Abstract
When used to measure blood flow, water leaves a residue in the vascular bed, which may contribute to the calculation of increased blood flow during functional activation of brain tissue. To assess the magnitude of this contribution with the two-compartment positron emission tomography (PET) method, we mapped the water clearance (K1) of the brain as an index of cerebral blood flow (CBF) and the apparent vascular distribution of nonextracted H215O (Vo). The latter map represented mainly the cerebral arterial and arteriolar volume. We also prepared subtraction maps (ΔK1, ΔVo) of the response to vibrotactile stimulation of the fingertips of the right hand of six normal volunteers. Using magnetic resonance (MR) images of all subjects, the data were rendered into Talairach's stereotaxic coordinates and the averaged subtraction images (activation minus baseline) merged with the corresponding averaged MRI image. The ΔK1 map revealed the expected response in the primary sensory hand area; the ΔVo response was located about 13 mm more anteriorly, close to the central fissure, most likely reflecting changes of the arteries feeding the primary sensory hand area. We conclude that cerebral perfusion and cerebrovascular responses to vibrotactile stimulation may occur in disparate locations that can be identified separately by using the two-compartment method.
The purpose of the present study is to reveal the contribution of vascular residues of water to the measured increases of blood flow in foci of functional activation of brain tissue. Positron emission tomography (PET) is a tool for the quantitative measurement of regional CBF. The i.v. H215O bolus method described by Herscovitch et al. (1983) and Raichle et al. (1983) is frequently used for that purpose. The underlying model is the single compartment initially proposed by Kety (1960) for ideally diffusible tracers. It is well known, however, that the diffusibility of water in the blood–brain barrier is limited. Kanno et al. (1984), Koeppe et al. (1987), and Ohta et al. (1996) demonstrated that a significant overestimation of the unidirectional water clearance from blood to brain, K1, occurs when the transmitted vascular component of H215O radioactivity is not taken into account. We therefore introduced a two-compartment model that gives a better description of the kinetics of water transit through cerebral tissue. This model accounts for vascular radioactivity and, in addition to the cerebral water clearance (K1), permits the imaging of the apparent vascular distribution volume (Vo) of the tracer, which is limited mainly to the arterial and arteriolar vascular tree (Ohta et al., 1996).
The measurement of regional CBF changes, most frequently using Kety's one-compartment model, is the basis of most blood flow activation studies presently being performed in a variety of fields such as physiology (Grafton et al., 1991; Zeki et al., 1991; Tempel and Perlmutter, 1992), cognitive neuroscience (Petersen et al., 1988; Cameron et al., 1990; Posner, 1993; Silbersweig et al., 1993), or psychiatry (Dolan et al., 1992; Friston et al., 1992). The primary interest in most of these studies is not in the magnitude of the blood flow change per se but rather in the location where this change occurs.
Although the observed blood flow or tissue perfusion changes are thought to reflect variations in neuronal synaptic activity (Roland et al., 1982) and, therefore, allow the identification of groups of neurons that are activated during a particular physiological or cognitive process, regional vascular changes (blood volume changes) may occur that reflect the hemodynamic adaptation of the cerebrovascular system to the redistribution of local energy demands. The locations where these vascular responses occur might well differ from the sites of perfusional changes, and the understanding of the exact nature and complexity of these responses is therefore essential to the correct interpretation of blood flow activation studies.
The parameters of water uptake identified by the two-compartment method include the clearance, K1, and the apparent vascular distribution, Vo. The clearance is an index of the perfusion of the tissue because it depends primarily on the blood flow. The present experiment was designed to identify separately foci of perfusional and vascular responses to unilateral vibrotactile stimulation of the fingertips. The two-compartment method is computationally sufficiently robust and efficient to generate a cerebral perfusion map (K1 map) as well as a map of the vascular distribution of the tracer (Vo map), which forms a part of the cerebral vascular volume, from a single PET study. With a one-compartment method, the two responses are confounded and are not separately distinguishable (in the present paper, we denoted this confounded blood flow value calculated with Kety's one-compartment method by the symbol F′). Finally, because both the perfusion and the vascular responses are obtained in the same PET study (same tracer injection), the method does not suffer from artefacts related to changes in equipment, movement of the subject, or changes of physiological state.
The simultaneous estimation of parameters affects the precision with which each can be determined. This particularly affects the estimates of Vo; however, the magnitudes and changes of Vo recorded in this study were subjected to rigorous statistical evaluation, including statistical analysis of the significance of the peaks of changes in Vo. In no instance was there reasonable doubt of the presence or absence of a change in Vo.
METHODS
H215O PET study
Six healthy right-handed volunteers were selected for this study after completion of a questionnaire that ruled out any apparent neurological deficits. Informed consent was obtained from each participant, and the protocol had been approved by the Research Ethics Committee of the Montreal Neurological Institute and Hospital.
The subjects were positioned in the tomograph with their heads immobilized by means of a customized foam headrest (Evans et al., 1988). A short in-dwelling catheter was placed in the left radial artery for blood sampling.
The tracer H215O was prepared with a Japanese Steel Works Medical Cyclotron (BC-107). The PET studies were performed on the Scanditronix PC-2048 15B, an 8-ring, 15-slice bismuth germanate (BGO) head tomograph with a transverse resolution of 5.8 to 6.4 mm and an axial resolution of 6.1 to 7.1 mm full-width at half maximum (FWHM). Its count rate efficiency was 9.7 Kcps/(μCi · ml–1) for the direct and 13.7 Kcps/(μCi · ml–1) for the cross slices (Evans et al., 1991). Reconstruction software included corrections for random and scattered events, detector efficiency variations, and dead time. An orbiting rod transmission source containing about 5 mCi of 68Ge was used for attenuation correction. For image-averaging purposes, a 20-mm FWHM Hanning filter was used during image reconstruction.
About 40 mCi of H215O were injected as a bolus into the antecubital vein of the right arm. Arterial blood sampling and dynamic imaging began at injection time. Dynamic scans with frame durations of 12 × 5 s, 6 × 10 s, and 3 × 20 s were obtained over 3 min and corrected for decay according to the method of Raichle et al. (1983). Arterial blood samples were collected manually at about 5-s intervals and assayed in a well counter, which was calibrated with respect to the tomograph. The delay in tracer arrival between the brain and the peripheral sampling site was corrected according to the global-fitting method of Iida et al. (1988). For dispersion correction, a time constant of 4 s was assumed (Iida et al., 1986; Meyer, 1989). The two-compartment model of Ohta et al. (1996), which accounts for intravascular radioactivity, was used for the generation of regional K1 maps as opposed to F′ maps based on Kety's one-compartment model. The two-compartment model at the same time provided regional maps of Vo, the initial distribution volume of the tracer. From the functional PET data volume, axial, sagittal, and coronal views were generated by three-dimensional (3-D) compositing and reformatting.
Vibrotactile stimulation was performed with a mechanical vibrator (Model 91, Daito, Osaka, Japan) fixed to the five fingertips of the right hand and held in place with tape to maintain a uniform pressure throughout the stimulation period. A vibration frequency of 110 Hz was chosen, and the vibrator was intermittently turned on and off at 1-s intervals (1 s on, 1 s off) to avoid adaptation (Applegate et al., 1988). The nominal vibration amplitude was 2 mm. The subjects were asked to focus their attention on the vibrotactile stimulus to maximize the change in CBF, which is known to be reduced by distraction (Meyer et al., 1991). Vibrotactile stimulation was initiated 30 s before the start of PET scanning and blood sampling and was continued throughout the 3-min scanning period.
All subjects also underwent a magnetic resonance imaging (MRI) examination on a Philips Gyroscan, 1.5 Tesla superconducting magnet system. The MRI data volume consisted of 64 contiguous slices of 2-mm thickness.
Data analysis
For analysis, each PET image dataset was first co-registered with its corresponding MRI image volume using a 3-D landmark-matching algorithm (Evans et al., 1989, 1991). The parametric images, calculated from the raw PET activity data, were projected into Talairach's stereotaxic coordinate space (Talairach and Tournoux, 1988) using the MRI volume for identification of the bicommissural plane (Evans et al., 1992). For each subject, subtraction images were obtained by subtracting baseline PET images from activation PET images after normalization of each scan with respect to its mean whole-brain value. Those subtraction images were then averaged across all subjects and analyzed according to the method of Worsley et al. (1992). This analysis yielded a mean percentage change image of the parameters under investigation (K1 and Vo) (for the purpose of comparison, subtraction images for F′ were generated as well). From that, a t statistic image was calculated by dividing the average change image by the average (pooled) standard deviation across voxels to allow identification of significant peaks in the subtraction image. Merging of the t statistic image with the mean stereotaxic MRI image yielded the activation peaks superimposed on the anatomical regions. Use of the mean MRI image, which is blurred by residual anatomical variability among subjects after stereotaxic transformation, provides a truer measure of the extent of compatibility of the individual subject's neuroanatomy than simple use of the idealized Talairach template. For a directed search in the cortex of the left postcentral gyrus which, in Talairach space, occupies approximately 24 cm3, activation peaks for the K1 and V0 subtraction images were considered statistically significant at p < 0.05 for t > 3.3 (Worsley et al., 1992). The stereotaxic coordinates of the significant K1, Vo, and F′ peaks were determined from both the averaged subtraction images and individual subtraction images. The latter was possible because of the strong primary sensory stimulus used in this experiment, which elicited K1, Vo and F′ responses above the noise level of individual subtraction images.
CT angiogram
To compare the appearance of the Vo maps with the anatomical location of major arteries, or groupings of smaller arteries, finely reconstructed CT images with a 1.5-mm slice thickness were taken from a single cadaver brain and composited into a 3-D angiogram. These images were obtained from postmortem scans in which the blood had been replaced with contrast agent (20% BaSO4 and 80% latex). The CT images were also transformed into stereotaxic space by identifying a series of landmarks in both angiogram and the average MRI volume so they could be compared with the PET and MRI images.
RESULTS
Figure 1 compares the CT angiographic images (left column) with the merged Vo PET images (right column) of the baseline scan, averaged over the six subjects after normalization with respect to the whole brain. The axial, coronal, and sagittal sections of corresponding CT and PET images were obtained at the same levels as indicated by the Talairach coordinates x, y, and z (in parentheses). Although the spatial resolution of the Vo PET images is significantly worse than that of the CT images as a result of the choice of a 20-mm FWHM reconstruction filter for averaging purposes, their high activity regions clearly correspond to areas of major arteries, including peripheral anterior cerebral arteries, such as the cingulate artery. Figure 1 therefore lends strong support to the claim that the vascular component (Vo) of the two-compartment model of water uptake predominantly represents the arterial vascular volume.
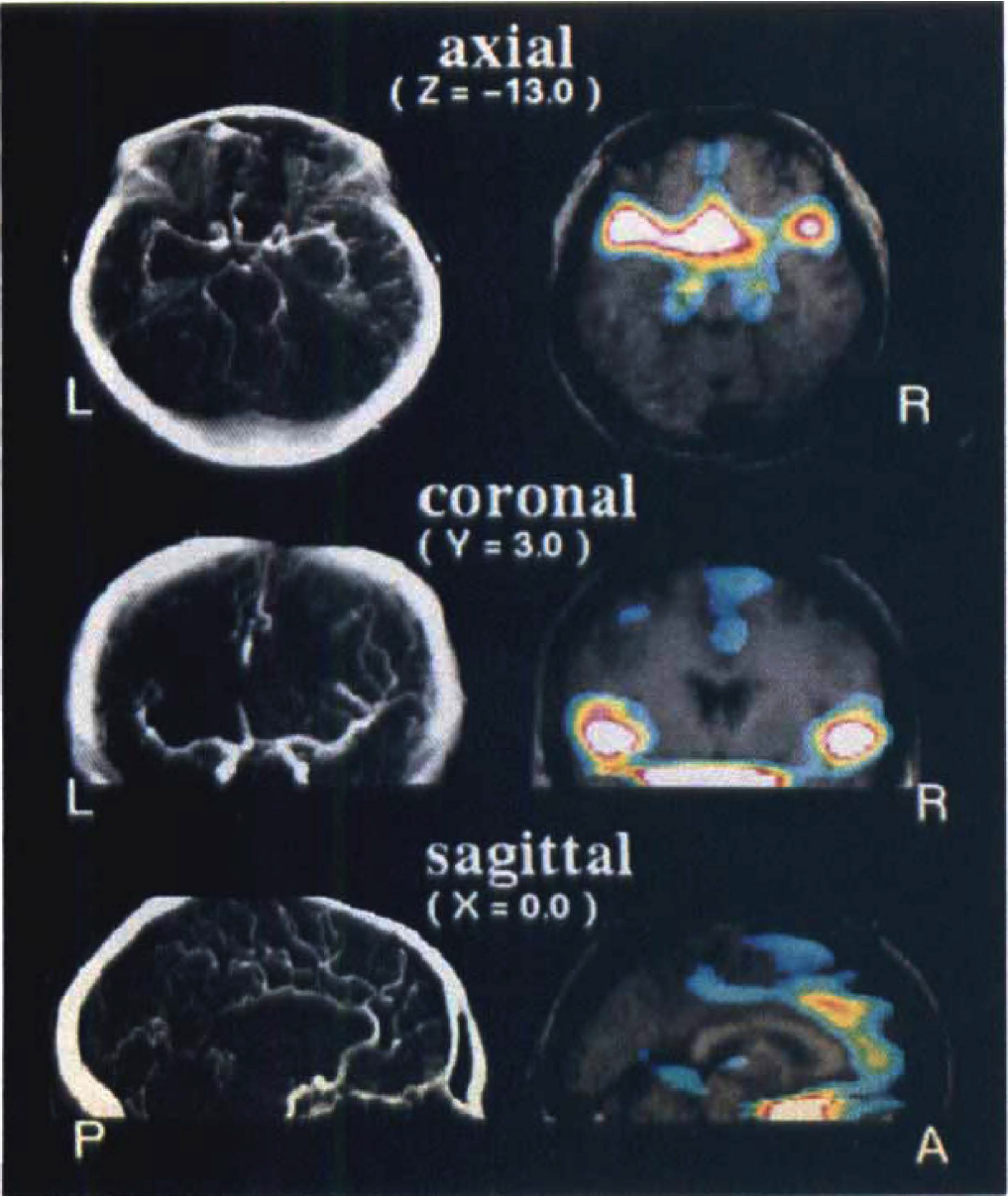
Comparison of computed tomography (CT) angiographic images obtained from a postmortem scan after replacing the blood with a contrast enhancing agent (20% BaSO4 and 80% latex) (left column) with averaged baseline Vo positron emission tomography (RET) images [20-mm full-width at half maximum (FWHM) reconstruction filter] superimposed on corresponding averaged MRI images (right column). The axial, coronal, and sagittal image planes of corresponding CT and PET images have the same Talairach coordinates x, y, and z (mm). The CT study was obtained from a single subject. Note the striking correspondence between the appearance of the Vo PET images and the arterial vascular tree displayed on the CT angiographic images.
The primary somatosensory response to the vibrotactile stimulus is shown in Fig. 2 in the form of t statistic maps. The focal perfusion change in the sensory hand area (SI) (ΔK1) and the focus of the associated, primarily arterial, vascular response (ΔVo), depicted together with their stereotaxic coordinates, are both statistically significant (t = 4.7 for ΔK1 and t = 3.3 for ΔVo for a directed search of the volume of the cortex of the left postcentral gyrus). Careful inspection of Fig. 2 further suggests that the vascular change (ΔVo) is located slightly more anteriorly and laterally than the perfusion change (ΔK1). The regional blood flow change derived from the one-compartment method (ΔF′) resulted in the most significant peak (t = 6.9). Its location was almost identical to that of the ΔK1 focus but, as expected, slightly displaced toward the ΔVo focus.
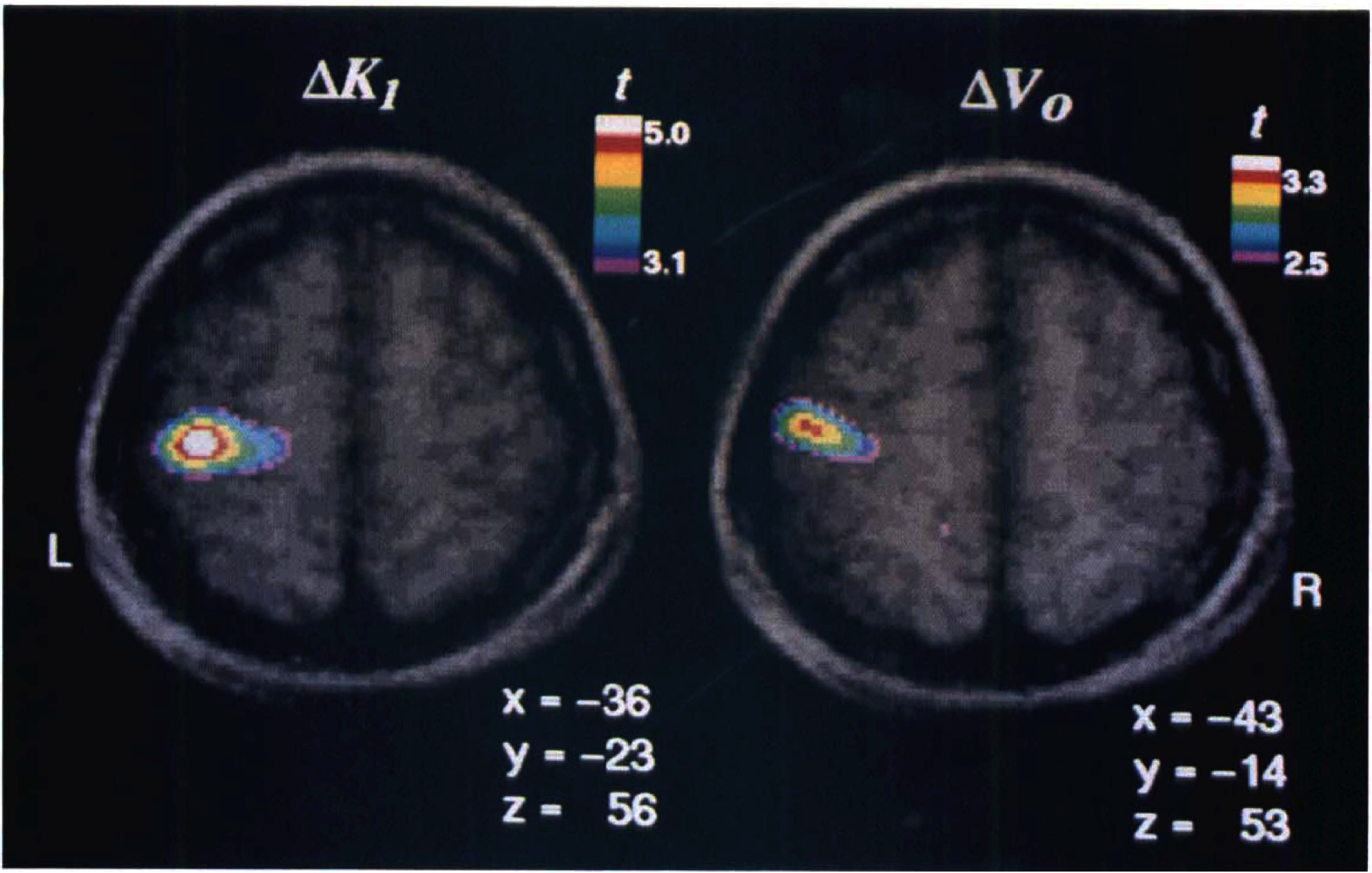
Averaged t statistic images superimposed on corresponding averaged MRI images. Both the ΔK1 and ΔVo images show a statistically significant activation focus in or close to the sensory hand area (SI). The two images have different t scales. The peak locations are characterized by their Talairach coordinates x (left–right), y (anterior–posterior) and z (rostral–caudal) in millimeters. Note the slightly more anterior and lateral position of the ΔVo focus relative to the ΔK1 focus.
These observations are further confirmed in Tables 1 and 2. Table 1 gives the individual Talairach coordinates of the three foci for each of the six cases studied. Table 2 shows the distance (in Talairach coordinates) between the three activation foci. Paired Student's t tests, performed separately between the x-, y-, and z coordinates of the ΔK1 and ΔVo foci, revealed a significant difference in the y coordinate (t = 3.86, df = 5, p < 0.02) but none for the other two (p > 0.1). In addition, a Hotelling's t2 test for a difference in all three dimensions proved to be just significant (t2 = 27.8, df = 3,3 and 0.05 < p < 0.1), indicating a significant spatial separation between these two foci. The coordinate difference between the ΔK1 and ΔF′ foci, on the other hand, was not statistically significant.
Coordinates of ΔF′, ΔK1, and ΔVo peaks
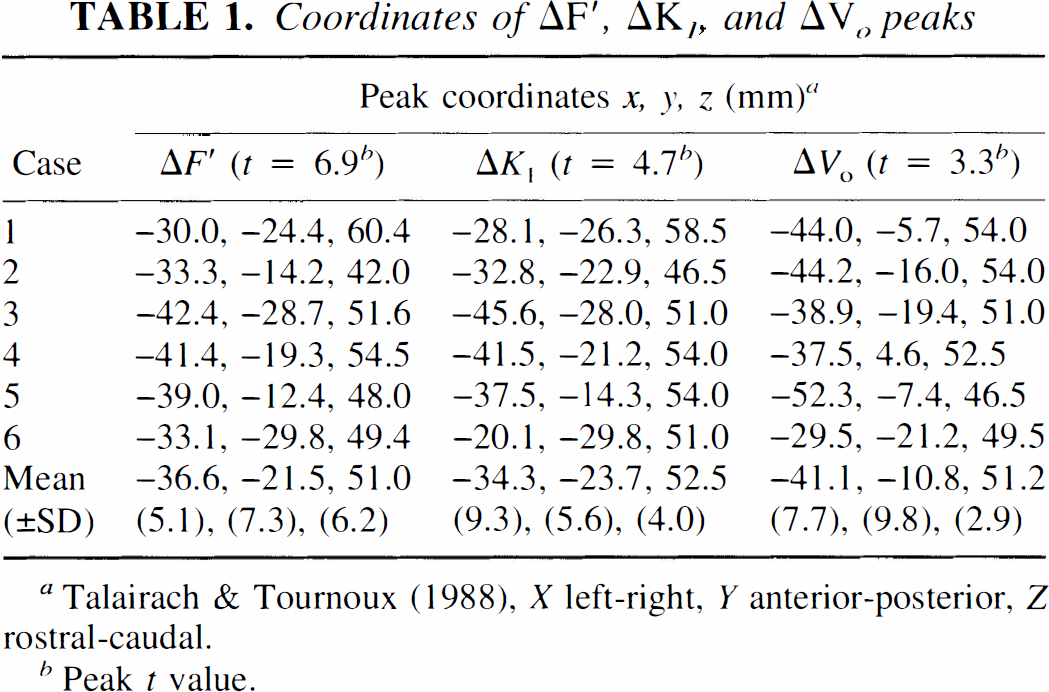
Talairach & Tournoux (1988), X left-right, Y anterior-posterior, Z rostral-caudal.
Peak t value.
Distances between ΔF′, ΔK1, and ΔVo peaks
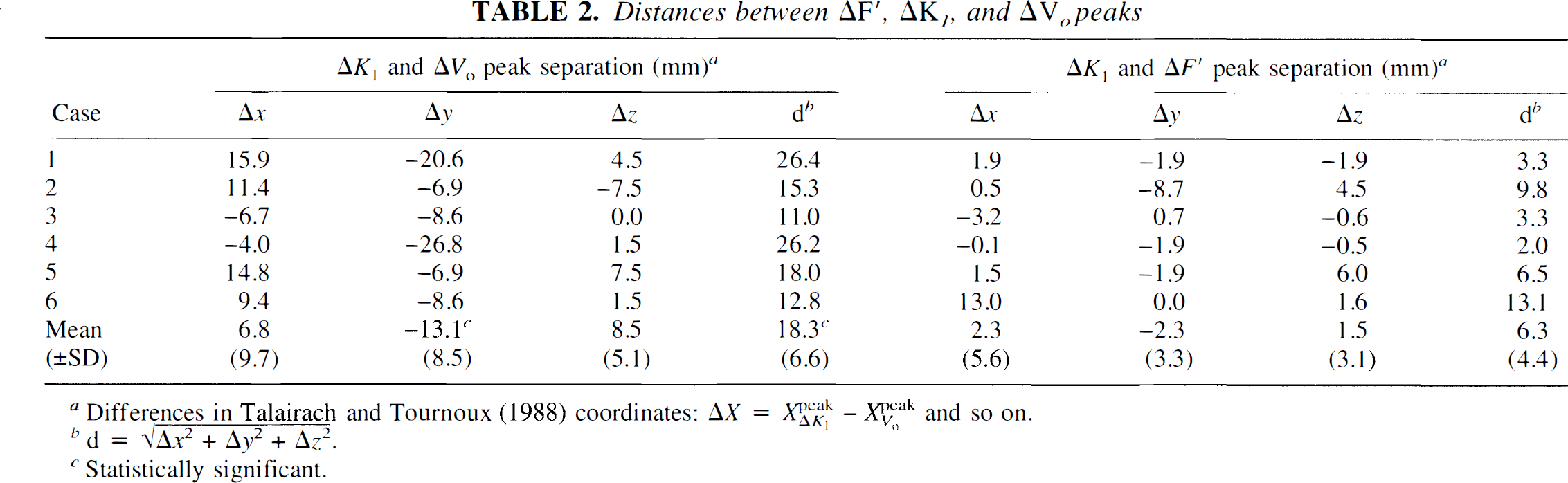
Differences in Talairach and Tournoux (1988) coordinates: 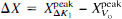 and so on.
and so on.
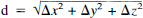 .
.
Statistically significant.
DISCUSSION
The two major findings described in this paper are (1) the observation that the known CBF or tissue perfusion response (K1) to vibrotactile stimulation of the fingertips is accompanied by a vascular response (Vo) mainly representing arteries, arterioles, and part of the capillary bed and (2) the suggestion that the locations of these two associated responses are different; that is, the stereotaxic coordinates of the ΔK1, peak are significantly different from those of the ΔVo peak.
The two-compartment method used here was described in a previous paper (Ohta et al., 1996). The properties of this model that allow us to determine simultaneously the tissue perfusion (K1) and initial distribution volume of the tracer (Vo) make the method particularly suitable for the investigation of the vascular contribution.
Because the apparent vascular distribution, Vo, is the important additional parameter derived from our model, we were particularly interested in identifying its properties. One may think of Vo in terms of the fraction of tracer that passes through the vasculature of a given tissue element without being extracted. This fraction therefore preserves its arterial concentration, Ca(t). For a tracer like C15O with a vanishingly small extraction fraction, Vo reflects the entire vascular volume (CBV). On the other hand, for a tracer with a relatively large first-pass extraction fraction, such as H215O (E0 ∼ 0.95), Vo is confined mainly to arteries, arterioles, and a small fraction of the capillary bed (Fujita et al., 1993). This conclusion finds strong qualitative support from the comparison of the postmortem computed tomography (CT) scan with contrast infusion and the averaged Vo baseline maps (Fig. 1). It should be noted that the CT scan is from a single cadaver, whereas the PET maps are averages from six normal subjects and are reconstructed with a relatively flat filter to account for residual intersubject variability after transformation into Talairach's stereotaxic space. Nevertheless, with this difference in spatial resolution in mind, the similarity between the two imaging modalities is striking. This comparison strongly suggests that the high activity regions of the Vo maps correspond to major arterial structures, such as the internal carotid arteries, anterior cerebral artery, bilateral middle cerebral arteries, and even the posterior cerebral artery.
Individual maps of the distribution volume are at present still beset with a considerable amount of uncertainty resulting from the strong sensitivity of Vo to tracer dispersion and delay effects. In the present study, tracer dispersion and delay were corrected for in a global manner; that is, a single fixed dispersion time constant of τ = 4 s was used, and individual delay corrections were applied to each of the 15 imaging planes using the methods of Iida et al. (1986, 1988). More regionalized methods for these two corrections are required to obtain reliable quantitative Vo maps. Based on the evidence discussed above, the averaged Vo maps used in this study faithfully reflect the transmitted tracer fraction. As a consequence, the introduction of Vo in the model provides a valid means of accounting for intravascular radioactivity. Therefore, the cerebral water clearance (K1), the other parameter provided by the model, is a more accurate index of tissue perfusion than the cerebral blood flow values (F′) obtained with the traditional one-compartment model.
We used the two-compartment model to analyze data from a blood flow activation PET study with vibrotactile stimulation of the fingertips of the right hand. As expected, a strong perfusion response (ΔK1) was found in the sensory hand area. Perhaps somewhat more surprising, although not completely unexpected, was the observation of a significant vascular response (ΔVo) in approximately the same area. There are two explanations that immediately present themselves. Either the transmitted tracer fraction, Vo, increases during vibrotactile stimulation, that is E0 decreases or Vo increases because of an increase in the arterial, arteriolar, and part of the capillary volume during the activation phase. The former explanation is difficult to sustain in the presence of the observed increase in water clearance, K1, during activation. We are therefore left with the suggestion that the observed increase in the initial distribution volume of H215O reflects a vascular volume increase in response to vibrotactile stimulation. If we adhere to the general belief that cerebral capillaries may be considered rigid, undistensible tubes (Mchedlishvili et al., 1987), this would mean that the vascular volume change would have to be attributed mainly to the arterioles and feeding arteries or to an increase in capillary density, a variable determined by Gjedde et al. (1990) and corroborated by Kuwabara et al. (1992), who showed a parallel increase of capillary density and cerebral blood flow during vibrotactile stimulation. Recent reports by Atkinson et al. (1990) and Duelli and Kuschinsky (1993) suggest that cerebral capillaries may in fact be distensible and that part of the observed volume increase may be the result of an increase in capillary volume in proximity to the activated brain region. The increase of Vo during vibrotactile stimulation most likely is due to a combination of the above mechanisms.
The study also identified disparate locations of the tissue perfusion response (ΔK1) and the vascular response (ΔV0). On visual inspection (Fig. 2), the ΔK1 peak is located clearly in the postcentral gyrus, whereas the ΔV0 peak appears more anteriorly and laterally, possibly coinciding with the central sulcus. Quantitatively (see Table 2), the difference between the y components (anterior-posterior) of the stereotaxic coordinates of the two peaks was statistically significant (13 mm) and so was the difference in all three dimensions (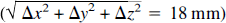 ) (see Results). The figures from individual subtraction images for each of the six subjects demonstrate the same trend (Table 1). Because the K1 and the V0 maps were obtained simultaneously from single PET scans, patient movement or other methodological artefacts can be ruled out. Therefore, we judge the observed difference in peak location to be real. A possible explanation for this observation might be as follows. The tracer, H215O, is transferred by the blood stream via the internal carotid and middle cerebral artery from the heart to the site of activation (i.e., the site of increased tissue perfusion), and the ΔV0 peak reflects a response of the arterial vascular bed located closer to the heart than the ΔK1 peak. In other words, the changes of ΔVo may arise before the capillary branching of the artery and arterioles that supply the activated area.
) (see Results). The figures from individual subtraction images for each of the six subjects demonstrate the same trend (Table 1). Because the K1 and the V0 maps were obtained simultaneously from single PET scans, patient movement or other methodological artefacts can be ruled out. Therefore, we judge the observed difference in peak location to be real. A possible explanation for this observation might be as follows. The tracer, H215O, is transferred by the blood stream via the internal carotid and middle cerebral artery from the heart to the site of activation (i.e., the site of increased tissue perfusion), and the ΔV0 peak reflects a response of the arterial vascular bed located closer to the heart than the ΔK1 peak. In other words, the changes of ΔVo may arise before the capillary branching of the artery and arterioles that supply the activated area.
It is important to realize that the two responses observed with the two-compartment method are confounded with the traditional one-compartment approach, which may result in a blurring of the observed peaks, depending on the spatial separation of the perfusion and vascular responses. A one-compartment analysis of the present study yielded a single strong focus (ΔF′) representing both perfusion (K1) and vascular (Vo) responses. It was therefore located in the space between the vascular and perfusion foci, obtained with the two-compartment method, in close proximity of the latter focus, reflecting the difference in magnitude of the two responses (tk1 > tv0). In other words, the magnitudes of cerebral blood flow changes (ΔF′) measured with the one-compartment model alone do not reflect changes in tissue perfusion because they receive contributions from a purely vascular change. We suggest that the use of our two-compartment method permits a more realistic interpretation of blood flow activation results. The method provides not only more accurate values for pure tissue perfusion, but the simultaneously derived vascular data permits additional insight into the processes underlying the relationship between cerebral blood flow and blood volume.
Footnotes
Acknowledgment:
This work was supported by MRC (Canada) grants PG-41 and SP-30, the Quebec Heart Foundation, and the Isaac Walton Killam Fellowship Fund of the Montreal Neurological Institute. We thank Dr. K. J. Worsley from the Department of Mathematics and Statistics, McGill University, for his help with the statistical analysis, Dr. G. D. Partlow, presently at the Department of Biomedical Sciences, University of Guelph, Ontario, Canada, for providing the computed tomography angiographic data and the technical staff of the McConnell Brain Imaging Centre for their assistance. Special thanks to Dr. Leonardo Ribeiro, Dr. Yoshifumi Yasuhara, Mr. Sean Marrett, and Mr. Manouchehr Vafaee for their help with the PET studies and to Mr. Peter Neelin, Mr. Sylvain Milot, and Ms. Paule-Joanne Toussaint for providing expert computer analysis support.
