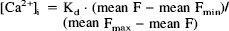Abstract
The effects of SB 206284A, 1-[7-(4-benzyloxyphenoxy)heptyl] piperidine hydrochloride, have been investigated in vitro on calcium and sodium currents in rat-cultured dorsal root ganglion (DRG) neurones and potassium-mediated calcium influx in rat synaptosomes. Cardiovascular hemodynamic effects in both anesthetized and conscious rats, and neuroprotective activity in in vivo cerebral ischemia models were also investigated. In the rat DRG cells, SB 206284A caused almost complete block of the sustained inward Ca2+ current (IC50 = 2.4μM), suggesting that the compound is an effective blocker of slowly inactivating, high-voltage calcium current. SB 206284A reduced locomotor hyperactivity in the gerbil bilateral carotid artery occlusion model without affecting ischemia-induced damage in the hippocampal CA1 region. In the rat middle cerebral artery occlusion model, SB 206284A reduced lesion volume in the posterior forebrain, and in the rat photochemical cortical lesion model, lesion volume was reduced even when treatment was delayed until 4 hours after occlusion. At neuroprotective doses, SB 206284A had no cardiovascular effects. These findings show that SB 206284A is a novel calcium channel antagonist that shows neuroprotective properties.
The use of calcium-channel antagonists in ischemic brain injury has been extensively studied in animal models (Sauter and Rudin, 1986; Van Reempts et al., 1987; Alps et al., 1988; Ito et al., 1994), but a consistently effective compound has yet to be discovered. For example, although the calcium-channel antagonist nimodipine has been reported to be neuroprotective in some studies (Gotoh et al., 1986; Germano et al., 1987), it had no effect on lesion volumes in other experiments (Snape et al., 1993; Womack et al., 1993). Similarly, isradipine (PN200-110)-reduced ischemic injury in some (Abe et al., 1988; Sauter et al., 1989), but not all (Marinov and Wassman, 1991), experiments. Both of these compounds are dihydropyridines, a class of calcium antagonists known for their preferential vasodilator properties, so it is possible that at least some of their apparent neuroprotective effects are caused by changes in blood pressure and cerebral blood flow rather than neuroprotection. This is especially true of isradipine, in which predosing in spontaneously hypertensive rats to the level at which their blood pressure was normalized, attenuated the neuroprotective efficacy of this drug (Sauter and Rudin, 1991). This feature is caused by the action of dihydropyridines at L-, and possibly T-, type channels, the only types found in smooth and cardiac muscle. However, neuronal tissue contains an additional number of neurone-specific channels (e.g., N and P), so an alternative approach to the development of L-channel selective compounds is to concentrate on those that show activity at all neuronal calcium channels. SB201823-A is such a compound because it is selective for calcium channels over sodium and potassium channels, but has little selectivity for the different subtypes of calcium channels (Benham et al., 1993). We have previously shown the neuroprotective efficacy of this novel, nonpeptide, calcium-channel antagonist in both the gerbil bilateral carotid artery occlusion (BCAO) and rat photothrombotic lesion models (Benham et al., 1993). In addition, SB201823-A reduced lesion volume in a rat middle cerebral artery occlusion (MCAO) model but also caused a decrease in heart rate at a neuroprotective dose (Barone et al., 1995).
In the present studies, we describe the neuroprotective profile of a new calcium antagonist, SB 206284A [1-[7-(4-benzyloxyphenoxy)heptyl] piperidine hydrochloride], in models of cerebral ischemia and also show its lack of systemic cardiovascular properties in the rat. Some of this work has recently been presented in abstract form (Campbell et al., 1995; Wood et al., 1995).
MATERIALS AND METHODS
All procedures used in this study were performed in accordance with either the Animals (Scientific Procedures) Act (1986) for experiments conducted in the United Kingdom, or the Guide for the Care and Use of Laboratory Animals (Department of Health, Education and Welfare, Bethesda, MD, U.S.A.) for studies conducted in the United States. In addition, all protocols were approved by a SmithKline Beecham internal ethics committee.
Electrophysiological studies
Sensory neurones from dorsal root ganglia (DRG) were dissociated from 1-day-old rat pups using the method of Forda and Kelly (1985). Cells were plated out onto glass coverslips and used within 24 hours for sodium current measurements to permit effective voltage clamp of the currents. Calcium currents (using barium as charge carrier) were measured in cells up to 4 days in culture. Whole-cell membrane currents were recorded using standard patch-clamp techniques.
The internal solution in the patch pipette contained 130 mM cesium chloride, 10 mM N-2-hydroxyethylpiperazine-N-2 ethanesulfonic acid (HEPES), 10 mM ethylene glycol-bis (β-aminoethyl ether)-N,N-tetraacetic acid (EGTA), 4 mM magnesium chloride, and 2 mM ATP, buffered to pH 7.2 with cesium hydroxide. For sodium current measurements 5 mM sodium chloride was added to the internal solution. The external solution for measuring sodium currents contained 145 mM sodium chloride, 2.5 mM potassium chloride, 10 mM glucose, 10 mM HEPES, 1.2 mM magnesium chloride, 0.15 mM calcium chloride, and 0.1 mM cadmium chloride buffered to pH 7.4 with sodium hydroxide. The external solution for recording calcium channel currents consisted of 10 mM barium chloride, 130 mM tetraethyl ammonium (TEA) TEA-Cl, 10 mM glucose, 10 mM HEPES, 1 mM magnesium chloride and 0.001 mM tetrodotoxin, buffered to pH 7.3 with TEA-OH.
All experiments were performed at 21–24°C. Test compounds were dissolved in dimethyl sulphoxide to make a 20 mM stock solution. At the maximum drug concentration used, the vehicle (0.1%) had no effect on calcium currents.
Calcium influx
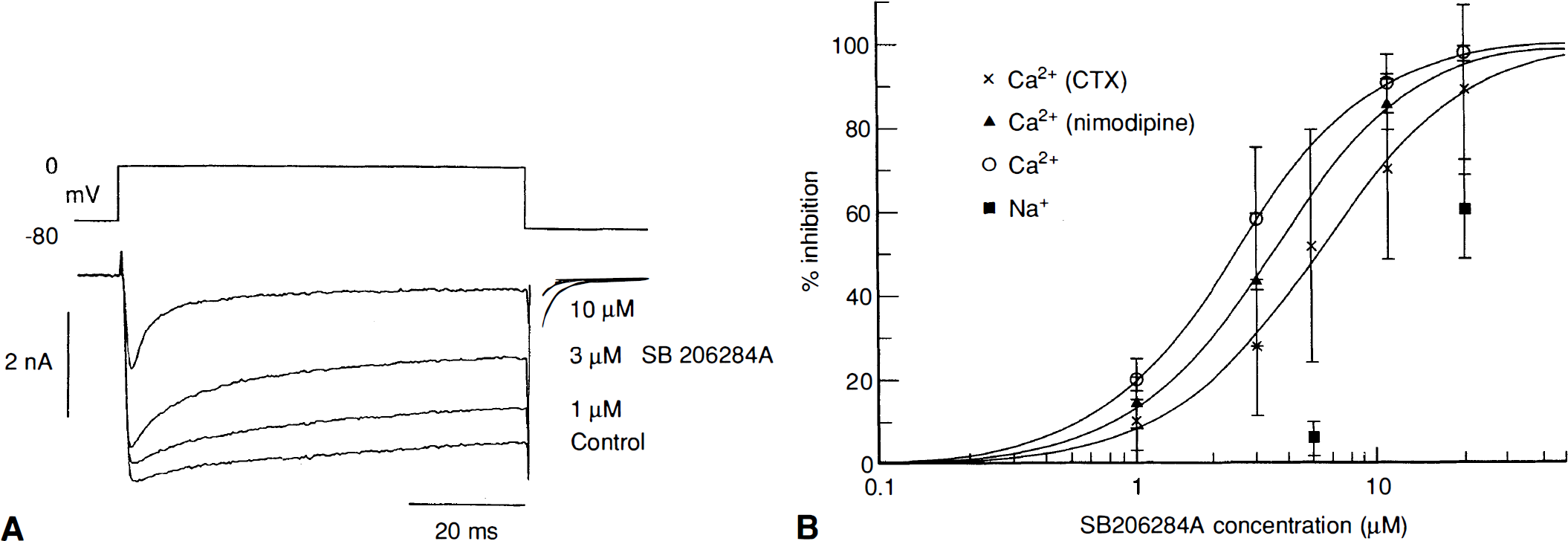
Synaptosomes were prepared from the cerebral cortex of male Sprague-Dawley rats (200–300 g), using isotonic sucrose/Percoll discontinuous density gradient centrifugation, by a method adapted from that of Dunkley et al. (1987). The synaptosomes were loaded with Fluo-3AM (Molecular Probes Inc., Eugene, OR, U.S.A.) (cell permeant acetoxymethyl ester of the calcium indicator, Fluo-3, at a final concentration of 10 μM in 1% dimethyl sulphoxide) in HEPES buffer (100 mM HEPES, 1.2 mM potassium dihydrogen phosphate, 1 mM magnesium chloride, 142 mM sodium chloride, 2.4 mM potassium chloride, 1 mM calcium chloride, 11.1 mM glucose, pH 7.4) containing 10 mg/ml bovine serum albumin, and were incubated at 30°C for 1 hour. Excess indicator and bovine serum albumin were removed by centrifugation and resuspension in HEPES buffer three times. Fluorescence measurements were performed at 25°C (excitation 485 nm, half bandwidth 20 nm; emission 530 nm, half bandwidth 25 nm) in a Millipore Cytofluor 2300 plate reader (Millipore Corp., Watford, Herts, U.K.) (20 μl synaptosomes/well, approximately 80 μg protein). The effect of SB 206284A or vehicle (distilled water) on resting internal calcium concentration [Ca2+]i and potassium-stimulated [Ca2+]i was measured after a 10-min preincubation period. Potassium stimulated changes in [Ca2+]i were observed about 15 seconds after addition of 50 mM potassium chloride. The [Ca2+]i at each concentration of the compound was calculated from the fluorescence intensities (F) using the following equation, where the dissociation constant of the Fluo-3-calcium complex (Kd) = 400nM (Merritt et al., 1990):
Fmax was determined in the presence of calcium on synaptosome lysis with 0.1% sodium dodecyl sulphate. Fmin was estimated from the fluorescence of wells in which synaptosomes were replaced by an equal volume of buffer. The additional contribution of unloaded synaptosomes was less than 8% of the total fluorescence signal. Statistical analysis was by Newman-Keuls method.
Cardiovascular data
Anesthetized rats.
Male Lister hooded rats (Charles River, UK) in the weight range 250–500 g were anesthetized by sodium pentobarbitone, 60mg/kg intraperitoneally (IP). The femoral artery was cannulated and connected to a blood-pressure transducer. Arterial blood pressure was recorded and captured using Macintosh IIci hardware (MACLAB Instruments Ltd, Hastings, East Sussex, UK), with heart rate being derived from the arterial pulse. Data were analyzed using MacLab/8 software.
After blood pressure and heart rate had equilibrated, SB 206284A (10mg/kg) or vehicle (10% hydroxypropyl β-cyclodextrin in saline) was infused over 30 min into a cannulated femoral vein. The animal was then killed by pentobarbitone overdose.
Conscious rats.
Male Lister hooded rats (Charles River, U.K.) in the weight range 300–400g, were treated with antibiotics (subcutaneously) 1 hour before surgery. Anesthesia was induced using medetomidine (Domitor, Pfizer, Sandwich, Kent, UK) and fentanyl (Sublimaze; Janssen Pharmaceutica, Inc., Piscataway, NJ, U.S.A), and cannulae were implanted in the femoral artery and vein and exteriorized between the scapulae. The cannulae were flushed with heparinized saline (250 U/ml) and heparinized dextrose (1000 U/ml heparin in 25% weight-to-volume ratio dextrose) to prevent thrombus formation at the tip of the cannulae. The anesthesia was reversed using atipamezole (Antisedan) and nalbuphine (Nubain; DuPont Pharm., Wilmington, DE, U.S.A.). After recovery from surgery, rats were housed individually. The day after surgery, and on alternate days thereafter, the cannulae were flushed. Rats were allowed to recover from surgery for at least 7 days before experimentation.
On the day of observation, rats were placed individually in conical restrainers and the cannulae were flushed with heparinized saline (250 U/ml). The arterial cannula was connected to a arterial pressure transducer and the arterial blood pressure and heart rate (electronically derived from the arterial blood pressure signal) were continuously recorded using a multichannel recorder. After a 1-hour stabilization period, SB206284A (10 mg/kg) or vehicle (as above) (1.5 ml) were infused intravenously [IV] during a 15-min period and mean arterial pressure and heart rate were monitored for 4 hours.
Ischemia models
All animals were housed singly under standardized environmental conditions (12 h light/dark cycle, 21 ± 1°C, and 55 ± 5% humidity) and allowed free access to food and water.
Gerbil bilateral carotid artery occlusion.
Male Mongolian gerbils (B & K, UK), in the weight range 50–70 g, were anesthetized with halothane in oxygen. Core body temperature was maintained at 37 ± 1°C with a heating blanket. The common carotid arteries were clamped with atraumatic aneurysm clips for 10 min. Blood flow was then reestablished on removal of the clips. The wound was then sutured closed and the animal allowed to recover for 2 hours in an incubator with an ambient temperature of 33–35°C. After recovery, the gerbils were housed individually with access to food and water ad libitum. Sham animals underwent the same surgical procedure but their carotid arteries were not isolated or clamped.
SB 206284A was administered IP at a dose of 30 mg/kg 30 min after occlusion, then at 10 mg/kg twice a day for 3 days. The vehicle was 10% hydroxypropyl β-cyclodextrin in saline.
Four days after surgery, 18 hours after the final dose of compound, locomotor activity was monitored by placing the gerbils in clear perspex boxes, where their movements were detected and recorded via the interruption of light beams directed across the box 3 cm above the base (ACE Software Ltd., Basingstoke, Hants., U.K.). Results were analyzed using analysis of variance statistics. After behavioral assessment, the animals were killed with an overdose of sodium pentobarbitone, and were then perfused via the left ventricle with neutral buffered formalin containing 5% sucrose. The brains were removed and placed in neutral buffered formalin for a minimum of 48 hours before sectioning and staining. A total of 12 serial coronal sections 12-μm thick were cut at the level of the dorsal hippocampus and stained with 1% cresyl fast violet. Cells in the CA1–CA3 region of the hippocampus were counted, using a DataCell image analysis system with Optimas software (Maidenhead, Berks, U.K.) and a cell-count program that was developed in-house. This assessment and subsequent analysis by analysis of variance were performed blind to the operator. In addition to counting cells in the hippocampus, other brain areas (e.g. entorhinal and somatosensory cortex) were examined for signs of neuronal damage.
Rat middle cerebral artery occlusion model.
Male Fisher F-344 rats, weighing 250–290 g, were anesthetized with isoflurane, and body temperature was maintained at 37 ± 1°C by a heated blanket with feedback control. The right middle cerebral artery was occluded by electrocoagulation (Aspen Labs Inc, Littleton, CO, U.S.A.; model MF 180 electrosurgical unit) at the level of the inferior cerebral vein. The right common carotid artery was then occluded by double ligation and transection. The right femoral vein was cannulated for compound administration. Animals were allowed to recover from anesthesia under a heating lamp, and were then returned to their cages.
SB 206284A was administered intravenously at a dose of 10 mg/kg over 1 hour, beginning 30 min after MCAO. Control animals received an equal volume of vehicle (10% hydroxypropyl-β-cyclodextrin in saline).
At 24 and 48 hours after surgery, a neurologic examination was performed, as previously described (Barone et al., 1995). Briefly, the animals were tested for hindlimb placement and degree of hemiparesis.
Forty-eight hours after MCAO, the rats were killed by sodium pentobarbitone overdose, and their brains removed. Seven coronal forebrain slices (2-mm thick) were made from the level of the olfactory bulbs to the cortical-cerebellar junction. The slices were immersed immediately in a 1% solution of triphenyltetrazolium chloride in phosphate buffer at 37°C for 20–30 min and then fixed by infiltration in 10% phosphate-buffered formalin. Color photographs of each triphenyltetrazolium chloride-stained section were then analyzed for quantification of ischemic damage by an Amersham RAS 3000 image analysis system (Loates Associates Inc., Westminster, MD, U.S.A.). Morphological changes after surgery were evaluated in the entire forebrain for each animal as previously described (Barone et al., 1992; 1994). Hemispheric swelling, infarct size, and infarct volume were determined for each slice. Hemispheric swelling was expressed as the percent increase in size of the ipsilateral hemisphere over the contralateral hemisphere. Infarct size was calculated as the percent infarcted tissue compared with the contralateral hemisphere. Total infarct volume was calculated by summation of the infarct area (in mm2) from all of the brain planar images that were considered 1-mm thick.
For statistical analysis of nonparametric data (behavioral tests), the X2 test was used. For parametric data, comparisons were made using an analysis of variance and/or t test.
Rat photothrombotic lesion model.
Male Lister hooded rats (Charles River, UK), weighing 250–280 g, were used in the following three experiments. Animals were anesthetized with halothane in oxygen, and rectal temperature was maintained at 37 ± 1°C by a heated blanket with feedback control. They were placed in a stereotaxic frame and a bifurcated fiber-optic light guide (diameter 3.0 mm) connected to a 300 W xenon arc lamp (Oriel Scientific Ltd) was positioned on the skull at the level of bregma, with the heads of the light guide centered 2.5 mm either side of the midline. An in-line water filter located between the arc lamp and light guide removed the heat from the light—control studies revealed no increase in skull temperature below the light guides (Wood, N. unpublished findings). Rose Bengal dye (Aldrich), 20 mg/kg, was infused during a period of 60 s into a lateral tail vein, and the skull illuminated for 5 min. The scalp was then sutured and the animals were returned to their home cages to recover.
The following drug dosing regimes were used (in all cases the vehicle was 10% hydroxypropyl b-cyclodextrin in saline): SB 206284A (10 mg/kg) or vehicle infused via a tail vein over a period of 15 min, beginning 10 min after illumination; SB 206284A (10 mg/kg) IP 2 hours after illumination, and twice daily for 3 days; SB 206284A (10mg/kg) IP 4 hours after illumination, and twice daily for 3 days. Histologic procedures are described below.
Marmoset photothrombotic lesion model.
Adult marmosets of both sexes (bred in-house), weighing 300–500 g were anesthetized initially with an intramuscular dose of alphaxalone/alphadolone (Saffan) and subsequently maintained at surgical anesthesia by intravenous administration of Saffan. The animals were placed in a stereotaxic frame with ear, palate, and orbital bars fitted, and lesion induction was performed using a single-headed light guide. The light guide was positioned on the skull at the following coordinates: anteroposterior zero plus 14.5 mm, midline minus 2.5 mm. Rose Bengal, 20 mg/kg, was infused during a period of 60 s into the intravenous cannula. The skull was then illuminated for 5 minutes, after which the light guide was moved to the other side of the skull (midline plus 2.5 mm), and the light switched on for an additional 5 minutes. The scalp wound was closed using subcuticular stitches and cyanoacrylate tissue adhesive was applied. Throughout the procedure, rectal temperature was maintained at 37 ± 1°C by a heated blanket with feedback control.
Drug administration was initiated 10 min after illumination; SB 206284A (10 mg/kg) or vehicle (10% hydroxypropyl-β-cyclodextrin) was infused via the intravenous cannula during a period of 15 min. The intravenous cannula was then removed, after which the animals were placed in recovery cages under infrared heating lamps to recover fully before being returned to their home cages.
Histologic procedures in photothrombotic model.
Histopathologic assessment was performed in the same manner for both rats and marmosets. Seven days after surgery all animals were killed with an overdose of sodium pentobarbitone, and were then perfused via the left ventricle with neutral buffered formalin containing 5% sucrose. The brains were removed and left in neutral buffered formalin/sucrose for a minimum of 48 hours. Coronal sections were cut serially through the length of the lesion at a thickness of 60 μm, and stained with cresyl fast violet. Lesions were measured planimetrically using a Quantimet 920 image analyser (Cambridge Instruments) and lesion volumes calculated using Simpson's rule. Statistical analysis was by analysis of variance.
RESULTS
Electrophysiologic studies
In DRG cells, application of SB 206284A caused a concentration-dependent reduction in calcium current. Figure 1(A) shows current recorded at a test potential of 0 mV under control conditions and in the presence of 1, 3, and 10 mM SB 206284A, with a 3-min contact time. SB 206284-A, 10 mM, produced almost complete block of the sustained inward current, indicating that SB 206284A is an effective blocker of slowly inactivating, high-voltage calcium current. The increased efficacy of SB 206284 in blocking sustained current relative to peak current might be explained by voltage- dependent block of the current. When applied to cells pretreated with 10 mM nimodipine (to block L type currents), and to other cells pretreated with 10 μM  -conotoxin GVIA (
-conotoxin GVIA ( ) (to block N-type currents), the inhibitory effects of SB 206284A show little difference in IC50 indicating little subtype selectivity of this compound (Table 1). However SB 206284A was less potent as a blocker of voltage-gated sodium currents evoked in DRG neurons (Table 1 and Figure 1B).
) (to block N-type currents), the inhibitory effects of SB 206284A show little difference in IC50 indicating little subtype selectivity of this compound (Table 1). However SB 206284A was less potent as a blocker of voltage-gated sodium currents evoked in DRG neurons (Table 1 and Figure 1B).
IC50 values for SB 206284A in rat DRG cells

CTX is ω-conotoxin GVIA.
Concentration-dependent block of calcium and sodium channels in sensory neurones by SB 206284A, 1 −30 μM. (
Intracellular administration of SB 206284A, by addition to the patch pipette internal solution, resulted in significantly weaker block of total DRG calcium current than when applied extracellularly. SB 206284A, 50 and 100 μM, was applied intracellularly to two separate batches of cells. The degree of block was assessed by measuring the calcium current 3 min after breakthrough relative to the first calcium current measured within 30 s of breakthrough. These two doses blocked 17 ± 12% (n = 4) and 14 ± 16% (n = 4) of the calcium current respectively. Current block of this magnitude cannot be accurately dissociated from possible cell rundown. When applied extracellularly to the same two batches of cells, 20 μM SB 206284A blocked 82 ± 24% (n = 4) and 83 ± 31% (n = 4) of the current respectively. The results suggest that the binding site for SB 206284A is located on the extracellular surface of these neurones and that the binding site cannot be accessed by this compound from an intracellular location, e.g., by traversing the cell membrane.
Calcium influx in synaptosomes
SB 206284A was tested for its potency in inhibiting potassium-stimulated calcium elevation in cortical synaptosomes, a model for presynaptic calcium entry. At concentrations up to 100 μM, SB 206284A had no significant effect on basal calcium in cortical synaptosomes (Fig. 2). However, SB 206284-A at concentrations of 50 and 100 μM caused a 30% reduction in potassium-stimulated calcium influx, with the lower concentration producing a significant decrease (p < 0.05). SB 206284A at concentrations of 100 μM did not inhibit calcium influx to any greater degree than did concentrations of 50 μM, which suggests that SB 206284A blocks a subpopulation of calcium channels that are present on cortical synaptosomes, but leaves unblocked a further subpopulation that is responsible for the remainder of the potassium-stimulated calcium influx. For comparison, SB201823-A, which is effective in three models of ischemia, blocks all of the depolarization-induced calcium influx at concentrations greater than 50 μM (Benham et al., 1993). Selective blockers of L- and N-type calcium channels inhibit only a small component of this calcium influx. Thus the maximal effect of the L-type channel antagonist nimodipine was a 7% block at a concentration of 10 μM.  GVIA, at 1 μM, a concentration that produces maximal block of N-type channels in rat DRG cells (Benham et al., 1993), blocked only a small proportion of the potassium stimulated calcium release (Meakin and Smith, 1993 and unpublished data).
GVIA, at 1 μM, a concentration that produces maximal block of N-type channels in rat DRG cells (Benham et al., 1993), blocked only a small proportion of the potassium stimulated calcium release (Meakin and Smith, 1993 and unpublished data).
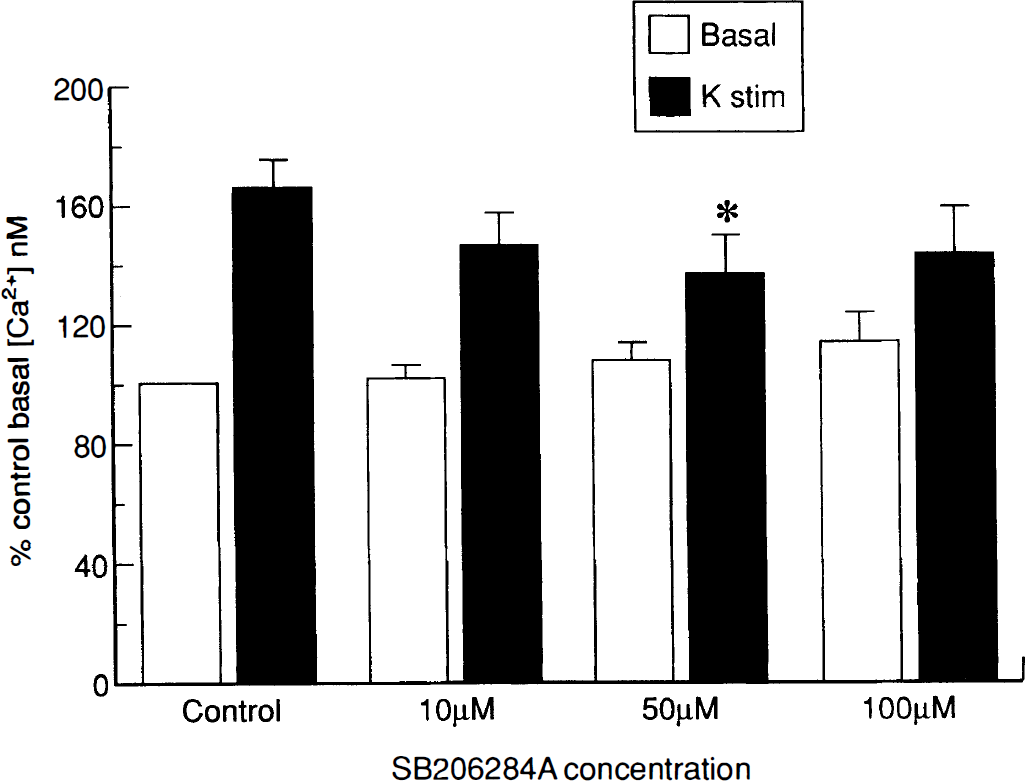
Effect of SB 206284-A, 10–100 μM, on basal calcium, and on potassium-stimulated synaptosomal calcium levels. Data were normalized to control Ca2+ of 412.6 ± 23 nM (n = 6). Values are mean ± SD. *p < 0.05.
Cardiovascular data
SB 206284-A (10 mg/kg IV over 30 min) did not significantly affect blood pressure or heart rate when compared with the respective vehicle group in both anesthetized and conscious rats (Tables 2 and 3).
Effect of SB 206284-A on cardiovascular parameters in anesthetized rats

Values are mean ± SD. SB 206284-A (10 mg/kg was infused i.v. over 30 min).
Effect of SB 206284-A on cardiovascular parameters in conscious rats

Values are mean ± SD. SB 206284-A (10 mg/kg was infused i.v. over 30 min).
Ischemia studies
Gerbil bilateral carotid artery occlusion model.
There was no difference in the extent of ischemic injury caused by 10 min BCAO in the hippocampal CA1 region between drug- (administered IP after occlusion) and vehicle-treated groups when determined 4 days after occlusion (Fig. 3A). However, SB 206284A caused a significant reduction in the hyperactivity induced by 10 min global ischemia. (Fig. 3B).
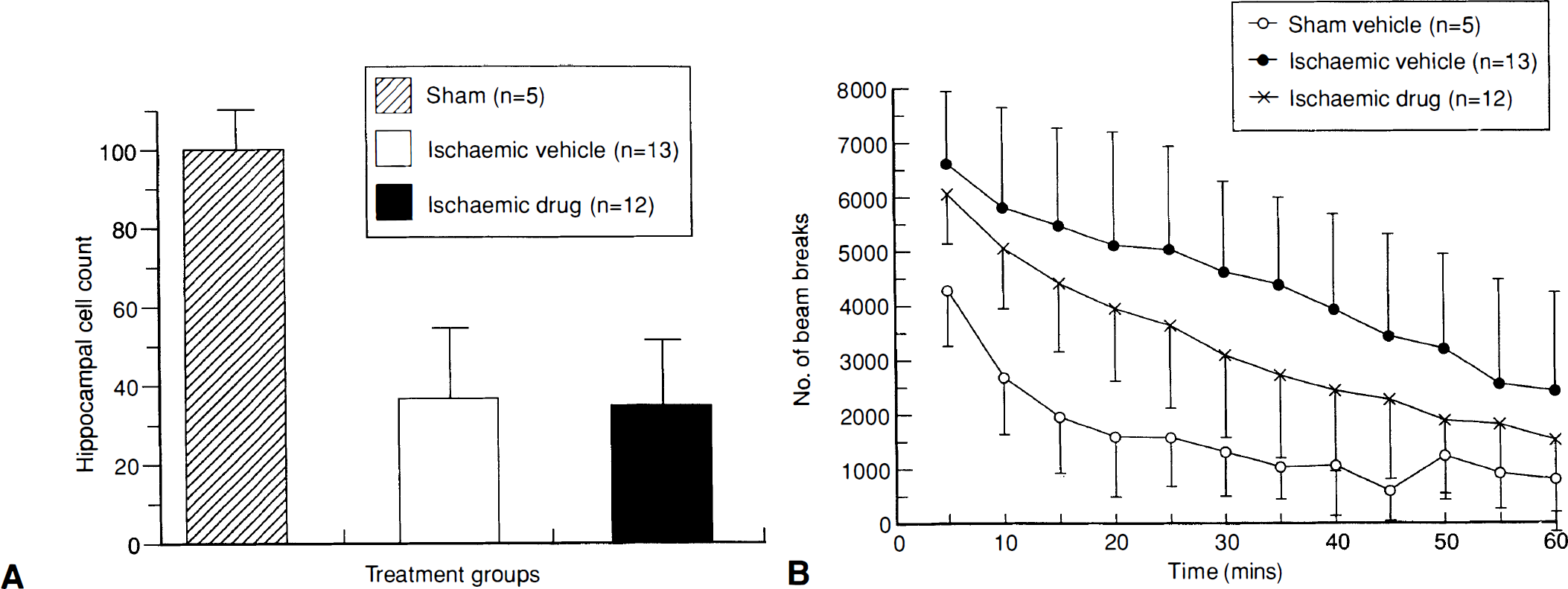
Histological and functional effects of SB 206284A (30 mg/kg IP 30 min after occlusion, then as 10 mg/kg IP twice daily for 3 days) in the gerbil bilateral carotid artery occlusion model. (
Rat middle cerebral artery occlusion model.
Intravenous infusion of SB 206284A, 10 mg/kg commencing 30 min after MCAO, significantly reduced both hemispheric swelling (Fig. 4A) and infarct size in some areas of the posterior forebrain at 48 h (Fig. 4B). SB 206284A did not cause an improvement in hindlimb neurologic deficits (data not shown).
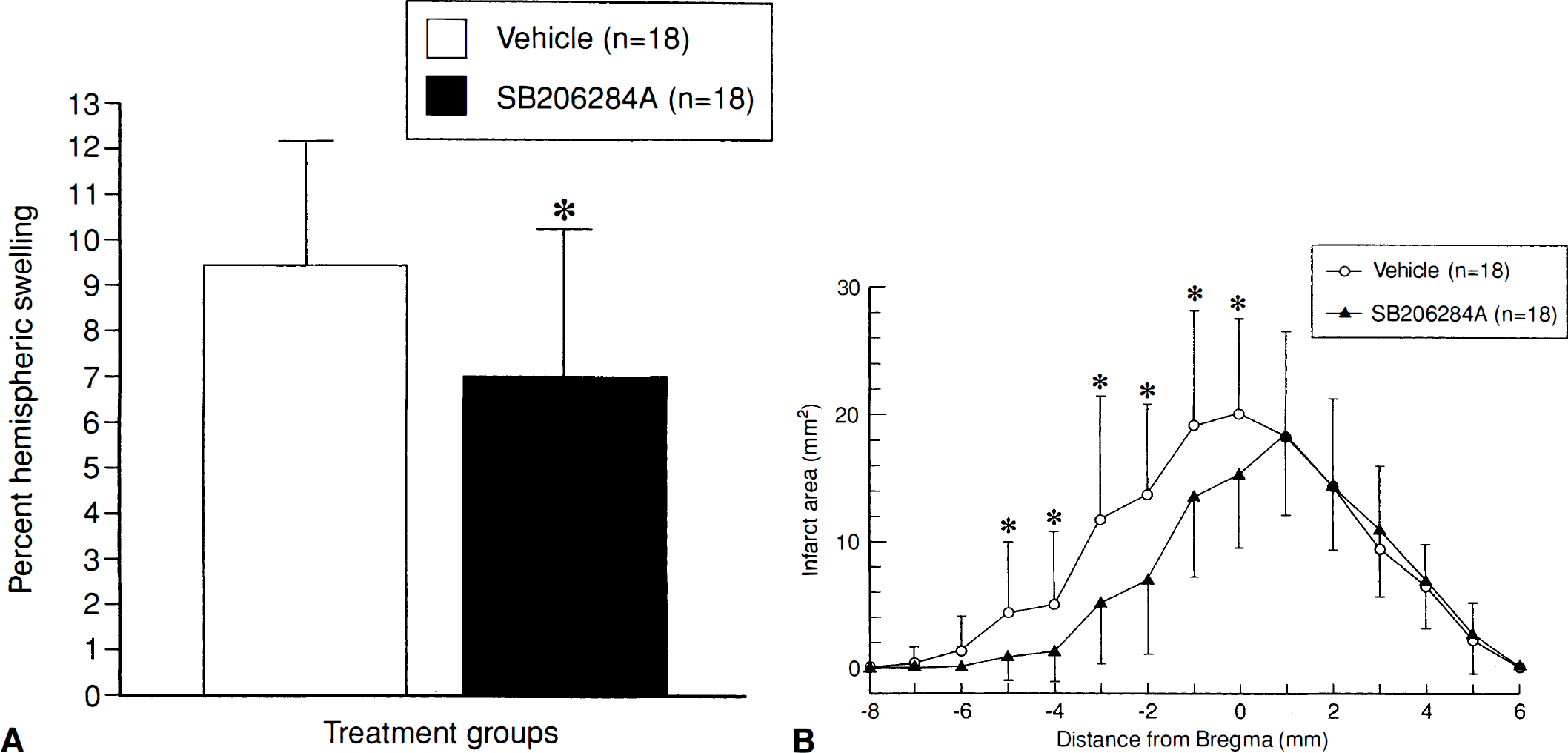
Effect of SB 206284-A, 10 mg/kg i.v, on (
Rat photothrombotic lesion model.
Under all dosing conditions SB 206284A significantly reduced lesion volume at 7 days in this model. The greatest effect (67% reduction in lesion volume) was produced when the compound was administered as an intravenous infusion of 10 mg/kg over 15 minutes, beginning 10 min after surgery. The compound was also neuroprotective when given as several IP bolus doses, even when the first dose was delayed until 2 or 4 hours after surgery, resulting in a 54% reduction in lesion volume (Table 4).
Effects of SB 206284-A, 10 mg/kg and vehicle, given post occlusion, on lesion volume (mean ± SD) at 7 days in photothrombotic models
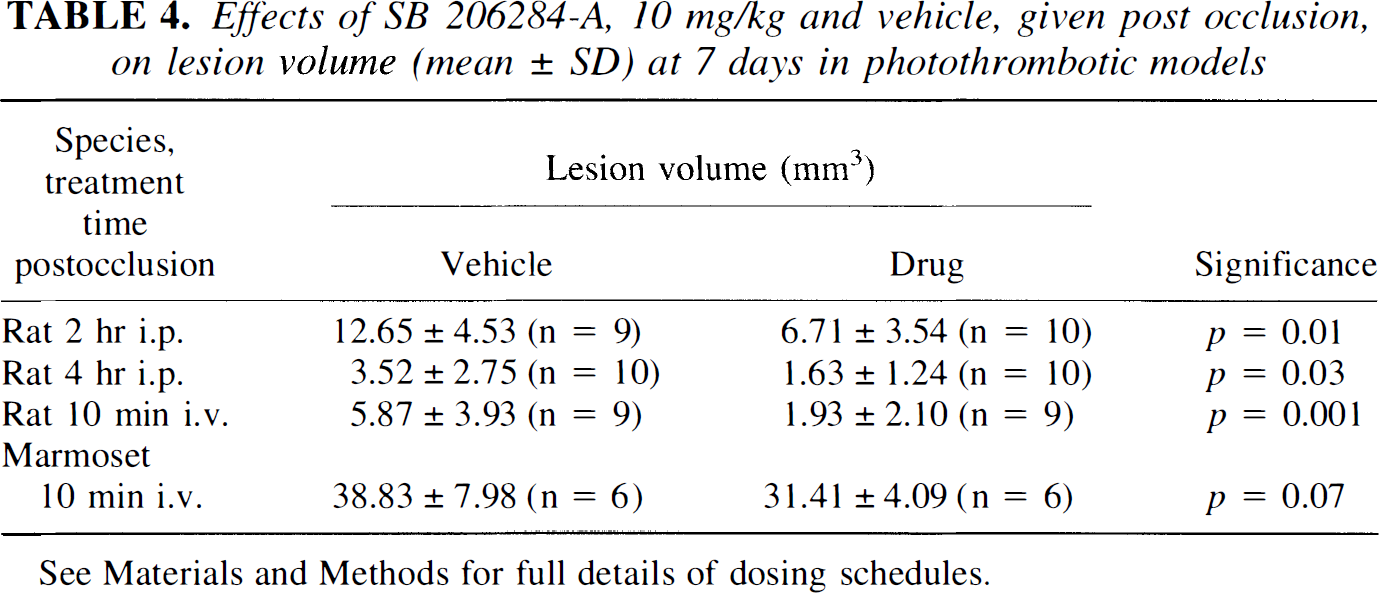
See Materials and Methods for full details of dosing schedules.
Marmoset photothrombotic lesion model.
SB 206284A, when administered as a 10 mg/kg infusion over 15 min beginning 10 min after surgery, produced a nonsignificant reduction in lesion volume at 7 days (Table 4).
DISCUSSION
These results show that the structurally novel neuronal calcium-channel antagonist SB 206284A is neuroprotective in three models of cerebral ischemia at doses that show no systemic cardiovascular effects. This profile represents an improvement over that of existing calcium-channel antagonists such as isradipine and SB201823-A which, although neuroprotective, significantly lower blood pressure (Sauter and Rudin, 1991) and heart rate (Barone et al., 1995), respectively. SB 206284A shows little selectivity for L- or N-type channels when blocking high threshold calcium channels in rat DRG cells and has less selectivity for sodium than calcium channels in comparison with SB201823A. Voltage-dependent calcium uptake into synaptosomes is mediated primarily by channels insensitive to dihydropyridines and  CTX GVIA (Thomas et al., 1994). This could explain why SB 206284A, which we have shown acts on these channel subtypes, is effective in blocking only part of the calcium influx into rat cortical synaptosomes. A component of synaptosomal calcium influx (approximately 30%) is reported to be blocked by the P-channel ligand,
CTX GVIA (Thomas et al., 1994). This could explain why SB 206284A, which we have shown acts on these channel subtypes, is effective in blocking only part of the calcium influx into rat cortical synaptosomes. A component of synaptosomal calcium influx (approximately 30%) is reported to be blocked by the P-channel ligand,  aga IVA (Thomas et al., 1994), but the present study did not examine whether SB 206284A does in fact block this channel subtype.
aga IVA (Thomas et al., 1994), but the present study did not examine whether SB 206284A does in fact block this channel subtype.
The effects of intravenously infused SB 206284A on blood pressure and heart rate in both anesthetized and conscious rats were insignificant. Thus, SB 206284A represents an important advance over SB201823-A, which produced a significant bradycardia (100 beats/min) when administered to conscious rats (Barone et al., 1995), because neuroprotective compounds devoid of systemic cardiovascular actions are preferred in clinical studies.
SB 206284A was tested in three different models of cerebral ischemia in three species—one global (gerbil BCAO), and two focal (MCAO in rats, photothrombotic lesions in rats and marmosets) models. When administered as several doses after occlusion in the gerbil, SB 206284A produced a marked reduction in ischemia-induced hyperlocomotion, but no amelioration of hippocampal damage. This was an unexpected finding, especially because it has been suggested that hyperlocomotion correlates with CA1 neuronal loss (Gerhardt and Boast, 1988). However, there is also evidence that it is possible to reduce CA1 damage without decreasing locomotor activity, implying that the two phenomena are not causally related (Gerhardt et al., 1986). Our data support this hypothesis by suggesting that the reverse is also true—it is possible to improve the functional deficit without preserving CA1 neurones. In this study no damage was detected in other brain areas, so the mechanism responsible for this ischemia-induced hyperlocomotion remains unclear. It has been suggested that damage to the entorhinal cortex, or to layers 2 and 3 of the somatosensory cortex, may be involved (Kuroiwa et al., 1991), but this was not observed in our study.
SB 206284A was effective in reducing the cortical injury generated in both the MCAO and photothrombotic models of focal cerebral ischemia in the rat. When administered as a 10 mg/kg intravenous infusion in the MCAO model, SB 206284A decreased total hemispheric swelling and infarct volume in the posterior forebrain. In the case of the photothrombotic model, the same dose of SB 206284A, regardless of the route of administration (IP or IV), reduced lesion volume and was effective even when the first dose was delayed until 4 hours after surgery. In the marmoset, the reduction in volume produced by 10 mg/kg IV SB 206284A did not reach statistical significance. Previously, we found that flunarizine at 10 mg/kg, administered intraperitoneally 30 min after ischemia and twite daily for 3 days, had no effect on lesion volume in this primate model (Milkowski et al., 1994). The failure of both SB 206284A and flunarizine to show neuroprotective efficacy may indicate that this primate variant of the photothrombotic lesion model is particularly resistant to drug treatment.
Although we have shown neuroprotection with SB201823-A (Benham et al., 1993), NBQX (Wood et al., 1993), and SB 206284A in the rat photothrombotic model, flunarizine (data not shown) was not effective. Similarly, isradipine failed to reduce lesion volume in either the gerbil BCAO or rat photothrombotic models (Bailey et al., 1995), although the drug had a positive effect in the rat MCAO model (White et al., 1994). These findings suggest that the BCAO and photothrombotic models are more resistant to amelioration by isradipine. Moreover, published data in the rat MCAO model are themselves equivocal (Sauter et al., 1989; Marinov and Wassman, 1991), showing that this dihydropyridine compound is not always effective even in the same model. Isradipine is not unique in this respect; the literature contains numerous examples of compounds that give positive results in some laboratories but not others. For example, the NMDA antagonist, MK-801, has been reported to be neuroprotective in some (Gill et al., 1987), but not other, experiments (Buchan & Pulsinelli, 1990; Li and Buchan, 1993). The fact that these studies used different models adds to, rather than detracts from, the problem. In the absence of evidence showing that some models are clinically more relevant than others, it is desirable that new compounds should be neuroprotective in as many models as possible. Even if consensus is reached on which is the best model, there remain the questions of the ideal dosing regime and time of killing.
The choice of dosing protocol (pre- vs postocclusion) is an important consideration, especially as some compounds have effects on vascular calcium channels. The dihydropyridines, in particular, reduce vascular resistance in the brain (Harper et al., 1981; Haws et al., 1983), and it has been shown that pretreatment with nimodipine will increase cerebral blood flow in the penumbral region and reduce lesion volume (Mohamed et al., 1985). Therefore, dosing compounds preischemia are likely, partially, to prevent formation of the lesion, rather than salvage the neurones after insult. Because it is debatable whether this reflects a chronic neuroprotection, and as prophylactic treatment is unlikely to be an option in the clinic, preocclusion administration of drug candidates is not the best option for drug selection and development. Therefore, SB 206284A possesses robust neuroprotective properties because it was found to be effective when administered after lesion induction, even when the first dose was delayed until 4 hours after surgery.
Finally, the time allowed to elapse after occlusion before the animals are killed will greatly affect the assessment of neuroprotection. The majority of published studies use short sacrifice times (48 hours or less), when lesion volume will be affected by the presence of edema. As calcium-channel antagonists such as the dihydropyridines (nimodipine, nicardipine, isradipine) and diphenylalkylamines (cinnarizine, flunarizine) also increase cerebral blood flow by dilating cerebral vessels, it is possible that the apparent reduction in lesion volume at short-time points, in part, may be caused by actions on vascular smooth muscle, increased cerebral blood flow, and subsequent enhanced clearance of edema. The diversity of results may therefore reflect the multiple roles of calcium channels in vascular reactivity, neuronal activity, and capillary transport mechanisms. Abe et al. (1988) suggested that the neuroprotective effect of isradipine may be at least partially explained by a reduction in edema formation caused by inhibition of calcium accumulation. Our choice of killing times was selected on the basis that by 4 days after surgery in the gerbil, and 7 days after surgery in the rat and marmoset, edema is largely cleared, and delayed neuronal cell death almost complete. Measurements of lesion volume are then not distorted by the presence of large amounts of water. This distortion was corrected for in the rat MCAO model, in which killing at 48 hours is justified by measuring hemispheric swelling as well as infarct size, the former measurement giving an indication of the extent of edema in addition to neuronal damage.
The choice of models, dosing regimes and end points utilized in the current studies was an attempt to address the criticisms mentioned above. Neuroprotection was shown in both permanent focal models and one transient global ischemia model, after a after infarct SB 206284A dosing regimen. Edema was assessed postmortem (rat MCAO) and killing was delayed long enough to allow for clearance of edema (gerbil BCAO, rat and marmoset photothrombotic models). The absence of cardiovascular side-effects, particularly in conscious rats, suggests that SB 206284A has a superior profile as a neuroprotectant.
In summary, our findings show that SB206284A is a novel neuroprotective agent with no cardiovascular effects at neuroprotective doses. Further work is needed to characterize fully the selectivity of this compound, and this should be possible using recombinant cell lines expressing specific subtypes of calcium channels.