Abstract
To examine the reliability of quantitative positron emission tomography studies in the rat (Rat–PET), we assessed the influence of radioactivity accumulated in the Harderian glands on PET CMRglc determination. We measured CMRglc by PET and ex vivo dissection methods by using 2-[18F]fluoro-2-deoxy-D-glucose in rats with and without focal brain ischemia. The CMRglc values obtained by PET, after correcting with recovery coefficients, were higher than those measured by the ex vivo method at rostral slices, and reduction of the CMRglc in the ischemic brain was not demonstrated by PET in the frontal cortex. The radioactivity accumulated in the Harderian glands prevents the quantitative determination of CMRglc using Rat–PET.
Positron emission tomography (PET) is more advantageous than in vitro or ex vivo studies because it enables noninvasive and repetitive determinations of regional biochemical processes in the same animal. On the other hand, the coarse spatial resolution of conventional PET scanners has hampered quantitative determinations in small animals. Recently, the development of high-resolution PET scanners (Litton et al., 1990; Wienhard et al., 1994) is making it feasible to apply PET to studies in rats (Rat–PET).
Rats have been widely used to examine the pathophysiology of brain ischemia by determining cerebral blood flow and metabolism with an ex vivo autoradiographic technique (Nedergaard et al., 1988; Kita et al., 1995) because several reproducible models have been established (Koizumi et al., 1986; Ginsberg and Busto, 1989; Zea Longa et al., 1989; Buchan et al., 1992). It may be useful to apply the PET technique for studying cerebral circulation and metabolism to these models.
Several authors have studied quantitative PET determination of CBF or CMRglc in rats (Brownell et al., 1991; Ingvar et al., 1991; Magata et al., 1995; Ouchi et al., 1996). These authors compared CBF or CMRglc values or both as determined by PET with those found using ex vivo methods reported by others (Nedergaard et al., 1988; Orzi et al., 1988; Sokoloff et al., 1977) and discussed the possibility of quantitative PET determinations. The actual CBF or CMRglc values obtained by PET vary, however, from study to study. These variations are often attributed to the different anesthetic conditions used. Thus, it remains unclear whether quantitative PET data are accurate in rats. Furthermore, we have found an extracranial accumulation of 2-[18F]fluoro-2-deoxy-
In the present study, we determined CMRglc by PET and conventional ex vivo techniques in the same rats and compared the values to examine the reliability of quantitative Rat–PET studies. We particularly assessed the influence of the extracranial radioactivity on PET-CMRglc measurements.
MATERIALS AND METHODS
We used male Sprague-Dawley rats weighing 273 to 335 g. All procedures were performed in accordance with institutional guidelines.
Animal studies
Rats assigned to middle cerebral artery (MCA) occlusion were anesthetized with 400 mg/kg body weight i.p. chloral hydrate. Right MCA was occluded using a method described previously (Minematsu et al., 1992; Kuge et al., 1995). The rats were permitted to recover from the anesthesia, and their neurological deficits were confirmed.
The PET studies were performed with a multislice PET (ECAT EXACT HR/47, Siemens/CTI, Knoxville, TN, U.S.A.) (Wienhard et al., 1994), which provided 47 tomographic images at 3.1-mm intervals per frame. The resolution at the center was 3.7 mm in-plane at full width at half maximum and 4.1 mm axially.
Before imaging, MCA-occluded (273–335 g, n = 8) and normal (285–307 g, n = 6) rats were prepared by venous and arterial catheterization under chloral hydrate anesthesia. Each rat was placed in the prone position, and the head was restrained by tooth and ear bars. The brain's position was standardized with the aid of laser beams. Anesthesia was maintained with 0.5 to 2.0% halothane.
To each rat 18F-FDG (18 to 48 MBq/kg body weight) synthesized by a standard procedure (Shiue et al., 1982) was administered via the venous catheter. In MCA-occluded rats, the average period from the MCA occlusion to the injection of 18F-FDG was 4.4 h (range, 2.5–5.7 h). Dynamic PET scanning (1 min × 5 frames, 5 min × 1 frame, 10 min × 2 frames, and 15 min × 1 frame) was initiated at the time of 18F-FDG injection. Arterial blood (<50 μl) was sampled intermittently, and plasma radioactivity and glucose concentration were determined.
Immediately after completion of the PET scanning, the rats were killed by an excess dose of i.v. pentobarbital and the brains dissected out and sectioned into eight blocks corresponding to the regions of interest (ROIs) on the PET images (see below). The radioactivity in the brain slices was measured with a well-type scintillation counter.
After the brains had been removed, a carcass of a normal rat was put back in the PET scanner, and scanning (20 min) was again performed. A PET scanning (30 min) was also done with another normal rat carcass whose brain, right eyeball, and right Harderian gland were removed.
Calculations
The PET images were reconstructed according to a standard filtered back-projection procedure using a Ramp filter. Semielliptical ROIs were placed on the left hemispheres of the four coronal brain slices at the levels of the frontal cortex (56 mm3), caudoputamen (127 mm3), hippocampus (170 mm3), and substantia nigra (188 mm3). The mirror image of each ROI was placed on the corresponding right hemisphere (Fig. 2). For the calculation of CMRglc, the radioactivity obtained 30 to 45 min postinjection was corrected for the recovery with recovery coefficients.
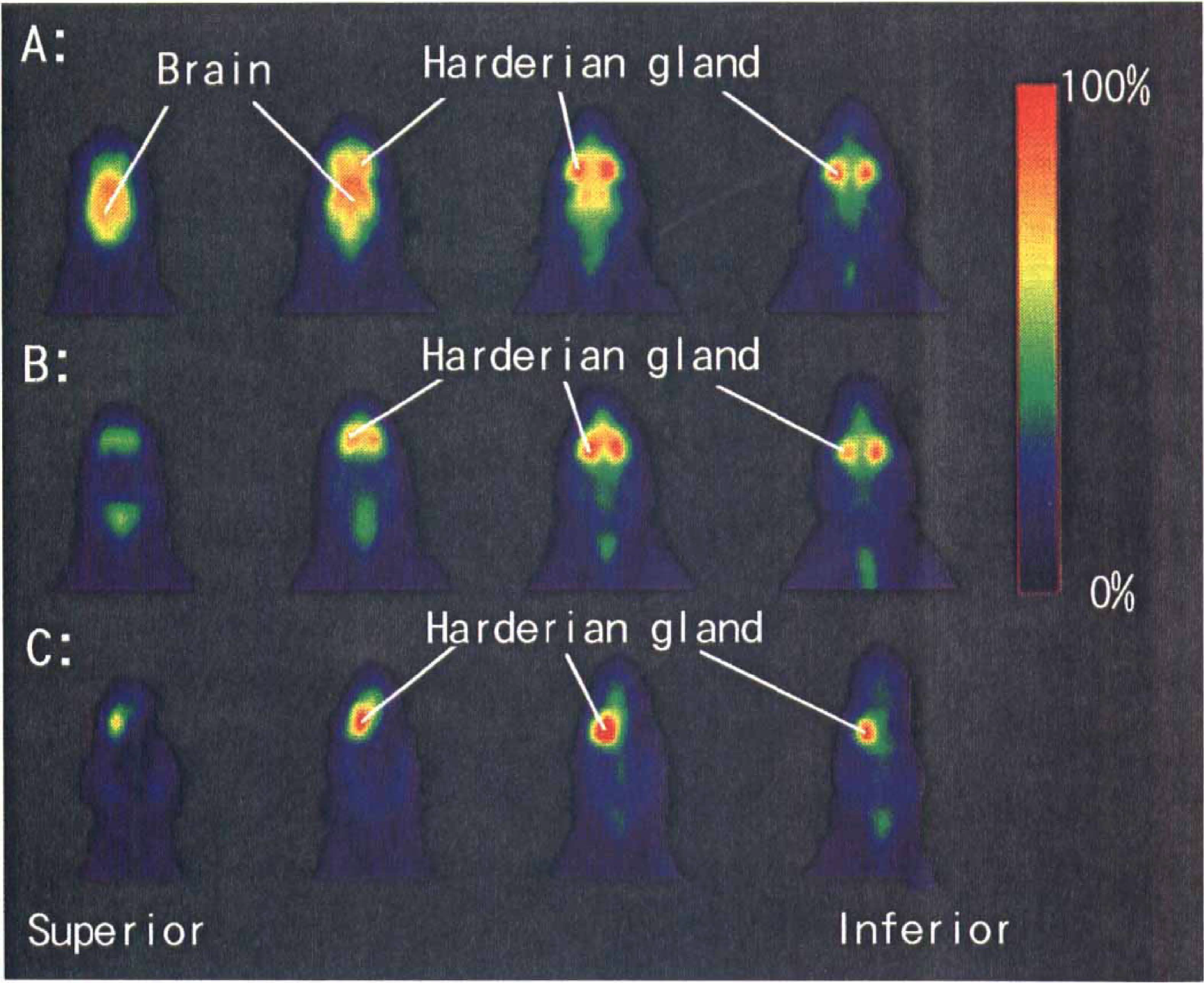
Positron emission tomography (PET) horizontal images of radioactivity in a rat obtained 30 to 45 min after the injection of 18F-FDG (
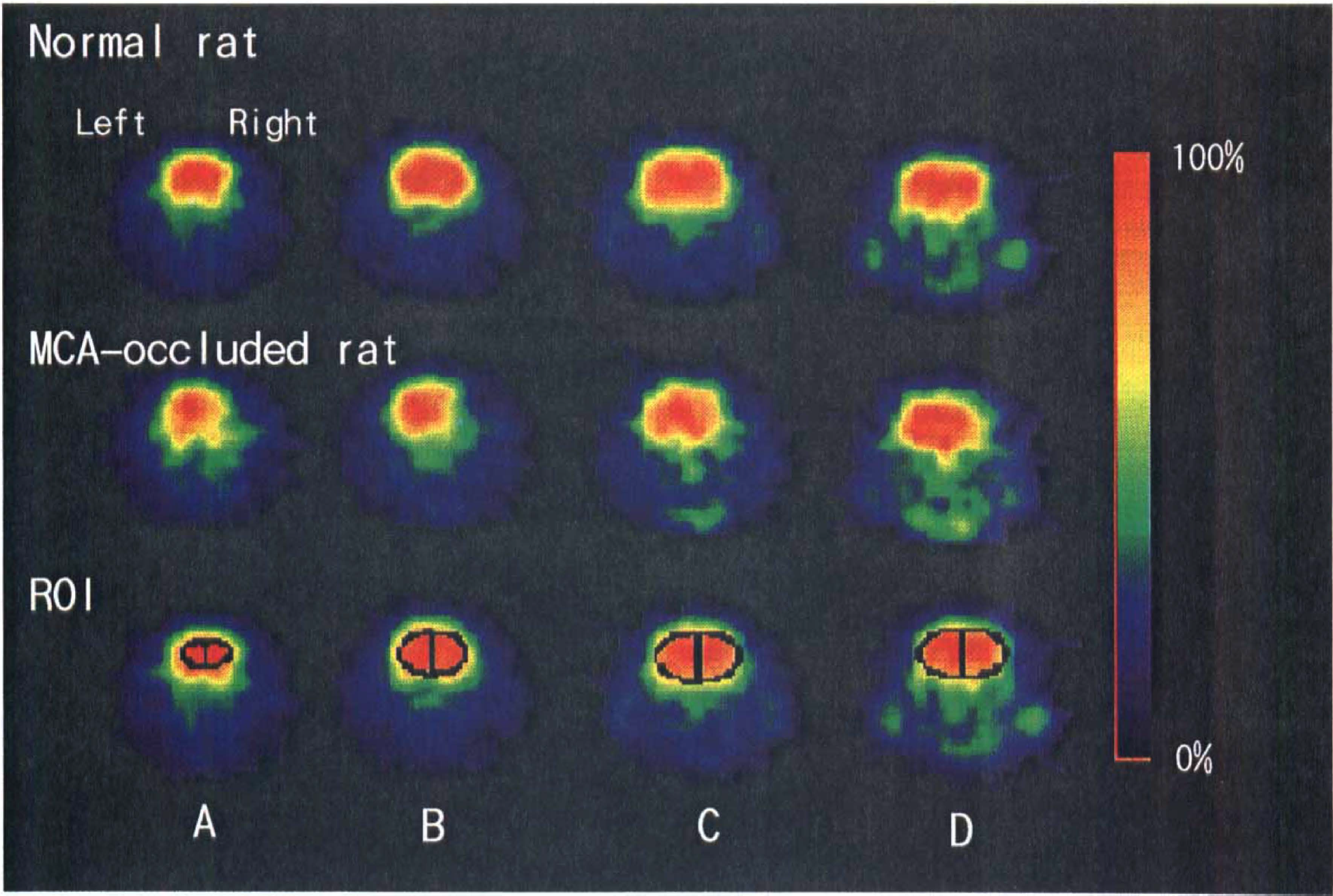
Positron emission tomography (PET) radioactivity images for four coronal slices of the brain in a normal (top row) and a MCA-occluded rat (middle row) obtained 30 to 45 min postinjection of 18F-FDG. Figures at the bottom row show where the ROIs are placed. The slices are at the frontal cortex level (
Recovery coefficients were determined using [18F]fluoride-solution in five cylindrical phantoms of diameters from 3 to 23 mm. Circular ROIs with the diameters as large as the phantoms were placed on a transverse image. PET radioactivity in each brain ROI was corrected by dividing it with the recovery coefficient corresponding to each ROI volume.
The CMRglc values were calculated according to the autoradiographic method (Sokoloff et al., 1977) in both PET and ex vivo experiments, adapting the values of 0.16 ml g–1 min–1, 0.32, and 0.1 min–1 for k1, k2, and k3, respectively, and the value of 0.60 for the lumped constant (Magata et al., 1995).
A paired t test was used to assess the significance of differences between the CMRglc values obtained by PET and those obtained by the ex vivo method, and an unpaired t test was used for comparison of ratios of CMRglc between the normal and ischemic brains. A two-tailed probability value <0.05 was considered significant.
RESULTS
Accumulations of radioactivity were detected in the brain and regions just anterior to the brain (Fig. 1A and B). The extracranial tissue with marked radioactivity was judged to be the Harderian glands because the radioactivity was markedly reduced in a carcass whose right Harderian gland was removed (Fig. 1C). As shown in Fig. 2, radioactivity was lower in the lesioned (right) than in the normal (left) hemisphere of the MCA-occluded rat.
Although good correlations (R2 = 0.929–0.998) were observed between the PET and ex vivo radioactivities in the normal and MCA-occluded brains, the PET-radioactivity in each hemispheric ROI was consistently lower than that obtained by the ex vivo method. The slopes of the regression lines ranged from 0.40 to 0.59.
The corrected CMRglcvalues obtained by PET were about twofold as high as those obtained by the ex vivo method at the level of the frontal cortex in both normal and MCA-occluded rats (Table 1). The increments in PET-CMRglc were 31 to 49% and 13 to 28% of those by the ex vivo method at the levels of the caudoputamen and hippocampus, respectively, and the differences were significant.
CMRglc obtained by PET and ex vivo methods in the normal and MCA-occluded rats a
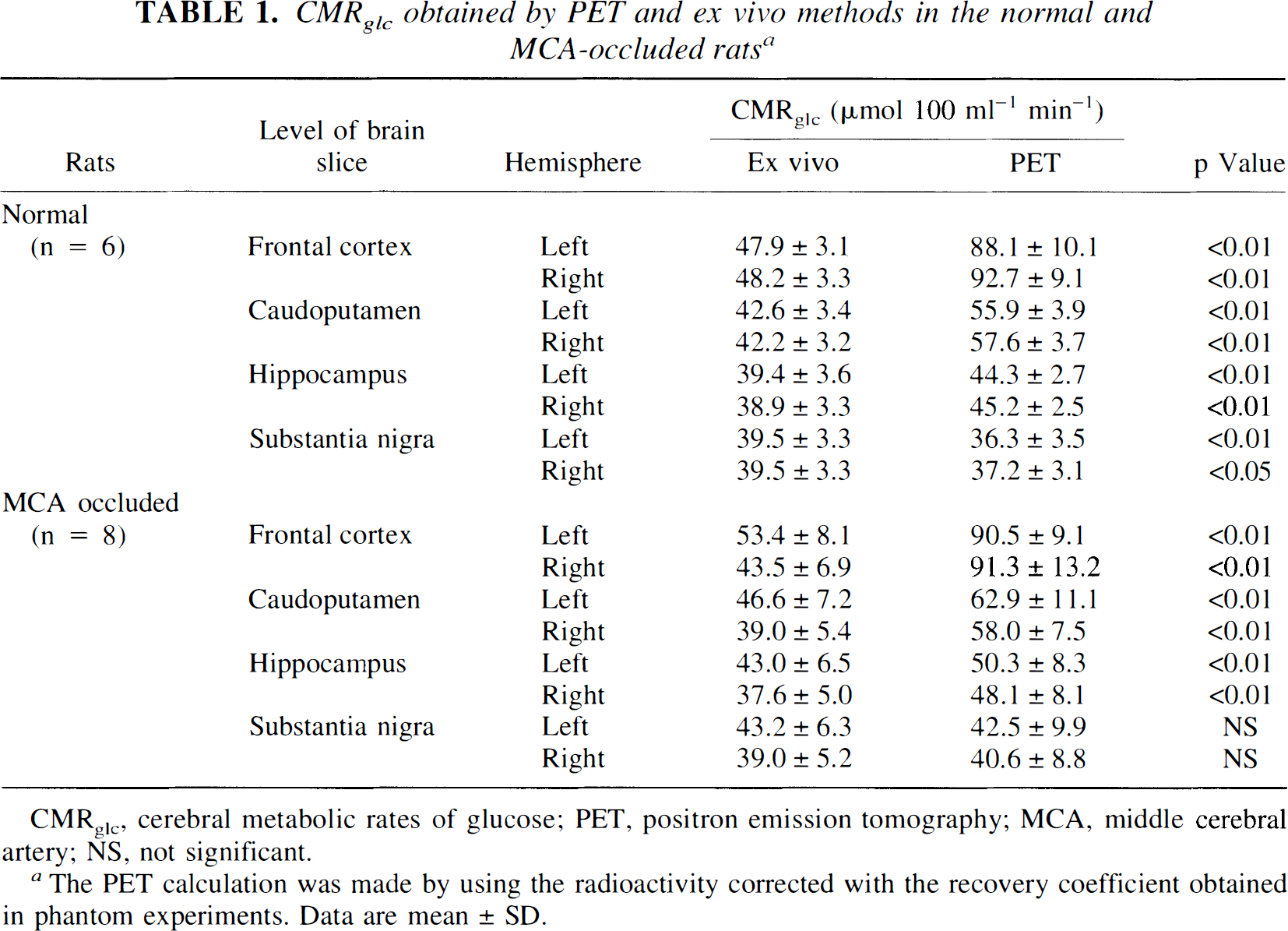
CMRglc, cerebral metabolic rates of glucose; PET, positron emission tomography; MCA, middle cerebral artery; NS, not significant.
The PET calculation was made by using the radioactivity corrected with the recovery coefficient obtained in phantom experiments. Data are mean ± SD.
The ratio of CMRglc in the lesioned to normal hemisphere was determined (Table 2). The ratio obtained by the ex vivo method was significantly lower in MCA-occluded rats than in normal rats at every slice (p < 0.01). Using the PET method, the ratios were significantly lower than those in normal rats at the caudal three levels (p < 0.05) but not at the level of the frontal cortex.
Ratios of CMRglc in the lesioned (right) to that in the normal (left) hemisphere
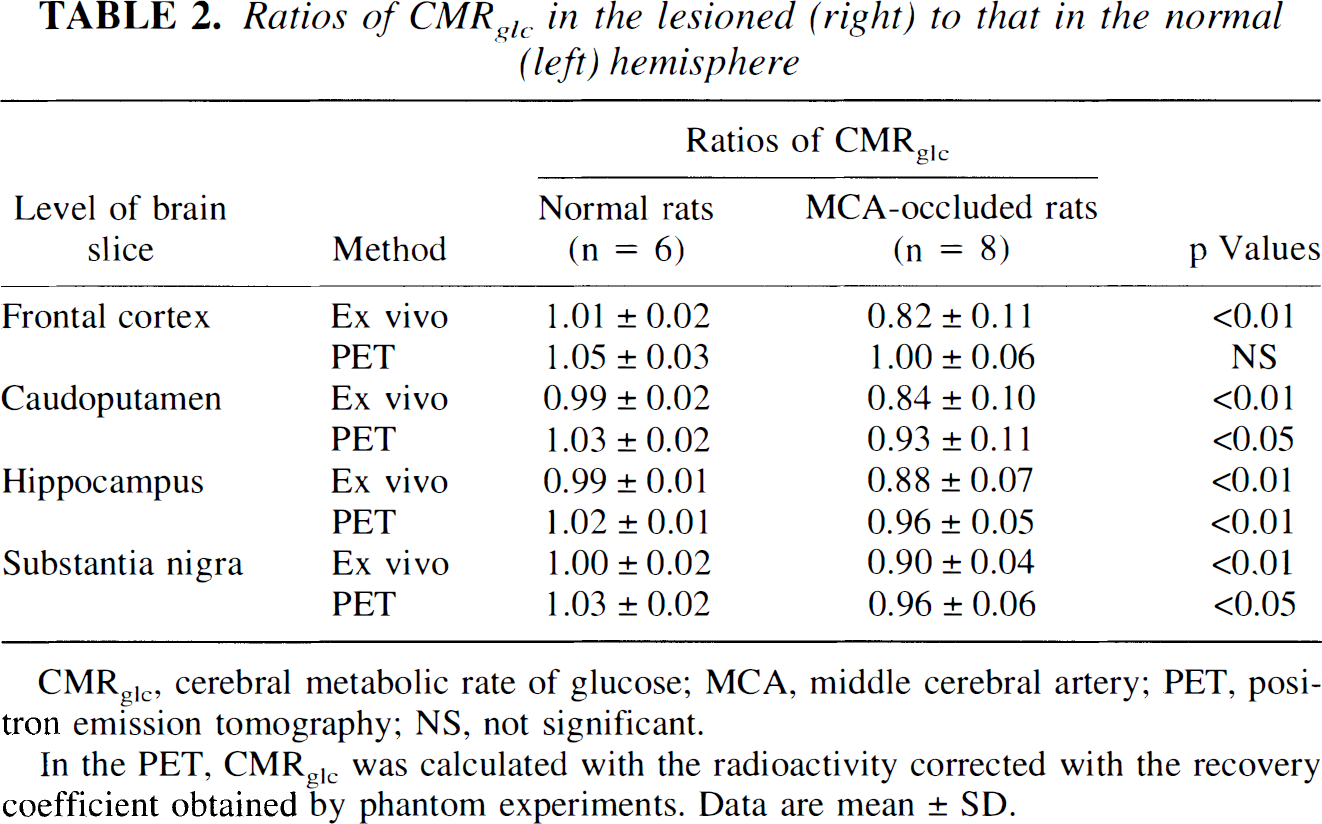
CMRglc, cerebral metabolic rate of glucose; MCA, middle cerebral artery; PET, positron emission tomography; NS, not significant.
In the PET, CMRglc was calculated with the radioactivity corrected with the recovery coefficient obtained by phantom experiments. Data are mean ± SD.
DISCUSSION
When the target tissue is small or heterogeneous, quantitative PET studies suffer from the problems that result from the limited resolution of the scanners (Hoffman and Phelps, 1986). The accumulation of radioactivity in the surrounding tissues makes it unfeasible to obtain quantitative values with PET. We demonstrated that 18F-FDG–derived radioactivity highly accumulated in the Harderian glands, located between the posterior part of the orbits and the frontal cortex (Fig. 1). Watanabe (1980) reported that in the mouse the Harderian glands have the highest radioactivity after administration of [14C]glucose compared with other organs. Although several authors (Brownell et al., 1991; Ingvar et al., 1991; Magata et al., 1995; Ouchi et al., 1996) have studied quantitative PET determination of CMRglc in rats, none mentioned the influence of radioactivity accumulated in the Harderian glands on the CMRglc measurements.
We found the radioactivity measured by the PET method to be consistently lower than that obtained by the ex vivo method, suggesting that a recovery correction is necessary. Most of the corrected CMRglc values were higher than those obtained by the ex vivo method (Table 1), suggesting the influence of the accumulated radioactivity in the surrounding tissues on the PET-CMRglc values. The discrepancies in CMRglc values between the PET and the ex vivo methods were larger at rostral levels than at caudal ones, indicating that the accumulated radioactivity in the Harderian glands disturbed the accuracy of PET values, mainly at the rostral levels. These differences may also be ascribed to the smaller ROI volumes at the rostral levels than at the caudal levels. In the frontal cortex, even the reduction of CMRglc in the ischemic brain was not demonstrated by PET (Table 2).
In conclusion, the influence of radioactivity accumulated in the Harderian glands on CMRglc determination could not be excluded even by using a high-resolution PET scanner. Quantitative PET determination of CMRglc is unreliable in rats because of the extracranial radioactivity and the limited resolution of scanners.
Footnotes
Acknowledgment:
This work was supported in part by a grant from the Research and Development Programs for Next-generation Spinhead Technologies of the Japan Health Science Foundation, Special Coordination Funds for Promoting Science and Technology (Encouragement System of COE) from the Science and Technology Agency of Japan, research grants for Cardiovascular Diseases 5A-5 and 6A-1 from the Ministry of Health and Welfare of Japan, and a research grant from the Japan Cardiovascular Research Foundation. We thank Dr. Marc Fisher (University of Massachusetts Medical School, Worcester, MA, U.S.A.) for reviewing the manuscript. We also thank Mr. Ejima Norimasa and Mr. Yamada Masanobu for technical assistance.
