Abstract
Myo-inositol is one of the major organic osmolytes in the brain. It is accumulated into cells through an Na+/myo-inositol cotransporter (SMIT) that is regulated by extracellular tonicity. To investigate the role of SMIT in the brain after cerebral ischemia, we examined expression of SMIT mRNA in the rat brain after middle cerebral artery occlusion, which would reflect alteration of extracellular tonicity. The expression of SMIT mRNA was markedly increased 12 h after surgery in the cortex of the affected side and lasted until the second day. Increased expression was also found in the contralateral cingulate cortex. Up-regulated expression was found predominantly in the neurons in remote areas, although nonneuronal cells adjacent to the ischemic core also expressed this mRNA. These results suggest that cerebral ischemia causes extensive osmotic stress in brain and that the neuronal cells respond to this stress by increasing SMIT expression.
A wide variety of pathological states, including cerebral infarction, and their treatment can alter both extracellular and intracellular volume in the central nervous system. It has been shown using animals with middle cerebral artery (MCA) occlusion that brain osmolality increases during ischemia (Schuier and Hossmann, 1980; Matsuoka and Hossmann, 1982; Hatashita et al., 1988), which results in formation of brain edema. During the early phase of ischemia, the increase in osmolality is due mainly to the accumulation of electrolytes. When the blood–brain barrier (BBB) remains still intact, ions move in and out across the BBB through the action of specific ion transporters and channels that are present in the brain capillary endothelial cells (Schielke, 1992). Once the barrier is broken, these exchanges occur through simple diffusion. The ratio of sodium gain to potassium loss increases to an even higher value, indicating a further relative enhancement of sodium uptake. The increased concentrations of cations, mainly sodium, generate osmotic forces that cause a net increase in water (Menzies et al., 1993) and induce brain edema. This mechanism of brain edema through disruption of the BBB is called “vasogenic edema.” Brain ischemia also causes changes in the brain cell volume, increasing brain cell solute content, which results in cytotoxic brain cell swelling.
Brain cells, like many kinds of cells, respond to extracellular hypertonicity by the accumulation of high concentrations of small organic solutes, which are referred to as “compatible osmolytes,” that protect cells from the perturbing effects of high intracellular concentrations of electrolytes (Yancey et al., 1982; Bagnasco et al., 1986). Most of the brain osmolytes have now been identified (Strange, 1992) and consist of various amino acids (glutamine, glutamate, taurine, and glycine), the polyols (myo-inositol), and the methylamines (glycero-phosphorylcholine and betaine). There have been many studies concerned with the effects of changes in serum Na+ concentration on brain organic osmolytes (Gullans et al., 1993). Osmolytes have been shown to be accumulated in the brain in response to elevation of serum osmolality (Lien et al., 1990), although long periods (approximately several days) are required for accumulation in significant amounts. One interesting hypothesis is that central pontine myelinolysis may be due to the delayed increase of organic osmolytes after rapid correction of chronic hyponatremia (Lien, 1995). There is, however, little information available regarding the osmoregulatory roles of organic osmolytes against the elevation of brain electrolyte concentration under ischemic conditions.
Most of the brain osmolytes are probably transported into cells from the extracellular fluid. The mechanism of transport of osmolytes such as myo-inositol, betaine, and taurine has been studied extensively in kidney-derived cultured cells. Cloning of the cDNAs encoding the osmolyte transporters (Kwon et al., 1992; Uchida et al., 1992; Yamauchi et al., 1992) may facilitate understanding of the molecular mechanisms underlying accumulation of these osmolytes. The abundance of the mRNAs for these transporters and the transcription rates of their genes increased when the cells were cultured in a hypertonic medium (Uchida et al., 1993; Yamauchi et al., 1993), suggesting that transcription is the primary step in regulation of these transporters by hypertonicity. We have recently shown using in situ hybridization (Yamauchi et al., 1995) that Na+/myo-inositol cotransporter (SMIT) mRNA expression in the kidney is regulated by NaCl loading and furosemide administration very rapidly. SMIT mRNA seems to be quite sensitive to changes in extracellular osmolality in vivo and may be a potent indicator of anisosmolality. Therefore, we would be able to monitor local osmotic changes by studying the expression of SMIT mRNA. It has been difficult to examine local osmotic changes in situ with traditional techniques such as the vapor pressure or the freeze-point depression method. It is possible that the changes in local osmolality may be different from those measured over wide areas by traditional methods. Changes in local osmolality might cause a net increase in water. Exacerbation of brain edema might induce further cellular damage. Our hypothesis is that brain ischemia causes an increase in local osmolality around the ischemic lesion, which results in the induction of SMIT expression. We speculate that myo-inositol functions as an osmolyte in cases of cerebral ischemia. To test this issue, we investigated the expression of SMIT mRNA in a model of focal brain ischemia.
METHODS
Animals with focal ischemia
Male Sprague-Dawley rats weighing 250 g were anesthetized with intraperitoneal chloral hydrate (300 mg/kg). Rectal temperature was maintained at 37.0–37,5°C with a thermostatically controlled heating pad. The left MCA was exposed with a modified procedure of the method described by Tamura and Graham (1981). Briefly, after a 2-cm vertical incision was made over the temporalis muscle, the muscle was split and its insertion into the mandible divided to reach the infratemporal fossa to expose the base of the skull from the foramen ovale to the foramen opticum. While preserving the zygoma, a drill was used to perform a small subtemporal craniectomy 2–3 mm rostral to the foramen ovale to expose the proximal part of the MCA, which is located under the dura. A 27-gauge needle was used to open the dura. The MCA was then irreversibly occluded by electrocoagulation over a distance of 2 mm, starting proximally at the medial margin of the olfactory tract. In sham-operated controls, the MCA was exposed but not coagulated (n = 3). Blood gases, pH, and MABP were monitored throughout the operation. Experiments were completed only if these physiological variables remained within normal limits. The normal values for MABP were set at 90–130 mm Hg, those for Pco2 at 30–50 mm Hg, for Po2 at 100–130 mm Hg, and for arterial blood pH at 7.25–7.45. After recovery from anesthesia, the animals were returned to their cages and allowed ad libitum normal rat chow and water. Two, 4, 6, and 12 h as well as 1, 2, 3, 4, and 7 days following MCA occlusion (n = 7 for 1 day and n = 3 for the other time intervals), (he animals were deeply anesthetized by intraperitoneal injection of pentobarbital (60 mg/kg) and their brains were removed and quickly frozen at −80°C. Serial coronal sections (5 μm thick) were obtained from the frozen brains with a cryostat and stored in a tightly closed case at −80°C.
Northern blot analysis
The entire cortex of the affected side including ischemic tissue samples was dissected out from the brains of rats with MCA occlusion at 2 h, 1 day, 3 days, and 7 days (n = 3 for each time interval), as well as from sham-operated controls (n = 3), and immediately frozen at −80°C. Total RNA was isolated by the acid guanidium isothiocyanate/phenol/chloroform method as described previously (Chomczynski and Sacchi, 1987). Aliquots (40 μg) of RNA were separated on 1% agarose formaldehyde gels and transferred onto nylon membranes (Hybond-N; Amersham Corp., Arlington Heights, IL, U.S.A.). A 490-bp rat SMIT cDNA (Yamauchi et al., 1995), which was 91% identical to the published MDCK SMIT cDNA (bases 808–1,297) (Kwon et al., 1992), and a nearly full-length rat GAPDH cDNA were labeled by random priming (Amersham) with [α-32P]dCTP (3,000 Ci/mmol; Amersham). Hybridization with the SMIT and GAPDH probes was carried out at 42°C overnight in 50% formamide, 5× SSC (1× SSC is 0.15 MNaCl and 0.015 M sodium citrate, pH 7.0), 0.1% sodium dodecyl sulfate (SDS), 50 mM sodium phosphate, 5× Denhardt solution, and 100 μg/ml salmon sperm DNA. The blots were washed three times at 42°C for 30 min in 1× SSC and 0.1% SDS. The membranes were placed in contact with x-ray film at −80°C for 5 days using intensifying screens. After quantification of the hybridized probe, it was removed from the membrane to be hybridized with the GAPDH probe. The hybridized probe was removed with a boiling solution of 0.1% SDS poured on the membrane and allowed to cool to room temperature. After the removal of the SMIT probe, hybridization and washing for the GAPDH probe were carried out in the same manner. Finally, x-ray film was placed on the membrane at −80°C for 16 h.
In situ hybridization
The antisense probe for SMIT was synthesized from a 490-bp rat SMIT cDNA (bases 808–1,297) insert cloned in the Novagen T-vector. The sense probe for SMIT was synthesized from a 490-bp rat SMIT cDNA insert cloned in the vector pSPORT 1. To synthesize hybridization riboprobes by in vitro transcription, these sequences were first linearized by digestion with restriction endonucleases of EcoR1 for both antisense and sense RNA synthesis. The linearized cDNA was then incubated at 37°C for 60 min with a mixture of reagents. This mixture consisted of 2 μl of transcription buffer (×5), 0.5 μl of 100 mM dithiothreitol, 0.5 μl of RNase inhibitor, 0.5 μl of 10 mM ATP, CTP, and GTP, 5 μl of [35S]UTP (NEG-039H; New England Nuclear), 0.5 μl of DNA template (1 μg/ml), with 1 μl of appropriate RNA polymerase (T7 RNA polymerase for antisense probe; SP6 RNA polymerase for sense probe). DNA digestion was achieved by the addition of 2 μl of DNase and incubation at 37°C for 10 min. Efficacy of labeling was estimated by quantification of radioactivity of the synthesized probes.
In situ hybridization techniques for SMIT mRNA (RNA probe) were based on those of Wilkinson et al. (1988) with some modifications. The sections were fixed in 4% paraformaldehyde in 0.1 M phosphate buffer for 20 min. After washing with phosphate-buffered saline, the sections were treated with 10 μg/ml of proteinase K in 50 mM Tris-HCl and 5 mM ethylenediaminetetraacetate (EDTA; pH 8.0) for 5 min at room temperature. They were fixed again in the same fixative, then acetylated with acetic anhydride in 0.1 M triethanolamine, rinsed with phosphate-buffered saline, dehydrated, and air dried. The 35S-labeled RNA probes (sense or antisense) were diluted in hybridization buffer, placed over the sections, and covered with siliconized coverslips. Hybridization was performed overnight in a humid chamber at 55°C. The hybridization buffer consisted of 50% deionized formamide, 0.3 M NaCl, 20 mM Tris-HCl, 5 mM EDTA, 10 mM phosphate buffer, 10% dextran sulfate, 1× Denhardt solution, 0.2% sarcosyl, 500 μg/ml yeast tRNA, and 200 μg/ml herring sperm DNA (pH 8.0). The probe concentration was 5 × 105 cpm/150 μl/slide. After hybridization, the slides were immersed in 5× SSC at 55 °C, and the coverslips were allowed to fall off. The sections were then incubated at 65°C in 50% deionized formamide with 2× SSC for 30 min. After rinsing with RNase buffer [0.5 M NaCl, 10 mM Tris-HCl, 5 mM EDTA (pH 8.0)] four times for 10 min each time at 37°C, the sections were treated with 1 μg/ml of RNase A in RNase buffer for 30 min at 37°C. After an additional wash in RNase buffer, the slides were incubated in 50% formamide with 2× SSC for 30 min at 65°C, rinsed with 2× SSC and 0.1× SSC for 10 min each at room temperature, dehydrated in an ascending alcohol series, and air dried.
x-Ray film was placed on the uncoated sections for 6 days. Next, the slides were coated with Ilford K-5 emulsion diluted in distilled water containing 2% glycerine (1:1). The slides were exposed for 3 weeks in a tightly sealed dark box at 4°C, developed in Kodak D-19, fixed with photographic fixer, stained with thionin, and coverslipped. After the x-ray macroautoradiogram had been studied, the tissue sections were examined under a regular light microscope.
For quantitative assessment of SMIT mRNA expression on the macroautoradiograms, the optical density of the cortex and thalamus was measured with a densitometer with an aperture of 0.5 mm (Sakura PDA-15; Konishiroku, Tokyo, Japan). The same area was measured five times and the mean value of the counts was used for further analysis. Optical density ratio of the cortex was calculated in comparison with the thalamus on the ipsilateral side. Statistical significance of results was determined using the nonparametric analysis of the Mann–Whitney U test with two-tailed probability.
RESULTS
Focal cerebral ischemic lesions are histologically well defined areas of necrosis appearing after 24 h of ischemia with disappearance of glial cells and nerve cells. Prior to 24 h, such lesions contain degenerating nerve cell perikarya with an increase in the neuropil area due to edema. The infarct is then surrounded by a thin zone of small cells, some of which extend into the lesion.
To determine whether expression of SMIT mRNA is altered by cerebral ischemia, total RNA was isolated from the ipsilateral cortex of the affected side following MCA occlusion and hybridized with the rat SMIT cDNA probe. Figure 1 shows a Northern blot revealing the time course of changes in cortical SMIT mRNA expression after MCA occlusion. The main band was clearly seen with a size of 10 kb, which is consistent with other reports (Kwon et al., 1992; Yamauchi et al., 1994). The level of this transcript was markedly increased, reaching a maximum 1 day after surgery. SMIT expression decreased thereafter.
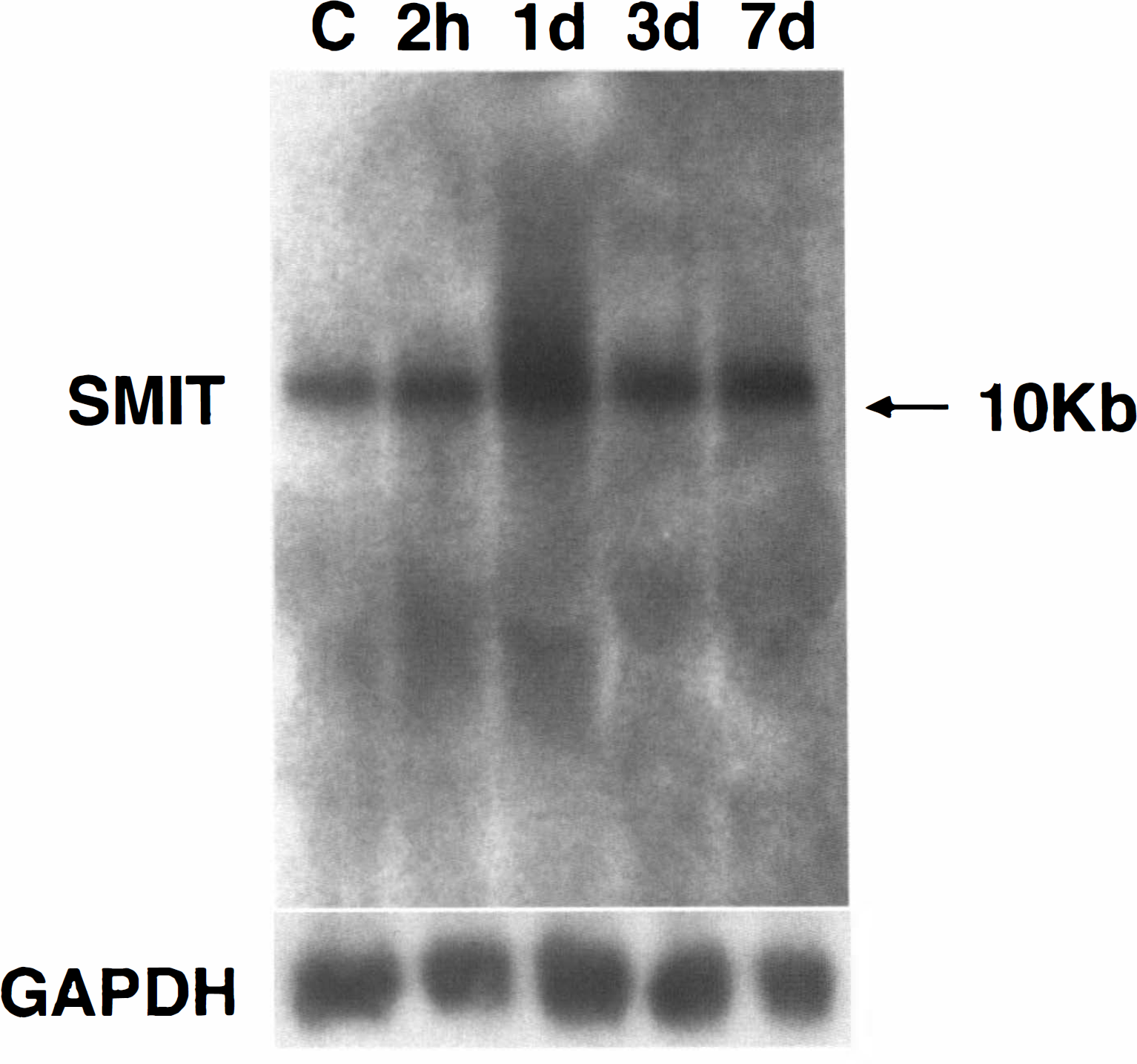
Northern blot analysis of Na+/myo-inositol cotransporter (SMIT) mRNA induction in the ischemic cortex following middle cerebral artery occlusion. The main band was clearly seen at a size of 10 kb. The level of this transcript was markedly increased, reaching a maximum 1 day after surgery. SMIT expression decreased thereafter.
To determine the localization of SMIT expression, we performed in situ hybridization using a 35S-labeled cRNA probe. Consecutive sections were hybridized with both sense and antisense probes to confirm the specificity of the hybridization signals of SMIT mRNA throughout this study (Fig. 2). Signals were observed only in the sections hybridized with the antisense probes. In sham-operated animals with normal serum osmolality, moderate expression of SMIT mRNA was detected in the choroid plexus, hippocampus, and suprachiasmatic nucleus. Microautoradiography showed that SMIT mRNA signals were scattered and located in both the neuronal cells and the small nonneuronal cells in the cerebral cortex, white matter, caudate–putamen, and thalamus.
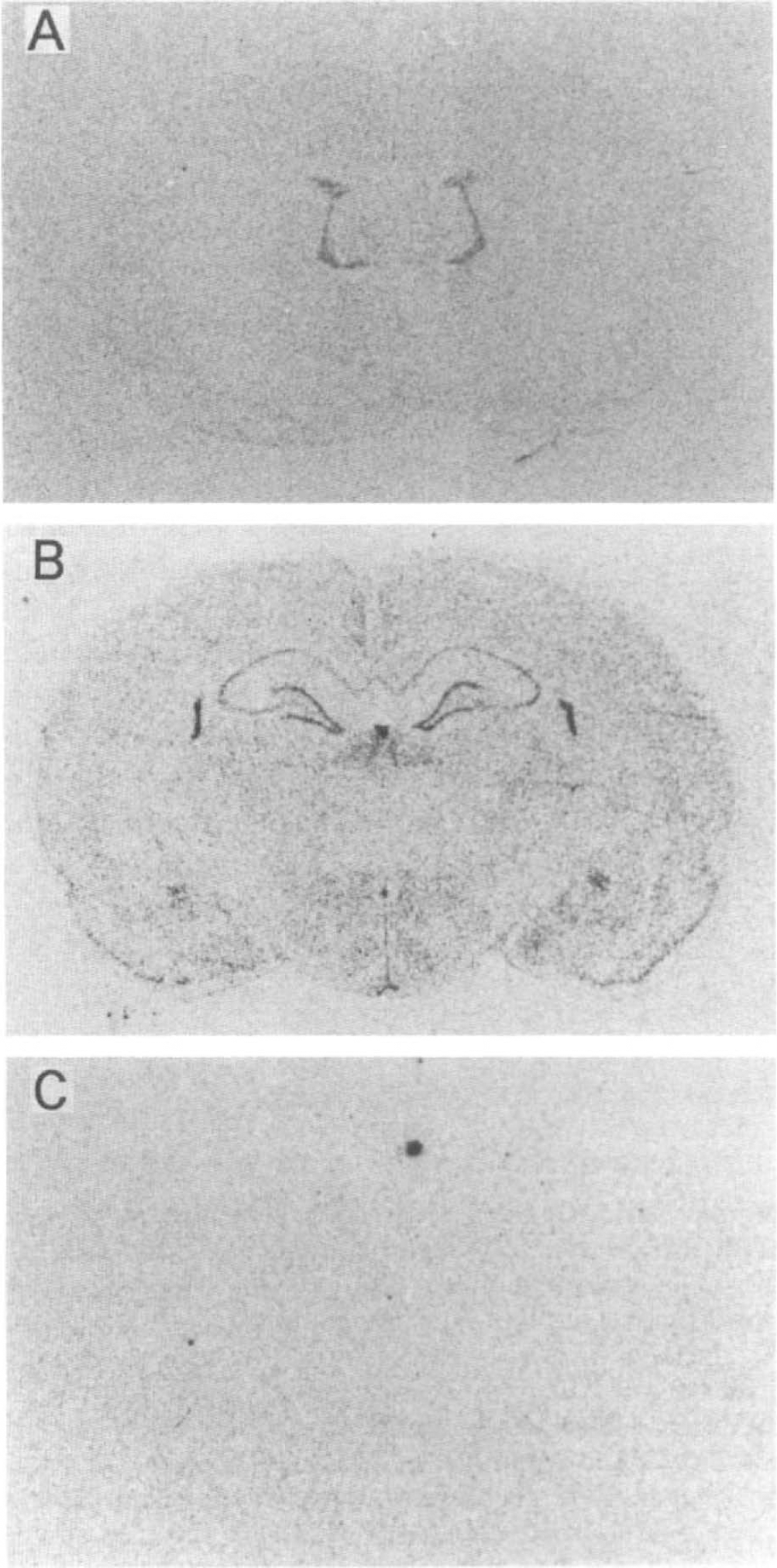
Macroautoradiogram of in situ hybridization for Na+/myo-inositol cotransporter (SMIT) mRNA. Antisense probe (
In animals with MCA occlusion, the expression of SMIT mRNA started to increase at 6 h and peaked at 12 h after surgery in the cortex of the affected side, including the cingulate, neocortex (frontal, parietal, and occipital) and piriform cortex (Fig. 3). Increased expression was also predominantly found in the periischemic area and caudate–putamen, but no increase was observed in the thalamus. Surprisingly, SMIT signals were seen in the contralateral side. The signals in the contralateral side were detected in the cingulate cortex, septal nucleus, and diagonal band. These signals in the contralateral side were increased and extended widely, and the increase in the hemisphere of the affected side was persistent at 1 day (Fig. 3). Microautoradiography showed that marked induction of SMIT mRNA signals was found mainly in medium- to large-sized cells (˜30 μm in diameter) with large nuclei in remote areas such as the piriform cortex (Fig. 4B), parietal cortex (Fig. 4C), and cingulate cortex, suggesting that these SMIT mRNA-positive cells are morphologically typical neuronal cells and up-regulation of the gene expression in these areas was due mainly to an increase in neuronal SMIT expression. No signals were observed in the core of the infarct at any time during the observation period, suggesting that the cells in this region were dying or had already died. SMIT mRNA signals of the cells also increased in the white matter adjacent to the ischemic area (Fig. 4D). Microglia or astrocytes also were responsible for a source of the mRNA in this area. The increase in SMIT mRNA expression peaked 1 day after surgery, remained elevated on the second day, and returned to almost control levels from the third day (Figs. 5 and 6). Signals in the contralateral piriform cortex (Fig. 4A) and white matter were similar to those in sham-operated animals. These findings suggest that osmotic stress may expand over a wide area, not only in regions adjacent to the site of infarction, but also in the contralateral cingulate cortex to the second day.
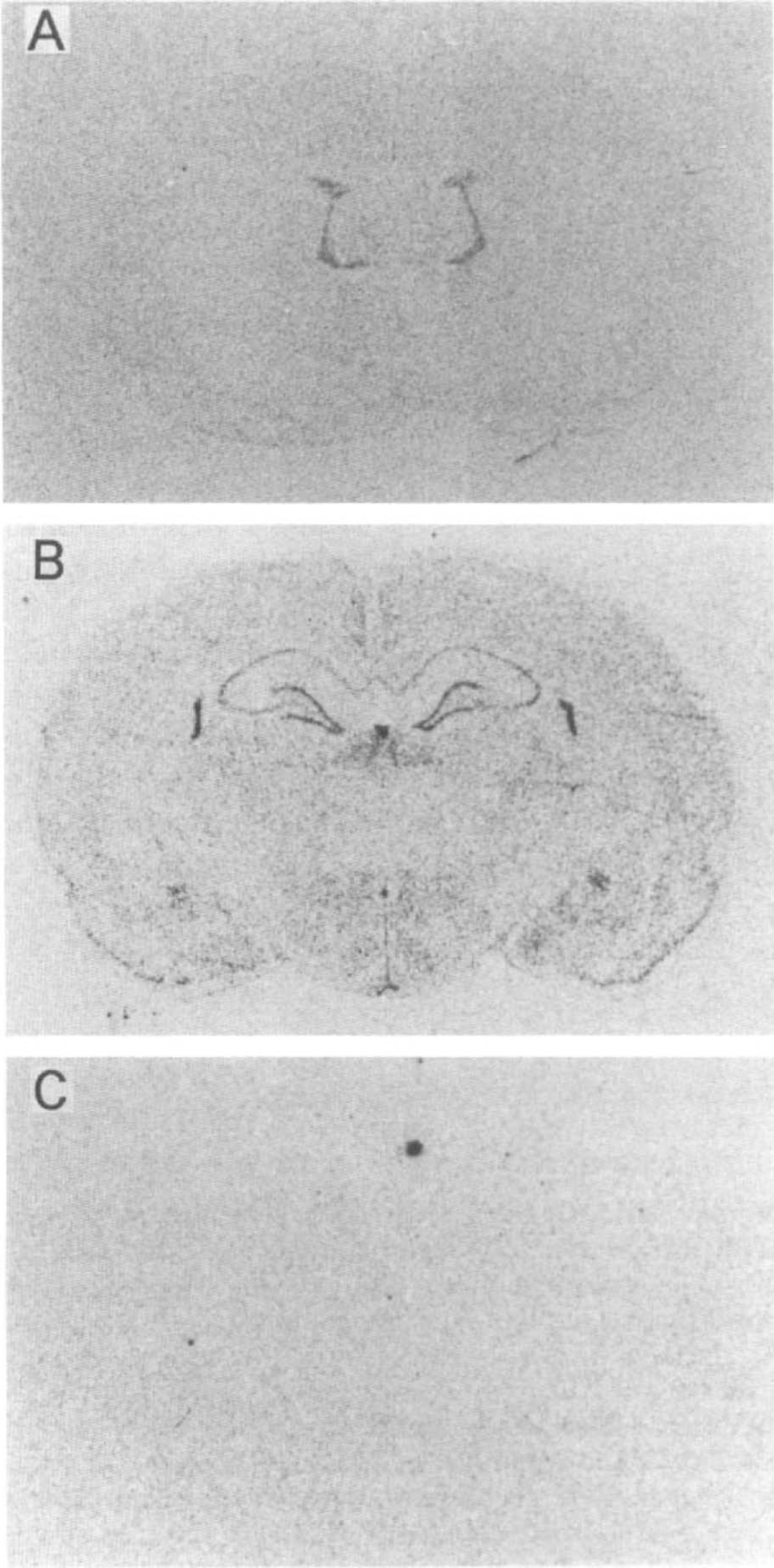
Macroautoradiograms obtained at different levels of the brain sections of animals with middle cerebral artery occlusion. Expression of Na+/myo-inositol cotransporter (SMIT) mRNA was markedly increased at 12 h (n = 3) after surgery in the cortex of the affected side, including the cingulate, neocortex (frontal, parietal, and occipital), and piriform cortex (arrow). There were no signals present in the core of the infarct. Increases in SMIT mRNA level in the contralateral cingulate cortex, septal nucleus, and diagonal band were also detected. On the first day (n = 7), these signals in the contralateral side (arrow) developed widely, while the increase in the hemisphere of the affected side was persistent.
DISCUSSION
Several studies in different species have shown that brain osmolality increases during ischemia. Hatashita et al. (1988) demonstrated that a significant increase in osmolality in the ischemic brain tissue was observed only at 3 and 6 h after MCA occlusion in rats using a vapor pressure osmometer. Schuier and Hossmann (1980) noted that brain osmolality, measured by determining freeze-point depression in tissue homogenates, increased in ischemic tissue within 4 h of MCA occlusion in cats. They indicated that cerebral ischemia caused an increase in the osmolality of ischemic brain tissue, but apparently only during the early stages of ischemia.
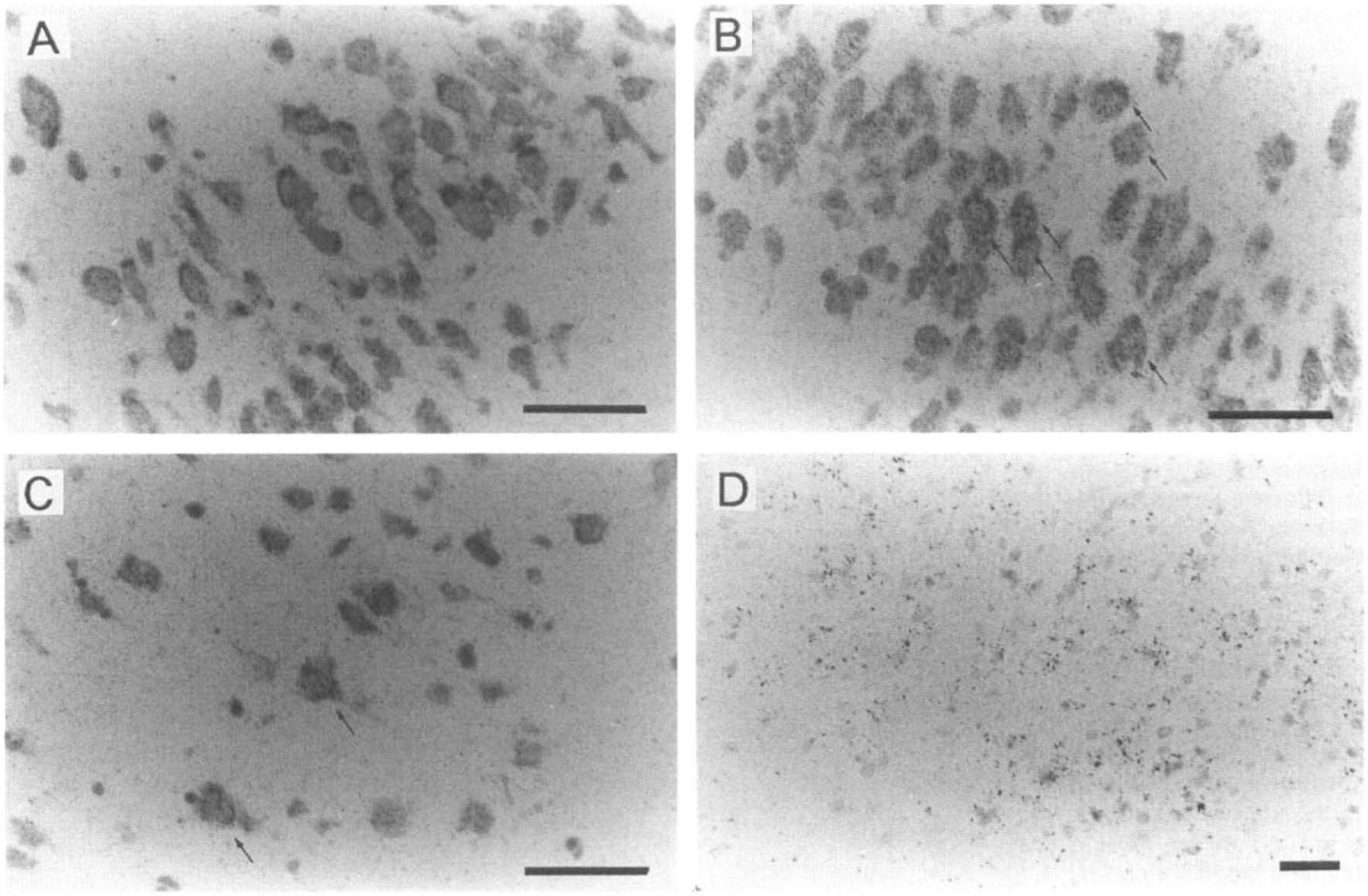
Bright-field microautoradiogram of in situ hybridization for Na+/myo-inositol cotransporter (SMIT) mRNA. In the contralateral cortex (
Our results seem to be inconsistent with these previous reports. Our recent observation suggested that SMIT expression reflects regional or local osmolality in the kidney (Yamauchi et al, 1995). The observation that SMIT expression continues to increase until 1 day and lasted at least 2 days after MCA occlusion suggests that the osmotic stress persisted over this period. This discrepancy is probably due to the sensitivity in detection of changes in local osmolality. As the previous reports described already examined only gross tissue osmolality, they could not detect the small changes in local osmolality. Furthermore, they did not pay attention to the remote areas from the ischemic core. Alternatively, the neuronal cells respond to osmotic stimuli much more slowly than kidney cells. Although there have been no reports regarding myo-inositol transport in the neuronal cells, myo-inositol transport in C6 glioma cells has been shown to increase very slowly in response to extracellular hypertonicity (Strange et al, 1991). Another possibility is that SMIT is regulated by unknown factor(s) other than osmolality. For example, some transcription factors produced after ischemia are known to activate transcription of wide varieties of genes. It is possible that the SMIT gene is activated by such transcription factors). There have been no reports, however, regarding the relationship between SMIT and these factors in spite of extensive studies of this transporter (Burg, 1995; Yamauchi et al, 1996). In either case, it was apparent that the neuronal cells in remote areas were obliged to express unusual genes for long periods after cerebral ischemia. Our results in the present study suggest that MCA occlusion induces osmotic stress over longer periods and more extensively than previous studies have shown.
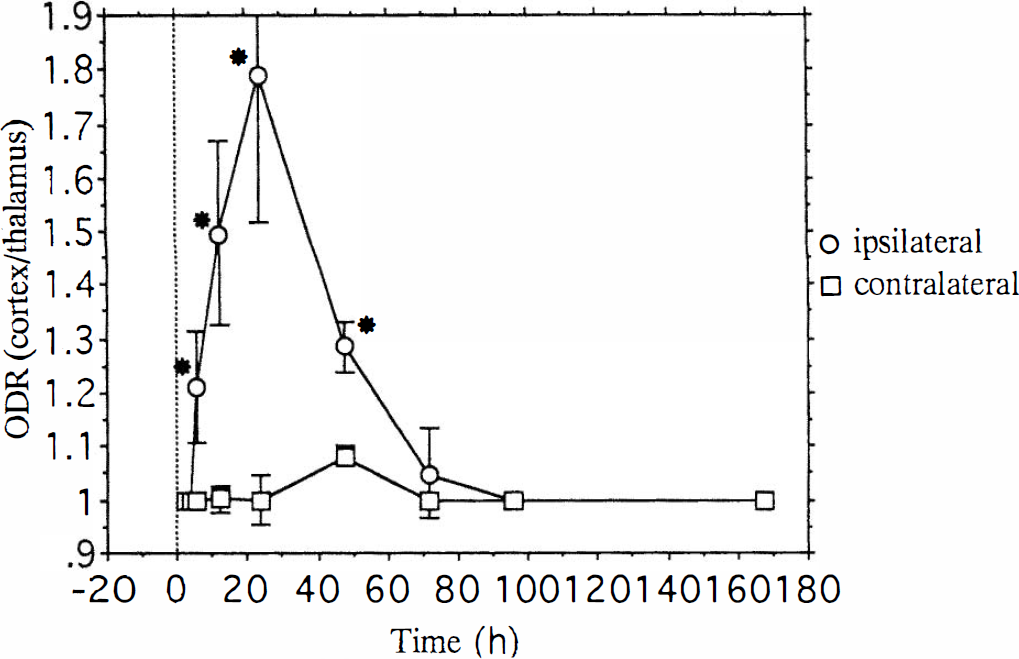
Time-dependent Na+/myo-inositol cotransporter (SMIT) mRNA expression after focal cerebral ischemia. Optical density ratio (ODR) of the piriform cortex was calculated in comparison with the ipsilateral thalamus. Data are presented as means ± SD for each time point. Nonparametricanalysis of the Mann–Whitney Utest with two-tailed probability was performed to demonstrate differences between cortex and thalamus. Significant (*p < 0.05) increases in SMIT mRNA level were observed in the affected side from 6 h to 2 days after surgery.
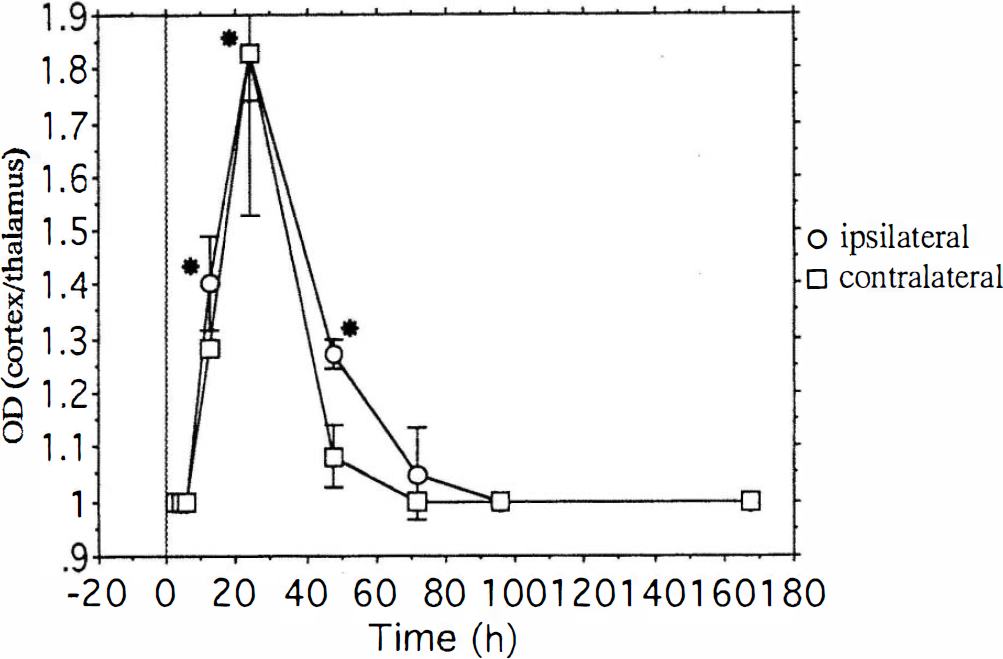
Time-dependent Na+/myo-inositol cotransporter (SMIT) mRNA expression after focal cerebral ischemia. Optical density ratio (OD) of the cingulate cortex was calculated in comparison with the ipsilateral thalamus. Data are presented as means ± SD for each time point. The expression of SMIT mRNA was markedly increased at 12 h after surgery in the cortex on the affected side. An increase in the contralateral cingulate cortex was also detected. It lasted until 2 days after surgery, and almost no increase in the expression of SMIT mRNA was found 3 days thereafter. Nonparametric analysis of the Mann–Whitney U test with two-tailed probability was performed to demonstrate differences between cortex and thalamus. Significant (*p < 0.05) increases in SMIT mRNA level were observed in the ipsilateral side from 12 h to 2 days after surgery.
The results of the present study indicate that, after MCA occlusion, SMIT expression extends over a wide area, not only adjacent to the site of infarction, but also in remote areas such as the contralateral cingulate cortex. These results suggest that the increased SMIT expression observed in areas remote from the ischemic core, where the BBB is presumably maintained, might reflect the increased osmotic substances diffusing from the ischemic core. In consideration with the time course of SMIT expression, not only Na+ but also osmotic substances other than Na+ might play major roles in inducing SMIT mRNA in cases of ischemia. Macromolecules from the blood flowing across the disrupted BBB and/or accumulation of lactate together with breakdown products of labile compounds might induce an increase in SMIT mRNA in remote areas. In other words, vasogenic brain edema may affect the cells in remote areas.
In the postischemic brain, alterations in protein synthesis could result in a lack of translation of the mRNA or in the production of a nonfunctional protein. Malfunction of SMIT might fail in protecting cells from the perturbing effects. The study of cellular mechanisms involved in regulation of these organic osmolyte concentrations will be an important area for further investigation of brain edema therapy.
In spite of the expansion of the expression of SMIT, there were only weak signals in ipsilateral thalamus and substantia nigra. Because infarction increased SMIT predominantly in neuronal cells in other regions, we speculate that neuronal cells in these regions may be less sensitive to osmotic stress or may have a different protection system against osmotic stress.
Our study suggested that the increased SMIT expression occurred predominantly in the neuronal cells in remote areas, although increased expression of nonneuronal small cells was found in periischemic areas. Neuronal cells are thought to be more susceptible to these kinds of pathological changes. The present results suggest that osmotic stress against the cells appears to be more extensive and longer-lasting than previously considered. The cells appear to initiate efforts to protect themselves from the perturbing effects of high intracellular concentrations of electrolytes by means of increasing myoinositol accumulation. Brain edema formation might have a much greater influence than has previously been supposed.
In summary, we examined SMIT expression in the brain after MCA occlusion. SMIT mRNA was markedly increased in the cortex of the affected side after the infarction. Increased expression was observed in both affected and unaffected sides of the brain and lasted until the second day, suggesting that extensive osmotic stress was induced by infarction. The extensive osmotic stress may be responsible for the prolonged and broad clinical symptoms after cerebral ischemia. This point should be elucidated in future studies. The results of our study offer new insight into volume regulation during cerebral infarction.
