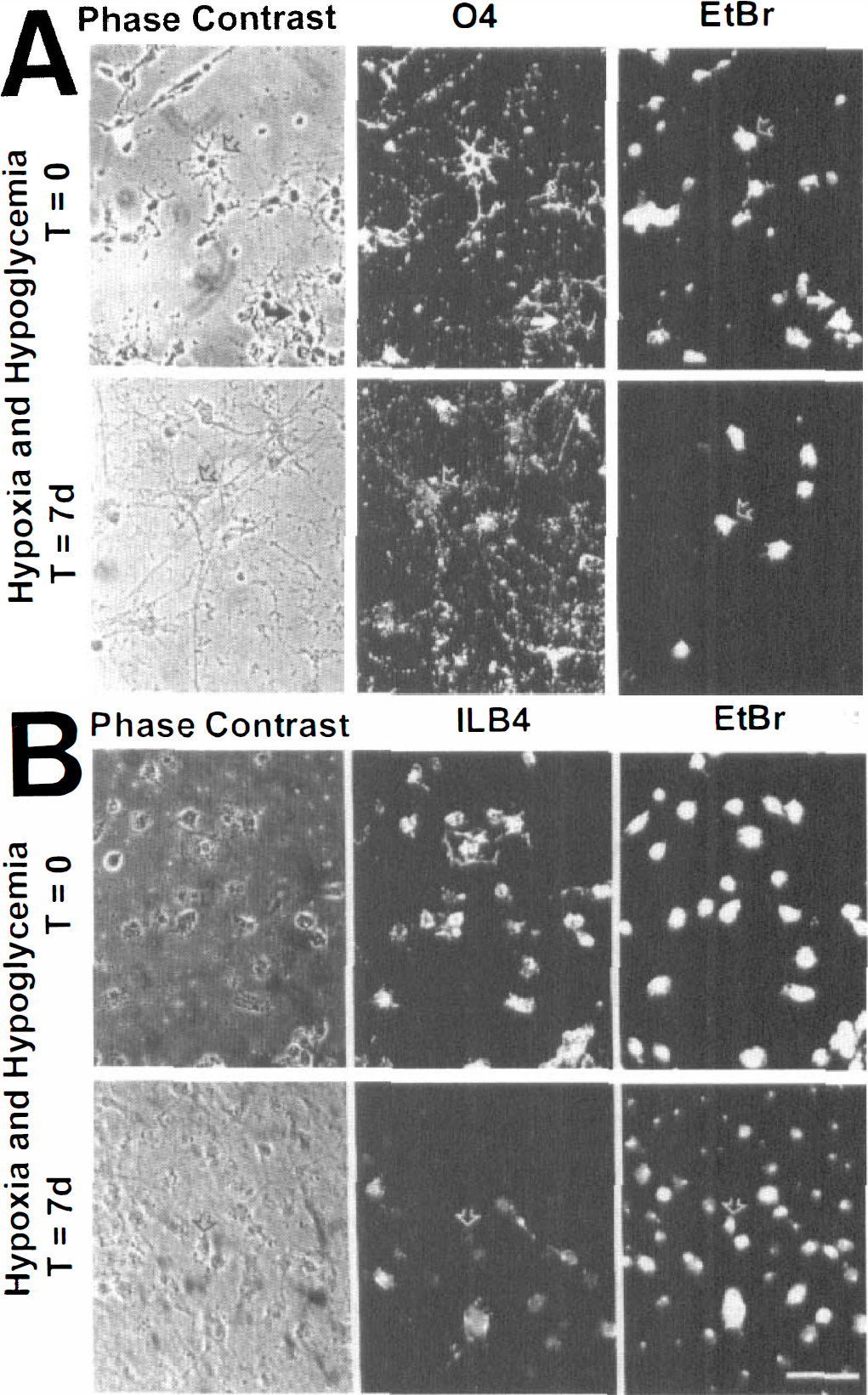
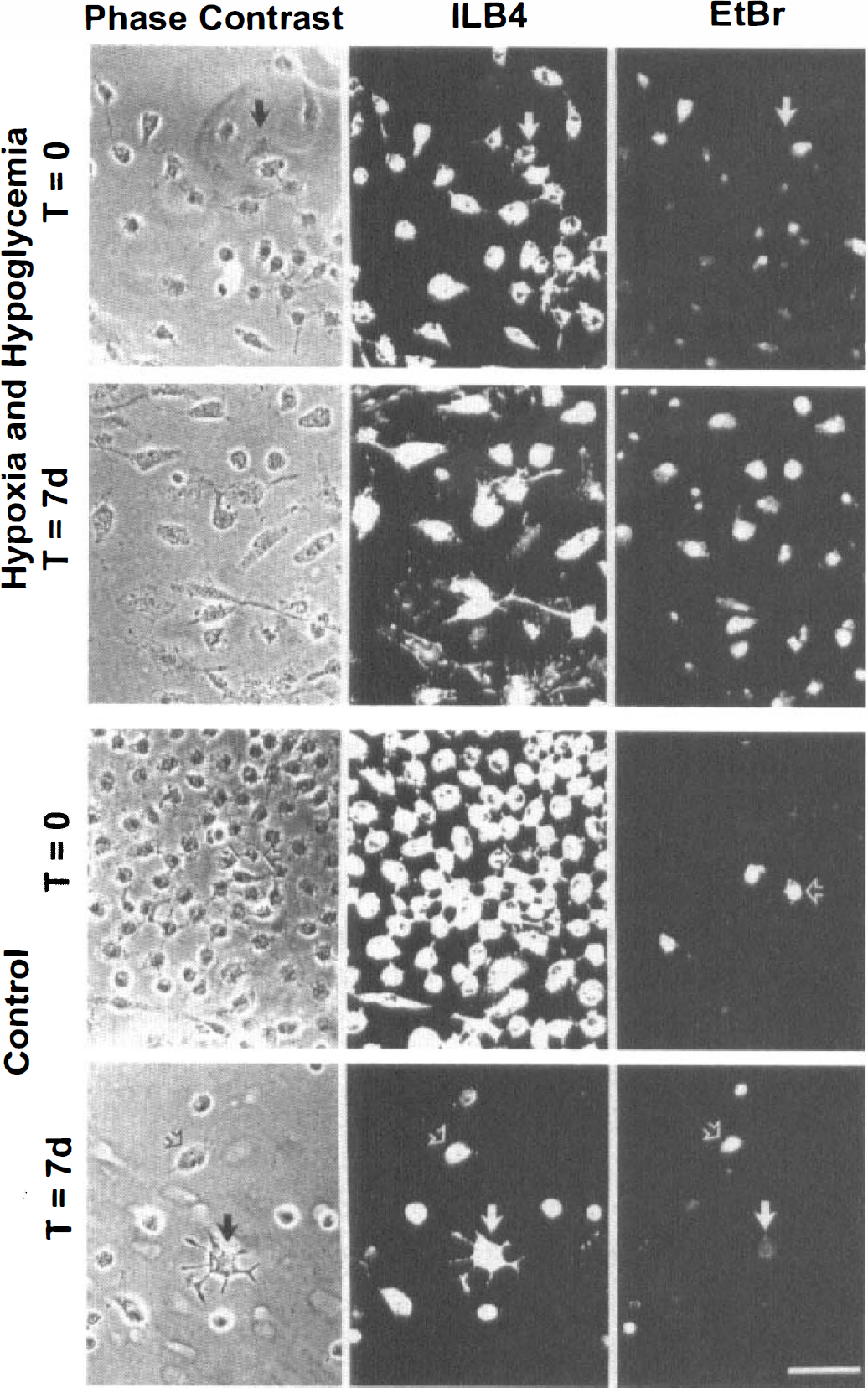
For oligodendrocytes and astrocytes, there were few differences in survival in the presence of mannitol when compared with 2-deoxyglucose results. Right after the insult at t = 0, 10% ± 3% (51/529) of oligodendrocytes survived as shown (Fig. 4A, solid arrows, O4+/EtBr–, top row). After 7 days, 95% ± 1% (330/347) of the 04+ cells were EtBr+ and had not survived (Fig. 4A, open arrows, second row). In control cultures from the same experiment, many live O4+/EtBr– cells were present (not shown, since control cells were similar in both 2-deoxyglucose and mannitol conditions). Total oligodendrocyte percentage of survival after 6 hours of combined hypoxia and hypoglycemia and concomitant LDH release for up to 7 days is shown in Fig. 5A.
Glial survival after 6 hours' combined hypoxia and hypoglycemia with mannitol substituted for glucose. (
Cultured microglia exhibit about 50% survival after 6 hours' exposure to combined hypoxia and hypoglycemia with mannitol present in the buffer. The phase-contrast ILB4+ microglia and EtBr+ dead cells are shown in the top row of photomicrographs immediately after 6 hours' exposure. The next row shows cells from the same experiment at the end of 7 days (examples of live cells are indicated with solid arrows). The lower two rows show examples of cultured microglia cultures from the control experiments at 0 and 7 days after 6 hours' exposure (EtBr+ cells are shown with open arrows). All photomicrographs are at equal magnification. Scale bar = 60 μm.
As for astrocytes, most survived immediately after the insult (99% ± 1%, 1381/1395) and the cell density was not affected 7 days afterward (not shown). Similarly, as in the presence of 2-deoxyglucose, total astrocyte and oligodendrocyte survival and the LDH release followed similar patterns after the insult (Fig. 5A and Fig. 5C). The LDH remained above control levels after 7 days in the astrocytes but was lower than controls in the experiments with oligodendrocytes. Thus, the overall impact on the astrocytes and oligodendrocytes was not substantially changed in any of the tested parameters when comparing 2-deoxyglucose and mannitol substitution.
However, the response of the microglia after the insult was different in the presence of mannitol in comparison with 2-deoxyglucose. In the presence of mannitol, about half of the microglial population survived the 6 hours of combined hypoxic and hypoglycemic treatment (49% ± 15%, 1116/2282 cells, n=5). This finding was based on the total number of ILB4+ cells that did not express EtBr staining (Fig. 6, solid arrows point to examples of live cells, top row). However, 7 days after the insult, there were few surviving microglial cells (3% ± 0.05%, 52/1745, all EtBr+, second row). In most of the dead cells, the nucleus appeared condensed, emphasized by the intensity of the EtBr staining (about 20 of 30 cells; Fig. 6, second row). Control cultures of microglia conversely exhibited only a few EtBr+ cells (Fig. 6, open arrows point to examples of dead cells, bottom rows). Total cell survival is shown, as well as the LDH release for both t = 0 and 7 days (Fig. 5B). The LDH release increased initially and decreased at 7 days in the same way with mannitol as with LDH release of microglia in the presence of 2-deoxyglucose (Fig. 5B).
Oligodendrocytes but not microglia exhibit increased cell death after hypoxia exposure
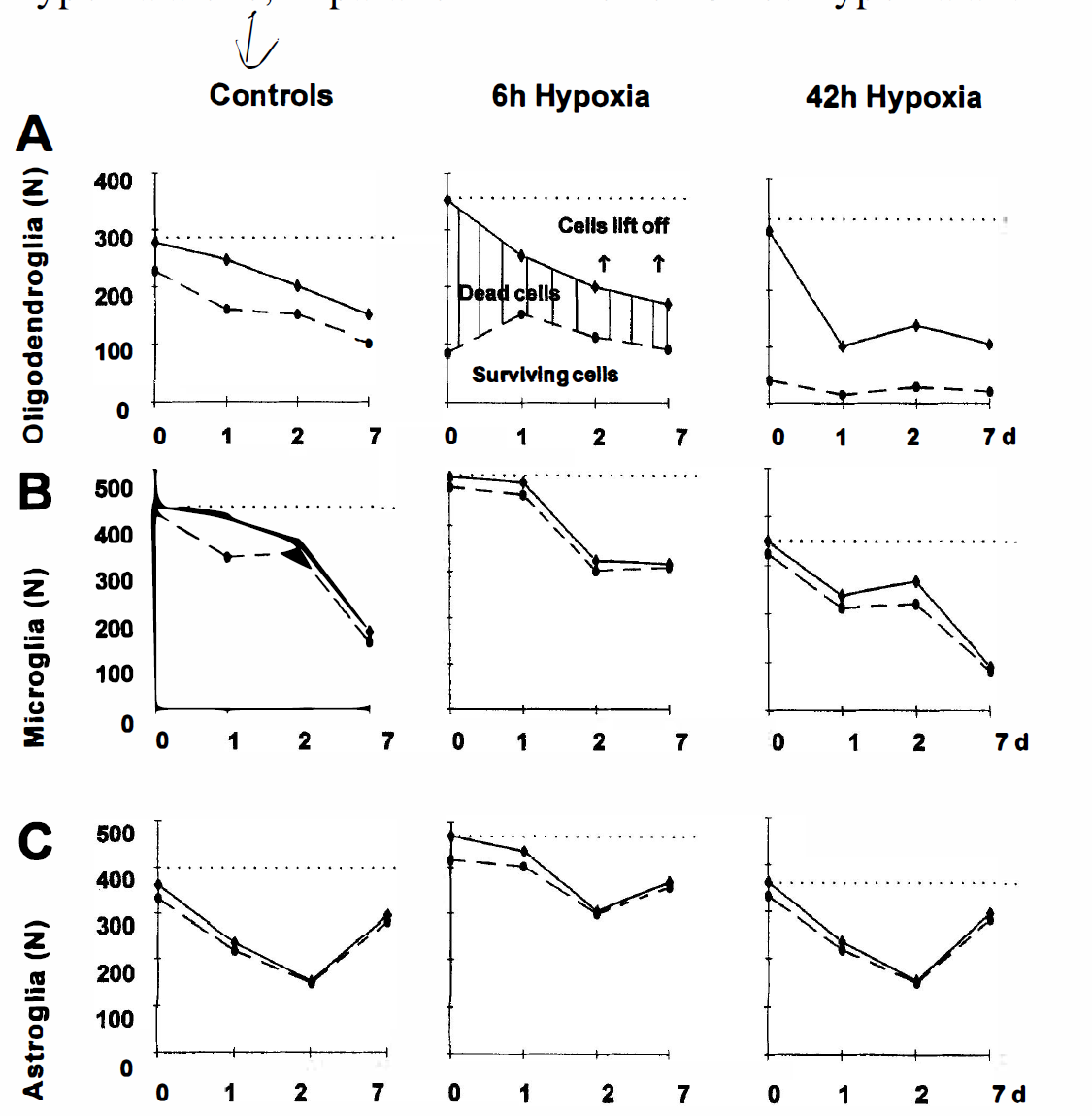
Oligodendrocytes were shown to be more susceptible to damage from hypoxia than microglia and astroglia, as evidenced by decreased cell survival after 6 hours' exposure time (middle column) when compared with the controls (Fig. 7A, first column). Immediately after 6 hours' exposure to hypoxia alone, only 24% of O4+ cells (85/353) survived. Seven days after the insult, the total cell number had decreased to about half of the original numbers, and of these, 54% (92/171) cells survived. We found a similar overall decline in cell number of the controls within 7 days. Under hypoxia-alone conditions, the LDH release for oligodendrocytes was only slightly less than combined hypoxia and hypoglycemia at all time points (data not shown).
Cultured oligodendrocytes (
Microglia survival was not as affected as oligodendrocyte survival after 6 hours of hypoxia when compared with the controls (Fig. 7B). The percentage of surviving cells at this first time point after 6 hours' hypoxia was 95% (485/508). Although microglia appear to be resistant to hypoxia death in culture, the total number of cells decreased over 1 week in culture, whether or not they were exposed to hypoxia for 6 hours. The LDH release for microglia was significantly less at all time points compared with combined hypoxia and hypoglycemia (data not shown).
Similarly, astrocytes appeared to be resistant to 6 hours' hypoxia in culture as seen in Fig. 7C, since the percentage of cell death after 6 hours' exposure to hypoxia was low and comparable with the control. The percentage of surviving cells immediately after 6 hours' hypoxia was 89% (417/470). Although the total number of cells varied at each time point, all increased in total cell number at 7 days. Under conditions of 6 hours' hypoxia alone, in parallel with the combined hypoxia and hypoglycemia experiments (without serum), the LDH release for astroglia also was significantly less at all time points (data not shown).
Long-term hypoxia resulted in the same survival pattern of glial subtypes
A 42-hour exposure of oligodendrocytes to hypoxic conditions resulted in severe damage in every experiment. Only 13% (40/306) of the cells were calcein green fluorescent (e.g., alive; Fig. 7A). Astrocytes and microglia, in contrast, were not effected concerning survival, after 42 hours' exposure to hypoxia when compared with controls (Fig. 7B and Fig. 7C).
Hypoglycemic conditions for 6 hours did not affect the survival of the different glial cell types
Glial cells were maintained in buffers for 6 hours at normoxic atmosphere where glucose was either replaced by mannitol or 2-deoxyglucose. Neither oligodendrocytes, astrocytes, nor microglial cells showed different survival patterns from controls, as determined by EtBr labeling in concert with the cell-type specific markers, O4, GFAP, and ILB4, respectively. In all cases, the LDH release was the same as control levels (data not shown). The cell survival was followed for 7 days to analyze possible delayed long-term damage. We conclude that all brain glial cell types can survive 6 hours' glucose-free conditions in culture.
DISCUSSION
Glial cells in culture are selectively vulnerable
Glial cells are thought to be much more resistant to hypoxic and hypoglycemic stress compared with neurons. Typically, reports of glial survival or injury to damage during or after ischemic injury refers primarily to astrocytes (Goldberg and Choi, 1993; Nedergaard et al., 1991). Here we confirm that astrocytes are more resistant to in vitro ischemic damage than other glia under these experimental conditions. Astrocytes with their glycogen stores have a beneficial effect on neuronal survival under glucose deprivation in culture (Swanson and Choi, 1993) and for themselves as well. Thus, the plastic and surviving astrocytes may serve as important entities necessary for the survival of the other brain cells. Our comparative approach in assaying the survival of microglia and oligodendrocytes, however, revealed that these populations are more susceptible to such insults. Thus, there is no common glial survival rate, but a rather distinct one for astrocytes, oligodendrocytes, and microglia, respectively. In general, the most damage to glial cells occurred immediately after combined hypoxia and hypoglycemia rather than at later time points. Whether this cell death is apoptotic or necrotic currently is being investigated.
LDH release is not a reliable assay for measuring glia toxicity
Typically, the release of LDH has been used as a standard method to analyze the survival rate of neurons and other cell types after different types of insults. In many studies, the rate of released LDH correlates well with the loss of neurons (Goldberg and Choi, 1993). Our data indicate that this assay cannot be used to study comparative glial survival. Whereas astrocytes exhibit a dramatic release in LDH after the ischemic/hypoxic insult, their survival is not affected. Astrocytes were stressed at first, but this did not affect the overall surviving numbers of astroglia or the long-term basal release of LDH. If we relied solely on LDH release data, we would conclude that astrocytes were the most susceptible to injury by combined hypoxia and hypoglycemia when compared with microglia and oligodendrocytes, when the opposite is true. The LDH release did not correlate with number of dead cells counted after the insult. We, therefore, had to base our studies on counting cells after live/dead staining or EtBr uptake into dead cells. This serves as caution to others when considering LDH release assays to measure cytotoxicity.
Whereas the LDH assay seems inappropriate to monitor glial death, it contains valuable information. Measuring LDH release is a technique indicating the time course of stress of a total population of cells, including those that die and lift off into the medium, which would never be counted by a staining technique. The LDH release was high initially after the insult in all cells. After the initial determination and a 30-minute reperfusion with glucose and at later time points, the LDH level returned toward control. This corresponds with LDH determination after the hypoxia insults, when the greatest damage occurs to cells after ischemia during the reperfusion period when radicals and proteases are released (Mertsch et al., 1995). In the case of the oligodendrocytes and microglia, LDH release fell, even below the control levels. This may indicate that the injury to the glial cells occurs primarily at the initial time point or that the level of LDH in the surviving cells is below the sensitivity of the assay.
Oligodendrocytes are the most vulnerable glial cell type
Oligodendrocytes appear to be nearly as sensitive as neurons to perturbations of oxygen and glucose in comparison with microglia and astrocytes under the conditions implemented in our experiments. Neurons in culture demonstrate similar responses to damage by hypoxia or hypoglycemia as shown by Goldberg and Choi (1993). Husain and Juurlink (1995) and Qi and Dawson (1993) analyzed the effect of hypoxia on oligodendrocytes and described extreme vulnerability with or without coculturing with astrocytes. It was expected that the removal of glucose (substituted by 2-deoxyglucose or mannitol) under the hypoxic conditions would exacerbate the damage to oligodendrocytes, which indeed, was the case. Although mannitol confers protection in some cell types under various stresses (Gillbe et al., 1996), cultured oligodendrocytes did not show an increase in survival after hypoxia and hypoglycemia in the presence of mannitol.
Oligodendrocyte susceptibility to damage in culture by hypoxia and combined hypoxia and hypoglycemia is not surprising, since they are difficult to culture and to maintain under normal conditions. Oligodendroglia exhibit sensitivity to various kinds of damage in culture, as demonstrated by others. Nitric oxide, released from endothelia, astrocytes, and microglia after being triggered by stimuli such as ischemia, causes lipid peroxidation in oligodendrocytes, as well as necrotic cell death (Merrill et al., 1993). In situ demonstration of oligodendroglial paranodal loops detaching from rat optic nerve axons after 1 hour of anoxia indicates selective vulnerability (Waxman et al., 1994). Under extreme conditions, white matter may be damaged immediately after a focal stroke, but usually the breakdown of white fiber tracts is found at later stages of ischemia. Models of white matter tract ischemia show destruction in the white matter, implicating the involvement of oligodendrocytes (Ferrer et al., 1994). Unfortunately, few data are available on the survival of oligodendrocytes in vivo after such an insult. Juurlink (1997) suggests in a review that oligodendrocytes may be more susceptible to oxidative stress after ischemia because of their low glutathione contents—important in reactive oxygen scavenging—and their higher iron contents, contributing to increased radical formation. Mandai and coworkers (1997) demonstrated, in a mouse model of middle cerebral artery occlusion, oligodendrocyte degeneration within 12 to 24 hours. This was followed by subsequent proliferation from oligodendrocytes 5 days after damage near the ischemic core. It would be interesting to learn about the survival of these adult oligodendrocyte progenitor cells, since this small cell pool is thought to be a source for generating new oligodendrocytes (Noble et al., 1992).
Microglia survive partially in the presence of mannitol
Microglia are strongly damaged after experiencing an ischemic insult in culture, nearly as much as oligodendrocytes or neurons. The presence of mannitol, however, partially protected microglial cells but not oligodendrocytes. Microglia exhibited about 50% survival under hypoxia with mannitol present in comparison to less than 10% survival after hypoxic exposure in the presence of 2-deoxyglucose. It seems most likely that mannitol is a free radical scavenger and thus protects microglia from their own products. Indeed, stimulation of microglia with lipopolysaccharide leads to an oxidative burst (Banati et al. 1991). It has not been investigated whether the combined hypoxia and hypoglycemia insult results in a similar response, but if so, microglial cells would damage each other and themselves. This would explain the lack of mannitol effect on oligodendrocyte survival, since these cells are not known to generate oxidative bursts.
We were unable to find reports regarding microglia death in vivo or in situ. It would require elegant work with double markers such as shown for dying and proliferating oligodendrocytes in mouse brain mentioned above by Mandai and associates (1997). Susceptibility of microglia in culture would conceivably be greater and more easily detected than in vivo. Cultures of microglia after combined hypoxia and hypoglycemia may create an environment of increased nitric acid accumulation, as well as cytokines or proteases, which would increase self-toxicity. The authors are aware of ongoing discussions concerning the fate of microglia in pathologic states as yet unsupported by convincing data.
CONCLUSION
It is apparent from these studies that the initial events are the most important to characterize. Long-term survival of glia in culture after exposure to hypoxia or hypoglycemia depends on the number of cells surviving the immediate damage. Evidently, there appears to be no massive delayed cell death for glia in culture, as is known for neurons in vivo, particularly in the CA1 region of the hippocampus. Of course, glial cells grown in cell culture differ in important ways from their in vivo counterparts. Further investigation is required to understand the interactions between glia and neurons within the complex cascade of events in ischemia. Nonetheless, this simpler model system can be used to ask questions about neuronal/glia signaling systems that change in response to injury. Later, the answers to these questions may be tested at the next biological levels of complexity, such as cocultures and brain slices.
Footnotes
Acknowledgments
The authors thank Brigitte Hunger for providing glial cultures and Regina Krauß for excellent technical assistance.
