Abstract
The effect of thrombolytic therapy was studied in rats submitted to thromboembolic stroke by intracarotid injection of autologous blood clots. Thrombolysis was initiated after 15 minutes with an intracarotid infusion of recombinant tissue-type activator (10 mg/kg body weight). Reperfusion was monitored for 3 hours using serial perfusion- and diffusion magnetic resonance imaging, and the outcome of treatment was quantified by pictorial measurements of ATP, tissue pH, and blood flow. In untreated animals, clot embolism resulted in an immediate decrease in blood flow and a sharp decrease in the apparent diffusion coefficient (ADC) that persisted throughout the observation period. Thrombolysis successfully recanalized the embolized middle cerebral artery origin and led to gradual improvement of blood flow and a slowly progressing reversal of ADC changes in the periphery of the ischemic territory, but only to transient and partial improvement in the center. Three hours after initiation of thrombolysis, the tissue volume with ADC values less than 80% of control was 39 ± 22% as compared to 61 ± 20% of ipsilateral hemisphere in untreated animals (means ± SD, P = .03) and the volume of ATP-depleted brain tissue was 25 ± 31% as compared to 46 ± 29% in untreated animals. Recovery of ischemic brain injury after thromboembolism is incomplete even when therapy is started as early as 15 minutes after clot embolism. Possible explanations for our findings include downstream displacement of clot material, microembolism of the vascular periphery, and events associated with reperfusion injury.
Thrombolytic therapy of ischemic stroke has received considerable attention during the past years. The most convincing investigation in support of a beneficial effect of this treatment is the multicenter randomized, double-blind National Institutes of Health trial which documented a modest but significant improvement in clinical outcome (The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group, 1995). With the advent of new thrombolytic agents such as human recombinant tissue plasminogen activator (rt-PA), and the strict adherence to well-defined selection criteria, notably a time limit of 3 hours and the exclusion of intracranial bleedings, safety of thrombolytic therapy has been substantially improved.
The concept of thrombolysis is clearly a progress in the treatment of stroke, but the number of patients who benefit from this treatment is still rather small (Furlan and Kanoti, 1997). Therefore, criteria that might predict a beneficial outcome of thrombolysis are currently under intense investigation. A further approach towards an improvement of thrombolysis is to clarify the complex pathophysiology of the reperfusion phase after rt-PA treatment. To date, the limiting factors for thrombolytic therapy are only partially understood. It is obvious that failure of the thrombolytic agent to reopen the occluded artery would not result in the reversal of ischemic injury, but other factors such as microembolism, breakdown of the blood-brain barrier, or reperfusion injury (Phillis, 1994), may play an additional role.
Many experimental studies have been devoted to reperfusion injury in reversible models of stroke, such as transient intraluminal thread occlusion of the middle cerebral artery (MCA) (Koizumi et al., 1986). In particular, the enhancement of inflammatory processes after reperfusion of the brain has been shown (Jiang et al., 1995; del Zoppo, 1994). The contribution of oxygen free radicals has been concluded from observations in Cu-Znsuperoxide overexpressing mice which exhibited less severe injury than their nontransgenic littermates (Chan et al., 1995). The pathophysiology of rt-PA—induced reperfusion can be expected to be even more complex. In addition to the aforementioned mechanisms it must be considered whether rt-PA treatment itself might induce specific complications that counteract the beneficial effects of reperfusion.
In this study we investigated the time course and spatial pattern of thrombolytic reperfusion in a modified experimental model that produces consistent embolism and recanalization of the proximal MCA (Busch et al., 1997b). Rt-PA treatment was started 15 minutes after clot embolism to minimize the impact of the primary ischemic injury and to focus the study on the effect of thrombolytic reperfusion. Moreover, we combined the magnetic resonance imaging study with a series of pictorial hemodynamic and biochemical measurements to obtain detailed spatial information about the final outcome of the ischemic injury.
MATERIALS AND METHODS
Surgical preparation and clot embolism
All animal handling and surgery was performed in accordance with animal protection guidelines and approved by local authorities.
Male Wistar rats (n = 16, 350 to 400 g) were anesthetized with 1.5% halothane in a 70%/30% mixture of N2O/O2. Catheters were inserted into a femoral artery and a femoral vein for blood sampling, blood pressure recording, and intravenous infusion of drugs. The animals were tracheotomized, immobilized with pancuronium bromide (0.3 mg/kg/h intravenously) and artificially ventilated. After the start of mechanical ventilation halothane was reduced to 0.8%.
Thromboembolic stroke was produced using a modification (Busch et al., 1997b) of the method described by Overgaard et al. (Overgaard et al., 1992). Fibrin-rich autologous blood clots were prepared at the beginning of the experiment by injecting 0.6 mL arterial blood with 0.15 mL thrombin (1 mg/mL) into polyethylen tubes with an inner diameter of 0.58 mm. After termination of surgery, clots were removed from the catheter, rinsed in saline to remove adherent erythrocytes, and inspected under the microscope for selection of fibrin-rich segments. The clots were cut into small cylindrical segments with a diameter and length of 0.35 mm and 1.5 mm, respectively. Twelve of these clots were suspended in a solution of albumin and phosphate buffered saline (1 mg/mL) and stored until embolization (see below).
After surgical preparation of the right carotid artery and ligation of the pterygopalatine and occipital arteries, a polyethylen tube (inner diameter 0.58 mm) was inserted into the external carotid artery, with its tip being located close to the carotid bifurcation. This tube was continuously rinsed with saline 1 mL/h. Additionally, a 3-0 suture was placed around the common carotid artery and both ends guided through a firm teflon tube with its tip being fixed close to the artery. By pulling both suture ends the common carotid artery could be temporarily closed. The tube containing the blood clots was connected to the catheter in the external carotid artery. The clots were injected over a period of 30 seconds during which the common carotid artery was temporarily occluded.
After injection into the carotid artery the low-pressure clots, which are loose in composition and flexible in shape, are compressed by the arterial pressure and are densely packed at the MCA origin. Some of the injected clot material may not reach the circle of Willis, but it may lodge in the extracranial part of the carotid artery, i.e., around the tip of the tube or at the origin of the pterygopalatine artery. Therefore, a sufficient amount of clot material must be injected to ensure reliable reduction in regional CBF (rCBF).
Physiologic monitoring
Physiologic monitoring was performed throughout the experiment. Arterial blood samples were taken at regular intervals for measurement of blood gases (Blood Gas System 288, Ciba Corning Diagnostics, Fernwald, FRG), blood glucose (Glucose Analyzer 2, Beckman Instruments, Fullerton, CA), and hematocrit. Body temperature was recorded with a rectal thermometer and maintained at 37 °C with a feedback-controlled warm-water blanket. Arterial blood pressure was recorded continuously using a miniature pressure transducer (Braun, Melsungen, FRG) connected to a polygraph (Gould Oscillograph 3000, Gould Inc., Cleveland, OH).
Rt-PA treatment and experimental protocol
Treated animals received an intracarotid infusion of 10 mg/kg human single-chain rt-PA, Actilyse (INN: Alteplas, Boehringer Ingelheim, Germany) dissolved in 2 mL of distilled water. Infusion started 15 minutes after embolization and continued for 45 minutes. Human rt-PA is 10 times less efficient in rats than in humans (Korninger and Collen, 1981; Karges et al., 1994); therefore, the 10-fold human dose was given to assure reliable recanalization. Untreated animals received 2 mL saline during the same period.
All animals were fixed in a prone position in a non-magnetic stereotaxic head-holder for accurate positioning. Embolization and rt-PA treatment was performed inside the nuclear magnetic resonance (NMR) tomograph (see below). After the NMR measurements, 3 hours after embolization, treated and untreated (each n = 8) animals were frozen in liquid nitrogen for further processing of the brains (see below).
Magnetic resonance imaging
The NMR recordings were performed on a 200 MHz BIO-SPEC MSL X-11 system (Bruker, Karlsruhe, FRG) with a 30-cm bore magnet and actively shielded gradient coils (maximum 100 mT/m; risetime < 250 microsecond). The radiofrequency (rf)-pulses were transmitted using a 12-cm Helmholtz coil arrangement. The NMR signal was acquired with a 16-mm diameter inductively coupled surface coil placed over the skull. The rf-coils were decoupled from each other, the transmitter coil actively, and the receiver coil passively.
Multislice apparent diffusion coefficient (ADC) maps were calculated from diffusion-weighted images (b-factors: 0, 750, 1500 seconds/mm2) acquired with a Stejskal-Tanner spin echo sequence (Stejskal and Tanner, 1965) (TE = 32 milliseconds, δ = 13.0 milliseconds, Δ = 15.1 milliseconds), resulting in an effective diffusion time of 10.8 milliseconds. Because the availability of high gradient power, the sequence was optimized for short TE, 32 milliseconds, therefore minimizing T2-dependent signal loss. The orientation of the diffusion-encoding gradient was always along the dorsal-ventral direction. Six coronal slices with a thickness of 1 mm and 0.75 mm interslice gap were recorded with a field of view of 40 mm. TR was 2325 milliseconds and matrix size 128 × 128. No averaging was done, resulting in an experimental time of 5 minutes for each multislice set.
Single-slice perfusion-weighted images through the center of the MCA territory were obtained with the arterial spin tagging technique (Detre et al., 1992). The perfusion-weighted imaging sequence consisted of two similar image acquisition intervals each of which comprised a magnetization preparation step of 3 seconds duration followed by snapshot fast low-angle shot imaging (TE = 3.5 milliseconds, TR = 7.4 milliseconds, field of view = 40 mm, slice thickness = 2 mm, scan time = 474 milliseconds for a 128 × 64 matrix). The images were recorded with centric phase encoding. During the first image acquisition (perfusion sensitive image), spins of blood flowing through the neck vessels were inverted adiabatically. During the second acquisition (control image), the magnetic field gradient was reversed so that inflowing spins were left undisturbed. Perfusion-weighted images were obtained by subtraction of both acquisitions (Kerskens et al., 1995). Eight subtraction images were averaged to improve the signal-to-noise ratio, resulting in a total scan time of 56 seconds for each perfusion-weighted image.
Three control ADC multislice sets and two perfusion-weighted images were obtained before embolization. One set of ADC maps and a perfusion-weighted image were acquired immediately after embolization, but before the infusion of rt-PA or saline. Eight further combinations of ADC and perfusion-weighted images were acquired during the following 3 hours.
Measurement of CBF
rCBF was measured 3 hours after MCA occlusion by quantitative autoradiography, using [14C]iodoantipyrine (35 μCi/mL; Amersham, Braunschweig, FRG) as the freely diffusible tracer (Sakurada et al., 1978). The tracer was infused intravenously over 60 seconds, and arterial blood samples were collected repeatedly on filter paper. At the end of tracer infusion, the rats were frozen in situ with liquid nitrogen.
Radioactivity of 20-μm thin cryostat sections was measured by quantitative autoradiography, using [14C] standards and a charge coupled device camera connected to an image processing system. [14C] radioactivity of blood was measured in a liquid scintillation counter (Wallace 1410, Pharmacia, Freiburg, FRG), and rCBF was calculated according to the algorithm of Sakurada et al.(1978).
Biochemical imaging
Cryostat brain sections adjacent to those used for autoradiography were collected for imaging of regional tissue pH and of the local distribution of ATP and glucose, using fluorescence and bioluminescence imaging techniques, respectively (Kogure and Furones Alonso, 1978; Csiba et al., 1983; Paschen et al., 1981). Images of tissue pH were calibrated with graded pH standards, and local metabolite content was quantified by correlating the optical density of bioluminescence images with metabolite values, measured in small tissue samples by conventional enzymatic techniques.
Image processing
ADC was calculated pixelwise by solving the monoexponential intravoxel incoherent motion model of Le Bihan (1991). For this purpose, the IDL (Interactive Data Language) software package (Creaso, Gilching, FRG) was used. Corrections were made for image-specific background noise and gradient crosstalk (Eis and Hoehn-Berlage, 1995). ADC maps and perfusion-weighted images were transferred to a Macintosh Power PC 7200/66 (Apple, Cupertino, CA). Image analysis was performed using the image processing software IMAGE (National Institutes of Health, Bethesda, MD).
For the evaluation of regional changes we defined the lesion core as the center of the MCA territory in which ATP was depleted and the borderzone as the peripheral parts of the lesion with tissue acidosis but preserved ATP (Kohno et al., 1995; Hoehn-Berlage et al., 1995). Therefore, signal intensities of perfusion-weighted images were measured in regions of interest placed in the lateral caudate-putamen (corresponding to areas with ATP-depletion: lesion center), the intermediate part of the cortex (corresponding to areas with tissue acidosis and preserved ATP: border zone), and in the frontoparietal cortex (nonischemic tissue). Signal intensities were expressed in percent of the mean control values.
Similarly, each ADC map was divided pixelwise by the average of three control ADC maps to allow evaluation of percent changes. The lesion area (defined as the area in which ADC decreased to less than 80% of control) was calculated for each time point using an intensity histogram algorithm. Additionally, the time course of ADC changes was determined in the same brain regions as chosen for the analysis of perfusion images.
The lesion areas visible in CBF, ATP, pH, and glucose images were calculated by thresholding with correction for tissue swelling (Swanson et al., 1990). Impaired ATP and glucose content was defined as the decrease to less than 30% of the mean value of the contralateral hemisphere. Tissue acidosis was defined as pH less than 6.3. Hemispheric lesion volumes were calculated according to Swanson et al. (1990) by multiplying the sum of the lesion areas of six slices by the interslice distance, and expressed as a percentage of the contralateral hemispheric volume.
Data are given as means ± SD. Statistical analysis between groups was performed by one-way analysis of variance, followed by Student's paired t-test; P < .05 was accepted as significant.
RESULTS
General physiologic variables
The physiologic variables before and after MCA occlusion are summarized in Table 1. Under control conditions, all general physiologic variables including body temperature were in the normal range. Rt-PA treatment resulted in a slight decrease in MABP (approximately 10% during the first 2 hours) but the other variables did not change. In particular, body temperature remained stable and did not differ between treated and untreated animals.
General physiologic variables
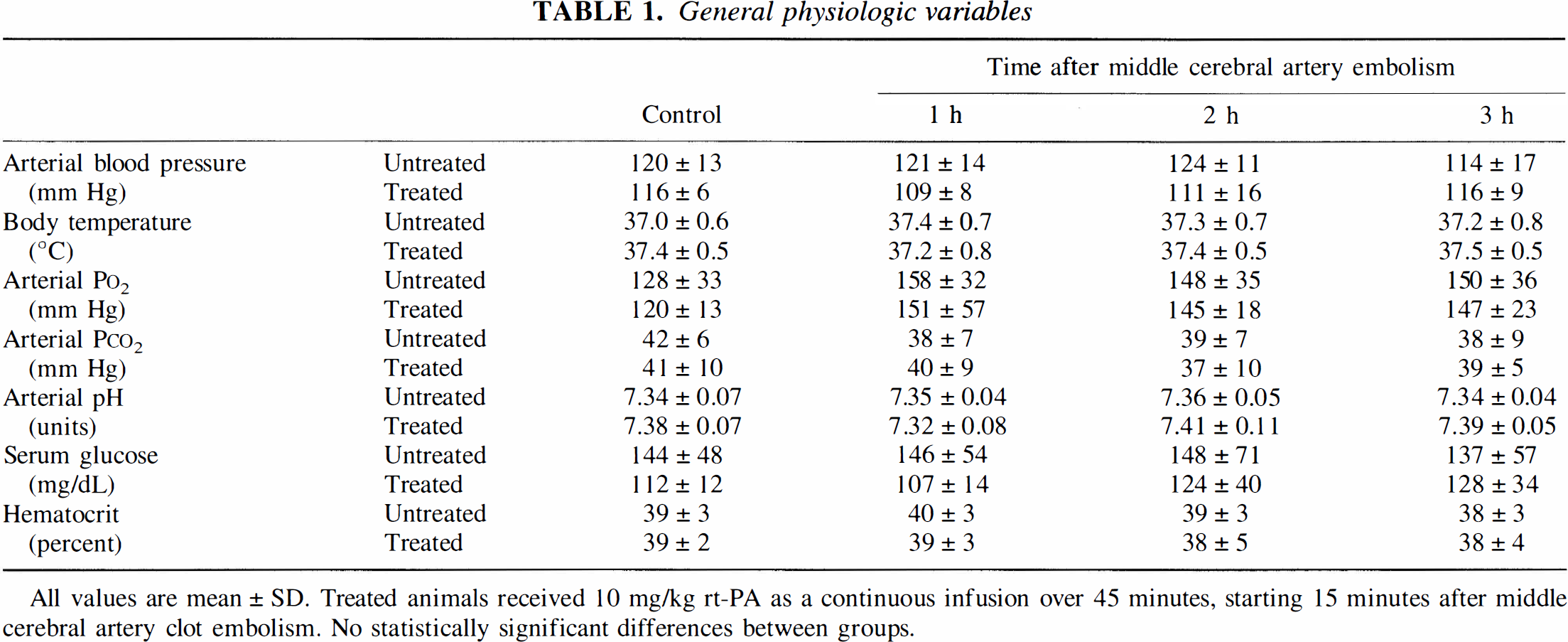
All values are mean ± SD. Treated animals received 10 mg/kg rt-PA as a continuous infusion over 45 minutes, starting 15 minutes after middle cerebral artery clot embolism. No statistically significant differences between groups.
Clot embolism without treatment
Perfusion imaging
Under control conditions, untreated animals (n = 8) did not exhibit interhemispheric asymmetries in the perfusion-weighted images. Immediately after injection of clots, a large area of reduced perfusion appeared in the right hemisphere and remained visible throughout the observation period of 3 hours (Fig. 1). Signal intensity decreased instantaneously to 34 ± 28% of control in the center of the MCA territory, and to 44 ± 28% of control in the border zone. These values remained nearly constant throughout the observation period. Signal intensity in a distant part of the fronto-parietal cortex was not affected by clot embolism (Fig. 2).
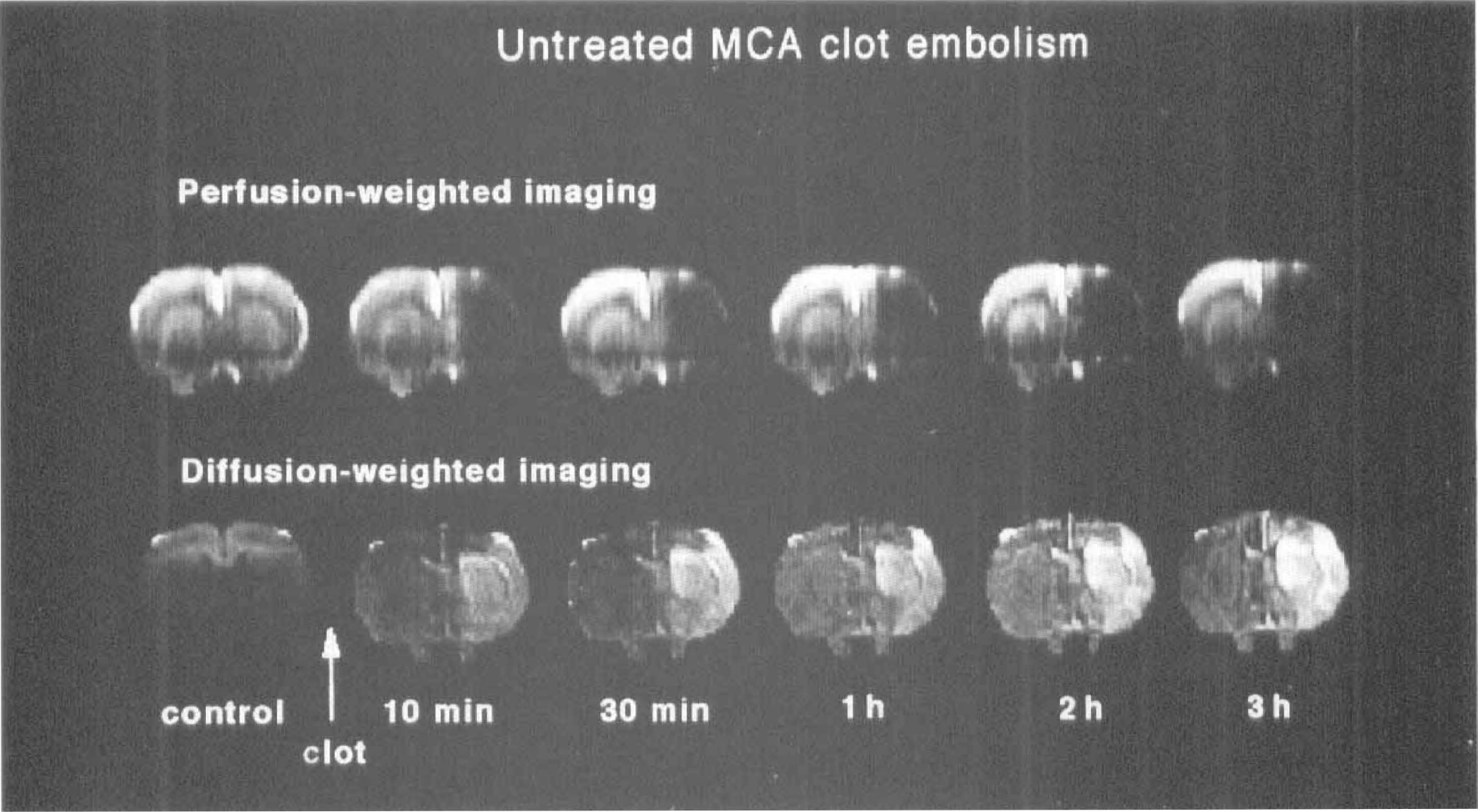
Perfusion- and diffusion-weighted images of a central brain slice of rat before and after clot embolism of the middle cerebral artery (MCA). After embolism the entire territory of the MCA is hypointense on perfusion images, indicating decreased regional perfusion. In diffusion-weighted images the same area shows gradually increasing signal intensity, indicating progressive tissue injury.
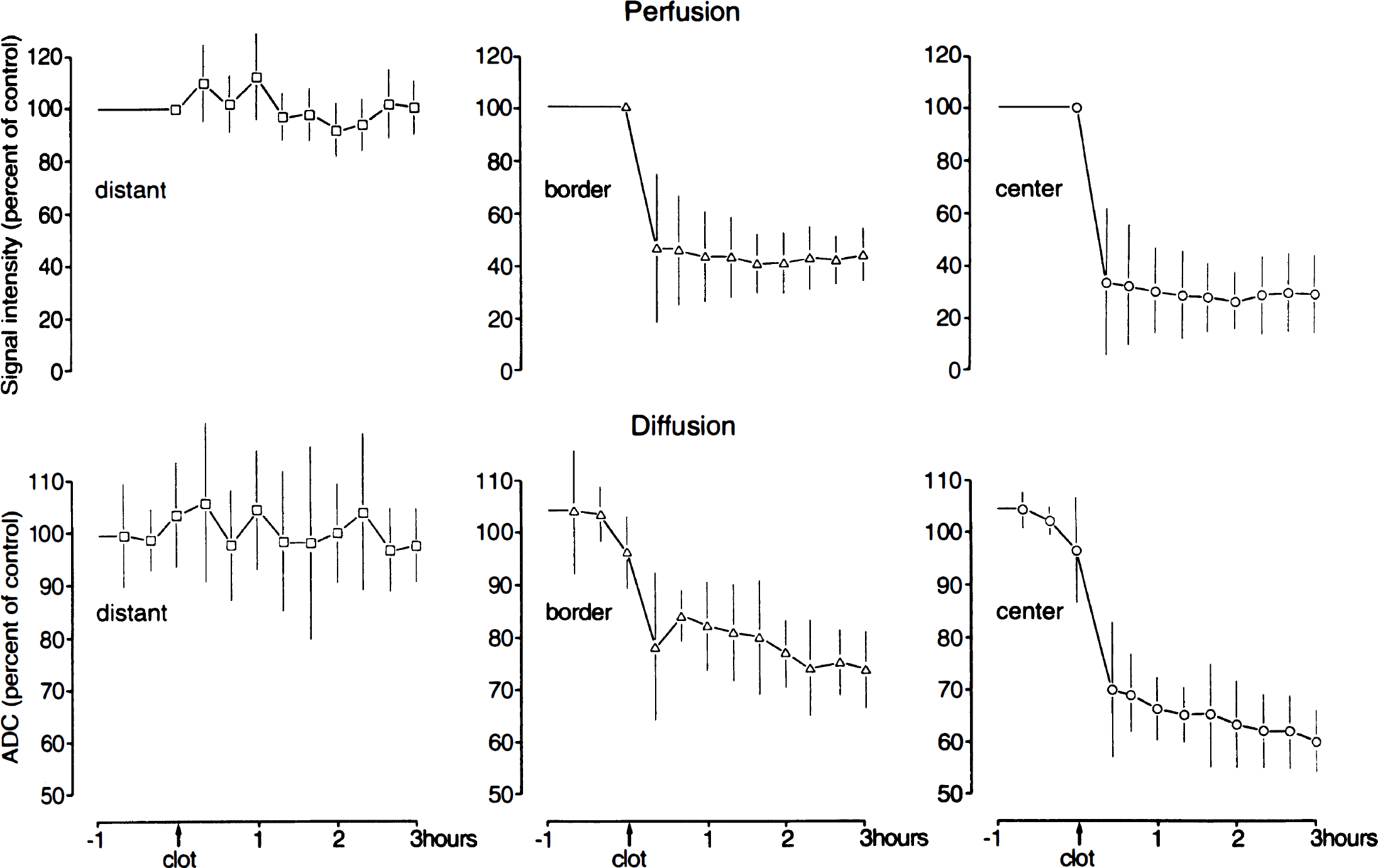
Mean regional changes of the signal intensity of perfusion-weighted images and of the apparent diffusion coefficient (ADC) in untreated animals (expressed as percent of control values ± SD, n = 8). Perfusion and ADC values sharply decrease in both the center and border zone of the middle cerebral artery territory immediately after embolism. Note progression of ADC—but not perfusion—changes over time.
ADC imaging
The ADC of water was determined in regions of interest in the parietal and lateral cortex and in the basal ganglia at the position of the caudate-putamen. The control ADC was 860 ± 12 × 10−6 mm2/s (parietal cortex) and 760 ± 14 × 10−6 mm2/s (basal ganglia), respectively. Immediately after embolism, ADC decreased in the center of the lesion to 70 ± 13% of control and decreased further to 60 ± 6% towards the end of the experiment (Fig. 2). In the border zone, ADC decreased initially to 78 ± 14% and it further decreased to 74 ± 7% of control. ADC did not change in distant parts of the hemisphere.
Ten minutes after injection of clots, the ADC lesion volume amounted to 43 ± 22% of ipsilateral hemisphere. This volume gradually increased over time to 61 ± 20% after 3 hours of embolization. The steady increase visible in Figure 3 suggests that the ADC lesion had not yet reached its final volume at the end of the observation period of 3 hours.
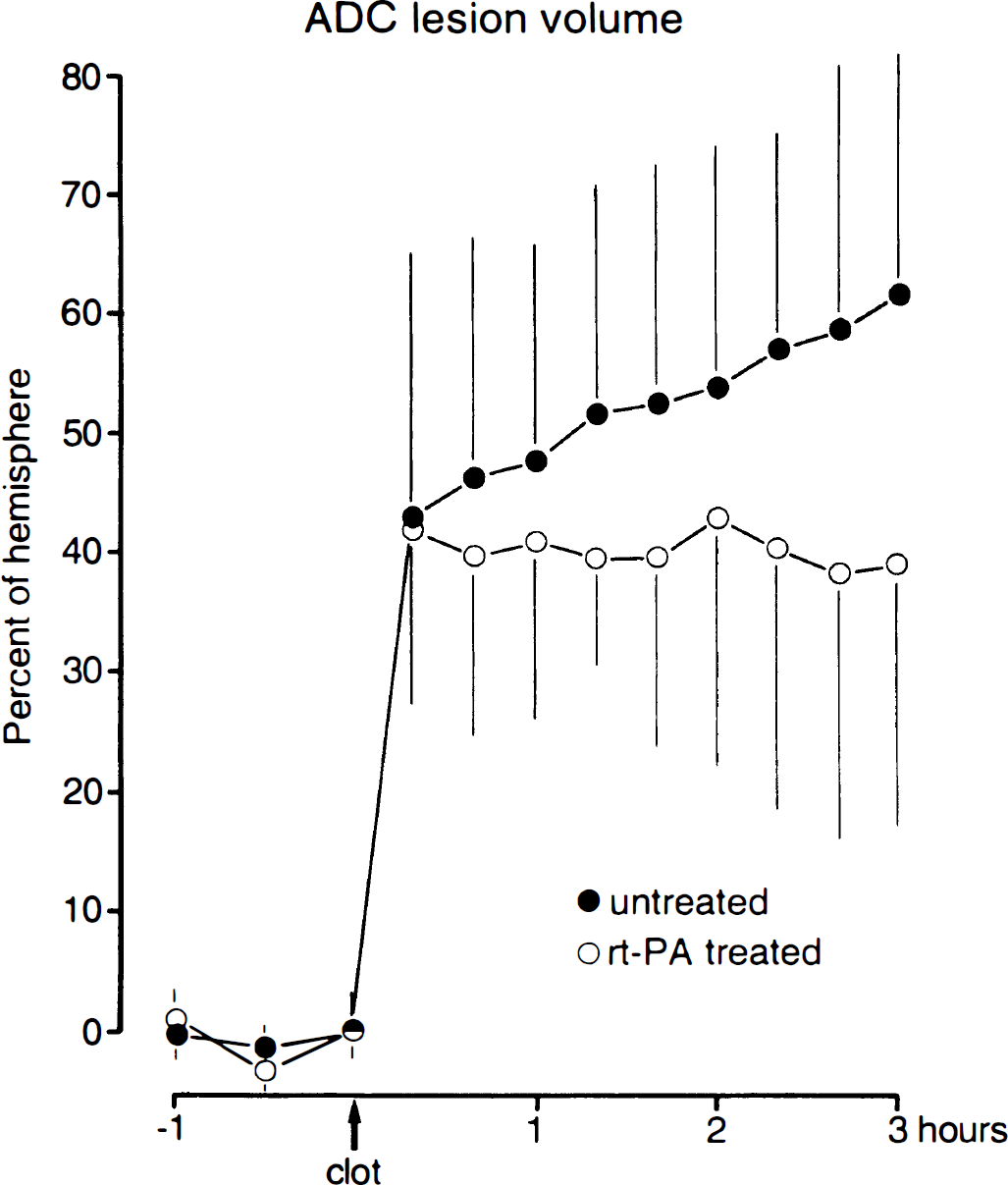
Changes of apparent diffusion coefficient lesion volume after clot embolism with and without thrombolytic therapy (means ± SD). Both groups show similar lesion volumes before onset of therapy. In untreated animals, there is further growth of the lesion volume during the following three hours. In recombinant tissue plasminogen activator treated animals, the lesion volume decreases slightly. The end points of both groups are significantly different (n = 8, P < .05).
Autoradiographic CBF measurements
rCBF(n = 5) was measured at 3 hours after clot embolism. Flow values gradually decreased from the border zone to the center of the ischemic territory (Fig. 4). rCBF was less than 50 mL/100 g/min in a volume corresponding to 66 ± 34% of the ipsilateral hemisphere, and less than 20 mL/100 g/min in 49 ± 30% of the hemisphere (Fig. 5).
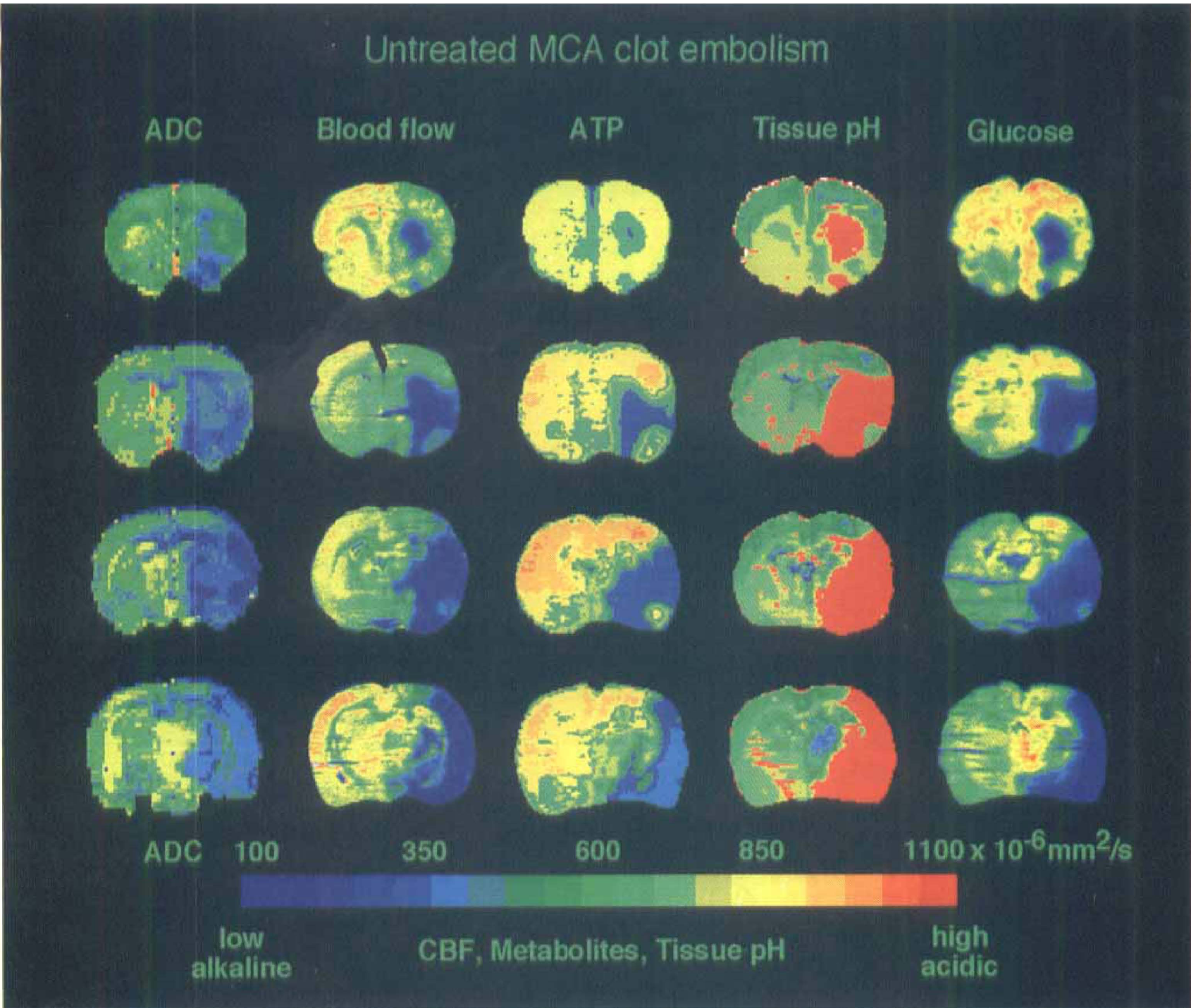
Comparison of apparent diffusion coefficient (ADC) maps of untreated rat with images of blood flow. ATP, tissue pH, and glucose content in corresponding cryostat sections of the same brain at 3 hours after middle cerebral artery (MCA) clot embolism. Presented are coronal slices (from top to bottom) at +1, 75 mm, 0 mm, −1, 75 mm, and −3, 5 mm, as related to the bregma. The region of ATP depletion is restricted to the core of the ischemic territory. The areas of reduced CBF, ADC decrease, and tissue acidosis cover almost the entire MCA territory, indicating the presence of a large penumbra.
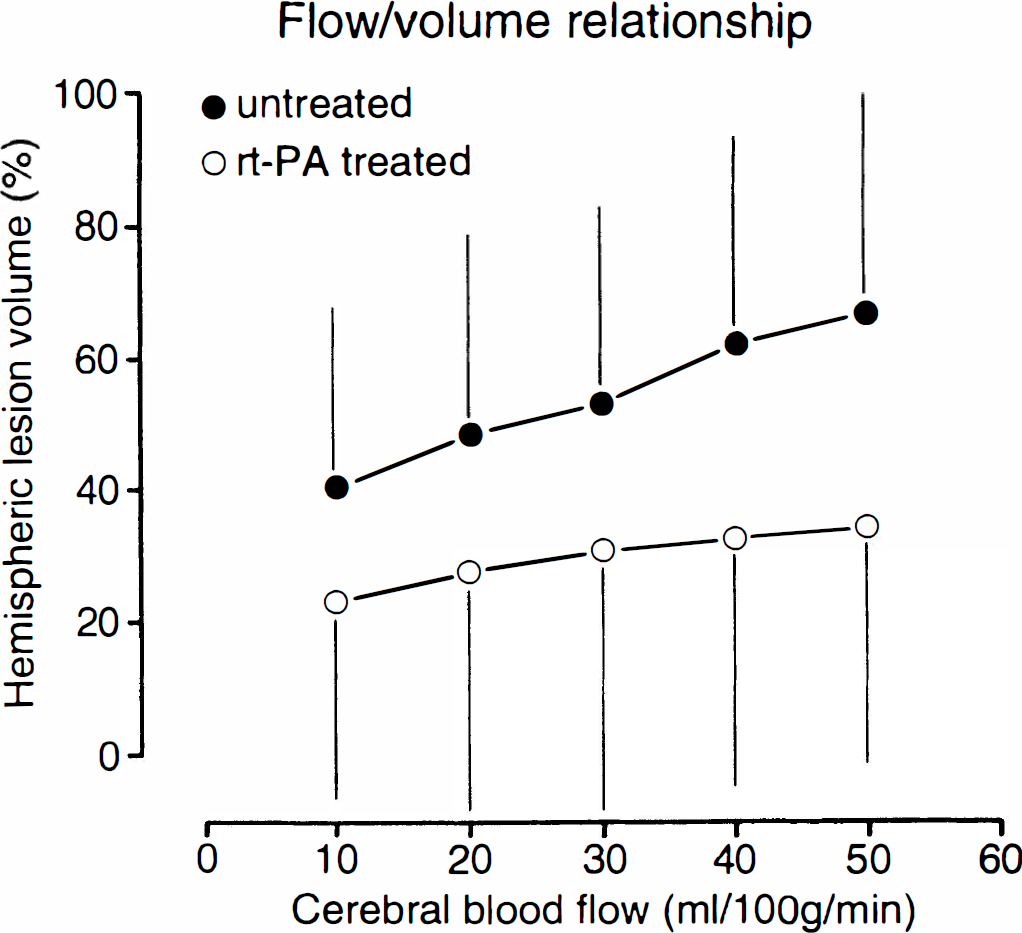
Volumes of reduced blood flow in untreated and recombinant tissue plasminogen activator (rt-PA) treated rats. The relationship between CBF and the corresponding hemispheric lesion volume were measured by quantitative autoradiography at 3 hours after clot embolism. Early thrombolysis with rt-PA reduces—but does not reverse—flow impairment. Values are means ± SD.
Biochemical imaging
Visual inspection of ATP, glucose, and pH images obtained at six coronal levels corresponding to the planes of ADC maps revealed consistent depletion of ATP and glucose as well as severe tissue acidosis in the center of the MCA territory (Fig. 4). Quantitative analysis (n = 7) of the hemispheric lesion volumes showed tissue acidosis in 56 ± 39% of ipsilateral hemisphere which was markedly larger than the lesion volumes of ATP, 46 ± 29%, or glucose depletion, 47 ± 30% (Fig. 6).
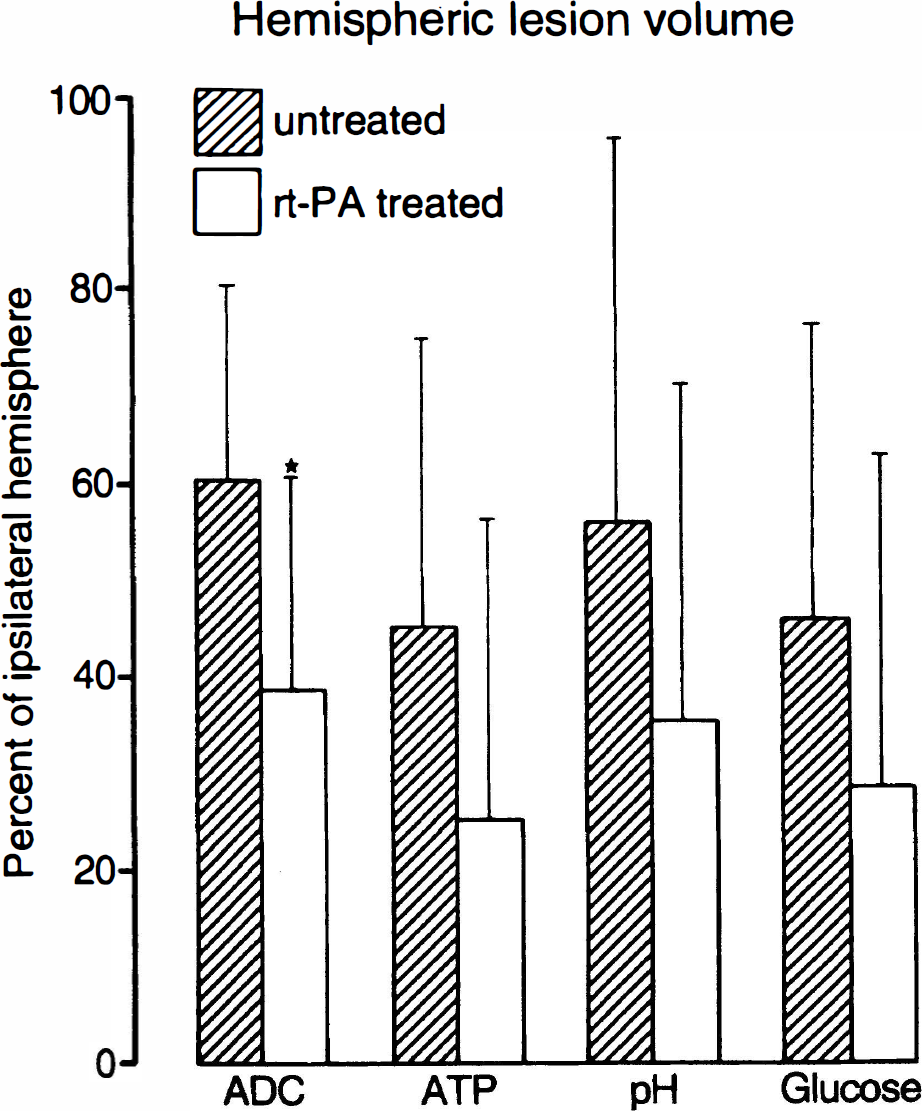
Comparison of hemispheric lesion volumes (means ± SD) defined by ADC decrease, ATP depletion, tissue acidosis and glucose depletion 3 hours after middle cerebral artery clot embolism in untreated (n = 7) and treated animals (n = 4). Thrombolysis reduces lesion volume defined by all four parameters but because of the limited number of animals, only the difference of apparent diffusion coefficient lesion volumes reaches statistical significance (P < .05).
Rt-PA treatment of clot embolism
Perfusion imaging
Intracarotid infusion of rt-PA started 15 minutes after injection of emboli and continued for 45 minutes. The first perfusion image, obtained right after embolism but before beginning of rt-PA infusion, confirmed successful clot embolization in all animals of this group. Rt-PA treatment resulted in improvement of the signal intensity in perfusion images (Fig. 7). In three of these animals, heterogeneous areas of hypoperfusion and hyperperfusion were transiently observed (Fig. 8).
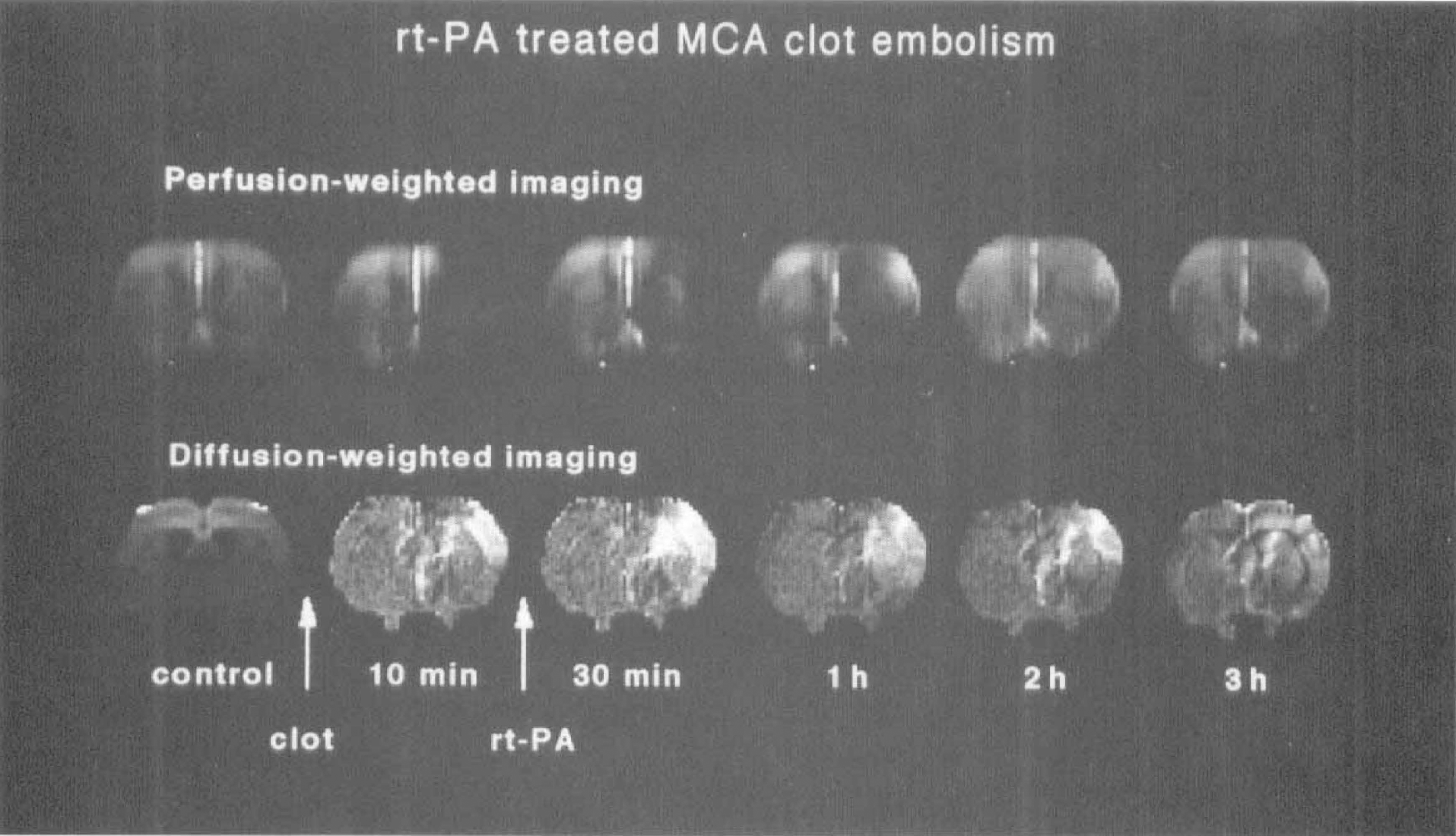
Perfusion- and diffusion-weighted images of a central brain slice of rat before and after clot embolism of the middle cerebral artery (MCA). Treatment with recombinant tissue plasminogen activator was started 15 minutes after embolism and continued for 45 minutes. Immediately after embolism, perfusion and diffusion images show ischemic alterations in the MCA territory. After thrombolysis both perfusion- and diffusion-weighted images show partial reversal of changes.
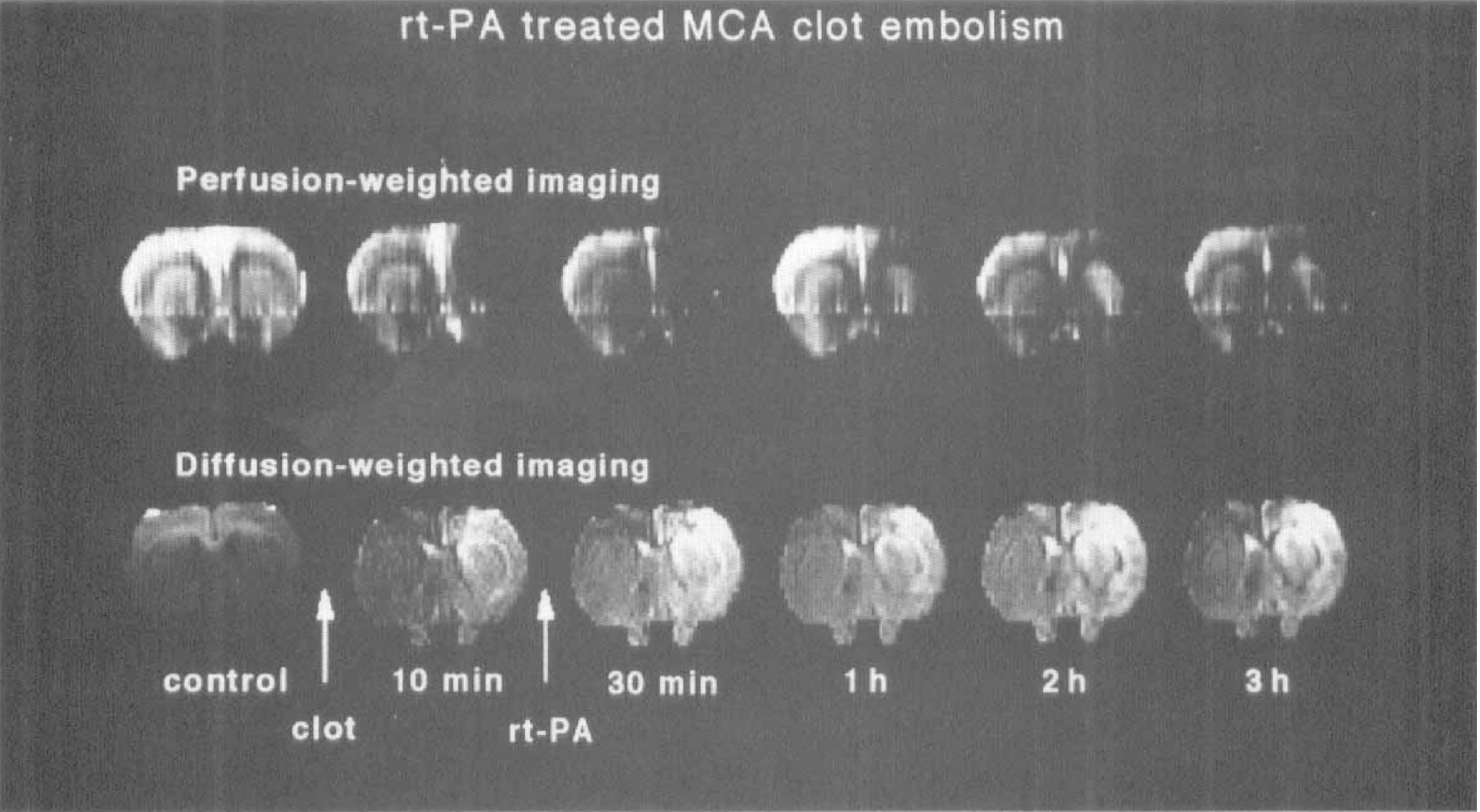
Example of a recombinant tissue plasminogen activator—treated animal with marked hyperperfusion in the basal ganglia—but not in cerebral cortex—at 1 to 2 hours after the onset of treatment. Diffusion-weighted images do not show recovery.
In the center of the MCA territory, the mean signal intensity of perfusion images decreased immediately after embolism to 30 ± 18% of control, which is not significantly different from untreated animals (Fig. 9). After the beginning of rt-PA treatment, signal intensity recovered to 52 ± 20% but afterwards it decreased again to reach a final value of 31 ± 12% of control. In the border zone, perfusion signal initially decreased to 33 ± 16% of control but then it steadily recovered to a final value of 70 ± 31% after 3 hours. The slope of the changes in perfusion suggests that recovery in the border zone had not reached its final value at the end of the 3 hours observation period (Fig. 9), indicating that even in the peripheral parts of the ischemic territory, restoration of blood flow is not promptly resumed after rt-PA treatment.
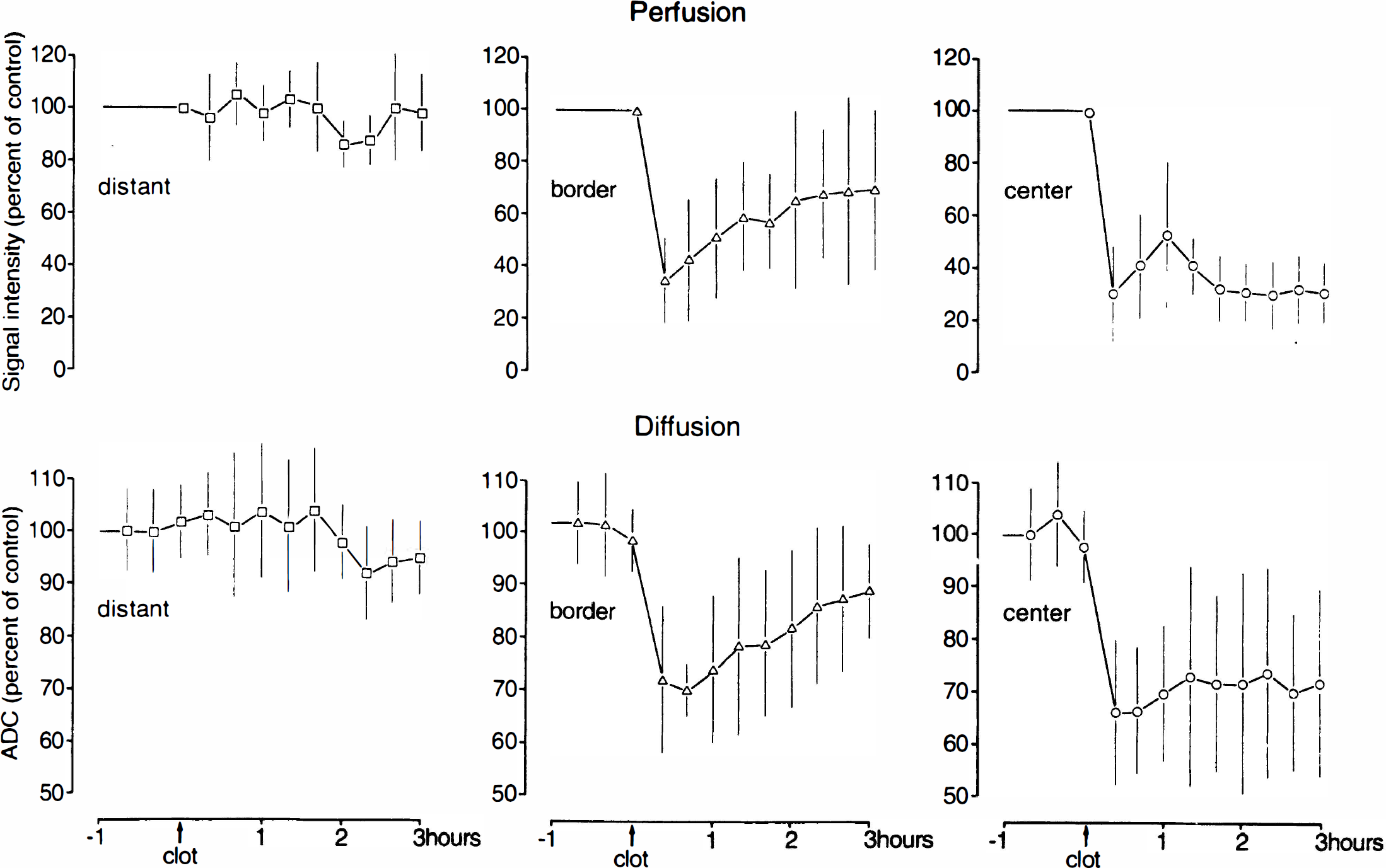
Mean regional changes of the signal intensity of perfusion-weighted images and of the apparent diffusion coefficient (ADC) in recombinant tissue plasminogen activator—treated animals (expressed as percent of control values ± SD, n = 8). Perfusion and ADC values decrease sharply in both the center and border zone of the middle cerebral artery territory immediately after embolism. In the center of the lesion perfusion begins to recoverduring treatment but then suffers a secondary decrease. In the borderzone, both perfusion and ADC steadily improve.
ADC imaging
Before embolism, the ADC was 883 ± 19 × 10−6 mm2/s in the parietal cortex and 784 ± 15 × 10−6 mm2/s in the basal ganglia. Immediately after embolism, the ADC decreased in the center to 66 ± 14% and in the border zone to 72 ± 14% of control. The ADC values before treatment were similar to the values in the untreated group. After thrombolysis, the ADC stabilized in the center at a final value of 72 ± 18% of control (versus 60 ± 6% in the untreated group). The border zone showed a marked improvement with a final value of 89 ± 9% of control (versus 74 ± 7% in the untreated group) (Fig. 9). No ADC change was observed in distant parts of the brain.
Immediately after embolism the ADC lesion volume of treated animals was very similar to the untreated group (42 ± 15% versus 43 ± 22% of ipsilateral hemisphere). However, in contrast to the untreated animals, this volume slightly decreased within 3 hours of rt-PA treatment to 39 ± 22% (Fig. 3). Because ADC lesion volume continued to grow in the untreated group, the difference between the two groups corresponded to a volume reduction by 36% (P = .03). One animal showed almost complete recovery, demonstrating that ischemic ADC changes are, in principle, fully reversible after successful thrombolysis (Fig. 10).
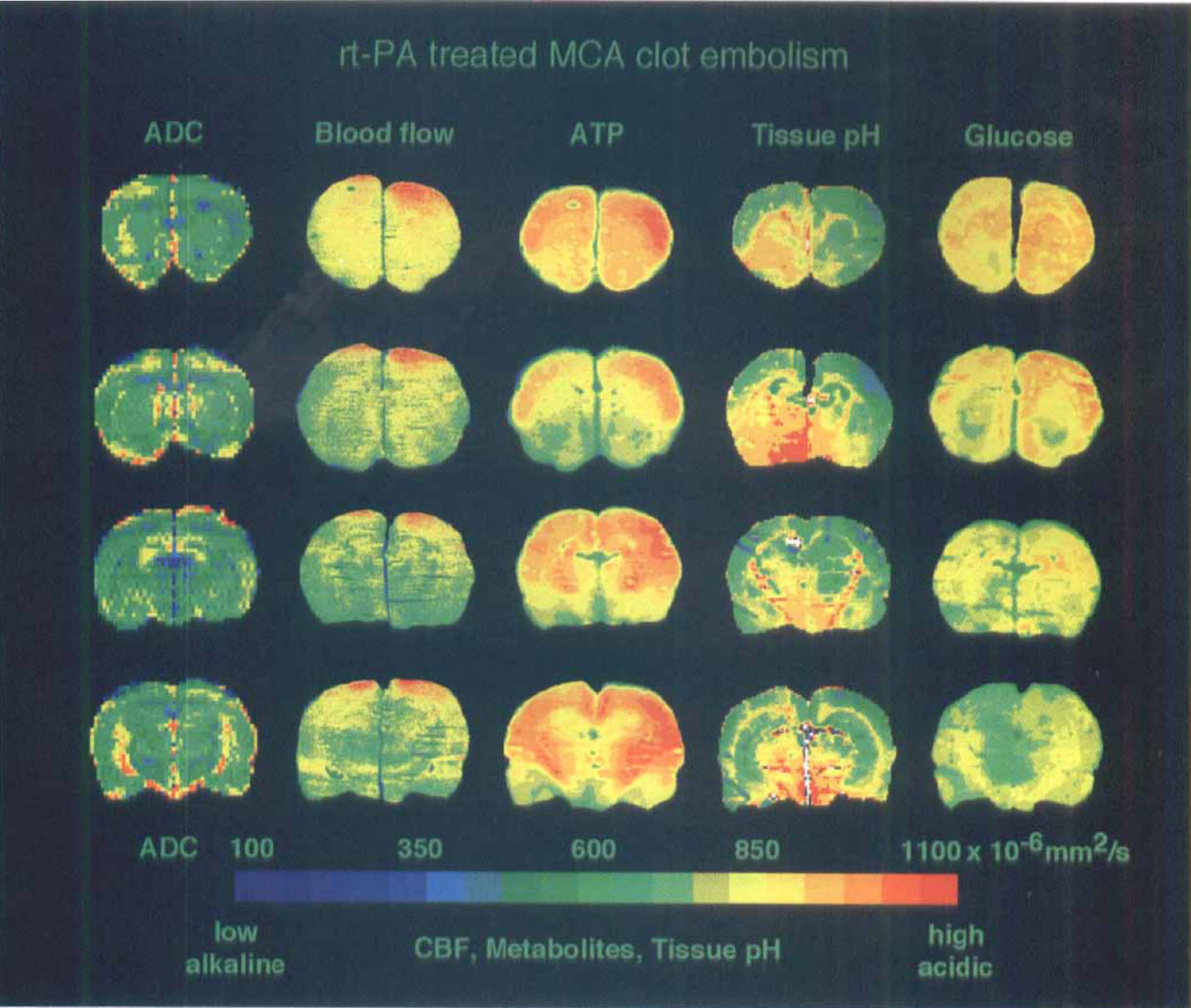
Comparison of apparent diffusion coefficient maps of recombinant tissue plasminogen activator—treated rat with images of blood flow, ATP, tissue pH, and glucose content in corresponding cryostat sections of the same brain at 3 hours after middle cerebral artery clot embolism. Note absence of changes, indicating full recovery after thrombolysis.
Autoradiographic CBF measurements
At 3 hours after clot embolism, the volumes of reduced blood flow, calculated for stepwise decreasing flow thresholds, were markedly smaller in rt-PA—treated animals (Fig. 5). The hemispheric volume with flow values less than 50 mL/100 mg/min was 33 ± 36% of the ipsilateral hemisphere as compared to 66 ± 34% (P = .17) in untreated animals, and the volume with flow values less than 20 mL/100 mg/min was 27 ± 37% as compared to 49 ± 30% (P = .31) in untreated animals. Thus, thrombolysis showed a trend towards reduction, but did not revert the flow impairment after clot embolism.
Biochemical imaging
Rt-PA treatment of clot embolism resulted in a reduction of lesion volume for both ATP and glucose depletion as well as for tissue acidosis (Fig. 6). For technical reasons, the full biochemical imaging protocol, i.e., evaluation of all six coronal sections required for calculation of hemispheric lesion volumes, could only be completed in four animals of this group. In one of these, biochemical changes were virtually absent (Fig. 10). In the other three rats, circumscribed lesions persisted in agreement with the deficits on perfusion and ADC images, but the size of these lesions was smaller than in untreated animals. The mean hemispheric lesion volumes were 25 ± 31% for ATP depletion, 29 ± 34% for glucose depletion, and 36 ± 34% for acidosis (Fig. 6). These values corresponded to a lesion reduction by 46% (P = .29), 38% (P = .38) and 36% (P = .38), respectively, compared to the untreated animals and, therefore, suggest reduction in lesion size for these parameters.
DISCUSSION
The results of this investigation showed that rt-PA treatment quickly improves blood flow, followed by a continuous increase over 40 minutes throughout the MCA territory. However, thereafter we found a secondary decrease of perfusion in the center of the lesion, whereas perfusion in the border zone further improved. As a result the volumes of both the ischemic core and the penumbra were reduced by about 40%, but ischemic injury could not be fully prevented. The comparison between CBF measurements (Fig. 5) and biochemical parameters (Fig. 6) suggests that the incomplete reversal of the lesion core, defined as area with ATP-depletion, was due to restricted reperfusion. Moreover, abnormalities in the periphery of the ischemic lesion, depicted as pH and ADC changes, partly persisted during the observation time, despite adequate reperfusion. These alterations are the more relevant as the changes in pH and ADC may have been attenuated by the developing brain edema. In fact, vasogenic edema causes alkalization of brain tissue and an increase in ADC, as previously documented in peritumoral brain edema (Hossmann et al., 1992; Els et al., 1995) or in the surrounding of a photothrombotic lesion (Lee et al., 1996).
The observation of restricted reperfusion in the core at the embolized vascular territory raises the question whether treatment failed to produce thrombolysis. Because of the fact that brains were frozen in situ, the cerebral vessels could not be inspected at the end of the experiment. However, the magnetic resonance-detectable improvement of blood perfusion throughout the MCA territory during the first 40 minutes after rt-PA infusion provides strong evidence that recanalization took place. In a previous study (Busch et al., 1997b) in which the present model of thromboembolic stroke and thrombolytic reperfusion was validated, we inspected the basal brain vessels under the operating microscope at 3 hours after embolism. Local, intra-arterial rt-PA treatment resulted in recanalization of the proximal part of the MCA in all cases. We could not exclude that some of the injected clot material was not dissolved by rt-PA. However, our finding of an initial reperfusion is supported by many studies, showing that systemically applied fibrinolytic agents are able to dissolve obstruction of large cerebral arteries (Phillips et al., 1988, Theron et al., 1989).
However, the possibility of irreversible primary injury also must be considered as an explanation for the incomplete recovery. Studies in global ischemia have shown that a 15-minute period of circulatory arrest does not cause brain infarction (Fischer et al., 1995). Reversible focal cerebral ischemia induced by intraluminal suture occlusion of the MCA was tolerated for as long as 60 minutes without necrotic brain lesions (Garcia, 1995; Kaplan et al., 1991). More recently, it was reported that delayed apoptotic cell death was observed in some neurons after MCA occlusion of as short as 10 to 20 minutes, but this was not accompanied by necrotic infarction (Chopp and Li, 1996; Li et al., 1995). In our study, rt-PA was given by infusion from 15 to 60 minutes after embolization. Recanalization of the MCA probably occurred during this time because the half-life of rt-PA is only several minutes (Verstraete et al., 1985). The initial improvement visible in the perfusion-weighted images is also in agreement with early recanalization. It is, therefore, unlikely that at the time of recanalization brain tissue was already irreversibly injured.
Mechanisms of ischemic injury
Recanalization of the proximal part of the MCA does not mean that clot material is removed from the arterial tree. It is likely that the clots, before complete dissolution, will be fragmented by thrombolysis and flushed downstream, resulting in two possible complications. Clot fragments could lodge in small arteries of the peripheral vascular bed with poor collateral flow, where they cannot be easily reached by rt-PA and may remain undissolved for a time long enough to cause tissue necrosis. Even smaller thrombotic particles could be trapped in the capillary bed, and such microemboli are known to produce breakdown of the blood brain barrier (Schuier et al., 1978). In fact, a previous study of blood brain barrier permeability in the present model of clot embolization revealed earlier and more pronounced barrier disturbances than after clip or suture occlusion (Busch et al., 1997a).
Furthermore, recanalization by thrombolysis is a slow process as compared to the sudden onset of reperfusion after removal of a surgical clip or an intraluminal suture. The induction of low pressure reperfusion during thrombolysis, in combination with downstream displacement of clot material, may, therefore, result in incomplete recirculation and ischemic cell swelling that could lead to microvascular compression and further impairment of blood flow.
A more speculative explanation for restricted reperfusion after thrombolysis is a reactive change of the coagulation system. It is known from studies of coronary thrombolysis (for review see Munkvad, 1993), that rt-PA induces paradoxical generation of systemic thrombin activity, probably due to the removal of fibrin deposits from the vascular intima leading to (reactive) activation of the coagulation system (Munkvad et al., 1990). Rt-PA also induces a prolonged depression of factor XII—dependent fibrinolytic activity that persists for at least 8 weeks (Munkvad et al., 1990). On the other hand, t-PA is expressed in brain capillaries suggesting that vascular endothelium plays a role in the regulation of the local hemostatic activity (Kittaka et al., 1996). Therefore, the procoagulant condition (Zlokovic et al., 1995; Catto and Grant, 1995) induced by cerebral ischemia could be aggravated by this effect. Because rt-PA has a half-life of only several minutes (Verstraete et al., 1985), these alterations could become important already during the first hours of treatment. Therefore, it is conceivable that rt-PA interferes with the endogenous hemostatic system of the capillary endothelium and contributes to impaired microcirculation.
In the rat, the 10-fold human dose of rt-PA must be given to achieve a comparable thrombolytic activity as in patients (Korninger and Collen, 1981; Karges et al., 1994). At present it is unknown whether this dose might create additional effects because t-PA is a catabolic enzyme. Future work should clarify whether there are direct and dose-dependent effects of rt-PA on the brain beyond its thrombolytic potential.
In the periphery of the ischemic lesion, acidosis and alterations of the ADC partly persisted despite improved blood flow (Figs. 5 and 6). Reversal of ischemic injury on reperfusion takes some time and the most likely explanation is that these alterations have not yet recovered during the observation period of 3 hours. However, it cannot be excluded that persisting acidosis and the associated alterations of ADC are indicators of the lasting disturbance of mitochondrial function which is compensated by increased anaerobic glycolysis. This could be due to peroxidative damage occurring during reperfusion (Chan et al., 1995). Reperfusion injury may, therefore, add to the hemodynamic disturbances responsible for the incomplete recovery observed in the present study.
Comparison with other studies on reperfusion
In contrast to thrombolysis of clot embolism, transient surgical occlusion of a mature brain artery does not produce reperfusion deficits unless duration of vascular occlusion is exceedingly long. After 30 minutes of MCA clip occlusion, rCBF recovered to about 50% of control after 15 minutes and returned to normal after 2 hours of reperfusion (Takagi et al., 1995). Another study observed a heterogeneous pattern of relative hyperperfusion and hypoperfusion after 1 hour of reversible ischemia induced by an intravascular thread occlusion (Dirnagl et al., 1989), but secondary flow impairments were not reported.
Flow measurements performed after thrombolytic reopening led to more divergent results. Papadopoulos et al. (1987) reported a rapid recovery of CBF, measured with the hydrogen clearance method, when treatment was started 2 hours after clot embolization. However, a recent study by Gross et al. (1995) showed only a marginal recovery of CBF in rabbits when treatment started 3 hours or later after embolism. Another study reported rCBF measurements by single-photon emission computed tomography after thrombolysis in five clinical cases of stroke (Herderschee et al., 1991). With the exception of one case, single-photon emission computed tomography measurements remained abnormal with further deterioration in two patients. CBF data acquired with positron-emission tomography on two patients after early rt-PA treatment showed only slight improvement of blood flow during the first hours but complete restoration after 1 day (Grond et al., 1996).
These measurements of CBF after thrombolysis could explain, similar to our investigation, the report of other studies that successful thrombolytic reopening of large cerebral arteries does not necessarily improve outcome. Phillips et al. (1989) investigated clot lysis by repeated angiography in a rabbit embolic stroke model where a single clot was placed into the intracranial part of the internal carotid artery using Seldinger catheterization. Treatment with an rt-PA analogue after 1 hour resulted in reversal of the angiographically visible occlusion but not in a reduction of infarct volume. Lee et al. (1989) showed reopening of the large arteries by digital subtraction angiography. Although magnetic resonance spectroscopy detected slight improvement of 31p changes, 2,3,5-triphenyl tetrazolium chloride staining did not show a difference in infarct size. The question as to whether thrombolytic reopening of large arteries correlates with improvement in outcome is also being discussed for organs other than the brain. In major trials on the thrombolytic therapy of myocardial infarction, different therapeutical strategies resulted in varying arterial patency rates but these differences were not reflected by changes in mortality (Stringer, 1994).
In conclusion, recovery of ischemic brain injury 3 hours after clot embolism and rt-PA treatment in the rat is incomplete even when thrombolysis is started as early as 15 minutes after vascular occlusion. We observed restricted reperfusion in the lesion core which may be partly explained by downstream displacement of clot material and microembolism of the vascular periphery. In the border zone we found incomplete tissue recovery despite sufficient reperfusion, which might be due to effects previously associated with reperfusion injury. Further understanding of the pathophysiologic mechanisms during and early after thrombolysis may have an impact on treatment strategies in stroke patients.
Footnotes
Abbreviations used
Acknowledgments
The authors thank Mrs. Ulla Uhlenkueken for help with MR data acquisition and analysis, and Mrs. Christiane Stratmann for processing the brains for bioluminescence imaging. The authors also thank Mrs. Doris Schewetzky and Mrs. Margret Hahmann for their secretarial assistance, Mrs. Ilka Muehlhoever for the illustrations. Mr. Bertram Huth is also gratefully acknowledged.
