Abstract
It has recently been shown, using either genetically engineered mutant mice (nitric oxide synthase [NOS] knockout) or specific pharmacological tools, that type I NOS (neuronal isoform of NOS, [nNOS]) participates in coupling cerebral blood flow to functional activation. However, it has not been clearly established whether the associated metabolic response was preserved under nNOS inhibition and whether this action was exerted homogeneously within the brain. To address these issues, we analyzed the combined circulatory and metabolic consequences of inhibiting the nNOS both at rest and during functional activation in the rat anesthetized with α-chloralose. Cerebral blood flow and cerebral glucose use (CGU) were measured autoradiographically using [14C]iodoantipyrine and [14C]2-deoxyglucose during trigeminal activation induced by unilateral whiskers stimulation in vehicle- and 7-nitroindazole-treated rats. Our data show that inhibition of nNOS globally decreased CBF without altering CGU, indicating that NO-releasing neurons play a significant role in maintaining a resting cerebrovascular tone in the whole brain. During whisker stimulation, nNOS inhibition totally abolished the cerebrovascular response only in the second order relay stations (thalamus and somatosensory cortex) of the trigeminal relay without altering the metabolic response. These findings provide evidence that the involvement of neurally-derived NO in coupling flow to somatosensory activation is region-dependent, and that under nNOS inhibition, CBF and CGU may vary independently during neuronal activation.
The integrity of brain function relies on the close coupling between functional activity and energy metabolism on one hand and between functional activity and blood flow on the other (Sokoloff, 1977a; Villringer and Dirnagl, 1995). It is clearly established that during functional activation, glucose consumption and blood flow increase in the specific neural structures that subserve the function.
In rodents, activation of the whisker-to-barrel pathway has long been used as an experimental model to study such relationships, because in this species, there exists a one-to-one relationship between the organization of the whiskers and specific units within the primary somatosensory cortex. Input from these peripheral sensory organs projects to the contralateral cortex via two major relay stations: the ipsilateral trigeminal nuclei and the contralateral ventral posteromedial thalamic nucleus. In the cortex, cell bodies in layer IV are arranged in anatomically distinct units called “barrels“, each barrel being supplied by one penetrating artery (Woolsey and Van der Loos, 1970; Cox et al., 1993; for review, Waite and Tracey, 1995). There are two important advantages of this model for investigations related to the coupling of blood flow, metabolism, and neuronal function: first, the physiological mode of activation and second, the large somatotopic representation of the posteromedial barrel subfield (20% of the primary somatosensory cortex in the rat) (Welker, 1971). Therefore, as previously mentioned by others (Greenberg et al., 1979; Ginsberg et al., 1987), this whisker-to-barrel system is ideally suited for the study of local flow/metabolism relationships.
Despite the large number of studies devoted to the characterization of the mechanisms underlying the coupling of cerebral blood flow to brain function, the mediators involved in this physiological coupling have yet to be fully elucidated (Iadecola, 1993 for references). The possible role of nitric oxide (NO) in the vasodilatory response elicited by vibrissal stimulation has been investigated by several authors using nonspecific inhibitors of the different isoforms of the NO synthase (NOS) (Dirnagl et al., 1993; Dirnagl et al., 1994; Wang et al., 1993a; Irikura et al., 1994; Adachi et al., 1994). The effect of inhibiting NOS on the hyperemia induced by stimulation of the trigeminal pathway in the rat was not conclusive. More recent studies using genetically engineered mutant mice lacking either the type I (neuronal) NOS gene or the type III (endothelial) NOS gene have provided more convincing evidence for an important role of neurally derived NO in the coupling of blood flow to brain function (Ma et al., 1996; Ayata et al., 1996).
The assumption that neuronal NOS (nNOS) mediates the vascular response to somatosensory stimulation in the somatosensory cortex is supported by our recent autoradiographic study in which we used 7-nitroindazole (7-NI) as a selective inhibitor of nNOS (Cholet et al., 1996). However, 7-NI did not modify the vascular response at the site of termination of primary afferents (trigeminal nuclei). This result underlines the fact that the role of nNOS in mediating the cerebrovascular response to a functional activation is not homogeneous within the central nervous system.
The mechanisms by which inhibition of nNOS attenuates the cerebrovascular response have not yet been elucidated. In particular, it has not been clearly established whether the metabolic response was preserved after administration of NOS inhibitors.
Therefore, in the current study, we used the quantitative autoradiographic technique with [14C]iodoantipyrine and [14C]2-deoxyglucose as tracers, to measure local cerebral blood flow and cerebral glucose use (CGU) respectively, during a functional activation induced by whisker stimulation. The consequences of inhibiting nNOS with 7-NI were analyzed on resting CBF and CGU (in selected nonactivated brain regions) and on CBF and CGU changes in all activated regions of the trigeminal pathway.
MATERIALS AND METHODS
Animals
The experimental protocols were approved by the Bureau de la Protection Animale of the Ministère de l'Agriculture (authorization Number 02935 for J.S. and 03001 for P.L.).
The experiments were performed on 48 adult male Sprague-Dawley (OFA) rats weighing 265 to 370 g provided from IFFA CREDO (France). The rats were housed in groups of 4, they were given food (Pietrement granulé ‘MS20’ entretien et élevage) and water ad libitum, in an ambient temperature of 22 ± 2°C (40% to 60% humidity) and a 12-hour light/12-hour darkness cycle.
Experimental procedure
The same anesthetic and surgical protocols were followed for the four experimental protocols used for CBF measurement (protocols 1 and 2), CGU measurement (protocol 3) and for NOS activity assay (protocol 4).
Anesthesia was induced with 3.5% halothane administered through a vaporizer (Halothane Vapor 19.3, Dräger, Germany), periodically calibrated using an Anesthetic Agent Monitor (7860, Kontron Instruments, England) in a gas mixture of 30% O2 −70% N2, and maintained under 1% to 1.5% halothane during the whole surgical procedure (30 to 35 minutes duration). Alpha-chloralose (40 mg/kg) was administered subcutaneously in the axillary brown adipose tissue 5 minutes after halothane induction, for maintenance of anesthesia after surgery. Such anesthetic regimen has been previously shown to induce a stable anesthetic state for more than 3 hours in the rat (Bonvento et al., 1994a). Body temperature was maintained at 37.5°C with a heating pad thermostatically controlled by a rectal probe (Homeothermic Blanket Control Unit, Harvard App., South Natick, MA, U.S.A.). Both femoral veins were cannulated for drug administration and replacement fluids. Cannulae were also placed in the femoral arteries to allow measurement of blood gas tensions and continuous monitoring of arterial blood pressure (Gould RS 3400, Gould Electronics, Cleveland, OH, U.S.A.). Lidocaïne gel (2%) was applied to the surgical wounds before closure and a tracheotomy was performed. Thereafter, the rats were placed in a stereotaxic frame (model 1400, Kopf Instruments), mechanical ventilation was started without curarizing agent (Rodent ventilator 683, Harvard App.) at 80 strokes per minute with 0.4 to 0.5 L/min of a gas of O2 enriched air (5% to 10%), and halothane was discontinued. All whiskers expect those of row C were clipped. Arterial PCO2, PO2, and pH were measured periodically (pH/Blood Gas Analyser 178, CIBA Corning Diagnostic S.A., France) and kept within the physiological range. The hematocrit level was determined at the end of each experiment.
Protocol 1.
Protocol 2.
Protocol 3.
Protocol 4.
Data and statistical analyses
Data are presented as mean ± SD. The effects of 7-NI administration on the time-course of CBF, MABP, and heart rate were analyzed by a one way analysis of variance and Dunnett's test.
The statistical significance of the effect of 7-NI on both the physiological parameters and CGU was evaluated by a Student's unpaired t-test.
The right and left CGU and CBF values for each regions were compared using a Student's paired t test. The activated regions are the ipsilateral Sp5c, Sp5i, Sp5o, Pr5, and contralateral VPM and FrPaSS cortex because the fibers cross before the VPM. The analysis of the CBF/CGU relationship was performed using a two-step procedure. First, the mean CBF and CGU values for each regions examined were plotted and a correlation coefficient was calculated from a linear regression analysis. Such analysis was made both in control conditions and under 7-NI (nonstimulated sites of the trigeminal pathway and averaged values (left and right) for the other brain regions). The slopes were compared using a Student's t-test (Zar, 1984). Second, a repeated-measures analysis of variance was performed on the log transform of the data sets as previously described by McCulloch et al. (1982).
RESULTS
Systemic effects of 7-nitroindazole
Intraperitonally administered 7-NI (25 mg/kg) did not significantly change either of the physiological variables measured, rectal temperature, arterial blood gas tensions and pH, glycemia, and hematocrit level (Table 1) as we previously observed (Cholet et al., 1996). Figure 1 also shows that 7-NI had no effect on MABP nor heart rate throughout the 30 minute-experiment, compared with vehicle-injected rats (Fig. 1, Table 1).
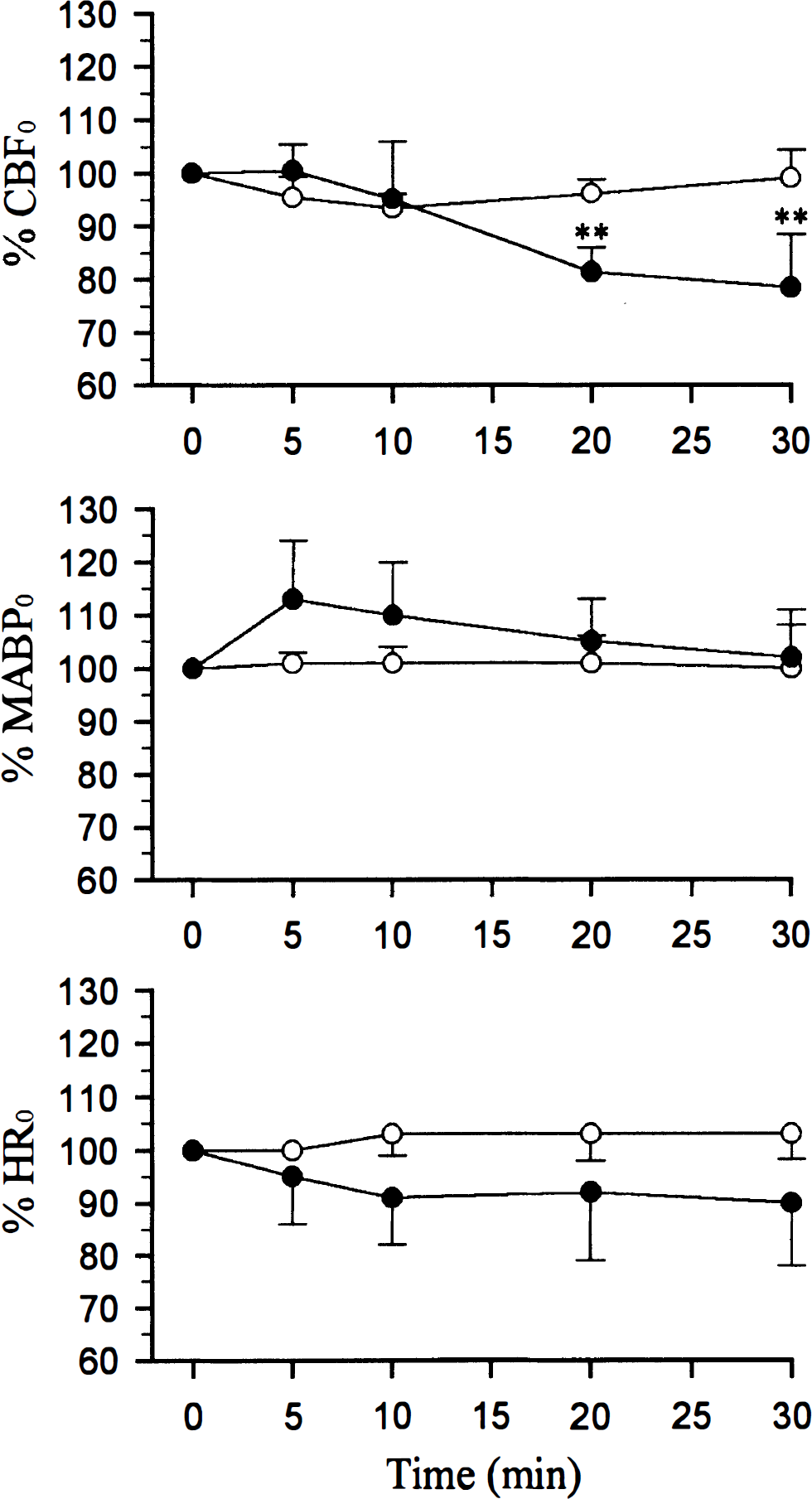
Time course of cerebral blood flow (CBF), mean arterial blood pressure (MABP), and heart rate (HR) changes after 25mg/kg ip injection of 7-nitroindazole (7-NI) (•) or vehicle (○). Values are expressed as mean percent changes ± SD (n = 5 in both groups). ** P < 0.01 significantly different from baseline (one-way analysis of variance followed by Dunett's test). HR0, MABP0, CBF0 are preinjection values. Control group: MABP0 = 117 ± 20 mm Hg and HR0 = 412 ± 33 bpm. 7-Nitroindazole (25 mg/kg) group: MABP0 = 119 ± 7 mm Hg and HR0 = 408 ± 30 bpm. Blood flow changes were calculated in percentage change versus the mean CBF value during the preceding 1-minute baseline condition (CBF0).
Effects of 7-nitroindazole on physiological variables
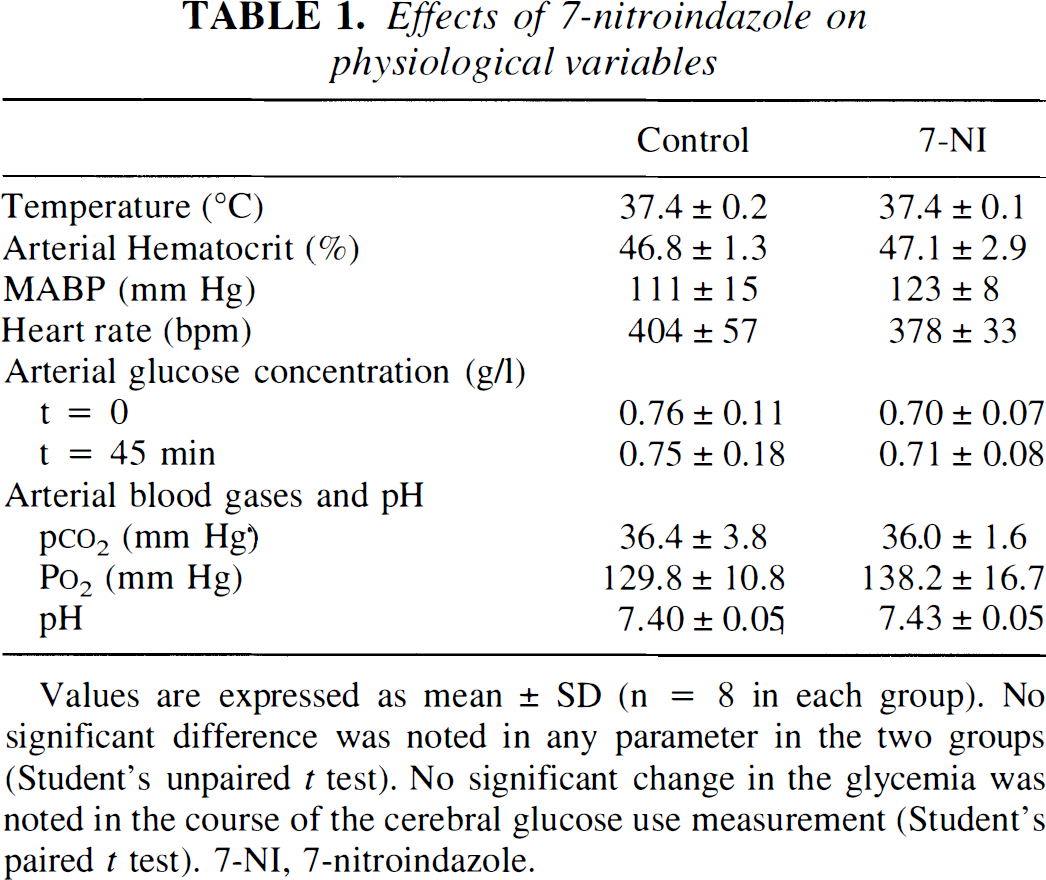
Values are expressed as mean ± SD (n = 8 in each group). No significant difference was noted in any parameter in the two groups (Student's unpaired t test). No significant change in the glycemia was noted in the course of the cerebral glucose use measurement (Student's paired t test). 7-NI, 7-nitroindazole.
Effect of 7-nitroindazole on resting local cerebral blood flow and cerebral glucose use
Figure 1 represents the time-course of the cortical blood flow changes measured by laser-Doppler flowmetry after treatment with 7-NI. Blood flow decreased progressively and significantly with maximal effect reached at 30 minutes (−22%) compared with vehicle-treated rats.
When measured using the autoradiographic technique, CBF significantly decreased in most of the brain structures examined (from −12 to −35%) 30 minutes after 7-NI injection (Tables 2, 3). In contrast, no significant effect of 7-NI on the CGU was noted either in the nonactivated sides of the relays of the trigeminal pathway (Table 4) or in the 13 regions examined outside of the trigeminal pathway (Table 5).
Effects of unilateral whiskers stimulation on CBF (mL 100g−1 min−1) in control conditions and after 7-nitroindazole, in the relays of the trigeminal pathway
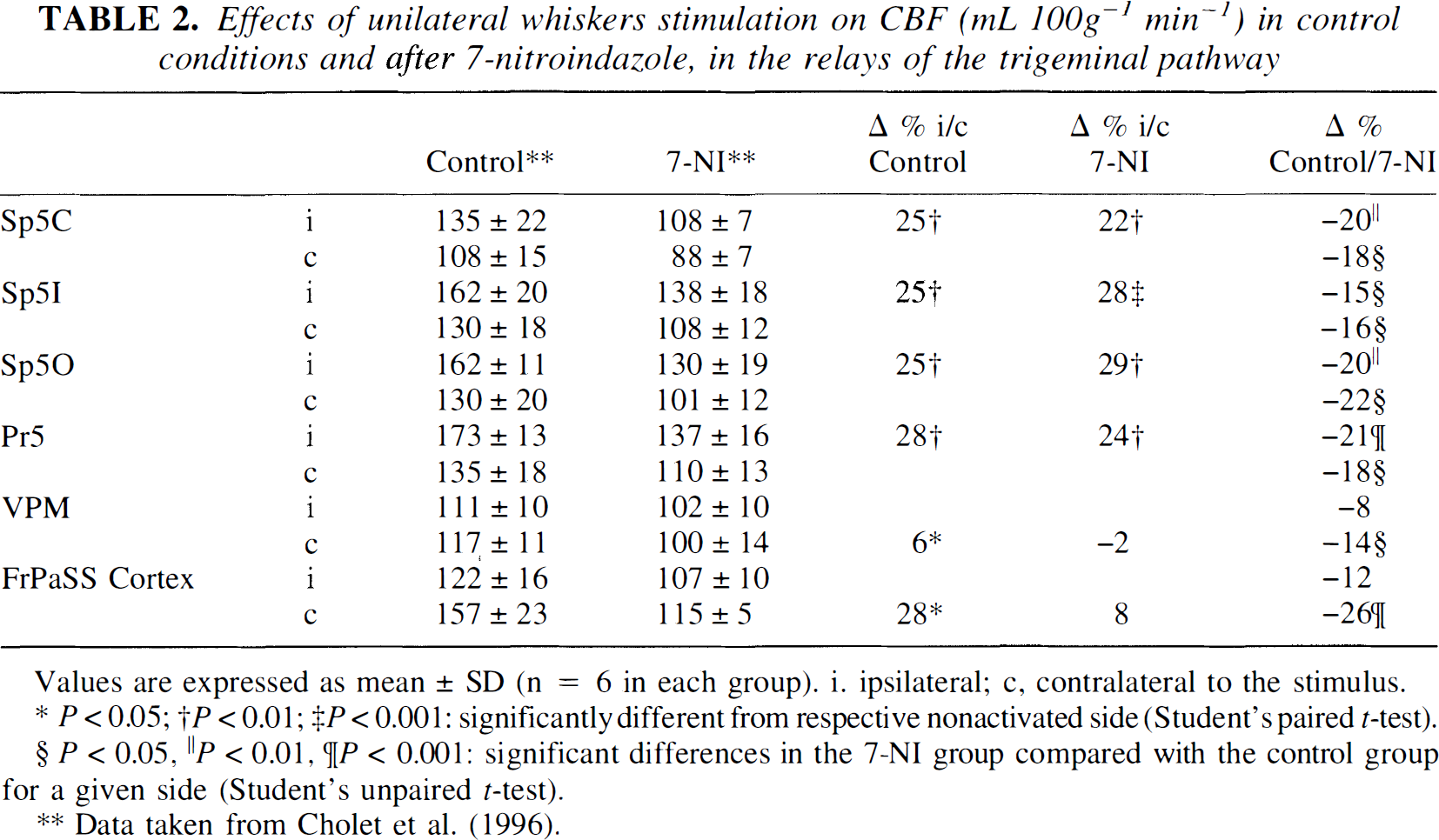
Values are expressed as mean ± SD (n = 6 in each group). i. ipsilateral; c, contralateral to the stimulus.
P < 0.05
P < 0.01
P < 0.001: significantly different from respective nonactivated side (Student's paired t-test).
P < 0.05
P < 0.01
P < 0.001: significant differences in the 7-NI group compared with the control group for a given side (Student's unpaired t-test).
Data taken from Cholet et al. (1996).
Effects of unilateral whiskers stimulation on CBF (ml 100g−1 min−1) in control conditions and after 7-nitroindazole, in the structures not involved in the trigeminal pathway
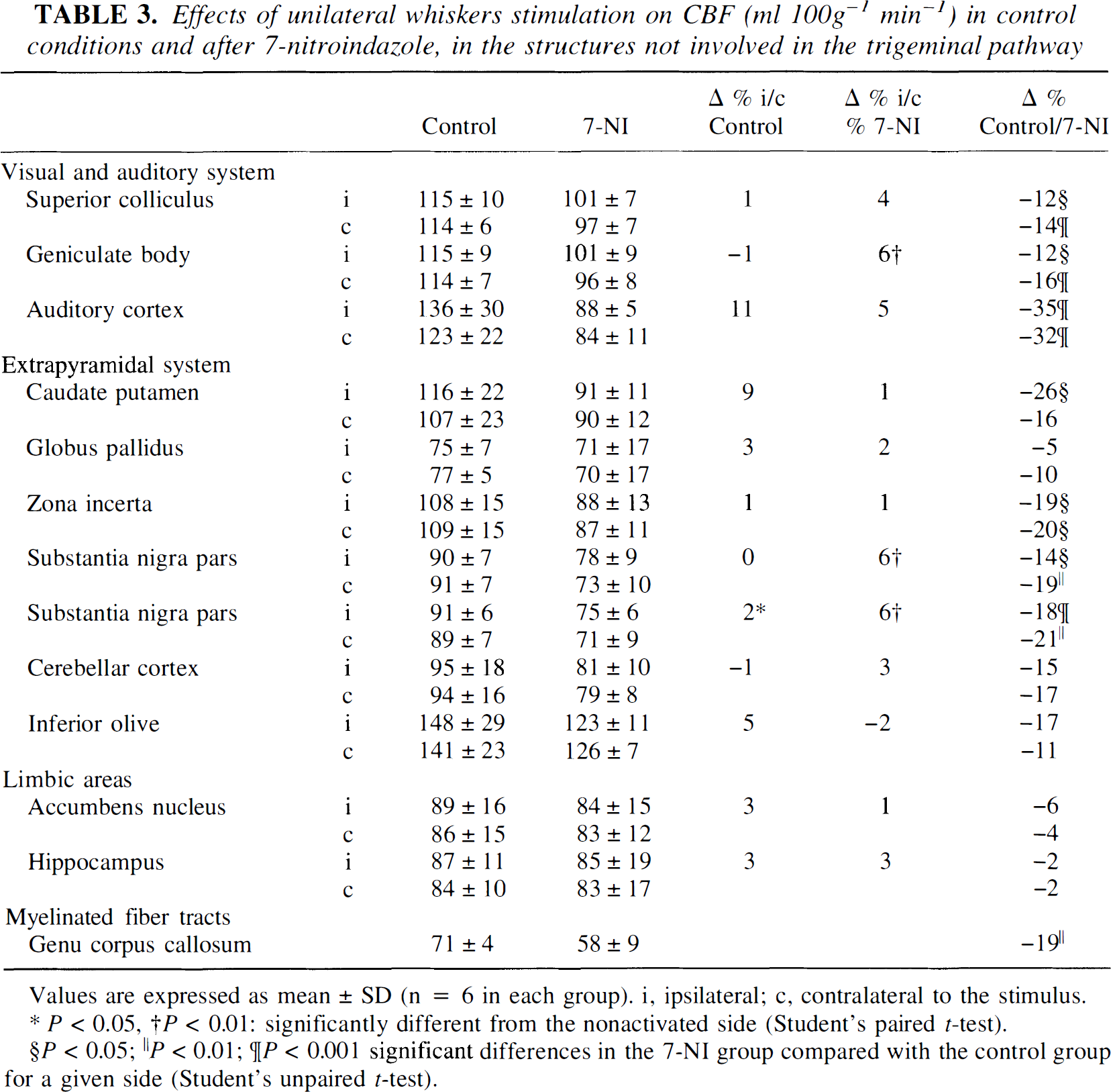
Values are expressed as mean ± SD (n = 6 in each group). i, ipsilateral; c, contralateral to the stimulus.
P < 0.05
P < 0.01: significantly different from the nonactivated side (Student's paired t-test).
P < 0.05
P < 0.01
P < 0.001 significant differences in the 7-NI group compared with the control group for a given side (Student's unpaired t-test).
Effects of unilateral whiskers stimulation on CGU (μmol 100g−1 min−1) in control conditions and after 7-nitroindazole, in the relays of the trigeminal pathway
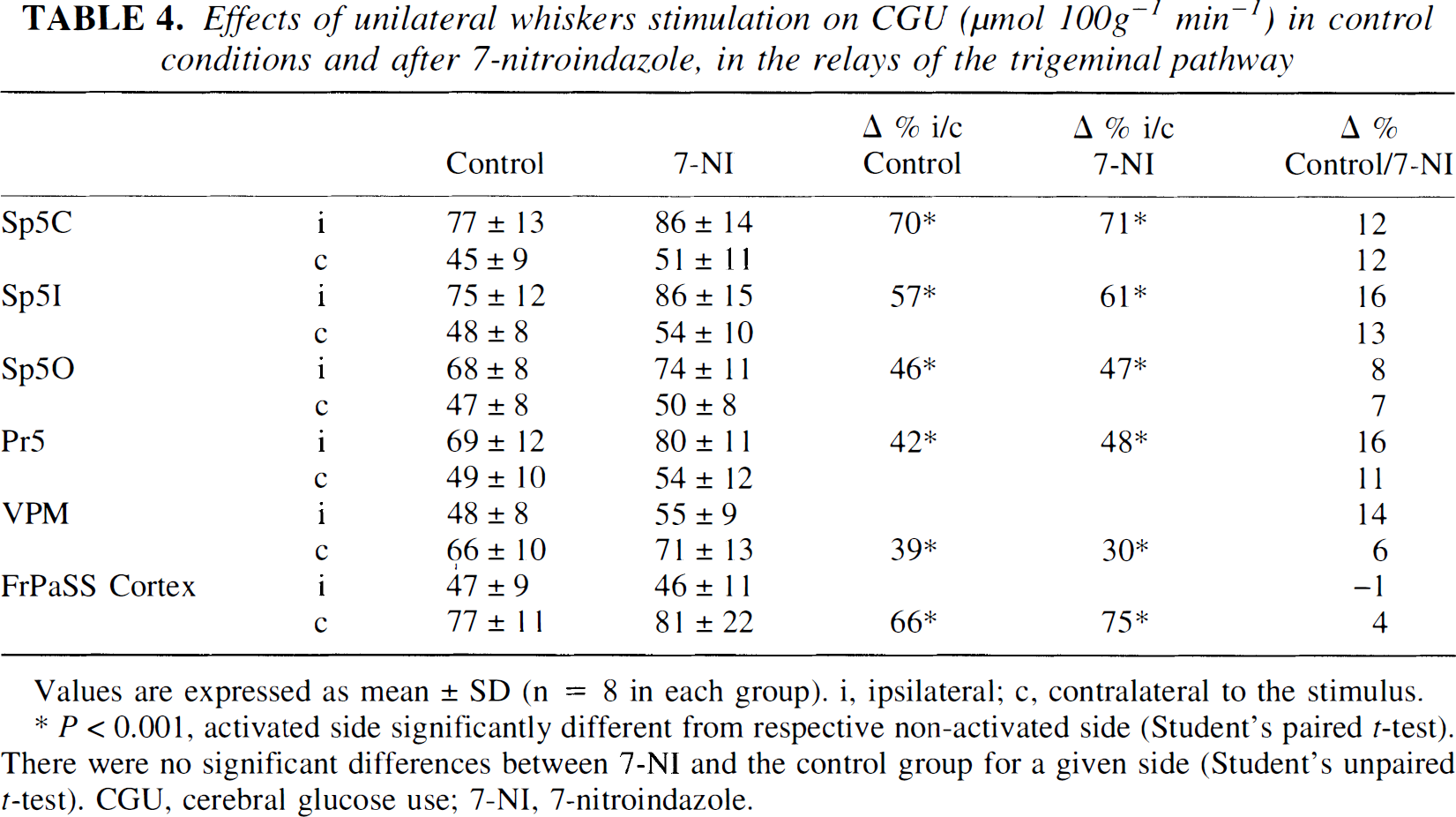
Values are expressed as mean ± SD (n = 8 in each group). i, ipsilateral; c, contralateral to the stimulus.
P < 0.001, activated side significantly different from respective non-activated side (Student's paired t-test). There were no significant differences between 7-NI and the control group for a given side (Student's unpaired t-test). CGU, cerebral glucose use; 7-NI, 7-nitroindazole.
Effects of unilateral whiskers stimulation on CGU (μmol 100g−1 min−1) in control conditions and after 7-nitroindazole, in structures outside of the trigeminal pathway
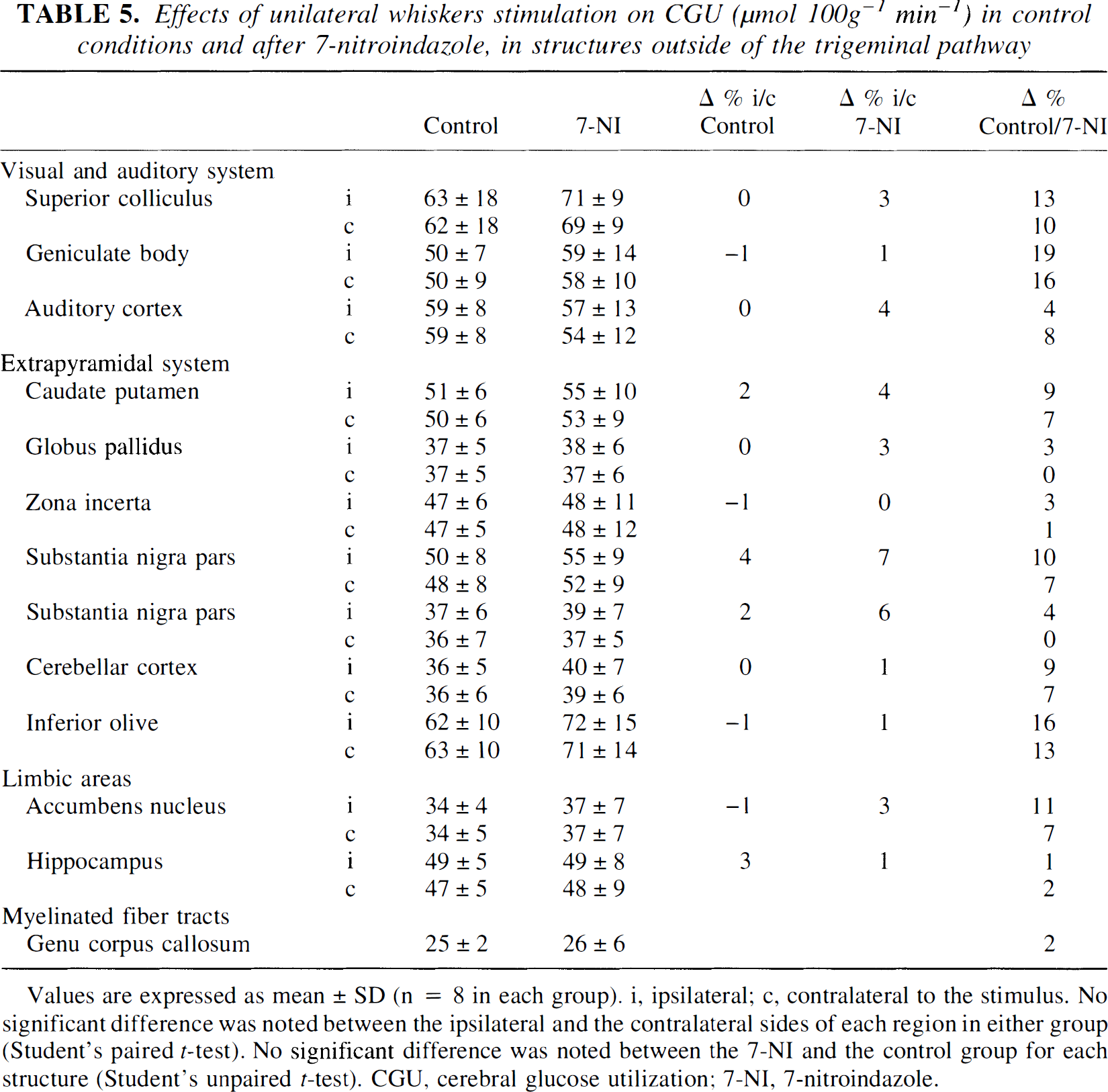
Values are expressed as mean ± SD (n = 8 in each group). i, ipsilateral; c, contralateral to the stimulus. No significant difference was noted between the ipsilateral and the contralateral sides of each region in either group (Student's paired t-test). No significant difference was noted between the 7-NI and the control group for each structure (Student's unpaired t-test). CGU, cerebral glucose utilization; 7-NI, 7-nitroindazole.
Because 7-NI decreased CBF without affecting CGU, there was a resetting of the flow-metabolism relationship in 7-NI-treated rats (Fig. 2). Significant correlation between CGU and CBF in control group (r = 0.79) was maintained in treated group (r = 0.67), but the slope of the correlation line was significantly decreased under 7-NI (1.8 versus 1.0, P < 0.05). Repeated-measures analysis of variance confirmed that the resetting of the flow-metabolism relationship was significant (P < 0.05).
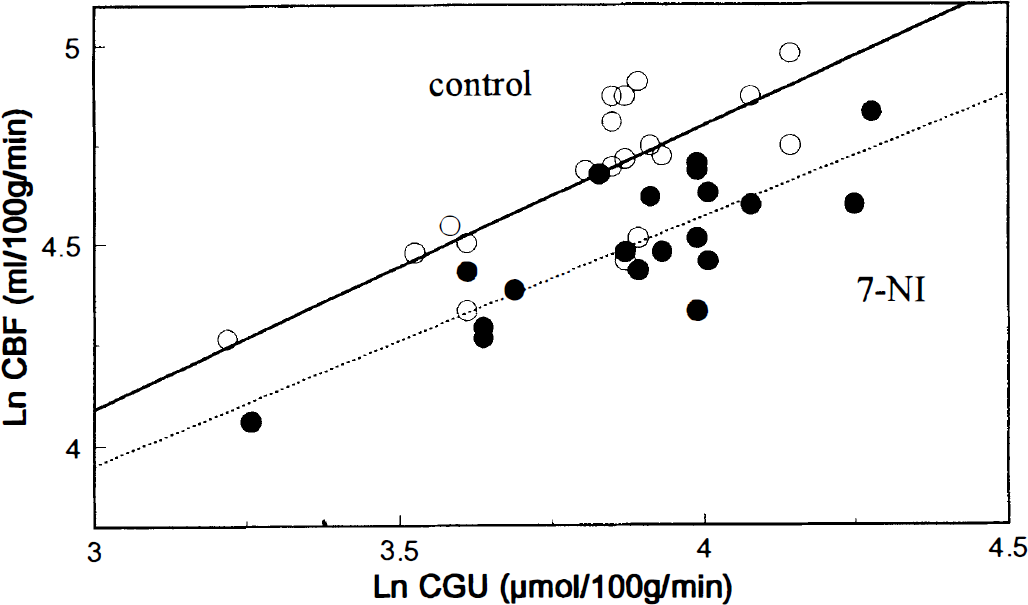
Relationship between resting CBF and cerebral glucose use (CGU) in control (○) and 7-NI-treated (•) animals. Each point represents mean CGU and mean CBF values (expressed in natural logarithms) obtained for one brain structure (nonstimulated sides of the trigeminal pathway and averaged values for the 13 other brain regions). Repeated-measures analysis of variance indicated that the resetting of the flow-metabolism relationship was significant (P < 0.05).
Effects of 7-nitroindazole on stimulus-induced changes in local cerebral blood flow and cerebral glucose use
In control conditions, unilateral C whisker stimulation resulted in distinct CBF increases from 25% to 28% in the ipsilateral trigeminal nuclei and of 6 and 29% in the contralateral VPM and the contralateral FrPaSS cortex, respectively (Table 2). Whisker stimulation induced corresponding localized CGU increases from 42% to 70% in the ipsilateral trigeminal nuclei (Sp5c, Sp5i, Sp5o, and Pr5) and of 39% and 66% in the contralateral VPM and FrPaSS cortex, respectively (Table 4, Fig. 3). Cerebral blood flow increases under 7-NI were similar to those in control rats in the ipsilateral trigeminal nuclei (from 22% to 29%) but no significant vasodilation was noted in the contralateral VPM and FrPaSS cortex (Table 2, Fig. 3). Compared with control animals, similar increases of CGU were observed in 7-NI-treated animals during stimulation with increases from 47% to 71% in the ipsilateral first-order relay stations (Sp5c, Sp5i, Sp5o, and Pr5) and of 30% and 75% in the contralateral second-order relay stations (VPM and FrPaSS cortex) (Table 4 and Fig. 3).
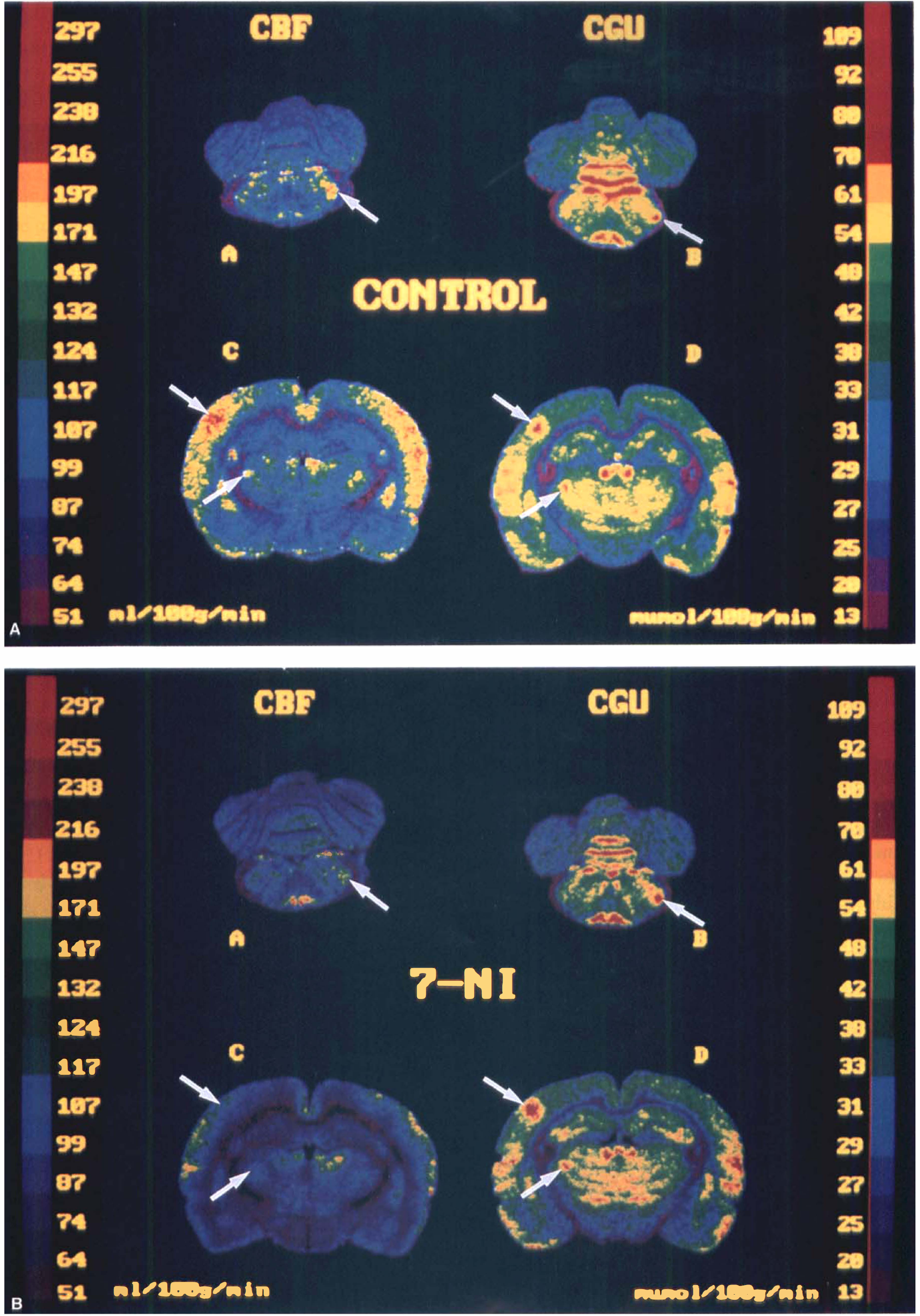
Representative color-coded autoradiograms of CBF (left), and CGU (right) during right C whiskers stimulation in control condition
Effect of 7-nitroindazole on nitric oxide synthase activity
In vehicle treated-rats, NOS activity was 31.7 ± 15.4, 5.4 ± 2.6, and 2.8 ± 0.9 pmol activity·mg tissue−1·min−1 in the cerebellum, pons-medulla, and somatosensory cortex, respectively. Administration of 7-NI significantly reduced NOS activity by 74%, 89%, and 79% in the same structures, respectively (Fig 4).
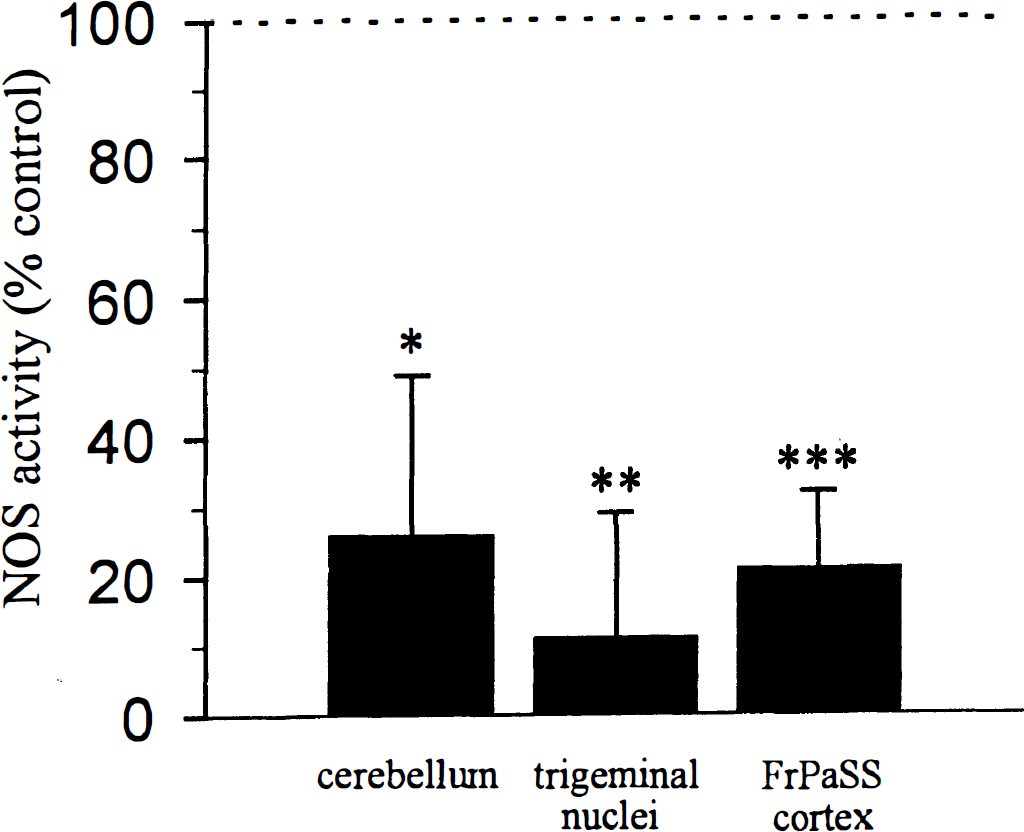
Effect of 7-NI (25 mg/kg) on nitric oxide synthase activity in cerebellum, pons/medulla including the trigeminal nuclei and somatosensory cortex. * P < 0.05, ** P < 0.01, *** P < 0.001 significantly different from respective vehicle-injected rats (n = 5 in both group).
DISCUSSION
This autoradiographic study analyzed the combined circulatory and metabolic consequences of inhibiting the nNOS during a functional activation in the α-chloralose anesthetized rat. We have provided evidence that (1) NO-releasing neurons play a significant role in maintaining a resting cerebrovascular tone in the whole brain; (2) in the somatosensory cortex, inhibition of nNOS abolishes the vascular response to whisker stimulation without affecting the metabolic activation; and (3) nNOS does not mediate the vascular and metabolic response to somatosensory activation in the first-order relay stations of the trigeminal pathway. These observations suggest that the involvement of neurally derived NO in coupling flow to somatosensory activation is region dependent and that CBF and CGU may vary independently under nNOS inhibition.
Differentiated effect of 7-nitroindazole on resting cerebral glucose use and cerebral blood flow
A number of recent studies have highlighted the critical importance of the dose, the route of administration, and the time elapsed after injection of a potential nNOS inhibitor to achieve an efficient blockade of the enzyme responsible for in vivo NO production (Yoshida et al., 1994; Mackenzie et al., 1994; Moore et al., 1993a).
In the present study, 7-NI was administered intraperitonally at 25 mg/kg and CBF and CGU were measured 30 minutes thereafter. Using these parameters, we indeed obtained a marked inhibition of nNOS (74% to 89%). We can thus reasonably assume that our data represent the optimal effect of 7-NI on cerebrovascular reactivity as also evidenced by our laser-Doppler flowmetry results. With regard to the specificity of 7-NI versus the neuronal form of NOS, we observed that the administration of 7-NI was devoid of any marked cardiovascular effects. This is consistent with previous studies using 7-NI (from 10 to 100 mg/kg) in several species either anesthetized or conscious (Yoshida et al., 1994; Mackenzie et al., 1994; Moore et al., 1993a; Kovach et al, 1994; Wang et al., 1995; Zhang et al., 1995; Iadecola et al., 1996a). Also, the lack of influence of 7-NI on the vasorelaxant action of acetylcholine (Yoshida et al., 1994; Zhang et al., 1995; Iadecola et al., 1996a) confirms that 7-NI is a selective inhibitor of neuronal NOS activity.
Alpha-chloralose is a suitable anesthetic for the study of cerebrovascular and metabolic responses to neuronal activation in experimental conditions that preclude the use of conscious animals (Bonvento et al., 1994a). It produces only minor and relatively homogeneous changes in CBF (Bonvento et al., 1994a; Bonvento et al., 1989; Lacombe et al., 1992) and glucose use (Dudley et al., 1982). Alpha-chloralose maintains the coupling between neuronal function and metabolism (Ueki et al., 1988, 1992). This is a unique feature because such coupling is not maintained under halothane, pentobarbital, and nitrous oxide anesthesia (Ueki et al., 1992). We also provided evidence that the magnitudes of the CGU responses to somatosensory stimulation (+39 to 70%) are of the same order as that reported in conscious rats (Dietrich et al., 1986). Alpha-chloralose at doses of up to 80 mg/kg has been shown to maintain the coupling between neuronal function and CBF (Ueki et al., 1988; Lindauer et al., 1993). Finally, the similarity of the slopes calculated from the linear regression of the CBF/CGU values in conscious (m = 2.0, Kelly et al., 1995) and chloralose-anesthetized rats (m = 1.8, present result) suggests that this anesthetic maintains a physiological CBF/CGU relationship.
Using laser-Doppler flowmetry and autoradiography, we have found that 7-NI decreases basal CBF, an observation supported by several other reports including ours, in which specific or nonspecific neuronal NOS inhibitors were used in conscious or anesthetized animals (Bonvento et al., 1994b; Montécot et al., 1997; Iadecola et al., 1994a). However, we have observed that 7-NI does not modify resting CGU in any brain regions examined as reported by Kelly et al. in conscious rats (1995). Consistently, other nonspecific inhibitors of NO synthase did not alter resting CGU either (Wang et al., 1993; Iadecola and Xu, 1994b; Takahashi et al., 1995). In agreement with this is the lack of influence of NOS inhibition on global cerebral oxygen consumption (Wang et al., 1993; Iadecola et al., 1994a). This is surprising in view of the well recognized role of NO as a major neuronal messenger involved in many physiological regulations including cerebrovascular reactivity (Takahashi et al., 1995; Iadecola et al., 1994a). These results collectively support the idea that 7-NI induces a global and appreciable decrease of resting CBF without altering brain metabolism.
The resetting of the flow-metabolism relationship observed after 7-NI treatment indicates that neurally-derived NO directly participate in the maintenance of a resting cerebrovascular tone, as observed by Kelly et al. (1995).
Paradoxical effects of 7-nitroindazole on stimulus-induced changes in cerebral blood flow and cerebral glucose use
We previously reported that with the use of 7-NI, the cerebrovascular reactivity elicited by whisker stimulation was abolished in the contralateral second-order relay stations, the VPM, and the FrPaSS cortex, but was preserved in the ipsilateral trigeminal nuclei (Cholet et al., 1996). This result contrasts with the previously reported unchanged percentage increases in CBF after the administration of L-NAME (a nonspecific NOS inhibitor) in conscious rats (Adachi et al., 1994). The explanation for such a discrepancy is unclear, but it seems unlikely that anesthesia is responsible for the differentiated cerebrovascular effects of NOS inhibition. Indeed, the involvement of NOS in either the maintenance of a resting cerebrovascular tone (see above) or in the cerebrovascular response to hypercapnia was shown to be identical in conscious (Bonvento et al., 1994b) and anesthetized animals (Iadecola et al., 1994).
We now provide evidence that this differential circulatory influence of nNOS inhibition is not a consequence of a differential metabolic effect because 7-NI did not modify the metabolic response to whisker stimulation in any of the six trigeminal relays studied. In agreement with this result, is the fact that NOS inhibition does not affect somatosensory-evoked potentials elicited by vibrissae activation in the chloralose anesthetized rat (Lindauer et al., 1996). Consequently, the close relationship that exists between CBF and CGU which is maintained during whisker stimulation in control rats, is disrupted in the second-order relay stations of the trigeminal pathway (VPM and FrPaSS cortex) after inhibition of the neuronal NO synthase activity, while it is preserved in the trigeminal nuclei. This indicates that the involvement of nNOS in the coupling of flow to functional activation is region dependent and is mainly effective in the cerebral cortex. It is noteworthy that nNOS has also been shown to mediate the vasodilation from cerebellar parallel fibers stimulation (Iadecola et al., 1996) suggesting that not only the cortical NO neurons participate in such coupling. Our results also suggest that in other brain structures, other mediators must be required to couple neuronal function to vasodilation, even within the same activated pathway.
The main finding of the current study is that with the use of 7-NI, the local cortical metabolic response is completely preserved despite an abolished flow response. This observation challenges the seminal but still prevailing hypothesis of Roy and Sherrington (1890), that the microvascular adjustments in response to changes in neuronal activity are designed to match the metabolic needs. Therefore, our results indicate that either these two variables are not inevitably linked, or that the CBF changes can serve purposes other than adjusting energy supply to increased neuronal metabolic demands. This latter hypothesis has long been known as ‘luxury perfusion’ (Lassen, 1966). This term implies that blood flow increases in large excess in face of the limited needs in supplementary substrates (glucose and oxygen) during a functional activation. According to Ueki et al. (1988), the brain has the capacity to increase its fractional arterial extraction of glucose and oxygen to some extent without requiring an increased blood supply. Such a hypothesis was recently substantiated by a study that shows a constant CBF response to a physiological brain activation in humans with progressive arterial hypoglycemia leading to a progressive reduction in glucose delivery to the activated area (Powers et al., 1996).
What are the putative sources of glucose in such conditions that preclude any blood flow changes? One hypothesis would be that glucose transport through endothelial cells can be modulated during functional activation. Indeed Farrel et al. (1991) observed that 40% of the GLUT1 isoform (55 kd), the specific endothelial glucose transporter, are located within the cytoplasmic compartment. The possible translocation of GLUT1 from the cytoplasm to the plasma membrane may provide one mechanism for rapid up-regulation of the endothelial glucose transporter activity. Another hypothesis is that glucose is derived from another source such as glycogen in astrocytes (Magistretti et al., 1993).
In the current report, we did not investigate the neuronal mechanisms of the metabolic and flow responses induced by somatosensory stimulation. However, recently accumulated evidence strongly supports the implication of glutamate release as a major step of the signaling process coupling neuronal activity to metabolism and CBF. This is of major interest because glutamate can be considered as one of the major neurotransmitters involved in the processing of somatosensory information (Zilles and Wree, 1995; Salt, 1986; Klockgether, 1987).
Indeed, it has been shown that glutamate exerts a metabolic function in astrocytes, by activating glycolysis and inducing the lactate synthesis, providing the activated neurons with energy-producing substrates (Pellerin and Magistretti, 1994).
Considering the vascular event, recent evidence suggests that activation of glutamate receptors participates in the mechanisms of functional hyperemia during cerebellar parallel fibers activation induced increased synaptic activity (Akgören, 1994; Iadecola et al., 1996b). One hypothesis of the mechanism by which such glutamate receptor activation leads to flow increase relies on an enhanced NO production via an increased Ca++ entry (Faraci and Brian, 1994). In view of all these observations, glutamate could be a suitable signal to increase both CGU and CBF during a functional activation via either its glycolysis-induced uptake of glucose in astrocytes and the stimulation of NOS activity.
In conclusion, our results provide evidence for a major and direct involvement of nNOS in the cortical flow coupling to somatosensory stimulation. In addition, the striking flow/metabolism uncoupling observed at the cortical level suggests that the vascular and metabolic responses, despite the fact that they probably share a common triggering signal (e.g., glutamate) are most likely controlled by distinct signaling processes.
