Abstract
Studies on the kinetics of 99mTc-D,L-hexamethylpropylene amine oxime (99mTc-HMPAO) in adults have shown that it is not an ideal tracer of CBF because it underestimates high-flow areas. Knowledge of the kinetics of the tracer is important in evaluating the studies. The kinetics of 99mTc-HMPAO in infants may be different from that in adults, therefore, we examined the cerebral uptake and retention of 99mTc-HMPAO in neonates and estimated the degree of brain-to-blood back diffusion by comparing corresponding 133Xe flow images and 99mTc-HMPAO distribution images. In addition, we measured the urinary excretion of 99mTc-HMPAO. Regional CBF was measured using a mobile brain dedicated, fast-rotating, four-head multidetector system specially designed for neonatal studies. Tracers were 99mTc-HMPAO (4 MBq/kg) and 133Xe (500 MBq/kg). Cerebral uptake and leak-out of 99mTc-HMPAO were measured by a single scintillation crystal placed over the frontoparietal part of the infant's head. The cerebral retention of 99mTc-HMPAO was analyzed in 50 infants. The mean gestational age and birth weight (95% confidence interval) were 34.4 weeks (32.2–35.7) and 2,326 g (1,954–2,995), respectively. The cerebral uptake of 99mTc-HMPAO was examined in 16 of the 50 infants, and activity during 24 h was monitored in five. In 11 infants, corresponding 133Xe studies were performed. Urinary excretion was studied in 12 infants. The maximal activity in the brain was reached 90s after i.v. injection and was 104% (98–111) of the stable level, which was reached approximately 3 min after the injection. The decay corrected leakout of the tracer during the following 24 h was 1.0% (0.4–1.5) per hour. The cerebral retention was calculated at 6.8% (6.1–7.6), highest in the group of ictal studies and lowest in premature infants with intracranial hemorrhage. The mean value of the fixation/clearance ratio α was estimated at 3.4 (2.8–4.4). The mean urinary excretion over 24 h was 19.5% (11.4–27.7) and was significantly related to renal function as indicated by serum urea (p = 0.02 r2 = 0.55). A four-compartment model describing the kinetics of 99mTc-HMPAO is shown to be valid in neonates. The cerebral retention of the tracer is higher in neonates because of higher extraction and lower initial back diffusion from brain to blood. In linearizing 99mTc-HMPAO distribution images, a smaller correction is necessary, and we propose a value of the correction factor of 3.4. In this way, 99mTc-HMPAO is a more reliable tracer of the distribution of CBF in neonates compared with adults. The urinary excretion is significantly reduced compared with adults, and the radiation dose to the bladder wall is reduced. The effective dose is 0.3 mSv/MBq/kg.
Since 1989 there have been reports on examinations of the distribution of regional CBF in infants using SPECT 99mTc-D-L-hexamethylpropylene amine oxime (99mTc-HMPAO) (Fockele et al., 1989). Studies on the kinetics of 99mTc-HMPAO in adults have shown that it is not an ideal tracer of CBF because it underestimates high-flow as a result of a back diffusion of the tracer from brain to blood (Andersen, 1989). Knowledge of the kinetics of the tracer is important in evaluation of these studies (Lassen et al., 1988). The kinetics of 99mTc-HMPAO may be different in infants because of the low CBF in this age group (Greisen, 1986). Hence, we examined the cerebral uptake and retention of 99mTc-HMPAO in neonates and estimated the degree of brain-to-blood back diffusion by comparison of 99mTc-HMPAO distribution images and 133Xe flow images.
In adults, 35% of the injected tracer is excreted into the urine, causing a high radiation dose to the bladder wall. As renal function is reduced in neonates, the radiation dose to the bladder wall may be lower because of lower urinary excretion. Therefore, we also measured the urinary excretion of 99mTc-HMPAO in neonates.
THEORY
HMPAO is a lipophilic compound that is rapidly converted into a hydrophilic form, presumably as a result of an action with gluthathione (Neirinx et al., 1988). The kinetics of HMPAO can be described by a four-compartment model (Andersen, 1989) (Fig. 1). The four compartments are lipophilic and hydrophilic tracer in the blood and in the brain, respectively, and the transfer between the compartments follows first-order kinetics with the rate constants k1ṫk6. The lipophilic tracer is freely diffusible between the compartments, whereas the conversion from lipophilic to hydrophilic form is assumed to be irreversible. Therefore, k4 and k6 are set to zero. Regarding a bolus injection, 1 U of lipophilic tracer and 1 U of brain tissue, a fraction E of the lipophilic tracer is extracted into the brain tissue as the bolus traverses the capillary. Washout of the lipophilic tracer is described by k2 = k1/λ, where λ is the partition coefficient; however, the amount of back diffusion can be described by the ratio α = k3/k2, where k3 is the conversion rate of lipophilic to hydrophilic tracer in the brain. A high value of α denotes a low degree of back diffusion and vice versa. Denoting brain mass and cardiac output mbrain and CO, respectively, the fraction of the injected tracer retained in the brain R may be calculated as
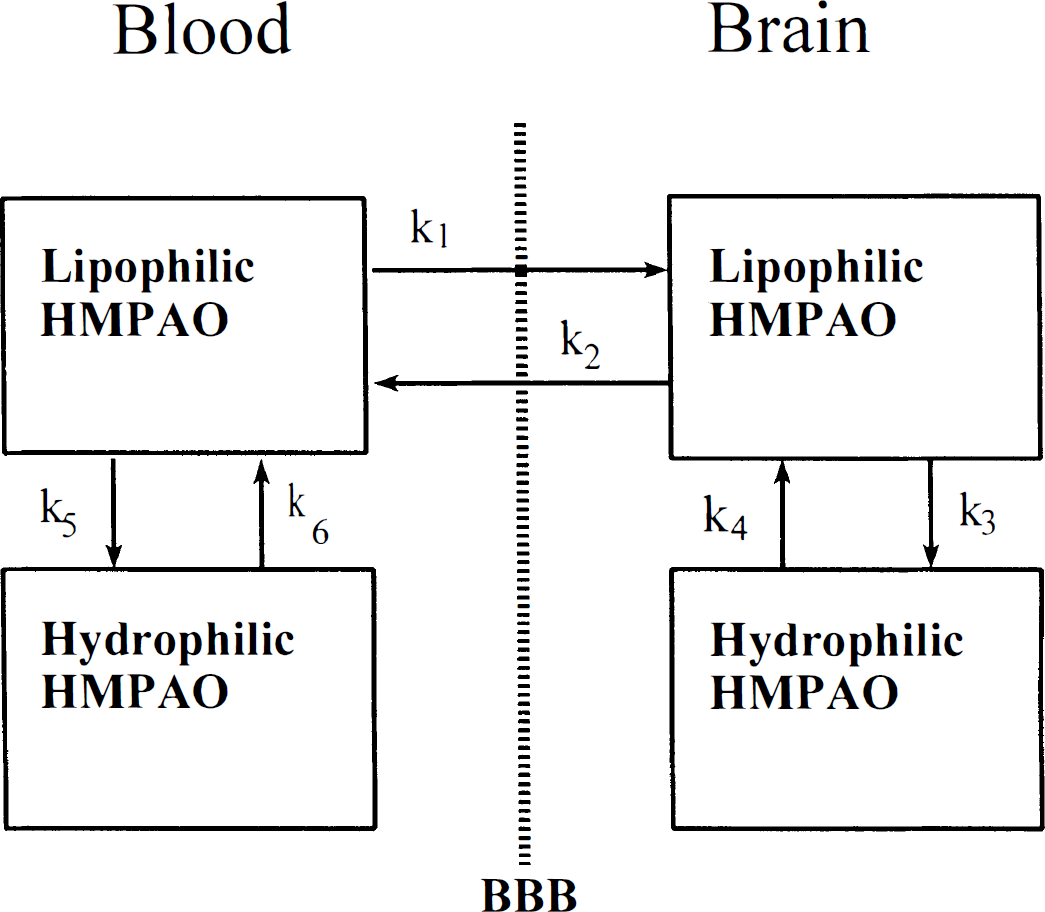
A four-compartment model describing the kinetics of 99mTc-HMPAO in the brain: k1 to k6 denotes constants of first-order kinetics. Lipophilic HMPAO is freely diffusible from blood to brain; hydrophilic HMPAO is nondiffusible. Since the conversion from the lipophilic to the hydrophilic form is almost irreversible k4 = k6 = 0. (BBB: blood-brain-barrier).
showing that the retention depends on the ratio CBF/CO, E, and α. Eqs. 1 and 2 yield the fraction of the injected dose retained in the brain, whereas the corresponding equations by Lassen et al. (1988) yield the retention of the tracer entering the cerebral circulation.
PATIENTS
The cerebral retention of 99mTc-HMPAO was analyzed in 50 infants of different ages and with different diagnoses (Table 1). The time course of the cerebral uptake of 99mTc-HMPAO was examined in 16 of these infants, and activity during 24 h was monitored in five of those. In 11 infants, corresponding 133Xe studies were performed (Table 2).
Characteristics of infants entering the study of cerebral retention of 99mTc-HMPAO

99mTc-HMPAO, 99mTc-D-L-hexamethylpropylene amine oxime; pCO2, partial pressure of carbon dioxide; CPAP, continuous positive airway pressure.
Statistically significant from the total group (analysis of variance with Tukey's post hoc correction for multiple comparisons).
Data presented as median (range).
Characteristics of 11 infants in which both a 99mTc-HMPAO and a 133Xe study were performed

99mTc-HMPAO, 99mTc-D-L-hexamethylpropylene amine oxime; CPAP, continuous positive airway pressure.
Urinary excretion was studied in 12 infants of different ages and diagnoses (Table 3). Serum values of urea determinated within ±12 h from the 99mTc-HMPAO study were available in the clinical records in nine of the 12 infants and used as a measure of renal function.
Characteristics of 12 infants entering the study of 24-h urinary excretion of 99mTc-HMPAO
99mTc-HMPAO, 99mTc-D-L-hexamethylpropylene amine oxime.
METHODS
Tracers
99mTc-labelled HMPAO was prepared by adding 80 MBq of freshly eluated 99mTc in 10 ml of isotonic saline into one vial (500 μg) of HMPAO (Ceretec, Amersham International). The tracer was injected intravenously in a dose of 4 MBq/kg body weight within 2 h after preparation. The radiation dose to the infant was calculated at 1.2 mSv/study (effective dose) (Vestergren et al., 1992).
For studies with 133Xe, we used a dose of 500 MBq/kg body weight in 1 ml of isotonic saline. These studies resulted in a radiation dose to the infant of 2.5 mSv/study (effective dose) (Greisen et al., 1984).
Procedure
For 99mTc-HMPAO studies, the infant was placed in the scanner within 1 to 2 h after injection of the tracer. The planes were parallel to the orbitomeatal plane and the position was secured by a light-positioning system. The acquisition time was 30 to 45 min per two slices. In total, two to four acquisitions, that is, four to eight slices, were obtained by moving the infant 10 to 20 mm in the longitudinal plane between each acquisition to cover the whole brain.
For 133Xe studies, the infant was placed in the scanner before the injection of tracer. After measurement of background activity, the uptake and washout of the tracer was measured in 15 acquisitions of 60 s each. The lung curve was obtained by a narrowly collimated scintilation crystal placed over the apical part of the right lung.
Reconstruction
The images were reconstructed by a filtered back-projection algorithm using a gaussian filter. In 133Xe studies, flow images were calculated from the first four images using the Kanno–Lassen algorithm (Kanno and Lassen, 1979; Celcis et al., 1981). The lung curve was corrected for the chest-wall component (Greisen and Pryds, 1988).
Data analysis
In SPECT, it has become standard to define the sensitivity (S) as the number of counts per second (cps) per slice obtained from a cylindrical phantom containing 1 MBq/ml of the radionuclide (Larsson, 1980; Kanno et al., 1981). Using this definition, the sensitivity has the unit cps·L·MBq–1·l and depends on the diameter of the phantom. An experiment using homogeneous phantoms with cross sectional areas ranging from 42 cm2 to 83 cm2 established a relation between the cross-sectional area (A) and the sensitivity (S) of
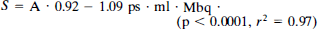
In the patient studies, the cross-sectional area of the brain slice was measured and using Eq. 3 S was estimated for each slice; the retained fraction in the brain of the tracer R was calculated by
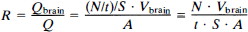
where Q and Qbrain are the injected activity and the activity in the brain, respectively. N/t denotes the count rate. S is given per volume (cps·L·MBq–1), and the multiplication with the brain volume Vbrain assumes that the part of the brain examined is representative for the whole brain. The retention was calculated as the mean retention of all brain slices. The volume of the brain was estimated at 12% of the body weight (Gruenveld and Minh, 1960) on the assumption that the specific gravity of the brain was 1.
Cerebral uptake and leakout of 99mTc-HMPAO were measured by a single scintillation crystal placed over the frontoparietal part of the infant's head. With sampling every 5 s, cerebral activity was measured during the first 15 min after the injection of tracer and hereafter with an interval of 4 to 6 h during the proceeding 24 h. In calculating uptake and leakout, the plateau phase 10 to 15 min after injection of the tracer was chosen as reference level. The brain-to-blood back diffusion was estimated by comparing three corresponding regions of interest (ROI) in high, intermediate, and low-count flow areas in 99mTc-HMPAO distribution and 133Xe flow images of the same infant. Both 99mTc-HMPAO and 133Xe images were normalized using the whole slice as a reference region. Thereby, the relative count rate (C/Cr) obtained from the 99mTc-HMPAO distribution images and the relative flow rate (F/Fr) obtained from the 133Xe flow images were comparable and, according to a four-compartment model (Fig. 1) (Andersen, 1989), the back diffusion described by α = k3/k2 was estimated from the expression
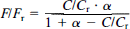
To avoid airway artifacts, ROIs were not placed in the frontal central part of the slice. By changing α in steps of 0.1 from 0 to 10, the value of α yielding the least-squared deviation between the observations and the model was determined. By the same method, the confidence limts were determined as the values of α resulting in a correlation coefficient between (C/Cr) and (F/Fr) not significantly different from 1.
In the first 24 h after injection of 99mTc-HMPAO, all nappies and clothing containing urine were collected. A reference source was made of the same volume of tracer from the original tracer vial as given to the infant. The nappies and clothing, were measured on a conventional gamma camera (General Electric Starcam 3000) together with the reference. After correcting for background activity, the amount excreted into the urine was calculated as a fraction of the injected dose.
RESULTS
The maximal activity in the brain was reached 90 s after i.v. injection of 99mTc-HMPAO and was 104% (98–111) of the stable level. The stable level was reched ∼3 min after the injection (Fig. 2). The decay-corrected leakout of tracer during the following 24 h was 1.0% (0.4–1.5) per hour. The cerebral retention was estimated at 6.8% (6.1–7.6) and was highest in the group of ictal studies and lowest in premature infants with intracranial hemorrhage (Table 1).
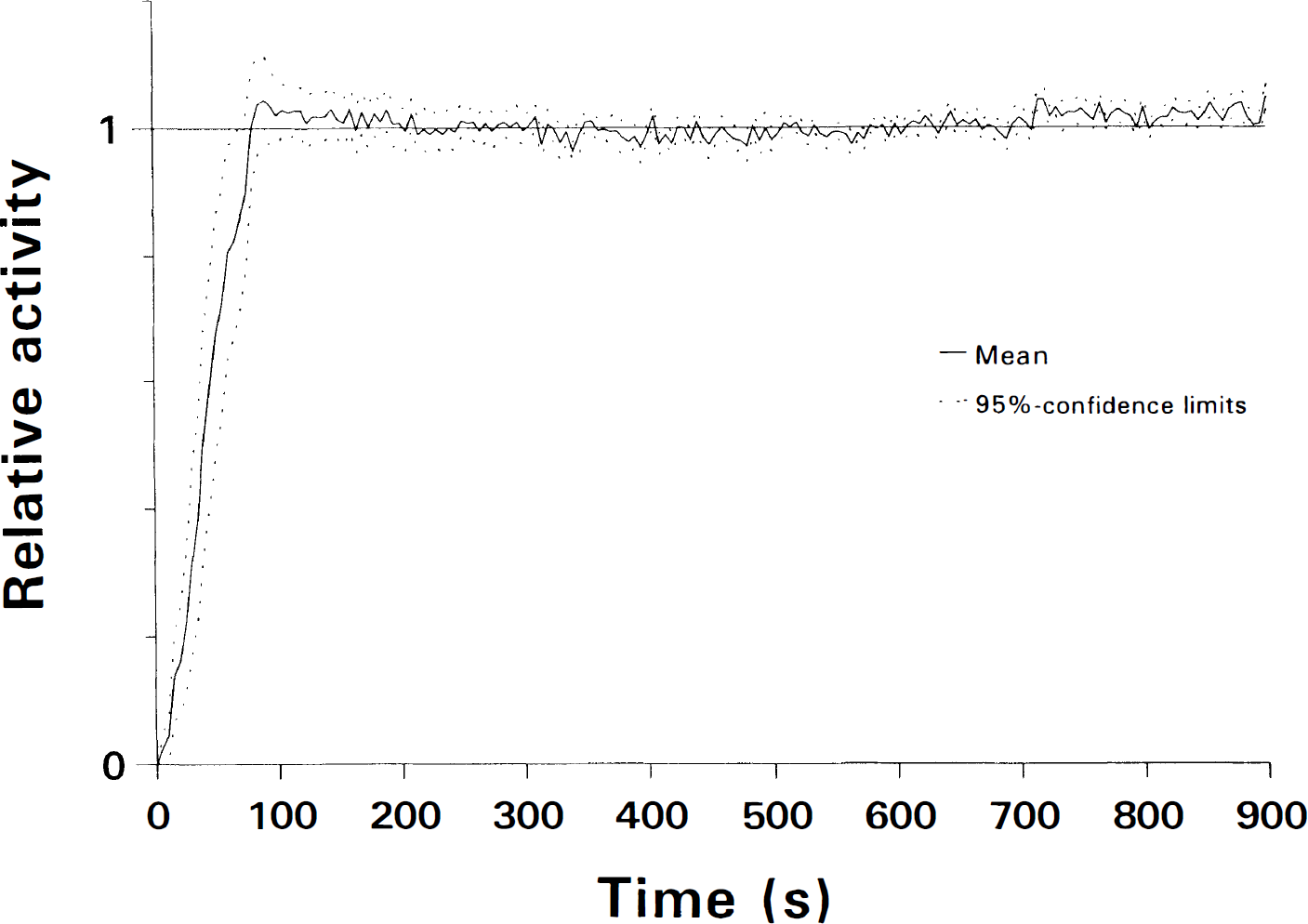
Cerebral uptake of 99mTc-HMPAO in 16 infants measured by a single scintillation crystal placed over placed over the frontoparietal part of the infant's head. Stable activity is reached after 3 to 5 min. During the following 24 h, the decay-corrected loss of activity was 1.0% (0.4–1.5) per hour.
The mean value of the coefficient of back diffusion α was estimated at 3.4 (2.8–4.4) (Fig. 3). The mean urinary excretion over 24 h was 20% (11.4–27.7). Renal function was evaluable in nine infants and excretion was significantly related to renal function evaluated by serum urea (p = 0.02, r2 = 0.55) (Fig. 4).
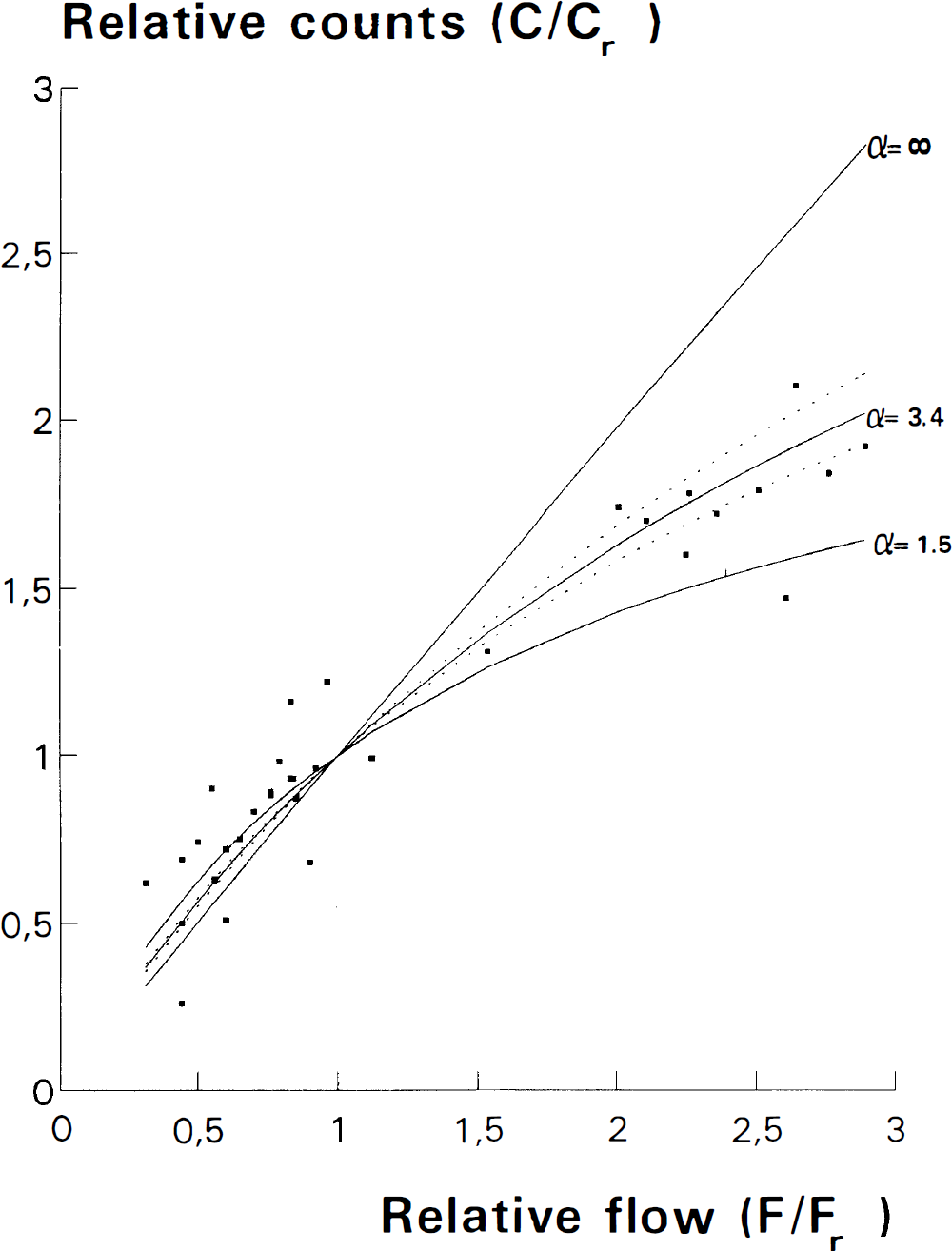
Relative number of counts (C/Cr) in a high an intermediate and a low activity region of 99mTc-HMPAO distribution images compared with relative flow (F/Fr) in the corresponding regions of 133Xe flow images in 11 infants. Because of back diffusion of tracer from brain to blood, which may be expressed by α (see text), 99mTc-HMPAO underestimates high-flow. The curve of best-fitted value of α with 95% confidence intervals for the observed values is shown together with the curves for α = 1.5 (adult value) and α = ∞ (ideal tracer).
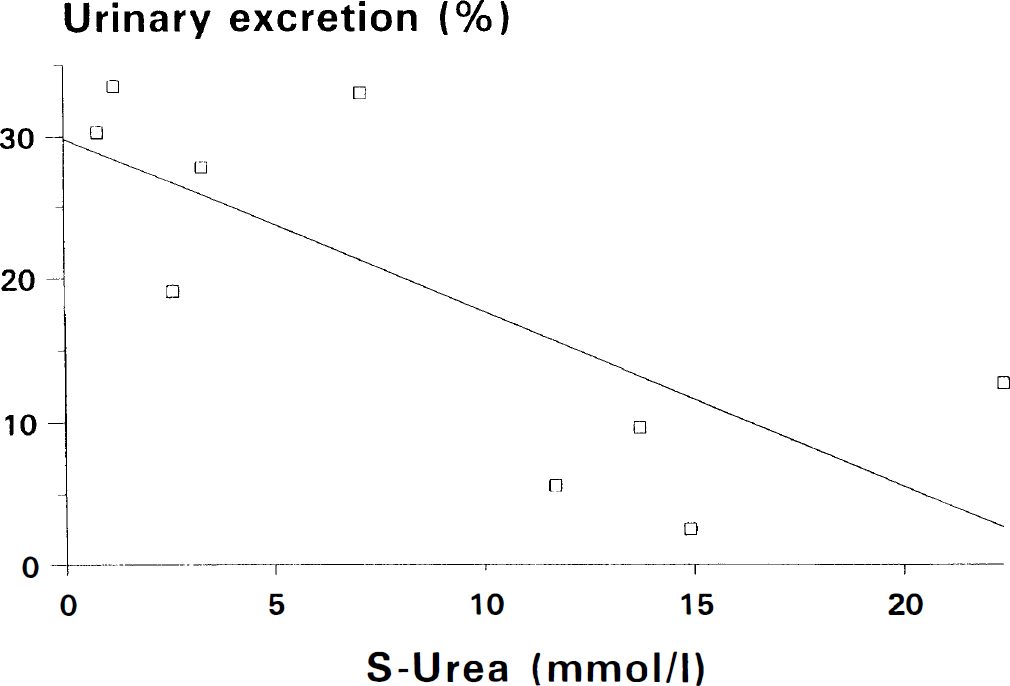
The figure shows a statistically significant correlation between urinary excretion of 99mTc-HMPAO and S-urea concentration. In infants with a normal renal function (S-urea < 5.5 mmol/l), the urinary excretion is of the same magnitude as in adults.
DISCUSSION
The study shows that, as in adults the tracer is trapped in the brain cells within 2 to 3 min; because of the lower CBF in infants, however, the maximal brain activity was only 104% (98–111) of the stable level, in contrast to adults, who have a maximal activity of 111% (Andersen et al. 1988a). During the next 24 h, the decay-corrected leakout was 1.0%/h (0.4–1.5), showing that the concentration of HMPAO in the brain remained almost constant.
In neonates, the fraction of cardiac output delivered to the brain is approximately 9%, assuming a cardiac output of 200 ml/kg, a brain mass of 12% of the body weight (Gruenwald and Minh, 1960), and a CBF of 15 ml 100 g–1 min–1 (Greisen, 1986). A study of adult patients with a CBF ranging from 39 to 133 ml 100 g–1 min–1 has shown a linear correlation of E to CBF (Andersen et al., 1988b)
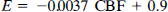
If this relationship is also valid in the neonatal flow range, that is, 10–20 ml/100 g–1/min–1 and, when inserting the observed value of α of 3.4, the model will predict, using Eq. 2, a cerebral retention of approximately 6%. This estimate corresponds with the observed cerebral retention of 99mTc-HMPAO in neonates of 6.8% (6.1–7.6), and the retention is significantly higher than in adults, who have retention rate of 4.2% (p < 0.01) (Sharp et al., 1986; Matsuda et al., 1988).
According to the kinetic model, the higher retention rate in infants may be caused by a combination of a higher fraction of cardiac output delivered to the brain, a higher extraction efficiency, and a lower degree of back diffusion. In our calculations, we assumed a linear relationship between count rate and cross-sectional area. 99mTc has an attenuation coefficient in water of 0.15 cm–1, and this results in an almost a linear relation between the count rate and cross-sectional area for phantoms below approximately 100 cm2 (Holm, 1988). Therefore, our assumption of a linear relationship is acceptable in infants.
Back diffusion from brain to blood causes a reduction of contrast, but knowing α it is possible to correct for this loss of contrast based solely on the steady-state 99mTc-HMPAO distribution image by Eq. 5. The basic assumptions for this method are that the extraction E, the conversion rate from lipophilic to hydrophilic HMPAO in the brain k3, and the partition coefficient λ, are the same in all regions (Lassen et al., 1988). The partition coefficient λ expresses the solubility of the tracer in tissue compared with blood and, because of the higher lipid content of white matter, for a lipophilic tracer such as 99m Tc-HMPAO, a higher value in this region might be expected. In adults, different values for white and gray matter have been shown (Matsuda et al., 1988); however, in infants the lipid content of the white matter is less and the same value of λ for white and gray matter can be assumed (Greisen 1986). Assuming the same value of k3 in all parts of the brain may be critical for the model. In damaged tissue, the conversion from lipophilic to hydrophilic HMPAO may be reduced, causing false low values in the flow distribution image. The rate constants k1, …, k6 and, by that also CBF and α, may be determined directly, but to do so demands catheterization of the internal jugular vein that was not acceptable in our patients. Because of the correction equation already described, it is possible to determine the degree of back diffusion α by an indirect method of comparing the contrast in 133Xe flow images and 99mTc-HMPAO distribution images. The assumptions for this method are the same as those for the correction algorithm and add to this that the compared images show identical parts of the brain and that the flow distribution does not change between the studies. These supplementary assumptions were ensured by performing the two studies with the infant in the same clinical situation and physical position. The ROIs in the flow images were not placed in regions with a risk of airway artifacts. Airway artifacts in the flow images would tend to increase the contrast in the high-flow areas and thereby a decreased estimate of α. In adults the reference region may be cerebellum or the whole brain (Syed et al., 1992). In neonates we chose the hemispheres as the reference region because the cerebellum is not so well defined and high perfused in neonates as in adults. In accordance with the lower flow in infants, we found that the fixation/clearance ratio α was higher in infants than in adults.
The urinary excretion of 99mTc-HMPAO was 20% in neonates, significantly less than in adults, who have a urinary excretion of 35% (p < 0.001) (Sharp et al. 1986), which may be caused mainly by reduced renal function in the first week of life (Modi 1988). In adults 60% of 99mTc-HMPAO in the blood is located in the red blood cells, and 40% is located in the plasma (Andersen et al., 1988c). The HMPAO in plasma is excreted renally and hepatically, whereas HMPAO in blood cells is excreted only hepatically. Therefore, the higher hematocrit in neonates may contribute to the reduced urinary excretion. Radiation to the bladder wall will be reduced by about 40%. As hepatic function may be impaired in neonates, decreased renal excretion may not lead to increased radiation to the liver, gallbladder and gut. Instead, it will cause an 8% increased radiation to all organs. In published calculations of the whole-body equivalent, the reduced urinary excretion was not considered; however, the increased radiation to all organs and the reduced radiation dose to the bladder wall have no net effect on the whole body equivalent, which is estimated at 0.3 mSv/MBq/kg (Vestergren et al., 1992).
We conclude that 99mTc-HMPAO is a more reliable tracer of the distribution of CBF in neonates compared with adults. A four-compartment model describing the kinetics of 99mTc-HMPAO is also valid in neonates. Cerebral retention of the tracer is higher in neonates because of higher extraction and lower initial back diffusion from brain to blood. In linearizing 99mTc-HMPAO distribution images, smaller correction is necessary, and we propose a value of the fixation/clearance ratio α of 3.4. Urinary excretion is significantly reduced compared with adults, and the radiation dose to the bladder wall is reduced.
Footnotes
Acknowledgment:
We thank Dr. Allan Rabøl, Department of Clinical Phsyiology and Nuclear Medicine, The National University Hospital, Copenhagen, Denmark, for technical assistance.
This study was supported by the A. P. Møller and Chastine McKinney Møller Foundation.


