Abstract
The intravenous infusion of rat adrenomedullin, at concentrations ranging from 0.1 to 1.0 μg/kg/min, for 60 min increased the regional cerebral blood flow (rCBF) in a dose-dependent manner in rats. rCBF was measured using a laser Doppler flowmetry device placed on the surface of the parietal cortex. The increase in rCBF induced by 1.0 μg/kg/min of adrenomedullin was up to 145 ± 10.8% of controls at 60 min (n = 5, p < 0.001). These concentrations of adrenomedullin did not affect systemic blood pressure or other physiologic parameters, including pH, PaCO2, PaO2, hemoglobin, and blood glucose. Repeated infusion of 1.0 μg/kg/min of adrenomedullin at 2-h intervals caused tachyphylaxis (n = 5, p < 0.01). Rat adrenomedullin (1.0 μg/kg/min) demonstrated a more potent effect than the same dose of human adrenomedullin. The C-terminal fragment of human adrenomedullin (0.5 and 5.0 μg/kg/min), adrenomedullin22–52, which did not affect rCBF alone, inhibited the effect of rat adrenomedullin (0.5 μg/kg/min) as a receptor antagonist in a dose-dependent manner. In a model of middle cerebral artery (MCA) occlusion in spontaneously hypertensive rats, pre- and postinfusion of 1.0 μg/kg/min of adrenomedullin suppressed the reduction in rCBF following MCA occlusion (control, 29 ± 15.1%; adrenomedullin group, 45 ± 14.4%; not significant) and decreased the volume of ischemic brain injury (control, 288 ± 35 mm3; adrenomedullin group, 232 ± 35 mm3; p < 0.05). These results suggest that adrenomedullin increases rCBF and prevents ischemic brain injury, partly by increasing the collateral circulation.
Keywords
Adrenomedullin is a new vasoactive peptide that was first isolated, purified, and sequenced from human pheochromocytoma (Kitamura et al., 1993a). This 52-amino-acid peptide has one intramolecular disulfide bond, indicating similarities with calcitonin gene–related peptide (CGRP), and induces cyclic AMP accumulation in various tissues. Adrenomedullin mRNA has been shown to be expressed mainly in the peripheral tissues, including the heart, lungs, kidneys, aorta, spleen, duodenum, and submandibular glands, and to a lesser extent in the central nervous system (Kitamura et al., 1993b; Ichiki et al., 1994). However, recent studies have shown that high concentrations of adrenomedullin are detected in many regions of human brain, with the highest concentrations in the thalamus and hypothalamus, and is also expressed in such pathological conditions as neoplasma or ischemic injury (Satoh et al., 1995; Wang et al., 1995). It has been demonstrated that the binding sites for adrenomedullin are localized with immunoreactive adrenomedullin in the same tissues (Owji et al., 1995).
It is known that the exogenous application of adrenomedullin elicits potent, long-lasting hypotension and vasodilation in the vascular system. Sugo et al. (1994a, b ) have reported that cultured vascular endothelial cells and smooth-muscle cells produce and secrete adrenomedullin and that its mRNA is constitutively expressed in both. This peptide may represent a new member of the group of endothelium-derived relaxing factors, owing to its actions at receptors positively coupled to cAMP formation in both endothelial and smooth-muscle cells.
Previous reports have stated that adrenomedullin as well as CGRP exert vasodilator activity in the cerebral circulation in vitro, in situ, and in vivo (Suzuki et al., 1989; Suzuki, 1993; Baskaya et al., 1995; Wang et al., 1995). This vasodilator activity of adrenomedullin was inhibited by CGRP8–37, an antagonist of CGRP, but was unaffected by NG-nitro-L-arginine methylester (L-NAME) or indomethacin administration in dogs (Baskaya et al., 1995; Wang et al., 1995). However, the direct evidence of the effect of adrenomedullin on the regional cerebral blood flow (rCBF) has not been available. The present investigation was therefore undertaken to study the effect of adrenomedullin on rCBF measured via a laser Doppler flowmeter device in rats. We also investigated the effect of the intravenous infusion of adrenomedullin on the volume of ischemic brain injury in a model of middle cerebral artery (MCA) occlusion in spontaneously hypertensive rats (SHRs).
METHODS
Preparation of animals
All protocols and surgical procedures were approved by the Institute for Laboratory Animal Research, Nagoya University School of Medicine. For this study, we used adult, male Sprague–Dawley (SD) rats, weighing 300–400 g and SHRs, weighing 250–300 g. Anesthesia consisted of 2% halothane for induction and 1.2% halothane plus 50% nitrous oxide–50% oxygen for maintenance. Animals were ventilated mechanically with a rodent ventilator (SN-480-7; Shinano, Tokyo, Japan) through an endotracheal tube (polyethylene tubing, PE-240). The respiratory volume and rate were adjusted to maintain the Pa
Regional CBF measurement
Changes of rCBF were recorded at the surface of the left parietal cortex using laser Doppler flowmeter probes attached to a laser flowmeter device (ALF 21R; Advance Co., Tokyo, Japan). After rats were placed in a stereotaxic frame, craniectomy (0.5 mm in diameter, 4–6 mm lateral and 1–2 mm caudal to the bregma) was performed with extreme care over the MCA territory, using a surgical blade to gently shave the bone. The dura was left intact. The probe of the laser Doppler flowmeter was advanced steadily, perpendicular to the cortical surface, using a micromanipulator (Z-1; Narishige, Tokyo, Japan). Care was taken to avoid placing the probe above the pial arteries and veins; although it touched, it did not indent the dura mater. To facilitate optimal laser Doppler flowmeter recording, the dura was kept moist with warmed saline. Changes in rCBF were expressed as the percentage from baseline rCBF assessed before drug administration.
Focal ischemia study
The left MCA and ipsilateral common carotid artery (CCA) of SHRs were occluded according to a modification of the Brint method, as previously described (Brint et al., 1988; Koketsu et al., 1992). Briefly, after the left cervical common carotid artery had been exposed and a 3-0 silk suture had been positioned under it for the purpose of ligation following MCA occlusion, a 1-cm skin incision was made at the midpoint between the left lateral canthus and anterior pinna in animals in the lateral position. A 3-mm craniectomy was performed at the junction of the zygoma and squamosal bone, and a 10-0 nylon suture was passed beneath the left MCA (just above the rhinal fissure) in preparation for ligation. Animals were then placed in a stereotaxic frame (SR-5; Narishige). A 3-mm burr hole was drilled with care over the cortical barrel fields (AP: 0–3.0 mm, ML: 4.0–7.0 mm). The rCBF was recorded on the surface of exposed dura. After stabilization, adrenomedullin or vehicle was infused intravenously for 60 min; then the left MCA was ligated, followed by ipsilateral CCA occlusion. The rCBF was monitored continuously for ≥2 h before ischemia and 2 h following ischemia.
Experimental protocol
A 1.0-ml volume of physiologic saline was used as a vehicle solution. Peptides were dissolved in the same amount of physiologic saline immediately before use. After a 1-h period of stabilization and assessment of the baseline rCBF, physiologic saline or the test solution was continuously infused intravenously. Rat adrenomedullin was injected at four different concentrations (0.05, 0.1, 0.5, and 1.0 μg/kg/min). Human adrenomedullin was used at doses of 0.5 and 1.0 μg/kg/min. To investigate the effect of human adrenomedullin22–52, a receptor antagonist, on the changes in rCBF induced by rat adrenomedullin, 0.5 and 5.0 μg/kg/min of human adrenomedullin22_52 were administered simultaneously with 0.5 μg/kg/min of rat adrenomedullin. In the rat MCA occlusion model, an intravenous infusion of 1 μg/kg/min of rat adrenomedullin was initiated 1 h before the ischemic event and was subsequently maintained for 1 h following MCA occlusion.
Measurement of the volume of ischemic brain injury
Animals were killed 24 h after the ischemia experiments. Their brains were quickly removed, placed in cold saline solution for 10 min, and then cut into 7 × 2-mm coronal slices in a rodent brain matrix (RBM 4000C; Activational Systems, Warren, MI, U.S.A.). Sections were stained with 2% TTC (2,3,5-triphenyltetrazolium chloride monohydrate) as previously described (Bederson et al., 1986; Koketsu et al., 1992). The volume of ischemic brain injury was measured using a Macintosh computer employing the public domain NIH Image program (written by Wayne Rasband at the U.S. National Institutes of Health and available from the Internet by anonymous ftp from zippy.nimh.nih.gov or a floppy disk from NTIS, 5285 Port Royal Rd., Springfield, VA 22161, part number PB93-504868). The volume of ischemic brain injury was calculated by the numeric integration of data from individual slices.
Materials
Rat adrenomedullin, human adrenomedullin, and human adrenomedullin22–52 were obtained from Peptide Institute (Osaka, Japan). TTC was obtained from Katayama Chemicals Co. (Nagoya, Japan). All other chemicals were reagent grade.
Statistical analysis
Data are expressed as the mean ± SD and were analyzed by a repeated-measures analysis of variance (ANOVA) followed by contrasts in repeated measures design and Student's t test (Super ANOVA; Abacus, CA, U.S.A.). A p value of <0.05 was considered statistically significant.
RESULTS
rCBF responses to rat adrenomedullin and human adrenomedullin
The intravenous infusion of 1.0 ml of saline for 60 min produced no significant change in rCBF (102 ± 7.6% of baseline, n = 5). The administration of 0.05 μg/kg/min of rat adrenomedullin for 60 min also did not result in a statistically significant change in rCBF compared with values obtained following saline infusion. However, the administration of 0.1, 0.5, and 1.0 μg/kg/min of rat adrenomedullin increased the rCBF in a dose-dependent manner (Fig. 1). There were no significant changes in pH, Pa
Physiological parameters in the cerebral blood flow study in SD rats and cerebral ischemia study in SHRs a
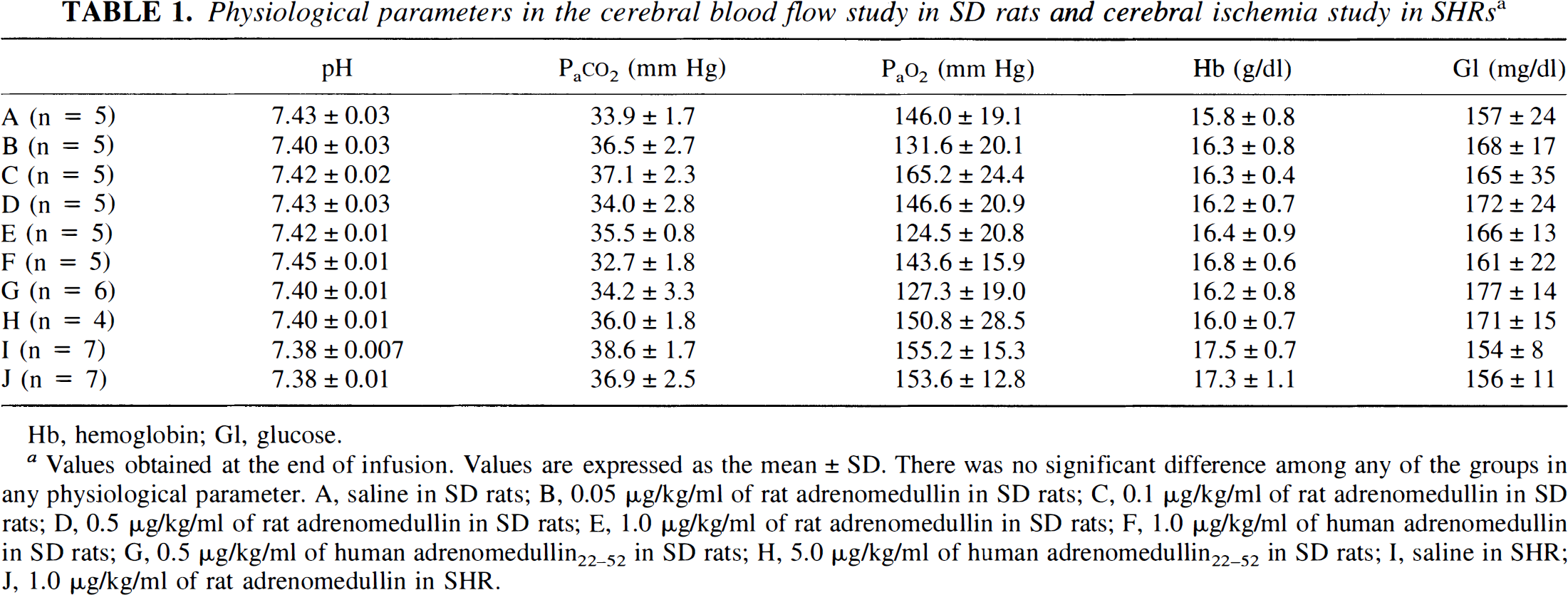
Hb, hemoglobin; Gl, glucose.
Values obtained at the end of infusion. Values are expressed as the mean ± SD. There was no significant difference among any of the groups in any physiological parameter. A, saline in SD rats; B, 0.05 μg/kg/ml of rat adrenomedullin in SD rats; C, 0.1 μg/kg/ml of rat adrenomedullin in SD rats; D, 0.5 μg/kg/ml of rat adrenomedullin in SD rats; E, 1.0 μg/kg/ml of rat adrenomedullin in SD rats; F, 1.0 μg/kg/ml of human adrenomedullin in SD rats; G, 0.5 μg/kg/ml of human adrenomedullin22–52 in SD rats; H, 5.0 μg/kg/ml of human adrenomedullin22–52 in SD rats; I, saline in SHR; J, 1.0 μg/kg/ml of rat adrenomedullin in SHR.
Mean arterial blood pressure (MABP) values in cerebral blood flow study in SD rats a

Preinf-MABP, mean arterial blood pressure just before starting the infusion; postinf-MABP, mean arterial blood pressure at the end of infusion.
Values are expressed as the mean ± SD. There was no significant difference among any of the groups. A, saline; B, 0.05 μg/kg/ml of rat adrenomedullin; C, 0.1 μg/kg/ml of rat adrenomedullin; D, 0.5 μg/kg/ml of rat adrenomedullin; E, 1.0 μg/kg/ml of rat adrenomedullin; F, 1.0 μg/kg/ml of human adrenomedullin; G, 0.5 μg/kg/ml of human adrenomedullin22–52; H, 5.0 μg/kg/ml of human adrenomedullin22–52.
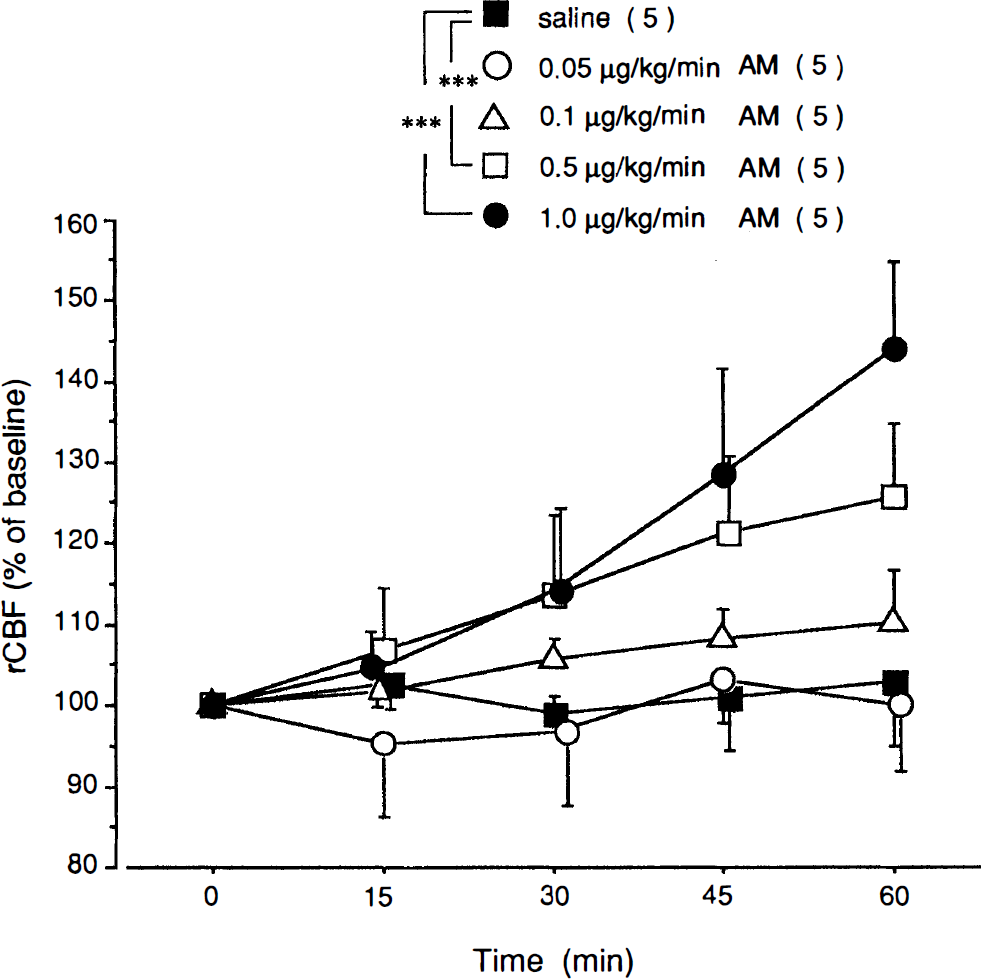
Effects of the intravenous administration of vehicle (saline, 1 ml/h) and four different concentrations of rat adrenomedullin (AM; 0.05, 0.1, 0.5, and 1.0 μg/kg/min) on regional cerebral blood flow (rCBF) in Sprague–Dawley rats. Changes in rCBF are expressed as a percentage of the baseline rCBF. Increases in rCBF in groups receiving 0.5 and 1.0 μg/kg/min of rat adrenomedullin were significantly greater than those in the group receiving saline, after 45 and 60 min of infusion. Values are means ± SD. The number of animals is indicated in parentheses. ***p < 0.001 versus control group by contrasts in a repeated-measures analysis of variance.
To investigate if rat adrenomedullin causes tachyphylaxis, we readministered 1.0 μg/kg/min of rat adrenomedullin 2 h after the end of the first infusion. This second infusion of rat adrenomedullin demonstrated a significantly decreased effect on rCBF compared with that of the first infusion (n = 5, p < 0.01). The intravenous infusion of human adrenomedullin at a dose of 1.0 μg/kg/min for 60 min produced less of an increase in rCBF compared with the same dose of rat adrenomedullin (Fig. 2). A significant difference existed between these two groups 60 min and beyond the start of the infusions. In the group of rats receiving human adrenomedullin, rCBF returned to values similar to baseline within 30 min of the end of the infusion. In the group receiving rat adrenomedullin, however, rCBF remained above baseline for an extended period (125 ± 6.2% of baseline at 90 min).
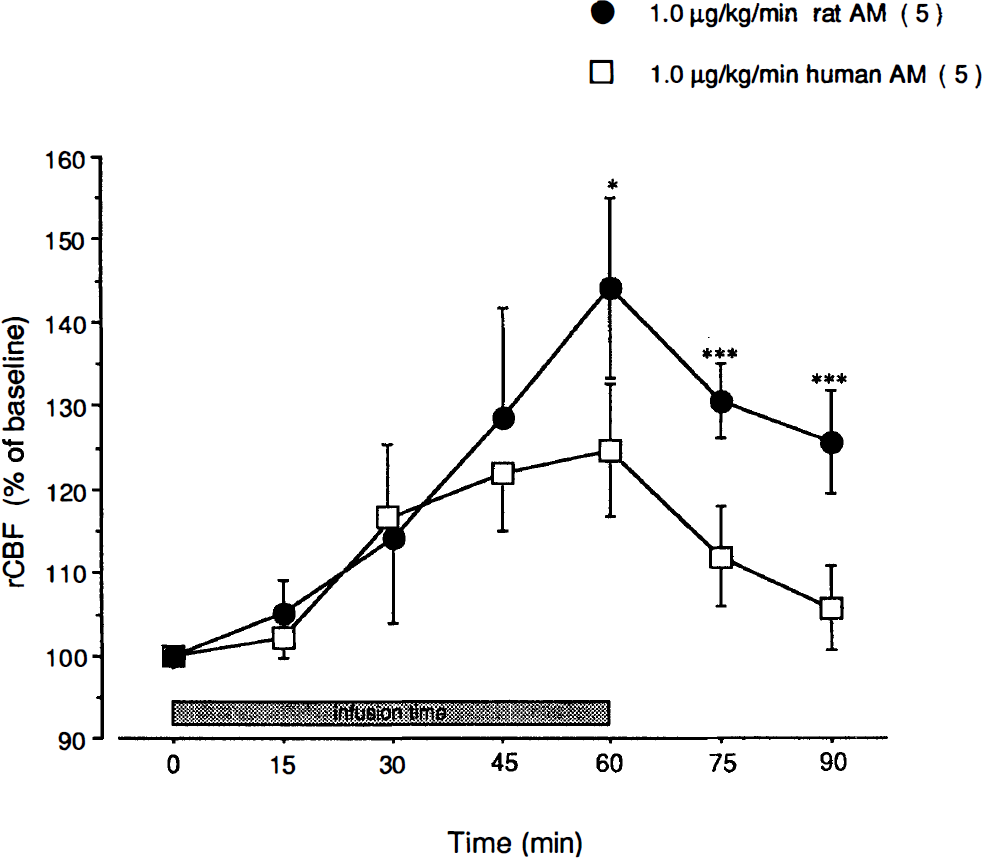
Comparison of the effects of the intravenous infusion of human adrenomedullin and rat adrenomedullin on regional cerebral blood flow (rCBF) in Sprague–Dawley rats. Changes in rCBF are expressed as a percentage of the baseline rCBF. The intravenous infusion of human adrenomedullin at a dose of 1.0 μg/kg/min for 60 min produced less of an increase in rCBF than the same dose of rat adrenomedullin. A significant difference between these two groups existed after 60 min of infusion. Values are means ± SD. The number of animals is indicated in parentheses. *p < 0.05; ***p < 0.01.
Effect of human adrenomedullin22–52 on rCBF responses to rat adrenomedullin
Administration of the C-terminal fragment of human adrenomedullin, human adrenomedullin22–52, did not cause a significant change in rCBF from baseline. However, the simultaneous administration of human adrenomedullin22–52 (0.5 and 5.0 μg/kg/min) and rat adrenomedullin (0.5 μg/kg/min) suppressed the increase in rCBF in response to rat adrenomedullin in a dose-dependent manner (Fig. 3).
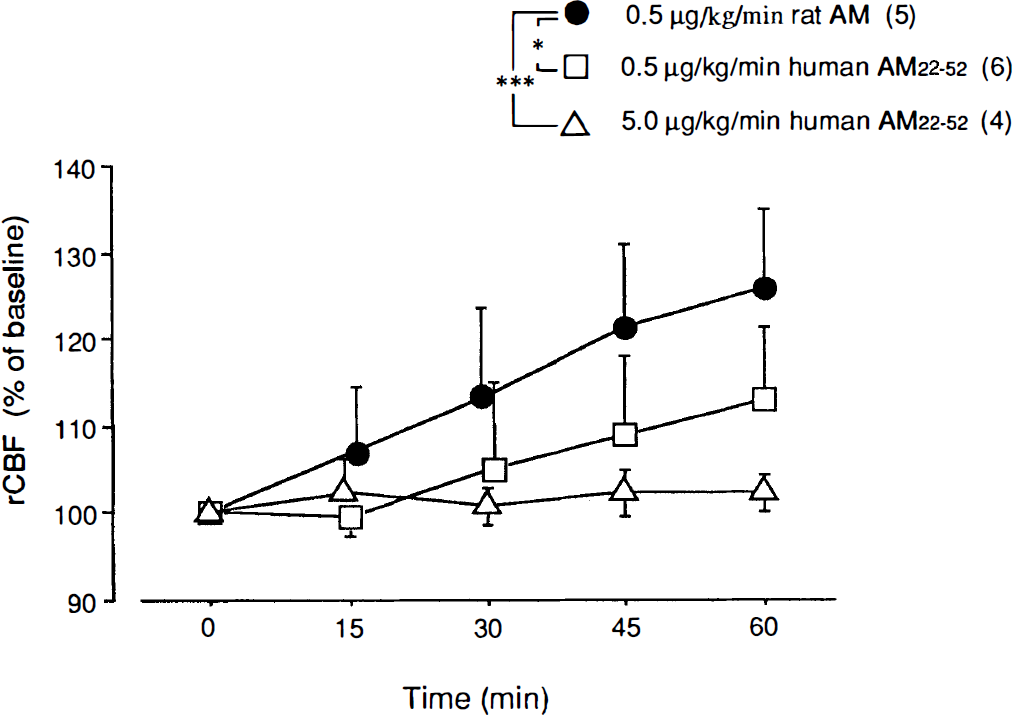
Effects of human adrenomedullin22–52 on the increased regional cerebral blood flow (rCBF) induced by rat adrenomedullin. Changes in rCBF are expressed as a percentage of the baseline rCBF. Increases in rCBF induced by 0.5 μg/kg/min of rat adrenomedullin (rat AM) were significantly suppressed in a dose-dependent manner when simultaneously administered with 0, 5, and 5.0 μg/kg/min of human adrenomedullin22–52. Values are expressed as the mean ± SD. The number of animals is indicated in parentheses. * p < 0.05 and *** p < 0.001 versus the rat adrenomedullin group by contrasts in a repeated-measures analysis of variance.
Effect of adrenomedullin on rCBF in the MCA occlusion model
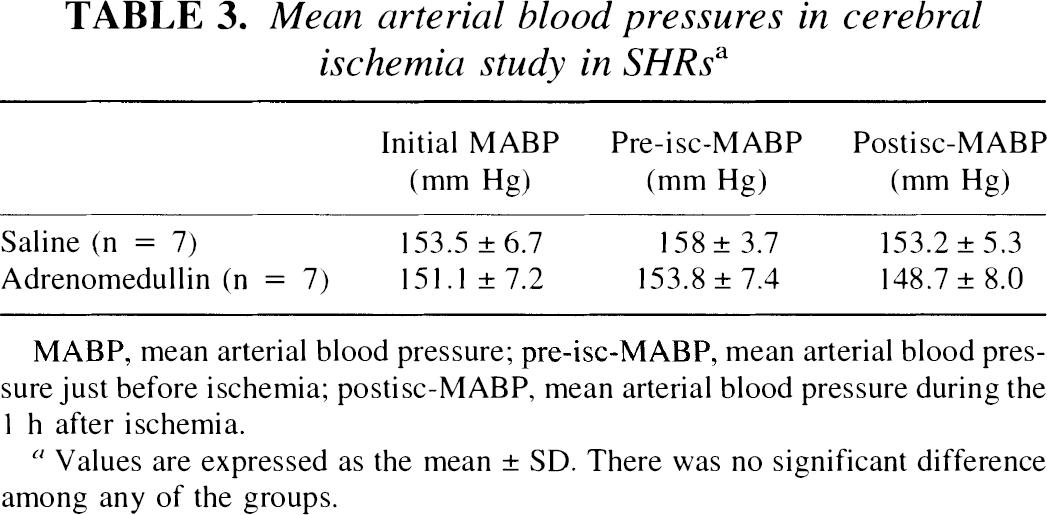
In the MCA occlusion model in SHRs, preinfusion of 1.0 μg/kg/min of rat adrenomedullin increased rCBF in the ischemic hemisphere to 118 ± 6.1% of baseline. After MCA occlusion, rCBF fell to 23 ± 11.3% of baseline in the ischemic hemisphere of the control rats. In the pretreated group, occlusion reduced rCBF to 35 ± 17.3% of baseline in the ischemic hemisphere. At the end of infusion (after 60 min of ischemia), rCBF values were 29 ± 15.1% and 45 ± 14.4% in the control and adrenomedullin groups, respectively (Fig. 4), which did not reflect a significant difference. Infusion of adrenomedullin in the MCA occlusion model did not affect the mean arterial blood pressure (Table 3).
Mean arterial blood pressures in cerebral ischemia study in SHRs a
MABP, mean arterial blood pressure; pre-isc-MABP, mean arterial blood pressure just before ischemia; postisc-MABP, mean arterial blood pressure during the 1 h after ischemia.
Values are expressed as the mean ± SD. There was no significant difference among any of the groups.
Effect of rat adrenomedullin on regional cerebral blood flow (rCBF) in the middle cerebral artery (MCA) occlusion model in spontaneously hypertensive rats. Changes in rCBF are expressed as a percentage of the baseline rCBF. Infusion of 1 ml/h of saline (control group) or 1 μg/kg/min of rat adrenomedullin (pretreated group) was started 1 h before and continued throughout 1 h of left MCA occlusion. In rats that received saline, rCBF remained essentially stable throughout the pre-ischemic infusion period (104 ± 5.5% of baseline); the intravenous infusion of AM produced a valuable increase in rCBF throughout the same period (118 ± 6.1% of baseline). During the ischemic period, in rats that received adrenomedullin (AM), the rCBF appeared to be maintained at a higher level than that in the saline (control) group, although a significant difference did not exist. Values are means ± SD. The number of animals is indicated in parentheses. *p < 0.05 versus control group.
Effect of adrenomedullin on ischemic brain injury volume
In SHRs, the MCA occlusion model induced a highly reproducible ischemic brain injury that affected both the cortex and striatum. The total volume of ischemic brain injury, assessed from TTC-stained brain slices, was 288 ± 35 mm3 in the control SHR group (n = 7). Treatment with 1.0 μg/kg/min of adrenomedullin for 60 min before occlusion and for 60 min following occlusion reduced the ischemic brain injury volume to 232 ± 35 mm3 (n = 7). A statistically significant difference existed between the two groups (p < 0.05). Thus, the total volume of ischemic brain injury was reduced by 19.5% with adrenomedullin treatment (Fig. 5).
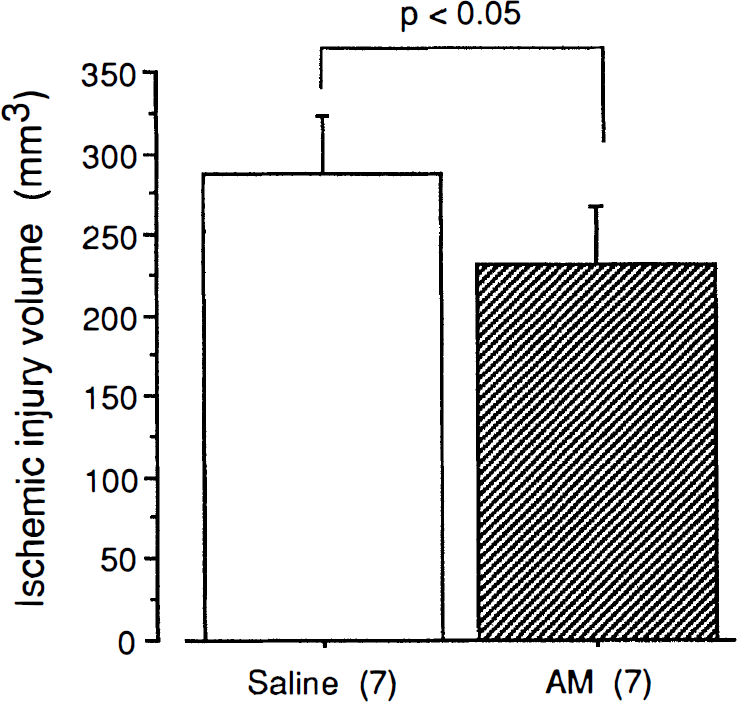
The effect of rat adrenomedullin (AM) on the volume of ischemic brain injury after 24 h of MCA occlusion in SHRs. Values are means ± SD. The number of animals is indicated in parentheses. *p < 0.05 versus control group.
DISCUSSION
This study has shown that the intravenous administration of rat adrenomedullin produces a dose-dependent increase in rCBF in anesthetized SD rats without affecting systemic blood pressure or blood gas values. The maximum increase in rCBF induced by adrenomedullin was ∼45%. In a previous study, we have shown that this peptide elicits an increase in vertebral artery blood flow and vasodilation in the major cerebral arteries on angiography in dogs in vivo (Baskaya et al., 1995). Vasodilator responses to adrenomedullin in isolated dog pial arteries (Baskaya et al., 1995; Wang et al., 1995), rat pial arteries in situ (Wang et al., 1995), and rat intracerebral arterioles (unpublished data) also have indicated the potent vasoactive effect of adrenomedullin on cerebral blood vessels. The evidence obtained in the present study supports the idea that adrenomedullin plays an important role as a vasoactive peptide in the control of CBF.
The increase in rCBF induced by adrenomedullin was inhibited in a dose-dependent manner by the simultaneous administration of the C-terminal fragment of adrenomedullin, adrenomedullin22–52, which lacks the common cyclic structure. It also has been demonstrated that the increase in vertebral artery blood flow caused by adrenomedullin and the vasodilator effect of adrenomedullin on the isolated basilar artery in dogs can be inhibited by an antagonist of the calcitonin gene–related peptide receptor, CGRP8–37 (Baskaya et al., 1995; Wang et al., 1995). These results suggest that adrenomedullin interacts with the common receptors of CGRP, a homologous peptide, in vascular smooth-muscle cells. A study with iodine 125–labeled rat adrenomedullin has established the presence of a single class of high-affinity binding sites for adrenomedullin in cultured vascular smooth-muscle cells from rat thoracic aorta (Eguchi et al., 1994). Furthermore, CGRP was shown to interact with these adrenomedullin receptors.
In contrast with the receptors in vascular smooth-muscle cells, those in endothelial cells seem to be more specific for adrenomedullin, since iodine 125–labeled adrenomedullin binding was not displaced by CGRP in bovine aortic endothelial cells (Shimekake et al., 1995). The vasodilator effects of adrenomedullin, therefore, may be dependent on at least two mechanisms. The first involves a direct action of adrenomedullin on vascular smooth-muscle cells mediated via receptors in which binding is coupled to the accumulation of intracellular cyclic AMP (Ishizaki et al., 1994). The second mechanism involves an indirect action of adrenomedullin on endothelial cells mediated through receptors in which binding is coupled to the release of nitric oxide, by increasing intracellular Ca2+ concentrations or activating phospholipase C, in addition to the accumulation of intracellular cyclic AMP (Shimekake et al., 1995).
The stimulating activity of adrenomedullin on the release of nitric oxide from endothelial cells has been confirmed in rat models using hindquarter vascular beds and isolated perfused kidneys (Feng et al., 1994; Hirata et al., 1995). The increase in nitric oxide stimulated by adrenomedullin was abolished by the application of the nitric oxide synthase inhibitors L-NAME (Feng et al., 1994) and NG-monomethyl-L-arginine (Hirata et al., 1995). However, pretreatment with L-NAME did not suppress the increase in vertebral artery blood flow induced by adrenomedullin, and the mechanical removal of endothelium did not change the vasorelaxant activity of adrenomedullin in isolated basilar and middle cerebral arteries in dogs (Baskaya et al., 1995). This discrepancy may reflect differences in the regional distribution of the effect of adrenomedullin or variations among species.
In this study, rat adrenomedullin was more potent than human adrenomedullin at the activity of increasing rCBF in rats. This phenomenon was similar to the results obtained in the measurement of its systemic depressor activity (Sakata et al., 1993; Lin et al., 1994). Recently, the cDNA encoding human, rat, and porcine adrenomedullin has been isolated and sequenced (Kitamura et al., 1993b; Sakata et al., 1993; Kitamura et al., 1994). The cDNA sequence of porcine adrenomedullin seems to be very similar to that of human adrenomedullin, although the sequence of rat adrenomedullin is distinct from that of humans with the deletion of two residues and the substitution of six (Kitamura et al., 1994). Therefore, human adrenomedullin may be less sensitive to adrenomedullin receptors in rats.
Interestingly, it is known that adrenomedullin is actively synthesized and secreted from both vascular endothelial and smooth-muscle cells of various species (Sugo et al., 1994a, b ). The secretion rate of adrenomedullin from endothelial cells seems to be almost comparable to that of endothelin-1, and the secretion rate from vascular smooth-muscle cells is less than that from endothelial cells. A large amount of adrenomedullin mRNA was detected by RNA blot analysis in cultured endothelial cells and vascular smooth-muscle cells, at an intensity higher than that in the adrenal gland. These data suggest that adrenomedullin belongs to the new category of vasoactive peptides and that adrenomedullin secreted from vascular smooth-muscle cells may function as an autocrine or paracrine regulator in vascular cell communication.
Infusion of adrenomedullin suppressed the reduction in rCBF on the lesioned side following MCA occlusion in SHRs, while retaining cortical blood flow distal to the occluded blood vessel. This effect was probably the result of increasing collateral circulation through various routes. The infusion of adrenomedullin reduced the volume of ischemic brain injury by 20% in this reproducible occlusion model. Since SHRs have fewer collateral vessels and are more refractory than SD rats to treatments aimed at reducing stroke size, the present findings indicate that the protection afforded by adrenomedullin is indeed substantial. We suggest that hemodynamic mechanisms, including increased collateral flow, may explain the reduced volume of ischemic brain injury after MCA occlusion following the intravenous infusion of adrenomedullin. It has been proposed that even small increases in rCBF may improve tissue outcome during focal ischemia, especially that involving the periischemic zone and ischemic penumbra. In addition, adrenomedullin may have differing protective effects against ischemic insults. A recent report showed that adrenomedullin mRNA expression is significantly increased in the ischemic cortex in the rat focal stroke model of MCA occlusion (Wang et al., 1995). The details regarding the specific involved mechanisms of adrenomedullin expressed in the ischemic brain will require further study.
In conclusion, adrenomedullin is a vasoactive peptide of a new category constitutively secreted from both vascular endothelial and smooth-muscle cells in addition to other tissues. Adrenomedullin of concentrations not affecting the systemic blood pressure may participate in the regulation of rCBF by adjusting the cerebral vascular tonus. This peptide suppresses the reduction in rCBF following an ischemic insult, hence preventing ischemic brain injury. The clinical relevance of adrenomedullin remains to be evaluated, especially in postocclusion treatment.
Footnotes
Acknowledgment:
We thank Dr. Mustafa K. Baskaya for his helpful advice.
