Abstract
Experimental data suggest that postischemic blood glucose concentration plays an important role in modulating both ischemic cerebral infarction and selective neuronal necrosis. This study investigated the association between functional neurological recovery and blood glucose concentrations in human cardiac arrest survivors. A group of 145 nondiabetic patients were evaluated after witnessed ventricular fibrillation cardiac arrest. Data regarding cardiac arrest were collected according to an internationally accepted protocol immediately after arrival. Blood glucose was measured on admission and 6, 12, and 24 h thereafter. To control for duration of cardiac arrest and cardiogenic shock, both known to influence outcome as well as blood glucose, levels, Spearman rank partial correlation was used. In this multivariate analysis, a high admission blood glucose level tended to be associated with poor neurological outcome (rs = −0.16, n = 142, p = 0.06). The association between high median blood glucose levels over 24 h and poor neurological outcome was strong and statistically significant (rs = −0.2, n = 145, p = 0.015). High blood glucose concentrations occurring over the first 24 h after cardiac arrest have deleterious effects on functional neurological recovery. Whether cardiac arrest survivors might benefit from reduction of blood glucose levels needs further investigation.
Keywords
Impaired neurological recovery is frequent in human cardiac arrest survivors (Cummins et al., 1991; AHA, 1992). Duration of cardiac arrest (BRCT I, 1985; Martens et al. 1993), preexisting disease (Mullie et al., 1989), and primary ECG rhythm (Pepe et al., 1993) are some of the known determinants of outcome. In the early postresuscitation period, a number of pathophysiologic alterations are caused not only by primary ischemia, but also by their combination with reoxygenation and recirculation (Negovsky and Gurvich, 1995; Safar, 1993). Important factors of this postresuscitation disease are disturbances of cerebral blood flow (Fischer et al., 1979; Sterz et al., 1992; Fischer and Hossmann, 1995; Cohan et al., 1989) and cerebral oxygen extraction (Müllner et al., 1986b; Takasu et al., 1995). The role of blood glucose in focal and global cerebral ischemia has only been partially elucidated. It is known that elevated preischemic blood glucose levels increase neuronal damage (Rehncrona et al., 1981; Gardiner et al., 1982; Wagner et al., 1992; Ginsberg et al., 1980). The role of blood glucose levels in the reperfusion phase after global cerebral ischemia is not clear. In human cardiac arrest survivors, elevated blood glucose levels on admission were predictive for unfavorable neurologic recovery (Longstreth et al., 1982; Longstreth and Inui, 1984). However, these findings were best explained by prolonged cardiopulmonary resuscitation (CRP), leading to both a greater increase in blood glucose level and a worse neurological outcome (Longstreth et al., 1986). Voll and Auer (1988) demonstrated in an animal model that postischemic blood glucose concentration plays an important role in modulating both ischemic infarction and selective neuronal necrosis. This study investigated the association between functional neurological recovery and blood glucose levels over the first 24 h after human cardiac arrest.
MATERIALS AND METHODS
Patients
Patients for this study were selected from the population served by the Department of Emergency Medicine at the Vienna General Hospital, a tertiary-care university hospital. The study period ranged from August 1, 1991, through May 31, 1994. Patients of >18 years of age who suffered a witnessed ventricular fibrillation cardiopulmonary arrest with return of spontaneous circulation for a duration ≥24 h were eligible for inclusion in the study. Patients whose cardiopulmonary arrest was associated with trauma, hypothermia, drowning, drug overdose, or a primary respiratory arrest were excluded from the study. Patients with a known history of diabetes and/or who received insulin and/or parenteral glucose during CPR or within the first 24 h after return of spontaneous circulation were excluded from the study. Further, we excluded patients whose functional neurological status could not be assessed; that is, if they died before the withdrawal of sedoanalgesia. Cardiopulmonary arrest was defined as the absence of both spontaneous respiration and palpable pulse. Return of spontaneous circulation was defined as electrical activity in the ECG and palpable pulses. Treatment in the field as well as in the hospital was in accordance with the American Heart Association guidelines for basic and advanced cardiac life support and for postresuscitation care (AHA, 1992). Out of hospital as well as in hospital, the administration of parenteral glucose was not a routine procedure: it was administered only if a patient had documented hypoglycemia. In the hospital, all patients received standard intensive-care treatment such as controlled mechanical ventilation, sedoanalgesia with midazolam (0.2 mg/kg/h), fentanyl (0.004 mg/kg/h) for at least 24 h and low-dose dopamine (1.5 γg/kg/min). Other medications were left to the discretion of the attending physician.
Study design and data collection
Data were acquired retrospectively according to the Utstein style, the recommended guidelines for uniform reporting of data on the arrival of each patient after out-of-hospital cardiac arrest (Cummins et al., 1991). Evaluated data included location of cardiac arrest (in or out of hospital), duration of the interval from time of collapse (presumed time of cardiac arrest) until basic and/or advanced life support and return of spontaneous circulation, first rhythm in the ECG, and recorded history for the individual patients, especially regarding the cause of cardiac arrest. The time from recognition of collapse until the emergency medical system was contacted was evaluated by one of the investigators who personally interviewed one or more witnesses. The time of cardiac arrest was estimated from the time that the emergency medical system was contacted and the time when the collapse was recognized. For the time interval from cardiac arrest the initiation of basic and/or advanced life support, we presummed that there was insufficient systemic blood flow (that is, duration of no blood flow). We presumed the time interval from initiation of life support until return of spontaneous circulation to be representative of the duration of low blood flow. Blood glucose and blood lactate levels were measured in fixed time intervals: on admission and 6, 12, and 24 h afterward. Measurements were carried out with an Hitachi 717 autoanalyzer (Boehringer Mannheim, Mannheim, Germany). In our laboratory, upper normal values for blood glucose and lactate concentration are 100 mg/dl and 2.2 mg/dl, respectively. Blood lactate was measured as a noninvasive estimate of cardiogenic shock severity (Carden et al., 1985; Mizock and Falk, 1992; Rackow and Weil, 1990).
Outcome measures
Cerebral function which was assessed prospectively on arrival and in regular intervals within 6 months after return of spontaneous circulation was expressed as cerebral performance category (CPC) (BRCT I, 1986; Cummins et al., 1991), which is based on the Glasgow overall performance categories (Jennett and Bond, 1975). The definitions are CPC 1, conscious and alert with normal function or only slight disability; CPC 2, conscious and alert with moderate disability; CPC 3, conscious with severe disability; CP4, comatose or in a persistent vegetative state; and CPC 5, brain death. The best CPC score achieved within 6 months was used for calculation. A CPC score of 1 or 2 was considered good and a CPC source of 3–5 as a bad neurological outcome. The investigators assessing the CPC score were blinded to data concerning resuscitation as well as to blood glucose levels of the patients.
Statistical analysis
Data were not normally distributed and thus expressed as median ± the interquartile range (IQR) according to the Utstein style (Cummins et al., 1991). For calculation, we used the admission values as well as the median of the four serial measurements for each patient. Percentages were determined for dichotomous variables. For the comparison of continuous variables, the Mann–Whitney U test was used to describe the groups but not the outcome measures of major interest, so we did not control for multiple comparison. To assess a trend across the ordered groups of CPCs, we applied the Kruskal–Wallis analysis of variance (H). The chisquared test was used for the comparison of dichotomous variables. All data were computed with SPSS for Windows (Chicago, IL U.S.A.). To assess how blood glucose on admission influenced neurological recovery, we used the Spearman rank partial correlation technique (rs) (Altman, 1991; Martens et al., 1993) to control for long durations of no and low systemic blood flow before return of spontaneous circulation during cardiac arrest and hyperlactatemia presumably caused by cardiogenic shock following cardiac arrest. A second model was developed to assess how median blood glucose over a 24-h period after cardiac arrest influenced neurological recovery. Besides median blood glucose levels over 24 h and neurological outcome, the variables in the second model were median lactate levels over 24 h and, again durations of no and low blood flow. For this analysis, neurological recovery was coded as follows: good functional neurological recovery (CPC 1 or 2) = 1, and bad functional neurological recovery (CPC 3–5) = 0. Thus, a negative association between functional recovery and glucose levels in the Spearman rank partial correlation indicates that high blood glucose levels are associated with bad neurological recovery. A p < 0.05 was considered statistically significant.
RESULTS
In an observational period of 46 months, 228 patients were brought to the emergency department in, or after witnessed, ventricular fibrillation cardiac arrest: 27 were not eligible because spontaneous circulation was not restored, another 30 were excluded because they died within the first 24 h under sedoanalgesia, and another four were excluded because they died thereafter, but still while under sedoanalgesia. Five patients were excluded because they received parenteral glucose during CPR or within the first 24 h after return of spontaneous circulation, and 17 patients had a history of diabetes mellitus.
A total of 145 patients fulfilled the inclusion criteria and were enrolled in the study. Their median age was 60 years (IQR, 52–69 years), 110 patients (76%) were men, and in 102 cases (70%) cardiac arrest occurred out of hospital. The median duration from collapse until begin of basic/advanced live support was 2 ± 7 min, and the duration of low systemic blood flow was 7 ± 16 min. The median blood glucose level over 24 h after cardiac arrest was 155 ± 71 mg/dl, and the median lactate over 24 h was 2.9 ± 3 mg/dl. There was statistical significant trend for higher median blood glucose levels over 24 h the more functional neurological recovery was impaired (H = 19.5, n = 145, p = 0.0006) (Fig. 1). Of 60 patients with unfavorable neurological recovery, 10 regained consciousness, but had severe neurological disability and were still considered as having a poor neurological outcome. Good functional neurological recovery within 6 months was observed in 85 patients (59%). A total of 60 patients (41%) died within 6 months. Patients with good functional recovery were slightly younger and had shorter durations of no and low systemic blood flow (Table 1). Blood glucose on admission as well as median blood glucose over 24 h were lower in patients with good functional neurological recovery (Table 2). On admission, blood lactate was higher in patients with unfavorable neurological recovery (8.7 ± 5.9 mg/dl, n = 59) than in patients with favorable neurological recovery (5.4 ± 5.3 mg/dl, n = 83) (p = 0.0001). Median blood lactate levels over 24 h also were higher in patients with unfavorable outcome (3.8 ±3.3 vs. 2.7 ±2.1 mg/dl, p = 0.0005).
Variables concerning demographics, cardiac arrest, and mortality in 145 patients with good and bad functional neurological recovery after witnessed ventricular fibrillation cardiac arrest
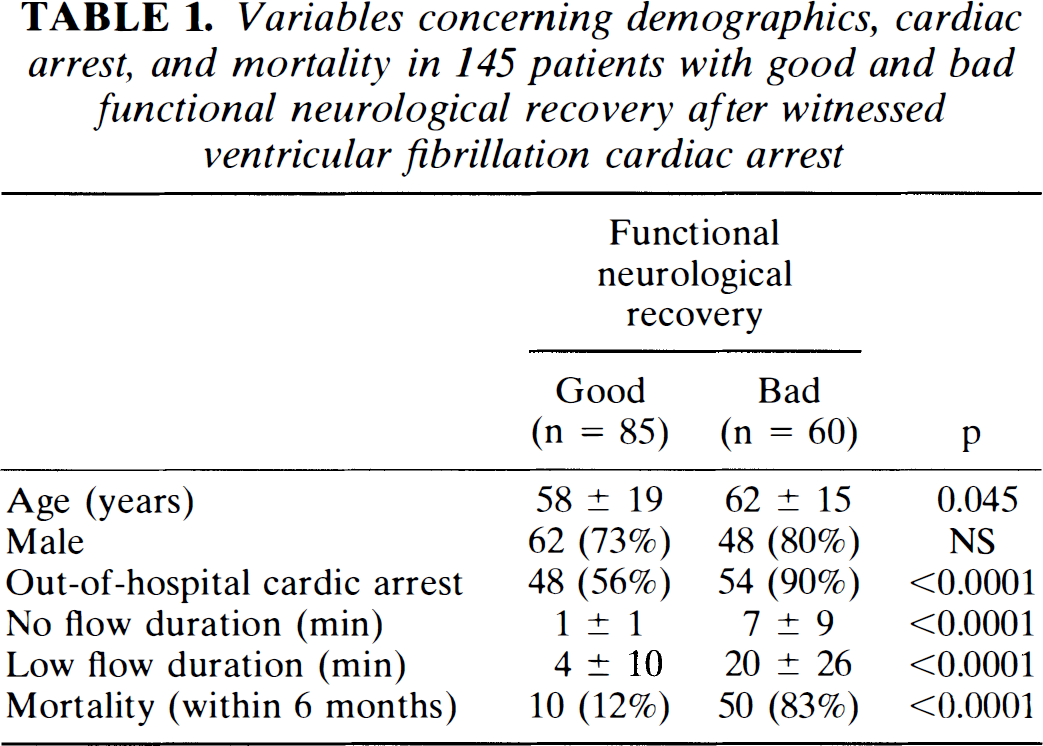
Functional neurological recovery: a cerebral performance category of 1 or 2 was considered as good functional recovery, and a cerebral performance category of 3–5 was considered as bad functional recovery; the best score achieved within 6 months after cardiac arrest was used for calculation. Low flow duration: time interval from the initiation of life support therapy to return of spontaneous circulation. No flow duration: time interval from collapse to the initiation of life-support therapy. Data are presented as median ± range from the 25th to the 75th quartile; there were no missing values among the variables presented.
Blood glucose concentrations in 145 patients with good and bad functional neurological recovery on admission and 6, 12, and 24 h after witnessed ventricular fibrillation cardiac arrest
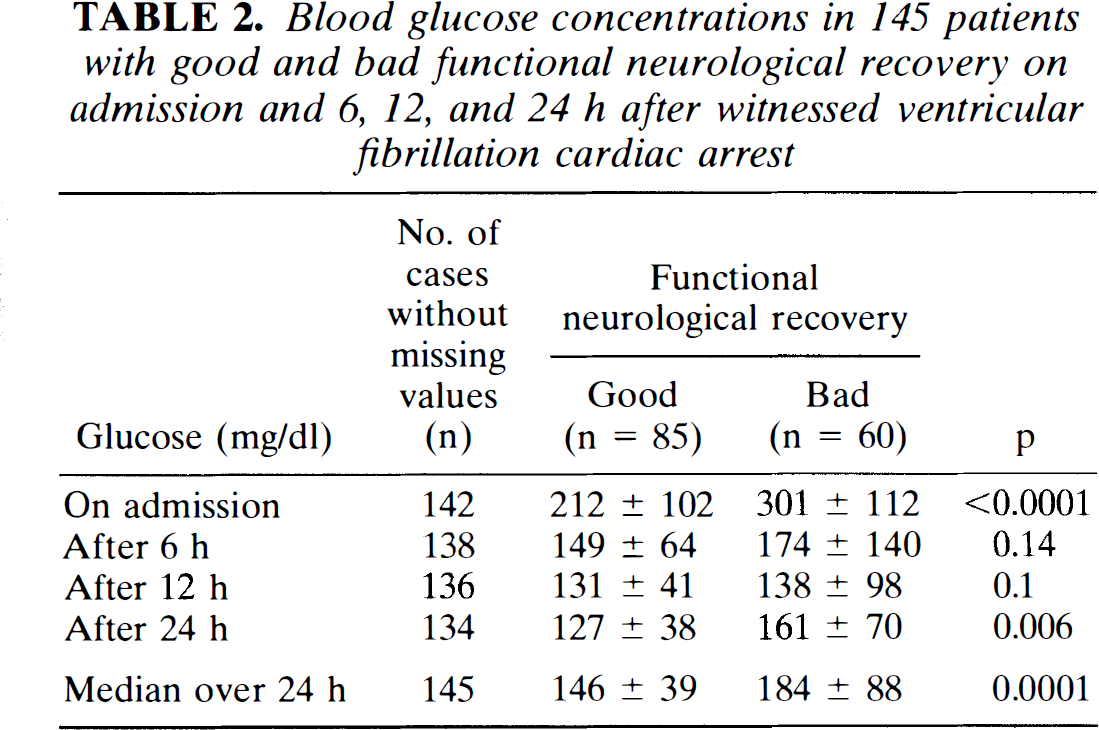
Functional neurological recovery: a cerebral performance category of 1 or 2 was considered as good functional recovery, and a cerebral performance category of 3–5 was considered as bad functional recovery; the best score achieved within 6 months after cardiac arrest was used for calculation: Data are presented as median ± range from the 25th to the 75th quartile.
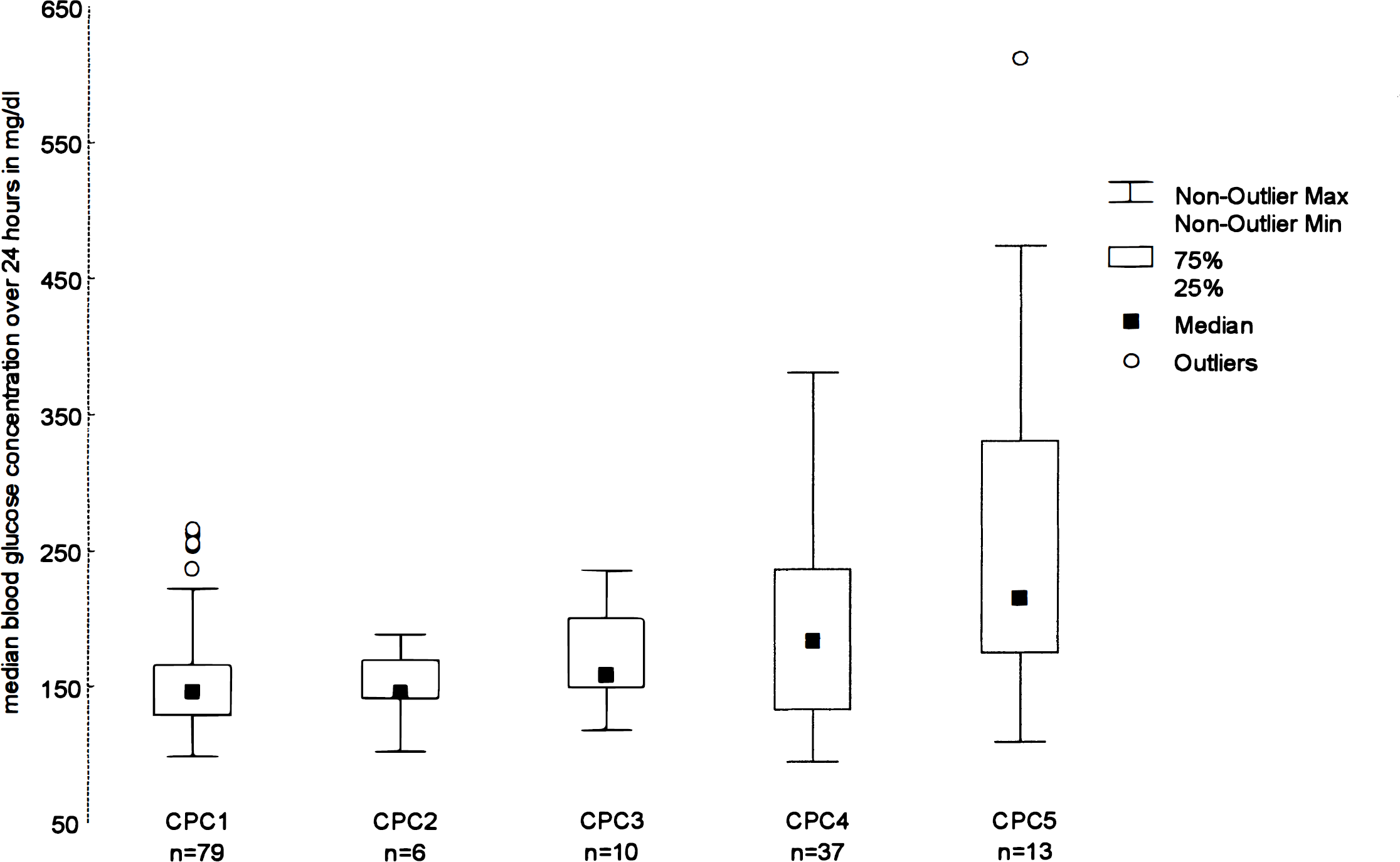
Median blood glucose concentrations over 24 h within the five cerebral performance categories (CPCs) in 145 patients after witnessed ventricular fibrillation cardiac arrest.
After controlling for duration of no or low systemic blood flow and admission lactate, parameters probably influencing outcome as well as blood glucose levels, the Spearman rank partial correlation showed a trend toward unfavorable neurological recovery and high admission blood glucose levels (rs = −0.16, n = 142, p = 0.06). The association between high median blood glucose levels measured over 24 h and unfavorable neurological recovery was stronger and statistically significant (rs −0.2, n = 145, p = 0.015) after controlling for the durations of no and low systemic blood flow and median lactate levels measured over 24 h.
DISCUSSION
The main finding of our study is that, in cardiac arrest survivors, high blood glucose levels over the first 24 h after return of spontaneous circulation are independently associated with unfavorable functional neurological recovery.
The impact of elevated blood glucose levels at the time of ischemia on neuronal injury has been well investigated in animal models. Hyperglycemia is known to impair the recovery of the cerebral energy state (Rehncrona et al., 1981; Gardiner et al., 1982), as well as cerebral perfusion (Ginsberg et al., 1980), and electrophysiologic recovery (Hossmann, 1988). Further hyperglycemic animals had increased cerebral morphological damage (Pulsinelli et al., 1982) and a higher neurological deficit score, seizure incidence, and mortality (Voll and Auer, 1988; LeMay et al., 1988). High glucose levels lead to increased brain tissue lactate accumulation during severe hypoxia (Gardiner et al., 1982). Mammalian astrocytes are known to become more acidotic than the interstitial microenvironment during hyperglycemic and complete ischemia (Kraig and Chesler, 1990). Cellular acidosis together with high lactate concentrations finally leads to the death of neurons (Kraig et al., 1987).
In humans with focal cerebral ischemic stroke, elevated blood glucose concentrations on admission were independently related to neurological deterioration (Davalos et al., 1990). In global cerebral ischemia, as seen in cardiac arrest survivors, elevated admission blood glucose levels are a prognostic indicator, but seem to be more the sequel of prolonged CPR than the cause of adverse neurological outcome (Longstreth and Inui, 1984; Longstreth et al. 1986, 1993). In a study by Longstreth et al. (1993), patients with out-of-hospital cardiac arrest were randomized to receive either 5% dextrose in water or half normal saline intravenously after the event. Overall, there was no difference in outcome between the groups. A subgroup of 220 patients with ventricular fibrillation or asystole cardiac arrest who never awakened had higher admission blood glucose levels than those who did awaken during their hospital stay. Awakening was defined as having comprehensible speech or following commands. Another recent study (Steingrub and Mundt, 1996) that also investigated admission blood glucose levels after cardiac arrest and outcome concluded that there is no association between the concentration of glucose after CPR and neurological outcome, even though there was a significant association between high glucose concentration and unfavorable neurological outcome in a small subset of 16 patients with CPR lasting for >4 min. This study included 80 patients, however, and only 26 had ventricular fibrillation, and neurological outcome was classified by the best CPC achieved within 48 h. Our patient population is smaller compared to the study by Longstreth et al. (1993), but much larger than the population investigated by Steingrub and Mundt (1996). Further, 100 patients (69%) of our study (data not presented) had total cardiac arrest durations of ≥5 min. However, our results are in concert with the data of the studies just cited: admission blood glucose levels are lower in patients with good neurological outcome, but seem to be more an epiphenomenon caused by duration of cardiac arrest and subsequent resuscitation.
Until now, it has never been determined whether postischemic blood glucose levels over an extended period in humans influence functional neurological recovery. We showed that there is an independent association between blood glucose levels in a 24 h postischemic period and functional neurological outcome. Why high blood glucose concentrations are deleterious in this setting remains speculative. It was shown that hyperglycemia in areas with incomplete ischemia may be even more deleterious than in complete ischemia (Rehncrona et al., 1981). Global and especially regional cerebral blood flow disturbances are known to occur after cardiac arrest (Fischer et al., 1979; Sterz et al., 1992; Fischer and Hossmann, 1995; Cohan et al., 1989). Both factors in conjunction could explain our observation.
The reason for elevated postischemic blood glucose levels in the post-CPR period without a history of diabetes is unknown. In the animal model of global cerebral ischemia, besides glucose administration, hyperglycemia is postulated to be an effect of intrinsic release and/or administration of catecholamines (Hossmann, 1988). In nondiabetic human patients with acute myocardial infarction, hyperglycemia was related to heart failure (Bellodi et al., 1989). Most likely, spontaneous hyperglycemia in humans in the post-CPR period is a sequel of the release of endogenous stress hormones like cortisol, glucagon, and epinephrine (Gore et al., 1993; Schultz et al., 1993). The exogenous administration of epinephrine also can cause hyperglycemia (Ensinger et al., 1994), which has also been shown in the setting of cardiac arrest (Longstreth et al., 1993; Calle et al., 1989). Even though hyperglycemia on admission after cardiac arrest might be a surrogate for the duration of the arrest (Longstreth et al., 1993), our data suggest a causal relation between blood glucose elevated over an extended period and neurological recovery, after controlling for the severity of this insult.
A reduced systemic blood flow as seen in heart failure after CPR might independently lead to reduced functional neurological recovery. In a recent study, we demonstrated that hypotension in cardiac arrest survivors within the first 2 h after return of spontaneous circulation is independently associated with unfavorable functional neurological recovery (Müllner et al., 1996a). For the present study, we considered blood pressure insufficient to predict systemic blood flow and thus heart failure (Wo et al., 1993). Blood lactate is known to be the best single objective measure of the severity of shock (Mizock and Falk, 1992; Rackow and Weil, 1990). Blood glucose and blood lactate are known to be interrelated via the pathway of anaerobic glycolysis. To control for heart failure as a potential cause of adverse neurological recovery and/or hyperglycemia, we included blood lactate in the analysis.
The remaining findings of this study have already been well described. There is evidence that durations of no and low systemic blood flow during cardiac arrest are major determinants of functional neurological recovery (BRCT I, 1985; Martens et al., 1993).
Limitations of the study
Difficulties in conducting clinical studies in cardiac arrest patients are numerous and well recognized (Cummins et al., 1991). By using an internationally recognized protocol, we eliminated some of the flaws of retrospective studies.
Imprecisions surrounding the exact time of collapse were minimized by tracing back from the accurate time of the emergency medical service activation to the time of recognition of collapse by one investigator personally interviewing the witnesses.
We had no better possibility to assess whether a patient was diabetic than by interviewing the patient's relatives and physician and by reviewing the patients medication. Further, we could not assess preischemic blood glucose concentrations. Thus, we can not control for preischemic blood glucose levels known to influence neurological outcome, at least in focal cerebral ischemia.
Implications of the study
We hypothesize that cardiac arrest survivors with hyperglycemia occurring within the first 24 h after ischemia might benefit from a reduction in blood glucose levels. Experimental data suggest that insulin mitigates neuronal damage in normoglycemic and hyperglycemic global cerebral ischemia in rats (LeMay et al., 1988; Voll and Auer, 1988, 1991). And even though Longstreth and colleagues (1993) demonstrated that the administration of limited amounts of glucose after cardiopulmonary arrest is not harmful for neurological recovery, further prospective studies are needed to determine the potential benefits of avoiding hyperglycemia following cardiac arrest.
