Abstract
Hibernation in mammals is associated with a regulated depression of global cellular functions accompanied by reductions of cerebral blood flow that would render the brain profoundly ischemic under normal conditions. Homeostatic control is preserved, however, and brain damage does not occur. We investigated the possibility that hibernation not only confers tolerance to profound hypothermia, but also to hypoxia and aglycemia independent of temperature. Hippocampal slices from ground squirrels Citellus tridecemlineatus in both the active and hibernating states and from rats were subjected to in vitro hypoxia and aglycemia at incubation temperatures of 36°C, 20°C, and 7°C and evaluated histologically. A binary bioassay was used to determine the duration of hypoxia/aglycemia tolerated in each group. At all temperatures, slices from hibernating animals were most tolerant compared with both active squirrels and rats. Slices from active ground squirrels were more tolerant than rat at 20°C and 7°C but not at 36°C indicating a species-specific difference that becomes manifest at lower temperatures. These results indicate that hibernation is associated not only with tolerance to profound hypothermia but also to deprivation of oxygen and glucose. Because tolerance was already demonstrable at the shortest duration of hibernation studied, rapid therapeutic induction of a similar state may be possible. Therefore, identification of the regulatory mechanisms underlying this tolerance may lead to novel neuroprotective strategies.
Hibernation in mammals is accompanied by reductions in cardiac output and cerebral blood flow that would render the brain profoundly ischemic under conditions of normal metabolic demand (Frerichs et al., 1994). Nevertheless, homeostatic control is preserved and brain damage does not ensue. Therefore, ischemic stress appears not to occur and an equilibrium between substrate availability and metabolic demand is maintained, despite a massive reduction in body temperature that is lethal in most species if induced artificially, including hibernators (Musacchia, 1984). This alone could be considered an indicator of ischemia resistance. It could be argued, however, that hibernation merely reflects a natural adaptation to cold with the implication that hibernating animals may be as vulnerable to CNS insults as their non-hibernating counterparts. To test the hypothesis that hibernation represents a state of neuroprotection, one would have to artificially impose an additional CNS insult. Resistance among hibernators to the artificial imposition of CNS insults, such as brain ischemia, in comparison with non-hibernating controls has not been demonstrated. It is difficult to make this comparison in vivo because control animals would have to be cooled to the same temperature as hibernators and, as indicated above, such artificially induced hypothermia is harmful. To circumvent this problem, we have used an in vitro model of ischemia in which hippocampal slices from hibernating and active ground squirrels and rats were exposed to hypoxia/aglycemia for varying periods. Demonstration of tolerance to an artificially imposed stress such as ischemia would lend considerable support to the idea that useful neuroprotective strategies could be derived from hibernation.
MATERIALS AND METHODS
Animals and surgical procedures
Experiments were performed in 18 male adult Sprague-Dawley rats (average weight, 364 ± 86 g) and 38 adult 13-lined ground squirrels Citellus tridecemlineatus (average weight, 198 ± 26 g) of either sex. Ground squirrels were captured in autumn by a United States Department of Agriculture–licensed trapper (TLS Research, Bartlett, IL). The experimental protocols were approved by the National Institute of Neurological Disorders and Stroke Animal Care and Use Committee.
Ground squirrels were anesthetized with ketamine (75 mg/kg, intraperitoneally) and xylazine (10.6 mg/kg, intraperitoneally) for implantation of telemetry devices as described previously (Frerichs et al., 1994) to allow continuous remote monitoring of the ECG and body temperature. Animals were allowed to recover for 48 hours before transfer into the hibernaculum which is kept in constant darkness at 5°C to simulate the natural environment conducive of hibernation. During the winter season, animals typically enter hibernation bouts within 2 to 3 days after being placed in this environment. Bout length increases toward the end of the natural hibernation season and ranges from hours to weeks.
Hippocampal slice preparation and in vitro hypoxia/aglycemia
Transverse hippocampal slices were prepared as previously described (Teyler, 1980). Rats and active ground squirrels were anesthetized with ketamine (squirrels 75 mg/kg, rats 100 mg/kg, intraperitoneally) before decapitation. Ketamine improves the physiologic and morphologic condition of the slice during preparation; blockade of N-methyl-D-aspartate (NMDA) receptors, however, is only short-lived (Taylor and Weber, 1993). Hibernating animals were gently lifted out of their cages and rapidly decapitated without prior intraperitoneal injection of ketamine to avoid arousal. Brains were rapidly removed and immersed in chilled (5°C) artificial CSF (aCSF) containing (mmol/L) NaCl 125, KCl 3, CaCl2 1.5, MgSO4 1.3, KH2PO4 1.4, NaHCO3 26, and glucose 5. Some investigators increase aCSF glucose to 10 mmol/L. Such high levels, however, may complicate studies of in vitro neuronal injury (Schurr et al., 1984) whereas 5 mmol/L of aCSF glucose adequately maintains physiologic slice responses (Alger et al., 1984) and approximates the normal physiologic level. The pH was adjusted to 7.4 after equilibration with 95% O2 and 5% CO2 before use. The osmolality of the incubation medium was 295 mOsm/kg and remained stable throughout the experiment (Wescor, Logan, UT). After allowing the brain to equilibrate in chilled aCSF for 30 seconds, the left hippocampus was quickly removed and sliced on the prechilled cutting stage of a tissue chopper (Stoelting, Wood Dale, IL) into 400-μm thick transverse sections. The entire cut hippocampus was then transferred into chilled aCSF for gentle separation of the individual slices under the microscope. A plastic pipette was used to transfer individual slices into an immersion-style brain slice chamber (two slices per chamber, six slices per hippocampus/animal) placed in a waterbath (Fig. 1). The slice chamber was designed to allow biochemical and morphologic studies over a wide range of temperatures (7°C, 20°C, and 36°C) and rapid exchange of the incubation medium (Fig. 1E). The medium was bubbled with the desired gas mixture at two locations without agitating the slices to ensure good mixing with minimal trauma to the slices by fluid waves. The medium was continuously exchanged by adding fresh medium (66 μL/min, Fig. 1B). Chamber volume was kept constant at 4 mL aCSF by controlled vacuum aspiration (Fig. 1C). The slices (two per chamber) rested on a glass filter mesh (pore size, 200 μm, Electron Microscopy Sciences, Ft. Washington, PA). Floating during medium exchange was prevented by a nylon mesh above the slices. Slices were kept at 20°C until 30 minutes after decapitation to optimize slice survival (Newman et al., 1992). Temperature was then gradually increased to 36°C over 30 minutes and maintained at 36°C for an additional 30 minutes. The temperature was then decreased to either 20°C or 7°C over 30 minutes or maintained at 36°C. At 2 hours after decapitation, the medium was rapidly exchanged with aCSF equilibrated with 95% N2/5% CO2 without glucose and the gas mixture was switched to 95% N2/5% CO2. This reduced the pO2 of the incubation medium from 464 ± 30 to 30 ± 9 mm Hg while the pCO2 remained unchanged at 35 ± 8 mm Hg (Corning 288, Corning, NY). In studies of control slices always conducted in parallel (two of the six slices per animal), a sham exchange with oxygenated aCSF was performed. After variable periods, glucose and oxygen was reintroduced and the gas mixture switched to 95% O2/5% CO2 and temperature increased to (or maintained at) 36°C in all groups over 30 minutes. All slices were incubated at 36°C for another 1.5 hours, which is sufficient to allow histologic damage to become visible while giving reversibly damaged cells time to recover (Izumi et al., 1996; Pizzi et al., 1995).
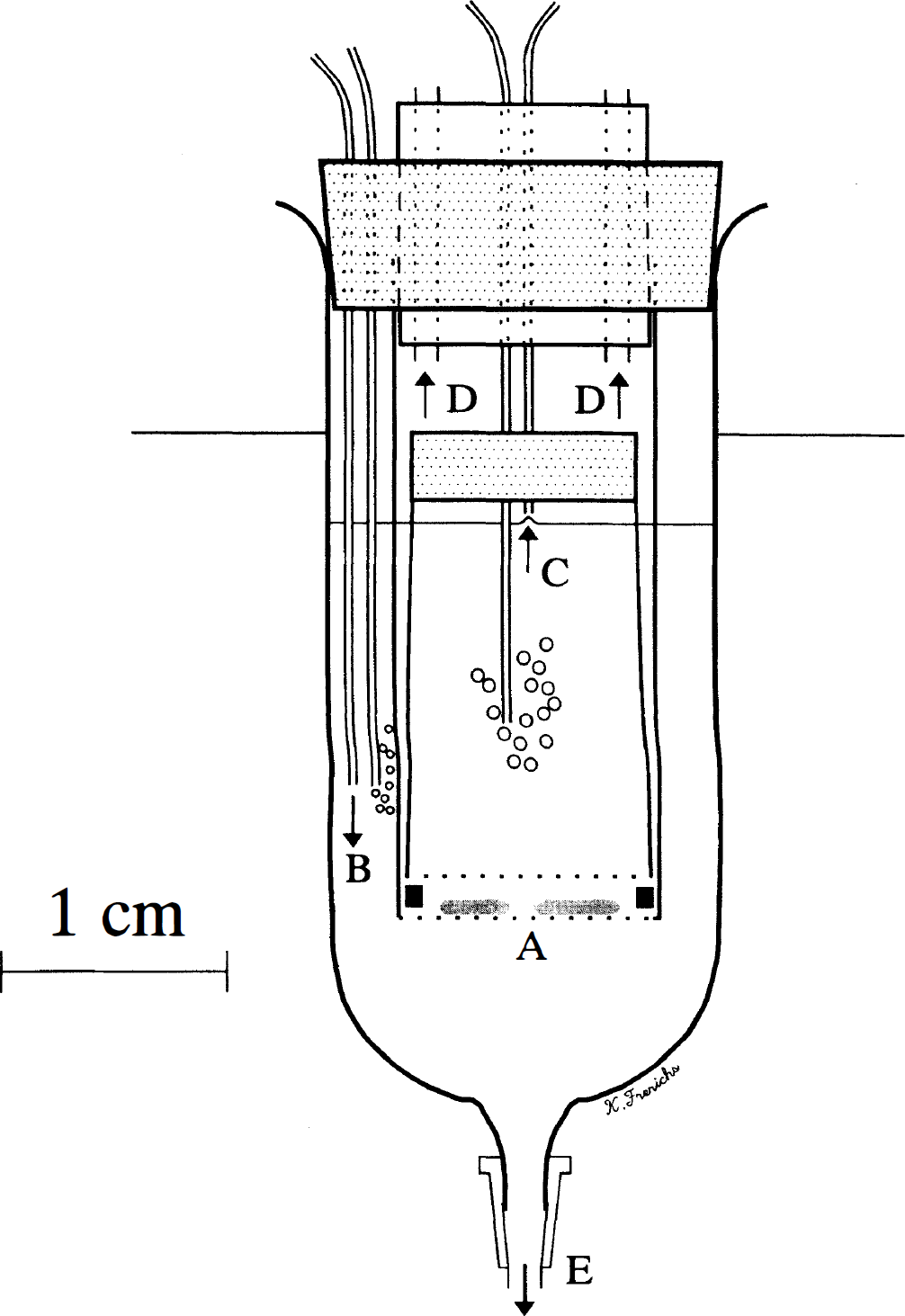
Diagram of the incubation chamber. Hippocampal slices rest on a glass filter mesh (A). Continuous oxygenation is provided at two locations and fresh medium is constantly added (B). Chamber volume is kept constant at 4 mL by vacuum aspiration (C). The chamber is adequately vented (D) and medium can be rapidly exchanged (E).
ATP
In preliminary experiments, slice ATP levels were determined to assess metabolic stability in our incubation system. Slices were frozen in isopentane cooled to −40°C and homogenized in 0.3 mmol/L of perchloric acid. The homogenate was centrifuged at 10,000g for 5 minutes. The pellet was resuspended in 1 N of NaOH and protein content determined (Bradford, 1976). The supernatant was neutralized with 3 mol/L of KHCO3 and centrifuged at 10,000g for 5 minutes. The resulting supernatant was assayed for ATP content by the luciferinluciferase technique (Lust et al., 1981).
Morphological analysis
Slices were immersion-fixed in freshly prepared paraformaldehyde (4% in phosphate-buffered saline, pH = 7.4) for 4 hours and then dehydrated in increasing ethanol concentrations (50%, 70%, 80%, and 90% for 20 minutes each, 95% for 20 minutes twice, and 100% for 20 minutes three times) for histologic processing. Paraffin-embedded slices were serially cut (7-μm thick sections) and stained with cresyl violet. The center portion of the slice, considered the most viable part (Misgeld and Frotscher, 1982), was examined by light microscopy. Data analysis was performed according to the method for quantal bioassays described by Zivin and Waud (1992). This approach is useful if an obvious quantal or binary effect can be identified in response to an experimental manipulation such as a neurologic insult. Then, instead of comparing outcomes (graded response) at a fixed insult severity between groups, it may be advantageous to vary the insult severity to compare different probabilities of the quantal response between groups. Examples of such binary responses include life or death, presence or absence of a neurologic deficit, or any other all or none end point. Analysis of binary response curves is analogous to the determination of the pharmacologic ‘LD50’ (the dose at which 50% of the subjects die) through nonlinear curve fitting. Differences in LD50 values can be compared statistically between groups. The mathematical basis of this analysis and the generation of binary response curves is explained in detail elsewhere (Zivin and Waud, 1992). In the current study, the two slices in each chamber were considered duplicates to assure consistency of slice responses. Therefore, the two slices were scored individually and then combined as a single determination, called a slice set for statistical purposes.
Analysis of variance followed by Scheffé‘s method was used to compare groups statistically. All data are presented as mean ± SD.
RESULTS
In the hibernating group, bout duration before decapitation ranged from 4 to 96 hours (average, 39 ± 28 hours). Body temperature during hibernation was 6.9 ± 0.4°C versus 36.2 ± 0.5°C in the active squirrels and 36.3 ± 0.4°C in rats. Viability of hippocampal slices under control conditions was confirmed morphologically and biochemically. ATP concentrations in slices maintained for 4 hours under control conditions at 36°C were similar in all three groups (rats: 12.2 ± 1.6 nmol/mg protein, n = 4; active squirrels: 11.5 ± 1.7 nmol/mg protein, n = 4; hibernating squirrels: 12.8 ± 2.8 nmol/mg protein, n = 4, P = 0.71) and similar to levels at 1 hour after decapitation. The ATP levels measured are in excellent agreement with those reported previously in hippocampal slices (Whittingham et al., 1984). Furthermore, the in vitro rate of protein synthesis in hippocampal slices incubated under these conditions approximates 40% of rates observed in vivo (Frerichs et al., 1997) which compares favorably to previously published rates (Lipton, 1987).
Preliminary experiments with increasing durations of in vitro hypoxia and aglycemia (IVHA) revealed that individual slices tended to be either severely damaged or relatively intact. An intact slice from an active ground squirrel subjected to a sublethal dose (3 minutes IVHA at 36°C) is shown in Figure 2A and C. A severely damaged slice (6 minutes IVHA at 36°C) characterized by poorly staining neurons with marked cytoplasmic swelling and condensed chromatin is shown in Figure 2B and D. Perikarya appear as large, nearly empty spaces. The appearance of the CA-1 sector correlated best with the duration of IVHA and was commonly the only sector exhibiting severe ischemic necrosis (Fig. 2B) especially after less severe insults. There was no correlation between the appearance of CA-3 and CA-1 or CA-4 or dentate gyrus and CA-1, but more severe insults usually led to the destruction of all sectors. These observations were made in slices from all groups of active and hibernating squirrels as well as rats. Therefore, a quantal bioassay for in vitro tolerance of the CA-1 sector to IVHA was devised. Individual slices were categorized in a binary fashion as either poor or good. The presence of occasional dark neurons did not exclude a slice from being classified as good. A representative comparative series in which this principle can be applied is shown in Figure 3. Hippocampal slices from rats and active and hibernating ground squirrels were subjected to increasing durations of IVHA at 36°C. The appearance of the CA-1 region in the rat series changed from good to poor after 5 minutes of IVHA. In the active ground squirrel group (non-hibernating), 5 minutes of IVHA were tolerated, but 6 minutes resulted in a poor outcome while in the hibernating ground squirrel group, up to 7 minutes of IVHA were still associated with a good outcome.
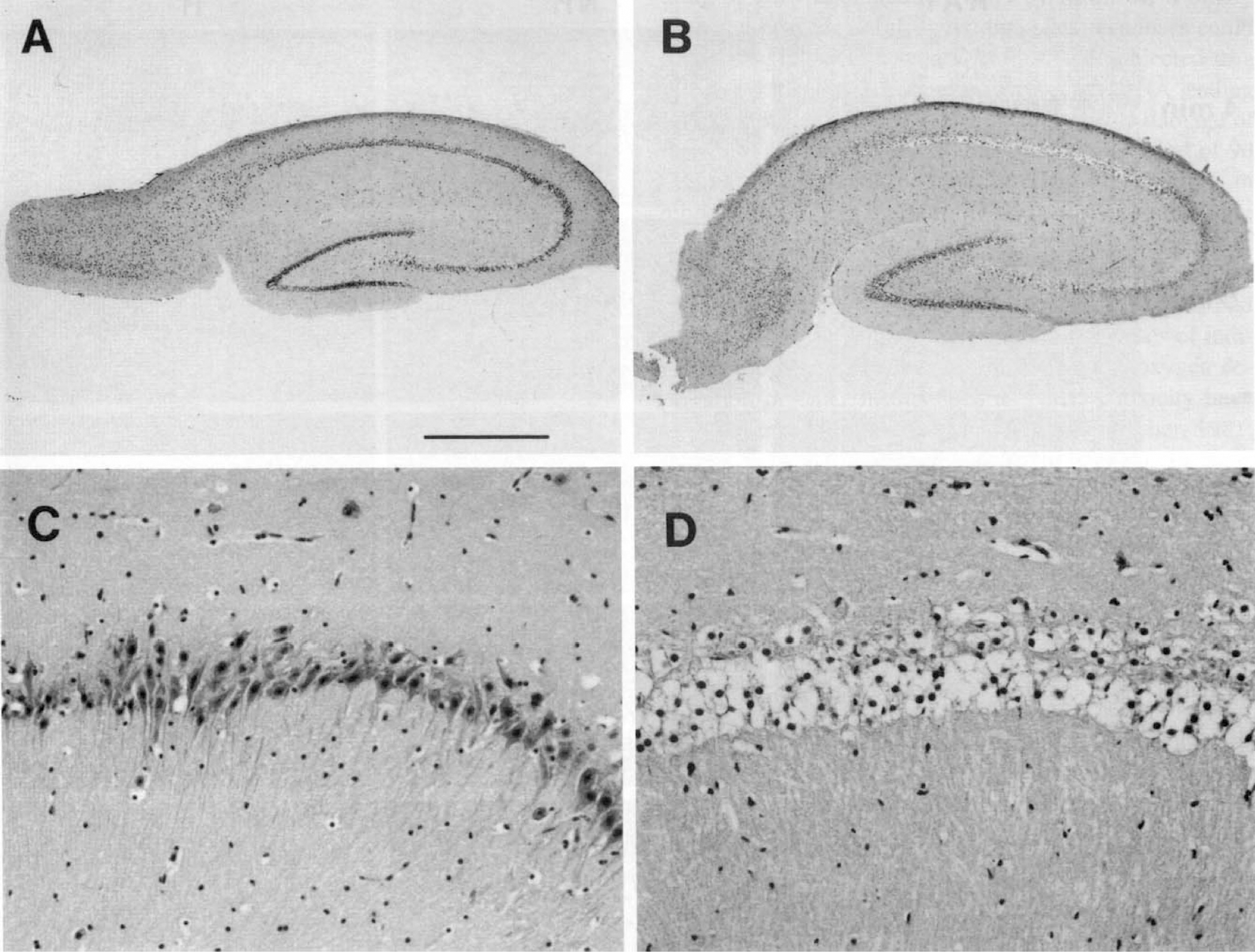
Cresyl violet-stained sections of hippocampal slices from ground squirrel. After a sublethal insult (3 minutes of IVHA), slices appear intact under low
Figure 4 summarizes the results at all three insult temperatures. Vertical bars at 36°C show the calculated duration of IVHA in minutes required to cause a poor outcome in half the slice sets (LD50 analogue) within the group. In all cases, both slices of each slice set showed identical morphologic responses, and the histologic appearance of all concurrently incubated control slices could be classified as good. The LD50 value could be derived from the analysis of the quantal dose-response curve through nonlinear curve fitting as outlined in the Materials and Methods section. Individual outcomes from all determinations are either shown as open (good) or filled (poor) circles. At 36°C, slices obtained from hibernating animals tolerated significantly more IVHA (7.4 ± 0.5 min, n = 12, nanimals = 6) than both active squirrels (5.6 ± 0.4 min, n = 16, nanimals = 8, P < .05) and rats (4.6 ± 0.3 min, n = 14, nanimals = 7, P < .05). No statistical difference was found between active squirrels and rats: At the lower insult temperatures, good and poor outcomes showed no overlap indicating low variability of slice responses. Therefore, curve fitting necessary to generate the LD50 value could not be performed. Data are expressed instead as the maximal duration of IVHA tolerated by slices in each group. After consultation with a local statistician, it was believed that no statistical/mathematical test appeared applicable or necessary to state that differences exist between the groups at the lower temperatures. The large differences in thresholds between animal groups within the same temperature group appear to outweigh any lack of precision in defining the exact threshold value within each group at 20°C and 7°C. More experiments close to the threshold duration within each group would likely generate data points that overlap around the true threshold within the group making statistical analysis as for the 36°C group possible. At 20°C, slices from rats tolerated up to 15 minutes, active squirrels 30 minutes, and hibernating animals 50 minutes of IVHA. At 7°C, species-specific differences were even greater but maximal tolerance was again observed in hibernating squirrels (rat 85 min, n = 6, nanimals = 3; active squirrel 210 min, n = 12, nanimals = 6; hibernating squirrel 240 min, n = 5, nanimals = 3).
DISCUSSION
The present study for the first time provides experimental evidence that mammalian hibernation confers tolerance to transient IVHA in an in vitro model of cerebral ischemia. We further conclude that, in some measure, the hibernation phenotype is preserved in vitro in hippocampal slices. The hippocampal slice preparation enabled us to compare effects of oxygen and glucose deprivation in vitro over a wide range of experimental temperatures that could not have been accomplished in vivo.
Hippocampal slices have been used extensively to study effects of oxygen and glucose deprivation in vitro. The slice system has several limitations and differs from the in vivo state in two major respects: (1) perfusion is absent and hence interaction with blood-borne elements can not occur, and (2) afferents and efferents have been severed. The brain slice preparation, however, gave superior control over the extracellular environment which was crucial for the current study.
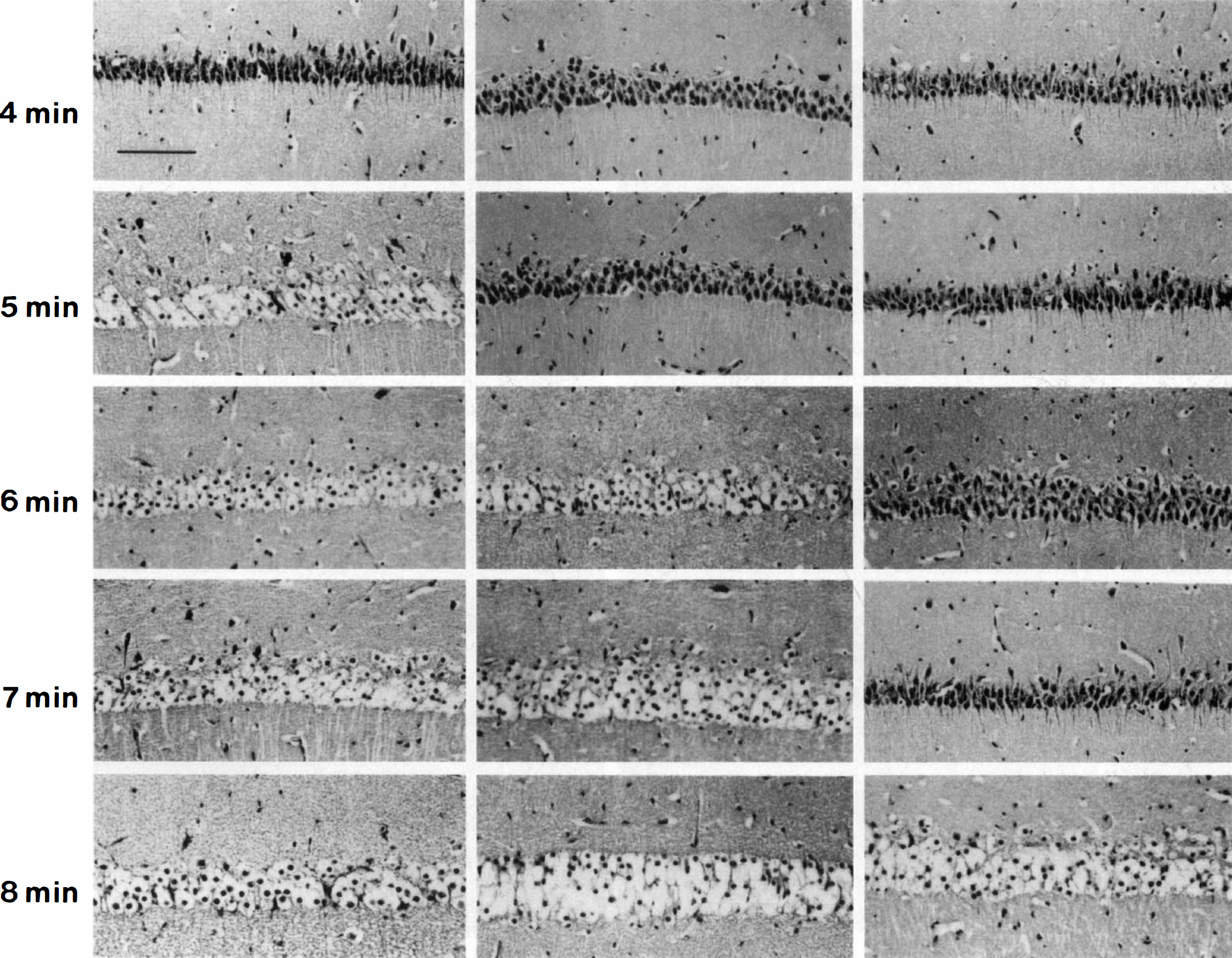
Appearance and binary response of the CA-1 sector in cresyl violet-stained sections of representative hippocampal slices from rats and active (NH) and hibernating (H) ground squirrels subjected to 4 to 8 minutes of IVHA. The appearance of the CA-1 sector after 4 minutes of IVHA in the rat slice was scored as good, after 5, 6, 7, and 8 minutes as poor, indicating that more than 4 minutes of IVHA were not tolerated by slices from rats. In active squirrels, up to 5 minutes and in hibernating squirrels, up to 7 minutes of IVHA, respectively, were associated with a good outcome in this representative set of animals. Scale bar = 20 μm.
The current chamber design was chosen to permit biochemical and morphologic evaluation of the slice and to facilitate rapid medium exchange for precise timing of the insult to minimize variability. Morphology is considered to be a powerful parameter of slice viability (Alger et al., 1984). The ways in which brain slices and their responses to in vitro insults are evaluated are far from standardized (for overview see Lipton et al., 1995, Aitken et al., 1995). Ideally, a set of morphologic (histology), metabolic (oxidative metabolism, protein biosynthesis), and electrophysiologic (population spikes, field potentials) criteria should be satisfied to accept results as representative for in-situ–like behavior (Lipton et al., 1995). In reality, however, this goal is very difficult to achieve and shortcomings and limitations lie within each of the three parameters. Correlation between different parameters, such as morphology and electrophysiology has been good, however. Preservation of synaptic activity appears to be well-correlated with a good histologic outcome and, conversely, loss of such activity tends to predict irreversible damage by morphologic criteria (Weber and Taylor, 1995; Misgeld and Frotscher, 1981). Viability of slices incubated in the current chamber was confirmed by histologic appearance, levels of ATP, and rates of protein biosynthesis (Frerichs et al., 1997), all of which were found to be consistent with previously published data (Whittingham et al., 1984; Lipton, 1987).
Our results accord with previous observations that the CA-1 sector exhibits relative vulnerability to metabolic insults such as IVHA (Aitken and Schiff, 1986). Evaluating in vitro tolerance to IVHA by means of a binary bioassay proved useful. Individual slice responses could be easily graded in a binary fashion and subjected to a quantal assay to determine the duration of IVHA leading to the morphologic equivalent of irreversible damage of the CA-1 sector in each group. A recovery period of 90 minutes is adequate to show acute neuronal injury in hippocampal slices by morphologic criteria (Izumi et al., 1996; Pizzi et al., 1995), in fact, histologic evidence of irreversible damage may appear as early as 20 minutes after an insult such as prolonged hypoxia (Misgeld and Frotscher, 1981). Binary morphologic responses of individual hippocampal slices to glucose and/or oxygen deprivation or other insults such as NMDA toxicity have been observed previously (Misgeld and Frotscher, 1981; Pizzi et al., 1995; Weber and Taylor, 1994). In these reports, morphologic responses were either dominated by the presence of healthy cells or the presence of poorly staining, severely ballooned neurons with very few interspersed intact cells. Good correlation of these morphologic outcomes with other parameters such as electrophysiologic responses were found. Little or no correlation was found with the number of dark neurons present.
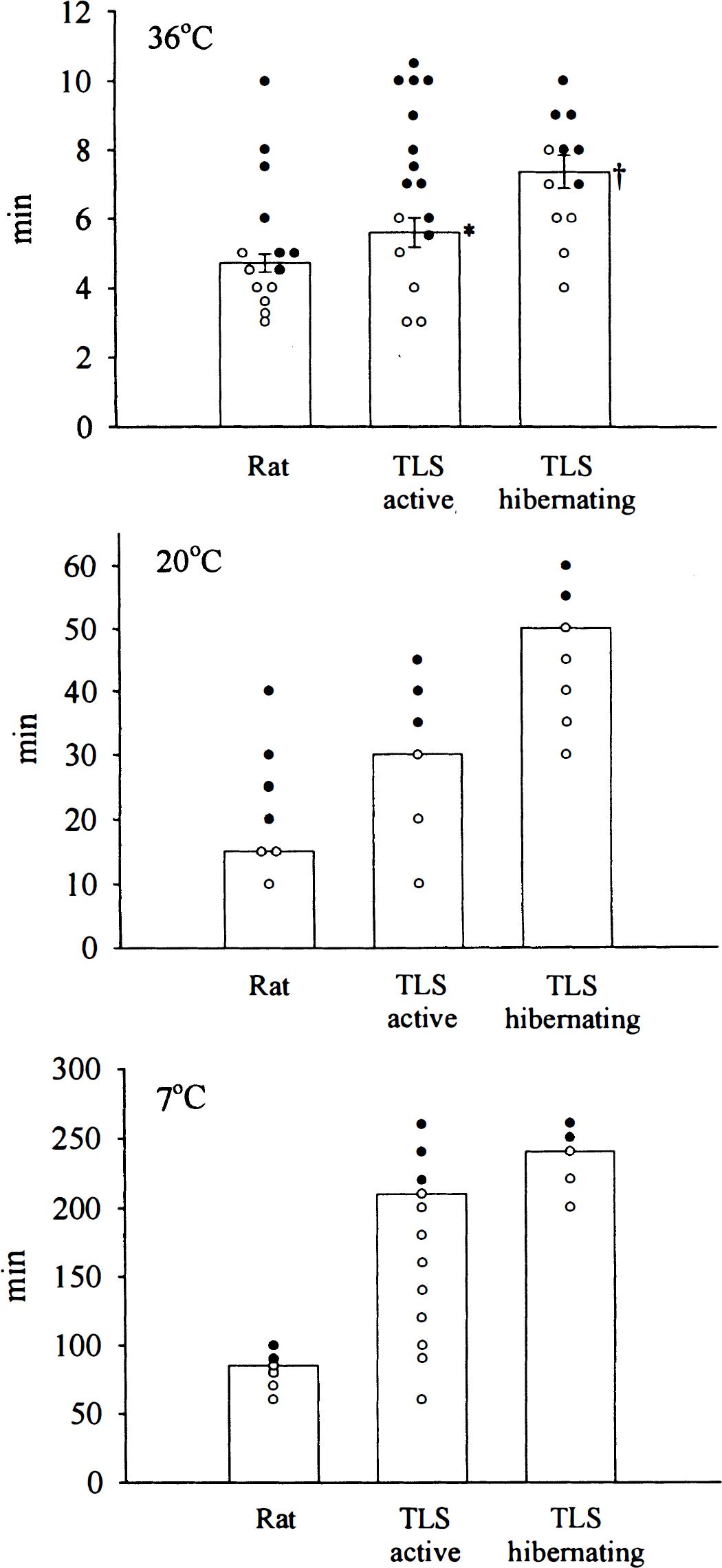
Effect of various durations of IVHA on slice outcome. Surviving slices are shown as open, poor slices as filled circles. Hypoxia and aglycemia was induced at three different temperatures, 36°C, 20°C, and 7°C. Slices were equilibrated at 36°C before the insult and allowed to recover at 36°C. Slices from hibernating animals were most tolerant in all three groups. At 36°C, no difference was found between active squirrels and rats. (*P < .05 active versus hibernating squirrels, †P < .05 hibernating squirrels versus rats). At the lower temperatures the order of increasing tolerance was rat < active squirrel < hibernating squirrel and there was no overlap of slice responses within animal groups.
Hippocampal slices obtained from hibernating animals were the most tolerant to IVHA compared with both active squirrels and rats at each of the experimental temperatures. This indicates that tolerance was induced specifically by the hibernation state because seasonal adaptations in preparation for the hibernation season had occurred in both active as well as hibernating squirrels. Furthermore, tolerance was noted at the earliest stages of the hibernation bout studied (4 hours) indicating that resistance to ischemia was induced rapidly. This would suggest that artificial induction of a similar tolerant state for therapeutic purposes in other species might be practicable. At 7°C, the difference in tolerance between slices from active and hibernating squirrels was small compared to the much greater difference between both squirrel groups and rats. Although small, the difference between hibernating and active animals reflects an additional hibernation-specific effect. The observation of increased tolerance in the active ground squirrel manifested at very low temperatures, however, is intriguing and may reflect a species-specific adaptation induced by cold alone. In fact, profound hypothermia in combination with deprivation of oxygen and glucose may have triggered some of the cellular changes that are responsible for increased tolerance compared with non-hibernating species. Previous work on hippocampal slices from hibernating species indicated that it might be possible to preserve features of hibernation in vitro. Functional survival of brain slices from hibernating squirrels was improved under conditions of prolonged hypothermia, an effect found to be both hibernation- and species-specific (Pakhotin et al., 1990). Murphy (1977) observed that cortical slices from hibernating golden hamsters preserved high-energy phosphate levels better during prolonged cold storage than active controls.
Hibernation is associated with tolerance to profound hypothermia. Many similarities exist between the detrimental effects of deep hypothermia and the effects of oxygen and glucose deprivation. Similar to ischemia, profound cold damages by disruption of cellular metabolic processes and interference with their regulation. Detrimental effects include alterations of metabolic regulatory enzymes, loss of ion homeostasis, disruption of membrane structure, and disturbance of receptor and transport functions, (Storey and Storey, 1990; Willis, 1987; Musacchia, 1984; Hochachka, 1986) and may explain some of the adverse reactions and poor outcomes associated with the clinical use of profound (<32°C) hypothermia (Wass and Lanier, 1996).
Numerous neuroprotective strategies have shown remarkable efficacy in models of cerebral ischemia and trauma. Success on the level of clinical trials has remained elusive, however. This may be due to the complexity of events that contribute to the propagation of damage. Identification of the regulatory pathways that confer resistance in animal models of natural tolerance to hypoxia and ischemia could offer an alternative approach to this difficult problem (Hallenbeck and Frerichs, 1993). Several specific adjustments occur during hibernation that may be involved in preservation of homeostasis and the induction of tolerance. Metabolic rate depression may be due to reversible modifications of several enzymes of glucose metabolism (Storey, 1987; Storey and Storey, 1990) and of the respiratory chain (Pehowich and Wang, 1984). While substrate availability is reduced it remains matched with residual metabolic demands. This coordinated cellular shutdown appears to favor energy consumption for critical processes such as ion homeostasis while suppressing others such as global protein synthesis. The most energy-consuming process in mammalian nervous tissue is the preservation of ion gradients. Preservation of ion homeostasis in hibernation may involve reversible changes in general membrane permeability as well as specific alterations of the properties of ion pumps such as the Na+-K+ ATPase (Houslay and Palmer, 1978). Igelmund et al. (1996) observed that hippocampal slices from hibernating hamsters exhibited significantly less NMDA-induced Ca2+ influx compared with active animals over a wide range of temperatures. Reduced sensitivity of NMDA receptor-mediated Ca2+ influx could protect hippocampal neurons during episodes of excessive neuroexcitation. Furthermore, endogenous opioids may be involved in the regulation of hibernation (Oeltgen et al., 1987; Cui et al., 1996) and may also have a central role in acute adaptations to hypoxia (Mayfield and D'Alecy, 1994). The massive reduction of all circulating while cell lines and platelets to about 10% of normal levels (Frerichs et al., 1994) implicates humoral factors in the pathways that regulate hibernation.
In summary, the current study provides evidence that hibernation is associated with tolerance to deprivation of oxygen and glucose in vitro. Efforts should be directed at identifying the mechanisms that regulate the coordinated suppression of cellular functions in hibernation. This could lead to new neuroprotective strategies.
